Artificial Floating Island with Vetiver for Treatment of Arsenic-Contaminated Water: A Real Scale Study in High-Andean Reservoir
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Materials
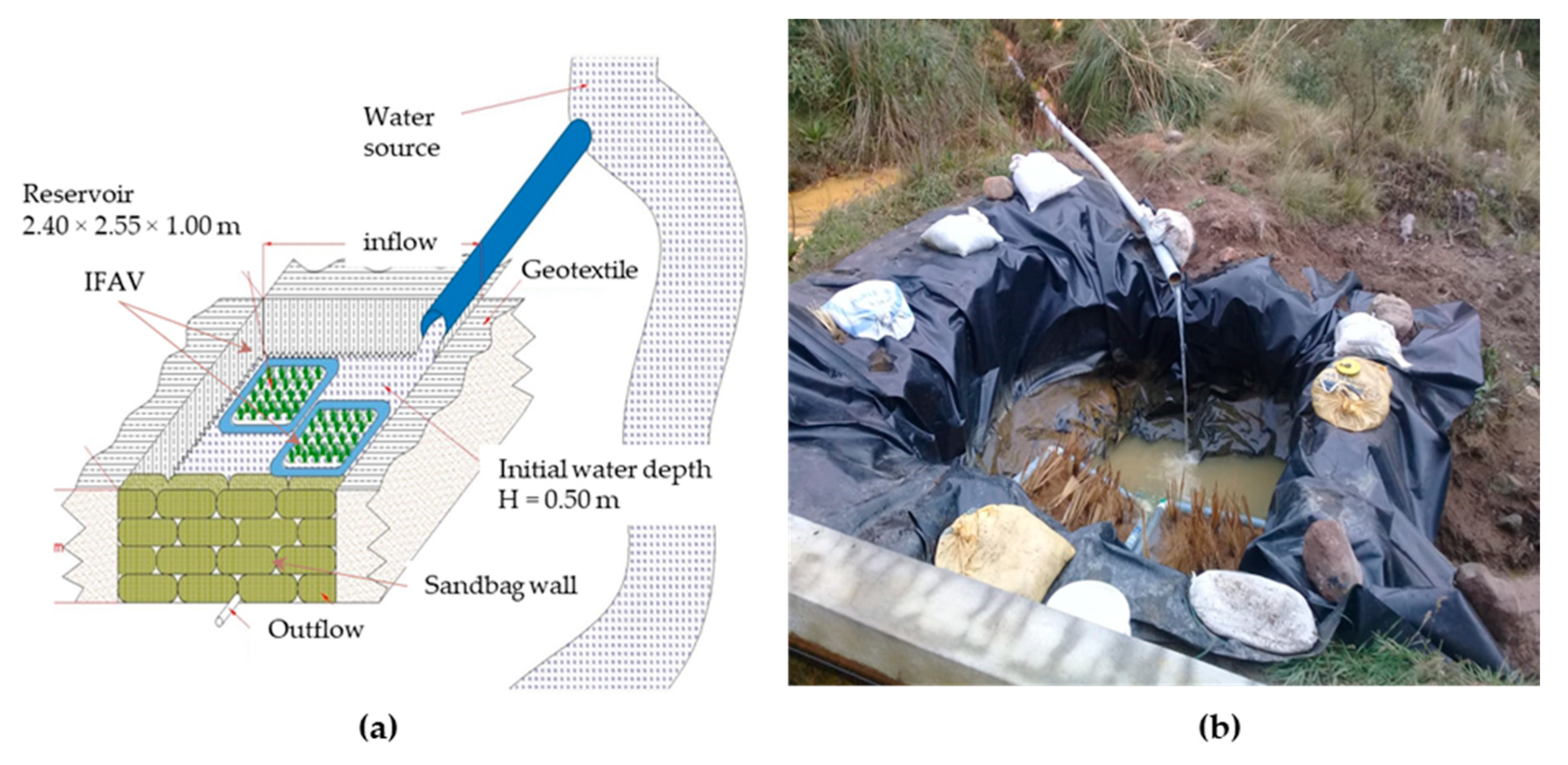
2.2. Experimental Setup and Procedure
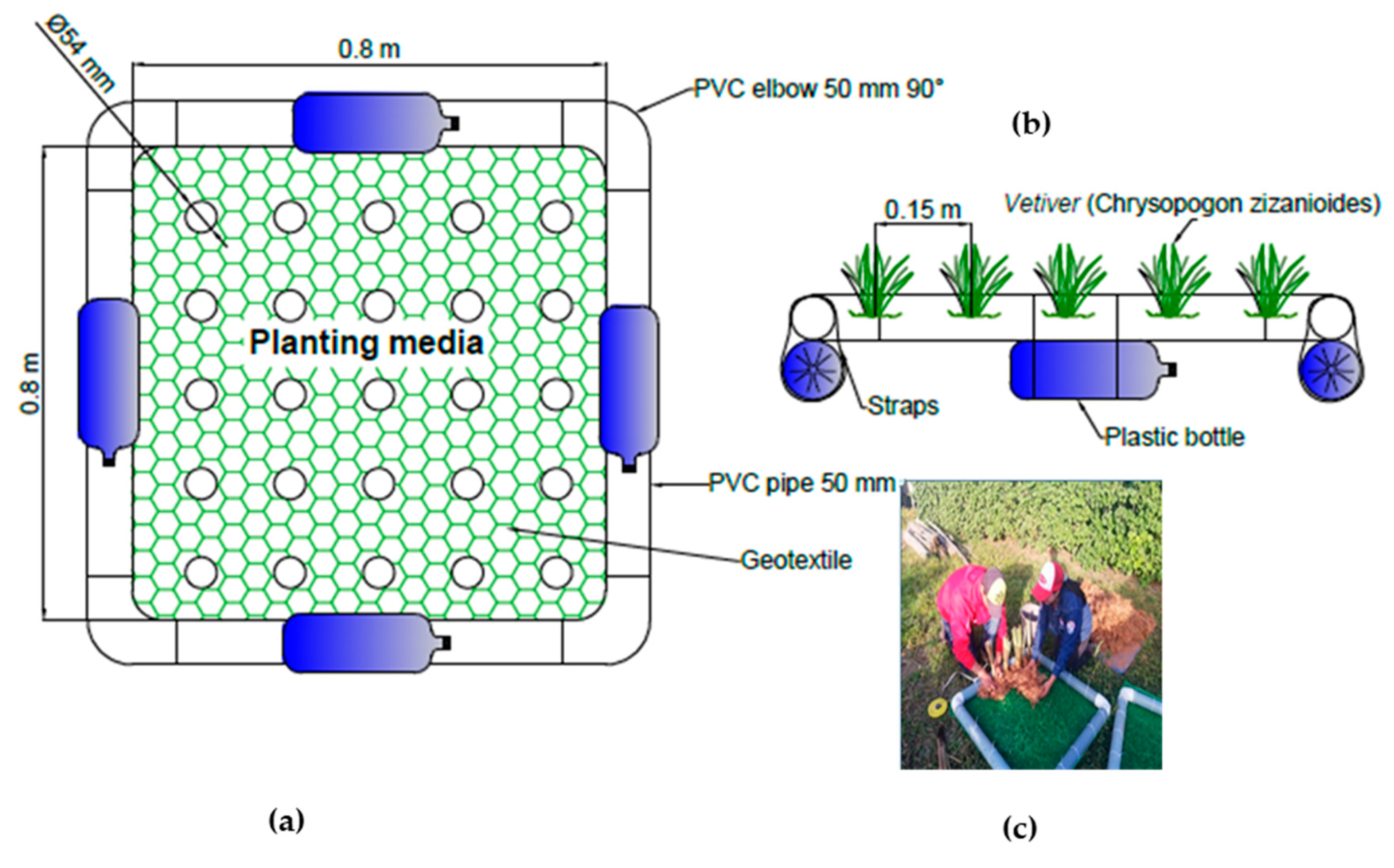
2.2.1. Floating Artificial Island Design and Construction
2.2.2. Adaptation of Chrysopogon zizanioides in the Páramo and Installation in the Floating System
2.3. Sample Analysis
2.3.1. Sample Collection
2.3.2. Water and Sediments Samples Analysis
2.3.3. Hydrometeorological Data
2.3.4. Estimation of heavy metals in Vetiver plants and its phytoextraction ability
3. Results
3.1. Plant Adaptation in the Floating System
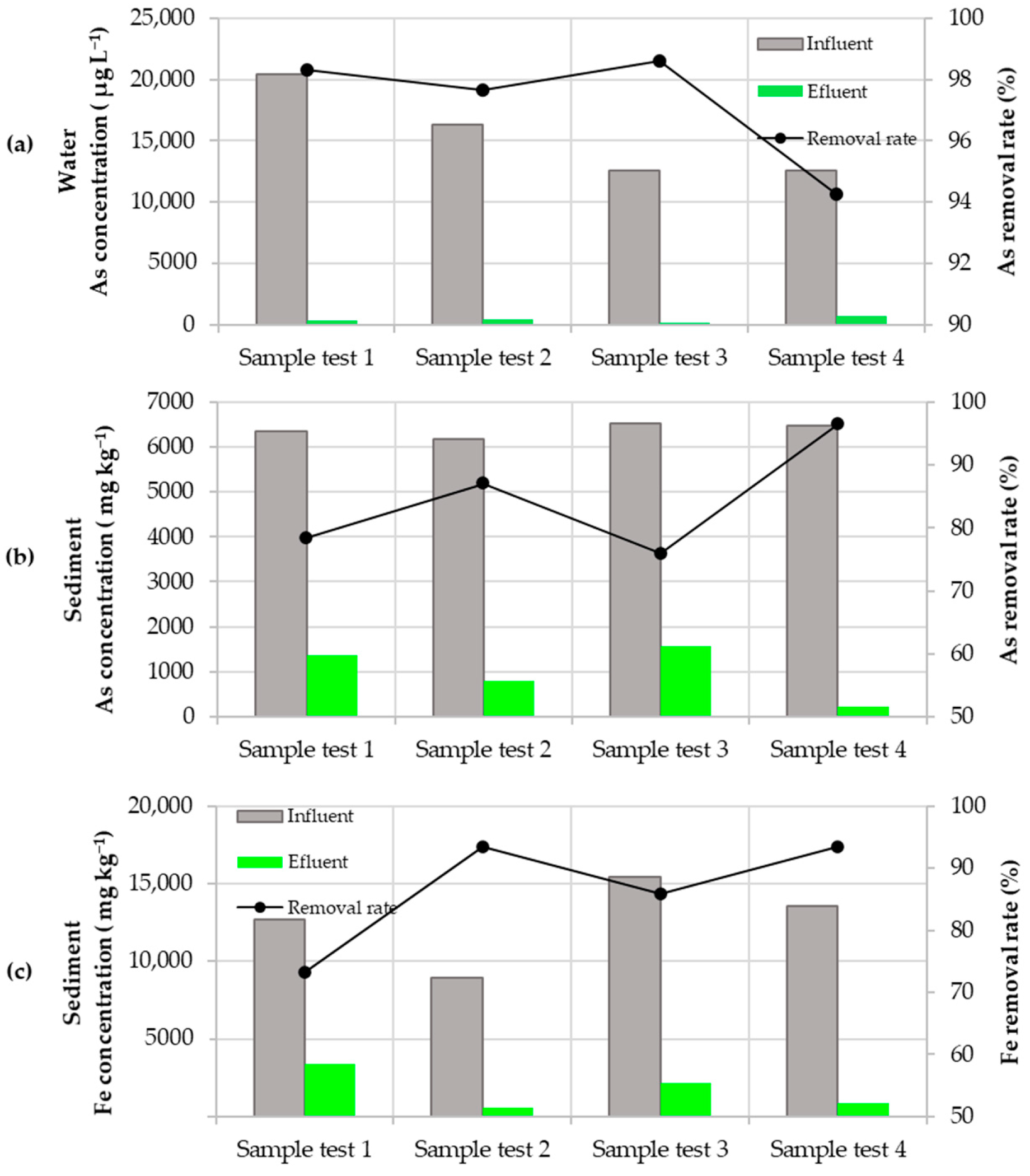
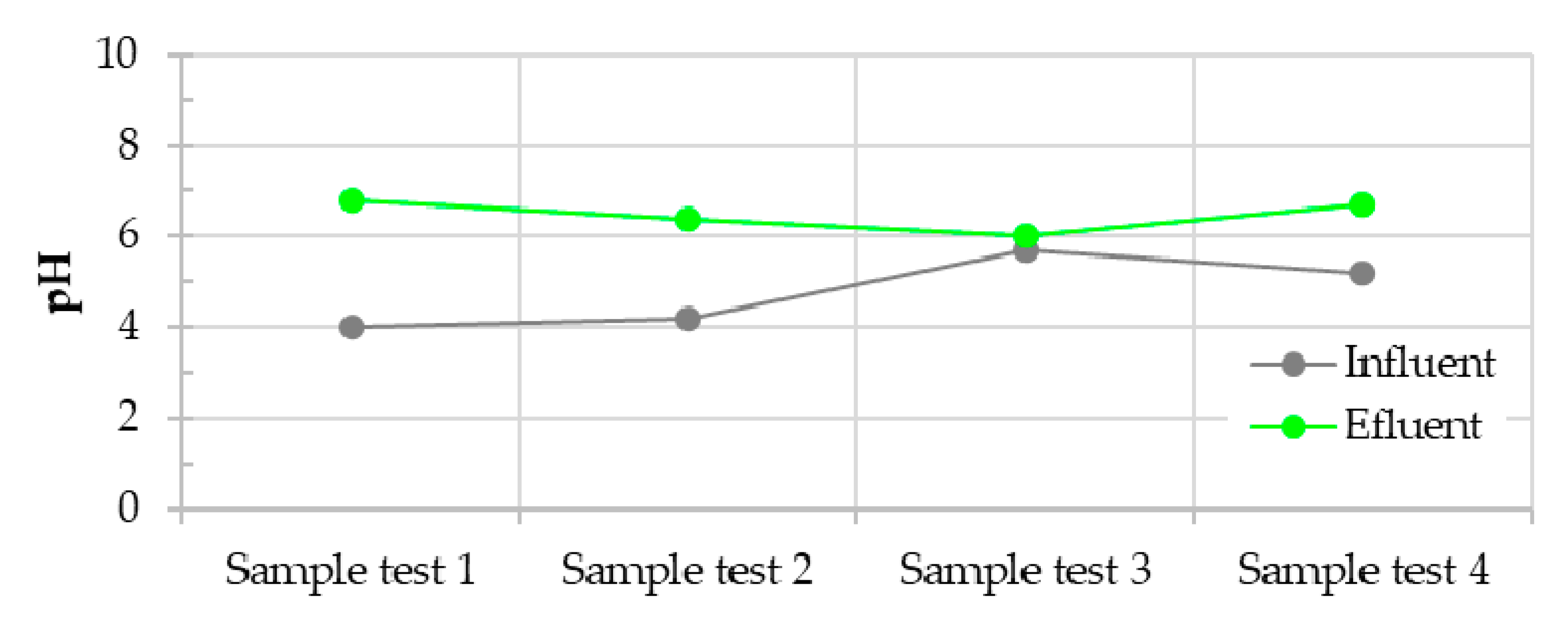
