Effect of Radio-Frequency Treatment on the Changes of Dissolved Organic Matter in Rainwater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Rainwater
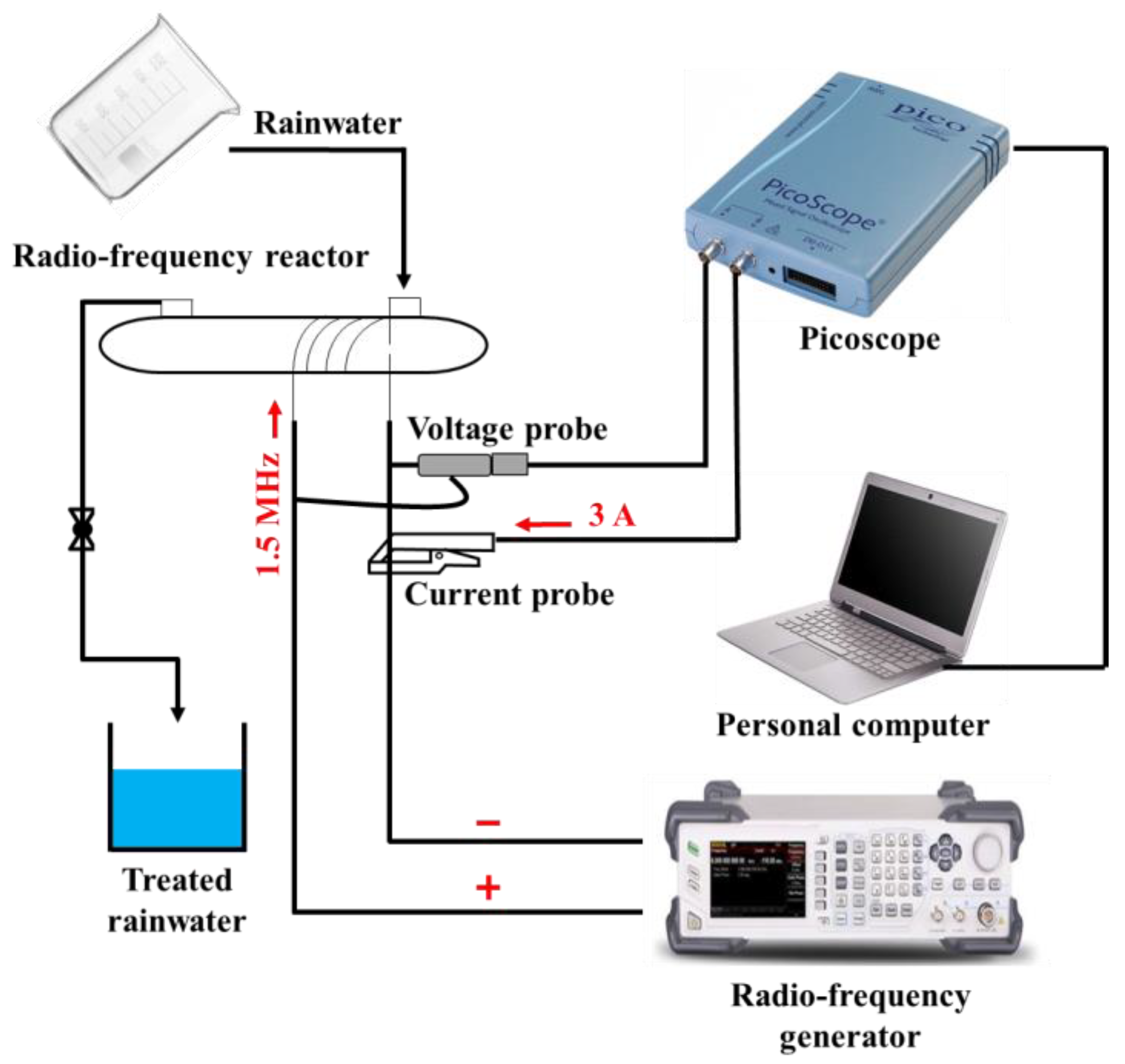
2.2. RF Treatment
2.3. DOM Analysis
3. Results and Discussion
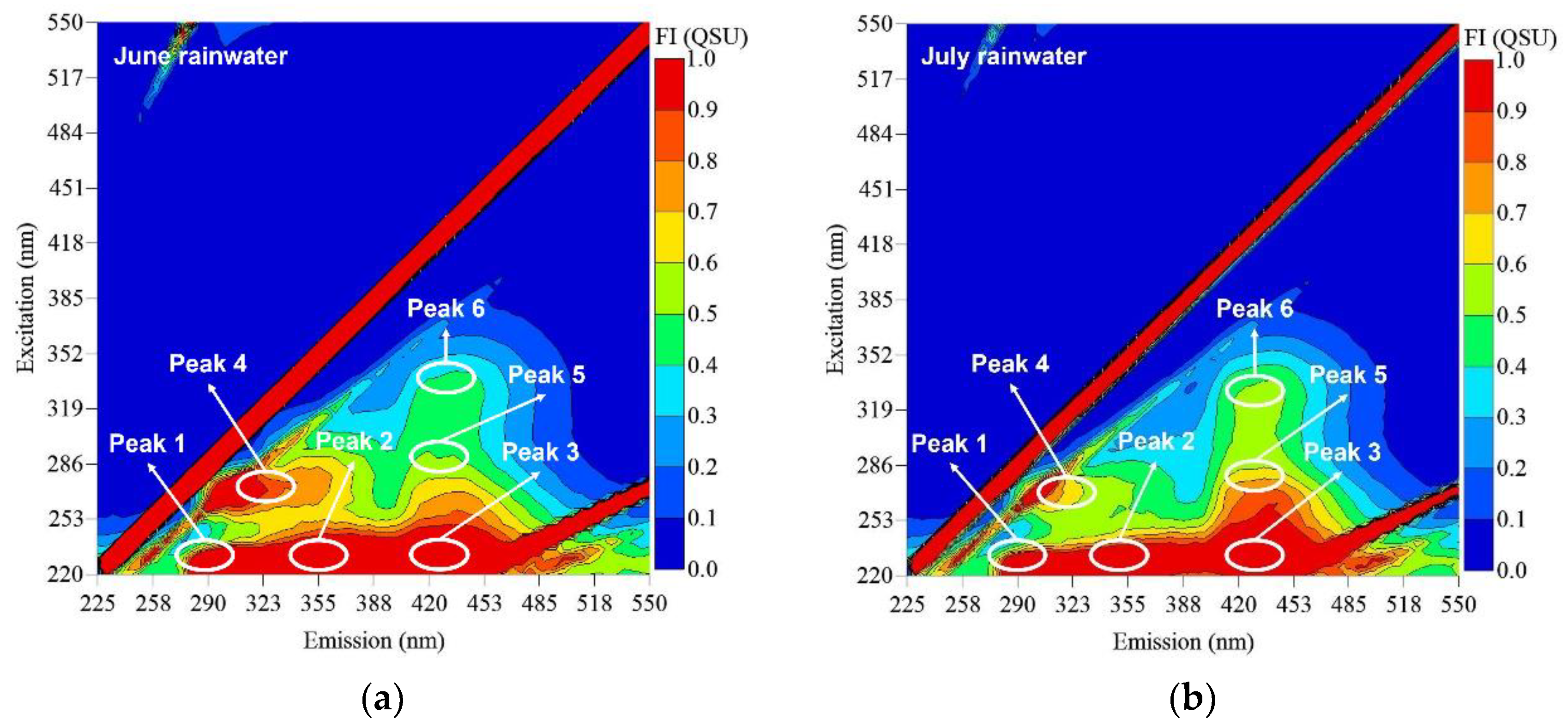
3.1. Characteristics of Rainwater
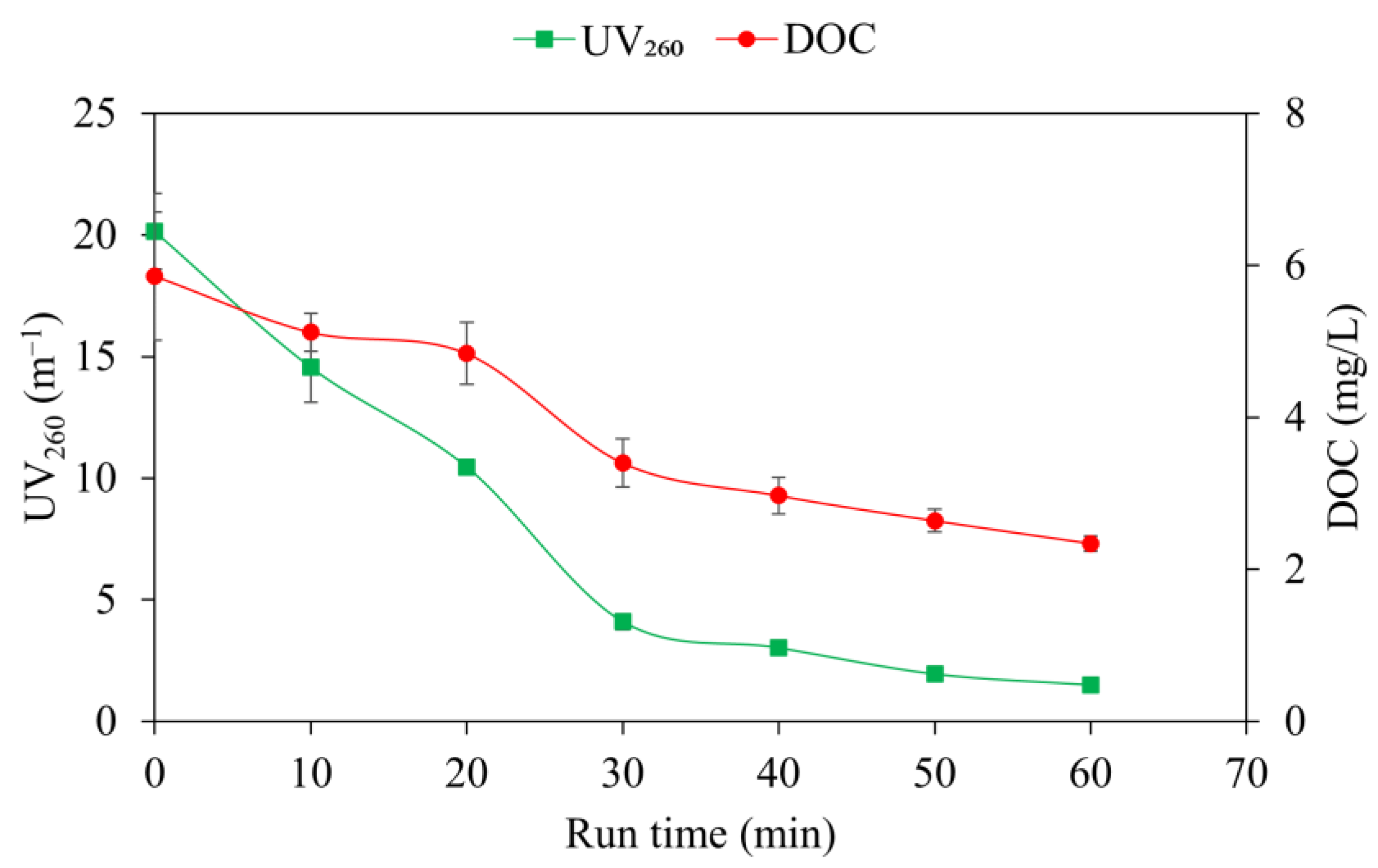
3.2. Impact of RF on DOC and UV260
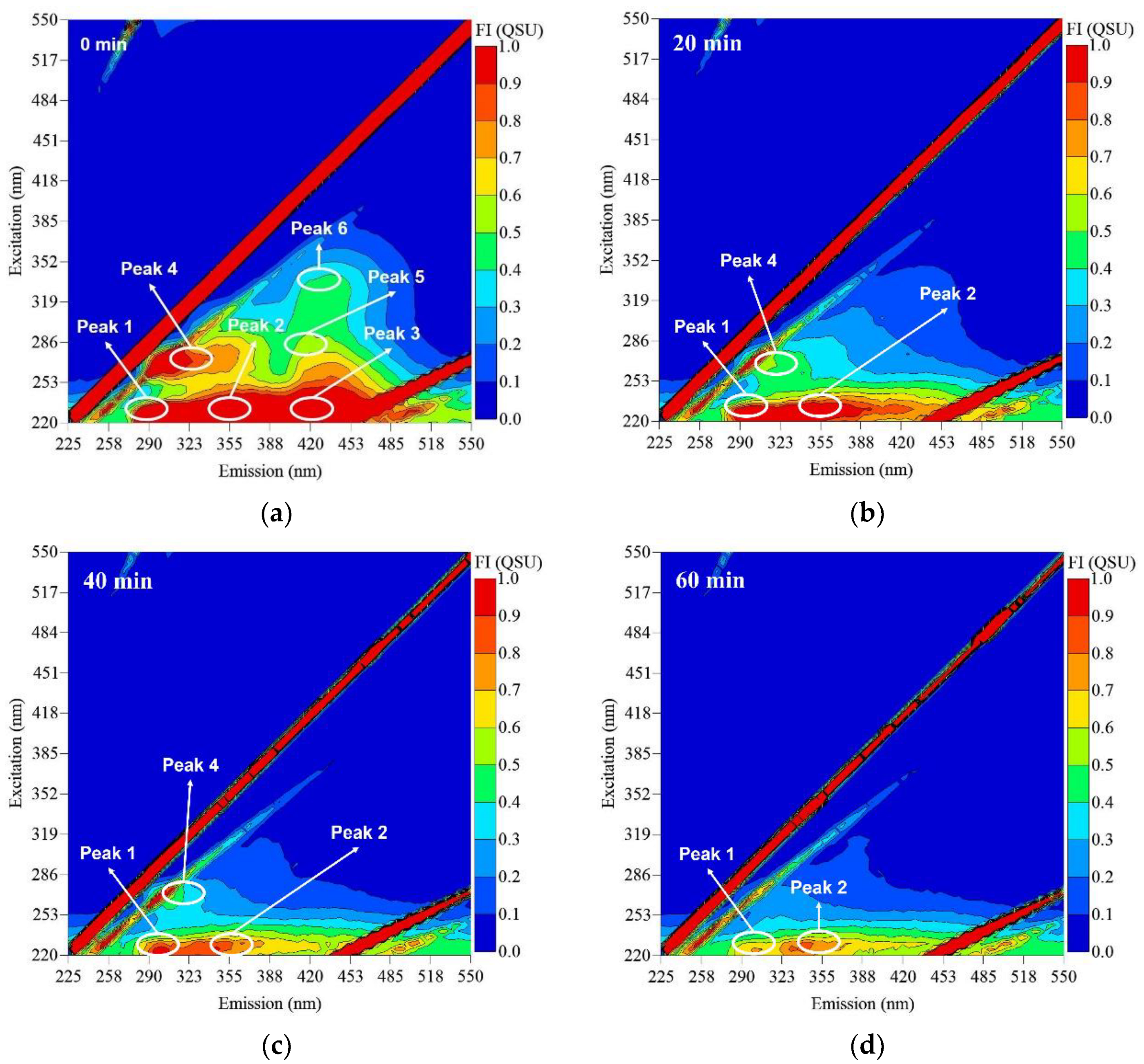
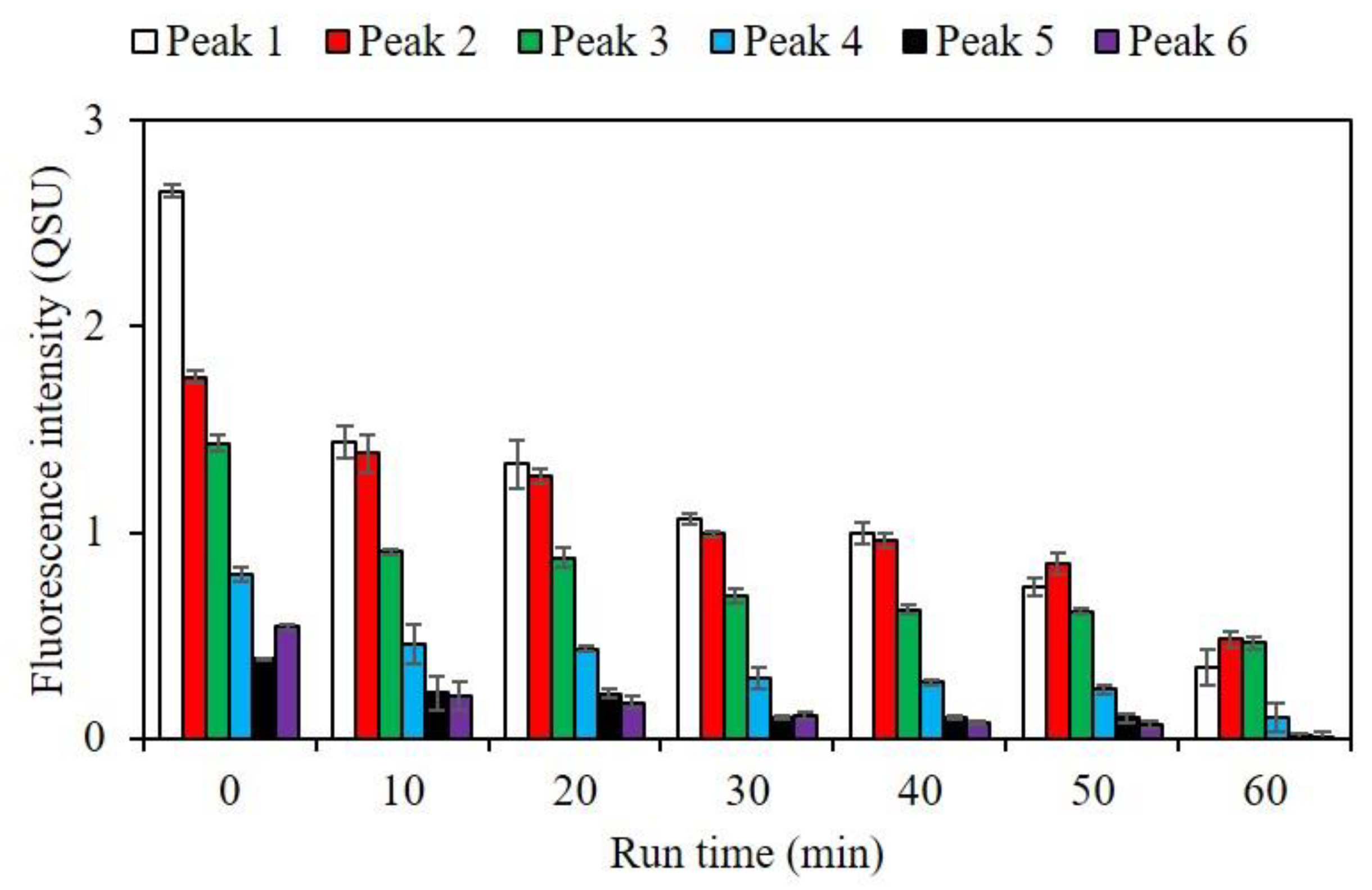
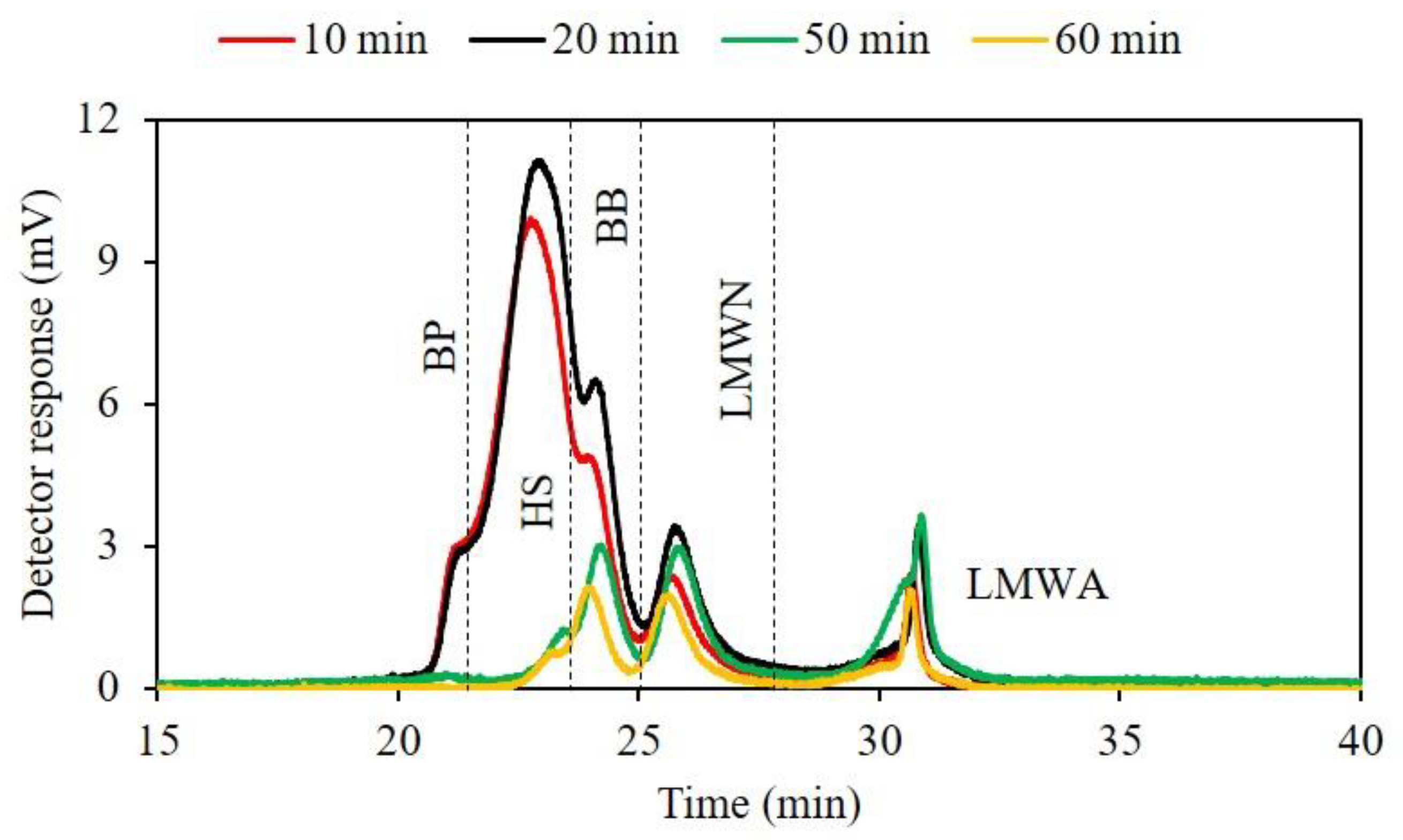
3.3. Impact of RF on Fluorescent DOM Composition
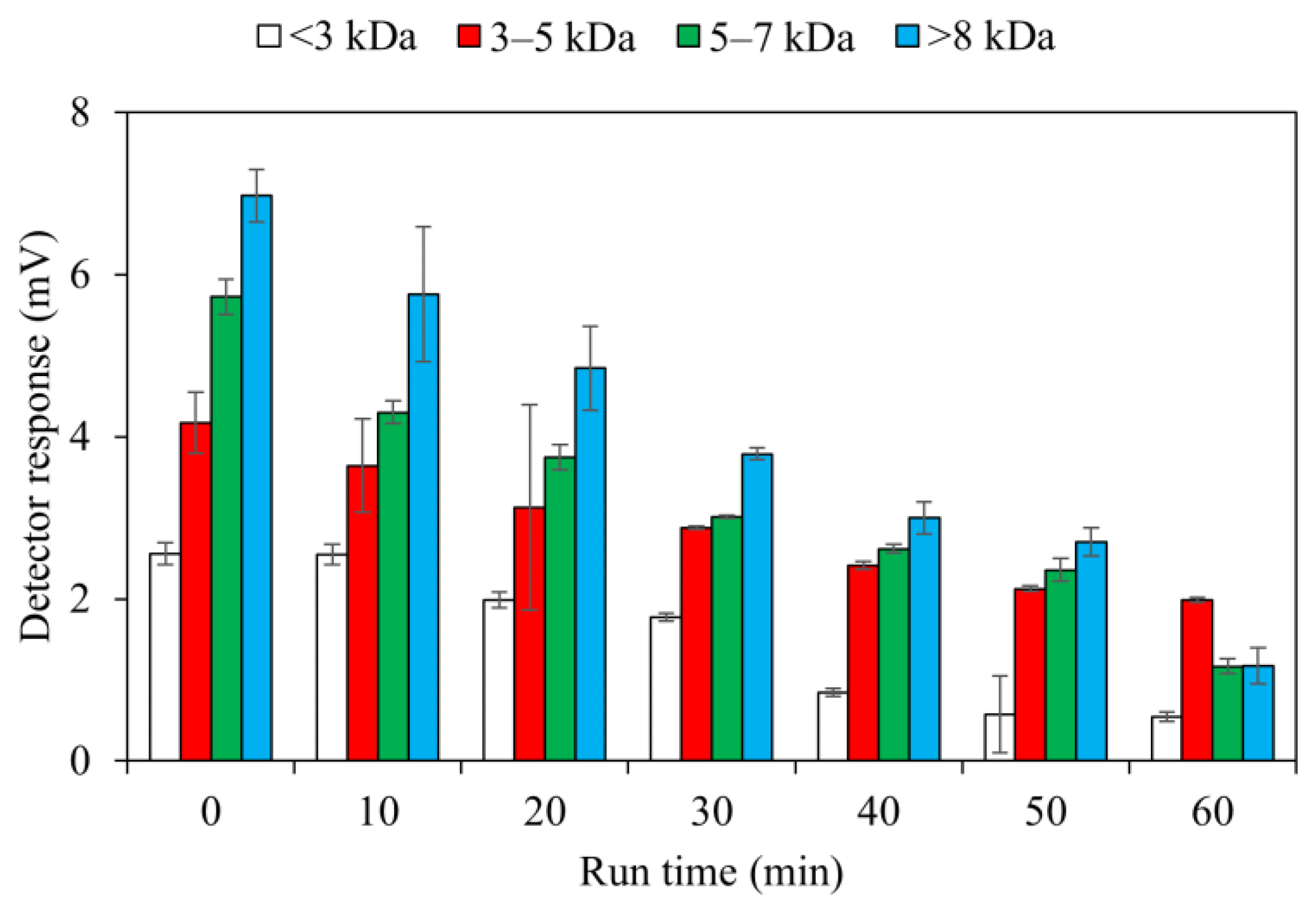
3.4. Impact of RF on MW of DOM
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Campasino, A.; Butler, D.; Ward, S.; Burns, M.J.; Friedler, E.; DeBusk, K.; Fisher-Jeffes, L.N.; Gishi, E.; Rahman, A.; Furumai, H.; et al. Urban rainwater harvesting systems: Research, implementation and future perspectives. Water Res. 2017, 115, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, J.; Yue, T.; Jing, X. Impacts of climate change on urban rainwater harvesting system. Sci. Total Environ. 2019, 665, 262–274. [Google Scholar] [CrossRef]
- Musayev, S.; Burgess, B.; Mellor, J. A global performance assessment of rainwater harvesting under climate change. Resour. Conserv. Recycl. 2018, 132, 62–70. [Google Scholar] [CrossRef]
- Chapa, F.; Krauss, M.; Hack, J. A multi-parameter method to quantify the potential of roof rainwater harvesting at regional levels in areas with limited rainfall data. Resour. Conserv. Recycl. 2010, 161, 104959. [Google Scholar] [CrossRef]
- Bui, T.T.; Nguyen, D.C.; Han, Y.; Kim, M.; Park, H. Rainwater as a source of drinking water: A resource recovery case study from Vietnam. J. Water Process Eng. 2011, 39, 101740. [Google Scholar] [CrossRef]
- Sharma, A.K.; Cook, S.; Gardner, T.; Tjandraatmadja, G. Rainwater tanks in modern cities: A review of current practices and research. J. Water Clim. Chang. 2016, 7, 445–466. [Google Scholar] [CrossRef]
- Nodle, E. Possibilities of rainwater utilisation in densely populated area including precipitation runoffs from traffic surfaces. Desalination 2017, 215, 1–11. [Google Scholar] [CrossRef]
- Villarreal, E.L.; Dixon, A. Analysis for a rainwater collection system for domestic water supply in Ringdansen, Norrkoping, Sweden. Build. Environ. 2015, 40, 1174–1184. [Google Scholar] [CrossRef]
- Kim, Y.K.; Han, M.Y.; Kabubi, J.; Sohn, H.G.; Nguyen, D.C. Community-based rainwater harvesting (CB-RWH) to supply drinking water in developing countries: Lessons learned from case studies in Africa and Asia. Water Supply 2016, 16, 1110–1121. [Google Scholar] [CrossRef]
