Genetic Changes in Mastocytes and Their Significance in Mast Cell Tumor Prognosis and Treatment
Abstract
1. Introduction
2. Characteristics of Mastocytes
- anaphylactic exocytosis (degranulation without de novo synthesis), observed after treatment with C3a and C5a complement peptides (on complement receptors), snake venom, UV, acrolein, titanium nanoparticles;
- selective exocytosis (degranulation) of cellular granules can occur after the action of specific stimulators, including antigen or IgE on FcεR1 (histamine is released), neuropeptides on NKR (cytokines, chemokines, and 5HT are released), 48/80 on Mrgprx2 (cytokines and chemokines are released), cathelicidin on G-protein-coupled receptor (GPCR, releasing histamine), defensins on GPCR (releasing histamine), pleurocidin on GPCR (releasing cytokines and chemokines), A23187 (releasing cytokines and chemokines), morphine and codeine on opioid receptor (releasing cytokines, chemokines, and hexosaminidase), and NGF on Trk receptor (releasing histamine and PGE2);
- constitutive exocytosis is associated with granule release without degranulation after treatment with zymosan on TLR2 (releasing GM-CSF, Il-1β), poly (I-C) on TLR (releasing cytokines), LPS on TLR4 and CD14 (releasing cytokines and chemokines), SCF on MAPK (releasing cytokines), and lectin on TIM3 (releasing cytokines);
- without degranulation and without de novo synthesis (exocytosis of exosomes). Exosomes are vesicles measuring 30 nm–100 nm in diameter formed in secretory granules. These vesicles penetrate the cell membrane, pass outside the cell, and after transferring in body fluids, they are engulfed by other cells. Exosomes are important for intercellular communication. They contain many proteins, up to 1000 different mRNAs, and more than 100 microRNA molecules [17,18,19].
Mast Cell Ligands and Receptors
3. Mast Cell Activation Syndrome
3.1. Systemic Mastocytosis
- cutaneous mastocytosis (CM)—most common in children—manifesting as urticaria pigmentosa as a diffuse or limited form of mastocytosis. Urticaria pigmentosa has good prognosis and usually resolves itself spontaneously;
- localized mastocytoma [95].
Targeted Therapies
3.2. Canine Systemic Mastocytosis
4. Mastocytoma
4.1. Diagnosis and Prognosis
4.2. Mastocytoma Cell Proteins
4.3. Mutations in Mastocytoma Cells
4.4. Differences in Benign and Malignant Forms of MCT (According to Kiupel)
4.5. Metastasis Formation in Mastocytoma
4.6. The Role of microRNAs
4.7. Treatment
4.7.1. Surgical Treatment
4.7.2. Pharmacological Treatment
4.7.3. Radiotherapy
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 5HT | 5-hydroxytryptamine |
| ALK | anaplastic lymphoma kinase |
| ASM | aggressive systemic mastocytosis |
| ASMAs | anti-smooth muscle antibodies |
| B/MCP | basophil and MC progenitor |
| BBMC | baboon bone marrow cells |
| BNDF | brain-derived neurotrophic factor |
| CAA | cancer-associated adipocytes |
| CAF | cancer-associated fibroblast |
| CAFs | cancer-associated fibroblasts |
| CCL | C-C motif ligand |
| CM | cutaneous mastocytosis |
| CMP | common myeloid progenitor |
| CNV | copy number variants |
| CPA3 | carboxypeptidase A3 |
| CSF-1R | colony-stimulating factor receptor |
| CSPs | competence sensing peptides |
| CTMCs | connective tissue mast cells |
| DOCK3 | dedicator of cytokinesis 3 |
| DSCAM | Down syndrome cell adhesion molecule |
| DSP | desmoplakin |
| dsRNA | double-stranded ribonucleic acid |
| ECM | extracellular matrix |
| EMP | erythroid–myeloid progenitors |
| EV | extracellular vesicles |
| FGF | fibroblast growth factor |
| FNAB | fine-needle aspiration biopsy |
| FSDMC | fetal-skin-derived mast cells |
| GM-CSF | granulocyte-macrophage colony stimulating factor |
| GMP | granulocyte-macrophage progenitor |
| GPCR | G-protein-coupled receptor |
| HalfaT | hereditary alpha-tryptasemia |
| HDM | house dust mites |
| HDP | heme detoxification protein |
| HemASC | hematopoietic adult stem cells |
| HR1 | histamine H1 receptor |
| hSM | human systemic mastocytosis |
| HSP | heat shock protein |
| Il | interleukin |
| ISM | indolent systemic mastocytosis |
| ITAM | immunoreceptor tyrosine-based activation motif |
| ITD | internal tandem duplication |
| JM domain | juxtamembrane domain |
| LPS | lipopolysaccharide |
| LYST | lysosomal trafficking regulator protein |
| MAPK | mitogen-activated protein kinase |
| MCAS | mast cell activation syndrome |
| MCL | mast cell leukemia |
| MCP4 | monocyte chemoattractant protein 4 |
| MCs | mast cells |
| MCT | mastocytoma |
| MDR1 | multi-drug resistance protein 1 |
| MDSCs | myeloid-derived suppressor cells |
| miRNA | micro-ribonucleic acid |
| MITF | microphthalmia-associated transcription factor |
| MMCs | mucosal mast cells |
| MMTV | mouse mammary tumor virus |
| MPP | multipotent progenitor |
| NF3 | nuclear factor 3 |
| NF4 | nuclear factor 4 |
| NGF | nerve growth factor |
| NGFR | nerve growth factor receptor |
| NK | natural killer |
| NKR | natural killer receptor |
| PAF | platelet-activating factor |
| PAMPs | pathogen-associated molecular patterns |
| PCDMC | peritoneal-cell-derived mast cells |
| PCNA | proliferating cell nuclear antigen |
| PD1/CD279 | programmed cell death protein |
| PDGF | platelet-derived growth factor |
| PDGFR | platelet-derived growth factor receptor |
| PD-L1 | programmed cell death ligand 1 |
| PEG-ZnPP | pegylated zinc protoporphyrin |
| PGE2 | prostaglandin E2 |
| PGN | peptidoglycan |
| PI3K | phosphoinositide 3-kinase |
| PKP1 | plakophilin 1 |
| PLC-γ | phospholipase C gamma |
| PMNs | premetastatic niches |
| QSMs | quorum sensing molecules |
| SCF | stem cell factor |
| SDPR | serum deprivation response |
| sEV | small extracellular vesicle |
| SM | systemic mastocytosis |
| SNARE | sensitive factor attachment protein receptor |
| ssRNA | single-stranded ribonucleic acid |
| TAFs | tumor-associated fibroblasts |
| TAM | tumor-associated macrophages |
| TGF | transforming growth factor |
| TIM3 | T-cell immunoglobulin and mucin domain 3 |
| TLRs | Toll-like receptors |
| TNF | tumor necrosis factor |
| Trk | tropomyosin receptor kinase |
| TSLP | thymic stromal lymphopoietin |
| TSLPR | thymic stromal lymphopoietin receptor |
| VEGF | vascular endothelial growth factor |
| VEGFR | vascular endothelial growth factor receptor |
| VIP | vasoactive intestinal peptide |
References
- Cohen, D.; Reif, J.S.; Brodey, R.S.; Keiser, H. Epidemiological analysis of the most prevalent sites and types of canine neoplasia observed in a veterinary hospital. Cancer Res. 1974, 34, 2859–2868. Available online: http://www.ncbi.nlm.nih.gov/pubmed/4529096 (accessed on 15 October 2023). [PubMed]
- Priester, W.A. Skin Tumors in Domestic Animals. Data from 12 United States and Canadian Colleges of Veterinary Medicine 2. JNCI J. Natl. Cancer Inst. 1973, 50, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Crivellato, E.; Beltrami, C.A.; Mallardi, F.; Ribatti, D. Paul Ehrlich’s doctoral thesis: A milestone in the study of mast cells. Br. J. Haematol. 2003, 123, 19–21. [Google Scholar] [CrossRef] [PubMed]
