Study on Heavy Metal Contamination in High Water Table Coal Mining Subsidence Ponds That Use Different Resource Reutilization Methods
Abstract
1. Introduction
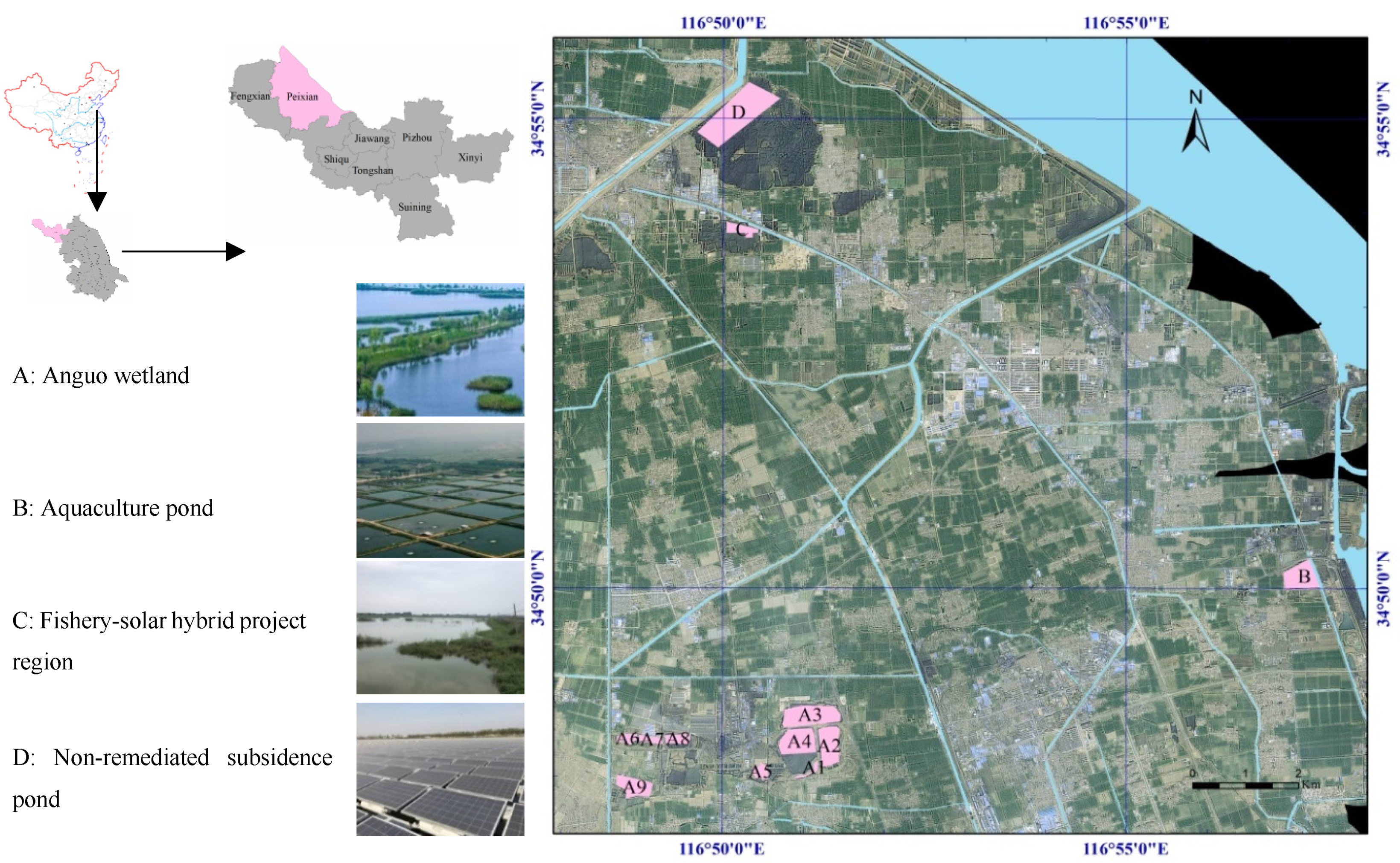
2. Study Area
3. Materials and Methods
3.1. Samples and Preparation
3.2. Pollution Assessment Methods
3.2.1. Sediment Quality Guidelines
3.2.2. Nemerow Synthetic Pollution Index
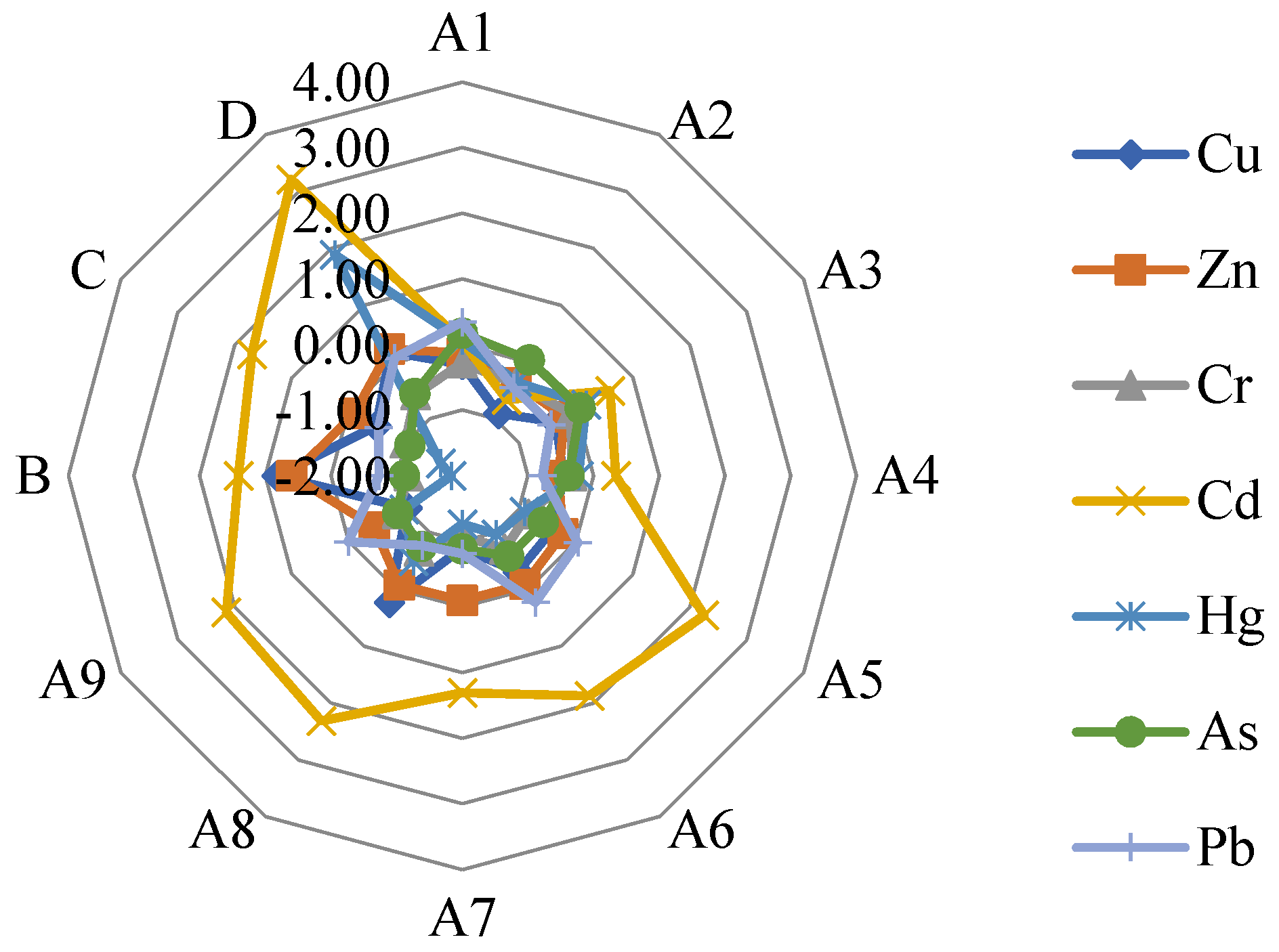
3.2.3. Geoaccumulation Index
3.3. Statistical Analyses
4. Results and Discussion
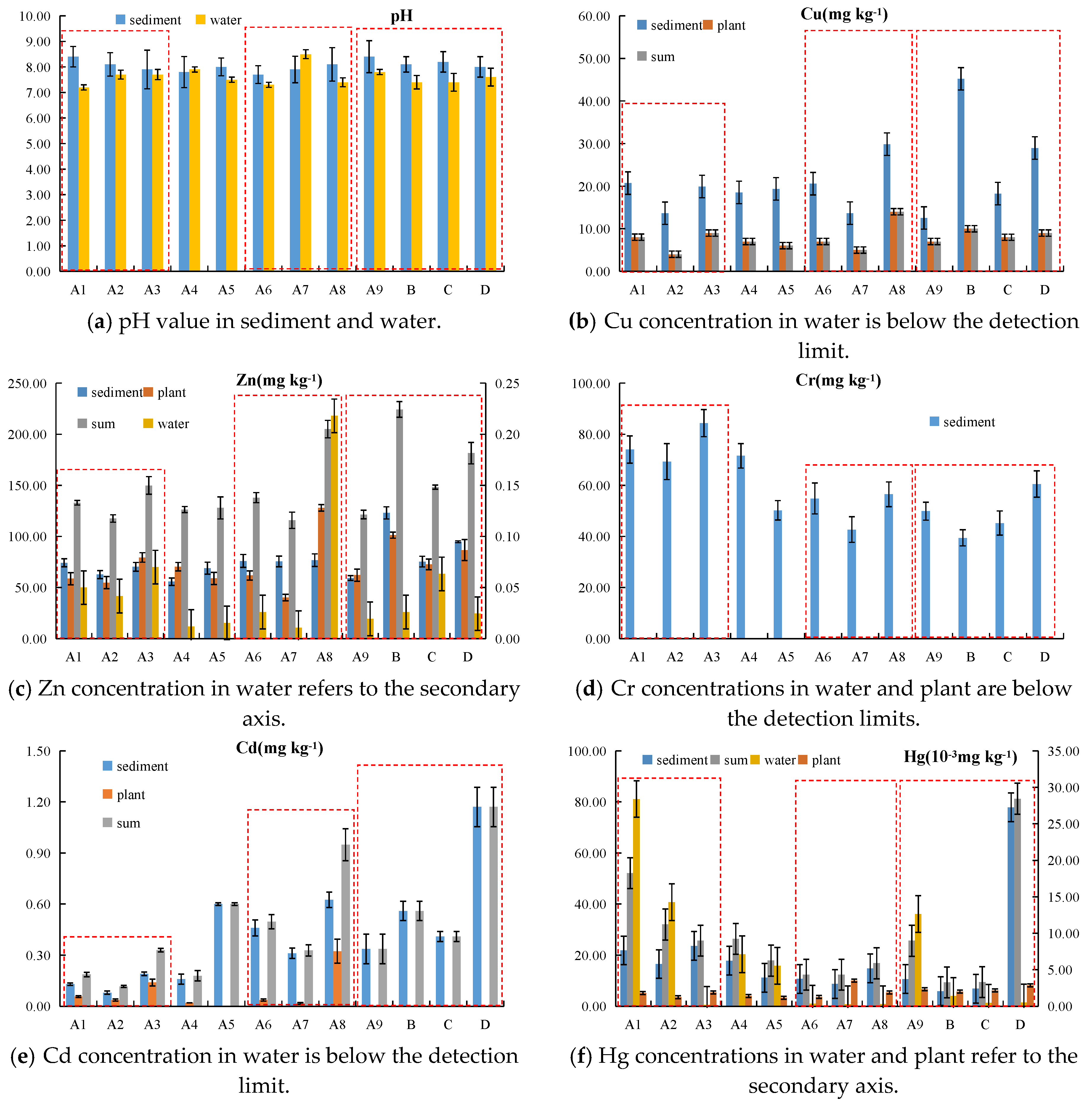
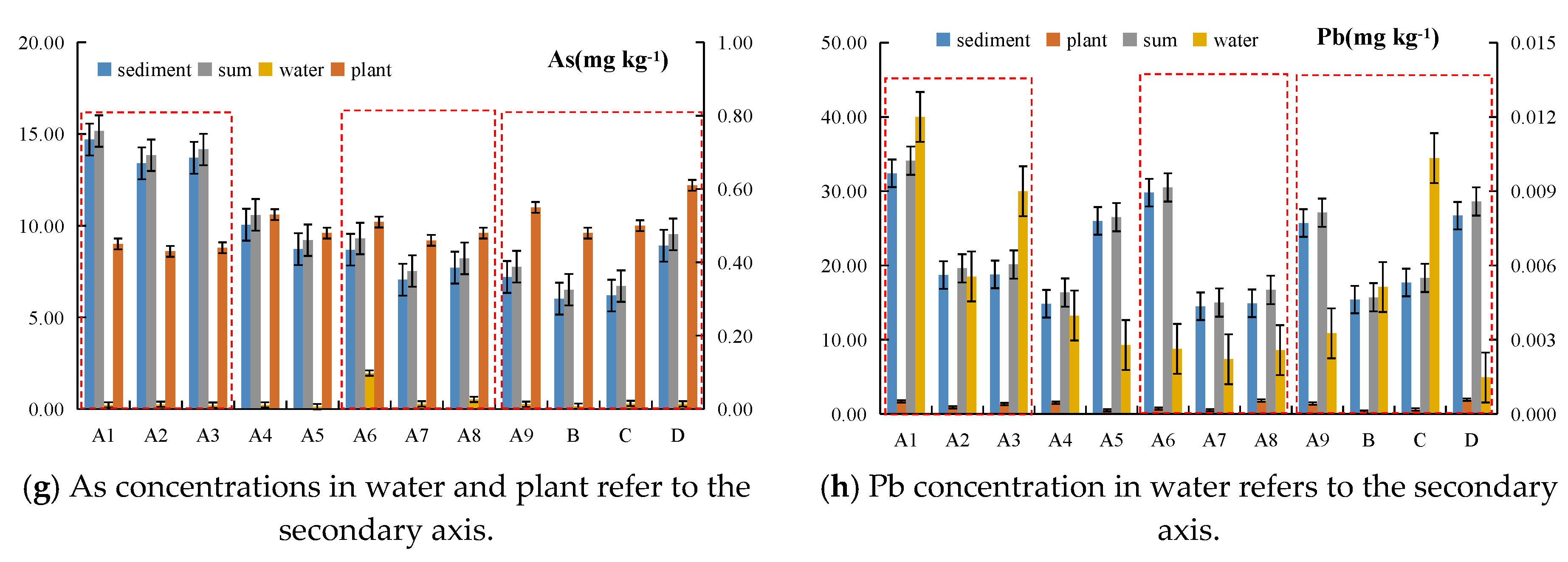
4.1. Assessment of Heavy Metal Contamination
4.2. Heavy Metal Spatial Distribution
4.3. Assessment of Nemerow’s Synthetic Pollution Index
4.4. Geostatistical Analyses of Sediment Heavy Metals
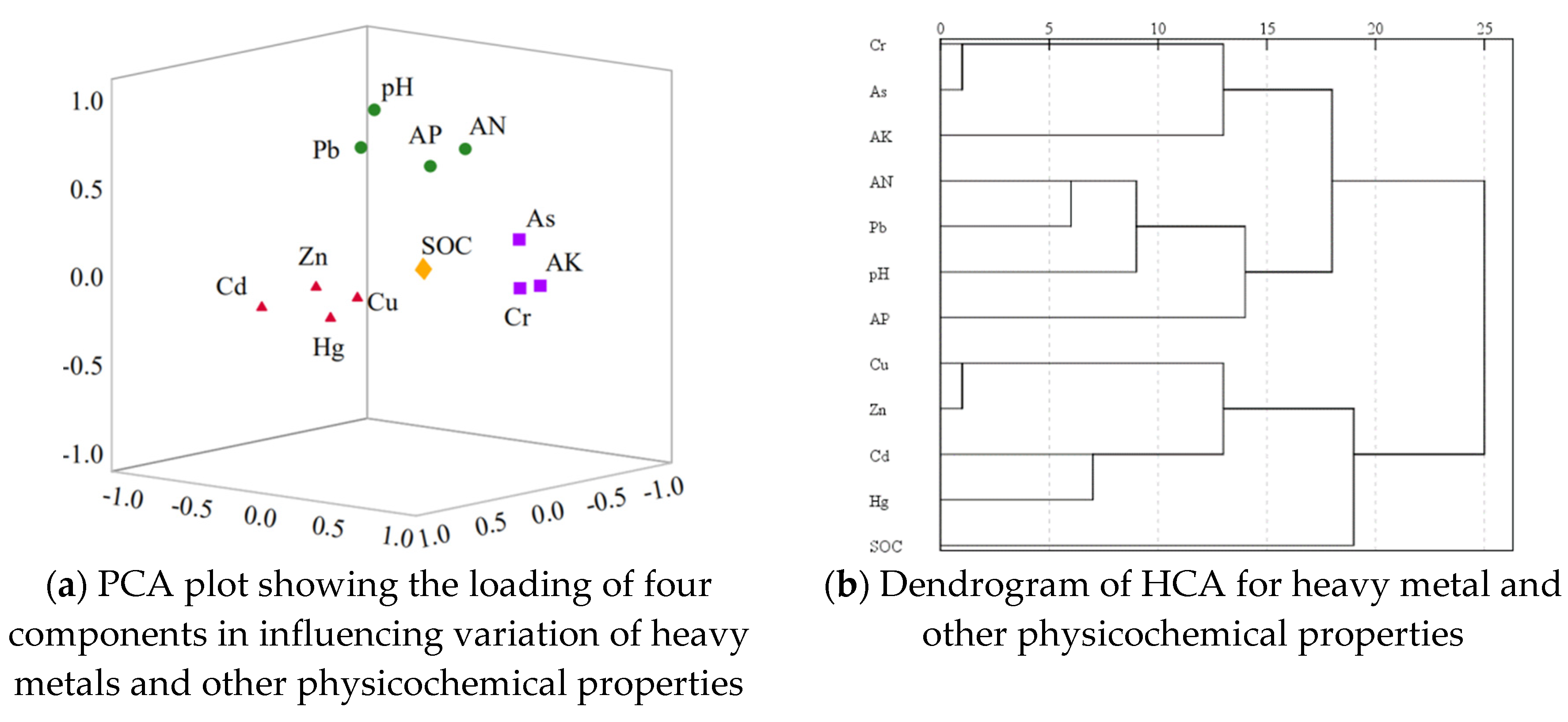
4.5. Pearson’s Correlation, PCA, and HCA
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xiao, W.; Hu, Z.; Chugh, Y.P.; Zhao, Y. Dynamic subsidence simulation and topsoil removal strategy in high groundwater table and underground coal mining area: A case study in Shandong Province. Int. J. Min. Reclam. Environ. 2013, 28, 250–263. [Google Scholar] [CrossRef]
- Hou, Q.; Yang, Z.; Ji, J.; Yu, T.; Chen, G.; Li, J.; Xia, X.; Zhang, M.; Yuan, X. Annual net input fluxes of heavy metals of the agro-ecosystem in the Yangtze River delta, China. J. Geochem. Explor. 2014, 139, 68–84. [Google Scholar] [CrossRef]
- Zhong, S.; Wei, C.; Liu, B.; Zhang, W.; Du, J.; Zhang, S. Restoration technologies of damaged paddy in hilly post-mining and subsidence-stable area of Southwest China. Int. J. Agric. Biol. Eng. 2015, 8, 46–57. [Google Scholar]
- Zhang, Q.; Wang, F.; Wang, R. Research Progress of Ecological Restoration for Wetlands in Coal Mine Areas. Procedia Environ. Sci. 2011, 10, 1933–1938. [Google Scholar] [CrossRef]
- Xie, K.; Zhang, Y.; Yi, Q.; Yan, J. Optimal resource utilization and ecological restoration of aquatic zones in the coal mining subsidence areas of the Huaibei Plain in Anhui Province, China. Desalin. Water Treat. 2013, 51, 4019–4027. [Google Scholar] [CrossRef]
