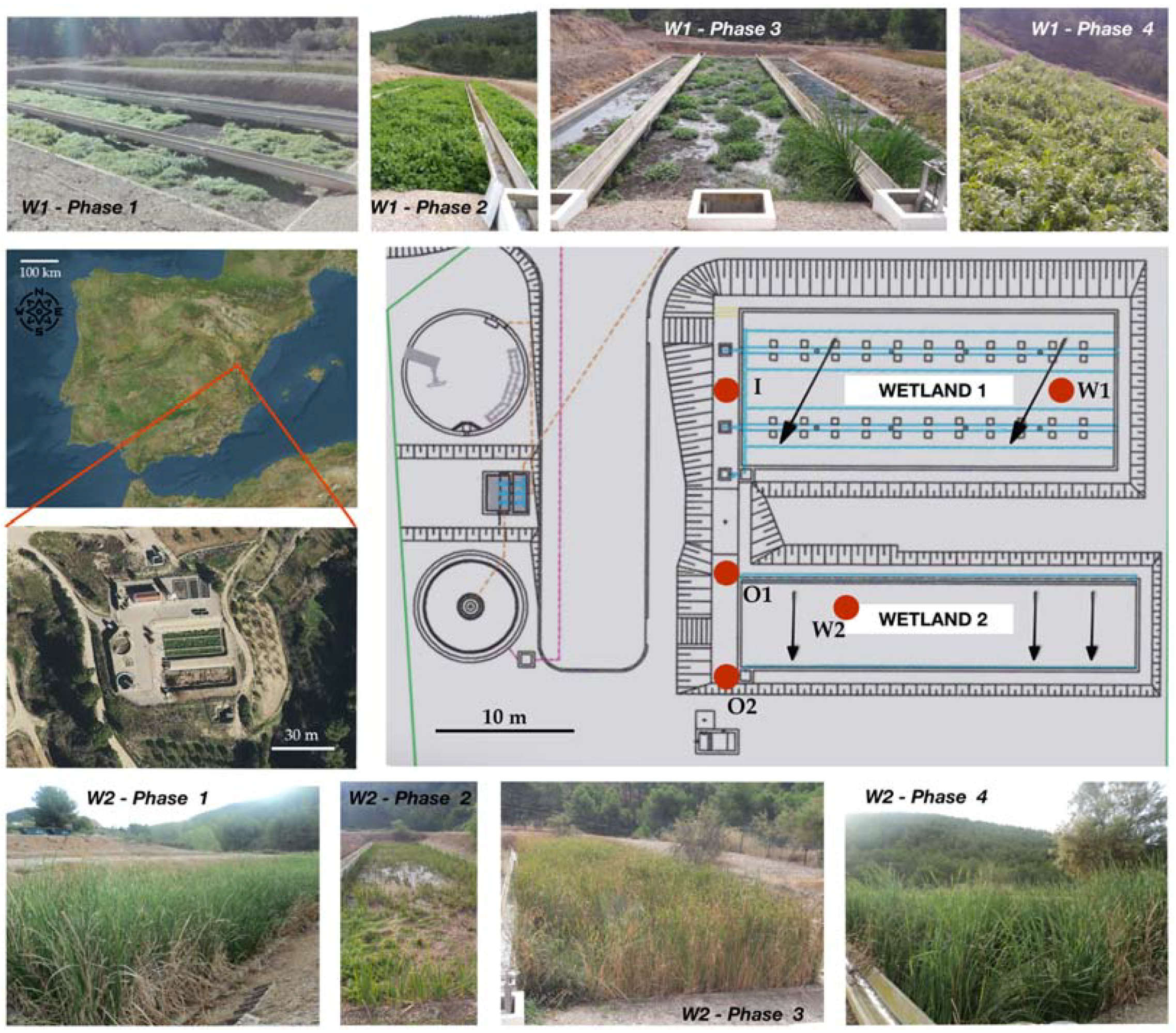
1. Introduction
Non-naturalized waters that originated from wastewater discharges, despite being treated in waste water treatment plants (WWTP) achieving the legislated quality standards, show quite different features to those of the natural environments where they are poured. This can cause a drop in the concentration of dissolved oxygen in the receiving environment, as well as promote eutrophication, the loss of biodiversity, and the release pathogenic microorganisms [
1,
2]. All of this causes a degradation of the natural environment, diminishing its ecological integrity and its capacity to provide ecosystem services [
3]. In Europe, the Directive 2013/39/EU has established the need to implement cost-effective purification treatments, which requires the development of innovative water treatment technologies, using more sustainable processes, such as artificial wetlands [
4,
5]. Furthermore, limits in the concentrations of the pollutants that have been fixed by European Directive 91/271/EEC for WWTP effluents may be excessively high for some deficit basins, such as those in the Mediterranean, where the discharge may constitute an important proportion of the total volume of the receiving water bodies. This problem, which has already been addressed by the European Commission, encumbers the achievement of the good ecological status of European water bodies that is required by the EU Water Framework Directive [
6].
