Chromosome Synapsis and Recombination in Male Hybrids between Two Chromosome Races of the Common Shrew (Sorex araneus L., Soricidae, Eulipotyphla)
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Karyotyping
2.3. Meiotic Chromosome Preparation, Staining and Identification
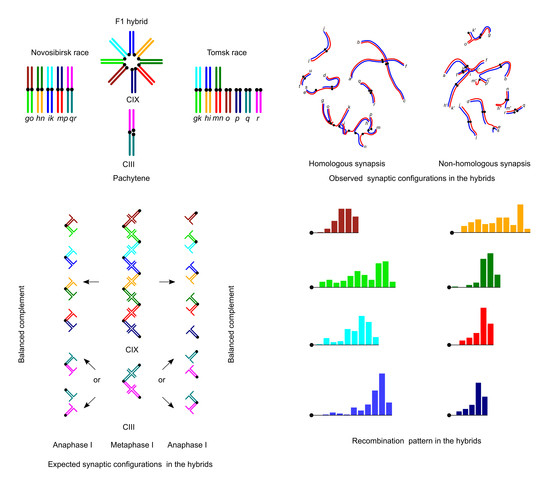
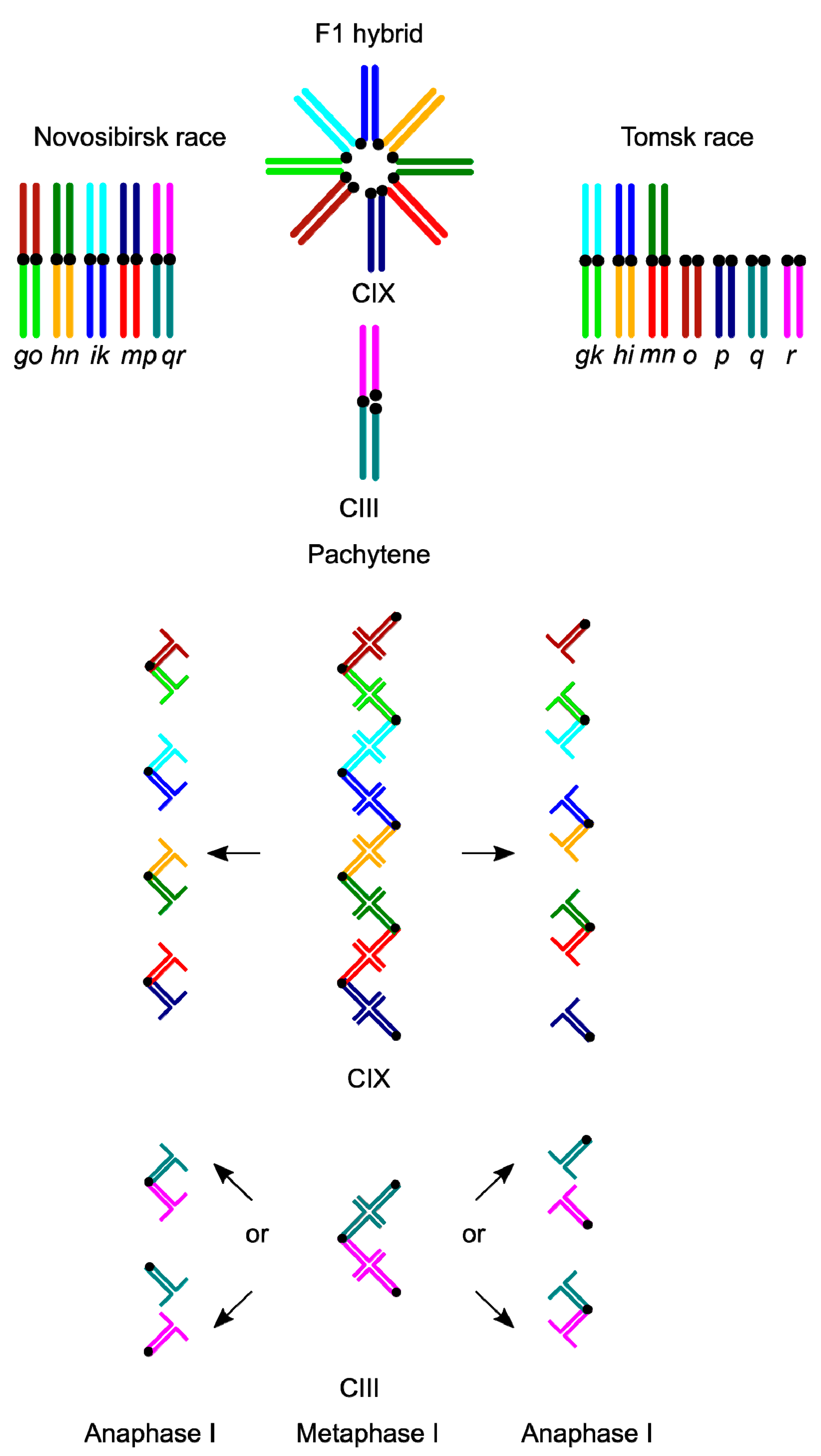
3. Results
3.1. Karyotype Variability in the Hybrid Zone
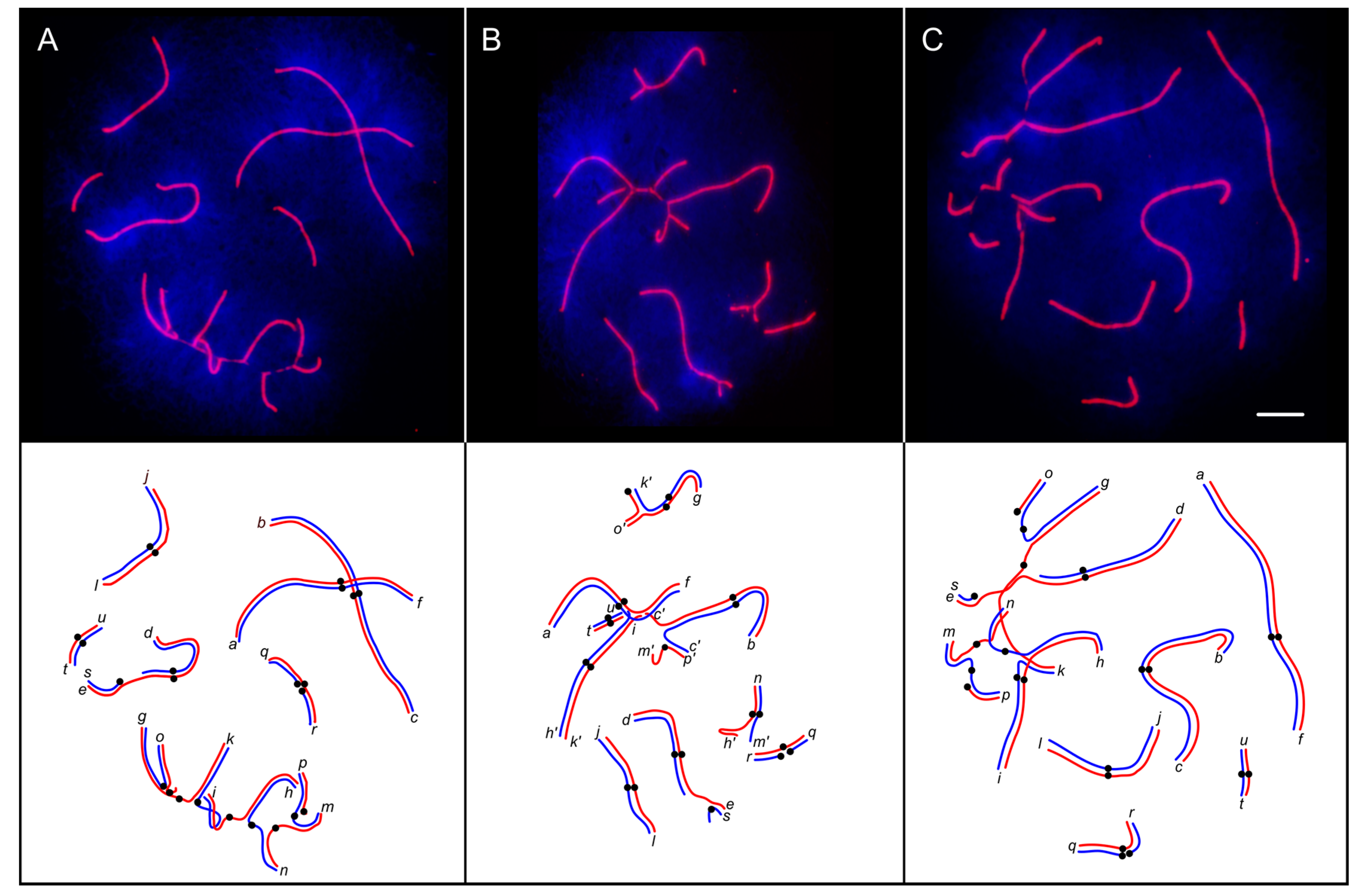
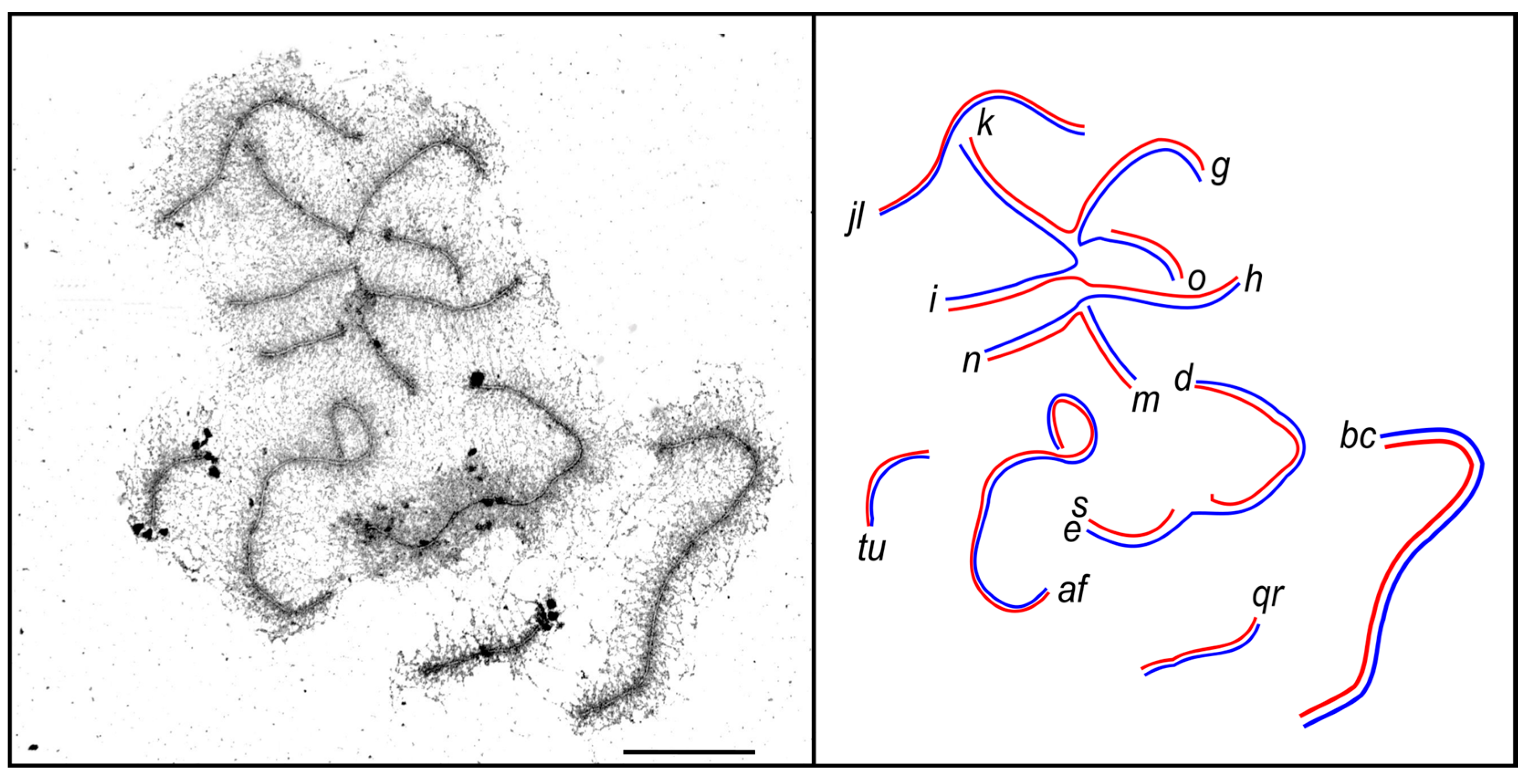
3.2. Synaptic Aberrations
3.3. MLH1 Foci Number and Distribution
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- King, M. Species Evolution: The Role of Chromosome Change; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Coyne, J.A.; Orr, H.A. Speciation; Sinauer Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Baker, R.J.; Bickham, J.W. Speciation by monobrachial centric fusions. Proc. Natl. Acad. Sci. USA 1986, 83, 8245–8248. [Google Scholar] [CrossRef] [PubMed]
- Borodin, P. Chromosomes and speciation. In Biosphere Origin and Evolution; Dobretsov, N., Kolchanov, N., Rozanov, A., Zavarzin, G., Eds.; Springer: Boston, MA, USA, 2008; pp. 315–325. [Google Scholar]
- Baird, A.B.; Hillis, D.M.; Patton, J.C.; Bickham, J.W. Speciation by monobrachial centric fusions: A test of the model using nuclear DNA sequences from the bat genus Rhogeessa. Mol. Phylogenet. Evol. 2009, 50, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, K.; Rieseberg, L. Chromosomal evolution and speciation: A recombination-based approach. New Phytol. 2004, 161, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, J.M.; Searle, J.B. The chromosome complement of Sorex granarius—The ancestral karyotype of the common shrew (Sorex araneus)? Heredity 1988, 61, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, J.M.; Borodin, P.M.; Fedyk, S.; Fredga, K.; Hausser, J.; Mishta, A.; Orlov, V.N.; Searle, J.B.; Volobouev, V.; Zima, J.; et al. The list of the chromosome races of the common shrew Sorex araneus (updated 2002). Mammalia 2003, 67, 169–178. [Google Scholar] [CrossRef]