- Vymazal, J.; Švehla, J. Iron and manganese in sediments of constructed wetlands with horizontal subsurface flow treating municipal sewage. Ecol. Eng. 2013, 50, 69–75. [Google Scholar] [CrossRef]
- Yang, J.; Yu, Z.; Yi, P.; Aldahan, A. Assessment of groundwater quality and 222Rn distribution in the Xuzhou region, China. Environ. Monit. Assess. 2018, 190, 549. [Google Scholar] [CrossRef]
- Wang, Z.; Hong, C.; Xing, Y.; Wang, K.; Li, Y.; Feng, L.; Ma, S. Spatial distribution and sources of heavy metals in natural pasture soil around copper-molybdenum mine in Northeast China. Ecotoxicol. Environ. Saf. 2018, 154, 329–336. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China (MEE). Soil Environmental Quality Risk Control Standard for Soil Contamination of Agricultural Land; GB15618-2018; Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2018.
- Zhen, H.; Jia, L.; Huang, C.; Qiao, Y.; Li, J.; Li, H.-F.; Chen, Q.; Wan, Y. Long-term effects of intensive application of manure on heavy metal pollution risk in protected-field vegetable production. Environ. Pollut. 2020, 263, 114552. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, Q.; Deng, M.; Japenga, J.; Li, T.; Yang, X.; He, Z. Heavy metal pollution and health risk assessment of agricultural soils in a typical peri-urban area in southeast China. J. Environ. Manag. 2018, 207, 159–168. [Google Scholar] [CrossRef]
- Muller, G. Index of Geoaccumulation in sediments of the Rhine River. Geo. J. 1969, 2, 108–118. [Google Scholar]
- Loska, K.; Wiechuła, D.; Korus, I. Metal contamination of farming soils affected by industry. Environ. Int. 2004, 30, 159–165. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, S.; Sun, Q.; Wadood, S.A.; Guo, B. Source identification and spatial distribution of arsenic and heavy metals in agricultural soil around Hunan industrial estate by positive matrix factorization model, principle components analysis and geo statistical analysis. Ecotoxicol. Environ. Saf. 2018, 159, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Young, F.J.; Hammer, R. Defining Geographic Soil Bodies by Landscape Position, Soil Taxonomy, and Cluster Analysis. Soil Sci. Soc. Am. J. 2000, 64, 989–998. [Google Scholar] [CrossRef]
- Steinhorst, R.K.; Williams, R.E. Discrimination of Groundwater Sources Using Cluster Analysis, MANOVA, Canonical Analysis and Discriminant Analysis. Water Resour. Res. 1985, 21, 1149–1156. [Google Scholar] [CrossRef]
- Ortega-Guerrero, A. Origin and geochemical evolution of groundwater in a closed-basin clayey aquitard, Northern Mexico. J. Hydrol. 2003, 284, 26–44. [Google Scholar] [CrossRef]
- Tan, M.; Zhao, H.; Li, G.; Qu, J.-F. Assessment of potentially toxic pollutants and urban livability in a typical resource-based city, China. Environ. Sci. Pollut. Res. 2020, 27, 18640–18649. [Google Scholar] [CrossRef]
- Cheng, W.; Bian, Z.; Dong, J.-H.; Lei, S.-G. Soil properties in reclaimed farmland by filling subsidence basin due to underground coal mining with mineral wastes in China. Trans. Nonferr. Met. Soc. China 2014, 24, 2627–2635. [Google Scholar] [CrossRef]
- Li, Y.; Chen, L.; Wen, H. Changes in the composition and diversity of bacterial communities 13 years after soil reclamation of abandoned mine land in eastern China. Ecol. Res. 2014, 30, 357–366. [Google Scholar] [CrossRef]
- Abakumov, E.V.; Cajthaml, T.; Brus, J.; Frouz, J. Humus accumulation, humification, and humic acid composition in soils of two post-mining chronosequences after coal mining. J. Soils Sediments 2012, 13, 491–500. [Google Scholar] [CrossRef]
- Slessarev, E.W.; Lin, Y.; Bingham, N.L.; Johnson, J.E.; Dai, Y.; Schimel, J.P.; Chadwick, O.A. Water balance creates a threshold in soil pH at the global scale. Nat. Cell Biol. 2016, 540, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Yang, J.; Xia, P.; Wu, S.; Lin, T.; Yi, Y. Contamination features and ecological risks of heavy metals in the farmland along shoreline of Caohai plateau wetland, China. Chemosphere 2020, 254, 126828. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.C.F.; Gusso-Choueri, P.; De Araujo, G.S.; Campos, B.G.; Abessa, D.M.D.S. Levels of metals and toxicity in sediments from a Ramsar site influenced by former mining activities. Ecotoxicol. Environ. Saf. 2019, 171, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Wang, Y.; Liu, A.; Yang, H.; Liu, A.; Li, M.; Niu, X. Historical Evolution of Sources and Pollution Levels of Heavy Metals in the Sediment of the Shuanglong Reservoir, China. Water 2020, 12, 1855. [Google Scholar] [CrossRef]
- Williams, C.; David, D. The effect of superphosphate on the cadmium content of soils and plants. Soil Res. 1973, 11, 43–56. [Google Scholar] [CrossRef]
- Bi, X.; Feng, X.; Yang, Y.; Li, X.; Shin, G.P.; Li, F.; Qiu, G.; Li, G.; Liu, T.; Fu, Z. Allocation and source attribution of lead and cadmium in maize (Zea mays L.) impacted by smelting emissions. Environ. Pollut. 2009, 157, 834–839. [Google Scholar] [CrossRef]
- Krčmar, D.; Grba, N.; Isakovski, M.K.; Varga, N.; Spasojević, J.; Agbaba, J.; Dalmacija, B.D. Multicriteria to estimate the environmental risk of sediment from the Obedska Bog (Northern Serbia), a reservation area on UNESCO’s list. Int. J. Sediment Res. 2020, 35, 527–539. [Google Scholar] [CrossRef]
- Gao, Z. Evaluation of heavy metal pollution and its ecological risk in one river reach of a gold mine in Inner Mongolia, Northern China. Int. Biodeterior. Biodegrad. 2018, 128, 94–99. [Google Scholar] [CrossRef]
- Eid, E.M.; Shaltout, K.H.; Al-Sodany, Y.M.; Haroun, S.A.; Galal, T.M.; Ayed, H.; Khedher, K.M.; Jensen, K. Common reed (Phragmites australis (Cav.) Trin. ex Steudel) as a candidate for predicting heavy metal contamination in Lake Burullus, Egypt: A biomonitoring approach. Ecol. Eng. 2020, 148, 148. [Google Scholar] [CrossRef]
- Ju, Y.-R.; Chen, C.-F.; Chuang, X.-Y.; Lim, Y.C.; Chen, C.-W.; Dong, C.-D. Biometry-dependent metal bioaccumulation in aquaculture shellfishes in southwest Taiwan and consumption risk. Chemosphere 2020, 253, 126685. [Google Scholar] [CrossRef]