- Yanase, Y.; Matsubara, D.; Takahagi, S.; Tanaka, A.; Ozawa, K.; Hide, M. Basophil Characteristics as a Marker of the Pathogenesis of Chronic Spontaneous Urticaria in Relation to the Coagulation and Complement Systems. Int. J. Mol. Sci. 2023, 24, 10320. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.; Silva, E.; Scroggie, A.E.; Fidlar, E.; Jaques, L.B. Liberation of Histamine and Heparin by Peptone from the Isolated Dog’s Liver. Exp. Biol. Med. 1947, 64, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Riley, J.F.; West, G.B. Histamine in tissue mast cells. J. Physiol. 1952, 117, 72P–73P. Available online: http://www.ncbi.nlm.nih.gov/pubmed/12991263 (accessed on 20 October 2023). [CrossRef] [PubMed]
- von KöCkritz-Blickwede, M.; Goldmann, O.; Thulin, P.; Heinemann, K.; Norrby-Teglund, A.; Rohde, M.; Medina, E. Phagocytosis-independent antimicrobial activity of mast cells by means of extracellular trap formation. Blood 2008, 111, 3070–3080. [Google Scholar] [CrossRef] [PubMed]
- Sobiepanek, A.; Kuryk, Ł.; Garofalo, M.; Kumar, S.; Baran, J.; Musolf, P.; Siebenhaar, F.; Fluhr, J.W.; Kobiela, T.; Plasenzotti, R.; et al. The Multifaceted Roles of Mast Cells in Immune Homeostasis, Infections and Cancers. Int. J. Mol. Sci. 2022, 23, 2249. [Google Scholar] [CrossRef]
- Dong, J.; Chen, L.; Zhang, Y.; Jayaswal, N.; Mezghani, I.; Zhang, W.; Veves, A. Mast Cells in Diabetes and Diabetic Wound Healing. Adv. Ther. 2020, 37, 4519–4537. [Google Scholar] [CrossRef]
- Bacci, S. Fine Regulation during Wound Healing by Mast Cells, a Physiological Role Not Yet Clarified. Int. J. Mol. Sci. 2022, 23, 1820. [Google Scholar] [CrossRef]
- Ryan, J.J.; Kashyap, M.; Bailey, D.; Kennedy, S.; Speiran, K.; Brenzovich, J.; Barnstein, B.; Oskeritzian, C.; Gomez, G. Mast Cell Homeostasis: A Fundamental Aspect of Allergic Disease. Crit. Rev. Immunol. 2007, 27, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Grimbaldeston, M.; Tsai, M. Immunomodulatory mast cells: Negative, as well as positive, regulators of immunity. Nat. Rev. Immunol. 2008, 8, 478–486. [Google Scholar] [CrossRef] [PubMed]
- da Silva, E.Z.M.; Jamur, M.C.; Oliver, C. Mast Cell Function. J. Histochem. Cytochem. 2014, 62, 698–738. [Google Scholar] [CrossRef] [PubMed]
- Moon, T.C.; Befus, A.D.; Kulka, M. Mast Cell Mediators: Their Differential Release and the Secretory Pathways Involved. Front. Immunol. 2014, 5, 569. [Google Scholar] [CrossRef] [PubMed]
- Artuc, M.; Hermes, B.; Stckelings, U.M.; Grützkau, A.; Henz, B.M. Mast cells and their mediators in cutaneous wound healing—Active participants or innocent bystanders? Exp. Dermatol. 1999, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lichterman, J.N.; Reddy, S.M. Mast Cells: A New Frontier for Cancer Immunotherapy. Cells 2021, 10, 1270. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Simpson, R.J.; Kalra, H.; Mathivanan, S. ExoCarta as a resource for exosomal research. J. Extracell. Vesicles 2012, 1, 18374. [Google Scholar] [CrossRef] [PubMed]
- Skokos, D.; Botros, H.G.; Demeure, C.; Morin, J.; Peronet, R.; Birkenmeier, G.; Boudaly, S.; Mécheri, S. Mast Cell-Derived Exosomes Induce Phenotypic and Functional Maturation of Dendritic Cells and Elicit Specific Immune Responses In Vivo. J. Immunol. 2003, 170, 3037–3045. [Google Scholar] [CrossRef]
- Elstak, E.D.; Neeft, M.; Nehme, N.T.; Voortman, J.; Cheung, M.; Goodarzifard, M.; Gerritsen, H.C.; Henegouwen, P.M.P.v.B.E.; Callebaut, I.; Basile, G.d.S.; et al. The munc13-4–rab27 complex is specifically required for tethering secretory lysosomes at the plasma membrane. Blood 2011, 118, 1570–1578. [Google Scholar] [CrossRef]
- Tadokoro, S.; Nakanishi, M.; Hirashima, N. Complexin II facilitates exocytotic release in mast cells by enhancing Ca2+ sensitivity of the fusion process. J. Cell Sci. 2005, 118, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Riedel, D.; Antonin, W.; Fernandez-Chacon, R.; de Toledo, G.A.; Jo, T.; Geppert, M.; Valentijn, J.A.; Valentijn, K.; Jamieson, J.D.; Südhof, T.C.; et al. Rab3D Is Not Required for Exocrine Exocytosis but for Maintenance of Normally Sized Secretory Granules. Mol. Cell. Biol. 2002, 22, 6487–6497. [Google Scholar] [CrossRef]
- Melicoff, E.; Sansores-Garcia, L.; Gomez, A.; Moreira, D.C.; Datta, P.; Thakur, P.; Petrova, Y.; Siddiqi, T.; Murthy, J.N.; Dickey, B.F.; et al. Synaptotagmin-2 Controls Regulated Exocytosis but Not Other Secretory Responses of Mast Cells. J. Biol. Chem. 2009, 284, 19445–19451. [Google Scholar] [CrossRef]
- Baier, A.; E Ndoh, V.N.; Lacy, P.; Eitzen, G. Rac1 and Rac2 control distinct events during antigen-stimulated mast cell exocytosis. J. Leukoc. Biol. 2014, 95, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, K.; Tanaka, Y.; Uruno, T.; Duan, X.; Harada, Y.; Sanematsu, F.; Yamamura, K.; Terasawa, M.; Nishikimi, A.; Côté, J.-F.; et al. DOCK5 functions as a key signaling adaptor that links FcεRI signals to microtubule dynamics during mast cell degranulation. J. Exp. Med. 2014, 211, 1407–1419. [Google Scholar] [CrossRef] [PubMed]
- Woska, J.R.; Gillespie, M.E. SNARE complex-mediated degranulation in mast cells. J. Cell. Mol. Med. 2012, 16, 649–656. [Google Scholar] [CrossRef]
- Li, Z.; Liu, S.; Xu, J.; Zhang, X.; Han, D.; Liu, J.; Xia, M.; Yi, L.; Shen, Q.; Xu, S.; et al. Adult Connective Tissue-Resident Mast Cells Originate from Late Erythro-Myeloid Progenitors. Immunity 2018, 49, 640–653.e5. [Google Scholar] [CrossRef] [PubMed]
- Huang, E.; Nocka, K.; Beier, D.R.; Chu, T.-Y.; Buck, J.; Lahm, H.-W.; Wellner, D.; Leder, P.; Besmer, P. The hematopoietic growth factor KL is encoded by the SI locus and is the ligand of the c-kit receptor, the gene product of the W locus. Cell 1990, 63, 225–233. [Google Scholar] [CrossRef]
- Mendoza, R.P.; Fudge, D.H.; Brown, J.M. Cellular Energetics of Mast Cell Development and Activation. Cells 2021, 10, 524. [Google Scholar] [CrossRef]
- MacDonald, C.A.; Qian, H.; Pundir, P.; Kulka, M. Sodium butyrate supresses malignant human mast cell proliferation, downregulates expression of KIT and promotes differentiation. Front. Allergy 2023, 4, 1109717. [Google Scholar] [CrossRef]
- Welle, M. Development, significance, and heterogeneity of mast cells with particular regard to the mast cell-specific proteases chymase and tryptase. J. Leukoc. Biol. 1997, 61, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Pejler, G.; Rönnberg, E.; Waern, I.; Wernersson, S. Mast cell proteases: Multifaceted regulators of inflammatory disease. Blood 2010, 115, 4981–4990. [Google Scholar] [CrossRef] [PubMed]
- A Irani, A.; Schechter, N.M.; Craig, S.S.; DeBlois, G.; Schwartz, L.B. Two types of human mast cells that have distinct neutral protease compositions. Proc. Natl. Acad. Sci. USA 1986, 83, 4464–4468. [Google Scholar] [CrossRef]
- Schwartz, L.B. Analysis of MC(T) and MC(TC) mast cells in tissue. Methods Mol. Biol. 2006, 315, 53–62. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16110148 (accessed on 20 October 2023).
