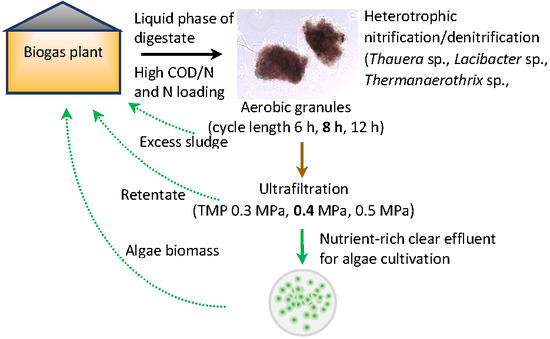
Treatment of Liquid Phase of Digestate from Agricultural Biogas Plant in a System with Aerobic Granules and Ultrafiltration
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate
2.2. GSBR Operation
2.3. Molecular Studies
2.4. Membrane Filtration
2.5. Statistical Analysis
3. Results and Discussion
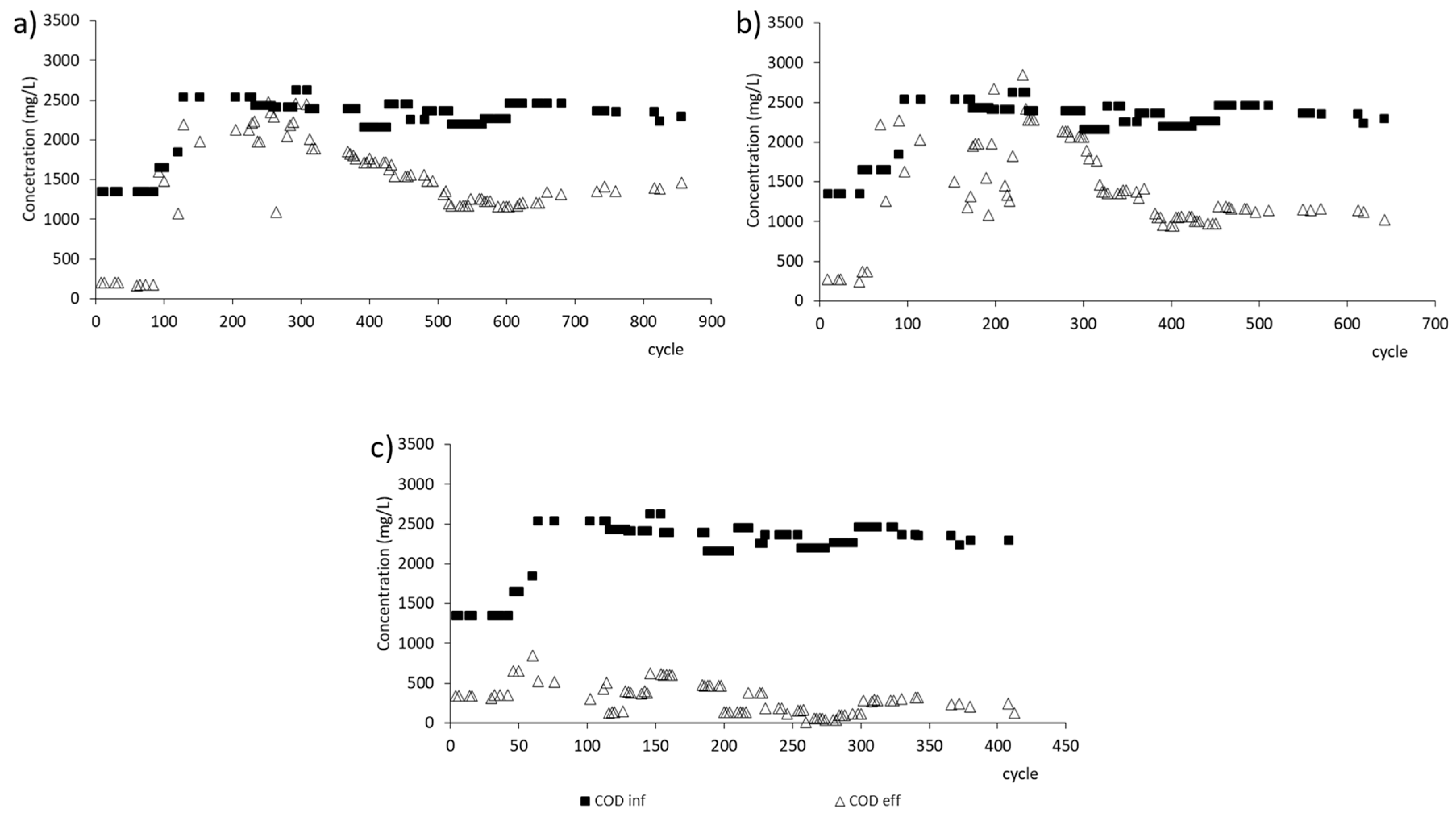
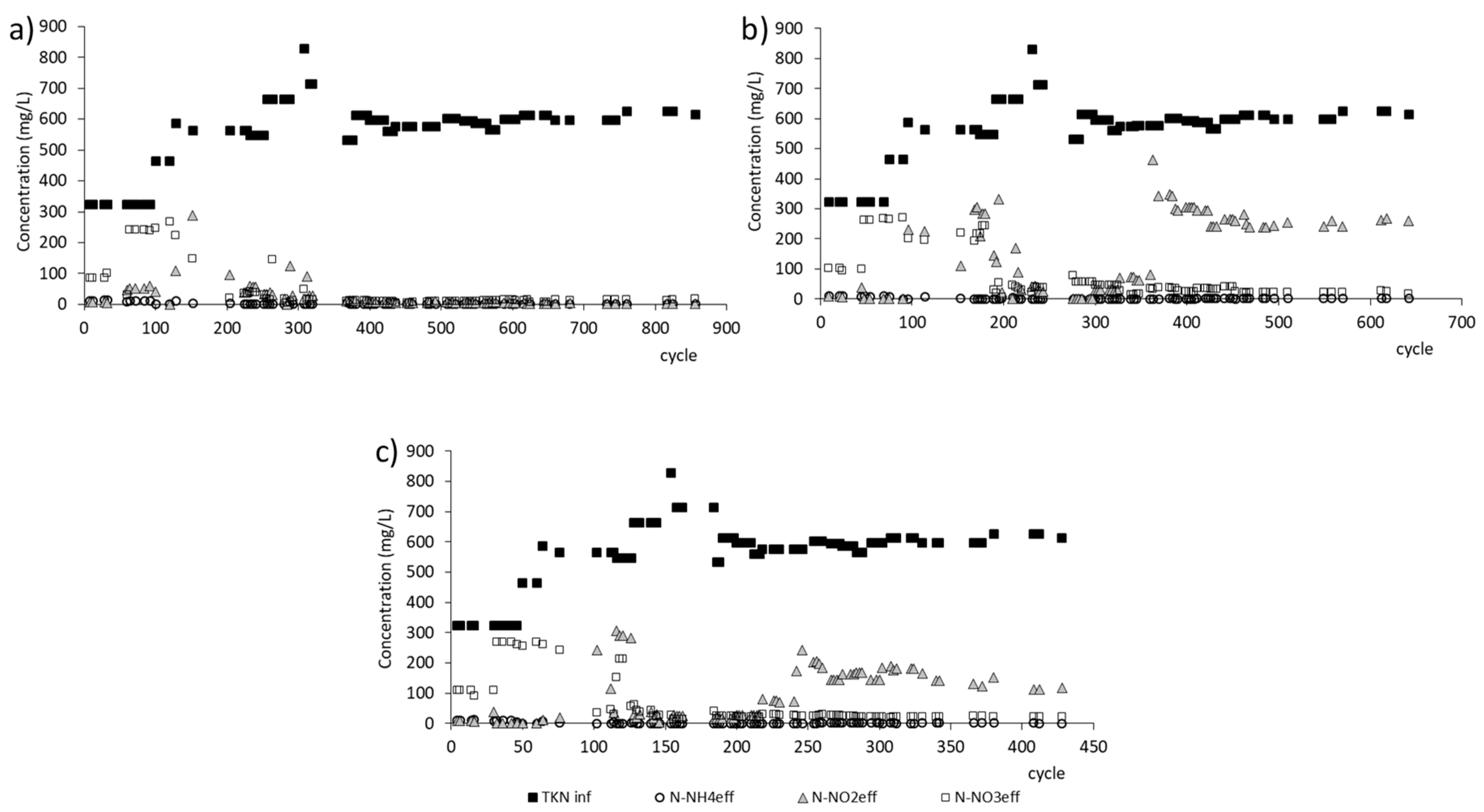
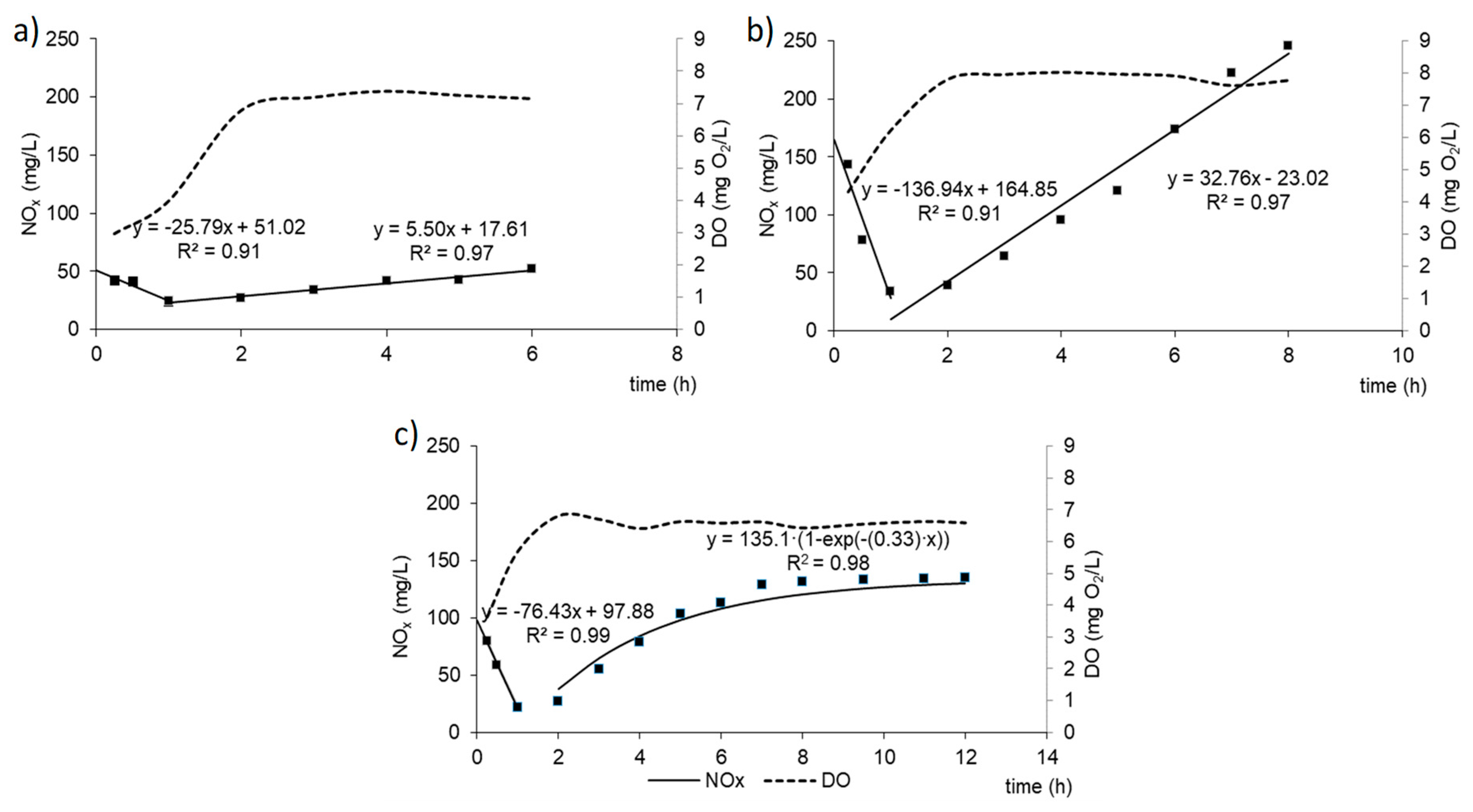
3.1. GSBR Operation
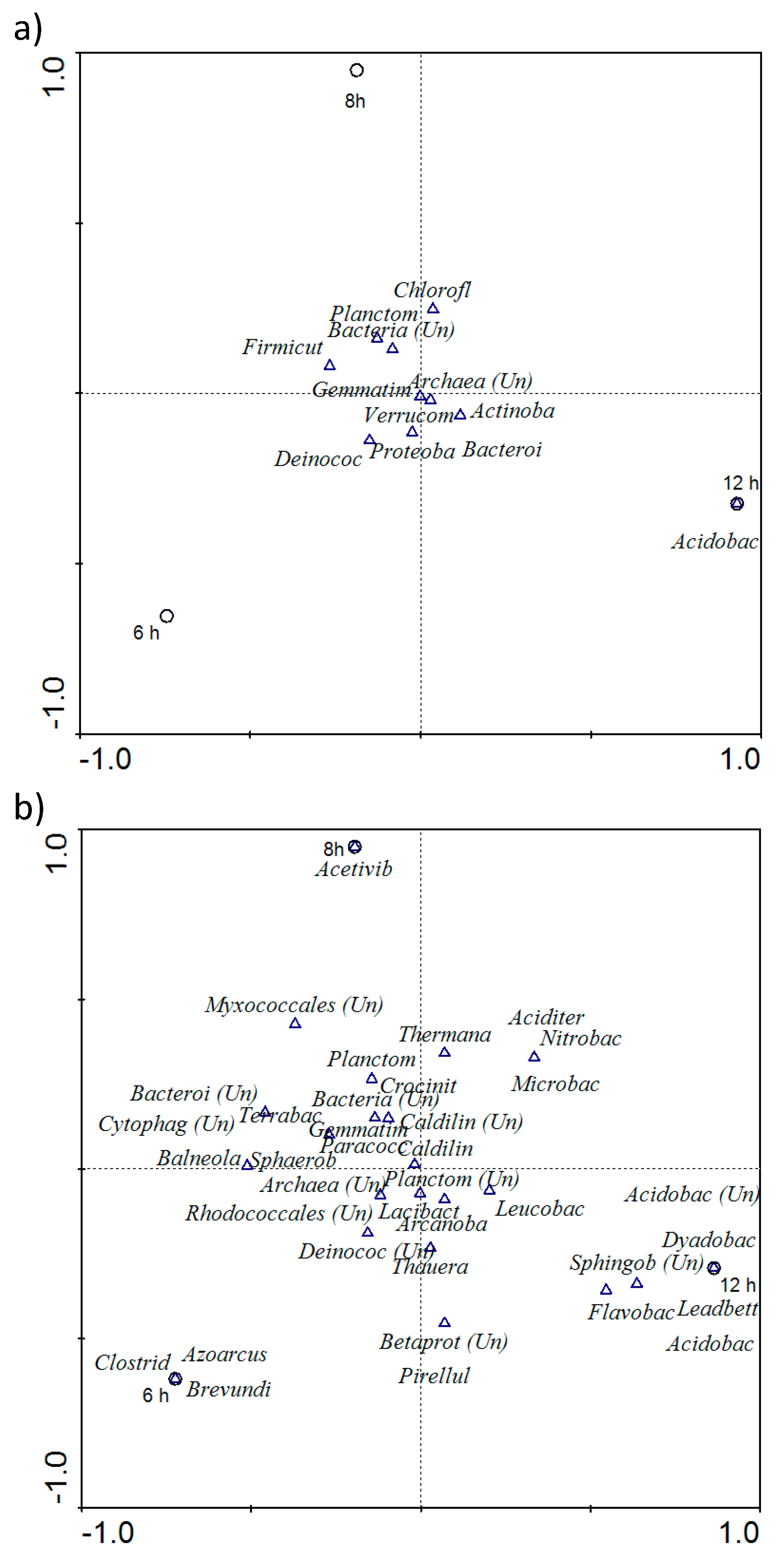
3.2. Molecular Studies
3.3. Membrane Filtration
3.4. Efficiency of Wastewater Treatment in the GSBR8h-UF System
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Biogas and Biomethane Report; European Biogas Association: Brussels, Belgium, 2015; Available online: http://european-biogas.eu/wp-content/uploads/2016/02/EBA-Annual-Report-2015-Web.pdf (accessed on 28 November 2018).
- Directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on Waste and Repealing Certain Directives. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32008L0098&from=lv (accessed on 28 November 2018).
- Pedrazzi, S.; Allesina, G.; Belló, T.; Rinaldini, C.A.; Tartarini, P. Digestate as bio-fuel in domestic furnaces. Fuel Process. Technol. 2015, 130, 172–178. [Google Scholar] [CrossRef]
- Tampio, E.; Ervasti, S.; Paavola, T.; Rintala, J. Use of laboratory anaerobic digesters to simulate the increase of treatment rate in full-scale high nitrogen content sewage sludge and co-digestion biogas plants. Bioresour. Technol. 2016, 220, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Menkveld, H.W.H.; Broeders, E. Recovery of ammonium from digestate as fertilizer. Water Pract. Technol. 2017, 12, 514–519. [Google Scholar] [CrossRef]
- Xia, A.; Murphy, J.D. Microalgal cultivation in treating liquid digestate from biogas systems. Trends Biotechnol. 2016, 34, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, L.; Peng, Y.; Zhang, Q. Effect of low COD/N ratios on stability of single-stage partial nitritation/anammox (SPN/A) process in a long-term operation. Bioresour. Technol. 2017, 244, 192–197. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, N.; Wang, L.; Zheng, K.; Fu, H.; Chen, T.; Zhang, H.; Yan, Y. Bioactivity and microbial community structure of nitrite-oxidizing bacteria in five membrane bioreactors operated as CANON process with different C/N ratio. Ecol. Eng. 2017, 99, 159–163. [Google Scholar] [CrossRef]
