Figure 1.
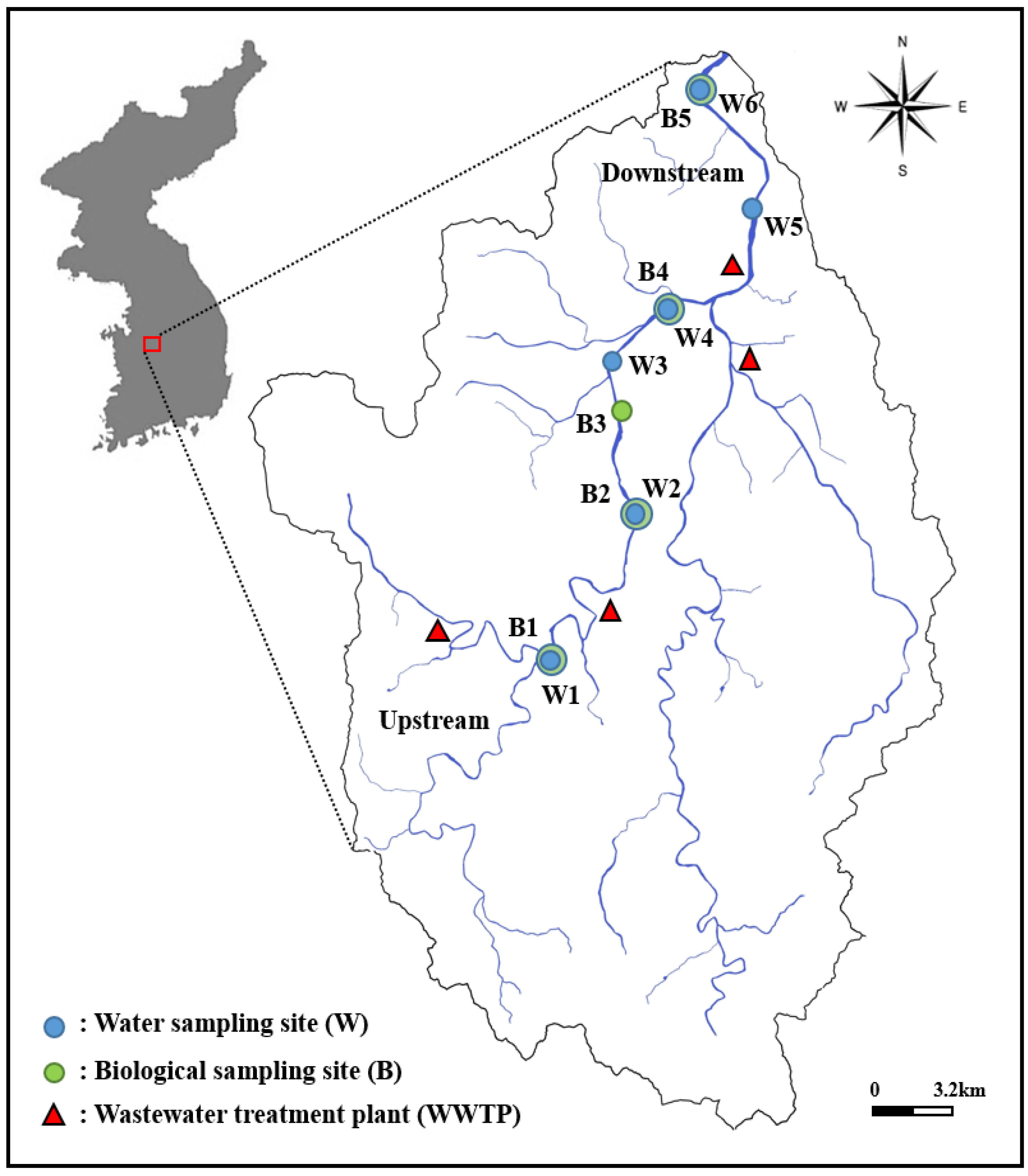
Watershed area and location of water (W) quality and biological (B) sampling sites in Gap Stream, South Korea. Where B1–B5 are the biological (B) sampling sites and W1–W6 are the water (W) quality monitoring sites.
Figure 1.
Watershed area and location of water (W) quality and biological (B) sampling sites in Gap Stream, South Korea. Where B1–B5 are the biological (B) sampling sites and W1–W6 are the water (W) quality monitoring sites.
Figure 2.
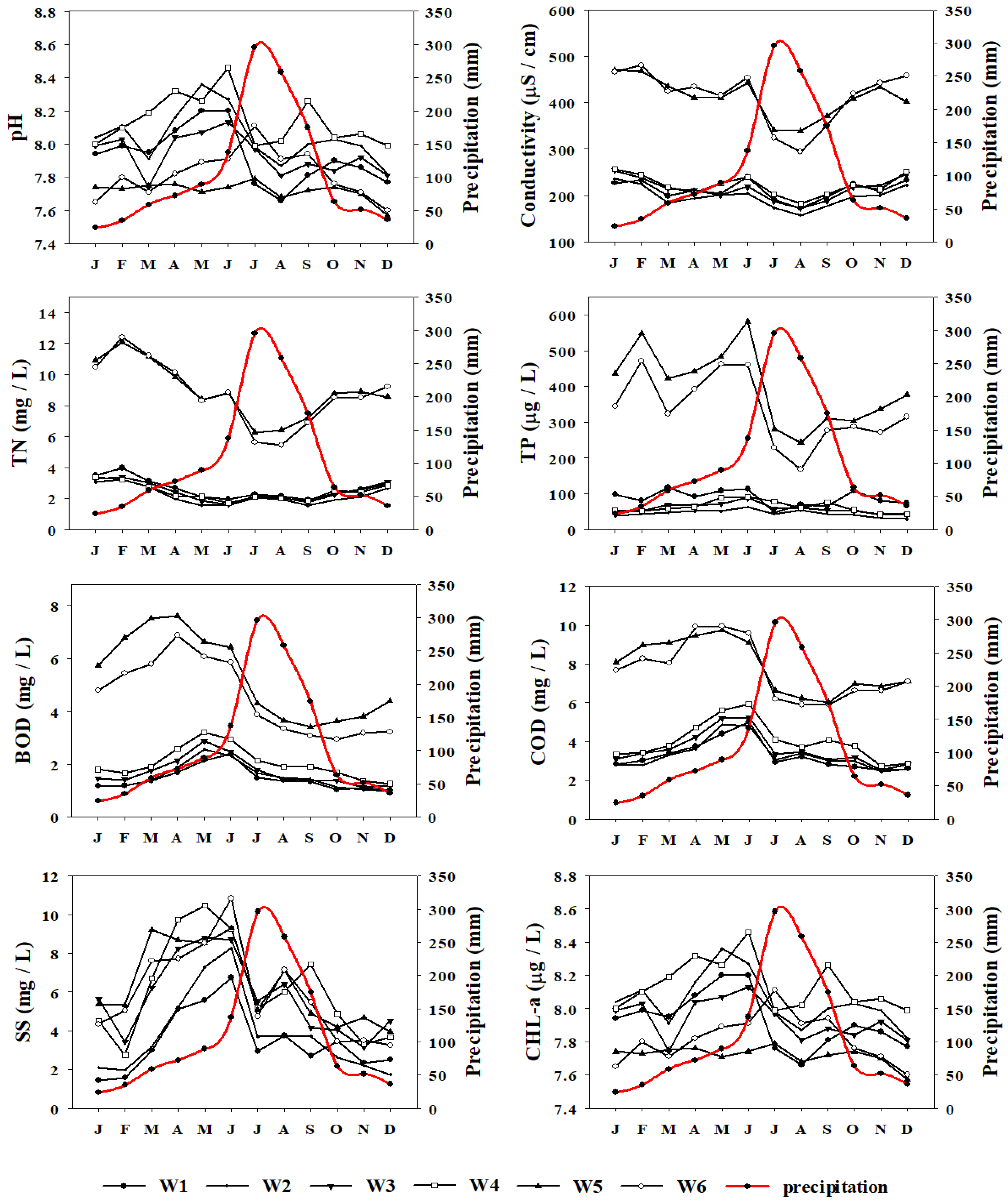
Monthly variation in selected water quality parameters in relation to monthly precipitation shown as (Spline red curve) in Gap stream during 2007–2016. TN = Total nitrogen, TP = Total phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a, W1–W6 = Water quality sites.
Figure 2.
Monthly variation in selected water quality parameters in relation to monthly precipitation shown as (Spline red curve) in Gap stream during 2007–2016. TN = Total nitrogen, TP = Total phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a, W1–W6 = Water quality sites.
