Effect of TSS Removal from Stormwater by Mixed Media Column on T-N, T-P, and Organic Material Removal
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of Media
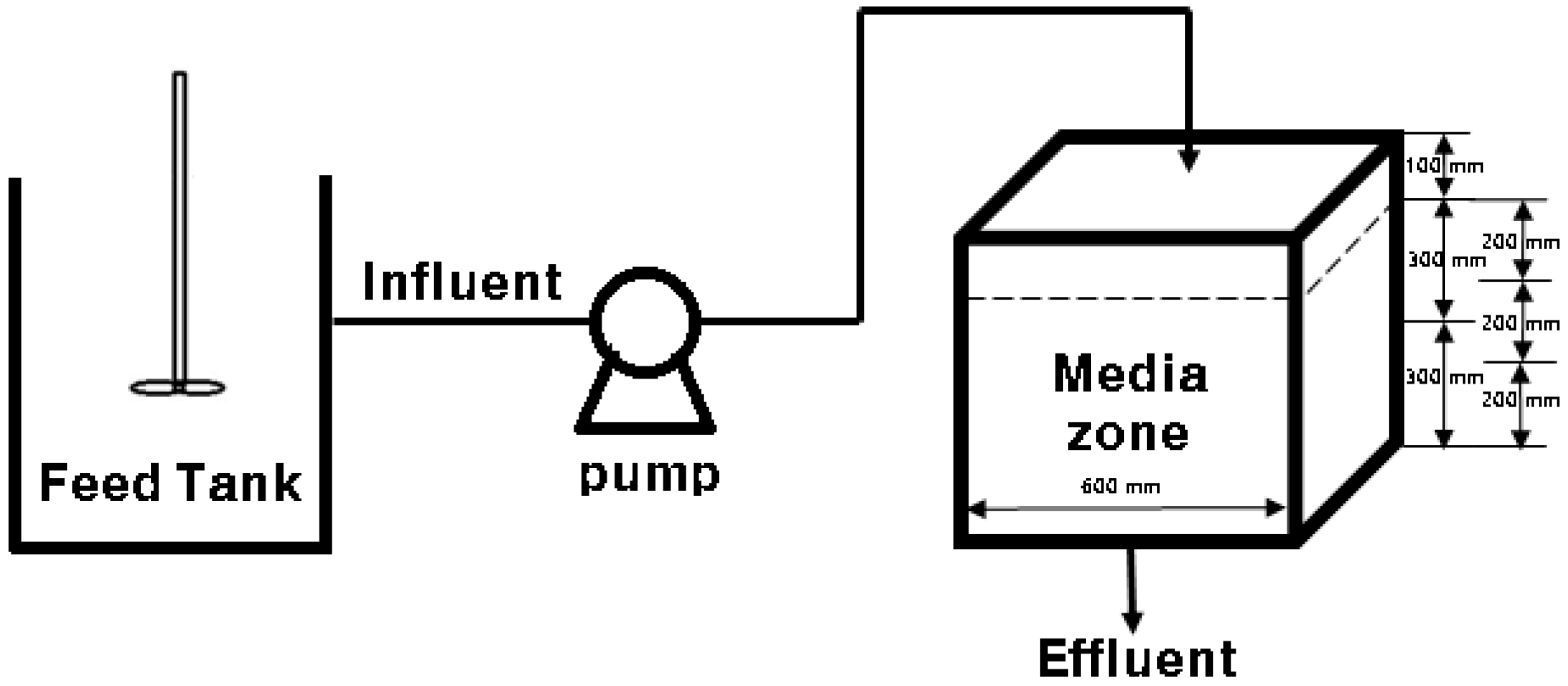
2.2. Lab-Scale Experiments
2.3. Operation Conditions
2.4. Contaminant Analysis
3. Results and Discussion
3.1. Removal Efficiency of Each Contaminant
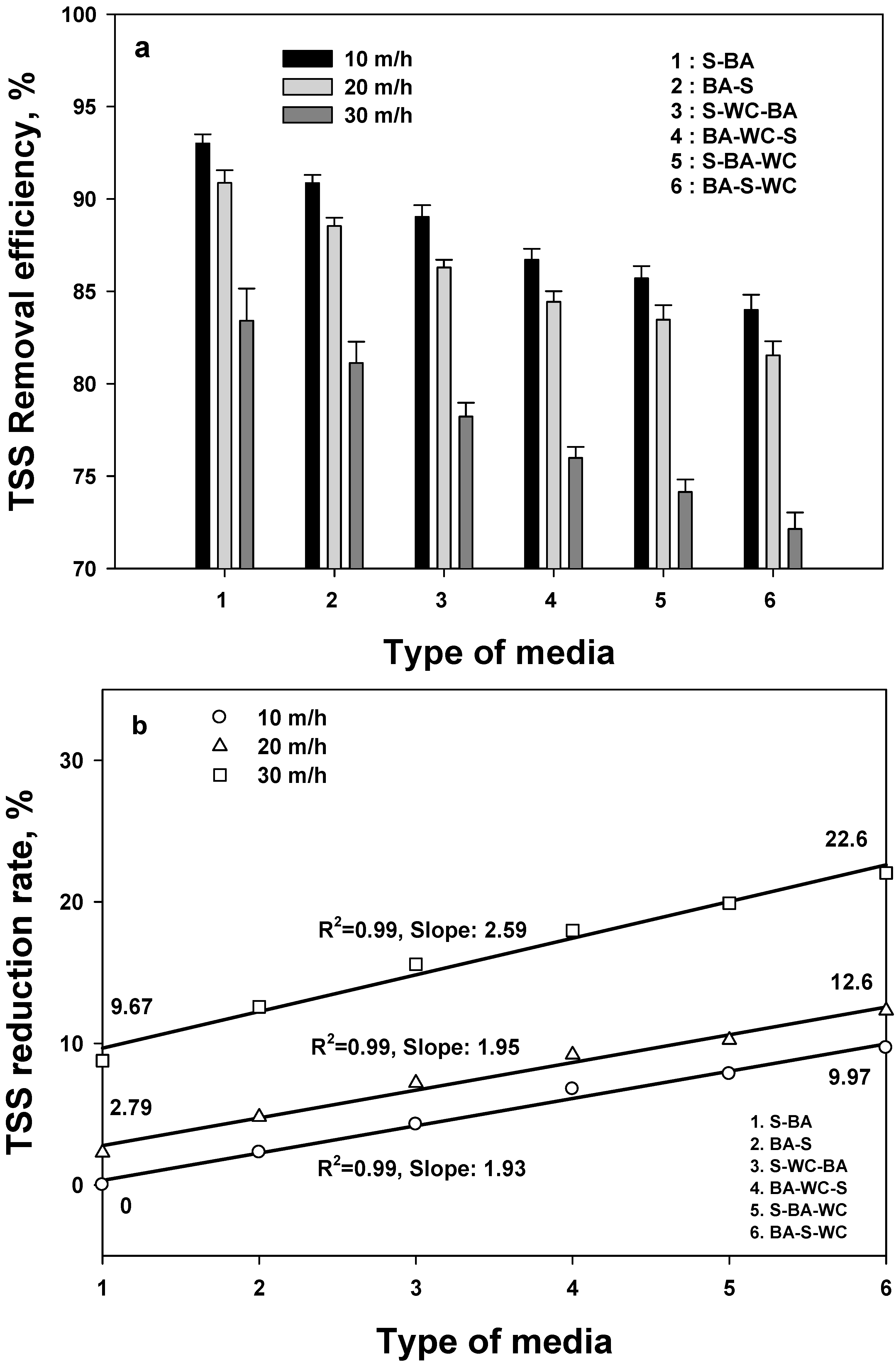
3.2. Removal of TSS
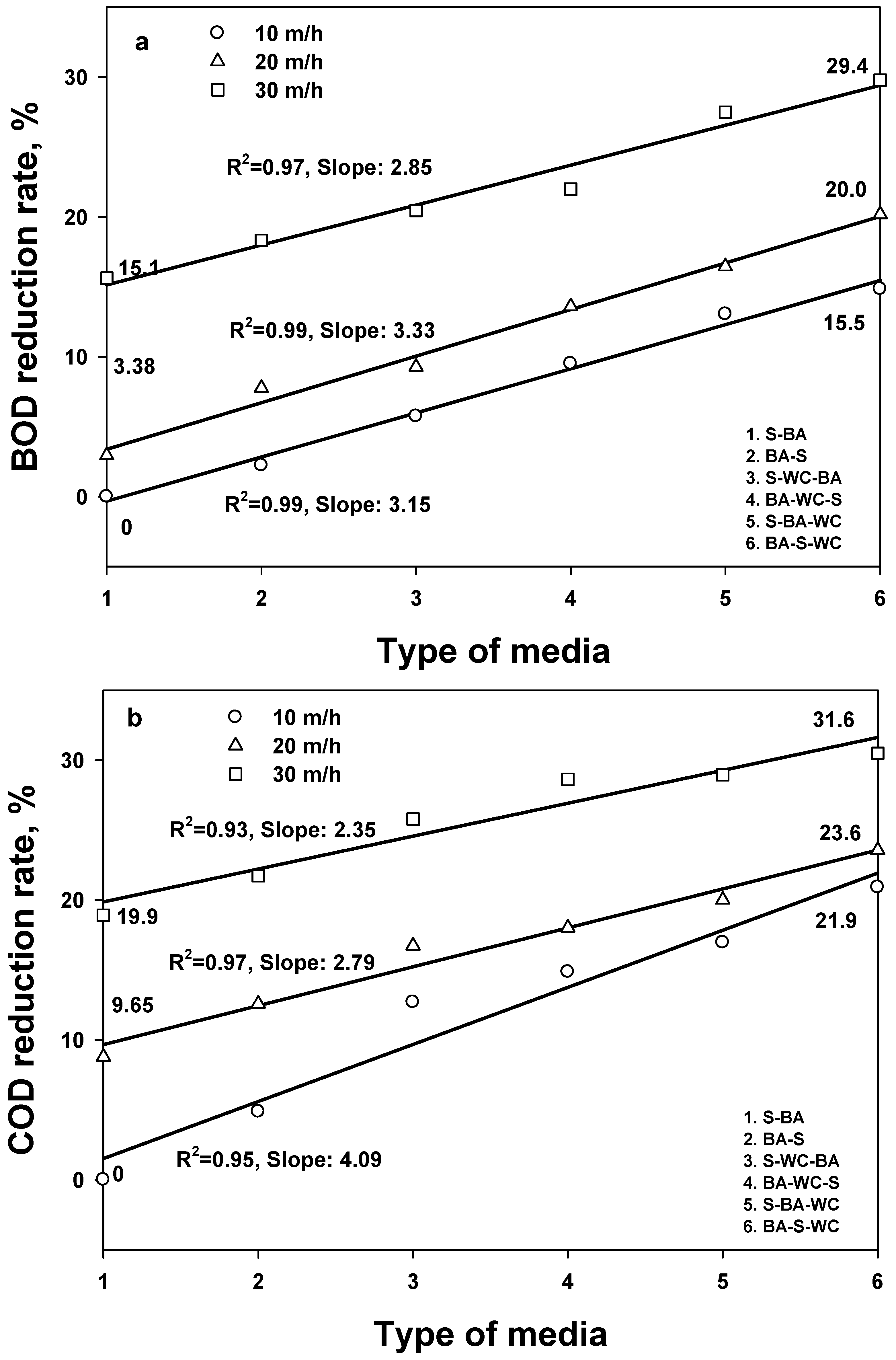
3.3. Removal of BOD and COD
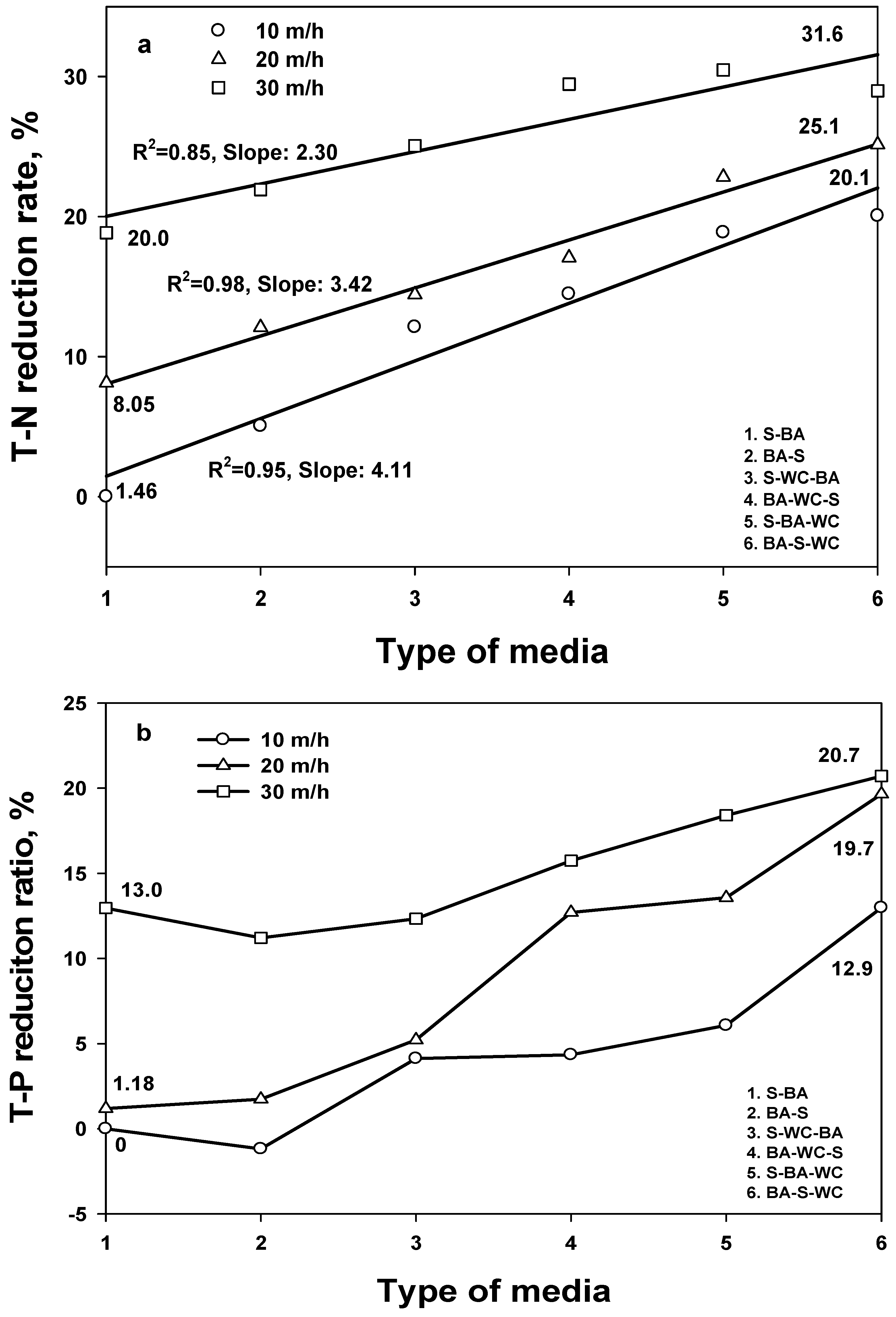