- Świątczak, P.; Cydzik-Kwiatkowska, A. Treatment of ammonium-rich digestate from methane fermentation using aerobic granular sludge. Water Air Soil Pollut. 2018, 229. [Google Scholar] [CrossRef]
- Cydzik-Kwiatkowska, A. Bacterial structure of aerobic granules is determined by aeration mode and nitrogen load in the reactor cycle. Bioresour. Technol. 2015, 181, 312–320. [Google Scholar] [CrossRef]
- Akhiar, A.; Battimelli, A.; Torrijos, M.; Carrere, H. Comprehensive characterization of the liquid fraction of digestates from full-scale anaerobic co-digestion. Waste Manag. 2017, 59, 118–128. [Google Scholar] [CrossRef]
- Iorhemen, O.T.; Hamza, R.A.; Zaghloul, M.S.; Tay, J.H. Simultaneous organics and nutrients removal in side-stream aerobic granular sludge membrane bioreactor (AGMBR). J. Water Process Eng. 2018, 21, 127–132. [Google Scholar] [CrossRef]
- Riley, S.M.; Ahoor, D.C.; Regnery, J.; Cath, T.Y. Tracking oil and gas wastewater-derived organic matter in a hybrid biofilter membrane treatment system: A multi-analytical approach. Sci. Total Environ. 2018, 613–614, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liang, W.; Chen, W.; Zhang, W.; Mo, J.; Liang, K.; Tang, B.; Zheng, Y.; Jiang, F. Integrated aerobic granular sludge and membrane process for enabling municipal wastewater treatment and reuse water production. Chem. Eng. J. 2018, 337, 300–311. [Google Scholar] [CrossRef]
- Sondhi, R.; Bhave, R.; Jung, G. Applications and benefits of ceramic membranes. Membr. Technol. 2003, 2003, 5–8. [Google Scholar] [CrossRef]
- APHA (American Public Health Association). Standard Methods for Examination of Water and Waste, 18th ed.; American Public Health Association: Washington, DC, USA, 1992. [Google Scholar]