- Abonia, J.P.; Blanchard, C.; Butz, B.B.; Rainey, H.F.; Collins, M.H.; Stringer, K.; Putnam, P.E.; Rothenberg, M.E. Involvement of mast cells in eosinophilic esophagitis. J. Allergy Clin. Immunol. 2010, 126, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Irani, A.M.; Craig, S.S.; DeBlois, G.; Elson, C.O.; Schechter, N.M.; Schwartz, L.B. Deficiency of the tryptase-positive, chymase-negative mast cell type in gastrointestinal mucosa of patients with defective T lymphocyte function. J. Immunol. 1987, 138, 4381–4386. Available online: http://www.ncbi.nlm.nih.gov/pubmed/3295046 (accessed on 20 October 2023). [CrossRef] [PubMed]
- Gilfillan, A.M.; Austin, S.J.; Metcalfe, D.D. Mast Cell Biology: Introduction and Overview. Adv. Exp. Med. Biol. 2011, 716, 2–12. [Google Scholar] [PubMed]
- Dahlin, J.S.; Hallgren, J. Mast cell progenitors: Origin, development and migration to tissues. Mol. Immunol. 2015, 63, 9–17. [Google Scholar] [CrossRef]
- Plum, T.; Wang, X.; Rettel, M.; Krijgsveld, J.; Feyerabend, T.B.; Rodewald, H.-R. Human Mast Cell Proteome Reveals Unique Lineage, Putative Functions, and Structural Basis for Cell Ablation. Immunity 2020, 52, 404–416.e5. [Google Scholar] [CrossRef]
- Kanagaratham, C.; El Ansari, Y.S.; Lewis, O.L.; Oettgen, H.C. IgE and IgG Antibodies as Regulators of Mast Cell and Basophil Functions in Food Allergy. Front. Immunol. 2020, 11, 603050. [Google Scholar] [CrossRef]
- Méndez-Enríquez, E.; Hallgren, J. Mast Cells and Their Progenitors in Allergic Asthma. Front. Immunol. 2019, 10, 821. [Google Scholar] [CrossRef] [PubMed]
- Oldford, S.A.; Marshall, J.S. Mast cells as targets for immunotherapy of solid tumors. Mol. Immunol. 2015, 63, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Yanase, Y.; Takahagi, S.; Ozawa, K.; Hide, M. The Role of Coagulation and Complement Factors for Mast Cell Activation in the Pathogenesis of Chronic Spontaneous Urticaria. Cells 2021, 10, 1759. [Google Scholar] [CrossRef] [PubMed]
- Ikuno, T.; Ito, S.; Inoue, T. Human induced pluripotent stem cell-derived mast cells useful for in vitro mast cell activation assay exhibiting phenotypes and morphological characteristics of human mast cells. J. Toxicol. Sci. 2019, 44, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Valent, P.; Akin, C.; Hartmann, K.; Nilsson, G.; Reiter, A.; Hermine, O.; Sotlar, K.; Sperr, W.R.; Escribano, L.; George, T.I.; et al. Mast cells as a unique hematopoietic lineage and cell system: From Paul Ehrlich’s visions to precision medicine concepts. Theranostics 2020, 10, 10743–10768. [Google Scholar] [CrossRef] [PubMed]
- Hubálek, Z.; Savage, H.; Halouzka, J.; Juăicová, Z.; Sanogo, Y.; Lusk, S. West Nile Virus Investigations in South Moravia, Czechland. Viral Immunol. 2000, 13, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Lamas, D.J.M.; Nicoud, M.B.; A Sterle, H.; A Cremaschi, G.; A Medina, V. Histamine: A potential cytoprotective agent to improve cancer therapy? Cell Death Dis. 2015, 6, e2029. [Google Scholar] [CrossRef]
- Blair, R.J.; Meng, H.; Marchese, M.J.; Ren, S.; Schwartz, L.B.; Tonnesen, M.G.; Gruber, B.L. Human mast cells stimulate vascular tube formation. Tryptase is a novel, potent angiogenic factor. J. Clin. Investig. 1997, 99, 2691–2700. [Google Scholar] [CrossRef]
- Scott, D.W.; Gascoyne, R.D. The tumour microenvironment in B cell lymphomas. Nat. Rev. Cancer 2014, 14, 517–534. [Google Scholar] [CrossRef]
- Conti, P.; Caraffa, A.; Tetè, G.; Gallenga, C.E.; Ross, R.; Kritas, S.K.; Frydas, I.; Younes, A.; Di Emidio, P.; Ronconi, G. Mast cells activated by SARS-CoV-2 release histamine which increases IL-1 levels causing cytokine storm and inflammatory reaction in COVID-19. J. Biol. Regul. Homeost. Agents 2020, 34, 1629–1632. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Lantz, C.S.; Oettgen, H.C.; Katona, I.M.; Fleming, T.; Miyajima, I.; Kinet, J.-P.; Galli, S.J. IgE Enhances Mouse Mast Cell FcεRI Expression In Vitro and In Vivo: Evidence for a Novel Amplification Mechanism in IgE-dependent Reactions. J. Exp. Med. 1997, 185, 663–672. [Google Scholar] [CrossRef] [PubMed]
- de Castro, R.O.; Zhang, J.; Jamur, M.C.; Oliver, C.; Siraganian, R.P. Tyrosines in the Carboxyl Terminus Regulate Syk Kinase Activity and Function. J. Biol. Chem. 2010, 285, 26674–26684. [Google Scholar] [CrossRef] [PubMed]
- Tkaczyk, C.; Jensen, B.M.; Iwaki, S.; Gilfillan, A.M. Adaptive and Innate Immune Reactions Regulating Mast Cell Activation: From Receptor-mediated Signaling to Responses. Immunol. Allergy Clin. N. Am. 2006, 26, 427–450. [Google Scholar] [CrossRef] [PubMed]
- Besmer, P.; Murphy, J.E.; George, P.C.; Qiu, F.; Bergold, P.J.; Lederman, L.; Snyder, H.W.; Brodeur, D.; Zuckerman, E.E.; Hardy, W.D. A new acute transforming feline retrovirus and relationship of its oncogene v-kit with the protein kinase gene family. Nature 1986, 320, 415–421. [Google Scholar] [CrossRef]
- Tsai, M.; Valent, P.; Galli, S.J. KIT as a master regulator of the mast cell lineage. J. Allergy Clin. Immunol. 2022, 149, 1845–1854. [Google Scholar] [CrossRef] [PubMed]
- Oriss, T.B.; Krishnamoorthy, N.; Ray, P.; Ray, A. Dendritic cell c-kit signaling and adaptive immunity. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Austen, K.F.; Friend, D.S.; Heidtman, M.; Boyce, J.A. Human Peripheral Blood Eosinophils Express a Functional c-kit Receptor for Stem Cell Factor that Stimulates Very Late Antigen 4 (VLA-4)–mediated Cell Adhesion to Fibronectin and Vascular Cell Adhesion Molecule 1 (VCAM-1). J. Exp. Med. 1997, 186, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Manova, K.; Bachvarova, R.; Huang, E.; Sanchez, S.; Pronovost, S.; Velazquez, E.; McGuire, B.; Besmer, P. c-kit receptor and ligand expression in postnatal development of the mouse cerebellum suggests a function for c-kit in inhibitory interneurons. J. Neurosci. 1992, 12, 4663–4676. [Google Scholar] [CrossRef][Green Version]
- Desai, A.; Sowerwine, K.; Liu, Y.; Lawrence, M.G.; Chovanec, J.; Hsu, A.P.; O’Connell, M.P.; Kim, J.; Boris, L.; Jones, N.; et al. GATA-2–deficient mast cells limit IgE-mediated immediate hypersensitivity reactions in human subjects. J. Allergy Clin. Immunol. 2019, 144, 613–617.e14. [Google Scholar] [CrossRef]
- Lemmon, M.A.; Schlessinger, J. Cell Signaling by Receptor Tyrosine Kinases. Cell 2010, 141, 1117–1134. [Google Scholar] [CrossRef]
- Liang, J.; Wu, Y.-L.; Chen, B.-J.; Zhang, W.; Tanaka, Y.; Sugiyama, H. The C-Kit Receptor-Mediated Signal Transduction and Tumor-Related Diseases. Int. J. Biol. Sci. 2013, 9, 435–443. [Google Scholar] [CrossRef]
- Anderson, D.M.; Williams, D.E.; Tushinski, R.; Gimpel, S.; Eisenman, J.; Cannizzaro, L.A.; Aronson, M.; Croce, C.M.; Huebner, K.; Cosman, D.; et al. Alternate splicing of mRNAs encoding human mast cell growth factor and localization of the gene to chromosome 12q22-q24. Cell Growth Differ. 1991, 2, 373–378. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1724381 (accessed on 23 October 2023). [PubMed]
- Anderson, A.C.; Joller, N.; Kuchroo, V.K. Lag-3, Tim-3, and TIGIT: Co-inhibitory Receptors with Specialized Functions in Immune Regulation. Immunity 2016, 44, 989–1004. [Google Scholar] [CrossRef] [PubMed]
- Youngblood, B.A.; Leung, J.; Falahati, R.; Williams, J.; Schanin, J.; Brock, E.C.; Singh, B.; Chang, A.T.; O’sullivan, J.A.; Schleimer, R.P.; et al. Discovery, Function, and Therapeutic Targeting of Siglec-8. Cells 2020, 10, 19. [Google Scholar] [CrossRef]