3.2. Water Quality Assessment
3.3. Translocation of As into the tissues of the Vetiver
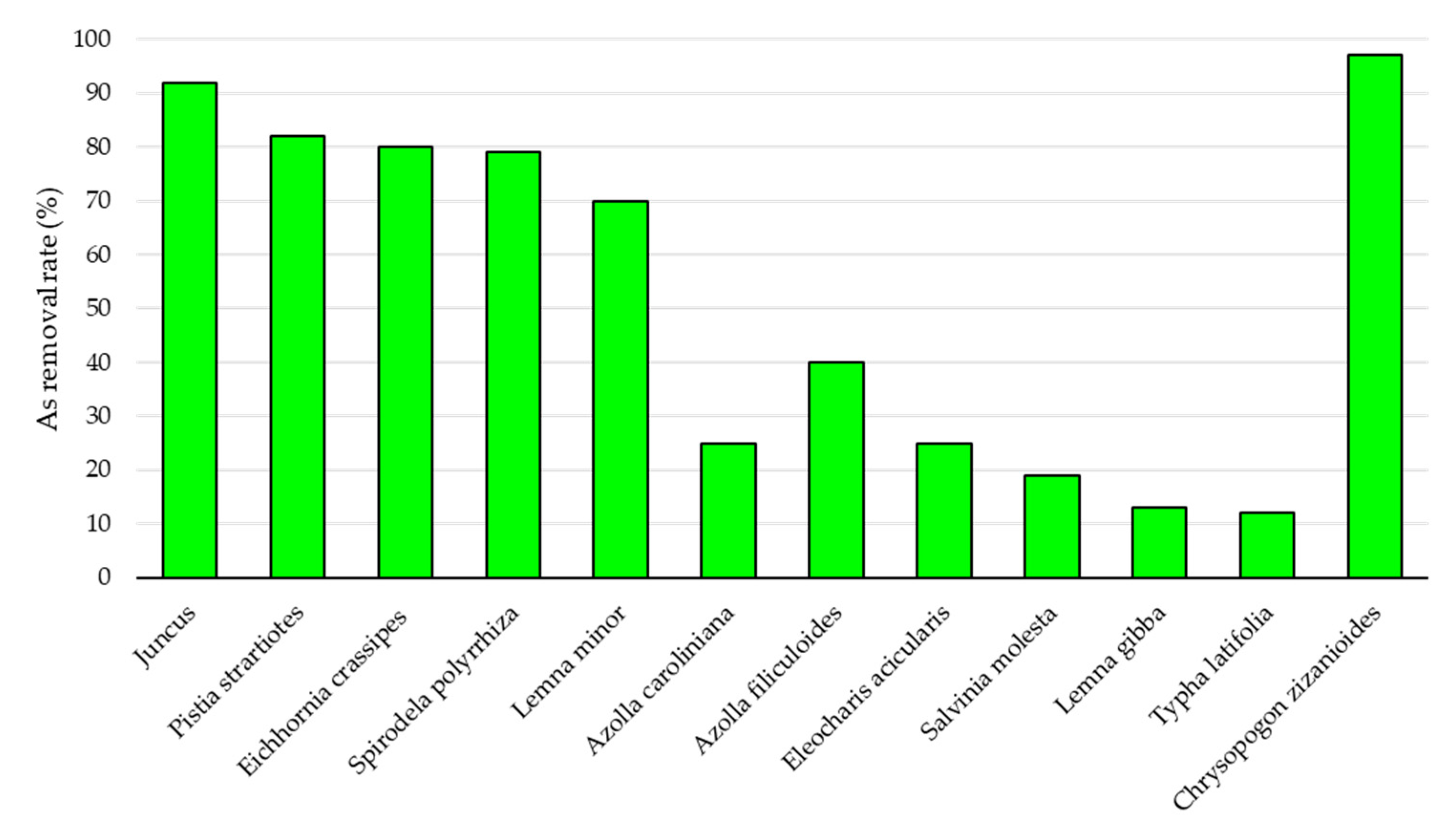
3.4. Use of AFIV in AWI of High Andean Ecosystems
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mondal, P.; Majumder, C.B.; Mohanty, B. Laboratory based approaches for arsenic remediation from contaminated water: Recent developments. J. Hazard. Mater. 2006, 137, 464–479. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, D.N.G. Chronic arsenic toxicity & human health. Indian J. Med. Res. 2008, 128, 12. [Google Scholar]
- Srivastava, S.; Suprasanna, P.; D’Souza, S.F. Mechanisms of arsenic tolerance and detoxification in plants and their application in transgenic technology: A critical appraisal. Int. J. Phytoremediat. 