Behavior of UV Filters, UV Blockers and Pharmaceuticals in High Rate Algal Ponds Treating Urban Wastewater
Abstract
1. Introduction
2. Materials and Methods
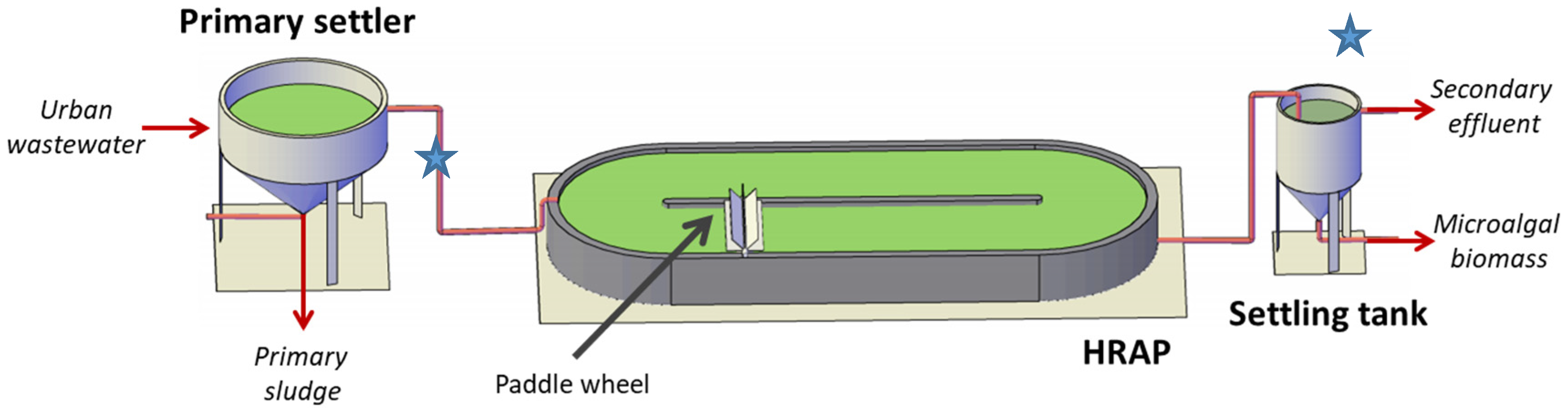
2.1. Microalgae-Based Treatment System Description and Operation
2.2. Sampling Campaign
2.3. Chemicals and Reagents
2.4. Analytical Methodologies
2.4.1. Physico-Chemical Characterization
2.4.2. Online-SPE-HPLC-MS/MS Analysis of the Target Compounds
2.5. Environmental Risk Assessment
3. Results
3.1. Physico-Chemical Water Quality Parameters
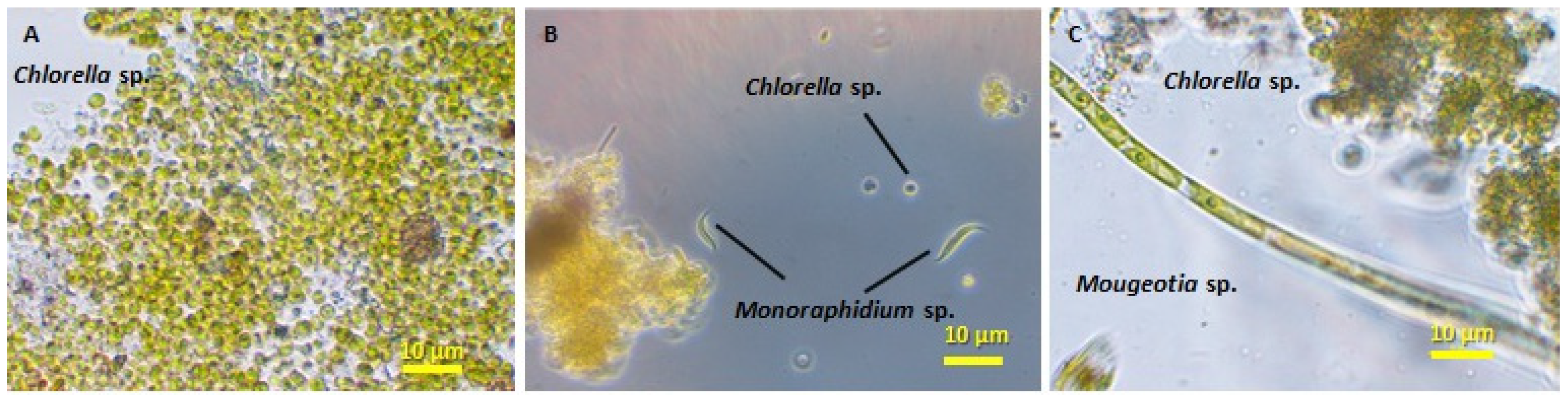
3.2. Biomass Productivity
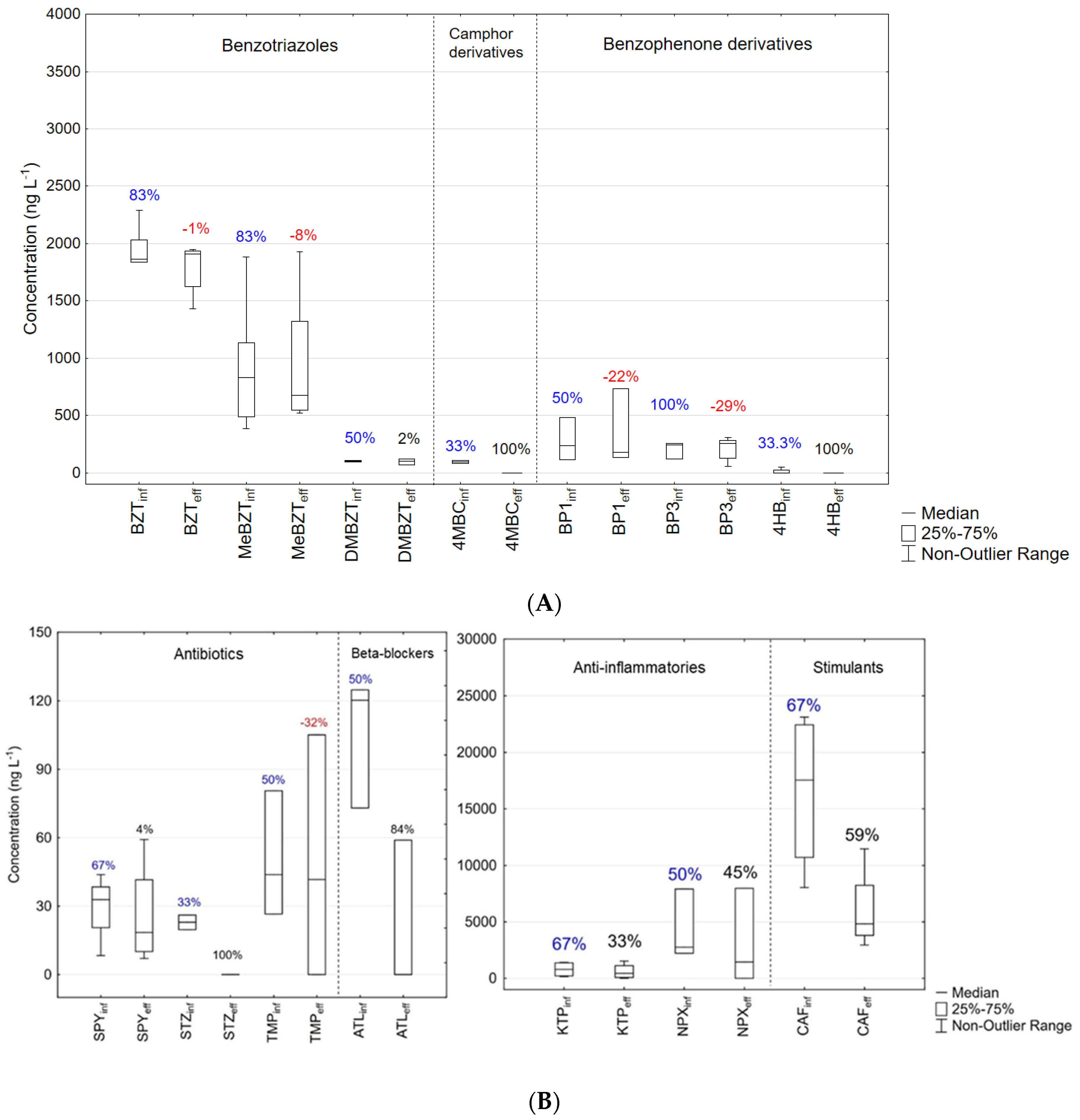
3.3. Occurrence of PPCPs in the HRAP Influent
3.3.1. Benzotriazoles and UV-Filters
3.3.2. Pharmaceuticals and Stimulants
3.4. Removal Efficiency in the HRAP
3.4.1. Benzotriazoles
3.4.2. UV Filters
3.4.3. Pharmaceuticals
3.5. Ecotoxicity in the HRAP Effluent
4. Discussion
Final Remarks
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chisti, Y. Raceways-based production of algal crude oil. Green 2013, 3, 195–216. [Google Scholar] [CrossRef]
- Oswald, W.J. Ponds in the twenty-first century. Water Sci. Technol. 1995, 31, 1–8. [Google Scholar] [CrossRef]
- García, J.; Green, B.F.; Lundquist, T.; Mujeriego, R.; Hernández-Mariné, M.; Oswald, W.J. Long term diurnal variations in contaminant removal in high rate ponds treating urban wastewater. Bioresour. Technol. 2006, 97, 1709–1715. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, R.; Guieysse, B. Algal-bacterial processes for the treatment of hazardous contaminants: A review. Water Res. 2006, 40, 2799–2815. [Google Scholar] [CrossRef] [PubMed]
- Arashiro, L.T.; Ferrer, I.; Pániker, C.C.; Gómez-Pinchetti, J.L.; Rousseau, D.P.L.; Van Hulle, S.W.H.; Garfí, M. Natural pigments and biogas recovery from microalgae grown in wastewater. ACS Sustain. Chem. Eng. 2020. [Google Scholar] [CrossRef] [PubMed]
- Arashiro, L.T.; Ferrer, I.; Rousseau, D.P.L.; Van Hulle, S.W.H.; Garfí, M. The effect of primary treatment of wastewater in high rate algal pond systems: Biomass and bioenergy recovery. Bioresour. Technol. 2019, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Sharma, G.K.; Malla, F.A.; Kumar, A.; Rashmi; Gupta, N. Microalgae based biofertilizers: A biorefinery approach to phycoremediate wastewater and harvest biodiesel and manure. J. Clean. Prod. 2019, 211, 1412–1419. [Google Scholar] [CrossRef]
- Rueda, E.; García-Galán, M.J.; Ortiz, A.; Uggetti, E.; Carretero, J.; García, J.; Díez-Montero, R. Bioremediation of agricultural runoff and biopolymers production from cyanobacteria cultured in demonstrative full-scale photobioreactors. Process Saf. Environ. Prot. 2020. [Google Scholar] [CrossRef]
- Rueda, E.; García-Galán, M.J.; Díez-Montero, R.; Vila, J.; Grifoll, M.; García, J. Polyhydroxybutyrate and glycogen production in photobioreactors inoculated with wastewater borne cyanobacteria monocultures. Bioresour. Technol. 2020, 295, 122233. [Google Scholar] [CrossRef] [PubMed]