- Searle, J.B. Chromosomal evolution: The case of Sorex araneus. In Evolution of Shrews; Wojcik, J.M., Wolsan, M., Eds.; Polish Academy of Sciences, Mammal Research Institute: Bialowieza, Poland, 1998; pp. 219–268. [Google Scholar]
- Polyakov, A.V.; Volobouev, V.T.; Borodin, P.M.; Searle, J.B. Karyotypic races of the common shrew (Sorex araneus) with exceptionally large ranges, the Novosibirsk and Tomsk races of Siberia. Hereditas 1996, 125, 109–115. [Google Scholar] [CrossRef]
- Polyakov, A.V.; White, T.A.; Jones, R.M.; Borodin, P.M.; Searle, J.B. Natural hybridization between extremely divergent chromosomal races of the common shrew (Sorex araneus, Soricidae, Soricomorpha): Hybrid zone in Siberia. J. Evol. Biol. 2011, 24, 1393–1402. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.; Basset, P.; Yannic, G.; Banaszek, A.; Borodin, P.M.; Bulatova, N.S.; Jadwiszczak, K.; Polyakov, A.V.; Ratkiewicz, M.; Searle, J.B.; et al. Chromosomal rearrangements do not seem to affect the gene flow in hybrid zones between karyotypic races of the common shrew (Sorex araneus). Evolution 2012, 66, 882–889. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, A.V; Onischenko, S.S.; Ilyashenko, V.B.; Searle, J.B.; Borodin, P.M. Morphometric difference between the Novosibirsk and Tomsk chromosome races of Sorex araneus in a zone of parapatry. Acta Theriol. 2002, 47, 381–387. [Google Scholar] [CrossRef]
