Concentration–Discharge Relationships in Runoff Components during Rainfall Events at the Hydrohill Experimental Catchment in Chuzhou, China
Abstract
1. Introduction
2. Materials and Methods
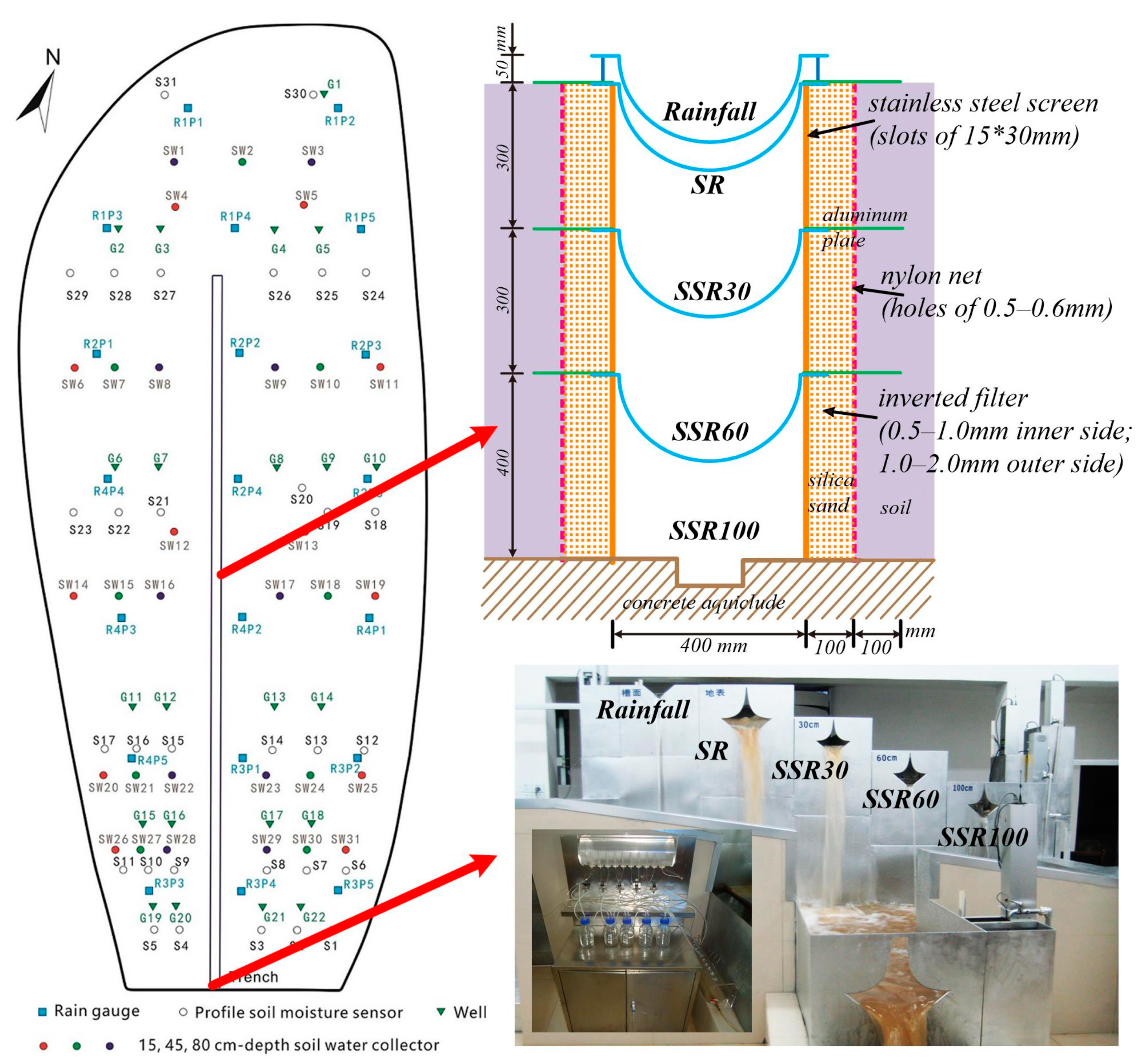
2.1. Study Site
2.2. Data Collection and Sample Analysis
2.3. Principal Component Analysis
3. Results
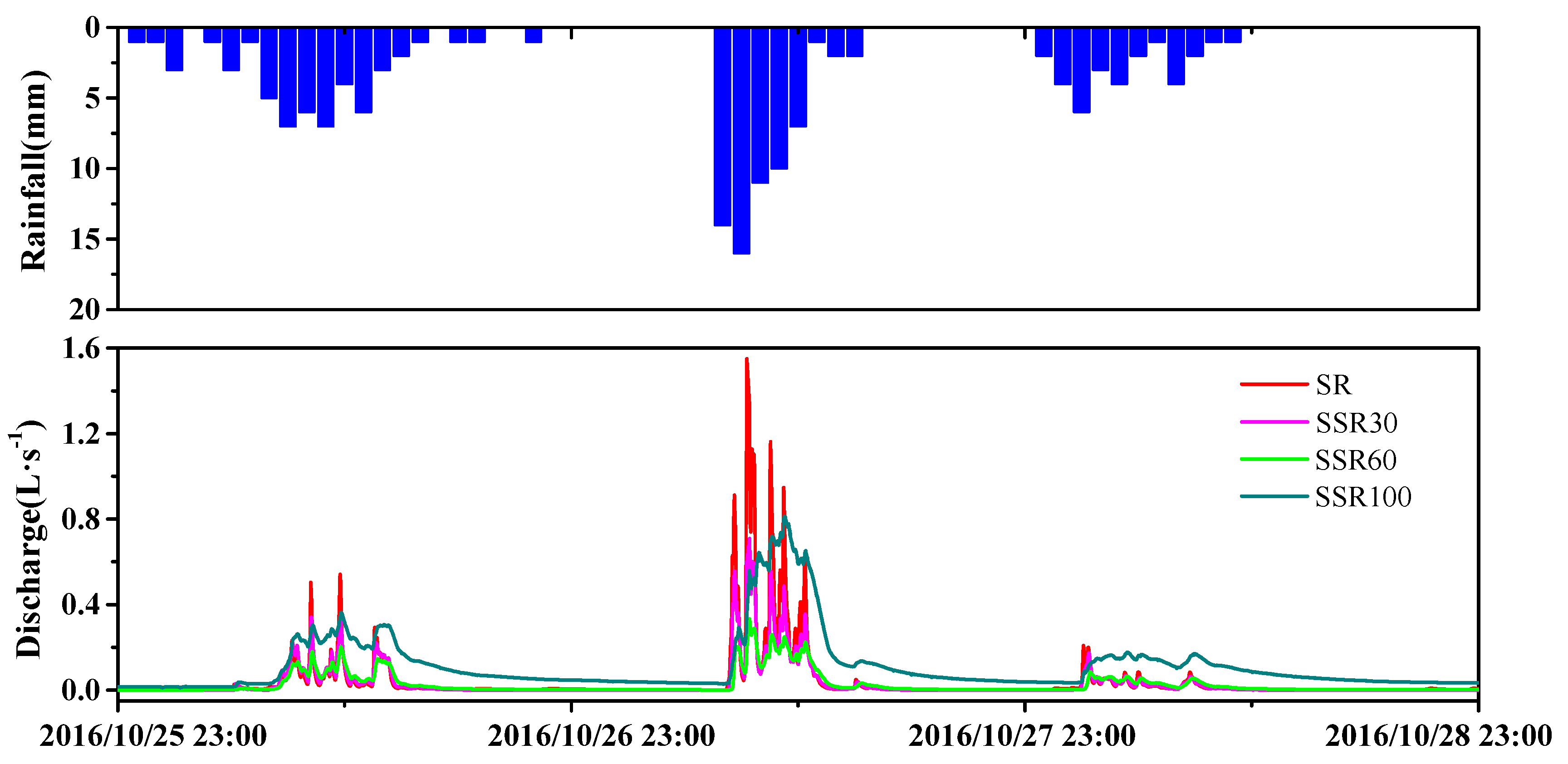
3.1. Hydrological Processes
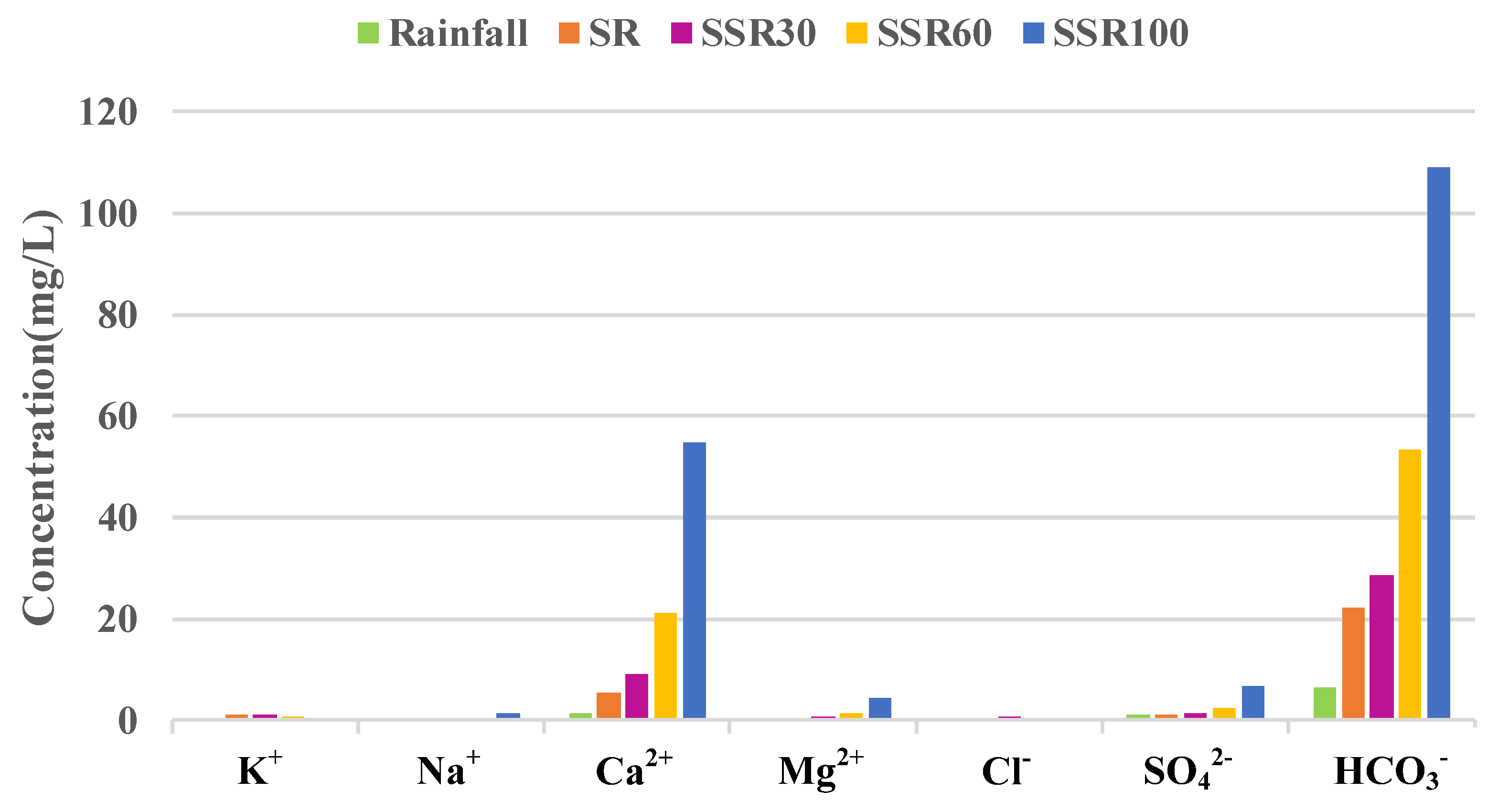
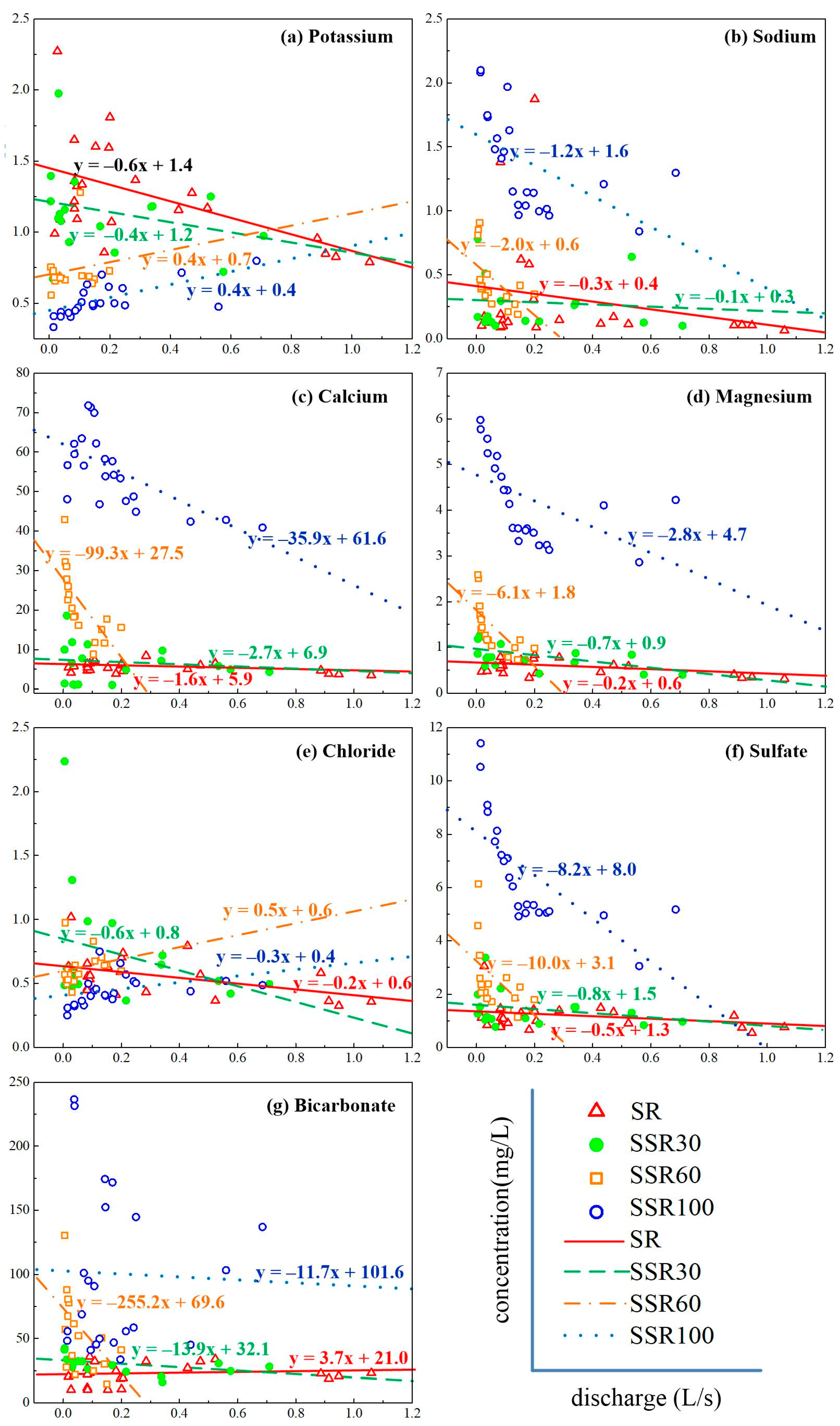
3.2. Concentration–Discharge Relationships
3.3. Principal Component Analysis
4. Discussion
4.1. Runoff Pathways and Concentration–Discharge Relationships
4.2. Principal Component Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chanat, J.G.; Rice, K.C.; Hornberger, G.M. Consistency of patterns in concentration-discharge plots. Water Resour. Res. 2002, 38, 1–10. [Google Scholar] [CrossRef]
- Chorover, J.; Derry, L.A.; McDowell, W.H. Concentration-discharge relations in the critical zone: Implications for resolving critical zone structure, function, and evolution. Water Resour. Res. 2017, 53, 8654–8659. [Google Scholar] [CrossRef]
- Hornberger, G.M.; Scanlon, T.M.; Raffensperger, J.P. Modelling transport of dissolved silica in a forested headwater catchment: The effect of hydrological and chemical time scales on hysteresis in the concentration–discharge relationship. Hydrol. Process. 2001, 15, 2029–2038. [Google Scholar] [CrossRef]