- Arashiro, L.T.; Montero, N.; Ferrer, I.; Acién, F.G.; Gómez, C.; Garfí, M. Life cycle assessment of high rate algal ponds for wastewater treatment and resource recovery. Sci. Total Environ. 2018. [Google Scholar] [CrossRef]
- Vassalle, L.; García-Galan, M.J.; De Aquinoc, S.F.; Afonso, R.J.; FrancoAfonso, R.J.d.; Ferrer, I.; Passos, F.; Filho, C.R.M. Can high rate algal ponds be used as post-treatment of UASB reactors to remove micropollutants? Chemosphere 2020, 125969. [Google Scholar] [CrossRef] [PubMed]
- Vassalle, L.; Díez-Montero, R.; Machado, A.T.R.; Moreira, C.; Ferrer, I.; Mota, C.R.; Passos, F. Upflow anaerobic sludge blanket in microalgae-based sewage treatment: Co-digestion for improving biogas production. Bioresour. Technol. 2020, 300, 9. [Google Scholar] [CrossRef] [PubMed]
- De Godos, I.; Arbib, Z.; Lara, E.; Rogalla, F. Evaluation of High Rate Algae Ponds for treatment of anaerobically digested wastewater: Effect of CO2 addition and modification of dilution rate. Bioresour. Technol. 2016, 220, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Craggs, R.J.; Tanner, C.C.; Sukias, J.P.S.; Davies-Colley, R.J. Dairy farm wastewater treatment by an advanced pond system. Water Sci. Technol. 2003, 48, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Solimeno, A.; García, J. Microalgae and bacteria dynamics in high rate algal ponds based on modelling results: Long-term application of BIO_ALGAE model. Sci. Total Environ. 2019, 650, 1818–1831. [Google Scholar] [CrossRef]
- García-Galán, M.J.; Arashiro, L.; Santos, L.H.M.L.M.; Insa, S.; Rodríguez-Mozaz, S.; Barceló, D.; Ferrer, I.; Garfi, M. Fate of priority pharmaceuticals and their main metabolites and transformation products in microalgae-based wastewater treatment systems. J. Hazard. Mater. 2020, 121771. [Google Scholar] [CrossRef]
- Matamoros, V.; Gutiérrez, R.; Ferrer, I.; García, J.; Bayona, J.M. Capability of microalgae-based wastewater treatment systems to remove emerging organic contaminants: A pilot-scale study. J. Hazard. Mater. 2015, 288, 34–42. [Google Scholar] [CrossRef]
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef]
- Van Wezel, A.P.; Van den Hurk, F.; Sjerps, R.M.A.; Meijers, E.M.; Roex, E.W.M.; Ter Laak, T.L. Impact of industrial waste water treatment plants on Dutch surface waters and drinking water sources. Sci. Total Environ. 2018, 640–641, 1489–1499. [Google Scholar] [CrossRef]
- Matamoros, V.; Uggetti, E.; García, J.; Bayona, J.M. Assessment of the mechanisms involved in the removal of emerging contaminants by microalgae from wastewater: A laboratory scale study. J. Hazard. Mater. 2016, 301, 197–205. [Google Scholar] [CrossRef]
- Ponsatí, L.; Corcoll, N.; Petrović, M.; Picó, Y.; Ginebreda, A.; Tornés, E.; Guasch, H.; Barceló, D.; Sabater, S. Multiple-stressor effects on river biofilms under different hydrological conditions. Freshw. Biol. 2016, 61, 2102–2115. [Google Scholar] [CrossRef]