Constructed wetlands (CWs) are engineered systems that are designed to treat different types of polluted water using natural biogeochemical processes. These systems can effectively treat raw, primary, secondary, or tertiary treated sewage, as well as different types of industrial wastewater, and they are considered environmentally friendly and sustainable options for wastewater treatment [
7], simulating the structure of natural wetlands. CWs are classified according to the wetland hydrology as free water surface and subsurface systems. Subsurface flow CWs are generally classified depending on the flow direction, as those with horizontal or vertical flow. The latter are usually fed intermittently and are generally capable of achieving higher nitrification rates because of the greater oxygenation of the wetland bed. The serial use of a combination of several types of wetlands is also common [
8]. On the other hand, CW may contain both emergent and/or submerged vegetation, which determines the wetland metabolism and performance [
9]. For instance, vegetation prevents the resuspension of solids and increases the oxygenation of the soil [
9]. Nutrient removal by vegetation may occur either by direct plant uptake, particularly nitrogen, or by facilitating the uptake by microorganisms [
10,
11] and chemical precipitation [
12]. Additionally, plants increase the available carbon in the sediment, which in turn can favour the nutrient-removal processes, such as denitrification [
11].
The selection of the vegetation cover for the CW depends of several factors, such as its geographical distribution, growth capacity, climatic adaptation, the type of contamination to be treated, the tolerance to CW conditions, and the costs of maintenance. Despite this high number of factors, there are few species of aquatic plants that are overrepresented in CWs. In relation to this, Vymazal [
13] describes the regular use of proliferative plants, such as those of the genera
Typha,
Scirpus,
Phragmites,
Juncus, and
Eleocharis, which are usually planted as monocultures. Monocultures, especially of exotic invasive species, might represent a risk for the conservation of natural biodiversity of aquatic ecosystems downstream, but also misprizes the potential functional complementarity of combining the different plant species [
14]. Moreover, some authors pointed out the suitability of using wild plant species for the treatment of effluents in constructed wetlands [
15], an issue that had not been sufficiently addressed so far. For instance, there has been no mention in the scientific literature for the use of wild species, such as
Helosciadum nodiflorum, which thrives in a wide variety of Mediterranean aquatic ecosystems, but more particularly, spreads within the nutrient-rich shores of Mediterranean streams.
In this study we evaluated the performance of a CW system, where two plant species (Helosciadum nodiflorum and Typha latifolia) were used separately in two different ponds that were working sequentially. This CW represents a tertiary treatment that receives a continuous flow from the outlet of a wastewater treatment plant (WWTP). Here, we assessed the pollution removal efficiency of the system and its capability to naturalize water, either by the simultaneous serial operation of both of the wetlands or by the single use of one of them. Our results will help to discern to what extent it is possible to gain advantages of the pollutants’ removal by using different plants species in combination, which shows a fast growth and have minimal maintenance costs. This would represent an inexpensive way to diminish the disturbance on the waterbody receiving the effluent, as the waters in the outlet of the CW system may then show more similar features to that of the receiving Mediterranean stream.
3. Results
The effect of both of the wetlands on the waters’ physical, chemical, and biological features differed throughout the different phases of the study, and was influenced by the operational mode. Some of the waters’ physical and chemical variables did not vary significantly when passing the wetlands, such as the water pH and electrical conductivity, whereas others showed significant variations (
Supplementary Figure S1). The dissolved oxygen concentrations in the outlet of the WWTP (I, inlet to Wetland 1) were usually comprised between 1.7 and 2.2 mg L
−1, although they occasionally dropped to values around 1 mg L
−1, or increased to maximum values in Phase 4 that reached up to 3.7 mg L
−1. The dissolved oxygen concentrations in the outlet of Wetland 1 (O1) commonly dropped slightly, to values around 1.6 mg L
−1, and this decrease made a significant difference (
p < 0.001) in the behaviour of the dissolved oxygen in Wetland 2 compared to Wetland 1, as in Wetland 2 they slightly increased to averages of around 2 mg L
−1, especially in phase 4. Concerning the pH, it was slightly alkaline, although no statistically significant changes were found in the pattern that was shown by both of the wetlands (
p = 0.843). The alkalinity was mostly maintained in Wetland 1, with respect to its inlet (I), and decreased slightly (2–17%) when passing through Wetland 2. Among the possible comparisons of the co-operational phases (1 & 4), this drop was significantly higher in Wetland 2 compared to Wetland 1 only in phase 4 (
p = 0.007), when Wetland 2 was operating under optimal conditions. The water electrical conductivity was highly variable and fluctuated in the outlet (I) of the WWTP that entered Wetland 1, from 430 to around 1000 μS cm
−1 and, as a general pattern, it dropped slightly (up to 6% maximum) in the transit through both of the wetlands, but without significant differences of the decreasing pattern when both of the wetlands were compared (
p = 0.435). The removal effect on suspended solids, however, fluctuated very much in Wetland 1, since they increased in phase 1, whereas they decreased between 31% (phase 4) and 42% (phase 2). Contrastingly, the suspended solids were continuously removed in Wetland 2, with removal percentages of around of 9% (phases 3 and 4) or even higher (20% in phase 1), but the contrasting variation pattern among both of the wetlands was statistically significant (
p = 0.015) only in Phase 1, when Wetland 2 reduced the suspended solids by 20%, whereas they increased by around 50% in Wetland 1. Nevertheless, the CW system generally contributed to the suspended solids removal. In summary, among these parameters, the combined effect of both of the wetlands contributed to a slightly decreased salt content, as it was revealed by the consistent decrease of conductivity, to a small drop in alkalinity and, especially, to the removal of the suspended solids, whereas both the dissolved oxygen and pH were mostly similar after the transit through the whole wetland system.