Figure 3.
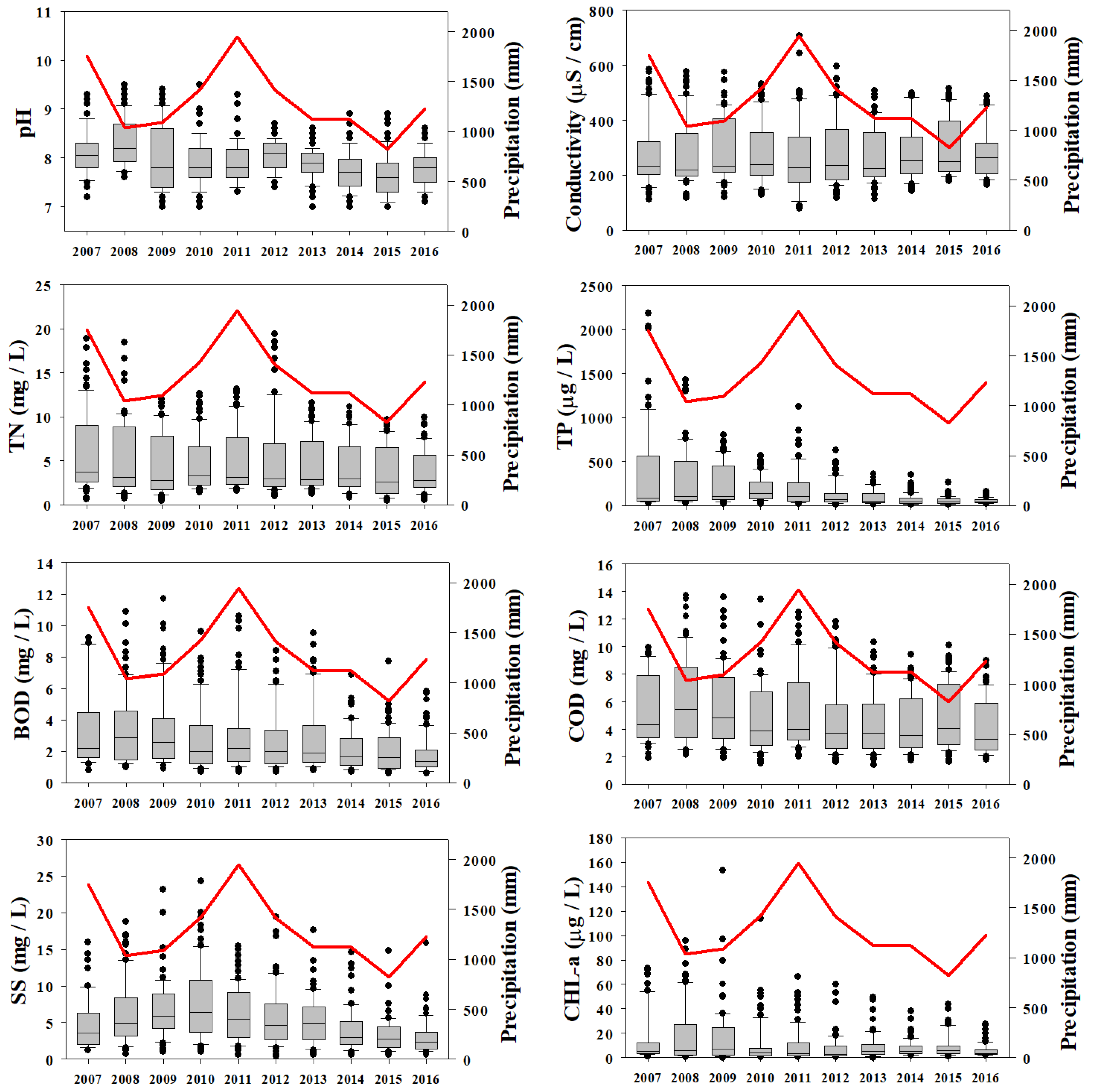
Annual variation in water quality parameters in relation to annual precipitation (shown as red curve) in Gap-stream during 2007–2016 (TN = Total nitrogen, TP = Total phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a).
Figure 3.
Annual variation in water quality parameters in relation to annual precipitation (shown as red curve) in Gap-stream during 2007–2016 (TN = Total nitrogen, TP = Total phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a).
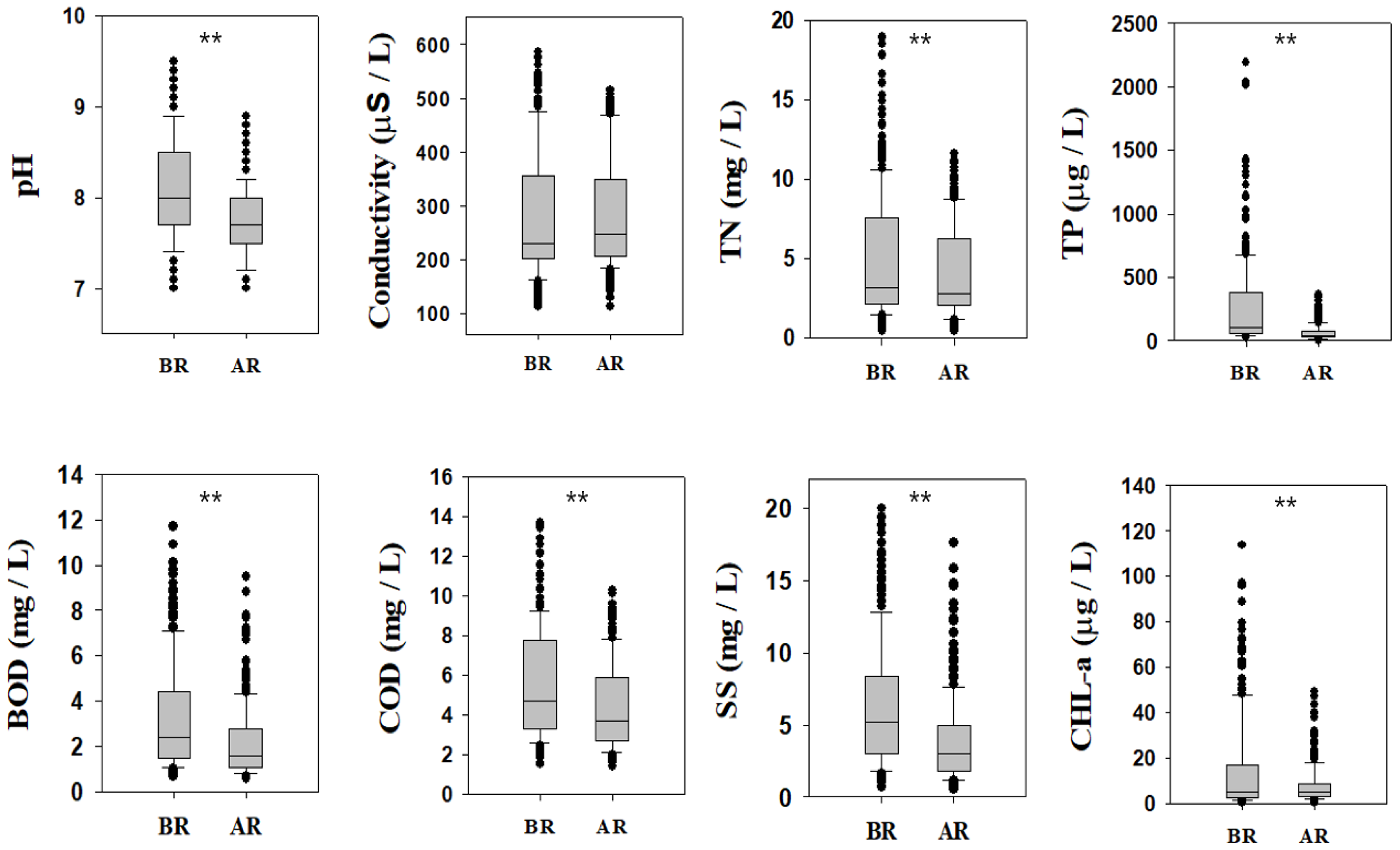
Figure 4.