- Kuzmanović, M.; Ginebreda, A.; Petrović, M.; Barceló, D. Risk assessment based prioritization of 200 organic micropollutants in 4 Iberian rivers. Sci. Total Environ. 2015, 503–504, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.L.; Wong, M.H. Pharmaceuticals and personal care products (PPCPs): A review on environmental contamination in China. Environ. Int. 2013, 59, 208–224. [Google Scholar] [CrossRef] [PubMed]
- Brausch, J.M.; Rand, G.M. A review of personal care products in the aquatic environment: Environmental concentrations and toxicity. Chemosphere 2011, 82, 1518–1532. [Google Scholar] [CrossRef]
- APHA-AWWA-WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, L.S., Eds.; American Water Works Association: Washington, DC, USA; American Public Health Association: Washington, DC, USA; Water Environment Federation: Washington, DC, USA, 2012; ISBN 9780875530130. [Google Scholar]
- Solórzano, L. Determination of ammonia in natural seawater by the phenol-hypochlorite method. Limnol. Oceanogr. 1969, 14, 799–801. [Google Scholar] [CrossRef]
- Bourrelly, P. Les Algues d’eau Douce. Les Algues Vertes; Boubée, Ed.; Editions N. Boubee & Cie: Paris, French, 1990. [Google Scholar]
- Palmer, C.M. Algas en Abastecimientos de Agua: Manual Ilustrado Acerca de la Identificación, Importancia y Control de Las Algas en Los Abastecimientos de Agua; Interamericana: Puerto Rico, México, 1962. [Google Scholar]
- Fisher, D.K.; Pringle, H.C., III. Evaluation of alternative methods for estimating reference evapotranspiration. Agric. Sci. 2013, 4, 51–60. [Google Scholar] [CrossRef]
- Gago-Ferrero, P.; Mastroianni, N.; Díaz-Cruz, M.S.; Barceló, D. Fully automated determination of nine ultraviolet filters and transformation products in natural waters and wastewaters by on-line solid phase extraction-liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2013. [Google Scholar] [CrossRef]
- García-Galán, M.J.; Díaz-Cruz, M.S.; Barceló, D. Determination of 19 sulfonamides in environmental water samples by automated on-line solid-phase extraction-liquid chromatography-tandem mass spectrometry (SPE-LC-MS/MS). Talanta 2010, 81, 355–366. [Google Scholar] [CrossRef]
- Sanderson, H.; Johnson, D.J.; Reitsma, T.; Brain, R.A.; Wilson, C.J.; Solomon, K.R. Ranking and prioritization of environmental risks of pharmaceuticals in surface waters. Regul. Toxicol. Pharmacol. 2004, 39, 158–183. [Google Scholar] [CrossRef]
- Santiago, A.F.; Calijuri, M.L.; Assemany, P.P.; Calijuri, M.D.C.; Reis, A.J.D. Dos Algal biomass production and wastewater treatment in high rate algal ponds receiving disinfected effluent. Environ. Technol. 2017, 34, 1877–1885. [Google Scholar] [CrossRef]