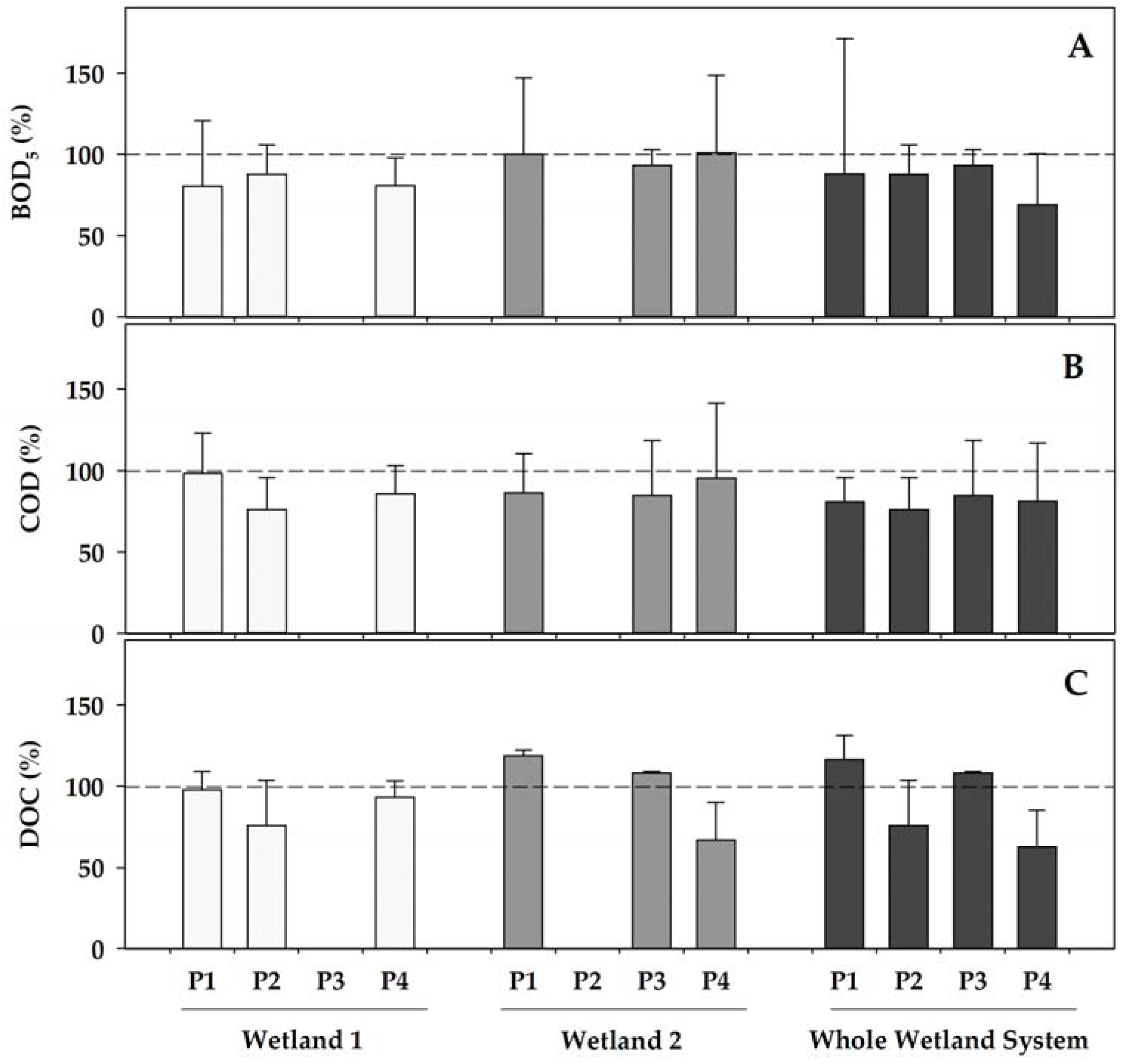
As a general pattern, both of the wetlands contributed to a further increase in the removal of the organic matter that was already initiated by the WWTP (
Figure 2). When operating (phases 1, 2, and 4) the concentrations of the indicators of organic matter were reduced in Wetland 1 by 13–20% (for BOD
5), by 2 to 23% (for COD), and by 2 to 24% (for DOC), with the highest decreases generally occurring in phase 2, when Wetland 1 was fully operational with a healthy coverage of
Helosciadum nodiflorum, and the temperatures became the highest during the two fully operational initial phases (phases 1 and 2) of Wetland 1. Contrastingly, Wetland 2, which was covered by
Typha latifolia, showed a more erratic pattern when operating, with small reductions (maximum 7% in Phase 3) in the BOD
5, decreases in the COD in all of the operating phases, from 5 to 15%, whereas the DOC increased by 8–16% in phases 1 and 3, but decreased by 33% in phase 4. Among these, only the highest DOC reduction in Wetland 2 compared to Wetland 1 in phase 4 was statistically significant (
p = 0.026) when comparing both of the co-operational phases (phase 1 and 4). In any case, the water organic content was reduced through its sequential transit over the combined constructed wetland showing total reductions ranging 7–30% for BOD
5, 15–24% for COD, and up to 37% for DOC.