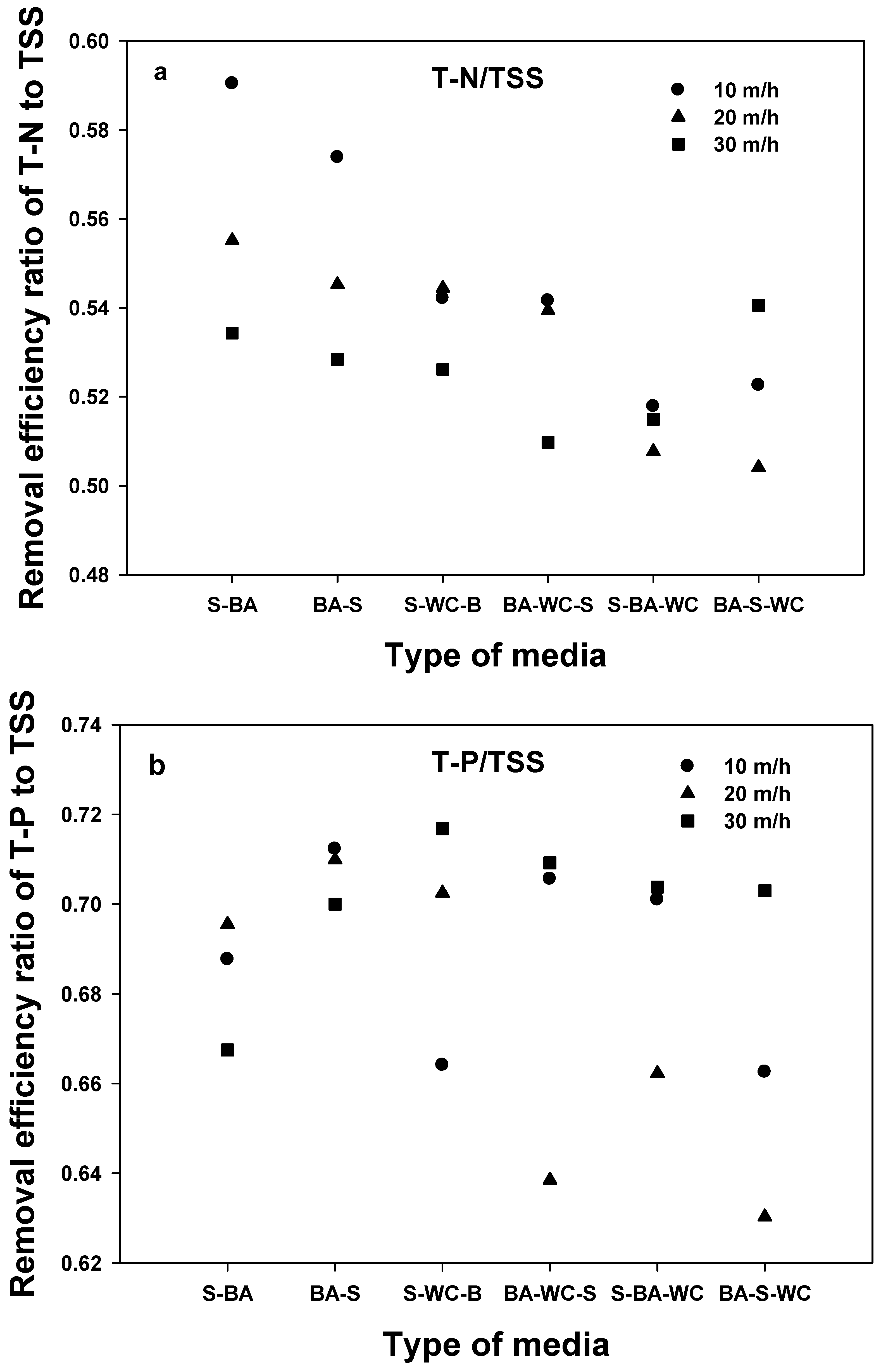
3.4. SS-T-N and SS-T-P
4. Conclusions
- Based on the TSS removal efficiency, all mixed filtration media obtained an 80% removal efficiency at 10 and 20 HC, which satisfies the Korean regulations for stormwater treatment. The two mixed media obtained at least a 15% higher removal efficiency than the three mixed media.
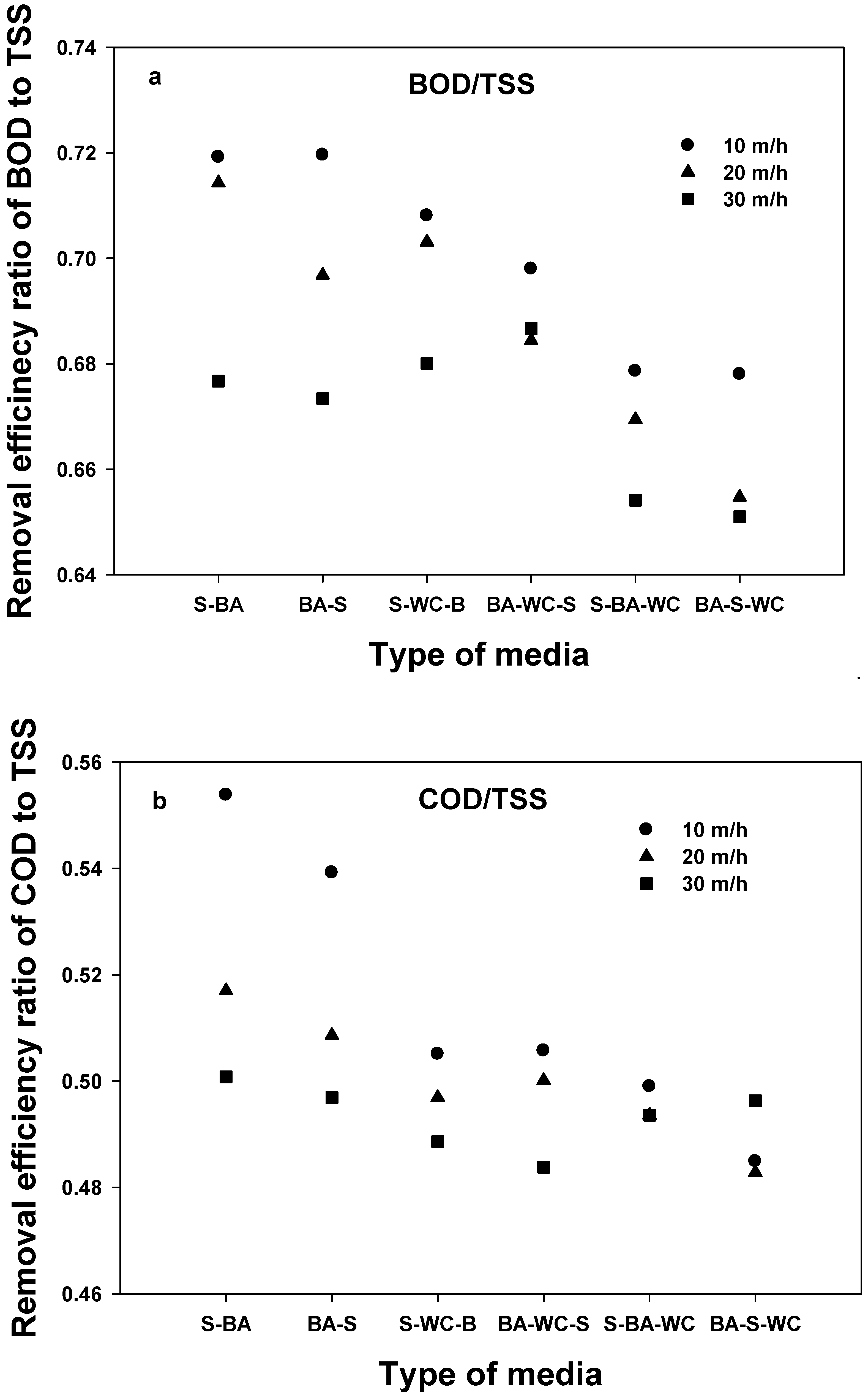
- The comparison of removal efficiencies for five different water quality indexes shows that TSS has the highest removal efficiency (regardless of filter media