2012, 14, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Brammer, H.; Ravenscroft, P. Arsenic in groundwater: A threat to sustainable agriculture in South and South-east Asia. Environ. Int. 2009, 35, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, D.K. Worldwide occurrences of arsenic in ground water. Sci. New Ser. 2002, 296, 2143–2145. [Google Scholar]
- Bundschuh, J.; Armienta, M.; Birkle, P.; Bhattacharya, P.; Matschullat, J.; Mukherjee, A. (Eds.) Natural Arsenic in Groundwaters of Latin America; CRC Press: London, UK, 2009. [Google Scholar] [CrossRef]
- Tovar, C.; Zapata, C.; Bernal, C.; Diaz, X. Anomalias de arsénico en las fuentes de agua de las zonas de la parroquia Toacaso aledañas al complejo volcanico los Ilinizas. In Proceedings of the I Congreso Latinoamericano y V Congreso Nacional de Manejo de Cuencas Hidrográficas, Mexico City, Mexico, 29–31 October 2019. [Google Scholar]
- Ministerio de Agricultura y Ganaderia. Informe de Rendimientos Objetivos de Papa en el Ecuador 2018; Ministerio de Agricultura y Ganaderia: Quito, Ecuador, 2018.
- Padmanabhan, P.; Sullivan, J.A.; Paliyath, G. Potatoes and related crops. In Encyclopedia of Food and Health; Elsevier: Oxford, UK, 2016; pp. 446–451. ISBN 978-0-12-384953-3. [Google Scholar]
- Ho, L.T.; Goethals, P.L.M. Opportunities and Challenges for the Sustainability of Lakes and Reservoirs in Relation to the Sustainable Development Goals (SDGs). Water 2019, 11, 1462. [Google Scholar] [CrossRef]
- Rasool, A.; Xiao, T.; Farooqi, A.; Shafeeque, M.; Masood, S.; Ali, S.; Fahad, S.; Nasim, W. Arsenic and heavy metal contaminations in the tube well water of Punjab, Pakistan and risk assessment: A case study. Ecol. Eng. 2016, 95, 90–100. [Google Scholar] [CrossRef]
- Banerjee, R. Vetiver grass is a potential candidate for phytoremediation of iron ore mine spoil dumps. Ecol. Eng. 2019, 132, 120–136. [Google Scholar] [CrossRef]
- Zhao, F.-J.; McGrath, S.P.; Meharg, A.A. Arsenic as a Food Chain Contaminant: Mechanisms of Plant Uptake and Metabolism and Mitigation Strategies. Annu. Rev. Plant Biol. 2010, 61, 535–559. [Google Scholar] [CrossRef]
- Pilon-Smits, E. Phytoremediation. Annu. Rev. Plant Biol. 2005, 56, 15–39. [Google Scholar] [CrossRef]
- Franco, E. Soluciones Basadas en la Naturaleza Son Clave Para Adaptarse al Cambio Climático. Available online: https://latinclima.org/articulos/soluciones-basadas-en-la-naturaleza-son-clave-para-adaptarse-al-cambio-climatico (accessed on 29 April 2020).