- Thangaraj, S.; Yuvaraj, P.; Damotharan, P.; Karthikeyan, V.; Jha, D.K.; Dharani, G.; Balasubramanian, T.; Kirubagaran, R. Assessment of trace metal contamination in the marine sediment, seawater, and bivalves of Parangipettai, southeast coast of India. Mar. Pollut. Bull. 2019, 149, 149. [Google Scholar] [CrossRef]
- Rahim, M.; Yoshino, J.; Yasuda, T. Evaluation of solar radiation abundance and electricity production capacity for application and development of solar energy. Int. J. Energy Environ. 2012, 3, 687–700. [Google Scholar]
- Şener, Ş.E.C.; Sharp, J.L.; Anctil, A. Factors impacting diverging paths of renewable energy: A review. Renew. Sustain. Energy Rev. 2018, 81, 2335–2342. [Google Scholar] [CrossRef]
- Chen, G.; Wang, X.; Wang, R.; Liu, G. Health risk assessment of potentially harmful elements in subsidence water bodies using a Monte Carlo approach: An example from the Huainan coal mining area, China. Ecotoxicol. Environ. Saf. 2019, 171, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, R.D.; Tripathi, P.; Dwivedi, S.; Kumar, A.; Mishra, A.; Chauhan, P.S.; Norton, G.J.; Nautiyal, C.S. Roles for root iron plaque in sequestration and uptake of heavy metals and metalloids in aquatic and wetland plants. Metallomics 2014, 6, 1789–1800. [Google Scholar] [CrossRef]
- Fawzy, M.; Badr, N.E.-S.; El-Khatib, A.; Abo-El-Kassem, A. Heavy metal biomonitoring and phytoremediation potentialities of aquatic macrophytes in River Nile. Environ. Monit. Assess. 2012, 184, 1753–1771. [Google Scholar] [CrossRef]
- Pramer, D. Soil Biology. Soil Sci. Soc. Am. J. 1968, 32, 345–368. [Google Scholar] [CrossRef]
- Kamala-Kannan, S.; Batvari, B.P.D.; Lee, K.J.; Kannan, N.; Krishnamoorthy, R.; Shanthi, K.; Jayaprakash, M. Assessment of heavy metals (Cd, Cr and Pb) in water, sediment and seaweed (Ulva lactuca) in the Pulicat Lake, South East India. Chemosphere 2008, 71, 1233–1240. [Google Scholar] [CrossRef]