- Villar-Navarro, E.; Baena-Nogueras, R.M.; Paniw, M.; Perales, J.A.; Lara-Martín, P.A. Removal of pharmaceuticals in urban wastewater: High rate algae pond (HRAP) based technologies as an alternative to activated sludge based processes. Water Res. 2018, 139, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Young, P.; Taylor, M.; Fallowfield, H.J. Mini-review: High rate algal ponds, flexible systems for sustainable wastewater treatment. World J. Microbiol. Biotechnol. 2017, 33. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, D.L.; Turnbull, M.H.; Craggs, R.J. Increased pond depth improves algal productivity and nutrient removal in wastewater treatment high rate algal ponds. Water Res. 2014, 53, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, A.G.; Ajibola, A.; Kannan, K.; Thomaidis, N.S. Occurrence and removal efficiencies of benzotriazoles and benzothiazoles in a wastewater treatment plant in Greece. Sci. Total Environ. 2013, 452–453, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.S.; Ying, G.G.; Shareef, A.; Kookana, R.S. Occurrence and removal of benzotriazoles and ultraviolet filters in a municipal wastewater treatment plant. Environ. Pollut. 2012, 165, 225–232. [Google Scholar] [CrossRef]
- Liu, Y.S.; Ying, G.G.; Shareef, A.; Kookana, R.S. Biodegradation of three selected benzotriazoles under aerobic and anaerobic conditions. Water Res. 2011, 45, 5005–5014. [Google Scholar] [CrossRef]
- Molins-Delgado, D.; Távora, J.; Silvia Díaz-Cruz, M.; Barceló, D. UV filters and benzotriazoles in urban aquatic ecosystems: The footprint of daily use products. Sci. Total Environ. 2017. [Google Scholar] [CrossRef]
- Langford, K.H.; Reid, M.J.; Fjeld, E.; Øxnevad, S.; Thomas, K.V. Environmental occurrence and risk of organic UV filters and stabilizers in multiple matrices in Norway. Environ. Int. 2015, 80, 1–7. [Google Scholar] [CrossRef]
- Wick, A.; Fink, G.; Ternes, T.A. Comparison of electrospray ionization and atmospheric pressure chemical ionization for multi-residue analysis of biocides, UV-filters and benzothiazoles in aqueous matrices and activated sludge by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2010. [Google Scholar] [CrossRef]
- Serra-Roig, M.P.; Jurado, A.; Díaz-Cruz, M.S.; Vázquez-Suñé, E.; Pujades, E.; Barceló, D. Occurrence, fate and risk assessment of personal care products in river–groundwater interface. Sci. Total Environ. 2016. [Google Scholar] [CrossRef]
- Kurissery, S.; Kanavillil, N.; Verenitch, S.; Mazumder, A. Caffeine as an anthropogenic marker of domestic waste: A study from Lake Simcoe watershed. Ecol. Indic. 2012, 23, 501–508. [Google Scholar] [CrossRef]
- Sidhu, J.P.S.; Ahmed, W.; Gernjak, W.; Aryal, R.; McCarthy, D.; Palmer, A.; Kolotelo, P.; Toze, S. Sewage pollution in urban stormwater runoff as evident from the widespread presence of multiple microbial and chemical source tracking markers. Sci. Total Environ. 2013, 463–464, 488–496. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; Hao, H.; Duc, L.; Ibney, F.; Zhang, J.; Liang, S.; Wang, X.C. Science of the Total Environment A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Garduño, B.; Pintado-Herrera, M.G.; Biel-Maeso, M.; Rueda-Márquez, J.J.; Lara-Martín, P.A.; Perales, J.A.; Manzano, M.A.; Garrido-Pérez, C.; Martín-Díaz, M.L. Environmental risk assessment of effluents as a whole emerging contaminant: Efficiency of alternative tertiary treatments for wastewater depuration. Water Res. 2017, 119, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Rodríguez-Mozaz, S.; Barceló, D. Fast and comprehensive multi-residue analysis of a broad range of human and veterinary pharmaceuticals and some of their metabolites in surface and treated waters by ultra-high-performance liquid chromatography coupled to quadrupole-linear ion trap tandem. J. Chromatogr. A 2012, 1248, 104–121. [Google Scholar] [CrossRef] [PubMed]