- Godsey, S.E.; Kirchner, J.W.; Clow, D.W. Concentration–discharge relationships reflect chemostatic characteristics of US catchments. Hydrol. Process. 2009, 23, 1844–1864. [Google Scholar] [CrossRef]
- Musolff, A.; Schmidt, C.; Selle, B.; Fleckenstein, J.H. Catchment controls on solute export. Adv. Water Resour. 2015, 86, 133–146. [Google Scholar] [CrossRef]
- Boyer, E.W.; Hornberger, G.M.; Bencala, K.E.; McKnight, D.M. Effects of asynchronous snowmelt on flushing of dissolved organic carbon: A mixing model approach. Hydrol. Process. 2000, 14, 3291–3308. [Google Scholar] [CrossRef]
- Burns, D.A. What do hydrologists mean when they use the term flushing? Hydrol. Process. 2005, 19, 1325–1327. [Google Scholar] [CrossRef]
- Rue, G.P.; Rock, N.D.; Gabor, R.S.; Pitlick, J.; Tfaily, M.; McKnight, D.M. Concentration-discharge relationships during an extreme event: Contrasting behavior of solutes and changes to chemical quality of dissolved organic material in the Boulder Creek Watershed during the September 2013 flood. Water Resour. Res. 2017, 53, 5276–5297. [Google Scholar] [CrossRef]
- Clow, D.W.; Drever, J.I. Weathering rates as a function of flow through an alpine soil. Chem. Geol. 1996, 132, 131–141. [Google Scholar] [CrossRef]
- House, W.A.; Warwick, M.S. Hysteresis of the solute concentration/discharge relationship in rivers during storms. Water Res. 1998, 32, 2279–2290. [Google Scholar] [CrossRef]
- Aguilera, R.; Melack, J.M. Concentration-discharge responses to storm events in coastal California watersheds. Water Resour. Res. 2018, 54, 407–424. [Google Scholar] [CrossRef]