- McNeil, B.D.; Pundir, P.; Meeker, S.; Han, L.; Undem, B.J.; Kulka, M.; Dong, X. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 2015, 519, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Wedi, B.; Gehring, M.; Kapp, A. The pseudoallergen receptor MRGPRX2 on peripheral blood basophils and eosinophils: Expression and function. Allergy Eur. J. Allergy Clin. Immunol. 2020, 75, 2229–2242. [Google Scholar] [CrossRef] [PubMed]
- Shtessel, M.; Limjunyawong, N.; Oliver, E.T.; Chichester, K.; Gao, L.; Dong, X.; Saini, S.S. MRGPRX2 Activation Causes Increased Skin Reactivity in Patients with Chronic Spontaneous Urticaria. J. Investig. Dermatol. 2021, 141, 678–681.e2. [Google Scholar] [CrossRef]
- Pundir, P.; Liu, R.; Vasavda, C.; Serhan, N.; Limjunyawong, N.; Yee, R.; Zhan, Y.; Dong, X.; Wu, X.; Zhang, Y.; et al. A Connective Tissue Mast-Cell-Specific Receptor Detects Bacterial Quorum-Sensing Molecules and Mediates Antibacterial Immunity. Cell Host Microbe 2019, 26, 114–122.e8. [Google Scholar] [CrossRef] [PubMed]
- Lorentz, A.; Sellge, G.; Bischoff, S.C. Isolation and Characterization of Human Intestinal Mast Cells. Methods Mol. Biol. 2015, 1220, 163–177. [Google Scholar]
- Trompette, A.; Divanovic, S.; Visintin, A.; Blanchard, C.; Hegde, R.S.; Madan, R.; Thorne, P.S.; Wills-Karp, M.; Gioannini, T.L.; Weiss, J.P.; et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature 2009, 457, 585–588. [Google Scholar] [CrossRef]
- Sandig, H.; Bulfone-Paus, S. TLR signaling in mast cells: Common and unique features. Front. Immunol. 2012, 3, 185. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Tsilioni, I.; Conti, P. Mast Cells May Regulate The Anti-Inflammatory Activity of IL-37. Int. J. Mol. Sci. 2019, 20, 3701. [Google Scholar] [CrossRef]
- Supajatura, V.; Ushio, H.; Nakao, A.; Okumura, K.; Ra, C.; Ogawa, H. Protective Roles of Mast Cells Against Enterobacterial Infection Are Mediated by Toll-Like Receptor 4. J. Immunol. 2001, 167, 2250–2256. [Google Scholar] [CrossRef]
- Supajatura, V.; Ushio, H.; Nakao, A.; Akira, S.; Okumura, K.; Ra, C.; Ogawa, H. Differential responses of mast cell Toll-like receptors 2 and 4 in allergy and innate immunity. J. Clin. Investig. 2002, 109, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- Gimenez, A.P.; Wu, Y.-Z.; Paya, M.; Delclaux, C.; Touqui, L.; Goossens, P.L. High Bactericidal Efficiency of Type IIA Phospholipase A2 against Bacillus anthracis and Inhibition of Its Secretion by the Lethal Toxin. J. Immunol. 2004, 173, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Soumelis, V.; Reche, P.A.; Kanzler, H.; Yuan, W.; Edward, G.; Homey, B.; Gilliet, M.; Ho, S.; Antonenko, S.; Lauerma, A.; et al. Human epithelial cells trigger dendritic cell–mediated allergic inflammation by producing TSLP. Nat. Immunol. 2002, 3, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Al-Sajee, D.; Oliveria, J.-P.; Sehmi, R.; Gauvreau, G.M. Antialarmins for treatment of asthma. Curr. Opin. Pulm. Med. 2018, 24, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an Interleukin-1-like Cytokine that Signals via the IL-1 Receptor-Related Protein ST2 and Induces T Helper Type 2-Associated Cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Iikura, M.; Suto, H.; Kajiwara, N.; Oboki, K.; Ohno, T.; Okayama, Y.; Saito, H.; Galli, S.J.; Nakae, S. IL-33 can promote survival, adhesion and cytokine production in human mast cells. Lab. Investig. 2007, 87, 971–978. [Google Scholar] [CrossRef]
- Spicuzza, L.; Di Maria, G.; Polosa, R. Adenosine in the airways: Implications and applications. Eur. J. Pharmacol. 2006, 533, 77–88. [Google Scholar] [CrossRef]
- Mohajeri, M.; Kovanen, P.T.; Bianconi, V.; Pirro, M.; Cicero, A.F.; Sahebkar, A. Mast cell tryptase—Marker and maker of cardiovascular diseases. Pharmacol. Ther. 2019, 199, 91–110. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Sprinzl, B.; Greiner, G.; Uyanik, G.; Arock, M.; Haferlach, T.; Sperr, W.R.; Valent, P.; Hoermann, G. Genetic Regulation of Tryptase Production and Clinical Impact: Hereditary α Tryptasemia, Mastocytosis and Beyond. Int. J. Mol. Sci. 2021, 22, 2458. [Google Scholar] [CrossRef] [PubMed]
- Greiner, G.; Sprinzl, B.; Górska, A.; Ratzinger, F.; Gurbisz, M.; Witzeneder, N.; Schmetterer, K.G.; Gisslinger, B.; Uyanik, G.; Hadzijusufovic, E.; et al. Hereditary α tryptasemia is a valid genetic biomarker for severe mediator-related symptoms in mastocytosis. Blood 2021, 137, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, G.; Schwartz, L.B.; Khan, D.A. Prolonged elevation of serum tryptase in idiopathic anaphylaxis. J. Allergy Clin. Immunol. 2006, 117, 950–951. [Google Scholar] [CrossRef] [PubMed]
- Omraninava, M.; Eslami, M.; Aslani, S.; Razi, B.; Imani, D.; Feyzinia, S. Interleukin 13 gene polymorphism and susceptibility to asthma: A meta-regression and meta-analysis. Eur. Ann. Allergy Clin. Immunol. 2022, 54, 150–167. [Google Scholar] [CrossRef] [PubMed]
- Okayama, Y.; Hagaman, D.D.; Metcalfe, D.D. A Comparison of Mediators Released or Generated by IFN-γ-Treated Human Mast Cells Following Aggregation of FcγRI or FcεRI. J. Immunol. 2001, 166, 4705–4712. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M.; Wershil, B.K. The c-kit receptor, stem cell factor, and mast cells. What each is teaching us about the others. Am. J. Pathol. 1993, 142, 965–974. Available online: http://www.ncbi.nlm.nih.gov/pubmed/7682764 (accessed on 23 October 2023).
- Marshall, J.S. Mast-cell responses to pathogens. Nat. Rev. Immunol. 2004, 4, 787–799. [Google Scholar] [CrossRef]
- Orinska, Z.; Bulanova, E.; Budagian, V.; Metz, M.; Maurer, M.; Bulfone-Paus, S.; Orinska, Z.; Bulanova, E.; Budagian, V.; Metz, M.; et al. TLR3-induced activation of mast cells modulates CD8+ T-cell recruitment. Blood 2005, 106, 978–987. [Google Scholar] [CrossRef]
- Nilsson, G.; Johnell, M.; Hammer, C.H.; Tiffany, H.L.; Nilsson, K.; Metcalfe, D.D.; Siegbahn, A.; Murphy, P.M. C3a and C5a are chemotaxins for human mast cells and act through distinct receptors via a pertussis toxin-sensitive signal transduction pathway. J. Immunol. 1996, 157, 1693–1698. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8759757 (accessed on 23 October 2023). [CrossRef] [PubMed]
- Horny, H.-P.; Sotlar, K.; Valent, P.; Hartmann, K. Mastocytosis—A Disease of the Hematopoietic Stem Cell. Dtsch. Arztebl. Int. 2008, 105, 686–692. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Valent, P.; Akin, C. Mast Cells, Mastocytosis, and Related Disorders. N. Engl. J. Med. 2015, 373, 1884–1886. [Google Scholar] [CrossRef]
- Metcalfe, D.D. Mast cells and mastocytosis. Blood 2008, 112, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Valent, P.; Akin, C.; Metcalfe, D.D. Mastocytosis: 2016 updated WHO classification and novel emerging treatment concepts. Blood 2017, 129, 1420–1427. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, K.; Escribano, L.; Grattan, C.; Brockow, K.; Carter, M.C.; Alvarez-Twose, I.; Matito, A.; Broesby-Olsen, S.; Siebenhaar, F.; Lange, M.; et al. Cutaneous manifestations in patients with mastocytosis: Consensus report of the European Competence Network on Mastocytosis; the American Academy of Allergy, Asthma & Immunology; and the European Academy of Allergology and Clinical Immunology. J. Allergy Clin. Immunol. 2016, 137, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Pardanani, A.; Lim, K.-H.; Lasho, T.L.; Finke, C.M.; McClure, R.F.; Li, C.-Y.; Tefferi, A. WHO subvariants of indolent mastocytosis: Clinical details and prognostic evaluation in 159 consecutive adults. Blood 2010, 115, 150–151. [Google Scholar] [CrossRef] [PubMed]