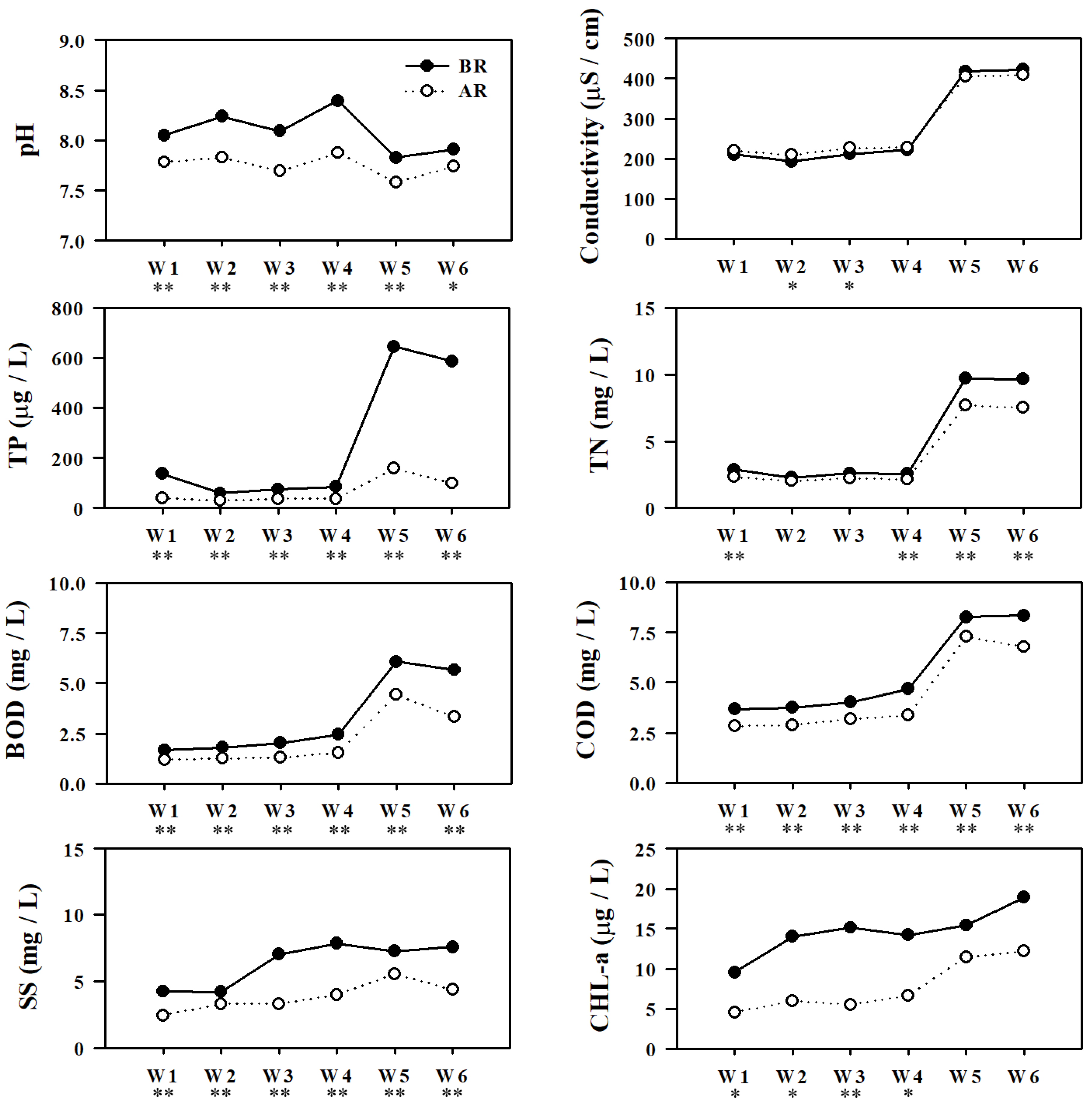
Comparison of water quality parameters to estimate the effect of restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, * = p < 0.05, ** = p < 0.01).
Figure 4.
Comparison of water quality parameters to estimate the effect of restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, * = p < 0.05, ** = p < 0.01).
Figure 5.
Spatial variation in selected water quality parameters at each sampling site of Gap-stream because of result of restoration measures. (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, ** = p < 0.01).
Figure 5.
Spatial variation in selected water quality parameters at each sampling site of Gap-stream because of result of restoration measures. (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, ** = p < 0.01).
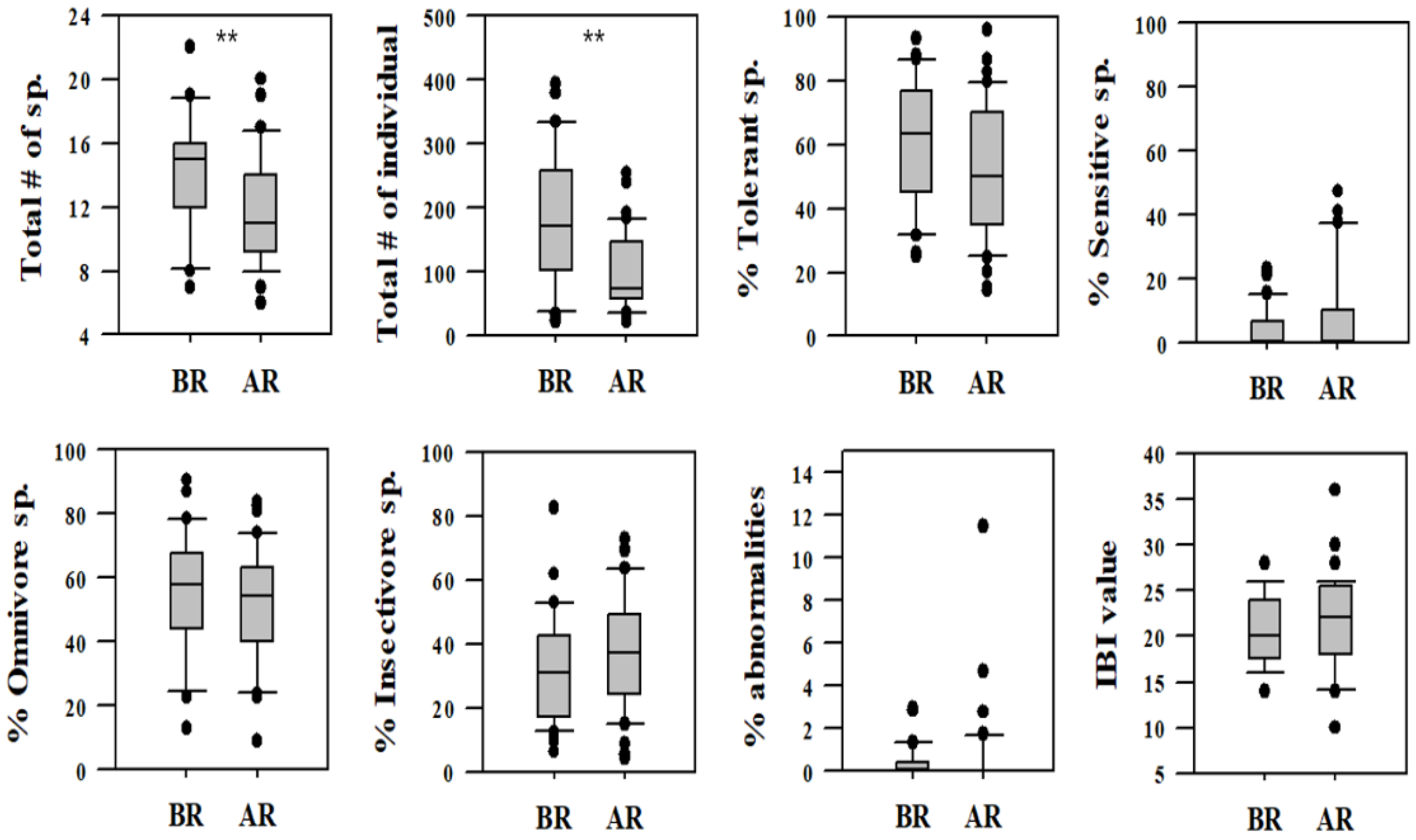
Figure 6.
Variation in different fish assemblages (trophic and tolerance guilds) as well as IBI values as ecological health parameters based on restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, ** = p < 0.01).
Figure 6.
Variation in different fish assemblages (trophic and tolerance guilds) as well as IBI values as ecological health parameters based on restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, ** = p < 0.01).