- Anderson, S.P.; Dietrich, W.E.; Torres, R.; Montgomery, D.R.; Loague, K. Concentration-discharge relationships in runoff from a steep, unchanneled catchment. Water Resour. Res. 1997, 33, 211–225. [Google Scholar] [CrossRef]
- Evans, C.; Davies, T.D. Causes of concentration/discharge hysteresis and its potential as a tool for analysis of episode hydrochemistry. Water Resour. Res. 1998, 34, 129–137. [Google Scholar] [CrossRef]
- Thompson, S.E.; Basu, N.B.; Lascurain, J.; Aubeneau, A.; Rao, P.S.C. Relative dominance of hydrologic versus biogeochemical factors on solute export across impact gradients. Water Resour. Res. 2011, 47. [Google Scholar] [CrossRef]
- Ameli, A.A.; Beven, K.; Erlandsson, M.; Creed, I.F.; McDonnell, J.J.; Bishop, K. Primary weathering rates, water transit times, and concentration-discharge relations: A theoretical analysis for the critical zone. Water Resour. Res. 2017, 53, 942–960. [Google Scholar] [CrossRef]
- Hoagland, B.; Russo, T.A.; Gu, X.; Hill, L.; Kaye, J.; Forsythe, B.; Brantley, S.L. Hyporheic zone influences on concentration-discharge relationships in a headwater sandstone stream. Water Resour. Res. 2017, 53, 4643–4667. [Google Scholar] [CrossRef]
- Basu, N.B.; Thompson, S.E.; Rao, P.S.C. Hydrologic and biogeochemical functioning of intensively managed catchments: A synthesis of top-down analyses. Water Resour. Res. 2012, 48. [Google Scholar] [CrossRef]