- Daniel, D.; Djohan, D.; Machairas, I.; Pande, S.; Arifin, A.; Al Djono, T.P.; Rietveld, L. Financial, institutional, environmental, technical, and social (FIETS) aspects of water, sanitation, and hygiene conditions in indigenous—Rural Indonesia. BMC Public Health 2021, 21, 1723. [Google Scholar] [CrossRef] [PubMed]
- Battin, T.J.; Luyssaert, S.; Kaplan, L.A.; Aufdenkampe, A.K.; Richter, A.; Tranvik, L.J. The boundless carbon cycle. Nat. Geosci. 2009, 2, 598–600. [Google Scholar] [CrossRef]
- Li, S.; Fan, R.; Luo, D.; Xue, Q.; Li, L.; Yu, X.; Huang, T.; Yang, H.; Huang, C. Variation in quantity and quality of rainwater dissolved organic matter (DOM) in a peri-urban region: Implications for the effect of seasonal patterns on DOM fates. Atmos. Environ. 2020, 239, 117769. [Google Scholar] [CrossRef]
- Soares, M.A.; Sabu, P.; Anilkumar, N.; Mishra, R.K.; Naik, R.K.; Bhaskar, P.V.; George, J.V.; Venkataramana, V.; Sarkar, A. variation of particulate organic matter characteristics in the upper water column of eddy-influenced waters at the subtropical front of the Indian sector of the Southern Ocean. J. Sea Res. 2021, 174, 102074. [Google Scholar] [CrossRef]
- Orlović-Leko, P.; Vidović, K.; Ciglenečki, I.; Omanović, D.; Sikirić, M.D.; Šimunić, I. Physico-chemical characterization of an urban rainwater (Zagreb, Croatia). Atmosphere 2020, 11, 144. [Google Scholar] [CrossRef] [Green Version]
- Gao, B.; Yu, J.Z.; Li, S.X.; Ding, X.; He, Q.F.; Wang, X.M. Roadside and rooftop measurements of polycyclic aromatic hydrocarbons in PM 2.5 in urban Guangzhou: Evaluation of vehicular and regional combustion source contribution. Atmos. Environ. 2011, 45, 7184–7191. [Google Scholar] [CrossRef]
- Zhang, Y.; Lang, J.; Cheng, S.; Li, S.; Zhou, Y.; Chen, D.; Zhang, H.; Wang, H. Chemical composition and sources of PM 1 and 2.5 in Beijing in autumn. Sci. Total Environ. 2018, 630, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.; He, K.; Duan, F.; Cheng, Y.; Ma, Y. Measurement of humic-like substances in aerosols: A review. Environ. Pollut. 2013, 188, 301–314. [Google Scholar] [CrossRef]
- Huo, M.Q.; Sato, K.; Ohizumi, T.; Akimoto, H.; Takahashi, K. Characteristics of carbonaceous components in precipitation and atmospheric particle at Japanese sites. Atmos. Environ. 2016, 146, 164–173. [Google Scholar] [CrossRef]
- Rafiq, M.; Mishra, A.K.; Meer, M.S. On land-use and land-cover changes over Lidder Valley in changing environment. Ann. GIS 2018, 24, 275–285. [Google Scholar] [CrossRef] [Green Version]
- Environmental Protection and Management, Law of the Republic of Indonesia 32. 2009. Available online: https://jdih.esdm.go.id (accessed on 30 December 2021).