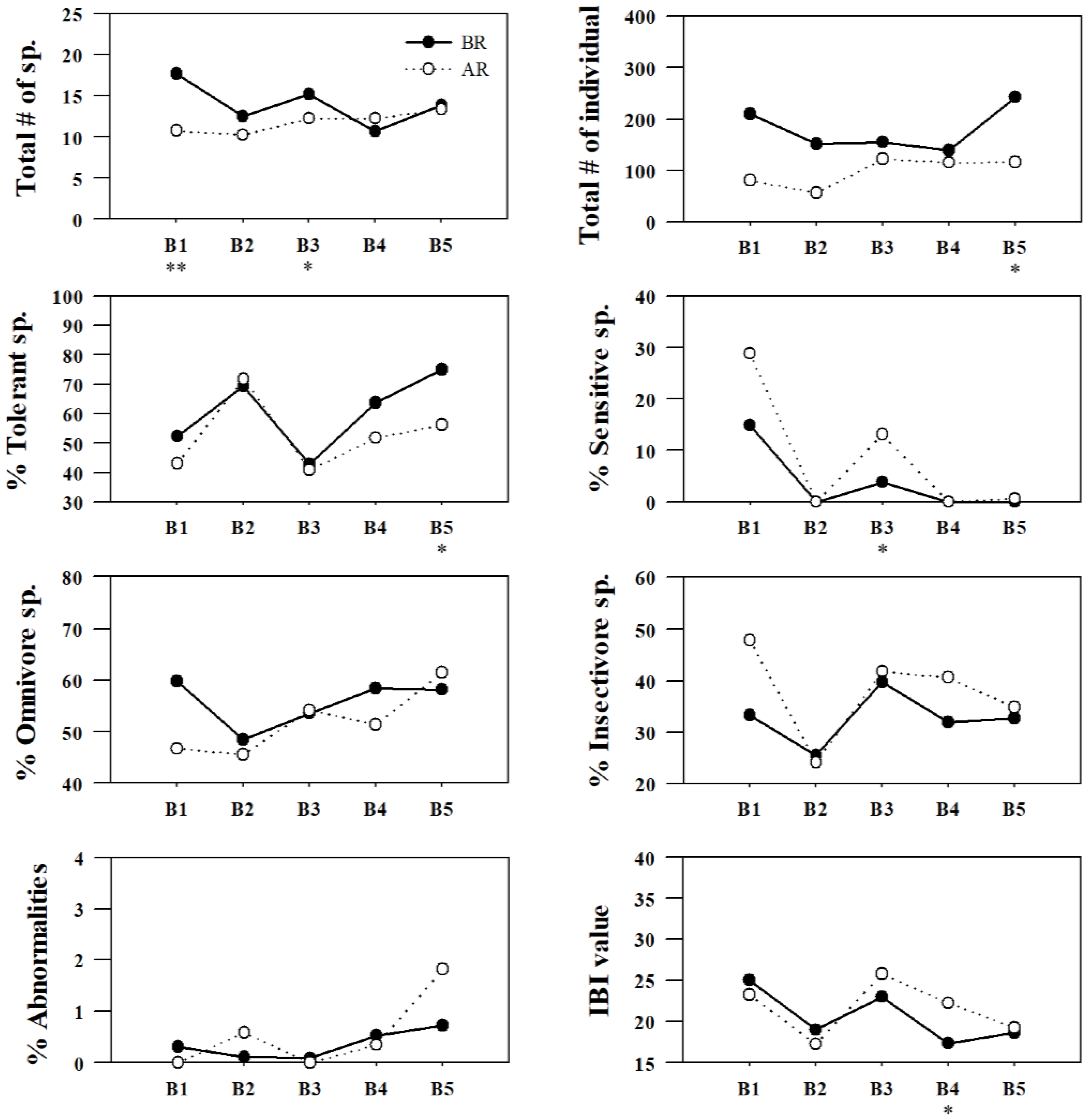
Figure 7.
Spatial variation in fish assemblages (trophic and tolerance guilds) as well as IBI values as ecological health parameters based on restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, * = p < 0.05, ** = p < 0.01).
Figure 7.
Spatial variation in fish assemblages (trophic and tolerance guilds) as well as IBI values as ecological health parameters based on restoration measures in Gap-stream (BR = before restoration, 2007–2010; AR = after restoration, 2013–2016, * = p < 0.05, ** = p < 0.01).
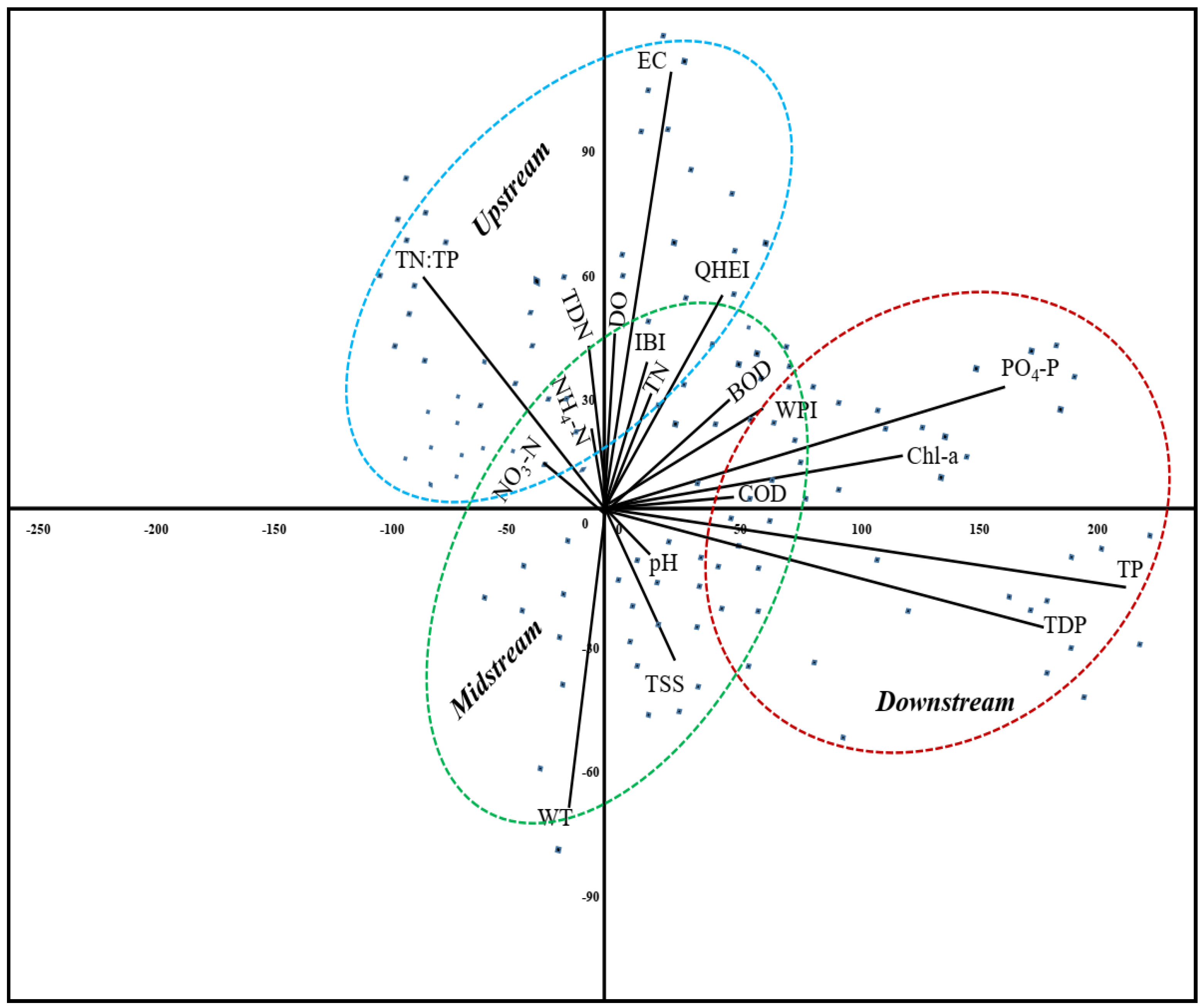
Figure 8.
PCA plot showing the settlement of water quality parameters as well as IBI, QHEI and WPI in the three main compartments of Gap Stream i.e., upstream, midstream and downstream during 2007–2016. (WT = Water temperature, EC = Electrical conductivity, DO = Dissolved oxygen, TDN = Total dissolved nitrogen, NH4-N = Ammonia nitrogen, NO3-N = Nitrate nitrogen, TN = Total nitrogen, TP = Total phosphorus, TDP = Total dissolved phosphorus, PO4-P =Phosphate phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a, WPI = Water pollution index, IBI = Index of biotic integrity, QHEI = Qualitative habitat evaluation index).
Figure 8.