- Van, M.K.J.; Basu, N.B.; Van, C.P. Two centuries of nitrogen dynamics: Legacy sources and sinks in the Mississippi and Susquehanna River Basins. Glob. Biogeochem. Cycles 2017, 31, 2–23. [Google Scholar] [CrossRef]
- Christopherson, N.; Hooper, R.P. Multivariate analysis of stream water chemical data: The use of principal components analysis for the end-member mixing problem. Water Resour. Res. 1992, 28, 99–107. [Google Scholar] [CrossRef]
- Carroll, S.; Goonetilleke, A. Assessment of high density of onsite wastewater treatment systems on a shallow groundwater coastal aquifer using PCA. Environmetrics 2005, 16, 257–274. [Google Scholar] [CrossRef]
- Lamouroux, C.; Hani, A. Identification of groundwater flow paths in complex aquifer systems. Hydrol. Process. 2006, 20, 2971–2987. [Google Scholar] [CrossRef]
- Rao, N.S.; Rao, J.P.; Subrahmanyam, A. Principal component analysis in groundwater quality in a developing urban area of Andhra Pradesh. J. Geol. Soc. India 2007, 69, 959. [Google Scholar]
- Yang, P.H.; Yuan, D.X.; Yuan, W.H.; Kuang, Y.L.; Jia, P.; He, Q.F. Formations of groundwater hydrogeochemistry in a karst system during storm events as revealed by PCA. Chin. Sci. Bull. 2010, 55, 1412–1422. [Google Scholar] [CrossRef]
- Gu, W.Z.; Liu, J.F.; Lin, H.; Lin, J.; Liu, H.W.; Liao, A.M.; Wang, N.; Wang, W.Z.; Ma, T.; Yang, N.; et al. Why hydrological maze: The hydropedological trigger? Review of experiments at Chuzhou hydrology laboratory. Vadose Zone J. 2018, 17, 1–27. [Google Scholar] [CrossRef]