- Cydzik-Kwiatkowska, A.; Białowiec, A.; Wojnowska-Baryła, I.; Smoczyński, L. Characteristic of granulated activated sludge fed with glycerin fraction from biodiesel production. Arch. Environ. Prot. 2009, 35, 41–52. [Google Scholar]
- Pellicer-Nàcher, C.; Domingo-Félez, C.; Mutlu, A.G.; Smets, B.F. Critical assessment of extracellular polymeric substances extraction methods from mixed culture biomass. Water Res. 2013, 47, 5564–5574. [Google Scholar] [CrossRef] [PubMed]
- Frølund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Lowry, O.; Rosenbrough, N.; Farr, A.; Randall, R. Protein measurement with the Folin Phenol reagent. J. Biol. Chem. 1951, 265–275. [Google Scholar]
- Gerhardt, P.; Murray, R.G.E.; Wood, W.A.; Krieg, N.R. Methods for General and Molecular Bacteriology; American Society for Microbiology: Washington, DC, USA, 1994; ISBN 978-1-55581-048-1. [Google Scholar]
- DeMaere, M.Z.; Williams, T.J.; Allen, M.A.; Brown, M.V.; Gibson, J.A.E.; Rich, J.; Lauro, F.M.; Dyall-Smith, M.; Davenport, K.W.; Woyke, T.; et al. High level of intergenera gene exchange shapes the evolution of haloarchaea in an isolated Antarctic lake. Proc. Natl. Acad. Sci. USA 2013, 110, 16939–16944. [Google Scholar] [CrossRef]
- Lane, D.J. 16S/23S rRNA sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; John Wiley & Sons: Chichester, UK, 1991; pp. 115–175. [Google Scholar]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Nawrocki, E.P.; Eddy, S.R. Infernal 1.1: 100-fold faster RNA homology searches. Bioinformatics 2013, 29, 2933–2935. [Google Scholar] [CrossRef]
- Zielińska, M.; Galik, M. Use of ceramic membranes in a membrane filtration supported by coagulation for the treatment of dairy wastewater. Water Air Soil Pollut. 2017, 228. [Google Scholar] [CrossRef] [PubMed]
- Bes-Piá, A.; Cuartas-Uribe, B.; Mendoza-Roca, J.-A.; Alcaina-Miranda, M.I. Study of the behaviour of different NF membranes for the reclamation of a secondary textile effluent in rinsing processes. J. Hazard. Mater. 2010, 178, 341–348. [Google Scholar] [CrossRef]