type), followed by T-P > BOD > COD = T-N, confirming that the filtration process is optimized for TSS.
- Similar removal efficiency trends of BOD, COD, and T-N with TSS indicate that the removal of BOD, COD, and T-N was primarily affected by the portion of the particle form.
- TSS was less sensitive to filter type and HC than other pollutants. Among a variety of experimental conditions, T-N had the highest variation, depending on the HC at S-BA and on the type of media at 10 HC.
- Through comparisons of BI, HRT, and C:N, it was found that biodegradation during filtration cannot occur.
- Unlike other pollutants, the highest removal efficiency was observed using BA-S for T-P. This corresponds to the adsorption effect for BA.
- Increased water quality using BA and WC demonstrated its capability and sustainability due to its low-cost and low-energy consumption factors, despite lower removal efficiency for water and wastewater treatment. In order to enhance the water quality and quantity treated, the system needs to be determined on the optimized operating parameters depending on influent concentrations or target pollutants.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecol. Appl. 1998, 8, 559–568. [Google Scholar] [CrossRef]
- Morée, A.L.; Beusen, A.H.W.; Bouwman, A.F.; Willems, W.J. Exploring global nitrogen and phosphorus flows in urban wastes during the twentieth century. Glob. Biogeochem. Cycles 2013, 27, 836–846. [Google Scholar] [CrossRef]
- USEPA. Guidance for Water Quality-Based Decisions, The TMDL Process, 1991. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi/00001KIO.PDF?Dockey=00001KIO (accessed on 11 July 2018).
- Ministry of Environment (ME). Key Projects of the Water Environment Policy Bureau, 2015. Available online: http://www.me.go.kr/home/file/readDownloadFile.do?fileId=116670&fileSeq=14&openYn=Y (accessed on 11 July 2018).
- Horner, R.R.; Skupien, J.J.; Livingston, E.H.; Shaver, H.E. Fundamentals of Urban Runoff Management: Technical and Institutional Issues; Terrene Institute: Washington, DC, USA, 1994; Available online: http://agris.fao.org/agris-search/search.do?recordID=US9536404 (accessed on 11 July 2018).