- Liu, J.F.; Liao, A.M.; Wang, N.; Lin, J.; Liu, H.W.; Wang, W.Z.; Ma, T.; Cai, Z.; Liao, M.H.; Li, X.G.; et al. Hydrology of artificial and controlled experiments. Practice on the watershed hydrological experimental system reconciling deterministic and stochastic subjects based on the system complexity: 2. Practice and test. In Hydrology of Artificial and Controlled Experiments; Liu, J.F., Gu, W.Z., Eds.; IntechOpen: London, UK, 2018; pp. 253–281. [Google Scholar] [CrossRef]
- Shrestha, S.; Kazama, F. Assessment of surface water quality using multivariate statistical techniques: A case study of the Fuji river basin, Japan. Environ. Model. Softw. 2007, 22, 464–475. [Google Scholar] [CrossRef]
- Field, A.P. Discovering Statistics Using SPSS, 2nd ed.; Sage: London, UK, 2005; pp. 574–575. Available online: sro.sussex.ac.uk/id/eprint/14911 (accessed on 28 October 2020).
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water, 3rd ed.; U.S. Geological Survey: Alexandria, VA, USA, 1985; p. 272.
- Yang, N.; Zhang, J.Y.; Liu, J.F.; Liu, G.D.; Liao, A.M.; Wang, G.Q. Analysis of event-based hydrological processes at the hydrohill catchment using hydrochemical and isotopic methods. In Proceedings of the International Association of Hydrological Sciences; Copernicus Publications: Bethel Park, PA, USA, 2020; Volume 383, pp. 99–110. [Google Scholar] [CrossRef]