- Polly, P.D.; Polyakov, A.V.; Ilyashenko, V.B.; Onischenko, S.S.; White, T.A.; Shchipanov, N.A.; Bulatova, N.S.; Pavlova, S.V.; Borodin, P.M.; Searle, J.B. Phenotypic variation across chromosomal hybrid zones of the common shrew (Sorex araneus) indicates reduced gene flow. PLoS ONE 2013, 8, e67455. [Google Scholar] [CrossRef] [PubMed]
- Capilla, L.; Garcia Caldés, M.; Ruiz-Herrera, A. Mammalian meiotic recombination: A toolbox for genome evolution. Cytogenet. Genome Res. 2016, 150, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.K.; Reeves, A.; Webb, L.M.; Ashley, T. Distribution of crossing over on mouse synaptonemal complexes using immunofluorescent localization of MLH1 protein. Genetics 1999, 151, 1569–1579. [Google Scholar] [PubMed]
- Segura, J.; Ferretti, L.; Ramos-Onsins, S.; Capilla, L.; Farré, M.; Reis, F.; Oliver-Bonet, M.; Fernández-Bellón, H.; Garcia, F.; Garcia-Caldés, M.; et al. Evolution of recombination in eutherian mammals: Insights into mechanisms that affect recombination rates and crossover interference. Proc. R. Soc. B. 2013, 280, 1945. [Google Scholar] [CrossRef] [PubMed]
- Borodin, P.M.; Karamysheva, T.V.; Belonogova, N.M.; Torgasheva, A.A.; Rubtsov, N.B.; Searle, J.B. Recombination map of the common shrew, Sorex araneus (Eulipotyphla, Mammalia). Genetics 2008, 178, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Searle, J.B. Meiotic studies of Robertsonian heterozygotes from natural populations of the common shrew, Sorex araneus L. Cytogenet. Genome Res. 1986, 41, 154–162. [Google Scholar] [CrossRef]
- Seabright, M. A rapid banding technique for human chromosomes. Lancet 1971, 2, 971–972. [Google Scholar] [CrossRef]
- Searle, J.B.; Fedyk, S.; Fredga, K.; Hausser, J.; Volobouev, V.T. Nomenclature for the chromosomes of the common shrew (Sorex araneus). Mém. Soc. Vaud. Sci. Nat. 1991, 19, 13–22. [Google Scholar] [CrossRef]