Among the fractions of the chromophoric dissolved organic matter (CDOM), the most noticeable reductions, although modest, occurred for the CDOM with a high molecular weight (S350–400), particularly in phase 4, when, as mentioned previously, Wetland 2 operated more efficiently in removing the dissolved organic carbon (
Figure 2C). During this period, the average decrease in the value of S350–400 that was attributable to this wetland was slightly above 10% (
Supplementary Figure S2). However, there was no statistically significant differential reductions for any of the wetlands in any of the phases (
p = 0.317). On the other hand, no significant differences (
p = 0.744) in the transformation patterns were observed between Wetland 1 and Wetland 2 when comparing the changes for the fraction of low molecular weight (S275–295) during the co-operating phases. Consequently, the changes of the Sr ratio mainly mimicked the variations of the high molecular weight CDOM.
With respect to the fluorescent fraction of the dissolved organic matter (FDOM), although representing a limited fraction of the total DOM, higher dynamisms appeared. The FDOM of types A and C, which are attributable to the presence of humic and fulvic acids, showed a clear increase during the earlier period (Phase 1), which eventually might have come from the leaching of the wetlands ground (
Supplementary Figure S2). On the contrary, from phase 2, the organic matter of these types that was entering the wetland system, particularly that of type C, began to be effectively removed, with maximum decreases close to 40% in Wetland 1 during phase 2, and around 25% for Wetland 2 in phase 4. This increased efficiency coincided with a progressive increase of the percentage of the bacteria with high DNA content (%HDNA). Nevertheless, the pattern of reduction for the humic types of FDOM was not significantly different for both of the wetlands, neither for type A (
p = 0.423) nor for type C (
p = 0.852). In parallel, the maximum removal of the FDOM of type T, which predominantly derives from tryptophan and tyrosine, thus indicating the presence of proteins, also reached its maximum in Wetland 1 during phase 2, with nearly 41% removal. Wetland 2, when operating, also removed efficiently this fraction of FDOM-T, especially in the warmer phase 4, when the removal reached up to 31% and was significantly higher (
p = 0.013) for Wetland 2 than that of Wetland 1.
Overall, the higher organic matter removal capacity of Wetland 1, according to all of the parameters that were related to the organic matter, was then achieved in phase 2, when this wetland was fully operative with a healthy cover of Helosciadum nodiflorum. Contrastingly, most of these parameters showed a higher removal efficiency in phase 4 for Wetland 2, when the health status of its Typha latifolia cover was optimum, with the plants actively growing under warmer temperatures.
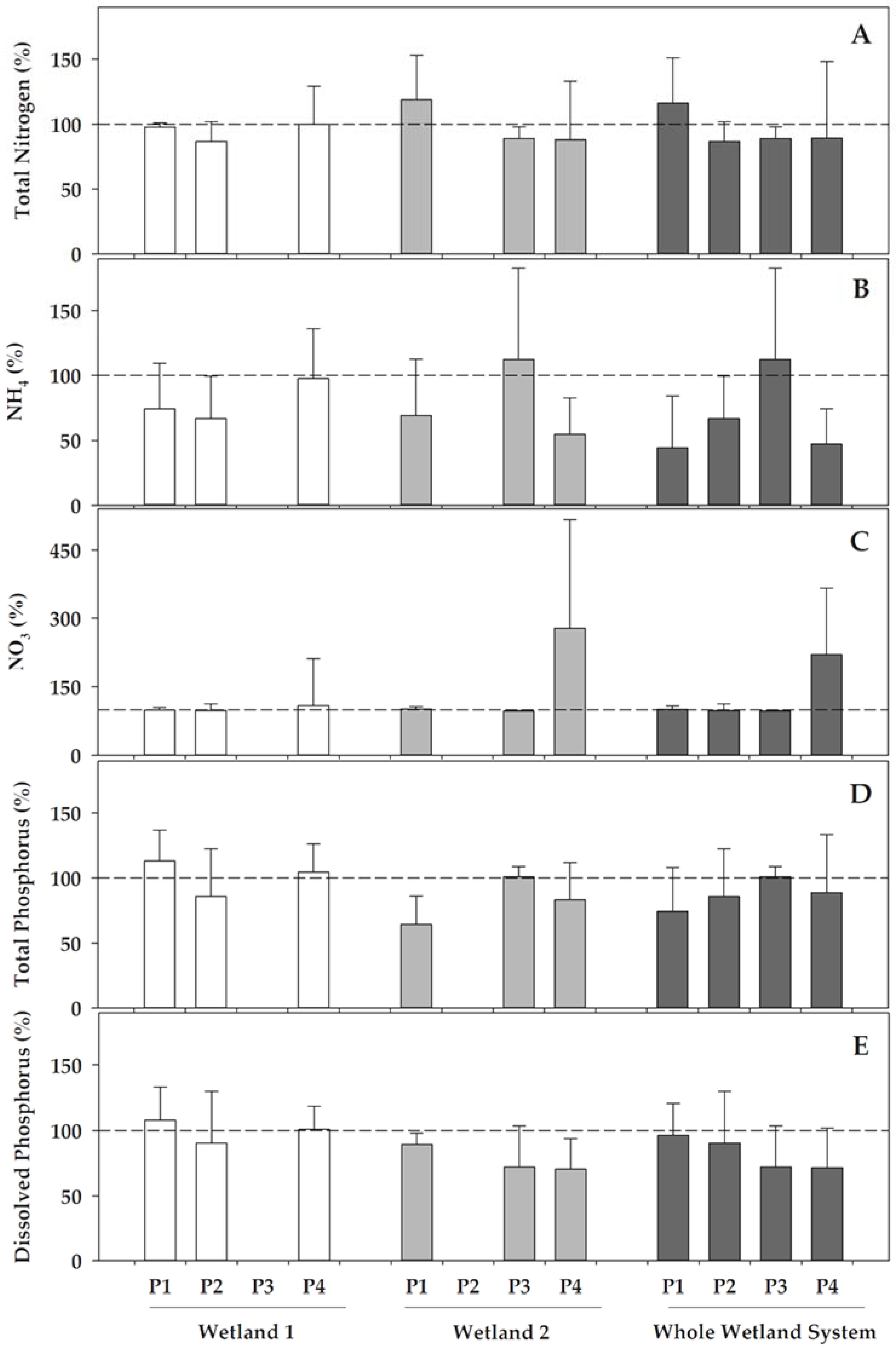
With regards to the nitrogen and phosphorus compounds, the studied constructed wetlands not only retained a part of these nutrients, but also influenced redox transformations (