- Langergraber, G.; Pucher, B.; Simperler, L.; Kisser, J.; Katsou, E.; Buehler, D.; Mateo, M.C.G.; Atanasova, N. Implementing nature-based solutions for creating a resourceful circular city. Blue Green Syst. 2020, 2, 173–185. [Google Scholar] [CrossRef]
- Colares, G.S.; Dell’Osbel, N.; Wiesel, P.G.; Oliveira, G.A.; Lemos, P.H.Z.; da Silva, F.P.; Lutterbeck, C.A.; Kist, L.T.; Machado, Ê.L. Floating treatment wetlands: A review and bibliometric analysis. Sci. Total Environ. 2020, 714, 136776. [Google Scholar] [CrossRef]
- Yeh, N.; Yeh, P.; Chang, Y.-H. Artificial floating islands for environmental improvement. Renew. Sustain. Energy Rev. 2015, 47, 616–622. [Google Scholar] [CrossRef]
- Martelo, J.; Borrero, J.A.L. Macrófitas flotantes en el tratamiento de aguas residuales: Una revisión del estado del arte. Ing. Cienc. 2012, 8, 221–243. [Google Scholar] [CrossRef]
- Stewart, F.M.; Muholland, T.; Cunningham, A.B.; Kania, B.G.; Osterlund, M.T. Floating islands as an alternative to constructed wetlands for treatment of excess nutrients from agricultural and municipal wastes–results of laboratory-scale tests. Land Contam. Reclam. 2008, 16, 25–33. [Google Scholar] [CrossRef]
- De Stefani, G.; Tocchetto, D.; Salvato, M.; Borin, M. Performance of a floating treatment wetland for in-stream water amelioration in NE Italy. Hydrobiologia 2011, 674, 157–167. [Google Scholar] [CrossRef]
- Garbett, P. An investigation into the application of floating reed bed and barley straw techniques for the remediation of eutrophic waters. Water Environ. J. 2005, 19, 174–180. [Google Scholar] [CrossRef]
- Tanner, C.C.; Headley, T.R. Components of floating emergent macrophyte treatment wetlands influencing removal of stormwater pollutants. Ecol. Eng. 2011, 37, 474–486. [Google Scholar] [CrossRef]
- Siyar, R.; Doulati Ardejani, F.; Farahbakhsh, M.; Norouzi, P.; Yavarzadeh, M.; Maghsoudy, S. Potential of Vetiver grass for the phytoremediation of a real multi-contaminated soil, assisted by electrokinetic. Chemosphere 2020, 246, 125802. [Google Scholar] [CrossRef]
- Shu, W.; Xia, H. Integrated vetiver technique for remediation of heavy metal contamination: Potential and practice. In Proceedings of the Third International Vetiver Conference, Guangzhou, China, 6–9 October 2003; p. 13. [Google Scholar]
- Wang, L.K.; Hung, Y.-T.; Tay, J.-H.; Tay, S.T.L. Environmental Bioengineering: Volume 11; Series: Handbook of environmental engineering; Humana Press: New York, NY, USA, 2010; ISBN 978-1-58829-493-7. [Google Scholar]
- Lara, S.; Navarro, R. Resultados y Lecciones en Sistema Vetiver para descontaminación de agua y aumento de su disponibilidad para riego; Serie Experiencias de Innovación para el Emprendimiento Agrario; Fundación Para la Innovación Agraria: Santiago de Chile, Chile, 2017; ISBN 978-956-328-213-9. [Google Scholar]
- Singh, S.; Sounderajan, S.; Kumar, K.; Fulzele, D.P. Investigation of arsenic accumulation and biochemical response of in vitro developed Vetiveria zizanoides plants. Ecotoxicol. Environ. Saf. 2017, 145, 50–56. [Google Scholar] [CrossRef]
- Smolcz, S.U.; Cortés, V.G. Remediation of boron contaminated water and soil with vetiver phytoremediation technology in Northern Chile. In Proceedings of the 6th International Conference on Vetiver (ICV6), Da Nang, Vietnam, 5–8 May 2015; pp. 5–8. [Google Scholar]
- Gobierno Autónomo Descentralizado de la Provincia de Cotopaxi. Plan de Desarrollo y Ordenamiento Territorial Cotopaxi 2015–2025; Cotopaxi: Latacunga, Ecuador, 2015. [Google Scholar]
- Pincha, G. Diagnóstico de la Parroquia de Toacaso; Gobierno Autónomo Descentralizado Parroquial Rural de Toacaso: Latacunga, Ecuador, 2015; p. 174.
- Hidalgo, S.; Monzier, M.; Martin, H.; Chazot, G.; Eissen, J.-P.; Cotten, J. Adakitic magmas in the Ecuadorian Volcanic Front: Petrogenesis of the Iliniza Volcanic Complex (Ecuador). J. Volcanol. Geotherm. Res. 2007, 159, 366–392. [Google Scholar] [CrossRef]
- Buytaert, W.; Célleri, R.; De Bièvre, B.; Cisneros, F.; Wyseure, G.; Deckers, J.; Hofstede, R. Human impact on the hydrology of the Andean páramos. Earth-Sci. Rev. 2006, 79, 53–72. [Google Scholar] [CrossRef]
- Janeau, J.L.; Grellier, S.; Podwojewski, P. Influence of rainfall interception by endemic plants versus short cycle crops on water infiltration in high altitude ecosystems of Ecuador. Hydrol. Res. 2015, 46, 1008–1018. [Google Scholar] [CrossRef]
- Tapia, J.; Murray, J.; Ormachea, M.; Tirado, N.; Nordstrom, D.K. Origin, distribution, and geochemistry of arsenic in the Altiplano-Puna plateau of Argentina, Bolivia, Chile, and Perú. Sci. Total Environ. 2019, 678, 309–325. [Google Scholar] [CrossRef] [PubMed]
- Prado, G.H.P.; Velasco, S.M. Fitodepuración de aguas residuales domesticas con poaceas: Brachiaria mutica, pennisetum purpureum y panicum maximun en el municipio de Popayán, Cauca. Biotecnol. En El Sect. Agropecu. Agroindustrial 2013, 11, 57–65. [Google Scholar]
- Instituto Ecuatoriano de Normalización. Agua. Calidad del Agua. Muestreo. Técnica de Muestreo; Instituto Ecuatoriano de Normalización: Quito, Ecuador, 2013. [Google Scholar]
- Lawrence, H.K. Environmental Sampling and Analysis: A Practical Guide; Lewis Publishers Inc.: Boca Raton, FL, USA, 1991. [Google Scholar]
- Agrocalidad. Instructivo de Muestreo de Aguas. Available online: https://www.agrocalidad.gob.ec/ (accessed on 25 September 2020).