- Peters, A.H.; Plug, A.W.; van Vugt, M.J.; de Boer, P. A drying-down technique for the spreading of mammalian meiocytes from the male and female germline. Chromosom. Res. 1997, 5, 66–68. [Google Scholar] [CrossRef]
- Howell, W.M.; Black, D.A. Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: A 1-step method. Experientia 1980, 36, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Belonogova, N.M.; Karamysheva, T.V.; Biltueva, L.S.; Perepelov, E.A.; Minina, J.M.; Polyakov, A.V.; Zhdanova, N.S.; Rubtsov, N.B.; Searle, J.B.; Borodin, P.M. Identification of all pachytene bivalents in the common shrew using DAPI-staining of synaptonemal complex spreads. Chromosome Res. 2006, 14, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Reeves, A. MicroMeasure: A new computer program for the collection and analysis of cytogenetic data. Genome 2001, 44, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Borodin, P.M. Synaptonemal complexes of the common shrew, Sorex araneus L., in spermatocyte spreads. Cytogenet. Cell Genet. 1991, 56, 61–62. [Google Scholar] [CrossRef] [PubMed]
- Pack, S.D.; Borodin, P.M.; Serov, O.L.; Searle, J.B. The X-autosome translocation in the common shrew (Sorex araneus L.): Late replication in female somatic cells and pairing in male meiosis. Chromosoma 1992, 102, 355–360. [Google Scholar] [CrossRef]
- Matveevsky, S.N.; Pavlova, S.V.; Acaeva, M.M.; Kolomiets, O.L. Synaptonemal complex analysis of interracial hybrids between the Moscow and Neroosa chromosomal races of the common shrew Sorex araneus showing regular formation of a complex meiotic configuration (ring-of-four). Comp. Cytogenet. 2012, 6, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Burgoyne, P.S.; Mahadevaiah, S.K.; Turner, J.M. The consequences of asynapsis for mammalian meiosis. Nat. Rev. Genet. 