- Nor-Anuar, A.; Ujang, Z.; Van Loosdrecht, M.C.M.; de Kreuk, M.K.; Olsson, G. Strength characteristics of aerobic granular sludge. Water Sci. Technol. 2012, 65, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Szabó, E.; Liébana, R.; Hermansson, M.; Modin, O.; Persson, F.; Wilén, B.-M. Microbial population dynamics and ecosystem functions of anoxic/aerobic granular sludge in sequencing batch reactors operated at different organic loading rates. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Ginige, M.P.; Kayaalp, A.S.; Cheng, K.Y.; Wylie, J.; Kaksonen, A.H. Biological phosphorus and nitrogen removal in sequencing batch reactors: Effects of cycle length, dissolved oxygen concentration and infuent particulate matter. Water Sci. Technol. 2013, 68, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Fernanda Ferraz, F.; Kang, A.J.; Yuan, Q. Treatment of old landfill leachate with high ammonium content using aerobic granular sludge. J. Biol. Eng. 2017, 11, 42. [Google Scholar] [CrossRef]
- Liu, X.; Dong, C. Simultaneous COD and nitrogen removal in a micro-aerobic granular sludge reactor for domestic wastewater treatment. Syst. Eng. Procedia 2011, 1, 99–105. [Google Scholar] [CrossRef]
- Cydzik-Kwiatkowska, A.; Zielińska, M.; Bernat, K.; Wojnowska-Baryła, I.; Truchan, T. Treatment of high-ammonium anaerobic digester supernatant by aerobic granular sludge and ultrafiltration processes. Chemosphere 2013, 90, 2208–2215. [Google Scholar] [CrossRef]
- Tomar, S.K.; Chakraborty, S. Characteristics of aerobic granules treating phenol and ammonium at different cycle time and up flow liquid velocity. Int. Biodeterior. Biodegrad. 2018, 127, 113–123. [Google Scholar] [CrossRef]
- Adav, S.S.; Lee, D.-J.; Tay, J.-H. Extracellular polymeric substances and structural stability of aerobic granule. Water Res. 2008, 42, 1644–1650. [Google Scholar] [CrossRef]
- Xing, W.; Li, J.; Li, D.; Hu, J.; Deng, S.; Cui, Y.; Yao, H. Stable-isotope probing reveals the activity and function of autotrophic and heterotrophic denitrifiers in nitrate removal from organic-limited wastewater. Environ. Sci. Technol. 2018, 52, 7867–7875. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wang, C.; Li, Y.; Wang, W.; Wei, J. Treatment of phenolic wastewater by combined UF and NF/RO processes. Desalination 2015, 355, 68–74. [Google Scholar] [CrossRef]