- Danh, L.; Truong, P.; Mammucari, R.; Pu, Y.; Foster, N. Phytoremediation of soils contaminated by heavy metals, metalloids, and radioactive materials using vetiver grass, Chrysopogon zizanioides. In Phytotechnologies; CRC Press: Boca Raton, FL, USA, 2012; pp. 255–280. ISBN 978-1-4398-7518-6. [Google Scholar]
- Sklenář, P. Presence of cushion plants increases community diversity in the high equatorial Andes. Flora-Morphol. Distrib. Funct. Ecol. Plants 2009, 204, 270–277. [Google Scholar] [CrossRef]
- Truong, P. Vetiver grass system: Potential applications for soil and water conservation in northern California. In Proceedings of the Stiff Grass Technology Seminar, Woodland, CA, USA, 9 May 2000; p. 10. [Google Scholar]
- Kumar, N. Vetiver Grass for Manifold Uses: A Critical Review. Int. J. Eng. Tech. Res. 2016, 4, 146–152. [Google Scholar]
- Wang, Y.-W. The root extension rate of vetiver under different temperature treatments. In Proceedings of the Second International Vetiver Conference, Phetchaburi, Thailand, 18–22 January 2000. [Google Scholar]
- Raman, J.K.; Gnansounou, E. A review on bioremediation potential of vetiver grass. In Waste Bioremediation; Varjani, S.J., Gnansounou, E., Gurunathan, B., Pant, D., Zakaria, Z.A., Eds.; Serie: Energy, Environment, and Sustainability; Springer: Singapore, 2018; pp. 127–140. ISBN 978-981-10-7412-7. [Google Scholar]
- Kiiskila, J.D.; Sarkar, D.; Panja, S.; Sahi, S.V.; Datta, R. Remediation of acid mine drainage-impacted water by vetiver grass (Chrysopogon zizanioides): A multiscale long-term study. Ecol. Eng. 2019, 129, 97–108. [Google Scholar] [CrossRef]
- Caviedes Rubio, D.I.; Delgado, D.R.; Olaya Amaya, A. Remoción de metales pesados comúnmente generados por la actividad industrial, empleando macrófitas neotropicales. Rev. P+L 2016, 11, 126–149. [Google Scholar] [CrossRef]
- Sarı, A.; Tuzen, M. Biosorption of As (III) and As (V) from aqueous solution by lichen (Xanthoria parietina) biomass. Sep. Sci. Technol. 2010, 45, 463–471. [Google Scholar] [CrossRef]
- Lee, B.-H.; Scholz, M. What is the role of Phragmites australis in experimental constructed wetland filters treating urban runoff? Ecol. Eng. 2007, 29, 87–95. [Google Scholar] [CrossRef]
- Sheoran, A.S.; Sheoran, V. Heavy metal removal mechanism of acid mine drainage in wetlands: A critical review. Miner. Eng. 2006, 19, 105–116. [Google Scholar] [CrossRef]
- Arteaga-Cortez, V.M.; Quevedo-Nolasco, A.; del Valle-Paniagua, D.H.; Castro-Popoca, M.; Bravo-Vinaja, Á.; Ramírez-Zierold, J.A. Estado del arte: Una revisión actual a los mecanismos que realizan los humedales artificiales para la remoción de nitrógeno y fósforo. Tecnol. Cienc. Agua 2019, 10, 319–342. [Google Scholar] [CrossRef]
- Interstate Technology and Regulatory Council (ITRC). Characterization and Remediation of Soils at Closed Small Arms Firing Ranges; Interstate Technology and Regulatory Council Small Arms Firing Range Team: Washintgon, DC, USA, 2003; pp. 1–2. [Google Scholar]
- Drever, J.I. The Geochemistry of Natural Waters; Prentice Hall: Englewood Cliffs, NJ, USA, 1988; Volume 437. [Google Scholar]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 978-1-118-59148-2. [Google Scholar]
- Horsnail, R.F.; Nichol, I.; Webb, J.S. Influence of variations in secondary environment on the metal content of drainage sediments. Q. Colo. Sch. Mines. 1969, 64, 307–322. [Google Scholar]
- Otte, M.L.; Kearns, C.C.; Doyle, M.O. Accumulation of arsenic and zinc in the rhizosphere of wetland plants. Bull. Environ. Contam. Toxicol. 1995, 55, 154–161. [Google Scholar] [CrossRef]
- Yang, C.; Ho, Y.-N.; Inoue, C.; Chien, M.-F. Long-term effectiveness of microbe-assisted arsenic phytoremediation by Pteris vittata in field trials. Sci. Total Environ. 2020, 740, 140137. [Google Scholar] [CrossRef]