2009, 10, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Li, X.C.; Barringer, B.C.; Barbash, D.A. The pachytene checkpoint and its relationship to evolutionary patterns of polyploidization and hybrid sterility. Heredity 2009, 102, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Roeder, G.S.; Bailis, J.M. The pachytene checkpoint. Trends Genet. 2000, 16, 395–403. [Google Scholar] [CrossRef]
- Tung, K.S.; Hong, E.J.; Roeder, G.S. The pachytene checkpoint prevents accumulation and phosphorylation of the meiosis-specific transcription factor Ndt80. Proc. Natl. Acad. Sci. USA 2000, 97, 12187–12192. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, S.V.; Bulatova, N.S.; Shchipanov, N.A. Cytogenetic control of a hybrid zone between two Sorex araneus chromosome races before breeding season. Russ. J. Genet. 2007, 43, 1357–1363. [Google Scholar] [CrossRef]
- Pavlova, S.V; Kolomiets, O.L.; Bulatova, N.S.; Searle, J.B. Demonstration of a WART in a hybrid zone of the common shrew (Sorex araneus Linnaeus, 1758). Comp. Cytogenet. 2008, 2, 115–120. [Google Scholar]
- Searle, J.B. Chromosomal hybrid zones in eutherian mammals. In Hybrid Zones and the Evolutionary Process; Harrison, R.G., Ed.; Oxford University Press: New York, NY, USA, 1993; pp. 309–353. [Google Scholar]
- Narain, Y.; Fredga, K. Spermatogenesis in common shrews, Sorex araneus, from a hybrid zone with extensive Robertsonian polymorphism. Cytogenet. Cell Genet. 1998, 80, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Narain, Y.; Fredga, K. Meiosis and fertility in common shrews, Sorex araneus, from a chromosomal hybrid zone in central Sweden. Cytogenet. Cell Genet. 1997, 78, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Jadwiszczak, K.A.; Banaszek, A. Fertility in the male common shrews, Sorex araneus, from the extremely narrow hybrid zone between chromosome races. Mamm. Biol. 2006, 71, 257–267. [Google Scholar] [CrossRef]