Figure 3). Concerning the nitrogen compounds, the most remarkable effect for both of the wetlands was that they generally favoured the ammonium nitrification, and this resulted in remarkable decreases of ammonium concentrations compared to the inlet (I) from the WWTP, which ranged 3–33% in Wetland 1 and 30–45% in Wetland 2, except, for the latter, in phase 3, where the ammonium did not decrease in the recently re-operating Wetland 2. The nitrification processes resulted in increases in the nitrate concentrations in water when passing through wetlands, although with a different behaviour in both of the wetlands. In Wetland 1, the denitrification should have occurred in phases 1 and 2, as the increase in nitrate concentration did not yet cover the decrease in ammonium, which was also reflected in the decrease of the total nitrogen, which ranged from 3 to 13% in these phases. Contrarily, the total nitrogen almost maintained its concentration in phase 4 in Wetland 1, and most of the ammonium was transformed into nitrate. Wetland 2 also presented nitrification, with ammonium decreases ranging from 30% and 45% in phases 1 and 4, respectively, although it did not decrease in phase 3, when
Typha latifolia was recently re-growing after the biomass withdrawal. In Wetland 2, part of the ammonium was nitrified in phase 1, but there was also an additional increase in the concentration of the total nitrogen compared to the water entering this wetland. Similarly, ammonium decreased in phase 4 and nitrate increased concomitantly, but the decreases in total nitrogen also revealed that part of the nitrogen could have been denitrified. When compared between both of the wetlands, denitrification was only significantly different (
p = 0.026) when it was linked to the higher reduction of the total nitrogen in Wetland 1 during phase 1. Contrastingly, nitrification, as deduced from the transformations of ammonium into nitrate was, in general, much more active in Wetland 2 during phase 4 than in Wetland 1 (
p = 0.007 for ammonium reduction and
p = 0.019 for nitrate increase).
Although the nitrogen was transformed and removed in Wetland 1, the phosphorus concentrations, both as orthophosphate and total phosphorus, were not reduced significantly in this wetland, but were instead varying with an unclear pattern, either increasing or decreasing compared to its the inlet (I) from the WWTP (
Figure 3). In contrast, Wetland 2 remarkably reduced both total P (17–35%, except in phase 3, when it maintained on average the concentrations entering this wetland), and orthophosphate (12 to 30%), when comparing its outlet (O2) with its inlet (O1). Overall, Wetland 2 was much more efficient in phosphorus removal than Wetland 1, both for the total phosphorus (
p = 0.001) and soluble orthophosphate, and accounted for most of the P-removal of the CW system.
Except for phase 1, both of the wetlands generally promoted a decrease in bacterial abundance, which was reduced by 15–50% in Wetland 1, and by 13–23% in Wetland 2. Concerning the bacterial activity, which was obtained from the relative abundance of the more active (HDNA) to less active (LDNA) bacteria, the relative abundance of the HDNA versus LDNA cells increased in all of the phases by 10 to 37% in Wetland 1 and by 7 to 65% in Wetland 2. Both trends were solid, and were not significantly different when comparing both of the wetlands (p = 0.394 for bacterial abundance, and p = 0.769 for the relative contribution of HDNA bacteria).