- Stein, L.Y. Heterotrophic Nitrification and Nitrifier Denitrification. In Nitrification; Klotz, M.G., Ward, B.B., Arp, D.J., Eds.; American Society of Microbiology: Washington, DC, USA, 2011; pp. 95–114. ISBN 978-1-55581-481-6. [Google Scholar]
- Winkler, M.-K.H.; Le, Q.H.; Volcke, E.I.P. Influence of partial denitrification and mixotrophic growth of nob on microbial distribution in aerobic granular sludge. Environ. Sci. Technol. 2015, 49, 11003–11010. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.-H.; Yuan, H.-L.; Yang, J.-S.; Li, H.-F.; Chen, N. Lacibacter cauensis gen. nov., sp. nov., a novel member of the phylum Bacteroidetes isolated from sediment of a eutrophic lake. Int. J. Syst. Evol. Microbiol. 2009, 59, 1153–1157. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.D.; Jones, D.; Schofield, G.M. Reclassification of ‘Corynebacterium haemolyticum’ (MacLean, Liebow & Rosenberg) in the genus Arcanobacterium gen. nov. as Arcanobacterium haemolyticum nom. rev., comb. nov. Microbiology 1982, 128, 1279–1281. [Google Scholar]
- Grégoire, P.; Fardeau, M.-L.; Joseph, M.; Guasco, S.; Hamaide, F.; Biasutti, S.; Michotey, V.; Bonin, P.; Ollivier, B. Isolation and characterization of Thermanaerothrix daxensis gen. nov., sp. nov., a thermophilic anaerobic bacterium pertaining to the phylum “Chloroflexi”, isolated from a deep hot aquifer in the Aquitaine Basin. Syst. Appl. Microbiol. 2011, 34, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Chouari, R.; Le Paslier, D.; Daegelen, P.; Ginestet, P.; Weissenbach, J.; Sghir, A. Molecular evidence for novel planctomycete diversity in a municipal wastewater treatment plant. Appl. Environ. Microbiol. 2003, 69, 7354–7363. [Google Scholar] [CrossRef]
- Lage, O.M.; Bondoso, J.; Viana, F. Isolation and characterization of Planctomycetes from the sediments of a fish farm wastewater treatment tank. Arch. Microbiol. 2012, 194, 879–885. [Google Scholar] [CrossRef]
- Kim, S.M.; Park, S.W.; Park, S.T.; Kim, Y.M. Terrabacter carboxydivorans sp. nov., a carbon monoxide-oxidizing actinomycete. Int. J. Syst. Evol. Microbiol. 2011, 61, 482–486. [Google Scholar] [CrossRef]
