Quantification of Daily Water Requirements of Container-Grown Calathea and Stromanthe Produced in a Shaded Greenhouse
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Location
2.2. Experimental Setup
2.3. Plant Materials and Their Growth
2.4. Data Collection
2.5. Modelling Plant Water Use
3. Results
3.1. Reference Evapotranspiration
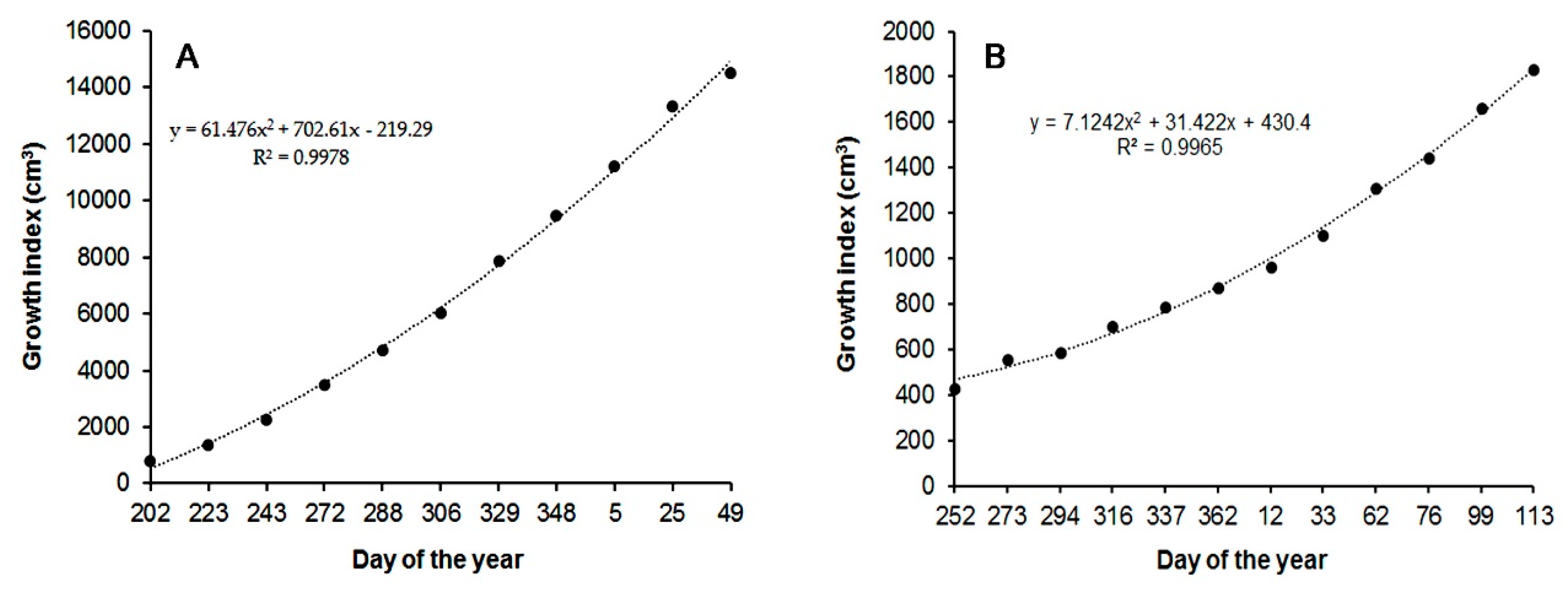
3.2. Plant Growth
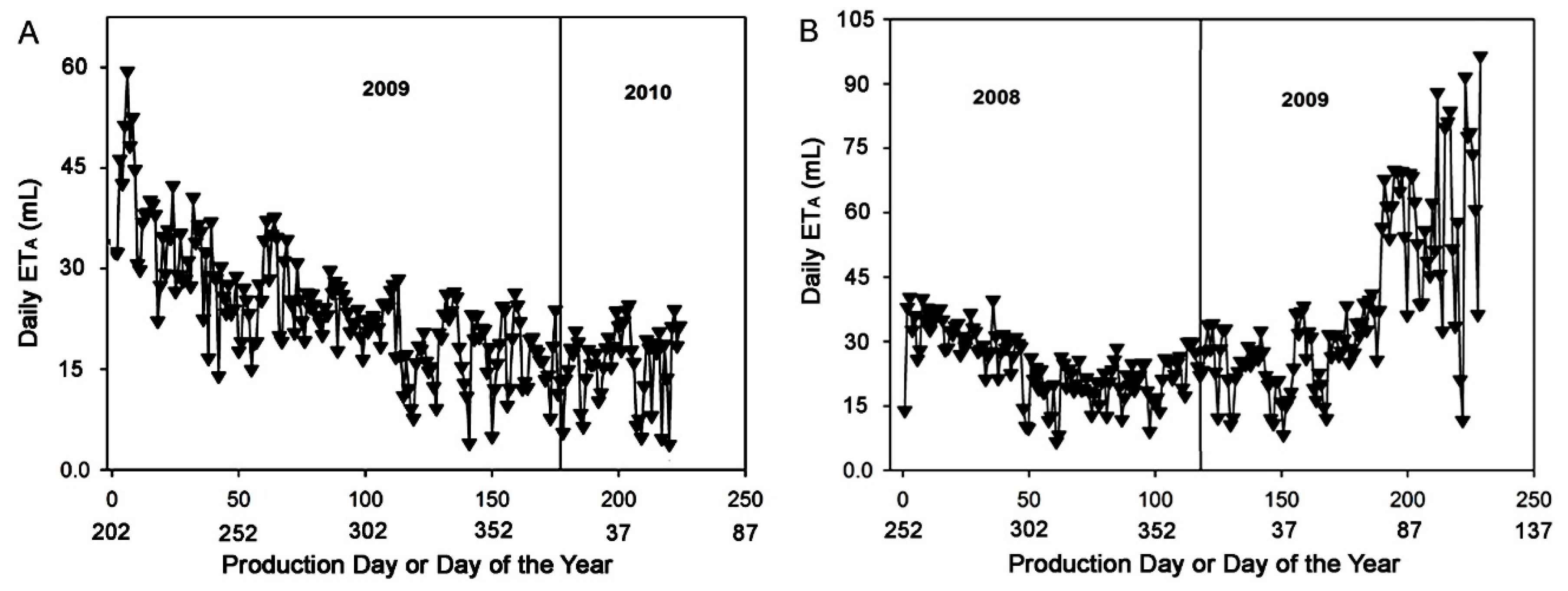
3.3. Plant Actual Evapotranspiration (ETA)
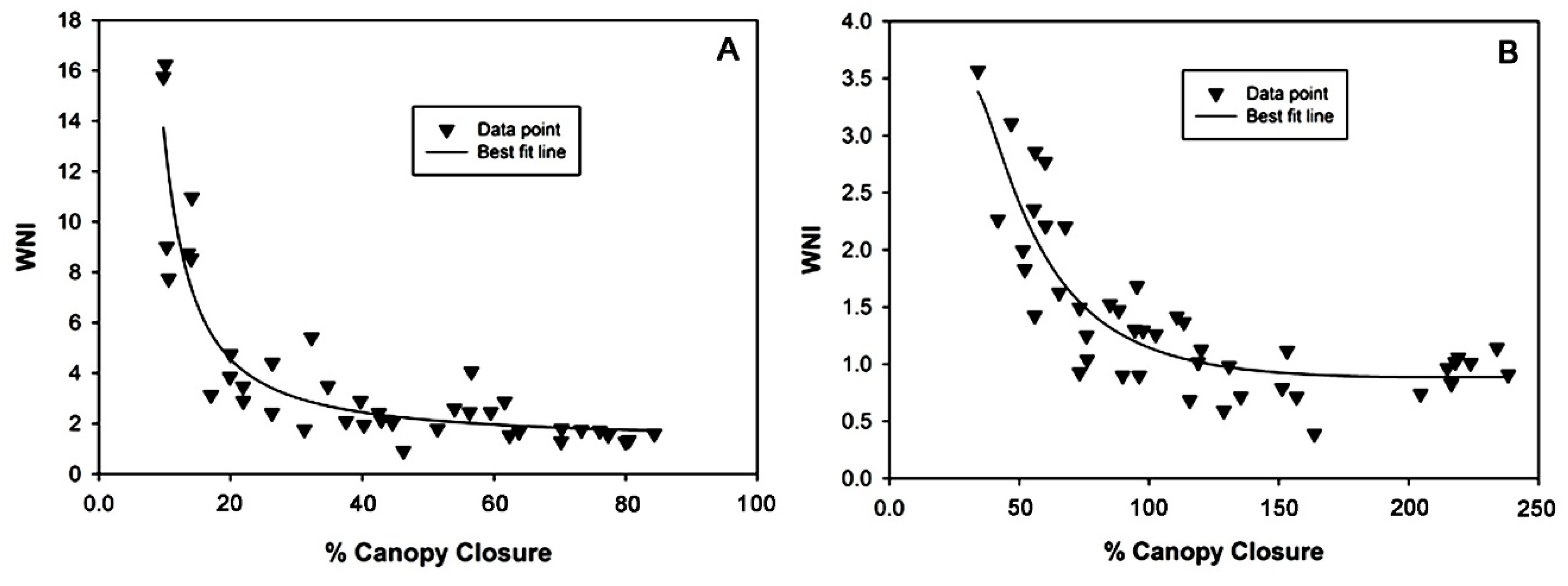
3.4. Data Analysis and Modeling
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Doll, P.; Fiedler, K.; Zhang, J. Global-scale analysis of river flow alterations due to water withdrawals and reservoirs. Hydrol. Earth Syst. Sci. 2009, 13, 2413–2432. [Google Scholar] [CrossRef]
- Food and Agricultural Organization of the United Nations. AQUASTAT—FAO’s Global Information System on Water and Agriculture; FAO: Rome, Italy, 2010. [Google Scholar]