4. Discussion
The release of the used waters into the environment requires effective treatments that could reduce the amount of pollutants in such a way that they cause no degradation to the receiving water bodies [
21]. However, even when these pollutants could have been reduced on a conventional WWTP, the physical, chemical, and biological features of these waters might differ very much to those from the receiving aquatic ecosystems [
22,
23], which strengthens the need to consider the role of constructed wetlands, not only as systems helping to reduce the amount of pollutants, but also, when these had already been reduced to acceptable levels, as driving these water features to others that are more similar to those of the receiving natural environment. Our work was conducted in a small constructed wetland that is annexed to a wastewater treatment plant (WWTP), the latter being efficient for pollutant removal. , Nevertheless, the conventional WWTP pours water of very different characteristics to those that circulate through the environment, in this case, a Mediterranean stream where the natural flows are very low (a few hundred litres per second, except during occasional storms) and, consequently, the effect of the treated waters on the environmental features could be strong. Our study provides evidences on how the ‘naturalization’ processes of the already treated waters benefits the receiving environment by decreasing the physical, chemical, and biological differences of the poured waters, compared to the small flowing waters of the receiving stream. These processes, for example, favour shifts to more oxidised compounds of certain elements (e.g., nitrogen), further degrading the dissolved organic matter towards more recalcitrant and inactive forms, and lowering bacterial loads. This type of studies, which is relatively uncommon on semiarid environments such as those of the Mediterranean basin, help by providing guidelines to avoid further deterioration of aquatic ecosystems, which is enforced by environmental laws such as, for Europe, the Water Framework Directive.
The studied system consists of two serial subsurface flow wetlands, one vertical and one horizontal, both of which contained different aquatic plant species (
Helosciadium nodiflorum in Wetland 1 and
Typha latifolia in Wetland 2). Apart from nicely illustrating the naturalization process of the already treated wastewater, the main novelty of our study was the use of
H. nodiflorum, a native macrophyte that regularly appears in the streams of Europe. This plant shows a large standing biomass in rivers, even when temperatures and nutrients content are low [
24], which makes it suitable for the water naturalization processes, even during the colder months under a Mediterranean climate. This naturalization consisted mainly in both the removal and the transformation of the remaining pollutants. As examples of the most remarkable effects on water renaturalization, the combined action of both of the wetlands allowed for the reduction of BOD
5 from averages close to 9.5 mg O
2 L
−1 in the effluent of the WWTP (I) to averages of 4.8 mg O
2 L
−1 in the outlet of the CW system (O2), which are closer to the typical averages of the receiving streams (2.2 mg O
2 L
−1) in the area, according to public data available from the Natura 2000 monitoring network (
http://www.agroambient.gva.es/documents/91061501/109939278/Evaluaci%C3%B3n+de+los+Datos+de+Calidad+de+Aguas+para+el+Seguimiento+de+Masas+de+Agua+Fluvial+en+los+Espacios+Red+Natura+2000/f9101f01-d819-4468-a6b7-e4fe823b6f08). Similarly, other pollutants, such as the suspended solids, dropped from higher concentrations in the WWTP effluent (8.4 mg L
−1) to values that were even lower in the CW outlet (3.5 mg L
−1) than those that are typically averaged in the receiving streams (5.8 mg L
−1). These and other positive effects of the CWs treatments minimised the impacts of the poured treated wastewaters on the receiving stream.
Any interpretation of the results that were obtained here need to bear in mind the different phases of the study. Only two of the four studied phases (1 and 4) were co-operative for both of the wetlands. In phase 1, in the fall, the temperatures were still relatively mild during the first weeks, so the temperature dependent processes were still quite operative. However, the healthy status of the
H. nodiflorum in Wetland 1 contrasted with the beginning of the autumnal senescence of
T. latifolia in Wetland 2, which implied a higher operational status of Wetland 1. This wetland, which was covered by
H. nodiflorum, was still fully operative (
Figure 2 and
Figure 3), even under the colder conditions at the end of phase 1 and the beginning of phase 2 (
Supplementary Table S1). This supports one of the main discoveries of our study, which is the possibility of using the native plant
H. nodiflorum for wastewater treatment during winter periods, when the other plants that are typically used in constructed wetlands cannot be operative because they remain in a vegetative status. The
T. latifolia cover of Wetland 2 was pruned in phase 2, and this wetland was closed, so only Wetland 1 was operating during this phase. On the contrary, Wetland 2 was re-opened in phase 3 and
T. latifolia started to grow, although slowly, whereas the
H. nodiflorum cover in Wetland 1 was pruned and this wetland was closed. Later, in phase 4, both of the wetlands were co-operating, but the
H. nodiflorum cover had just started to recover in Wetland 1, whereas Wetland 2 was already recolonized by a healthy and dense
T. latifolia cover. This implies that, comparatively, the operational status of the latter was healthier than that of
H. nodiflorum in Wetland 1, which coincided with the highest (summer) temperatures (
Supplementary Table S1). Additionally, the higher hydraulic load that was experienced by the smaller Wetland 2 (
Table 1) implies that the reductions in the pollution loads that were achieved in this wetland were relatively higher at equal values, when compared to those of Wetland 1 in terms of areal efficiency, although the
T. latifolia cover of Wetland 2 was quite inefficient during the colder periods. Overall, our results also demonstrate the complementarity of both plants that were used in our wetland system, with Wetland 1 covered by
H. nodiflorum being more capable of treating the WWTP during the colder months, contrasting with Wetland 2 covered by
T. latifolia being more efficient during the warmer period. This makes an important advance in overcoming one of the main problems of the constructed wetlands that are used in wastewater treatment, namely its much lower efficiency in colder months because of the low operational status of the typically used plants.