- Gros, M.; Petrovic, M.; Ginebreda, A.; Barceló, D. Removal of pharmaceuticals during wastewater treatment and environmental risk assessment using hazard indexes. Environ. Int. 2010, 36, 15–26. [Google Scholar] [CrossRef] [PubMed]
- López-Serna, R.; Marín-de-Jesús, D.; Irusta-Mata, R.; García-Encina, P.A.; Lebrero, R.; Fdez-Polanco, M.; Muñoz, R. Multiresidue analytical method for pharmaceuticals and personal care products in sewage and sewage sludge by online direct immersion SPME on-fiber derivatization—GCMS. Talanta 2018, 186, 506–512. [Google Scholar] [CrossRef]
- Gros, M.; Rodríguez-Mozaz, S.; Barceló, D. Rapid analysis of multiclass antibiotic residues and some of their metabolites in hospital, urban wastewater and river water by ultra-high-performance liquid chromatography coupled to quadrupole-linear ion trap tandem mass spectrometry. J. Chromatogr. A 2013, 1292, 173–188. [Google Scholar] [CrossRef]
- Szymańska, U.; Wiergowski, M.; Sołtyszewski, I.; Kuzemko, J.; Wiergowska, G.; Woźniak, M.K. Presence of antibiotics in the aquatic environment in Europe and their analytical monitoring: Recent trends and perspectives. Microchem. J. 2019, 147, 729–740. [Google Scholar] [CrossRef]
- García-Galán, M.J.; Díaz-Cruz, M.S.; Barceló, D. Occurrence of sulfonamide residues along the Ebro river basin. Removal in wastewater treatment plants and environmental impact assessment. Environ. Int. 2011, 37, 462–473. [Google Scholar] [CrossRef]
- Paíga, P.; Correia, M.; Fernandes, M.J.; Silva, A.; Carvalho, M.; Vieira, J.; Jorge, S.; Silva, J.G.; Freire, C.; Delerue-Matos, C. Assessment of 83 pharmaceuticals in WWTP influent and effluent samples by UHPLC-MS/MS: Hourly variation. Sci. Total Environ. 2019, 648, 582–600. [Google Scholar] [CrossRef] [PubMed]
- García-Galán, M.J.; González Blanco, S.; López Roldán, R.; Díaz-Cruz, S.; Barceló, D. Ecotoxicity evaluation and removal of sulfonamides and their acetylated metabolites during conventional wastewater treatment. Sci. Total Environ. 2012, 437, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.Z.; Glady-Croué, J.; Croué, J.P. Photodegradation of sulfathiazole under simulated sunlight: Kinetics, photo-induced structural rearrangement, and antimicrobial activities of photoproducts. Water Res. 2017, 124, 576–583. [Google Scholar] [CrossRef]