- Polyakov, A.V; Panov, V.V. Study of male—Mediated gene flow across a hybrid zone in the common shrew (Sorex araneus) using Y chromosome. Comp. Cytogenet. 2017, 11, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Hunt, P.A.; Hassold, T.J. Sex matters in meiosis. Science 2002, 296, 2181–2183. [Google Scholar] [CrossRef] [PubMed]
- Schwander, T.; Libbrecht, R.; Keller, L. Supergenes and complex phenotypes. Curr. Biol. 2014, 24, R288–R294. [Google Scholar] [CrossRef] [PubMed]
- Franchini, P.; Colangelo, P.; Solano, E.; Capanna, E.; Verheyen, E.; Castiglia, R. Reduced gene flow at pericentromeric loci in a hybrid zone involving chromosomal races of the house mouse Mus musculus domesticus. Evolution 2010, 64, 2020–2032. [Google Scholar] [PubMed]
- Giménez, M.D.; White, T.A.; Hauffe, H.C.; Panithanarak, T.; Searle, J.B. Understanding the basis of diminished gene flow between hybridizing chromosome races of the house mouse. Evolution 2013, 67, 1446–1462. [Google Scholar] [CrossRef] [PubMed]
- Merico, V.; Giménez, M.D.; Vasco, C.; Zuccotti, M.; Searle, J.B.; Hauffe, H.C.; Garagna, S. Chromosomal speciation in mice: A cytogenetic analysis of recombination. Chromosom. Res. 2013, 21, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, A.V; Volobouev, V.T.; Aniskin, V.M.; Zima, J.; Searles, J.B.; Borodin, P.M. Altitudinal partitioning of two chromosome races of the common shrew (Sorex araneus) in West Siberia. Mammalia 2003, 67, 201–207. [Google Scholar] [CrossRef]

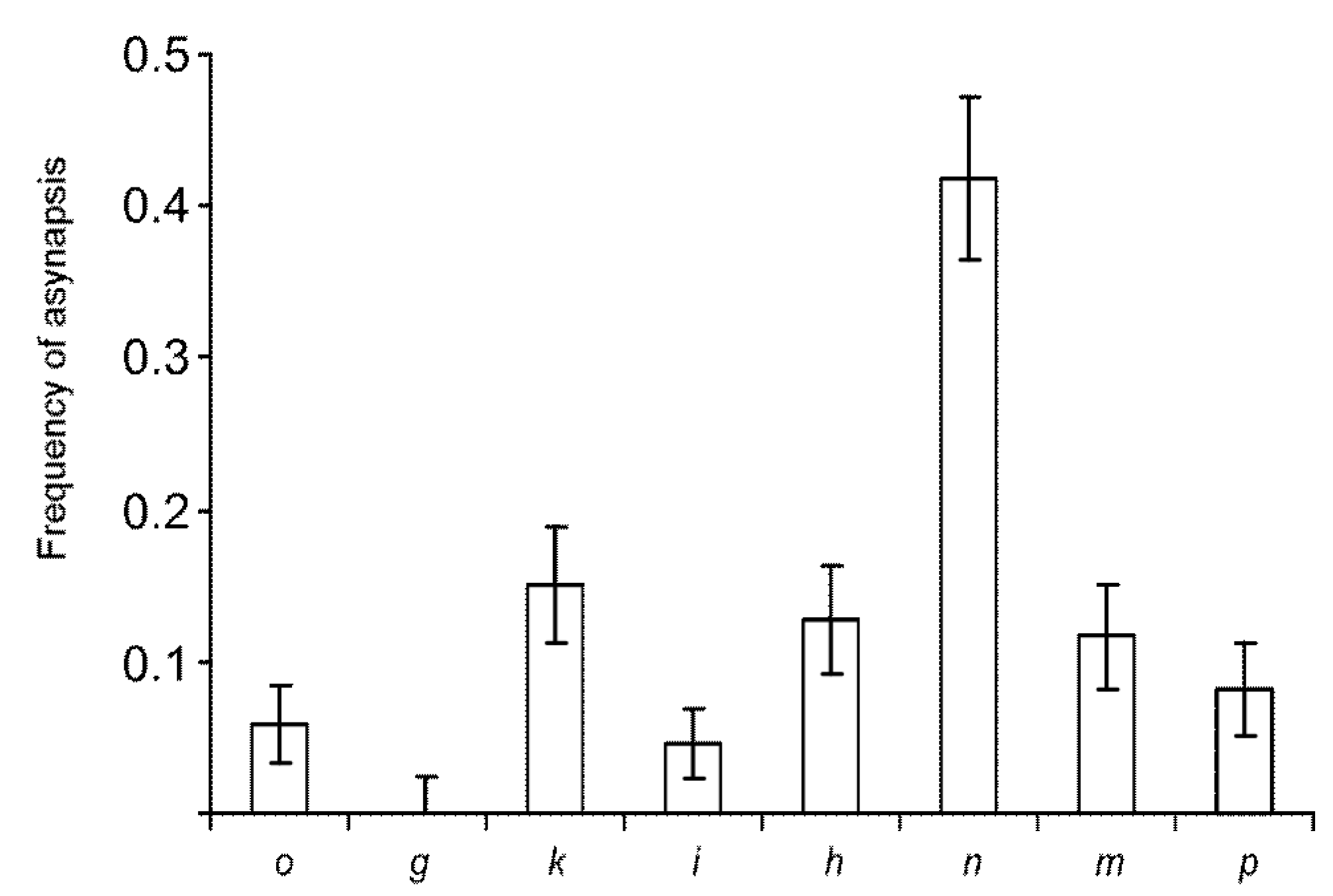

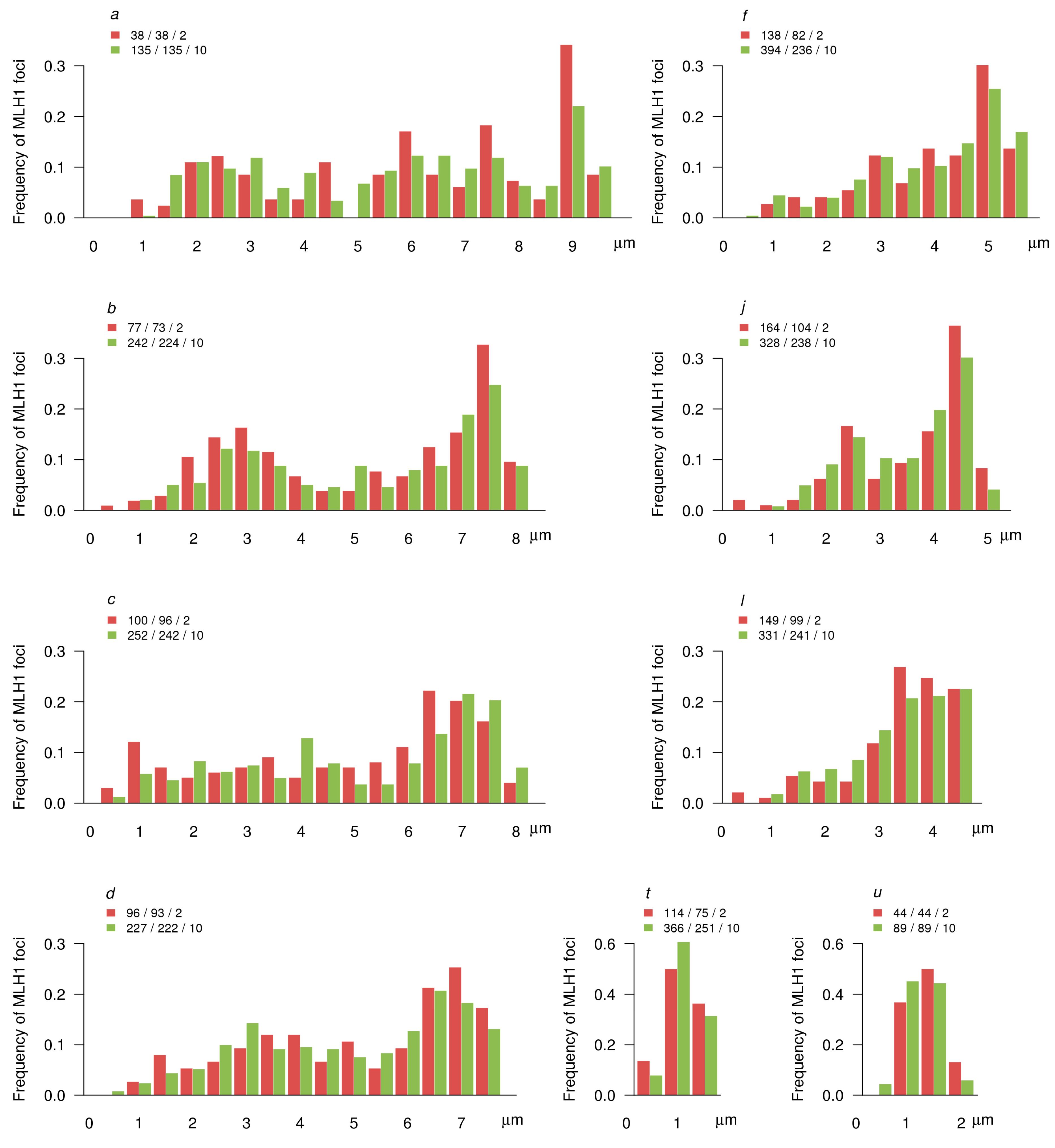
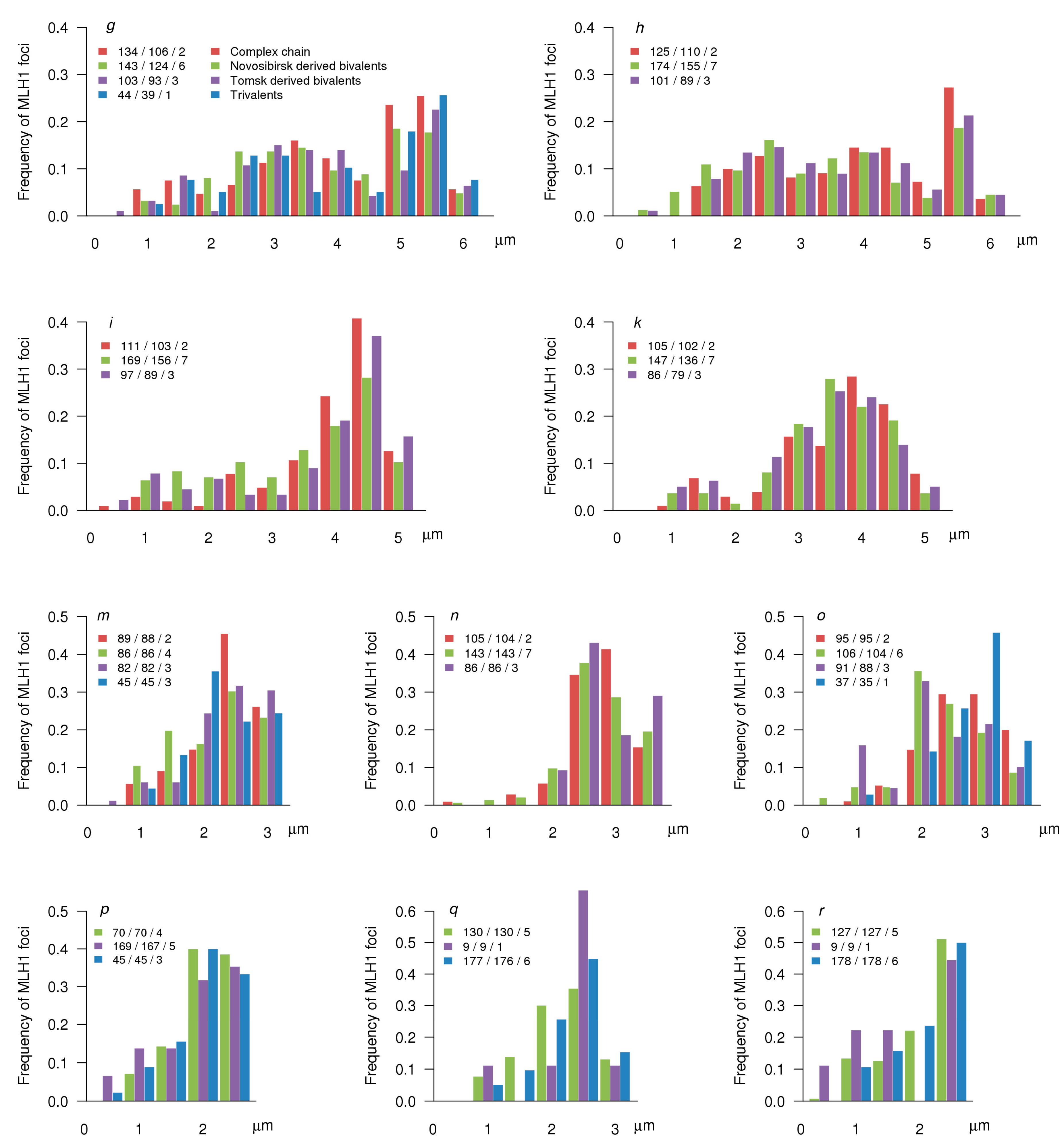
| ID | Synaptic Type (Autosomes) | Diagnostic Chromosome Set | Karyotype | 2n | Number of Cells Scored | Frequency of Cells with Synaptic Aberrations |