- Chiu, A.; Orazi, A. Mastocytosis and related disorders. Semin. Diagn. Pathol. 2012, 29, 19–30. [Google Scholar] [CrossRef]
- Worobec, A.S.; Metcalfe, D.D. Mastocytosis: Current Treatment Concepts. Int. Arch. Allergy Immunol. 2002, 127, 153–155. [Google Scholar] [CrossRef]
- Jordan, J.-H.; Walchshofer, S.; Jurecka, W.; Mosberger, I.; Sperr, W.R.; Wolff, K.; Chott, A.; Bühring, H.-J.; Lechner, K.; Horny, H.-P.; et al. Immunohistochemical properties of bone marrow mast cells in systemic mastocytosis: Evidence for expression of CD2, CD117/Kit, and bcl-xL. Hum. Pathol. 2001, 32, 545–552. [Google Scholar] [CrossRef]
- Krokowski, M.; Sotlar, K.; Krauth, M.-T.; Födinger, M.; Valent, P.; Horny, H.-P. Delineation of Patterns of Bone Marrow Mast Cell Infiltration in Systemic Mastocytosis. Am. J. Clin. Pathol. 2005, 124, 560–568. [Google Scholar] [CrossRef]
- van Anrooij, B.; Kluin, P.M.; Elberink, J.N.O.; Kluin-Nelemans, J.C. CD30 in Systemic Mastocytosis. Immunol. Allergy Clin. N. Am. 2014, 34, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Minato, N.; Hattori, M.; Hamazaki, Y. Physiology and pathology of T-cell aging. Int. Immunol. 2020, 32, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Kluin-Nelemans, H.C.; Jawhar, M.; Reiter, A.; van Anrooij, B.; Gotlib, J.; Hartmann, K.; Illerhaus, A.; Elberink, H.N.O.; Gorska, A.; Niedoszytko, M.; et al. Cytogenetic and molecular aberrations and worse outcome for male patients in systemic mastocytosis. Theranostics 2021, 11, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Arock, M.; Akin, C.; Hermine, O.; Valent, P. Current treatment options in patients with mastocytosis: Status in 2015 and future perspectives. Eur. J. Haematol. 2015, 94, 474–490. [Google Scholar] [CrossRef] [PubMed]
- Palacios, F.; Abreu, C.; Prieto, D.; Morande, P.; Ruiz, S.; Fernández-Calero, T.; Naya, H.; Libisch, G.; Robello, C.; I Landoni, A.; et al. Activation of the PI3K/AKT pathway by microRNA-22 results in CLL B-cell proliferation. Leukemia 2015, 29, 115–125. [Google Scholar] [CrossRef]
- Garcia-Montero, A.C.; Jara-Acevedo, M.; Teodosio, C.; Sanchez, M.L.; Nunez, R.; Prados, A.; Aldanondo, I.; Sanchez, L.; Dominguez, M.; Botana, L.M.; et al. KIT mutation in mast cells and other bone marrow hematopoietic cell lineages in systemic mast cell disorders: A prospective study of the Spanish Network on Mastocytosis (REMA) in a series of 113 patients. Blood 2006, 108, 2366–2372. [Google Scholar] [CrossRef]
- Gotlib, J.; Kluin-Nelemans, H.C.; Akin, C.; Hartmann, K.; Valent, P.; Reiter, A. Practical management of adverse events in patients with advanced systemic mastocytosis receiving midostaurin. Expert Opin. Biol. Ther. 2021, 21, 487–498. [Google Scholar] [CrossRef]
- Li, Z.; Beutel, G.; Rhein, M.; Meyer, J.; Koenecke, C.; Neumann, T.; Yang, M.; Krauter, J.; von Neuhoff, N.; Heuser, M.; et al. High-affinity neurotrophin receptors and ligands promote leukemogenesis. Blood 2009, 113, 2028–2037. [Google Scholar] [CrossRef]
- Sawada, J.; Itakura, A.; Tanaka, A.; Furusaka, T.; Matsuda, H. Nerve growth factor functions as a chemoattractant for mast cells through both mitogen-activated protein kinase and phosphatidylinositol 3-kinase signaling pathways. Blood 2000, 95, 2052–2058. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10706874 (accessed on 27 October 2023). [CrossRef]
- Peng, W.-M.; Maintz, L.; Allam, J.-P.; Raap, U.; Gütgemann, I.; Kirfel, J.; Wardelmann, E.; Perner, S.; Zhao, W.; Fimmers, R.; et al. Increased circulating levels of neurotrophins and elevated expression of their high-affinity receptors on skin and gut mast cells in mastocytosis. Blood 2013, 122, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Pan, Z.; Huang, K.; Büsche, G.; Feuerhake, F.; Chaturvedi, A.; Nie, D.; Heuser, M.; Thol, F.; von Neuhoff, N.; et al. Activation of TRKA receptor elicits mastocytosis in mice and is involved in the development of resistance to KIT-targeted therapy. Oncotarget 2017, 8, 73871–73883. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Smrž, D.; Kim, M.-S.; Zhang, S.; Mock, B.A.; Smržová, Š.; DuBois, W.; Simakova, O.; Maric, I.; Wilson, T.M.; Metcalfe, D.D.; et al. mTORC1 and mTORC2 differentially regulate homeostasis of neoplastic and non-neoplastic human mast cells. Blood 2011, 118, 6803–6813. [Google Scholar] [CrossRef] [PubMed]
- Gabillot-Carré, M.; Lepelletier, Y.; Humbert, M.; de Sepuvelda, P.; Ben Hamouda, N.; Zappulla, J.P.; Liblau, R.; Ribadeau-Dumas, A.; Machavoine, F.; Letard, S.; et al. Rapamycin inhibits growth and survival of D816V-mutated c-kit mast cells. Blood 2006, 108, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Itzykson, R.; Kosmider, O.; Fenaux, P. Somatic mutations and epigenetic abnormalities in myelodysplastic syndromes. Best Pract. Res. Clin. Haematol. 2013, 26, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Schevzov, G.; Kee, A.J.; Wang, B.; Sequeira, V.B.; Hook, J.; Coombes, J.D.; Lucas, C.A.; Stehn, J.R.; Musgrove, E.A.; Cretu, A.; et al. Regulation of cell proliferation by ERK and signal-dependent nuclear translocation of ERK is dependent on Tm5NM1-containing actin filaments. Mol. Biol. Cell 2015, 26, 2475–2490. [Google Scholar] [CrossRef]
- Hanssens, K.; Brenet, F.; Agopian, J.; Georgin-Lavialle, S.; Damaj, G.; Cabaret, L.; Chandesris, M.O.; de Sepulveda, P.; Hermine, O.; Dubreuil, P.; et al. SRSF2-p95 hotspot mutation is highly associated with advanced forms of mastocytosis and mutations in epigenetic regulator genes. Haematologica 2014, 99, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Jawhar, M.; Schwaab, J.; Schnittger, S.; Meggendorfer, M.; Pfirrmann, M.; Sotlar, K.; Horny, H.-P.; Metzgeroth, G.; Kluger, S.; Naumann, N.; et al. Additional mutations in SRSF2, ASXL1 and/or RUNX1 identify a high-risk group of patients with KIT D816V+ advanced systemic mastocytosis. Leukemia 2016, 30, 136–143. [Google Scholar] [CrossRef]
- Wilson, T.M.; Maric, I.; Simakova, O.; Bai, Y.; Chan, E.C.; Olivares, N.; Carter, M.; Maric, D.; Robyn, J.; Metcalfe, D.D. Clonal analysis of NRAS activating mutations in KIT-D816V systemic mastocytosis. Haematologica 2011, 96, 459–463. [Google Scholar] [CrossRef]
- Li, Z. New Insights into the Pathogenesis of Systemic Mastocytosis. Int. J. Mol. Sci. 2021, 22, 4900. [Google Scholar] [CrossRef]