- Khan, U.A.; Kujala, K.; Nieminen, S.P.; Räisänen, M.L.; Ronkanen, A.-K. Arsenic, antimony, and nickel leaching from northern peatlands treating mining influenced water in cold climate. Sci. Total Environ. 2019, 657, 1161–1172. [Google Scholar] [CrossRef]
- Palmer, M.J.; Chételat, J.; Jamieson, H.E.; Richardson, M.; Amyot, M. Hydrologic control on winter dissolved oxygen mediates arsenic cycling in a small subarctic lake: Under-ice as cycling in a shallow subarctic lake. Limnol. Oceanogr. 2020, 9999, 1–17. [Google Scholar] [CrossRef]
- Banerjee, R.; Goswami, P.; Pathak, K.; Mukherjee, A. Vetiver grass: An environment clean-up tool for heavy metal contaminated iron ore mine-soil. Ecol. Eng. 2016, 90, 25–34. [Google Scholar] [CrossRef]
- Chen, Y.; Shen, Z.; Li, X. The use of vetiver grass (Vetiveria zizanioides) in the phytoremediation of soils contaminated with heavy metals. Appl. Geochem. 2004, 19, 1553–1565. [Google Scholar] [CrossRef]
- Ghosh, M.; Paul, J.; Jana, A.; De, A.; Mukherjee, A. Use of the grass, Vetiveria zizanioides (L.) Nash for detoxification and phytoremediation of soils contaminated with fly ash from thermal power plants. Ecol. Eng. 2015, 74, 258–265. [Google Scholar] [CrossRef]
- Shaw, A.J. Heavy Metal Tolerance in Plants: Evolutionary Aspects; CRC Press: Boca Raton, FL, USA, 1990; ISBN 978-0-8493-6852-3. [Google Scholar]
- Brush, S.; Carney, H.; Huamán, Z. Dynamics of Andean potato agriculture. Econ. Bot. 1981, 35, 70–88. [Google Scholar] [CrossRef]
- Samal, A.C.; Kar, S.; Bhattacharya, P.; Santra, S.C. Human exposure to arsenic through foodstuffs cultivated using arsenic contaminated groundwater in areas of West Bengal, India. J. Environ. Sci. Health Part A 2011, 46, 1259–1265. [Google Scholar] [CrossRef]
- Pumisacho, M.; Aherwood, S. El Cultivo de la Papa en Ecuador, 1st ed.; INIAP-CIP: Quito, Ecuador, 2002. [Google Scholar]
- Suquilanda, M. Producción Orgánica de Cultivos Andinos; Ministerio de Agricultura, Ganaderia, Acuacultura y Pesca: Manta, Ecuador, 2012.


| Period | As Concentration (mg kg−1) | Fe Concentration (mg kg−1)) | ||||
|---|---|---|---|---|---|---|
| Root | Shoots | Leaves | Root | Shoots | Leaves | |
| Before the installation of the AFIV system | ND | ND | ND | 3721.30 | NA | NA |
| After treatment period | 494.50 | 71.92 | 30.05 | 3377.09 | 1037.49 | 362.77 |
| Translocation factor (TF) | 0.15 | NA | 0.31 | NA | ||
| Plant metal content mg | 298.23 | 2388.67 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fonseca Largo, K.M.; Ruiz Depablos, J.L.; Espitia-Sarmiento, E.F.; Llugsha Moreta, N.M. Artificial Floating Island with Vetiver for Treatment of Arsenic-Contaminated Water: A Real Scale Study in High-Andean Reservoir. Water 2020, 12, 3086. https://doi.org/10.3390/w12113086
Fonseca Largo KM, Ruiz Depablos JL, Espitia-Sarmiento EF, Llugsha Moreta NM. Artificial Floating Island with Vetiver for Treatment of Arsenic-Contaminated Water: A Real Scale Study in High-Andean Reservoir. Water. 2020; 12(11):3086. https://doi.org/10.3390/w12113086
Chicago/Turabian StyleFonseca Largo, Kalina Marcela, Joseline Luisa Ruiz Depablos, Edgar Fabián Espitia-Sarmiento, and Nataly Marisol Llugsha Moreta. 2020. "Artificial Floating Island with Vetiver for Treatment of Arsenic-Contaminated Water: A Real Scale Study in High-Andean Reservoir" Water 12, no. 11: 3086. https://doi.org/10.3390/w12113086
APA StyleFonseca Largo, K. M., Ruiz Depablos, J. L., Espitia-Sarmiento, E. F., & Llugsha Moreta, N. M. (2020). Artificial Floating Island with Vetiver for Treatment of Arsenic-Contaminated Water: A Real Scale Study in High-Andean Reservoir. Water, 12(11), 3086. https://doi.org/10.3390/w12113086