- Gu, W.Z.; Lu, J.J.; Zhao, X.; Norman, E.P. Responses of hydrochemical inorganic ions in the rainfall-runoff processes of the experimental catchments and its significance for tracing. Adv. Water Resour. 2007, 18, 1–7. (in Chinese). [Google Scholar] [CrossRef]
- Nimick, D.A.; Cleasby, T.E.; McCleskey, R.B. Seasonality of diel cycles of dissolved trace-metal concentrations in a Rocky Mountain stream. Environ. Geol. 2005, 47, 603–614. [Google Scholar] [CrossRef]
- Liu, H.Y.; Huang, J.G.; Guo, Y.N. Impacts of different vegetation types and soil properties on runoff chemical characteristics. Environ. Sci. 2006, 27, 654–660. [Google Scholar] [CrossRef]
- Qiao, B.Q.; Chen, K.P. The chemical composition in different categories of soil water. J. Nanjing Univ. Nat. Sci. 2015, 3, 604–611. [Google Scholar]
- Zhang, R.Q. Fundamentals of Hydrogeology, 7th ed.; Geological Publishing House: Beijing, China, 2011. [Google Scholar]
- Guo, L.; Lin, H. Addressing two bottlenecks to advance the understanding of preferential flow in soils. Adv. Agron. 2018, 147, 61–117. [Google Scholar] [CrossRef]
- Jarvis, N.J. A review of non-equilibrium water flow and solute transport in soil macropores: Principles, controlling factors and consequences for water quality. Eur. J. Soil Sci. 2007, 58, 523–546. [Google Scholar] [CrossRef]