- De Vita, S.; Schneider, R.K.; Garcia, M.; Wood, J.; Gavillet, M.; Ebert, B.L.; Gerbaulet, A.; Roers, A.; Levine, R.L.; Mullally, A.; et al. Loss of Function of TET2 Cooperates with Constitutively Active KIT in Murine and Human Models of Mastocytosis. PLoS ONE 2014, 9, e96209. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, G.; Mancini, M.; De Benedittis, C.; Rondoni, M.; Papayannidis, C.; Manfrini, M.; Meggendorfer, M.; Calogero, R.; Guadagnuolo, V.; Fontana, M.C.; et al. SETD2 and histone H3 lysine 36 methylation deficiency in advanced systemic mastocytosis. Leukemia 2018, 32, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Nagase, R.; Inoue, D.; Pastore, A.; Fujino, T.; Hou, H.-A.; Yamasaki, N.; Goyama, S.; Saika, M.; Kanai, A.; Sera, Y.; et al. Expression of mutant Asxl1 perturbs hematopoiesis and promotes susceptibility to leukemic transformation. J. Exp. Med. 2018, 215, 1729–1747. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Y.; Smith, M.L.; Schultheis, B.; Fitzgibbon, J.; Lister, T.A.; Melo, J.V.; Cross, N.C.; Cavenagh, J.D. A novel K509I mutation of KIT identified in familial mastocytosis—In vitro and in vivo responsiveness to imatinib therapy. Leuk. Res. 2006, 30, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Boxer, M.; Drummond, A.; Ogston, P.; Hodgins, M.; Burden, A.D. A germline mutation in KIT in familial diffuse cutaneous mastocytosis. J. Med. Genet. 2004, 41, e88. [Google Scholar] [CrossRef] [PubMed]
- Wasag, B.; Niedoszytko, M.; Piskorz, A.; Lange, M.; Renke, J.; Jassem, E.; Biernat, W.; Debiec-Rychter, M.; Limon, J. Novel, activating KIT-N822I mutation in familial cutaneous mastocytosis. Exp. Hematol. 2011, 39, 859–865.e2. [Google Scholar] [CrossRef] [PubMed]
- Galatà, G.; García-Montero, A.C.; Kristensen, T.; Dawoud, A.A.; Muñoz-González, J.I.; Meggendorfer, M.; Guglielmelli, P.; Hoade, Y.; Alvarez-Twose, I.; Gieger, C.; et al. Genome-wide association study identifies novel susceptibility loci for KIT D816V positive mastocytosis. Am. J. Hum. Genet. 2021, 108, 284–294. [Google Scholar] [CrossRef]
- Ammendola, M.; Sacco, R.; Sammarco, G.; Luposella, M.; Patruno, R.; Gadaleta, C.D.; De Sarro, G.; Ranieri, G. Mast Cell-Targeted Strategies in Cancer Therapy. Transfus. Med. Hemotherapy 2016, 43, 109–113. [Google Scholar] [CrossRef]
- Faustino-Rocha, A.I.; Gama, A.; Oliveira, P.A.; Vanderperren, K.; Saunders, J.H.; Pires, M.J.; Ferreira, R.; Ginja, M. Modulation of mammary tumor vascularization by mast cells: Ultrasonographic and histopathological approaches. Life Sci. 2017, 176, 35–41. [Google Scholar] [CrossRef]
- Iqbal, N.; Iqbal, N. Imatinib: A Breakthrough of Targeted Therapy in Cancer. Chemother. Res. Pract. 2014, 2014, 357027. [Google Scholar] [CrossRef]
- Cimpean, A.M.; Raica, M. The Hidden Side of Disodium Cromolyn: From Mast Cell Stabilizer to an Angiogenic Factor and Antitumor Agent. Arch. Immunol. Ther. Exp. 2016, 64, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Marech, I.; Ammendola, M.; Gadaleta, C.; Zizzo, N.; Oakley, C.; Gadaleta, C.D.; Ranieri, G. Possible biological and translational significance of mast cells density in colorectal cancer. World J. Gastroenterol. 2014, 20, 8910–8920. [Google Scholar] [CrossRef] [PubMed]
- Coelho, Y.N.B.; Soldi, L.R.; da Silva, P.H.R.; Mesquita, C.M.; Paranhos, L.R.; dos Santos, T.R.; Silva, M.J.B. Tyrosine kinase inhibitors as an alternative treatment in canine mast cell tumor. Front. Vet. Sci. 2023, 10, 1188795. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zeng, S.; Metcalfe, D.D.; Akin, C.; Dimitrijevic, S.; Butterfield, J.H.; McMahon, G.; Longley, B.J. The c-KIT mutation causing human mastocytosis is resistant to STI571 and other KIT kinase inhibitors; kinases with enzymatic site mutations show different inhibitor sensitivity profiles than wild-type kinases and those with regulatory-type mutations. Blood 2002, 99, 1741–1744. [Google Scholar] [CrossRef] [PubMed]
- Droogendijk, H.J.; Kluin-Nelemans, H.J.C.; van Doormaal, J.J.; Oranje, A.P.; van de Loosdrecht, A.A.; van Daele, P.L.A. Imatinib mesylate in the treatment of systemic mastocytosis. Cancer 2006, 107, 345–351. [Google Scholar] [CrossRef]
- Pagano, L.; Valentini, C.G.; Caira, M.; Rondoni, M.; Van Lint, M.T.; Candoni, A.; Allione, B.; Cattaneo, C.; Marbello, L.; Caramatti, C.; et al. Advanced mast cell disease: An Italian Hematological Multicenter experience. Int. J. Hematol. 2008, 88, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Vega-Ruiz, A.; Cortes, J.E.; Sever, M.; Manshouri, T.; Quintás-Cardama, A.; Luthra, R.; Kantarjian, H.M.; Verstovsek, S. Phase II study of imatinib mesylate as therapy for patients with systemic mastocytosis. Leuk. Res. 2009, 33, 1481–1484. [Google Scholar] [CrossRef]
- Pardanani, A.; Elliott, M.; Reeder, T.; Li, C.-Y.; Baxter, E.; Cross, N.; Tefferi, A. Imatinib for systemic mast-cell disease. Lancet 2003, 362, 535–537. [Google Scholar] [CrossRef]
- Dubreuil, P.; Letard, S.; Ciufolini, M.; Gros, L.; Humbert, M.; Castéran, N.; Borge, L.; Hajem, B.; Lermet, A.; Sippl, W.; et al. Masitinib (AB1010), a Potent and Selective Tyrosine Kinase Inhibitor Targeting KIT. PLoS ONE 2009, 4, e7258. [Google Scholar] [CrossRef]
- Lortholary, O.; Chandesris, M.O.; Livideanu, C.B.; Paul, C.; Guillet, G.; Jassem, E.; Niedoszytko, M.; Barete, S.; Verstovsek, S.; Grattan, C.; et al. Masitinib for treatment of severely symptomatic indolent systemic mastocytosis: A randomised, placebo-controlled, phase 3 study. Lancet 2017, 389, 612–620. [Google Scholar] [CrossRef]
- Paul, C.; Sans, B.; Suarez, F.; Casassus, P.; Barete, S.; Lanternier, F.; Grandpeix-Guyodo, C.; Dubreuil, P.; Palmérini, F.; Mansfield, C.D.; et al. Masitinib for the treatment of systemic and cutaneous mastocytosis with handicap: A phase 2a study. Am. J. Hematol. 2010, 85, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Fabbro, D.; Ruetz, S.; Bodis, S.; Pruschy, M.; Csermak, K.; Man, A.; Campochiaro, P.; Wood, J.; O’Reilly, T.; Meyer, T. PKC412--a protein kinase inhibitor with a broad therapeutic potential. Anticancer. Drug Des. 2000, 15, 17–28. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10888033 (accessed on 27 October 2023).
- Evans, E.K.; Gardino, A.K.; Kim, J.L.; Hodous, B.L.; Shutes, A.; Davis, A.; Zhu, X.J.; Schmidt-Kittler, O.; Wilson, D.; Wilson, K.; et al. A precision therapy against cancers driven by KIT/PDGFRA mutations. Sci. Transl. Med. 2017, 9, eaao1690. [Google Scholar] [CrossRef]
- Dave, N.; Devlin, M.; Rodstrom, J.; Yu, B.; Foley, M.; He, K.; Rasmussen, S.; Boral, A.; Si, T.D. Abstract CT122: Safety and pharmacokinetics of BLU-263, a next-generation KIT inhibitor, in normal healthy volunteers. Cancer Res 2021, 81, CT122. [Google Scholar] [CrossRef]
- Guarnieri, A.; Chicarelli, M.; Cable, L.; Bouhana, K.; Sullivan, F.; Ball, H.; Sachs, J.; Winski, S.; Robinson, J. Preclinical Data with KIT D816V Inhibitor Bezuclastinib (CGT9486) Demonstrates High Selectivity and Minimal Brain Penetrance. Blood 2021, 138 (Suppl. S1), 4595. [Google Scholar] [CrossRef]
- Sledge, D.G.; Webster, J.; Kiupel, M. Canine cutaneous mast cell tumors: A combined clinical and pathologic approach to diagnosis, prognosis, and treatment selection. Vet. J. 2016, 215, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Aceino, A.; Jeffery, U.; Piccione, J.; Hodo, C.L. Systemic mastocytosis with subcutaneous hemorrhage and edema in a Greyhound dog: Case report and review of diagnostic criteria. J. Vet. Diagn. Investig. 2020, 33, 95–100. [Google Scholar] [CrossRef]
- Zorzan, E.; Hanssens, K.; Giantin, M.; Dacasto, M.; Dubreuil, P. Mutational Hotspot of TET2, IDH1, IDH2, SRSF2, SF3B1, KRAS, and NRAS from Human Systemic Mastocytosis Are Not Conserved in Canine Mast Cell Tumors. PLoS ONE 2015, 10, e0142450. [Google Scholar] [CrossRef]
- Pariser, M.S.; Gram, D.W. Urticaria pigmentosa-like disease in a dog. Can. Vet. J. 2015, 56, 245–248. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25750443 (accessed on 27 October 2023).