- Berkman, H.E.; Rabeni, C.F. Effects of siltation on stream fish communities. Environ. Biol. Fish. 1987, 18, 285–294. [Google Scholar] [CrossRef]
- Matilainen, A.; Vepsäläinen, M.; Sillanpää, M. Natural organic matter removal by coagulation during drinking water treatment: A review. Adv. Colloid Interface Sci. 2010, 159, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.H.; Lee, S.H.; Choi, J.W.; Hong, S.W.; An, B. Enhanced removal of phosphate on modified ion exchanger with competing ion. J. Korean Soc. Water Wastewater 2013, 27, 121–128. [Google Scholar] [CrossRef]
- Hipp, J.A.; Ogunseitan, O.; Lejano, R.; Smith, C.S. Optimization of stormwater filtration at the urban/watershed interface. Environ. Sci. Technol. 2006, 40, 4794–4801. [Google Scholar] [CrossRef] [PubMed]
- Hatt, B.E.; Fletcher, T.D.; Deletic, A. Hydraulic and pollutant removal performance of fine media stormwater filtration systems. Environ. Sci. Technol. 2008, 42, 2535–2541. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Shokouhian, M.; Sharma, H.; Minami, C. Laboratory study of biological retention for urban stormwater management. Water Environ. Res. 2001, 73, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Somdee, T.; Wibuloutai, J.; Somdee, T.; Somdee, A. Biodegradation of the cyanobacterial hepatotoxin [Dha7] microcystin-LR within a biologically active sand filter. Water Sci. Technol. 2014, 14, 672–680. [Google Scholar] [CrossRef]
- Cho, K.W.; Song, K.G.; Cho, J.W.; Kim, T.G.; Ahn, K.H. Removal of nitrogen by a layered soil infiltration system during intermittent storm events. Chemosphere 2009, 76, 690–696. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Yang, D.H. Removal of pollutants from wastewater by coal bottom ash. J. Environ. Sci. Health 2002, 37, 1509–1522. [Google Scholar] [CrossRef]
- Kandel, S.; Vogel, J.; Penn, C.; Brown, G. Phosphorus retention by fly ash amended filter media in aged bioretention cells. Water 2017, 9, 746. [Google Scholar] [CrossRef]
- Gupta, V.K.; Mittal, A.; Jhare, D.; Mitaal, J. Batch and bulk removal of hazardous colouring agent rose Bengal by adsorption techniques using bottom ash as adsorbent. RSC Adv. 2012, 2, 8381–8389. [Google Scholar] [CrossRef]
- Schipper, L.A.; Robertson, W.D.; Gold, A.J.; Jaynes, D.B.; Cameron, S.C. Denitrifying bioreactors-an approach for reducing nitrate loads to receiving waters. Ecol. Eng. 2010, 36, 1532–1543. [Google Scholar] [CrossRef]
- Blowes, D.W.; Robertson, W.D.; Ptacek, C.J.; Merkley, C. Removal of agricultural nitrate from tile-drainage effluent water using in-line bioreactors. J. Contam. Hydrol. 1994, 15, 207–221. [Google Scholar] [CrossRef]
- Goh, H.W.; Zakaria, N.A.; Lau, T.L.; Foo, K.Y.; Chang, C.K.; Leow, C.S. Mesocosm study of enhanced bioretention media in treating nutrient rich stormwater for mixed development area. Urban Water J. 2017, 14, 134–142. [Google Scholar] [CrossRef]
- Geronimo, F.K.F.; Maniquiz-Redillas, M.C.; Kim, L.-H. Fate and removal of nutrients in bioretention systems. Desalt. Water Treat 2014, 53, 3072–3079. [Google Scholar] [CrossRef]
- Li, W.W.; Yu, H.Q.; He, Z. Towards sustainable wastewater treatment by using microbial fuel cells-centered technologies. Energy Environ. Sci. 2014, 7, 911–924. [Google Scholar]
- Razali, M.; Kim, J.F.; Attfield, M.; Budd, P.M.; Drioli, E.; Lee, Y.M.; Szekely, G. Sustainable wastewater treatment and recycling in membrane manufacturing. Green Chem. 2015, 17, 5196–5205. [Google Scholar] [CrossRef]
- Kim, T.; Lee, J.; Lee, D.; Shin, H.; Kim, H.; Kwon, S. Verification of the Filter Media Applied to Filter Type Facility Considering the Treatment Efficiency Factor. J. Korean Soc. Water Environ. 2017, 33, 572–579. [Google Scholar]
- Ministry of Environment (ME), Guideline Manual of Installation and Operation Management for Nonpoint Facility. 2014. Available online: https://www.me.go.kr/home/web/policy_data/read.do?menuId=10259&seq=4070 (accessed on 10 August 2018).