- Chen, X.; Wang, Y.; Li, W.Y.; Zhang, J.P.; Qi, W.Q.; Lu, Y.F.; Ding, Z.B. Coupling changes of disinfectant and bacteria induced stagnation and disinfection strategy. Chemosphere 2020, 242, 125190. [Google Scholar] [CrossRef]
- Vilhunen, S.; Silanpää, M. Recent developments in photochemical and chemical AOPs in water treatment: A mini-review. Rev. Environ. Sci. Bio/Technol. 2010, 9, 323–330. [Google Scholar] [CrossRef]
- Li, H.; Zhao, Z.; Xiouras, C.; Stefanidis, D.; Li, X.; Gao, X. Fundamentals and applications of microwave heating to chemical separation processes. Renew. Sustain. Energy Rev. 2019, 114, 109316. [Google Scholar] [CrossRef]
- Kovaleva, L.; Zinnatullin, R.; Gabdrafikov, A.; Sultanguzhin, R.; Kireev, V. Influence of radio-frequency and microwave electromagnetic treatment on water-in-oil emulsion separation. Colloids Surf. A Physicochem. Eng. Asp. 2021, 614, 126081. [Google Scholar] [CrossRef]
- Desmiarti, R.; Hazmi, A.; Trianda, Y.; Sari, E. Removal of pathogenic bacteria from water by radio-frequency thermal plasma treatment. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 889–897. [Google Scholar]
- Jiang, B.; Zheng, J.; Liu, Q.; Wu, M. Degradation of organic dye by pulsed discharge non-thermal plasma technology assisted with modified activated carbon fibers. Chem. Eng. J. 2013, 215–216, 969–978. [Google Scholar] [CrossRef]
- De la Hoz, A.; Díaz-Ortiz, A.; Moreno, A. Review on non-thermal effects of microwave irradiation in organic synthesis. J. Microw. Power Electromagn. Energy 2007, 41, 45–66. [Google Scholar] [CrossRef]
- Subedi, D.P.; Tyata, R.B.; Khadgi, A.; Wong, S.C. Treatment of water by dielectric barrier discharge. J. Sci. Technol. Trop. 2009, 5, 117–123. [Google Scholar]
- Sun, B.; Sato, M.; Clements, J.D. Optical study of active species produced by a pulsed streamer corona discharge in water. J. Electrost. 1997, 39, 189–202. [Google Scholar] [CrossRef]
- Desmiarti, R.; Sari, E.; Vallepi, R.R.; Wahyeni, F.S.; Rosadi, M.Y.; Hazmi, A. Drinking water production from rainwater using radio frequency plasma system. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020; Volume 990, p. 012019. [Google Scholar] [CrossRef]
- Desmiarti, R.; Hazmi, A.; Trianda, Y.; Ramayandi, R.; Yamada, T.; Li, F. Enhancement of drinking water treatment by combined filtration-ICPS: Integrated, based on EEMS, DOC, UV260 and removal pathogenic bacteria. Int. J. Technol. 2019, 10, 593–602. [Google Scholar] [CrossRef] [Green Version]
- Vorontsov, A.V. Advancing Fenton and photo-Fenton water treatment through the catalyst design. J. Hazard. Mater. 2019, 372, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Ya, V.; Chen, Y.C.; Chou, Y.H.; Choo, K.H.; Liu, J.C.; Mameda, N.; Li, C.W. Cryolite (Na3AlF6) crystallization for fluoride recovery using electrolytic process equipped with a sacrificial aluminum anode. J. Hazard. Mater. 2019, 326, 90–96. [Google Scholar] [CrossRef]
- Yacouba, Z.A.; Mendret, J.; Lesage, G.; Zaviska, F.; Brosillon, S. Removal of organic micropollutants from domestic wastewater: The effect of ozone-based advanced oxidation process on nanofiltration. J. Water Process Eng. 2021, 39, 101869. [Google Scholar] [CrossRef]
- PSDA Provinsi Sumatra Barat, Rainfall Statistics. 2019. Available online: http://psda.sumbarprov.go.id/details/hujan (accessed on 15 October 2019).