- da Silva, M.S.; Klaser, W.; Scherer, F.; Santos, D.D.; Garcia, C.; Bortolini, E.; Costa, M. Metastatic Intranasal Mastocytoma in a Dog. Acta Sci. Vet. 2023, 51, 892. [Google Scholar] [CrossRef]
- Thompson, J.J.; Pearl, D.L.; Yager, J.A.; Best, S.J.; Coomber, B.L.; Foster, R.A. Canine Subcutaneous Mast Cell Tumor. Vet. Pathol. 2011, 48, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Bostock, D.E. The prognosis following surgical removal of mastocytomas in dogs. J. Small Anim. Pract. 1973, 14, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, A.K.; Ehler, W.J.; MacEwen, E.G. Canine Cutaneous Mast Cell Tumor: Morphologic Grading and Survival Time in 83 Dogs. Vet. Pathol. 1984, 21, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Kiupel, M.; Webster, J.D.; Bailey, K.L.; Best, S.; DeLay, J.; Detrisac, C.J.; Fitzgerald, S.D.; Gamble, D.; Ginn, P.E.; Goldschmidt, M.H.; et al. Proposal of a 2-Tier Histologic Grading System for Canine Cutaneous Mast Cell Tumors to More Accurately Predict Biological Behavior. Vet. Pathol. 2011, 48, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Stefanello, D.; Buracco, P.; Sabattini, S.; Finotello, R.; Giudice, C.; Grieco, V.; Iussich, S.; Tursi, M.; Scase, T.; Di Palma, S.; et al. Comparison of 2- and 3-category histologic grading systems for predicting the presence of metastasis at the time of initial evaluation in dogs with cutaneous mast cell tumors: 386 cases (2009–2014). J. Am. Vet. Med. Assoc. 2015, 246, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Northrup, N.C.; Howerth, E.W.; Harmon, B.G.; Brown, C.A.; Carmicheal, K.P.; Garcia, A.P.; Latimer, K.S.; Munday, J.S.; Rakich, P.M.; Richey, L.J.; et al. Variation among Pathologists in the Histologic Grading of Canine Cutaneous Mast Cell Tumors with Uniform Use of a Single Grading Reference. J. Vet. Diagn. Investig. 2005, 17, 561–564. [Google Scholar] [CrossRef]
- Ozaki, K.; Yamagami, T.; Nomura, K.; Narama, I. Mast cell tumors of the gastrointestinal tract in 39 dogs. Vet. Pathol. 2002, 39, 557–564. [Google Scholar] [CrossRef]
- Ejima, E.; Fujiyama, K.; Kiriyama, T.; Eguchi, K. [Bone changes in thyrotoxicosis]. Nihon Rinsho. 1998, 56, 1587–1590. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9648486 (accessed on 28 October 2023).
- Elston, L.B.; Sueiro, F.A.; Cavalcanti, J.N.; Metze, K. Letter to the Editor: The Importance of the Mitotic Index as a Prognostic Factor for Survival of Canine Cutaneous Mast Cell Tumors: A Validation Study. Vet. Pathol. 2009, 46, 362–364. [Google Scholar] [CrossRef]
- Marcos, R.; Almeida, J.; Marques, J.; Moreira, R.; Dias-Pereira, P.; Correia-Gomes, C.; Santos, M. Canine mast cell tumors: Utility of stereologic tools in cytology. J. Vet. Diagn. Investig. 2022, 34, 263–267. [Google Scholar] [CrossRef]
- Hosseini, E.; Pedram, B.; Bahrami, A.M.; Moghaddam, M.H.J.; Javanbakht, J.; Ghomi, F.E.; Moghaddam, N.J.; Koohestani, M.; Shafiee, R. RETRACTED ARTICLE: Cutaneous mast cell tumor (Mastocytoma): Cyto- histopathological and haematological investigations. Diagn. Pathol. 2014, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Cruz, V.S.; Borges, J.C.A.; Nepomuceno, L.L.; Gonçalves, P.A.M.; Prado, Y.C.L.; Bianchi, C.; Fioravanti, M.C.S.; Araújo, E.G. Histological classification and expression of markers of canine mast cell tumors. Vet. World 2020, 13, 1627–1637. [Google Scholar] [CrossRef] [PubMed]
- Hillman, L.A.; Garrett, L.D.; de Lorimier, L.-P.; Charney, S.C.; Borst, L.B.; Fan, T.M. Biological behavior of oral and perioral mast cell tumors in dogs: 44 cases (1996–2006). J. Am. Vet. Med. Assoc. 2010, 237, 936–942. [Google Scholar] [CrossRef]
- Graf, R.; Pospischil, A.; Guscetti, F.; Meier, D.; Welle, M.; Dettwiler, M. Cutaneous Tumors in Swiss Dogs: Retrospective Data From the Swiss Canine Cancer Registry, 2008–2013. Vet. Pathol. 2018, 55, 809–820. [Google Scholar] [CrossRef] [PubMed]
- Pejler, G.; Åbrink, M.; Ringvall, M.; Wernersson, S. Mast Cell Proteases. Adv. Immunol. 2007, 95, 167–255. [Google Scholar]
- Sakai, H.; Noda, A.; Shirai, N.; Iidaka, T.; Yanai, T.; Masegi, T. Proliferative Activity of Canine Mast Cell Tumours Evaluated by Bromodeoxyuridine Incorporation and Ki-67 Expression. J. Comp. Pathol. 2002, 127, 233–238. [Google Scholar] [CrossRef]
- Simoes, J.P.C.; Schoning, P.; Butine, M. Prognosis of Canine Mast Cell Tumors: A Comparison of Three Methods. Vet. Pathol. 1994, 31, 637–647. [Google Scholar] [CrossRef]
- Kravis, L.D.; Vail, D.M.; Kisseberth, W.C.; Ogilvie, G.K.; Volk, L.M. Frequency of argyrophilic nucleolar organizer regions in fine-needle aspirates and biopsy specimens from mast cell tumors in dogs. J. Am. Vet. Med. Assoc. 1996, 209, 1418–1420. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8870737 (accessed on 28 October 2023). [CrossRef] [PubMed]
- Ayl, R.D.; Couto, C.G.; Hammer, A.S.; Weisbrode, S.; Ericson, J.G.; Mathes, L. Correlation of DNA Ploidy to Tumor Histologic Grade, Clinical Variables, and Survival in Dogs with Mast Cell Tumors. Vet. Pathol. 1992, 29, 386–390. [Google Scholar] [CrossRef]
- Jaffe, M.H.; Hosgood, G.; Taylor, H.W.; Kerwin, S.C.; Hedlund, C.S.; Lopez, M.K.; Davidson, J.R.; Miller, D.M.; Paranjpe, M. Immunohistochemical and Clinical Evaluation of p53 in Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2000, 37, 40–46. [Google Scholar] [CrossRef]
- Webster, J.D.; Kiupel, M.; Yuzbasiyan-Gurkan, V. Evaluation of the kinase domain of c-KIT in canine cutaneous mast cell tumors. BMC Cancer 2006, 6, 85. [Google Scholar] [CrossRef]
- Strefezzi, R.D.F.; Xavier, J.G.; Catão-Dias, J.L. Morphometry of Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2003, 40, 268–275. [Google Scholar] [CrossRef]
- Kiupel, M.; Webster, J.D.; Miller, R.A.; Kaneene, J.B. Impact of Tumour Depth, Tumour Location and Multiple Synchronous Masses on the Prognosis of Canine Cutaneous Mast Cell Tumours. J. Vet. Med. Ser. A 2005, 52, 280–286. [Google Scholar] [CrossRef]
- Cahalane, A.K.; Payne, S.; Barber, L.G.; Duda, L.E.; Henry, C.J.; Mauldin, G.E.; Frimberger, A.E.; Cotter, S.M.; Moore, A.S. Prognostic factors for survival of dogs with inguinal and perineal mast cell tumors treated surgically with or without adjunctive treatment: 68 cases (1994–2002). J. Am. Vet. Med. Assoc. 2004, 225, 401–408. [Google Scholar] [CrossRef]
- Kiupel, M.; Webster, J.D.; Kaneene, J.B.; Miller, R.; Yuzbasiyan-Gurkan, V. The Use of KIT and Tryptase Expression Patterns as Prognostic Tools for Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2004, 41, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.J.; Morrison, J.A.; Pearl, D.L.; Boston, S.E.; Wood, G.A.; Foster, R.A.; Coomber, B.L. Receptor Tyrosine Kinase Expression Profiles in Canine Cutaneous and Subcutaneous Mast Cell Tumors. Vet. Pathol. 2016, 53, 545–558. [Google Scholar] [CrossRef]
- Webster, J.D.; Yuzbasiyan-Gurkan, V.; Miller, R.A.; Kaneene, J.B.; Kiupel, M. Cellular Proliferation in Canine Cutaneous Mast Cell Tumors: Associations with c-KIT and Its Role in Prognostication. Vet. Pathol. 2007, 44, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Kiupel, M.; Farrelly, J.; Cohen, R.; Olmsted, G.; Kirpensteijn, J.; Brocks, B.; Post, G. Recurrence rates and clinical outcome for dogs with grade II mast cell tumours with a low AgNOR count and Ki67 index treated with surgery alone. Vet. Comp. Oncol. 2017, 15, 36–45. [Google Scholar] [CrossRef] [PubMed]