PCA plot showing the settlement of water quality parameters as well as IBI, QHEI and WPI in the three main compartments of Gap Stream i.e., upstream, midstream and downstream during 2007–2016. (WT = Water temperature, EC = Electrical conductivity, DO = Dissolved oxygen, TDN = Total dissolved nitrogen, NH4-N = Ammonia nitrogen, NO3-N = Nitrate nitrogen, TN = Total nitrogen, TP = Total phosphorus, TDP = Total dissolved phosphorus, PO4-P =Phosphate phosphorus, BOD = Biological oxygen demand, COD = Chemical oxygen demand, SS = Suspended solids, CHL-a = Chlorophyll-a, WPI = Water pollution index, IBI = Index of biotic integrity, QHEI = Qualitative habitat evaluation index).
Table 1.
Geographical location coordinates of water quality monitoring and biological sampling sites in Gap Stream, South Korea.
Table 1.
Geographical location coordinates of water quality monitoring and biological sampling sites in Gap Stream, South Korea.
| Sites | Site Location | Latitude | Longitude |
|---|
| B1, W1 | Bonggok 2, Bonggok-dong, Seo-gu, Daejeon | 36°15′11.75″ N | 127°19′24.91″ E |
| B2, W2 | Singing bridge of Junglim-dong, Seo-gu, Daejeon | 36°18′23.67″ N | 127°21′32.29″ E |
| B3 | Wolpin Park in Gomma-dong, Seo-gu, Daejeon | 36°19′34.31″ N | 127°21′15.15″ E |
| B4, W4 | Daecheon Bridge, Samcheon-dong, Seo-gu, Daejeon | 36°22′16.24″ N | 127°22′32.09″ E |
| B5, W6 | Daejeon Metropolitan City | 36°26′48.13″ N | 127°23′23.06″ E |
| W3 | Daejeon Metropolitan City Wolpyeong-dong | 36°20′35.50″ N | 127°21′06.16″ E |
| W5 | Gapcheon Bridge in Jeonmin-dong, Yuseong-gu, Daejeon | 36°24′15.25″ N | 127°24′48.87″ E |
Table 2.
Summary statistics of water chemistry factors in Gap Stream during 2007–2016.
Table 2.
Summary statistics of water chemistry factors in Gap Stream during 2007–2016.
| Sites | Attributes | pH | DO | WT | BOD | COD | TSS | EC | TN | TP | TN:TP | TDN | NH4-N | NO3-N | TDP | PO4-P | CHL-a |
|---|
| W1 | Min. | 7.1 | 7.9 | 2.0 | 0.6 | 1.4 | 0.4 | 80 | 1.0 | 9.0 | 6.1 | 0.9 | 0.0 | 0.5 | 5.0 | 0.0 | 0.0 |
| Max. | 9.1 | 16.6 | 30 | 4.0 | 6.6 | 13.6 | 326 | 6.4 | 291 | 321 | 5.5 | 1.0 | 4.6 | 271 | 216 | 66 |
| Mean | 7.9 | 11.4 | 15.5 | 1.4 | 3.3 | 3.4 | 212 | 2.6 | 87.9 | 52.9 | 2.4 | 0.1 | 1.9 | 73.5 | 50.9 | 7.3 |
| Stand. dev. | 0.4 | 2.0 | 8.5 | 0.7 | 1.1 | 2.6 | 44 | 0.9 | 67.7 | 52.5 | 0.8 | 0.1 | 0.8 | 62.2 | 53.2 | 11.9 |
| W2 | Min. | 7.1 | 7.2 | 0.0 | 0.6 | 1.6 | 0.6 | 78 | 0.4 | 8.0 | 7.4 | 0.3 | 0.0 | 0.1 | 3.0 | 0.0 | 0.3 |
| Max. | 9.5 | 15.6 | 30.8 | 4.1 | 8.0 | 14.6 | 298 | 5.7 | 132 | 324.5 | 4.4 | 0.3 | 3.6 | 100 | 94 | 113.7 |
| Mean | 8.0 | 11.2 | 15 | 1.5 | 3.3 | 3.8 | 198.2 | 2.2 | 44.6 | 69.6 | 1.9 | 0.0 | 1.4 | 30.3 | 18.3 | 9.6 |
| Stand. dev. | 0.5 | 2.2 | 9.2 | 0.7 | 1.2 | 2.8 | 39.9 | 0.9 | 23 | 62.9 | 0.9 | 0.0 | 0.8 | 20 | 18.6 | 15.9 |
| W3 | Min. | 7.0 | 7.1 | 0.0 | 0.8 | 1.6 | 0.8 | 88 | 0.7 | 11 | 10.7 | 0.2 | 0.0 | 0.1 | 5.0 | 0.0 | 0.0 |
| Max. | 9.4 | 15.2 | 29 | 4.4 | 8.8 | 24.3 | 322 | 6.4 | 168 | 279.9 | 4.2 | 1.0 | 3.6 | 100 | 90 | 79.3 |
| Mean | 7.9 | 10.8 | 14.4 | 1.7 | 3.6 | 5.7 | 214.6 | 2.4 | 58.6 | 58.3 | 2.1 | 0.1 | 1.6 | 36.7 | 23.7 | 9.6 |
| Stand. dev. | 0.5 | 2.1 | 8.9 | 0.8 | 1.3 | 4.6 | 42.5 | 0.9 | 31.7 | 47.8 | 0.8 | 0.2 | 0.8 | 20.6 | 19.5 | 15.8 |
| W4 | Min. | 7.1 | 7.4 | 1.0 | 0.8 | 2.0 | 0.8 | 104 | 0.5 | 7.0 | 7.3 | 0.4 | 0.0 | 0.0 | 2.0 | 0.0 | 0.5 |
| Max. | 9.5 | 16.5 | 31 | 5.5 | 9.7 | 19.4 | 327 | 5.2 | 235 | 356.1 | 4.2 | 1.0 | 3.5 | 230 | 211 | 95.8 |
| Mean | 8.1 | 11.2 | 15.8 | 2.0 | 4.0 | 6.2 | 222.4 | 2.4 | 63.4 | 61.4 | 2.1 | 0.1 | 1.5 | 40.6 | 23 | 10 |
| Stand. dev. | 0.6 | 2.2 | 9.2 | 1.0 | 1.6 | 4.6 | 44.9 | 0.9 | 42.9 | 61.9 | 0.8 | 0.2 | 0.8 | 34.7 | 29.7 | 14.6 |
| W5 | Min. | 7.0 | 7.1 | 5.0 | 1.5 | 3.6 | 0.8 | 185 | 3.8 | 62 | 5.7 | 3.4 | 0.0 | 0.8 | 31 | 24 | 0.0 |
| Max. | 9.1 | 15.6 | 31 | 10.9 | 13.4 | 20 | 707 | 18.5 | 2188 | 103.8 | 18.3 | 15.9 | 10.3 | 1964 | 1159 | 96.8 |
| Mean | 7.7 | 10.1 | 18.4 | 5.3 | 7.9 | 6.4 | 411.3 | 8.9 | 397.2 | 36.7 | 8.1 | 2.3 | 4.4 | 327.8 | 218.8 | 12.3 |
| Stand. dev. | 0.4 | 1.6 | 7.7 | 2.2 | 1.9 | 3.5 | 89.6 | 2.9 | 357.3 | 24 | 2.7 | 2.7 | 1.8 | 318.7 | 206.7 | 15.9 |
| W6 | Min. | 7.0 | 7.6 | 3.0 | 1.5 | 3.0 | 1.2 | 126 | 2.1 | 50 | 5.4 | 2.0 | 0.0 | 1.3 | 37 | 3.0 | 0.1 |
| Max. | 9.1 | 16.2 | 31.7 | 11.7 | 13.7 | 23.2 | 644 | 19.5 | 2011 | 158.3 | 18.6 | 13.7 | 10 | 1953 | 1270 | 153.4 |
| Mean | 7.8 | 11 | 18.3 | 4.5 | 7.7 | 6.0 | 413.8 | 8.8 | 333.6 | 49.7 | 8.1 | 1.9 | 5.0 | 282.3 | 185.6 | 14.9 |
| Stand. dev. | 0.4 | 1.6 | 8.0 | 2.2 | 2.2 | 3.9 | 100.7 | 3.2 | 336.1 | 35.8 | 3.0 | 2.7 | 1.9 | 312.8 | 216.2 | 20.3 |
Table 3.
Annual results of water pollution index (WPI) and water quality status evaluation in Gap Stream during 2007–2016.
Table 3.