- Wan, C.; Sun, S.; Lee, D.-J.; Liu, X.; Wang, L.; Yang, X.; Pan, X. Partial nitrification using aerobic granules in continuous-flow reactor: Rapid startup. Bioresour. Technol. 2013, 142, 517–522. [Google Scholar] [CrossRef]
- Xie, B.; Gu, J.; Lu, J. Surface properties of bacteria from activated sludge in relation to bioflocculation. J. Environ. Sci. 2010, 22, 1840–1845. [Google Scholar] [CrossRef]
- Kragelund, C.; Thomsen, T.R.; Mielczarek, A.T.; Nielsen, P.H. Eikelboom’s morphotype 0803 in activated sludge belongs to the genus Caldilinea in the phylum Chloroflexi: Eikelboom’s morphotype 0803 belongs to phylum Chloroflexi. FEMS Microbiol. Ecol. 2011, 76, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Sun, H.; Chen, Z.; Wang, Y.-H.; Huo, Z.-Y.; Ikuno, N.; Ishii, K.; Jin, Y.; Hu, H.-Y.; Wu, Y.-H.; et al. Different bacterial species and their extracellular polymeric substances (EPSs) significantly affected reverse osmosis (RO) membrane fouling potentials in wastewater reclamation. Sci. Total Environ. 2018, 644, 486–493. [Google Scholar] [CrossRef] [PubMed]
- Lesjean, B.; Rosenberger, S.; Laabs, C.; Jekel, M.; Gnirss, R.; Amy, G. Correlation between membrane fouling and soluble/colloidal organic substances in membrane bioreactors for municipal wastewater treatment. Water Sci. Technol. J. Int. Assoc. Water Pollut. Res. 2005, 51, 1–8. [Google Scholar] [CrossRef]
- Wang, F.; Lu, S.; Wei, Y.; Ji, M. Characteristics of aerobic granule and nitrogen and phosphorus removal in a SBR. J. Hazard. Mater. 2009, 164, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Aouni, A.; Fersi, C.; Cuartas-Uribe, B.; Bes-Pia, A.; Alcaina-Miranda, M.I.; Dhahbi, M. Study of membrane fouling using synthetic model solutions in UF and NF processes. Chem. Eng. J. 2011, 175, 192–200. [Google Scholar] [CrossRef]
- Lim, A. Membrane fouling and cleaning in microfiltration of activated sludge wastewater. J. Membr. Sci. 2003, 216, 279–290. [Google Scholar] [CrossRef]
- Muthukumaran, S.; Nguyen, D.A.; Baskaran, K. Performance evaluation of different ultrafiltration membranes for the reclamation and reuse of secondary effluent. Desalination 2011, 279, 383–389. [Google Scholar] [CrossRef]