|---|---|---|---|---|---|---|
| 1 | Bivalents only | Novosibirsk | go, hn, ik, mp, qr | 21 | 29 | 0.07 |
| 2 | Bivalents only | Novosibirsk | go, hn, ik, mp, qr | 21 | 67 | 0.03 |
| 3 | Bivalents only | Novosibirsk | go, hn, ik, mp, qr | 21 | 31 | 0.06 |
| 4 | Bivalents only | Tomsk | gk, hi, mn, o, p, qr | 23 | 76 | 0.00 |
| 5 | Bivalents only | Tomsk | gk, hi, mn, o, p, q, r | 25 | 13 | 0.23 |
| 6 | Single trivalents CIII | Tomsk | gk, hi, mn, o, p, q/r | 24 | 74 | 0.07 |
| 7 | Single trivalents CIII | Novosibirsk | go, hn, ik, mp, q/r | 22 | 44 | 0.00 |
| 8 | Single trivalents CIII | Novosibirsk | g/o, hn, ik, mp, qr | 22 | 21 | 0.10 |
| 9 | Single trivalents CIII | Novosibirsk | go, hn, ik, m/p, qr | 22 | 78 | 0.03 |
| 10 | Double trivalents CIII | Novosibirsk | go, hn, ik, m/p, q/r | 23 | 21 | 0.10 |
| 11 | Double trivalents CIII | Novosibirsk | go, hn, ik, m/p, q/r | 23 | 19 | 0.00 |
| 12 | Double trivalents CIII | Novosibirsk | g/o, hn, ik, mp, q/r | 23 | 94 | 0.10 |
| 13 | Double trivalents CIII | Novosibirsk | go, hn, ik, m/p, q/r | 23 | 12 | 0.33 |
| 14 | Complex chains CVIII | Hybrid | o/og/gk/ki/ih/hn/nm/m, p, qr | 23 | 69 | 0.48 |
| 15 | Complex chains CVIII + CIII | Hybrid | o/og/gk/ki/ih/hn/nm/m, p, q/r | 24 | 39 | 0.44 |
| 16 | Complex chains CIX + CIII | Hybrid | o/og/gk/ki/ih/hn/nm/mp/p, q/r | 23 | 126 | 0.79 |
| ID | Synaptic Type (Autosomes) | Karyotype | Number of Cells Scored | Average Number of MLH1 Foci per Cell ± S.D. | Expected Number of Obligate Chiasmata |
|---|---|---|---|---|---|
| 1 | Bivalents only | go, hn, ik, mp, qr | 6 | 18.7 ± 3.1 | 11 |
| 2 | Bivalents only | go, hn, ik, mp, qr | 18 | 18.6 ± 3.3 | 11 |
| 4 | Bivalents only | gk, hi, mn, o, p, qr | 6 | 19.5 ± 2.4 | 12 |
| 5 | Bivalents only | gk, hi, mn, o, p, q, r | 7 | 19.8 ± 2.1 | 13 |