Annual results of water pollution index (WPI) and water quality status evaluation in Gap Stream during 2007–2016.
| Category | Metrics (M) | Mean and Stand. Deviation (Obtained Metric Score) |
|---|
| 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|
| Nutrient Regime | M1: Total Nitrogen (mg/L) | 5.75 ± 4.52 | 3.78 ± 3.21 | 2.88 ± 2.62 | 3.74 ± 2.23 | 3.77 ± 2.98 | 3.90 ± 4.19 | 3.50 ± 2.25 | 3.15 ± 1.91 | 2.80 ± 2.40 | 2.79 ± 1.63 |
| (1) | (1) | (3) | (1) | (1) | (1) | (1) | (1) | (3) | (3) |
| M2: Total Phosphorus (µg/L) | 356.2 ± 505 | 242 ± 313 | 182 ± 164 | 117 ± 63.1 | 131.2 ± 86.8 | 100 ± 82.1 | 69.6 ± 41.5 | 59.4 ± 29.8 | 45.9 ± 22.1 | 41.5 ± 19.1 |
| (1) | (1) | (1) | (1) | (1) | (3) | (3) | (3) | (3) | (3) |
| M3: TN:TP ratio | 40.15 ± 35.5 | 32.2 ± 18.5 | 37.2 ± 15.9 | 44.1 ± 14.8 | 48.6 ± 24.5 | 39.4 ± 26.1 | 39.2 ± 31.1 | 37.8 ± 22.2 | 48.2 ± 22.5 | 47.2 ± 24.6 |
| (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) |
| Organic Matter | M4: Biological Oxygen Demand (mg/L) | 5.35 ± 2.4 | 3.46 ± 1.83 | 3.68 ± 2.15 | 1.98 ± 1.42 | 2.83 ± 1.91 | 2.85 ± 1.33 | 2.46 ± 1.51 | 1.87 ± 0.74 | 1.89 ± 0.77 | 0.78 ± 0.99 |
| (1) | (1) | (1) | (3) | (1) | (1) | (3) | (5) | (3) | (5) |
| Ionic Contents & Solids | M5: Total Suspended Solids (mg/L) | 4.8 ± 3.5 | 8.21 ± 3.15 | 7.31 ± 2.54 | 8.17 ± 4.03 | 6.93 ± 2.53 | 7.31 ± 2.20 | 5.61 ± 2.50 | 5.60 ± 1.26 | 3.31 ± 0.87 | 3.36 ± 1.46 |
| (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (5) | (5) |
| M6: Electrical Conductivity (µS/cm) | 279 ± 127 | 242 ± 109 | 232.8 ± 79.2 | 208.9 ± 70.9 | 245.6 ± 110 | 236.9 ± 83.8 | 226.7 ± 72.3 | 247.9 ± 76.6 | 268.7 ± 88.7 | 242.8 ± 68.4 |
| (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) |
| Primary Production | M7: Sestonic Chlorophyll-a (µg/L) | 13.42 ± 19.3 | 30.2 ± 16.2 | 26.1 ± 12.8 | 4.88 ± 4.14 | 16.7 ± 8.48 | 12.7 ± 6.74 | 11.2 ± 5.47 | 9.08 ± 3.70 | 9.87 ± 7.3 | 6.22 ± 5.53 |
| (1) | (1) | (1) | (3) | (1) | (1) | (1) | (3) | (3) | (3) |
| Final Scores of WPI Water Quality Criteria | 13 | 13 | 15 | 17 | 13 | 16 | 16 | 21 | 23 | 25 |
| Poor | Poor | Poor | Poor | Poor | Poor | Poor | Fair | Fair | Good |
Table 4.
Annual results of qualitative habitat evaluation index (QHEI) and habit status evaluation in Gap Stream during 2007–2016.
Table 4.
Annual results of qualitative habitat evaluation index (QHEI) and habit status evaluation in Gap Stream during 2007–2016.
| Model Metrics (M) | Mean and Stand. Deviation |
|---|
| 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|
| M1: Epifaunal substrate, available cover | 13.3 ± 4 | 13.4 ± 4.1 | 13.4 ± 4.5 | 13.4 ± 4.5 | 13.1 ± 4.3 | 13.4 ± 4.5 | 13.4 ± 4.5 | 13.6 ± 4.6 | 13.6 ± 4.6 | 13.6 ± 4.6 |
| M2: Embeddedness/pool substrate characterization | 15.1 ± 3.7 | 15.0 ± 3.9 | 16.2 ± 3.2 | 15.2 ± 4.1 | 15.2 ± 3.7 | 15.8 ± 3.5 | 15.8 ± 3.5 | 15.8 ± 3.5 | 15.6 ± 3.3 | 15.8 ± 3.5 |
| M3: Velocity, depth combination/pool variability | 16.1 ± 2.5 | 16.4 ± 2.4 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 | 16.8 ± 2.5 |
| M4: Sediment deposition | 18.2 ± 2.6 | 18.4 ± 2.8 | 17.1 ± 2.7 | 17.6 ± 2.8 | 16.7 ± 3.1 | 18.4 ± 2.8 | 18.4 ± 2.8 | 18.2 ± 2.9 | 17.1 ± 3.1 | 18.2 ± 2.9 |
| M5: Channel flow status | 16.4 ± 2.9 | 17.4 ± 2.7 | 16.7 ± 2.4 | 17.3 ± 2.7 | 17.2 ± 2.9 | 17.4 ± 2.5 | 17.6 ± 2.6 | 17.6 ± 2.6 | 16.7 ± 3.3 | 17.4 ± 2.7 |
| M6: Channel alteration | 10.5 ± 6.2 | 10.8 ± 6.1 | 10.8 ± 6.1 | 10.8 ± 6.1 | 10.3 ± 6.0 | 10.8 ± 6.1 | 10.8 ± 6.1 | 10.8 ± 6.1 | 10.8 ± 6.1 | 10.8 ± 6.1 |
| M7: Frequency of riffles or bends/channel sinuosity | 9.1 ± 5.5 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 | 9.2 ± 5.6 |
| M8: Bank stability | 10.4 ± 5.9 | 10.4 ± 5.9 | 10.4 ± 5.9 | 10.4 ± 5.9 | 10.2 ± 5.7 | 10.0 ± 5.5 | 10.2 ± 5.7 | 10.4 ± 5.9 | 10.6 ± 6.1 | 10.8 ± 6.3 |
| M9: Bank vegetative protection | 10.1 ± 7.4 | 10.0 ± 7.5 | 10.2 ± 7.3 | 10.0 ± 7.5 | 10.6 ± 6.6 | 10.8 ± 6.8 | 10.8 ± 6.8 | 11.0 ± 6.6 | 11.0 ± 6.6 | 10.8 ± 6.8 |
| M10: Riparian vegetative zone width | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 | 11.8 ± 5.7 |
| M11: Small scale weir construction | 11.0 ± 5.6 | 11.0 ± 5.6 | 11.3 ± 5.4 | 12.0 ± 4.4 | 11.2 ± 4.0 | 12.0 ± 4.4 | 12.0 ± 4.4 | 12.4 ± 4.6 | 12.4 ± 4.6 | 12.4 ± 4.6 |
| QHEI Score | 142 ± 42.5 | 143.8 ± 43.7 | 143.9 ± 42.4 | 144.5 ± 41.8 | 142.3 ± 38.5 | 146.4 ± 41.4 | 146.8 ± 41.5 | 147.6 ± 41.8 | 145.6 ± 39.8 | 147.6 ± 42.3 |
| QHEI Criteria | Good | Good | Good | Good | Good | Good | Good | Good | Good | Good |
Table 5.
Spatial distribution of fish species sampled from Gap Stream and their difference before and after restoration.
Table 5.