Despite the strong reduction, the waters that were treated in the WWTP still contained higher levels of certain pollutants than those of the clean water that was previously taken from the environment that was to be used by population. This includes, for example, higher levels (and different relative amounts) of inorganic nitrogen and phosphorus forms, which are primary nutrients promoting eutrophication. For sure, the nutrient loads that entered the constructed wetland system from the WWTP of Higueruelas were not very high compared to those where the wastewater has not been so intensively treated [
25]. However, these waters still showed the characteristic composition of effluents of urban wastewaters, with, for example, a relatively high concentration of ammonium and soluble phosphorus. Thus, the main function of this wetland system would be to naturalize the effluent, discharging in a small Mediterranean stream located in a mountain area, which is a sub-tributary of the Túria River, the main fluvial axis of the region. To this respect, the huge removal of ammonium and suspended solids in the constructed wetland system, if compared with the WWTP effluent, although being variable over time depending on the growth of the plants, equalled or was even higher than that shown by other studies that were conducted in south Europe aiming to recover the natural features of streams, which also involved the use of constructed wetlands as tertiary treatments. For example, Huertas et al. [
26] described the use of recycled water for stream flow augmentation, which, as in our case, has the potential to improve the stream habitat in water courses suffering strong water abstractions, particularly in the Mediterranean region (East of Iberian Peninsula), where renewable water use is an important concern.
The design of the wetlands (either vertical or horizontal), together with the choice of the aquatic plant in each wetland, may be the main factors in determining the yields of nutrients and organic matter reduction, in such a way that a combination of horizontal and vertical flow wetlands could remove up to 90% of the organic load, and of the total N and P of the untreated sewage [
11,
27]. In addition, the horizontal flow wetlands have an advantage in long-term P-removal, because P is bound to organic substances [
28]. Concerning phosphorus, we also found a similar behaviour, with most of the phosphorus removal occurring in the horizontal flow Wetland 2, and with no consistent P-removal in the vertically flowing Wetland 1, where the newly used of
H. nodiflorum did not significantly contribute to a significant increase in phosphorus removal. The higher phosphorus reductions occurred in Wetland 2, during the periods of the healthier growing of
Typha latifolia. This is also consistent with other studies, which also showed the role on P-removal involving this species [
29,
30]. However, P-retention by plants could not be the most effective option, since aquatic plant biomass may become P-saturated after a relatively short time [
28], or it might even become an important P-source of after plant senescence [
31]. By contrast, the P-retention capacity could be determined by the adsorption of phosphorus to soil particles, or by chemical precipitation, with either inorganic (i.e., Ca, Al, and Fe) and organic compounds, as previously mentioned. The calcareous regional lithology, providing water in the area with huge amounts of dissolved Ca, suggest that P bound to Ca was likely a prevailing form for P-retention in the soils of the studied CW.
Nitrification occurred in both of the wetlands, but with a higher transformation of ammonium into nitrate in Wetland 1 when both of the wetlands were operating simultaneously under similar operative conditions (Phase 1). This also agrees with the higher nitrification rates that were generally reported for vertical flow subsurface wetlands, compared to those with horizontal flow [
32], which, in our case, is also demonstrated when the riparian
H. nodiflorum is newly used. Additionally, both of the wetlands were capable of efficiently removing nitrogen through denitrification, but Wetland 1, especially in phase 1, presented higher removal rates. In this wetland, the anaerobic consumption of the remaining organic matter using nitrate (both that coming from the WWTP and the newly created by nitrification in the upper wetland layers) as an electron acceptor [
33], was favoured by the lower oxygen content in phase 1. In this co-operating period, only Wetland 1 was capable of nitrogen removal through denitrification. However, during the other co-operative period (Phase 4), Wetland 2, which was then more healthfully operating than Wetland 1, was more efficient than Wetland 1 in removing nitrogen, although nitrification was the dominating nitrogen pathway in this horizontally flowing subsurface wetland covered by
T. latifolia. Nevertheless, the removal of ammonium by nitrification, with further partial denitrification to remove nitrogen from the poured waters, occurred in both of the wetlands when each was fully functional. This also enhances the importance of the novelty of our study in showing the utility of