- Hamoda, M.F.; Al-Ghusain, I.; AL-Mutairi, N.Z. Sand filtration of wastewater for tertiary treatment and water reuse. Desalination 2004, 164, 203–211. [Google Scholar] [CrossRef]
- MWH. Water Treatment Principles and Design; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; ISBN 978-0471110187. [Google Scholar]
- Kim, S. Variation of Water Quality and Storm Runoff Impact in the Suyeong River. Ph.D. Thesis, Pukyong National University, Busan, Korea, August 2016. [Google Scholar]
- McDowell, W.H.; Fisher, S.G. Autumnal processing of dissolved organic matter in a small woodland stream. Ecology 1976, 57, 561–569. [Google Scholar] [CrossRef]
- Huber, S.A.; Huber, W.; Frimmel, F.H. Summarische Parameter für Organische Verbindungen; R. Oldenbourg: Munchen, Germany, 1993; pp. 47–67. [Google Scholar]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering Treatment and Reuse; McGraw-Hill Higher Education: Boston, MA, USA, 2003; ISBN 978-0070418783. [Google Scholar]
- Oppenlöander, T. Photochemical Purification of Water and Air: Advanced Oxidation Processes; John Wiley & Sons: Weinheim, Germany, 2007; ISBN 9783527305636. [Google Scholar]
- Abdalla, K.Z.; Hammam, G. Correlation between biochemical oxygen demand and chemical oxygen demand for various wastewater treatment plants in Egypt to obtain the biodegradability indices. Int. J. Sci. Basic Appl. Res. 2014, 13, 42–48. [Google Scholar]
- Ho, L.; Hoefel, D.; Bock, F.; Saint, C.P.; Newcombe, G. Biodegradation rates of 2-methylisoborneol (MIB) and geosmin through sand filters and in bioreactors. Chemosphere 2007, 66, 2210–2218. [Google Scholar] [CrossRef] [PubMed]
- Davise, P.S. The Biological Basis of Wastewater Treatment; Strathkelvin Instruments LTD: Glasgow, UK, 2005; Available online: http://www.s-can.nl/media/1000154/thebiologicalbasisofwastewatertreatment.pdf (accessed on 11 July 2018).
- Qualls, R.G.; Haines, B.L. Measuring adsorption isotherms using continuous, unsaturated flow through intact soil cores. Soil Sci. Soc. Am. J. 1992, 56, 456–460. [Google Scholar] [CrossRef]
- Kalbitz, K.; Solinger, S.; Park, J.H.; Michalzik, B.; Matzner, E. Controls on the dynamics of dissolved organic matter in soils: A review. Soil Sci. 2000, 165, 277–304. [Google Scholar] [CrossRef]
- Snoeyink, V.L.; Jenkins, D. Water Chemistry; Wiley: New York, NY, USA, 1980; ISBN 978-0471051961. [Google Scholar]
- Wan, Z.; Li, T.; Shi, Z. A layered bioretention system for inhibiting nitrate and organic matters leaching. Ecol. Eng. 2017, 107, 233–238. [Google Scholar] [CrossRef]
- Saad, R.; Belkacemi, K.; Hamoudi, S. Adsorption of phosphate and nitrate anions on ammonium-functionalized MCM-48: Effects of experimental conditions. J. Colloid Interface Sci. 2007, 311, 375–381. [Google Scholar] [CrossRef] [PubMed]

| Type of Filter Media | Sand | Bottom Ash | Wood Chip |
|---|---|---|---|
| Main Composition a | SiO2: 70% Na2O: 13.3% CaO: 10% | SiO2: 55% Al2O3: 22.5% Fe2O3: 10% | Water and Organic: 95% K2O: 1% CaO: 2% MgO: 1% |
| Particle size | 0.85–2 mm | 0.5–1.2 mm | 2.00–4.76 mm |
| Uniformity coefficient | 1.2 | 1.8 | 1.6 |
| Porosity | 35–40% | 45–55% | 70–80% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.J.; Choi, J.-W.; Kim, T.-H.; Park, J.-S.; An, B. Effect of TSS Removal from Stormwater by Mixed Media Column on T-N, T-P, and Organic Material Removal. Water 2018, 10, 1069. https://doi.org/10.3390/w10081069
Kim HJ, Choi J-W, Kim T-H, Park J-S, An B. Effect of TSS Removal from Stormwater by Mixed Media Column on T-N, T-P, and Organic Material Removal. Water. 2018; 10(8):1069. https://doi.org/10.3390/w10081069
Chicago/Turabian StyleKim, Hoi Jin, Jae-Woo Choi, Tae-Han Kim, Jong-Sup Park, and Byungryul An. 2018. "Effect of TSS Removal from Stormwater by Mixed Media Column on T-N, T-P, and Organic Material Removal" Water 10, no. 8: 1069. https://doi.org/10.3390/w10081069
APA StyleKim, H. J., Choi, J.-W., Kim, T.-H., Park, J.-S., & An, B. (2018). Effect of TSS Removal from Stormwater by Mixed Media Column on T-N, T-P, and Organic Material Removal. Water, 10(8), 1069. https://doi.org/10.3390/w10081069