Spatial distribution of fish species sampled from Gap Stream and their difference before and after restoration.
| Fish Species | Fish Guilds | Before Restoration (BR) | After Restoration (AR) | TNI | RA (%) |
|---|
| TO | TR | HA | B1 | B2 | B3 | B4 | B5 | TNI | RA (%) | B1 | B2 | B3 | B4 | B5 | TNI | RA (%) |
|---|
| Zacco platypus | | | | 221 | 69 | 93 | 116 | 184 | 683 | 37.6 | 55 | 26 | 78 | 69 | 53 | 281 | 27.7 | 482 | 33.7 |
| Pseudogobio esocinus | IS | I | - | 4 | 30 | 49 | 12 | 25 | 120 | 6.6 | 4 | 7 | 11 | 18 | 38 | 78 | 7.7 | 99 | 6.9 |
| Carassius auratus | TS | O | - | 4 | 38 | 6 | 34 | 50 | 132 | 7.3 | 6 | 8 | 1 | 23 | 19 | 57 | 5.6 | 94.5 | 6.6 |
| Acheilognathus lanceolatus | IS | O | - | 20 | 16 | 29 | 28 | 19 | 112 | 6.2 | 8 | 5 | 15 | 17 | 23 | 68 | 6.7 | 90 | 6.3 |
| Hemibarbus labeo | TS | I | - | | 13 | 14 | 17 | 73 | 117 | 6.4 | | 9 | 3 | 22 | 26 | 60 | 5.9 | 88.5 | 6.2 |
| Opsarichthys uncirostris amurensis | TS | C | - | 1 | 40 | 12 | 17 | 42 | 112 | 6.2 | 4 | 9 | 5 | 6 | 1 | 25 | 2.5 | 68.5 | 4.8 |
| Rhinogobius brunneus | IS | I | RB | 9 | 6 | 10 | 3 | 3 | 31 | 1.7 | 7 | 2 | 39 | 13 | 6 | 67 | 6.6 | 49 | 3.4 |
| Zacco koreanus | SS | I | - | 48 | | 1 | | | 49 | 2.7 | 40 | | | | | 40 | 3.9 | 44.5 | 3.1 |
| Pungtungia herzi | IS | I | - | 10 | 1 | 7 | 4 | 3 | 25 | 1.4 | 12 | 3 | 10 | 17 | 3 | 45 | 4.4 | 35 | 2.4 |
| Microphysogobio yaluensis | IS | O | RB | 7 | | 13 | 1 | 2 | 23 | 1.3 | 3 | | 22 | 4 | 7 | 36 | 3.6 | 29.5 | 2.1 |
| Hemibarbus lon girostris | IS | I | - | 6 | 6 | 11 | 10 | 6 | 39 | 2.1 | 2 | 1 | 3 | 9 | 4 | 19 | 1.9 | 29 | 2.0 |
| Coreoleuciscus splendidus | SS | I | RB | 6 | | 7 | | | 13 | 0.7 | 2 | | 37 | | 1 | 40 | 3.9 | 26.5 | 1.9 |
| Micropterus salmoides | TS | C | - | 4 | 26 | 5 | 1 | 1 | 37 | 2.0 | 1 | 6 | 2 | 3 | 2 | 14 | 1.4 | 25.5 | 1.8 |
| Pseudorasbora parva | TS | O | - | | 3 | | 11 | 14 | 28 | 1.5 | 1 | 1 | | 1 | 17 | 20 | 2.0 | 24 | 1.7 |
| Acanthorhodeus macropterus | IS | O | - | 1 | 1 | | 2 | 23 | 27 | 1.5 | 1 | 1 | 1 | 10 | 3 | 16 | 1.6 | 21.5 | 1.5 |
| Acanthorhodeus gracilis | IS | O | - | 1 | | 20 | 4 | 3 | 28 | 1.5 | | 1 | 1 | 8 | 1 | 11 | 1.1 | 19.5 | 1.4 |
| Odontobutis interrupta | IS | C | - | 6 | 10 | 4 | 1 | 4 | 25 | 1.4 | 3 | 5 | 1 | 1 | 2 | 12 | 1.2 | 18.5 | 1.3 |
| Misgurnus anguillicaudatus | TS | O | - | 2 | 19 | 1 | 1 | 1 | 24 | 1.3 | 1 | 5 | | | | 6 | 0.6 | 15 | 1.0 |
| Gnathopogon strigatus | IS | I | - | 5 | 2 | 3 | 2 | 5 | 17 | 0.9 | | | | 2 | 11 | 13 | 1.3 | 15 | 1.0 |
| Acheilognathus rhombeus | IS | O | - | 1 | | 6 | 1 | 1 | 9 | 0.5 | | 2 | 10 | 3 | 5 | 20 | 2.0 | 14.5 | 1.0 |
| Squalidus gracilis majimae | IS | I | - | 23 | | | | | 23 | 1.3 | 3 | | | | | 3 | 0.3 | 13 | 0.9 |
| Iksookimia koreensis | IS | I | RB | 9 | 1 | 10 | | | 20 | 1.1 | 3 | | 1 | | | 4 | 0.4 | 12 | 0.8 |
| Lepomis macrochirus | TS | I | - | | | | | | | | | 20 | | 1 | 1 | 22 | 2.2 | 22 | 1.5 |
| Pseudobagrus koreanus | IS | I | RB | 2 | | 6 | | 1 | 9 | 0.5 | 4 | | 3 | 1 | 1 | 9 | 0.9 | 9 | 0.6 |
| Misgurnus mizolepis | TS | O | - | 2 | 15 | | | | 17 | 0.9 | | 1 | | | | 1 | 0.1 | 9 | 0.6 |
| Cyprinus carpio | TS | O | - | | | 1 | 9 | 3 | 13 | 0.7 | | 1 | 1 | 1 | 1 | 4 | 0.4 | 8.5 | 0.6 |
| Squaliobarbus curriculus | IS | O | - | | | | | 13 | 13 | 0.7 | | | | | 2 | 2 | 0.2 | 7.5 | 0.5 |
| Odontobutis platycephala | SS | C | - | 8 | | | | | 8 | 0.4 | 1 | | 1 | | 1 | 3 | 0.3 | 5.5 | 0.4 |
| Cobitis lutheri | IS | I | - | 5 | 2 | 1 | | | 8 | 0.4 | 1 | | 2 | | | 3 | 0.3 | 5.5 | 0.4 |
| Zacco temminckii | SS | I | - | 6 | | | | | 6 | 0.3 | 2 | | | | | 2 | 0.2 | 4 | 0.3 |
| Tridentiger brevispinis | IS | I | RB | 1 | | 1 | | 5 | 7 | 0.4 | | | | | 1 | 1 | 0.1 | 4 | 0.3 |
| Rhodeus uyekii | IS | O | - | 6 | | | | | 6 | 0.3 | | | 1 | | | 1 | 0.1 | 3.5 | 0.2 |
| Pseudobagrus fulvidraco | TS | I | - | 1 | 2 | 1 | | 1 | 5 | 0.3 | | | 1 | 1 | | 2 | 0.2 | 3.5 | 0.2 |
| Squalidus japonicus coreanus | TS | O | - | | | | | 1 | 1 | 0.1 | | | | 2 | 4 | 6 | 0.6 | 3.5 | 0.2 |
| Siniperca scherzeri | IS | C | - | | | | 2 | 1 | 3 | 0.2 | | | 1 | 2 | 1 | 4 | 0.4 | 3.5 | 0.2 |
| Sarcocheilichthys nigripinnis morii | IS | I | - | 1 | 1 | | 1 | | 3 | 0.2 | | 1 | | 1 | 1 | 3 | 0.3 | 3 | 0.2 |
| Squalidus chankaensis tsuchigae | IS | O | - | | | | 4 | | 4 | 0.2 | | | | | 2 | 2 | 0.2 | 3 | 0.2 |
| Silurus asotus | TS | C | - | 1 | 1 | 1 | | | 3 | 0.2 | | 1 | | | 1 | 2 | 0.2 | 2.5 | 0.2 |
| Channa argus | TS | C | - | | 1 | | 1 | | 2 | 0.1 | | 1 | | | 1 | 2 | 0.2 | 2 | 0.1 |
| Carassius cuvieri | TS | O | - | | | | 1 | 3 | 4 | 0.2 | | | | | | | | 4 | 0.3 |
| Erythroculter erythropterus | TS | C | - | | | | | 1 | 1 | 0.1 | | | | 1 | 1 | 2 | 0.2 | 1.5 | 0.1 |
| Iksookimia choii | SS | I | - | | | 3 | | | 3 | 0.2 | | | | | | | | 3 | 0.2 |
| Acheilognathus koreensis | IS | O | - | 1 | | | | | 1 | 0.1 | 1 | | | | 1 | 2 | 0.2 | 1.5 | 0.1 |
| Rhodeus notatus | IS | O | - | | 2 | | | | 2 | 0.1 | | | | | | | | 2 | 0.1 |
| Sarcocheilichthys variegatus wakiyae | IS | I | | | | | | 1 | 1 | 0.1 | | 1 | | | | 1 | 0.1 | 1 | 0.1 |
| Rhinogobius giurinus | TS | O | - | | | | | | | | | | | 1 | | 1 | 0.1 | 1 | 0.1 |
| Pseudopungtungia nigra | SS | I | - | | | | | | | | | | 1 | | | 1 | 0.1 | 1 | 0.1 |
| Oryzias sinensis | TS | O | - | | | | | | | | | | | | 1 | 1 | 0.1 | 1 | 0.1 |
| Monopterus albus | TS | C | - | | | | | | | | | | 1 | | | 1 | 0.1 | 1 | 0.1 |
| Macropodus ocellatus | TS | I | - | | 1 | | | | 1 | 0.1 | | | | | | | | 1 | 0.1 |
| Total Number of Species | | | | 31 | 24 | 26 | 24 | 27 | 45 | | 23 | 23 | 26 | 26 | 33 | 46 | | 50 | |
| Total Number of individuals | | | | 422 | 306 | 315 | 283 | 489 | 1815 | | 165 | 117 | 252 | 237 | 242 | 1013 | | 1432 | |
Table 6.