- Kenny, J.F.; Barber, N.L.; Hutson, S.S.; Linsey, K.S.; Lovelace, J.K.; Maupin, M.A. Estimated Water Use in the United States in 2005; U.S. Geological Survey Circular: Reston, VA, USA, 2009.
- Chen, J.; Wei, X. Controlled-released fertilizers as a means to reduce nitrogen leaching and runoff in container-grown plant production. In Nitrogen in Agriculture-Updates; Khan, A., Fahad, S., Eds.; InTech Open: Rijeka, Croatia, 2018. [Google Scholar]
- Parrella, M.P.; Wagner, A.; Fujino, D.W. The floriculture and nursery industry’s struggle with invasive species. Am. Entomol. 2015, 61, 39–50. [Google Scholar] [CrossRef]
- CENSUS of Agriculture, United States Summary and State Data. 2007. Available online: http://www.agcensus.usda.gov/Publications/2007/Full_Report/usv1.pdf (accessed on 28 July 2018).
- Belayneh, B.E.; Lea-Cox, J.D.; Lichtenberg, E. Costs and benefits of implementing sensor-controlled irrigation in a commercial pot-in-pot container nursery. HortTechnology 2013, 23, 760–769. [Google Scholar]
- Chen, J.; Huang, Y.; Caldwell, R.D. Best management practices for minimizing nitrate leaching from container-grown nurseries. Sci. World J. 2001, 1, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Beeson, R.C., Jr.; Yeager, T.H.; Stamps, R.H.; Felter, L.A. Evaluation of captured rainwater and irrigation runoff for greenhouse foliage and bedding plant production. HortScience 2003, 38, 228–233. [Google Scholar]
- Beeson, R.C., Jr. Suspension lysimeter systems for quantifying water use and modulating water stress for crops grown in organic substrates. Agric. Water Manag. 2011, 98, 967–976. [Google Scholar] [CrossRef]
- Majsztrik, J.C.; Ristvey, A.G.; Lea-Cox, J.D. Water and nutrient management in the production of container-grown ornamentals. Hortic. Rev. 2011, 38, 253–297. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration: Guidelines for Computing Crop Water Requirements—FAO Irrigation and Drainage Paper 56; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998. [Google Scholar]
- Howell, T.A.; Evett, S.R. The Penman-Monteith Method. 2006. Available online: http://www.cprl.ars.usda.gov/wmru/pdfs/PM%20COLO%20Bar%202004%20cor-rected%209apr04.pdf (accessed on 4 September 2018).