| 6 | Single trivalents CIII | gk, hi, mn, o, p, q/r | 15 | 22.1 ± 3.7 | 13 |
| 10 | Double trivalents CIII | go, hn, ik, m/p, q/r | 14 | 20.6 ± 3.4 | 13 |
| 12 | Double trivalents CIII | go, hn, ik, m/p, q/r | 27 | 19.1 ± 1.9 | 13 |
| 14 | Complex chains CVIII | o/og/gk/ki/ih/hn/nm/m, p, qr | 19 | 21.1 ± 2.5 | 15 |
| 15 | Complex chains CVIII + CIII | o/og/gk/ki/ih/hn/nm/m, p, q/r | 17 | 22.1 ± 2.8 | 16 |
| 16 | Complex chains CIX + CIII | o/og/gk/ki/ih/hn/nm/mp/p, q/r | 5 | 14.1 ± 2.5 | 16 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belonogova, N.M.; Polyakov, A.V.; Karamysheva, T.V.; Torgasheva, A.A.; Searle, J.B.; Borodin, P.M. Chromosome Synapsis and Recombination in Male Hybrids between Two Chromosome Races of the Common Shrew (Sorex araneus L., Soricidae, Eulipotyphla). Genes 2017, 8, 282. https://doi.org/10.3390/genes8100282
Belonogova NM, Polyakov AV, Karamysheva TV, Torgasheva AA, Searle JB, Borodin PM. Chromosome Synapsis and Recombination in Male Hybrids between Two Chromosome Races of the Common Shrew (Sorex araneus L., Soricidae, Eulipotyphla). Genes. 2017; 8(10):282. https://doi.org/10.3390/genes8100282
Chicago/Turabian StyleBelonogova, Nadezhda M., Andrei V. Polyakov, Tatyana V. Karamysheva, Anna A. Torgasheva, Jeremy B. Searle, and Pavel M. Borodin. 2017. "Chromosome Synapsis and Recombination in Male Hybrids between Two Chromosome Races of the Common Shrew (Sorex araneus L., Soricidae, Eulipotyphla)" Genes 8, no. 10: 282. https://doi.org/10.3390/genes8100282
APA StyleBelonogova, N. M., Polyakov, A. V., Karamysheva, T. V., Torgasheva, A. A., Searle, J. B., & Borodin, P. M. (2017). Chromosome Synapsis and Recombination in Male Hybrids between Two Chromosome Races of the Common Shrew (Sorex araneus L., Soricidae, Eulipotyphla). Genes, 8(10), 282. https://doi.org/10.3390/genes8100282