- Barlas, N.; Akbulut, N.; Aydoğan, M. Assessment of Heavy Metal Residues in the Sediment and Water Samples of Uluabat Lake, Turkey. Bull. Environ. Contam. Toxicol. 2005, 74, 286–293. [Google Scholar] [CrossRef]
- MacKenzie, A.; Pulford, I. Investigation of contaminant metal dispersal from a disused mine site at Tyndrum, Scotland, using concentration gradients and stable Pb isotope ratios. Appl. Geochem. 2002, 17, 1093–1103. [Google Scholar] [CrossRef]
- Li, Q.; Cheng, H.; Zhou, T.; Lin, C.; Guo, S. The estimated atmospheric lead emissions in China, 1990–2009. Atmos. Environ. 2012, 60, 1–8. [Google Scholar] [CrossRef]
- Feng, J.; Wang, Y.; Zhao, J.; Zhu, L.; Bian, X.; Zhang, W. Source attributions of heavy metals in rice plant along highway in Eastern China. J. Environ. Sci. 2011, 23, 1158–1164. [Google Scholar] [CrossRef]
- Li, B.; Xiao, R.; Wang, C.-Q.; Cao, L.; Zhang, Y.; Zheng, S.; Yang, L.; Guo, Y. Spatial distribution of soil cadmium and its influencing factors in peri-urban farmland: A case study in the Jingyang District, Sichuan, China. Environ. Monit. Assess. 2016, 189. [Google Scholar] [CrossRef] [PubMed]
- Equeenuddin, S.M.; Tripathy, S.; Sahoo, P.K.; Panigrahi, M. Metal behavior in sediment associated with acid mine drainage stream: Role of pH. J. Geochem. Explor. 2013, 124, 230–237. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Y.; Chen, Q.; Li, Y.; Guo, D.; Nie, X.; Peng, X. Assessment of heavy metal pollution and the effect on bacterial community in acidic and neutral soils. Ecol. Indic. 2020, 117, 106626. [Google Scholar] [CrossRef]
- Imoto, Y.; Yasutaka, T. Comparison of the impacts of the experimental parameters and soil properties on the prediction of the soil sorption of Cd and Pb. Geoderma 2020, 376, 114538. [Google Scholar] [CrossRef]
- Bang, J.; Hesterberg, D. Dissolution of Trace Element Contaminants from Two Coastal Plain Soils as Affected by pH. J. Environ. Qual. 2004, 33, 891–901. [Google Scholar] [CrossRef]