- Cucu, A.; Tiliakos, A.; Tanase, I.; Serban, C.E.; Stamatin, I.; Ciocanea, A.; Nichita, C. Microbial fuel cell for nitrate reduction. Energy Procedia 2016, 85, 156–161. [Google Scholar] [CrossRef]
- Guldhe, A.; Ansari, F.A.; Singh, P.; Bux, F. Heterotrophic cultivation of microalgae using aquaculture wastewater: A biorefinery concept for biomass production and nutrient remediation. Ecol. Eng. 2017, 99, 47–53. [Google Scholar] [CrossRef]
- An, J.-Y.; Sim, S.-J.; Lee, J.S.; Kim, B.W. Hydrocarbon production from secondarily treated piggery wastewater by the green alga Botryococcus braunii. J. Appl. Phycol. 2003, 15, 185–191. [Google Scholar] [CrossRef]
- Bick, A.; Gillerman, L.; Manor, Y.; Oron, G. Economic assessment of an integrated membrane system for secondary effluent polishing for unrestricted reuse. Water 2012, 4, 219–236. [Google Scholar] [CrossRef]
- Chew, C.M.; Aroua, M.K.; Hussain, M.A.; Ismail, W.M.Z.W. Evaluation of ultrafiltration and conventional water treatment systems for sustainable development: An industrial scale case study. J. Clean. Prod. 2016, 112, 3152–3163. [Google Scholar] [CrossRef]
- Oron, G.; Gillerman, L.; Bick, A.; Manor, Y.; Buriakovsky, N.; Hagin, J. Membrane technology for sustainable treated wastewater reuse: Agricultural; environmental and hydrological considerations. Water Sci. Technol. 2008, 57, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
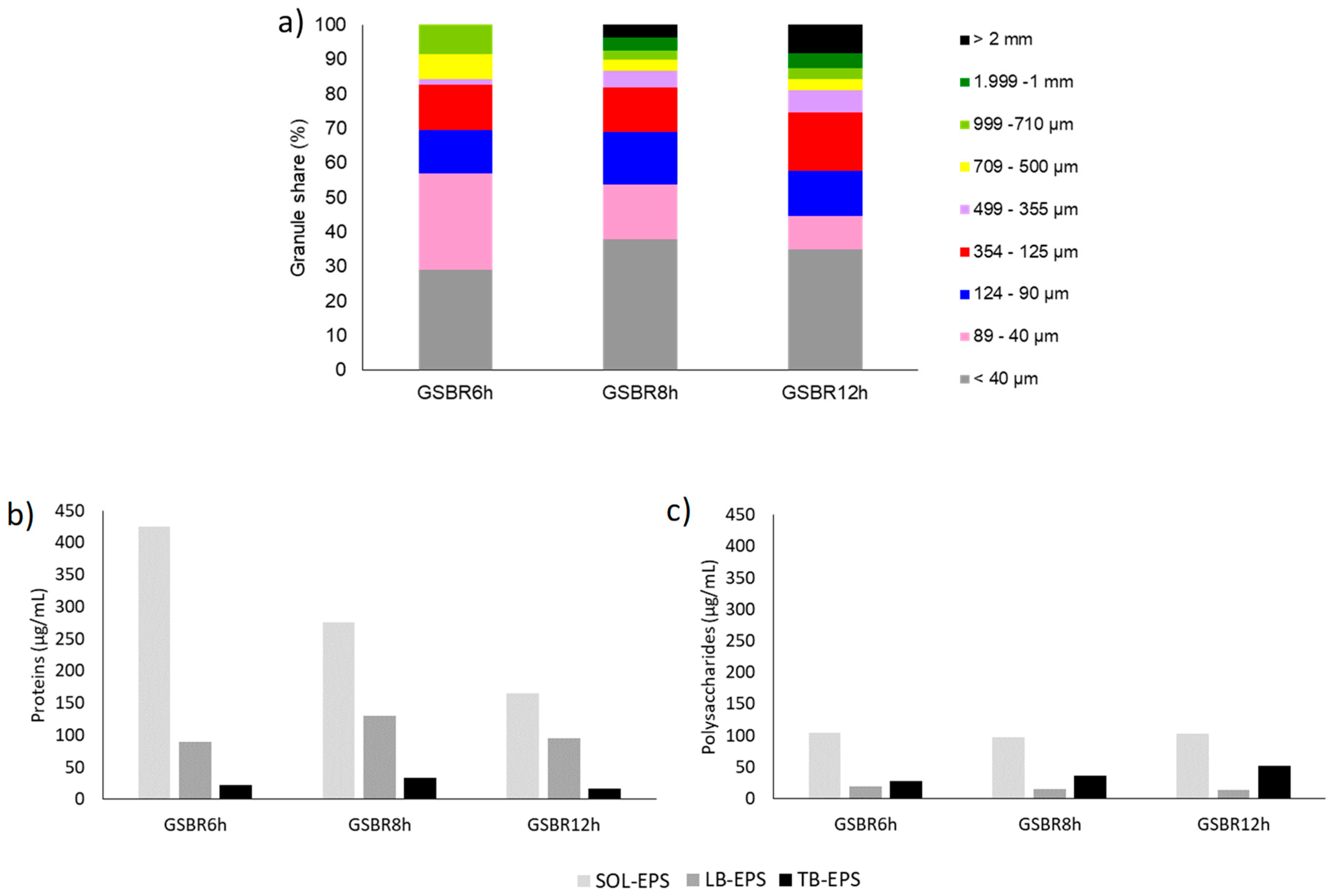

| Parameter | Unit | GSBR6h | GSBR8h | GSBR12h |
|---|---|---|---|---|
| Efficiency of ammonium removal | % | 97.2 ± 10.7 | 97.5 ± 9.5 | 96.7 ± 11.4 |
| Efficiency of nitrification | % | 94.4 ± 0.5 | 88.6 ± 14.8 | 97.5 ± 0.5 |
| Efficiency of nitrogen removal | % | 58.4 ± 7.6 | 46.1 ± 10.9 | 60.0 ± 3.1 |
| Nitrogen removed per day | g/d | 1.9 | 0.8 | 0.2 |
| Efficiency of organics removal | % | 46.2 ± 8.7 | 51.8 ± 5.2 | 88.8 ± 5.9 |
| Efficiency of phosphorus removal | % | 17.1 ± 5.4 | 27.0 ± 9.6 | 21.7 ± 8.1 |
| Ammonium removal rate | mg/(L·h) | 107.7 | 72.5 | 49.3 |
| Organics removal rate | mg/(L·h) | 923.4 | 725.9 | 193.5 |