Annual results of index of biotic integrity (IBI) and biological health evaluation in Gap Stream during 2007–2016.
Table 6.
Annual results of index of biotic integrity (IBI) and biological health evaluation in Gap Stream during 2007–2016.
| Category | Metric | Mean and Stand. Deviation (Obtained Metric Score) |
|---|
| 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|
| Species composition | M1: Total number of native fish species | 13.9 ± 2.1 | 13.1 ± 2.3 | 14.8 ± 3.0 | 12.4 ± 4.8 | 14.4 ± 5.3 | 12.9 ± 2.9 | 9.8 ± 2.9 | 11.6 ± 3.0 | 13.5 ± 3.3 | 9.3 ± 2.4 |
| (5) | (5) | (5) | (5) | (5) | (5) | (3) | (5) | (5) | (3) |
| M2: Number of riffle benthic species | 2.2 ± 1.5 | 2.0 ± 1.3 | 2.3 ± 1.8 | 2.2 ± 2.1 | 1.8 ± 1.5 | 2.2 ± 1.8 | 2.2 ± 1.8 | 1.8 ± 1.4 | 2.6 ± 1.8 | 1.5 ± 1.4 |
| (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (3) | (1) |
| M3: Number of sensitive species | 0.9 ± 1.1 | 0.8 ± 1.2 | 0.7 ± 0.9 | 1.0 ± 1.3 | 0.4 ± 0.7 | 0.9 ± 1.0 | 0.5 ± 0.5 | 0.6 ± 0.8 | 1.0 ± 1.4 | 0.6 ± 0.7 |
| (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) |
| M4: Percent individuals as tolerant species (%) | 60.7 ± 25.9 | 61.7 ± 25.8 | 58.9 ± 16.1 | 61.4 ± 14.7 | 50.7 ± 19.4 | 59.2 ± 22.2 | 46.0 ± 20.8 | 49.6 ± 16.0 | 49.0 ± 21.8 | 66.5 ± 19.6 |
| (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) |
| Trophic composition | M5: Proportion of individuals as native omnivores (%) | 56.7 ± 17.8 | 58.7 ± 17.2 | 55.5 ± 14.5 | 53.0 ± 24.3 | 53.4 ± 20.0 | 62.3 ± 15.2 | 49.0 ± 23.1 | 51.8 ± 15.7 | 50.3 ± 16.4 | 56.3 ± 15.3 |
| (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) | (1) |
| M6: Proportion of individuals as native insectivores (%) | 32.1 ± 15.5 | 31.0 ± 15.9 | 32.2 ± 12.7 | 34.8 ± 23.2 | 36.9 ± 19.2 | 31.6 ± 18.0 | 42.3 ± 20.6 | 40.8 ± 17.2 | 38.2 ± 17.6 | 30.2 ± 15.8 |
| (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) | (3) |
| Fish abundance and condition | M7: Total number of individuals | 192.2 ± 79.1 | 189.2 ± 79.8 | 165.3 ± 88.2 | 185 ± 139.4 | 138.0 ± 62.7 | 154.5 ± 50.7 | 69.3 ± 44.1 | 94.7 ± 47.8 | 143.9 ± 72.4 | 85.6 ± 41.9 |
| (3) | (3) | (3) | (3) | (3) | (3) | (1) | (1) | (3) | (1) |
| M8: Portion of individuals with abnormalities (%) | 0.3 ± 0.4 | 0.4 ± 0.5 | 0.4 ± 0.9 | 0.3 ± 0.9 | 0.0 ± 0.1 | 0.4 ± 0.6 | 1.1 ± 3.6 | 1.1 ± 1.6 | 0 | 0 |
| (3) | (3) | (3) | (3) | (3) | (3) | (1) | (1) | (5) | (5) |
| Sum of IBI Scores | 18 | 18 | 18 | 18 | 18 | 18 | 12 | 14 | 22 | 16 |
| Health status | Poor | Poor | Poor | Poor | Poor | Poor | V. poor | V. poor | Poor | Poor |
Table 7.
Correlation analysis on selected water quality parameters and indices used in Gap Stream during 2007–2016.
Table 7.
Correlation analysis on selected water quality parameters and indices used in Gap Stream during 2007–2016.
| Parameters | pH | EC | TN | TP | BOD | COD | TSS | CHL-a | WPI | QHEI | IBI |
|---|
| pH | 1 | | | | | | | | | | |
| EC | −0.194 | 1 | | | | | | | | | |
| TN | −0.115 | 0.878 ** | 1 | | | | | | | | |
| TP | 0.086 | 0.634 ** | 0.689 ** | 1 | | | | | | | |
| BOD | 0.094 | 0.781 ** | 0.826 ** | 0.789 ** | 1 | | | | | | |
| COD | 0.120 | 0.858 ** | 0.840 ** | 0.736 ** | 0.925 ** | 1 | | | | | |
| TSS | 0.390 ** | 0.161 | 0.264 * | 0.406 ** | 0.481 ** | 0.428 ** | 1 | | | | |
| CHL-a | 0.391 ** | 0.358 ** | 0.290 ** | 0.517 ** | 0.580 ** | 0.638 ** | 0.444 ** | 1 | | | |
| WPI | −0.263 * | −0.623 ** | −0.617 ** | −0.666 ** | −0.787 ** | −0.783 ** | −0.647 ** | −0.633 ** | 1 | | |
| QHEI | −0.230 * | −0.105 | −0.084 | −0.065 | −0.178 | −0.190 | −0.122 | −0.081 | 0.109 | 1 | |
| IBI | −0.089 | −0.139 | −0.231 * | −0.145 | −0.256 * | −0.229 * | −0.233 * | −0.120 | 0.167 | 0.569 ** | 1 |
Table 8.
Loading values, eigenvalues and variances of factors obtained from PCA in Gap Stream.
Table 8.
Loading values, eigenvalues and variances of factors obtained from PCA in Gap Stream.
| Parameters | PC 1 | PC 2 | PC 3 | PC 4 | PC 5 |
|---|
| pH | 0.001 | −0.001 | −0.463 | −0.001 | −0.002 |
| DO | 0.001 | 0.013 | −0.005 | 0.018 | −0.004 |
| BOD | 0.004 | 0.518 | −0.001 | −0.006 | −0.002 |
| COD | 0.575 | 0.006 | 0.002 | −0.011 | −0.005 |
| TSS | 0.006 | −0.012 | 0.011 | −0.029 | 0.002 |
| TN | 0.003 | 0.010 | −0.008 | 0.011 | 0.001 |
| TP | 0.690 | −0.078 | −0.294 | −0.069 | −0.032 |
| TN:TP | −0.102 | 0.224 | −0.228 | 0.910 | 0.061 |
| WT | −0.001 | −0.074 | 0.029 | −0.123 | −0.002 |
| EC | 0.072 | 0.935 | −0.138 | −0.235 | −0.196 |
| TDN | 0.002 | 0.010 | −0.008 | 0.008 | −0.001 |
| NH4-N | 0.002 | 0.005 | −0.005 | −0.465 | 0.002 |
| NO3-N | 0.000 | 0.003 | −0.002 | 0.646 | −0.005 |
| TDP | 0.612 | −0.080 | −0.220 | 0.135 | 0.076 |
| PO4-P | 0.356 | 0.119 | 0.870 | 0.237 | −0.204 |
| CHL-a | 0.032 | 0.010 | 0.033 | −0.118 | 0.655 |
| WPI | 0.008 | 0.008 | 0.023 | −0.004 | 0.872 |
| QHEI | 0.074 | 0.208 | 0.183 | −0.076 | 0.950 |
| IBI | 0.004 | 0.014 | 0.947 | −0.012 | 0.058 |
| Eigenvalue | 33,483.1 | 2985.62 | 1890.21 | 1112.01 | 1019.19 |
| % variance | 81.99 | 7.311 | 4.63 | 2.72 | 2.49 |
| % CV | 81.99 | 89.30 | 93.93 | 96.65 | 99.15 |