- Guo, L.; Hallett, P.D.; Müller, K. Landmark Papers: No. 9 Jarvis, NJ 2007. A review of non-equilibrium water flow and solute transport in soil macropores: Principles, controlling factors and consequences for water quality. European Journal of Soil Science, 58, 523–546: Commentary on the impact of Jarvis (2007) by L. Guo, PD Hallett and K. Müller. PD Hallett and K. Müller. Eur. J. Soil Sci. 2020, 71, 308–315. [Google Scholar] [CrossRef]
- Guo, L.; Lin, H. Critical zone research and observatories: Current status and future perspectives. Vadose Zone J. 2016, 15. [Google Scholar] [CrossRef]
- Hencher, S.R. Preferential flow paths through soil and rock and their association with landslides. Hydrol. Process. 2010, 24, 1610–1630. [Google Scholar] [CrossRef]
- Freeze, R.A.; Cherry, J.A. Groundwater; Prentice Hall: Englewood Cliffs, NJ, USA, 1979; p. 398. [Google Scholar]

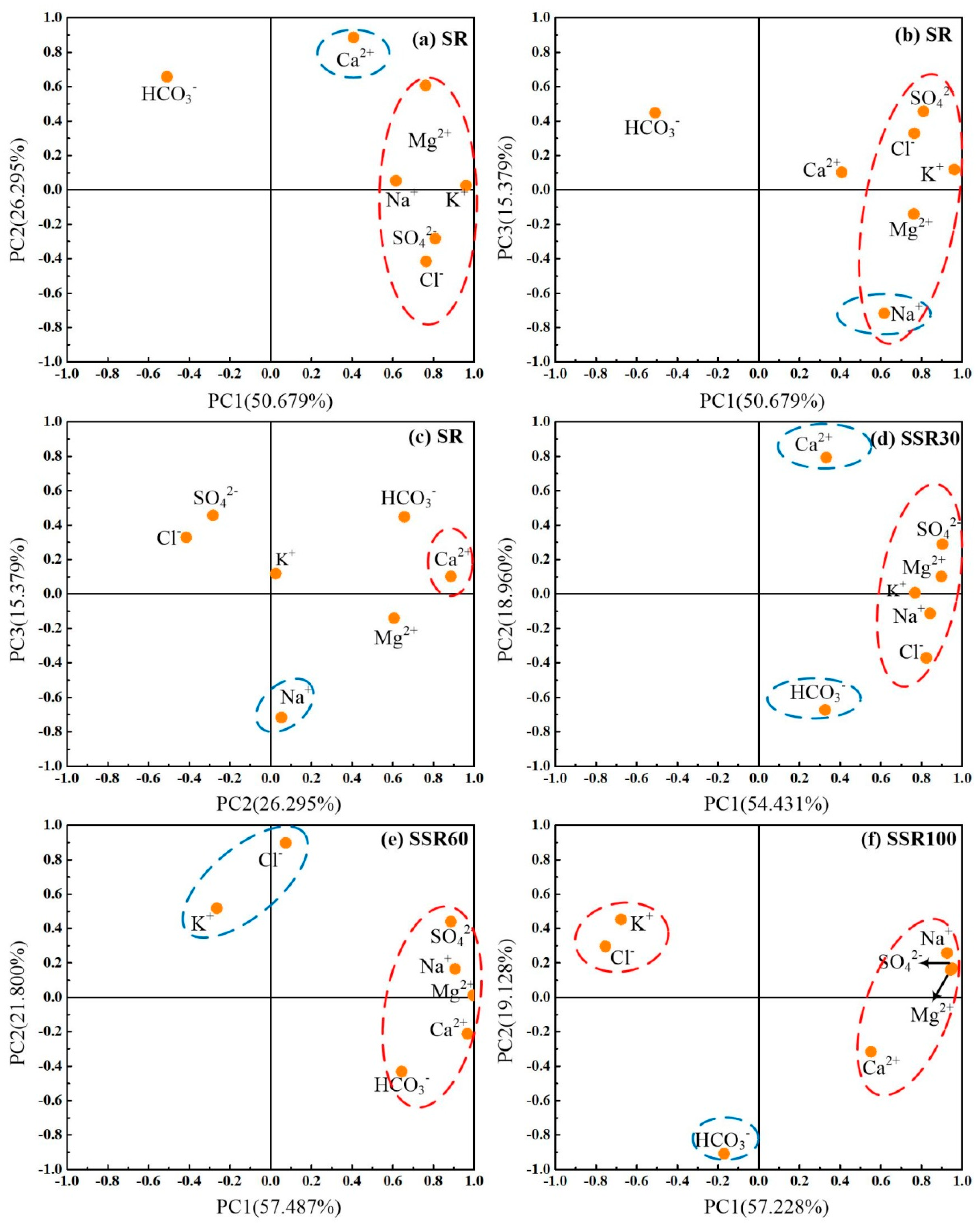
| Parameters | SR | SSR30 | SSR60 | SSR100 | |||||
|---|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC1 | PC2 | PC1 | PC2 | PC1 | PC2 | |
| K+ | 0.961 | 0.025 | 0.119 | 0.768 | 0.007 | −0.264 | 0.518 | −0.676 | 0.453 |
| Na+ | 0.616 | 0.053 | −0.717 | 0.842 | −0.114 | 0.907 | 0.166 | 0.925 | 0.257 |
| Ca2+ | 0.408 | 0.886 | 0.103 | 0.331 | 0.793 | 0.967 | −0.211 | 0.551 | −0.316 |
| Mg2+ | 0.762 | 0.607 | −0.140 | 0.897 | 0.102 | 0.996 | 0.012 | 0.943 | 0.160 |
| Cl− | 0.764 | −0.415 | 0.329 | 0.823 | −0.372 | 0.074 | 0.898 | −0.754 | 0.297 |
| SO42− | 0.809 | −0.283 | 0.457 | 0.902 | 0.289 | 0.886 | 0.440 | 0.950 | 0.170 |
| HCO3− | −0.509 | 0.657 | 0.449 | 0.326 | −0.673 | 0.664 | −0.431 | −0.171 | −0.908 |