- Dolar, D.; Gros, M.; Rodriguez-Mozaz, S.; Moreno, J.; Comas, J.; Rodriguez-Roda, I.; Barceló, D. Removal of emerging contaminants from municipal wastewater with an integrated membrane system, MBR-RO. J. Hazard. Mater. 2012, 239–240, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Vassalle, L.; Sunyer Caldú, A.; Uggetti, E.; Díez-Montero, R.; Díaz-Cruz, M.S.; García, J.; García-Galán, M.J. Bioremediation of emerging micropollutants in irrigation water. The alternative of microalgae-based treatments. J. Environ. Manag. 2020. [Google Scholar] [CrossRef]
- Reemtsma, T.; Miehe, U.; Duennbier, U.; Jekel, M. Polar pollutants in municipal wastewater and the water cycle: Occurrence and removal of benzotriazoles. Water Res. 2010, 44, 596–604. [Google Scholar] [CrossRef]
- Liu, Y.S.; Ying, G.G.; Shareef, A.; Kookana, R.S. Photolysis of benzotriazole and formation of its polymerised photoproducts in aqueous solutions under UV irradiation. Environ. Chem. 2011, 8, 174–181. [Google Scholar] [CrossRef]
- Loos, R.; Gawlik, B.M.; Locoro, G.; Rimaviciute, E.; Contini, S.; Bidoglio, G. EU-wide survey of polar organic persistent pollutants in European river waters. Environ. Pollut. 2009, 157, 561–568. [Google Scholar] [CrossRef]
- Mizukawa, A.; Molins-Delgado, D.; De Azevedo, J.C.R.; Fernandes, C.V.S.; Díaz-Cruz, S.; Barceló, D. Sediments as a sink for UV filters and benzotriazoles: The case study of Upper Iguaçu watershed, Curitiba (Brazil). Environ. Sci. Pollut. Res. 2017, 24, 18284–18294. [Google Scholar] [CrossRef]
- Molins-Delgado, D.; Silvia Díaz-Cruz, M.; Barceló, D. Removal of polar UV stabilizers in biological wastewater treatments and ecotoxicological implications. Chemosphere 2015, 119, S51–S57. [Google Scholar] [CrossRef]
- Díaz-Cruz, M.S.; Gago-Ferrero, P.; Llorca, M.; Barceló, D. Analysis of UV filters in tap water and other clean waters in Spain. Anal. Bioanal. Chem. 2012, 402, 2325–2333. [Google Scholar] [CrossRef] [PubMed]
- Gago-Ferrero, P.; Díaz-Cruz, M.S.; Barceló, D. Occurrence of multiclass UV filters in treated sewage sludge from wastewater treatment plants. Chemosphere 2011. [Google Scholar] [CrossRef] [PubMed]
- Gago-Ferrero, P.; Badia-Fabregat, M.; Olivares, A.; Piña, B.; Blánquez, P.; Vicent, T.; Caminal, G.; Díaz-Cruz, M.S.; Barceló, D. Evaluation of fungal- and photo-degradation as potential treatments for the removal of sunscreens BP3 and BP1. Sci. Total Environ. 2012, 427–428, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Acharya, K. Removal of trimethoprim, sulfamethoxazole, and triclosan by the green alga Nannochloris sp. J. Hazard. Mater. 2016, 315, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Gentili, F.G.; Fick, J. Algal cultivation in urban wastewater: An efficient way to reduce pharmaceutical pollutants. J. Appl. Phycol. 2017, 29, 255–262. [Google Scholar] [CrossRef]
- Tolboom, S.N.; Carrillo-Nieves, D.; De Jesús Rostro-Alanis, M.; De la Cruz Quiroz, R.; Barceló, D.; Iqbal, H.M.N.; Parra-Saldivar, R. Algal-based removal strategies for hazardous contaminants from the environment—A review. Sci. Total Environ. 2019, 665, 358–366. [Google Scholar] [CrossRef]