| Phosphorus removal rate | mg/(L·h) | 1.3 | 1.2 | 1.1 |
| The rate constant for ammonium removal | mg/(L·h) | 0.40 | 0.42 | 0.43 |
| The rate constant for organics removal | 1/h | 0.82 | 0.60 | 0.28 |
| The rate constant for phosphorus removal | 1/h | 0.24 | 0.26 | 0.20 |
| Pressure (MPa) | Indicator | Concentration in Permeate | Efficiency of Membrane Filtration (%) | Efficiency of System GSBR-UF (%) |
|---|---|---|---|---|
| 0.3 | Organics (mg/L) | 267.5 ± 132.2 | 76.0 | 88.5 |
| 0.4 | 286.5 ± 4.94 | 74.4 | 87.6 | |
| 0.5 | 285.5 ± 7.8 | 74.5 | 87.7 | |
| 0.3 | Total Nitrogen (mg/L) | 3.9 ± 0.8 | 86.9 | 99.3 |
| 0.4 | 7.6 ± 1.1 | 73.7 | 98.7 | |
| 0.5 | 4.6 ± 0.6 | 84.3 | 99.2 | |
| 0.3 | Total Phosphorus (mg/L) | 9.1 ± 1.5 | 54.6 | 70.3 |
| 0.4 | 11.9 ± 2.1 | 44.7 | 61.1 | |
| 0.5 | 12.9 ± 2.6 | 40.1 | 57.8 | |
| 0.3 | NOx (mg/L) | 259.0 ± 12.6 | 11.1 | - |
| 0.4 | 253.1 ± 21.0 | 13.0 | - | |
| 0.5 | 256.1 ± 18.3 | 12.0 | - | |
| 0.3 | Color * | 0.10 ± 0.00 | 78.3 | 95.5 |
| 0.4 | 0.15 ± 0.00 | 50.0 | 93.3 | |
| 0.5 | 0.05 ± 0.00 | 81.5 | 97.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Świątczak, P.; Cydzik-Kwiatkowska, A.; Zielińska, M. Treatment of Liquid Phase of Digestate from Agricultural Biogas Plant in a System with Aerobic Granules and Ultrafiltration. Water 2019, 11, 104. https://doi.org/10.3390/w11010104
Świątczak P, Cydzik-Kwiatkowska A, Zielińska M. Treatment of Liquid Phase of Digestate from Agricultural Biogas Plant in a System with Aerobic Granules and Ultrafiltration. Water. 2019; 11(1):104. https://doi.org/10.3390/w11010104
Chicago/Turabian StyleŚwiątczak, Piotr, Agnieszka Cydzik-Kwiatkowska, and Magdalena Zielińska. 2019. "Treatment of Liquid Phase of Digestate from Agricultural Biogas Plant in a System with Aerobic Granules and Ultrafiltration" Water 11, no. 1: 104. https://doi.org/10.3390/w11010104
APA StyleŚwiątczak, P., Cydzik-Kwiatkowska, A., & Zielińska, M. (2019). Treatment of Liquid Phase of Digestate from Agricultural Biogas Plant in a System with Aerobic Granules and Ultrafiltration. Water, 11(1), 104. https://doi.org/10.3390/w11010104