| Eigenvalue | 3.547 | 1.841 | 1.077 | 3.810 | 1.327 | 4.024 | 1.526 | 4.006 | 1.339 |
| Variance explained by PCs (%) | 50.68 | 26.30 | 15.38 | 54.43 | 18.96 | 57.49 | 21.80 | 57.23 | 19.13 |
| Cumulative variance explained by PCs (%) | 50.68 | 76.97 | 92.35 | 54.43 | 73.39 | 57.49 | 79.29 | 57.23 | 76.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, N.; Zhang, J.; Liu, J.; Liu, G.; Boyer, E.W.; Guo, L.; Wang, G. Concentration–Discharge Relationships in Runoff Components during Rainfall Events at the Hydrohill Experimental Catchment in Chuzhou, China. Water 2020, 12, 3033. https://doi.org/10.3390/w12113033
Yang N, Zhang J, Liu J, Liu G, Boyer EW, Guo L, Wang G. Concentration–Discharge Relationships in Runoff Components during Rainfall Events at the Hydrohill Experimental Catchment in Chuzhou, China. Water. 2020; 12(11):3033. https://doi.org/10.3390/w12113033
Chicago/Turabian StyleYang, Na, Jianyun Zhang, Jiufu Liu, Guodong Liu, Elizabeth W. Boyer, Li Guo, and Guoqing Wang. 2020. "Concentration–Discharge Relationships in Runoff Components during Rainfall Events at the Hydrohill Experimental Catchment in Chuzhou, China" Water 12, no. 11: 3033. https://doi.org/10.3390/w12113033
APA StyleYang, N., Zhang, J., Liu, J., Liu, G., Boyer, E. W., Guo, L., & Wang, G. (2020). Concentration–Discharge Relationships in Runoff Components during Rainfall Events at the Hydrohill Experimental Catchment in Chuzhou, China. Water, 12(11), 3033. https://doi.org/10.3390/w12113033