- Buchter, B.; Davidoff, B.; Amacher, M.C.; Hinz, C.; Iskandar, I.K.; Selim, H.M. Correlation of freundlich Kd and n retention parameters with soils and elements. Soil Sci. 1989, 148, 370–379. [Google Scholar] [CrossRef]
- Elbana, T.A.; Selim, H.; Akrami, N.; Newman, A.; Shaheen, S.M.; Rinklebe, J. Freundlich sorption parameters for cadmium, copper, nickel, lead, and zinc for different soils: Influence of kinetics. Geoderma 2018, 324, 80–88. [Google Scholar] [CrossRef]
- Yu, H.-Y.; Liu, C.; Zhu, J.; Li, F.; Deng, D.-M.; Wang, Q.; Liu, C. Cadmium availability in rice paddy fields from a mining area: The effects of soil properties highlighting iron fractions and pH value. Environ. Pollut. 2016, 209, 38–45. [Google Scholar] [CrossRef]
- Caporale, A.G.; Violante, A. Chemical Processes Affecting the Mobility of Heavy Metals and Metalloids in Soil Environments. Curr. Pollut. Rep. 2016, 2, 15–27. [Google Scholar] [CrossRef]
- Liao, J.; Deng, S.; Liu, X.; Lin, H.; Yu, C.; Wei, C. Influence of soil evolution on the heavy metal risk in three kinds of intertidal zone of the Pearl River Estuary. Land Degrad. Dev. 2020. [Google Scholar] [CrossRef]
- Juhos, K.; Czigány, S.; Madarász, B.; Ladányi, M. Interpretation of soil quality indicators for land suitability assessment—A multivariate approach for Central European arable soils. Ecol. Indic. 2019, 99, 261–272. [Google Scholar] [CrossRef]
- Mestrinho, S.S.P.; Reitemajer, D.; Porciúncula, D.C.; Lyrio, R. Evaluation of the surface water quality in the Itapicuru river basin—State of Bahia, Brazil. Water Pollut. VIII Model. Monit. Manag. 2007, 103, 133–141. [Google Scholar] [CrossRef]
| Psediment | Pwater | Pplant | P | |
|---|---|---|---|---|
| A1 | 0.55 | 0.84 | 12.56 | 4.65 |
| A2 | 0.49 | 0.94 | 6.68 | 2.70 |
| A3 | 0.51 | 0.77 | 9.85 | 3.71 |
| A4 | 0.38 | 0.81 | 11.07 | 4.09 |
| A5 | 0.56 | 0.48 | 3.81 | 1.62 |
| A6 | 0.44 | 7.19 | 5.22 | 4.28 |
| A7 | 0.30 | 1.03 | 3.75 | 1.69 |
| A8 | 0.58 | 1.92 | 13.51 | 5.33 |
| A9 | 0.52 | 0.96 | 10.54 | 4.01 |