- Salgado, R.; Pereira, V.J.; Carvalho, G.; Soeiro, R.; Gaffney, V.; Almeida, C.; Cardoso, V.V.; Ferreira, E.; Benoliel, M.J.; Ternes, T.A.; et al. Photodegradation kinetics and transformation products of ketoprofen, diclofenac and atenolol in pure water and treated wastewater. J. Hazard. Mater. 2013, 244–245, 516–527. [Google Scholar] [CrossRef]
- Suarez, S.; Lema, J.M.; Omil, F. Removal of Pharmaceutical and Personal Care Products (PPCPs) under nitrifying and denitrifying conditions. Water Res. 2010, 44, 3214–3224. [Google Scholar] [CrossRef]
- Mira, Č.; Gros, M.; Farré, M.; Barceló, D.; Petrovi, M. Pharmaceuticals as chemical markers of wastewater contamination in the vulnerable area of the Ebro Delta (Spain). Sci. Total Environ. 2019, 652, 952–963. [Google Scholar]
- Guzik, U. Naproxen in the environment: Its occurrence, toxicity to nontarget organisms and biodegradation. Appl. Microbiol. Biotechnol. 2020, 104, 1849–1857. [Google Scholar]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.K.; Sharma, S.K.; Abel, C.D.T.; Magic-Knezev, A.; Amy, G.L. Role of biodegradation in the removal of pharmaceutically active compounds with different bulk organic matter characteristics through managed aquifer recharge: Batch and column studies. Water Res. 2011, 45, 4722–4736. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Zhao, Q.; Wu, Y.; Wu, C.; Xiang, W. Removal of nutrients and pharmaceuticals and personal care products from wastewater using periphyton photobioreactors. Bioresour. Technol. 2018, 248, 113–119. [Google Scholar] [CrossRef]
- Ávila, C.; Matamoros, V.; Reyes-Contreras, C.; Piña, B.; Casado, M.; Mita, L.; Rivetti, C.; Barata, C.; García, J.; Bayona, J.M. Attenuation of emerging organic contaminants in a hybrid constructed wetland system under different hydraulic loading rates and their associated toxicological effects in wastewater. Sci. Total Environ. 2014, 470–471, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Valcárcel, Y.; González Alonso, S.; Rodríguez-Gil, J.L.; Gil, A.; Catalá, M. Detection of pharmaceutically active compounds in the rivers and tap water of the Madrid Region (Spain) and potential ecotoxicological risk. Chemosphere 2011, 84, 1336–1348. [Google Scholar] [CrossRef]
- De Wilt, A.; Butkovskyi, A.; Tuantet, K.; Leal, L.H.; Fernandes, T.V.; Langenhoff, A.; Zeeman, G. Micropollutant removal in an algal treatment system fed with source separated wastewater streams. J. Hazard. Mater. 2016, 304, 84–92. [Google Scholar] [CrossRef]
- Xiong, J.Q.; Kurade, M.B.; Abou-Shanab, R.A.I.; Ji, M.K.; Choi, J.; Kim, J.O.; Jeon, B.H. Biodegradation of carbamazepine using freshwater microalgae Chlamydomonas mexicana and Scenedesmus obliquus and the determination of its metabolic fate. Bioresour. Technol. 2016, 205, 183–190. [Google Scholar] [CrossRef]
| Parameters (n = 6) | Sample Type | |
|---|---|---|
| HRAPinfluent | HRAPeffuent | |
| Mean ± SD | Mean ± SD | |
| TSS (mg L−1) | 192.44 ± 114.65 | 323.15 ± 88.4 */42.59 ± 12.8 |
| VSS (mg L−1) | 154.67 ± 102.9 | 274.30 ± 77.5 */42.89 ± 13.9 |
| COD (mgO2 L−1) | 199.92 ± 75.11 | 78.85 ±33.13 |
| pH | 8.2 ± 0.2 | 8.4 ± 0.4 |