- Campbell Scientific. Application Note 4-D; Campbell Scientific Ltd.: Logan, UT, USA, 1991. [Google Scholar]
- Jensen, M.E.; Burman, R.D.; Allen, R.G. Evapotranspiration and Irrigation Water Requirements. 1990. Available online: http://agris.fao.org/agris-search/search.do?recordID=US19910106366 (accessed on 28 July 2018).
- Allen, R.G.; Wright, J.L.; Pruitt, W.O.; Pereira, L.S. Water requirements. In Design and Operation of Farm Irrigation Systems; ASAE: St. Joseph, MN, USA, 2007. [Google Scholar]
- Bryla, D.R.; Trout, T.J.; Ayars, J.E. Weighing lysimeters for developing crop coefficients and efficient irrigation practices for vegetable crops. HortScience 2010, 45, 1597–1604. [Google Scholar]
- Thornthwaite, C.W. An approach toward a rational classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- Fitzpatrick, G. Water budget determinations for container-grown ornamental plants. Proc. Fla. State Hortic. Soc. 1980, 93, 166–168. [Google Scholar]
- Fitzpatrick, G. Relative water demand in container-grown ornamental plants. HortScience 1983, 18, 760–762. [Google Scholar]
- Burger, D.W.; Hartin, J.S.; Hodel, D.R.; Lukaszewski, T.A.; Tjosvoid, S.A.; Wagner, S.A. Water use in California’s ornamental nurseries. Calif. Agric. 1987, 41, 7–8. [Google Scholar]
- Doorenbos, J.; Pruitt, W.O. Guidelines for Predicting Crop Water Requirements—FAO Irrigation and Drainage Paper 24; Food and Agriculture Organization of the United Nations: Rome, Italy, 1977. [Google Scholar]
- Beeson, R.C., Jr. Penman crop Coefficients for container growth landscape ornamentals. In Evapotranspiration and Irrigation Scheduling; Camp, C.R., Sadler, E.J., Yoder, R.E., Eds.; American Society of Agricultural Engineers: San Antonio, TX, USA, 1996. [Google Scholar]
- Beeson, R.C., Jr. Modeling actual evapotranspiration of Ligustrum japonicum from rooted cuttings to commercially marketable plants in 12 liter black polyethylene containers. Acta Hortic. 2004, 664, 71–77. [Google Scholar] [CrossRef]
- Beeson, R.C., Jr. Modeling irrigation requirements for landscape ornamentals. HortTechnology 2005, 15, 18–22. [Google Scholar]
- Beeson, R.C., Jr. Response of evapotranspiration of Viburnum odoratissimum to canopy closure and the implications for water conservation during production and in landscapes. HortScience 2010, 45, 359–364. [Google Scholar]
- Beeson, R.C., Jr. Modeling actual evapotranspiration of Viburnum odoratissimum during production from rooted cuttings to market size plants in 11.4-L containers. HortScience 2010, 45, 1260–1264. [Google Scholar]
- Beeson, R.C., Jr. Development of a simple reference evapotranspiration model for irrigation of woody ornamentals. HortScience 2012, 47, 264–268. [Google Scholar]
- Chen, J.; Beeson, R.C., Jr. Actual evapotranspiration of Asplenium nidus and Chamaedorea elegans during production from liners to marketable plants. Acta Hortic. 2013, 990, 339–344. [Google Scholar] [CrossRef]
- Hagen, E.; Mambuthiri, S.; Fulcher, A.; Geneve, R. Comparing substrate moisture-based daily water use and on-demand irrigation regimes for oakleaf hydrangea grown in two container sizes. Sci. Hortic. 2014, 179, 132–139. [Google Scholar] [CrossRef]
- Pershey, N.A.; Cregg, B.N.; Andresen, J.A.; Fernandez, R.T. Irrigating based on daily water use reduces nursery runoff volume and nutrient load without reducing growth of four conifers. HortScience 2015, 50, 1553–1561. [Google Scholar]
- Chen, J.; McConnell, D.B.; Norman, D.L.; Henny, R.J. The foliage plant industry. Hortic. Rev. 2004, 31, 47–112. [Google Scholar]
- USDA, USDA National Agricultural Statistics Service. Floriculture Crops 2015 Summary; USDA: Washington, DC, USA, 2016.