| B | 0.33 | 0.56 | 2.34 | 1.08 |
| C | 0.39 | 1.11 | 4.57 | 2.02 |
| D | 1.06 | 1.03 | 14.43 | 5.51 |
| pH | AN | AP | AK | SOC | Cu | Zn | Cr | Cd | Hg | As | Pb | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| pH | 1.00 | |||||||||||
| AN | 0.51 | 1.00 | ||||||||||
| AP | 0.28 | 0.34 | 1.00 | |||||||||
| AK | −0.09 | 0.15 | −0.01 | 1.00 | ||||||||
| SOC | 0.08 | −0.12 | −0.03 | 0.20 | 1.00 | |||||||
| Cu | −0.02 | −0.35 | −0.26 | 0.08 | 0.36 | 1.00 | ||||||
| Zn | 0.01 | −0.35 | −0.28 | −0.15 | 0.10 | 0.90 ** | 1.00 | |||||
| Cr | −0.10 | 0.49 | 0.16 | 0.38 | −0.07 | −0.26 | −0.44 | 1.00 | ||||
| Cd | −0.11 | −0.40 | −0.28 | −0.48 | 0.21 | 0.53 | 0.55 | −0.39 | 1.00 | |||
| Hg | −0.07 | −0.03 | −0.20 | −0.42 | −0.10 | 0.17 | 0.19 | 0.33 | 0.64 * | 1.00 | ||
| As | 0.06 | 0.65 * | 0.43 | 0.36 | −0.21 | −0.30 | −0.38 | 0.91 ** | −0.50 | 0.22 | 1.00 | |
| Pb | 0.24 | 0.69 * | 0.41 | −0.30 | −0.33 | −0.17 | −0.10 | 0.18 | 0.16 | 0.31 | 0.33 | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, M.; Wang, K.; Xu, Z.; Li, H.; Qu, J. Study on Heavy Metal Contamination in High Water Table Coal Mining Subsidence Ponds That Use Different Resource Reutilization Methods. Water 2020, 12, 3348. https://doi.org/10.3390/w12123348
Tan M, Wang K, Xu Z, Li H, Qu J. Study on Heavy Metal Contamination in High Water Table Coal Mining Subsidence Ponds That Use Different Resource Reutilization Methods. Water. 2020; 12(12):3348. https://doi.org/10.3390/w12123348
Chicago/Turabian StyleTan, Min, Kun Wang, Zhou Xu, Hanghe Li, and Junfeng Qu. 2020. "Study on Heavy Metal Contamination in High Water Table Coal Mining Subsidence Ponds That Use Different Resource Reutilization Methods" Water 12, no. 12: 3348. https://doi.org/10.3390/w12123348
APA StyleTan, M., Wang, K., Xu, Z., Li, H., & Qu, J. (2020). Study on Heavy Metal Contamination in High Water Table Coal Mining Subsidence Ponds That Use Different Resource Reutilization Methods. Water, 12(12), 3348. https://doi.org/10.3390/w12123348