| DO (mg L−1) | - | 8.80 ± 1.7 |
| Temperature (°C) | 19.5 ± 1.3 | 18.9 ± 1.6 |
| N-NH4+ (mg L−1) | 24.1 ± 3.0 | 0.6 ± 0.4 |
| TN (mg L−1) | 56.9 ± 17.8 | 38.7 ± 9.4 |
| TIN (mg L−1) | 29.3 ± 9.94 | 17.4 ± 6.5 |
| TC (mg L−1) | 253.5 ± 95.6 | 160.6 ± 21.6 |
| N-NO2− (mg L−1) | 5.2 ± 4.5 | 1.8 ± 2.1 |
| N-NO3− (mg L−1) | 0.15 ± 0.36 | 15.2 ± 8.6 |
| P-PO43− (mg L−1) | 3.9 ± 2.1 | 3.2 ± 2.1 |
| S-SO42− (mg L−1) | 76.2 ± 84.9 | 39.9 ± 20.1 |
| Toxicity Endpoints (mg L−1) | Hazard Quotients (HQs) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Effluent Concentration (mg L−1) | Green Algae | Crustaceans | Fish | Ref. | Green Algae | Crustaceans | Fish | ||
| Benzotriazoles | 1H-benzotriazole (BZT) | 0.001769 | 2.51 | 107 | 654 | [1] | 7.07 × 10−1 | 1.65 × 10−2 | 2.72 × 10−2 |
| 5-methyl-1-H-benzotriazole (MeBZT) | 0.000998 | 2.861 | 51.6 | 224 | [1] | 3.49 × 10−1 | 1.93 × 10−2 | 4.54 × 10−2 | |
| Benzophenones | BP1 | 0.000346 | 10.52 | 3.9 | 49,194 | [2] | 3.3 × 10−2 | 8.87 × 10−2 | 7.03 × 10−5 |
| BP3 | 0.000239 | 0.961 | 1.6 | 7495 | [2] | 2.49 × 10−1 | 1.49 × 10−1 | 3.19 × 10−4 | |
| Camphor derivatives | 4MBC* | 0.000049 | – | 2.3 | 4155 | [2] | – | 2.13 × 10−2 | 1.18 × 10−4 |
| Antibiotics | Sulfapyridine (SPY) | 0.000026 | 1912 | – | <39.836 | [1] | 1.29 × 10−4 | – | 6.21 × 10−4 |
| Trimethoprim (TMP) | 0.000073 | 80.32 | 92 | >1007 | [1] | 9 × 10−4 | 7.98 × 10−4 | 7.34 × 10−4 | |
| Analgesics/Anti- inflammatories | Ketoprofen (KPF) | 0.000954 | 1642 | 248 | 32 | [3] | 5.8 × 10−3 | 3.85 × 10−3 | 2.40 × 10−2 |
| Naproxen (NPX) | 0.005373 | 223 | 15 | 34 | [3] | 2.44 × 10−1 | 3.583 × 10−2 | 1.58 × 10−1 | |
| Stimulants | Caffeine (CAF) | 0.004924 | >1502 | 160 | 1514 | [1] | 3.28 × 10−2 | 3.08 × 10−2 | 3.26 × 10−2 |
| Cumulative HQs | 1.62 | 0.36 | 0.27 | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vassalle, L.; Sunyer-Caldú, A.; Díaz-Cruz, M.S.; Arashiro, L.T.; Ferrer, I.; Garfí, M.; García-Galán, M.J. Behavior of UV Filters, UV Blockers and Pharmaceuticals in High Rate Algal Ponds Treating Urban Wastewater. Water 2020, 12, 2658. https://doi.org/10.3390/w12102658
Vassalle L, Sunyer-Caldú A, Díaz-Cruz MS, Arashiro LT, Ferrer I, Garfí M, García-Galán MJ. Behavior of UV Filters, UV Blockers and Pharmaceuticals in High Rate Algal Ponds Treating Urban Wastewater. Water. 2020; 12(10):2658. https://doi.org/10.3390/w12102658
Chicago/Turabian StyleVassalle, Lucas, Adrià Sunyer-Caldú, M. Silvia Díaz-Cruz, Larissa Terumi Arashiro, Ivet Ferrer, Marianna Garfí, and Mª Jesús García-Galán. 2020. "Behavior of UV Filters, UV Blockers and Pharmaceuticals in High Rate Algal Ponds Treating Urban Wastewater" Water 12, no. 10: 2658. https://doi.org/10.3390/w12102658
APA StyleVassalle, L., Sunyer-Caldú, A., Díaz-Cruz, M. S., Arashiro, L. T., Ferrer, I., Garfí, M., & García-Galán, M. J. (2020). Behavior of UV Filters, UV Blockers and Pharmaceuticals in High Rate Algal Ponds Treating Urban Wastewater. Water, 12(10), 2658. https://doi.org/10.3390/w12102658