H. nodiflorum as a plant that can be usable in treatment wetlands.
As a general pattern, the studied CW system contributed to further enhancing the removal and transformation of organic matter that was already performed by the WWTP, with the removal in the wetlands system reaching up to 40% (depending on the studied variable) of the organic loads that were already reduced to low levels by the WWTP. The changes that were observed in the optical properties of organic matter might shed light on these transformations. With regards to the colourless dissolved organic matter (CDOM), most of the variability was found for the fraction of high molecular weight, which was parametrized by the slope between wavelengths of 350 and 400 nm (
S350–400), when compared to that of the low molecular weight (
S275–295), which indeed remained almost unchanged. The increases of
S275–295 values are typically related with photodegradation processes, which cause bond cleavage and/or the formation of photoproducts with a low molecular weight [
34]. Accordingly, the limited exposure of wastewater to light in the studied wetland system would explain the minimal variations that were observed for this DOM fraction. A high exposure of DOM to sun light would have caused a shift from high to low molecular compounds that distinctly would yield an increase of
Sr values, which did not occur in our case. The decreases that were observed during some of the periods in the slopes at the higher wavelength range (
S350–400) seem to be, however, more related to the biodegradation processes, as also indicated by the net loss of DOC, COD, and BOD
5 that were observed during these periods. Instead, the observed net increment in the outlet of the
S350–400 values in phase 2 might have been caused by the increase of the lignin derived from vegetation, which was likely originating from the
H. nodiflorum that was present in Wetland 1, which experienced a slightly less healthy status during the winter period.
The reductions of the organic matter loads were also visualized by the concomitant reductions in the fluorescent DOM compositional indicators (FDOM). In our study, these fluorescence parameters displayed net average decreases of around 40%, starting from phase 2. The fluorescence signals of type A and C denote the presence of humic-like substances, and the fluorophores of the type T are indicative of the occurrence of aromatic amino acids (i.e., proteins) and/or undegraded polyphenols in the sample [
34]. The former are then proxies of more recalcitrant DOM, whereas the signal of type T indicated a fresh-like (i.e., labile) DOM. In our case, the ratio between the fluorescence peaks of type A and type T decreased slightly along the studied period, from mean values in phases 1 and 4 of 8.2 and 6.4, respectively, which would have accordingly indicate greater aromatic content in the initial DOM pool.
Both of the wetlands promoted notable decreases of bacterial abundances, which thus indicate a good capability to act as biofilters. The physical retention of the bacteria through the filtration substrate may explain these reductions, as well as the low survival rates of the allochthonous microbes that were entering with the wastewater. These might have included pathogens as well as the bacteria from the activated sludge, which had a low functional relation with the wetland environment [
35]. Additionally, the retention of bacteria by filtration can be improved with the presence of plants in the wetland [
36], which likely occurred in our studied system. Nevertheless, the reduction of the total bacterial numbers in the studied CW reached up to 80% in some periods, which indicates removal efficiencies that were comparable to other subsurface flow constructed wetlands [
37]. Interestingly, and despite this net reduction in the bacterial numbers, the wetland performance enhanced the microbial activity of the remaining or newly growing community, as indicated by the increase of the proportion of the bacterial cells with a high DNA content [
20,
38]. This demonstrates the high degradative capacity of the pollutants by the wetland’s microbiota. Several studies indicated that microbial activity is higher in the subsurface (vertical flow) systems compared to the surface flow systems [
39], which agrees with observations that showed how the wetland design mainly determines the activity and structure of the microbial assemblage, rather than the characteristics of the wastewater influent [
40]. This is a promising finding in our study, as it indicates that the microbial community that was leaving the wetland was not largely influenced by the composition of its inlet, but it could still play its role in pollutants removal. Future research would then be related to the molecular and functional diversity of the microbial community involved in the tertiary treatment.
The role of aquatic plants in constructed wetlands has been widely studied [
9,
41], although the knowledge on the relative relevance of the species used has always been more limited [
42]. Vymazal [
13] reported an exhaustive revision of the aquatic plants that are used in constructed wetlands, some of them, such as the species of genera
Typha,
Scirpus,
Phragmites,
Juncus, and
Eleocharis, being widely used.
Typha latifolia, which grew in our Wetland 2, is one of the most used plants in constructed wetlands, commonly presenting a high efficiency in inorganic nutrient and organic matter removal [
29]. However,
H. nodiflorum, the riparian plant covering Wetland 1 in our study, was not mentioned in this detailed revision, neither in successive studies, so we could assume that our study represented the first that was conducted with this species in these types of constructed wetlands. Thus, our contributions implied an important step for the basic knowledge to be applied when constructing wetlands for wastewater treatment.
H. nodiflorum had been only used sporadically for the reduction of heavy metals in soils [
43] and its antimicrobial activity has also been reported [
44]. However, it was never demonstrated so far as being usable in treatment wetlands, and even less was known on its capacity for offering good efficiencies under the more problematic winter periods, which enhances the importance of our results. The biomass production of
H. nodiflorum in the vertical flow Wetland 1 was greater than that of
T. latifolia. This might have responded to a lower capacity of
T. latifolia to adapt to the lack of water occurring in the unsaturated vertical system [
45]. Additionally,
H. nodiflorum is a wild plant that resists moderate pollution levels and relatively cold temperatures, thus being appropriate for the environmental context where the studied CW system is located. In this sense, there is a growing evidence in the importance of selecting native plants species for constructed wetlands to maximize the removal and purification of effluents [
10,
15] in such way that these benefits could be compromised if the selection of the plants is not appropriate, particularly in monoculture systems [
45]. Here we demonstrated how a native plant such as
H. nodiflorum, which is well adapted to the climate and local conditions, could reach adequate levels of efficiency for its use in these types of wetlands, with operational advantages and cost reduction. This plant might then have a relevant role in the naturalization of treated waters, especially during periods when most of the plants that are typically used in treatment wetlands cannot be fully operative.