- Beeson, R.C., Jr. Relationship of plant growth and actual evapotranspiration to irrigation frequency based on managed allowable deficits for container nursery stock. J. Am. Soc. Hortic. Sci. 2006, 131, 140–148. [Google Scholar]
- Henny, R.J.; Holm, J.R.; Chen, J.; Scheiber, M. In vitro induction of tetraploids in Dieffenbachia x ‘Star Bright M-1’ by colchicine. HortScience 2009, 44, 646–650. [Google Scholar]
- Stanhill, G. Water use efficiency. Adv. Agron. 1987, 39, 53–85. [Google Scholar]
- Poole, R.T.; Conover, C.A. Fertilizers levels and medium affect foliage plant growth in an ebb and flow irrigation system. J. Environ. Hortic. 1992, 10, 81–86. [Google Scholar]
- Kim, J.; van Iersel, M.W.; Burnett, S.E. Estimating daily water use of two Petunia cultivars based on plant and environmental factors. HortScience 2011, 46, 1287–1293. [Google Scholar]
- Henny, R.J.; Chen, J. Cultivar development of ornamental foliage plants. Plant Breed. Rev. 2003, 23, 245–290. [Google Scholar]
- Wang, Q.; Chen, J. Variation in photosynthetic characteristics and leaf area contributes to Spathiphyllum cultivar differences in biomass production. Photosynthetica 2003, 41, 443–447. [Google Scholar] [CrossRef]
- Neal, C.A.; Henley, R.W. Water use and runoff comparisons of greenhouse irrigation systems. Proc. Fla. State Hortic. Soc. 1992, 105, 191–194. [Google Scholar]
| Plant | Mean Leaf No | Leaf Area (cm2) | Shoot Fresh Weight (g) | Root Fresh Weight (g) | Shoot Dry Weight (g) | Root Dry Weight (g) | Water Use Efficiency (g L−1) y |
|---|---|---|---|---|---|---|---|
| Calathea | 19.4 ± 0.59 | 852.0 ± 54.60 | 33.8 ± 2.40 | 5.6 ± 0.37 | 3.7 ± 0.33 | 0.5 ± 0.04 | 0.87 |
| Stromanthe | 84.0 ± 2.49 | 2729.2 ± 68.36 | 110.7 ± 3.10 | 42.2 ± 1.35 | 14.4 ± 0.17 | 3.0 ± 0.08 | 2.56 |
| Species | Model Equation | r2 |
|---|---|---|
| Calathea | WNI = 1.213 + 0.383/%CC + 0.032/(%CC)2 + 0.005/(%CC)3 | 0.82 |
| Stromanthe | WNI = 1.136 − 1.131/%CC + 1.395/(%CC)2 − 0.255/(%CC)3 | 0.73 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beeson, R.C., Jr.; Chen, J. Quantification of Daily Water Requirements of Container-Grown Calathea and Stromanthe Produced in a Shaded Greenhouse. Water 2018, 10, 1194. https://doi.org/10.3390/w10091194
Beeson RC Jr., Chen J. Quantification of Daily Water Requirements of Container-Grown Calathea and Stromanthe Produced in a Shaded Greenhouse. Water. 2018; 10(9):1194. https://doi.org/10.3390/w10091194
Chicago/Turabian StyleBeeson, Richard C., Jr., and Jianjun Chen. 2018. "Quantification of Daily Water Requirements of Container-Grown Calathea and Stromanthe Produced in a Shaded Greenhouse" Water 10, no. 9: 1194. https://doi.org/10.3390/w10091194
APA StyleBeeson, R. C., Jr., & Chen, J. (2018). Quantification of Daily Water Requirements of Container-Grown Calathea and Stromanthe Produced in a Shaded Greenhouse. Water, 10(9), 1194. https://doi.org/10.3390/w10091194