- Zhang, Y.; Gao, G.; Shi, K.; Niu, C.; Zhou, Y.; Qin, B.; Liu, X. Absorption and fluorescence characteristics of rainwater CDOM and contribution to Lake Taihu, China. Atmos. Environ. 2014, 98, 483–491. [Google Scholar] [CrossRef]
- Coble, P.G.; Del Castillo, C.E.; Avril, B. Distribution of CDOM in the Arabian Sea during the 1995 Southwest Monsoon. Deep-Sea Res. Part II 1998, 45, 2195–2223. [Google Scholar] [CrossRef]
- Kowalczuk, P.; Durako, M.J.; Young, H.; Kahn, A.E.; Cooper, W.J.; Gonsior, M. Characterization of dissolved organic matter fluorescence in the South Atlantic Bight with use of PARAFAC model: Interannual variability. Mar. Chem. 2009, 113, 182–196. [Google Scholar] [CrossRef]
- Fellman, J.B.; Hood, E.; Spencer, R.G.M. Fluorescence spectroscopy opens new windows into dissolved organic matter dynamics in freshwater ecosystems: A review. Limnol. Oceanogr. 2010, 55, 2452–2462. [Google Scholar] [CrossRef]
- Salve, P.R.; Lohkare, H.; Gobre, T.; Bodhe, G.; Krupadam, R.J.; Ramteke, D.S.; Wate, S.R. Characterization of chromophoric dissolved organic matter (CDOM) in rainwater using fluorescence spectrophotometry. Bull. Environ. Contam. Toxicol. 2012, 88, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Santos, P.S.; Duarte, R.M.; Duarte, A.C. Absorption and fluorescence properties of rainwater during the cold season at a town in Western Portugal. J. Atmos. Chem. 2009, 62, 45–57. [Google Scholar] [CrossRef]
- Liu, J.F.; Song, Z.G.; Xu, T. Study on ionic composition of rainwater at Guangzhou and the primary factors of rainwater acidity. Huan Jing Ke Xue = Huanjing Kexue 2002, 27, 1998–2002. [Google Scholar]
- Lamsal, R.; Walsh, M.E.; Gagnon, G.A. Comparison of advanced oxidation process for the removal of natural organic matter. Water Res. 2011, 45, 3263–3269. [Google Scholar] [CrossRef]
- Lee, M.; Zimmermann-Steffens, S.G.; Arey, J.S.; Fenner, K.; von Gunten, U. Development of prediction models for the reactivity of organic compounds with ozone in aqueous solution by quantum chemical calculations: The role of delocalized and localized molecular orbitals. Environ. Sci. Technol. 2015, 49, 9925–9935. [Google Scholar] [CrossRef]
- Azaïs, A.; Mendret, J.; Cazals, G.; Petit, E.; Brosillon, S. Ozonation as a pretreatment process for nanofiltration brines: Monitoring of transformation products and toxicity evaluation. J. Hazard. Mater. 2017, 338, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.; Taurozzi, J.S.; Tarabara, V.V. Ozonation as pretreatment for nanofiltration: Effect of oxidation pathway on the permeate flux. Sep. Purif. Technol. 2015, 149, 174–182. [Google Scholar] [CrossRef]
- Ike, I.A.; Lee, Y.; Hur, J. Impacts of advanced oxidation processes on disinfection by-products from dissolved organic matter upon post-chlor(am)ination: A critical review. Chem. Eng. J. 2019, 375, 121929. [Google Scholar] [CrossRef]
- Zhang, S.; Rouge, V.; Gutierrez, L.; Croue, J.P. Reactivity of chromophoric dissolved organic matter (CDOM) to sulfate radicals: Reaction kinetics and structural transformation. Water Res. 2019, 163, 114846. [Google Scholar] [CrossRef]
- Wang, H.; Quan, B.; Bo, G.; Zhang, Y.; Liu, L.; Zhang, J.; Zhang, X.; Zhang, C. Advanced oxidation treatment of dissolved organic matter from wastewater treatment plant secondary effluent using scattering electrical reactor. J. Clean. Prod. 2020, 267, 122258. [Google Scholar] [CrossRef]
- Maghsoodi, M.; Jacquin, C.; Teychené, B.; Heran, M.; Tarabara, V.V.; Lesage, G.; Snow, S.D. Emerging investigator series: Photocatalysis for MBR effluent post-treatment: Assessing the effects of effluent organic matter characteristics. Environ. Sci. Water Res. Technol. 2019, 5, 482–484. [Google Scholar] [CrossRef]
- Sid, S.; Volant, A.; Lesage, G.; Heran, M. Cost minimization in a full-scale conventional wastewater treatment plant: Associated costs of biological energy consumption versus sludge production. Water Sci. Technol. 2017, 76, 2473–2481. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Li, P.; Tang, X.; Korshin, G.V. Ozonation effects on emerging micropollutants and effluent organic matter in wastewater: Characterization using changes of three-dimensional HP-SEC and EEM fluorescence data. Environ. Sci. Pollut. Res. 2016, 23, 20567–20579. [Google Scholar] [CrossRef]
- Rozario-Ortiz, F.L.; Wert, E.C.; Snyder, S.A. Evaluation of UV/H2O2 treatment for the oxidation of pharmaceuticals in wastewater. Water Res. 2010, 44, 1440–1448. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazmi, A.; Rosadi, M.Y.; Desmiarti, R.; Li, F. Effect of Radio-Frequency Treatment on the Changes of Dissolved Organic Matter in Rainwater. Water 2022, 14, 111. https://doi.org/10.3390/w14010111
Hazmi A, Rosadi MY, Desmiarti R, Li F. Effect of Radio-Frequency Treatment on the Changes of Dissolved Organic Matter in Rainwater. Water. 2022; 14(1):111. https://doi.org/10.3390/w14010111
Chicago/Turabian StyleHazmi, Ariadi, Maulana Yusup Rosadi, Reni Desmiarti, and Fusheng Li. 2022. "Effect of Radio-Frequency Treatment on the Changes of Dissolved Organic Matter in Rainwater" Water 14, no. 1: 111. https://doi.org/10.3390/w14010111
APA StyleHazmi, A., Rosadi, M. Y., Desmiarti, R., & Li, F. (2022). Effect of Radio-Frequency Treatment on the Changes of Dissolved Organic Matter in Rainwater. Water, 14(1), 111. https://doi.org/10.3390/w14010111