- de Nardi, A.B.; Horta, R.d.S.; Fonseca-Alves, C.E.; de Paiva, F.N.; Linhares, L.C.M.; Firmo, B.F.; Sueiro, F.A.R.; de Oliveira, K.D.; Lourenço, S.V.; Strefezzi, R.D.F.; et al. Diagnosis, Prognosis and Treatment of Canine Cutaneous and Subcutaneous Mast Cell Tumors. Cells 2022, 11, 618. [Google Scholar] [CrossRef]
- Meuten, D.J. (Ed.) Tumors in Domestic Animals; Wiley: Hoboken, NJ, USA, 2016. [Google Scholar]
- Book, A.P.; Fidel, J.; Wills, T.; Bryan, J.; Sellon, R.; Mattoon, J. Correlation of Ultrasound Findings, Liver and Spleen Cytology, and Prognosis in the Clinical Staging of High Metastatic Risk Canine Mast Cell Tumors. Vet. Radiol. Ultrasound 2011, 52, 548–554. [Google Scholar] [CrossRef]
- Sulce, M.; Marconato, L.; Martano, M.; Iussich, S.; Dentini, A.; Melega, M.; Miniscalco, B.; Riondato, F. Utility of flow cytometry in canine primary cutaneous and matched nodal mast cell tumor. Vet. J. 2018, 242, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Chiarle, R.; Podda, A.; Prolla, G.; Gong, J.; Thorbecke, G.; Inghirami, G. CD30 in Normal and Neoplastic Cells. Clin. Immunol. 1999, 90, 157–164. [Google Scholar] [CrossRef]
- Bauer, K.; Hadzijusufovic, E.; Cerny-Reiterer, S.; Hoermann, G.; Reifinger, M.; Pirker, A.; Valent, P.; Willmann, M. IL-4 downregulates expression of the target receptor CD30 in neoplastic canine mast cells. Vet. Comp. Oncol. 2017, 15, 1240–1256. [Google Scholar] [CrossRef] [PubMed]
- Uxa, S.; Castillo-Binder, P.; Kohler, R.; Stangner, K.; Müller, G.A.; Engeland, K. Ki-67 gene expression. Cell Death Differ. 2021, 28, 3357–3370. [Google Scholar] [CrossRef]
- Booth, D.G.; Takagi, M.; Sanchez-Pulido, L.; Petfalski, E.; Vargiu, G.; Samejima, K.; Imamoto, N.; Ponting, C.P.; Tollervey, D.; Earnshaw, W.C.; et al. Ki-67 is a PP1-interacting protein that organises the mitotic chromosome periphery. eLife 2014, 3, e01641. [Google Scholar] [CrossRef]
- Mrouj, K.; Andrés-Sánchez, N.; Dubra, G.; Singh, P.; Sobecki, M.; Chahar, D.; Al Ghoul, E.; Aznar, A.B.; Prieto, S.; Pirot, N.; et al. Ki-67 regulates global gene expression and promotes sequential stages of carcinogenesis. Proc. Natl. Acad. Sci. USA 2021, 118, e2026507118. [Google Scholar] [CrossRef] [PubMed]
- Makii, R.; Cook, H.; Louke, D.; Breitbach, J.; Jennings, R.; Premanandan, C.; Green, E.M.; Fenger, J.M. Characterization of WWOX expression and function in canine mast cell tumors and malignant mast cell lines. BMC Vet. Res. 2020, 16, 415. [Google Scholar] [CrossRef]
- Aqeilan, R.I.; Trapasso, F.; Hussain, S.; Costinean, S.; Marshall, D.; Pekarsky, Y.; Hagan, J.P.; Zanesi, N.; Kaou, M.; Stein, G.S.; et al. Targeted deletion of Wwox reveals a tumor suppressor function. Proc. Natl. Acad. Sci. USA 2007, 104, 3949–3954. [Google Scholar] [CrossRef]
- Schrock, M.S.; Batar, B.; Lee, J.; Druck, T.; Ferguson, B.; Cho, J.H.; Akakpo, K.; Hagrass, H.; A Heerema, N.; Xia, F.; et al. Wwox–Brca1 interaction: Role in DNA repair pathway choice. Oncogene 2017, 36, 2215–2227. [Google Scholar] [CrossRef]
- Gamperl, S.; Stefanzl, G.; Willmann, M.; Valent, P.; Hadzijusufovic, E. In vitro effects of histamine receptor 1 antagonists on proliferation and histamine release in canine neoplastic mast cells. Vet. Med. Sci. 2021, 7, 57–68. [Google Scholar] [CrossRef]
- Gonzalez-Maganña, A.; Blanco, F.J. Human PCNA Structure, Function and Interactions. Biomolecules 2020, 10, 570. [Google Scholar] [CrossRef] [PubMed]
- Gill, V.; Leibman, N.; Monette, S.; Craft, D.M.; Bergman, P.J. Prognostic Indicators and Clinical Outcome in Dogs with Subcutaneous Mast Cell Tumors Treated with Surgery Alone: 43 Cases. J. Am. Anim. Hosp. Assoc. 2020, 56, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Ohashi, E.; Nakagawa, T.; Mochizuki, M.; Nishimura, R.; Sasaki, N. Role of β1 Integrins in Adhesion of Canine Mastocytoma Cells to Extracellular Matrix Proteins. J. Vet. Med. Sci. 2007, 69, 495–499. [Google Scholar] [CrossRef][Green Version]
- Houtman, R.; Koster, A.S.; Nijkamp, F.P. Integrin VLA-5: Modulator and activator of mast cells. Clin. Exp. Allergy 2001, 31, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.S.; Jones, R.E.; Ransom, R.C.; Longaker, M.T.; Norton, J.A. The evolving relationship of wound healing and tumor stroma. JCI Insight 2018, 3, e99911. [Google Scholar] [CrossRef] [PubMed]
- Hadzijusufovic, E.; Rebuzzi, L.; Gleixner, K.V.; Ferenc, V.; Peter, B.; Kondo, R.; Gruze, A.; Kneidinger, M.; Krauth, M.-T.; Mayerhofer, M.; et al. Targeting of heat-shock protein 32/heme oxygenase-1 in canine mastocytoma cells is associated with reduced growth and induction of apoptosis. Exp. Hematol. 2008, 36, 1461–1470. [Google Scholar] [CrossRef]
- Kondo, R.; Gleixner, K.V.; Mayerhofer, M.; Vales, A.; Gruze, A.; Samorapoompichit, P.; Greish, K.; Krauth, M.-T.; Aichberger, K.J.; Pickl, W.F.; et al. Identification of heat shock protein 32 (Hsp32) as a novel survival factor and therapeutic target in neoplastic mast cells. Blood 2007, 110, 661–669. [Google Scholar] [CrossRef]
- Kamal, A.; Thao, L.; Sensintaffar, J.; Zhang, L.; Boehm, M.F.; Fritz, L.C.; Burrows, F.J. A high-affinity conformation of Hsp90 confers tumour selectivity on Hsp90 inhibitors. Nature 2003, 425, 407–410. [Google Scholar] [CrossRef]
- Whitesell, L.; Lindquist, S.L. HSP90 and the chaperoning of cancer. Nat. Rev. Cancer 2005, 5, 761–772. [Google Scholar] [CrossRef]
- Xu, W.; Neckers, L. Targeting the Molecular Chaperone Heat Shock Protein 90 Provides a Multifaceted Effect on Diverse Cell Signaling Pathways of Cancer Cells. Clin. Cancer Res. 2007, 13, 1625–1629. [Google Scholar] [CrossRef]
- Lin, T.-Y.; Bear, M.; Du, Z.; Foley, K.P.; Ying, W.; Barsoum, J.; London, C. The novel HSP90 inhibitor STA-9090 exhibits activity against Kit-dependent and -independent malignant mast cell tumors. Exp. Hematol. 2008, 36, 1266–1277. [Google Scholar] [CrossRef]
- Akgul, C. Mcl-1 is a potential therapeutic target in multiple types of cancer. Cell. Mol. Life Sci. 2009, 66, 1326–1336. [Google Scholar] [CrossRef]
- Moulding, D.A.; Giles, R.V.; Spiller, D.G.; White, M.R.; Tidd, D.M.; Edwards, S.W. Apoptosis is rapidly triggered by antisense depletion of MCL-1 in differentiating U937 cells. Blood 2000, 96, 1756–1763. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10961874 (accessed on 29 October 2023). [CrossRef] [PubMed]
- Maekawa, N.; Konnai, S.; Ikebuchi, R.; Okagawa, T.; Adachi, M.; Takagi, S.; Kagawa, Y.; Nakajima, C.; Suzuki, Y.; Murata, S.; et al. Expression of PD-L1 on Canine Tumor Cells and Enhancement of IFN-γ Production from Tumor-Infiltrating Cells by PD-L1 Blockade. PLoS ONE 2014, 9, e98415. [Google Scholar] [CrossRef] [PubMed]
- Geng, L.; Huang, D.; Liu, J.; Qian, Y.; Deng, J.; Li, D.; Hu, Z.; Zhang, J.; Jiang, G.; Zheng, S. B7-H1 up-regulated expression in human pancreatic carcinoma tissue associates with tumor progression. J. Cancer Res. Clin. Oncol. 2008, 134, 1021–1027. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.M.; Hwu, W.-J.; Topalian, S.L.; Hwu, P.; Drake, C.G.; Camacho, L.H.; Kauh, J.; Odunsi, K.; et al. Safety and Activity of Anti-PD-L1 Antibody in Patients with Advanced Cancer. N. Engl. J. Med. 2012, 366, 2455–2465. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Gao, X.; Londono, D.; Devroy, S.E.; Mauldin, K.N.; Frankel, J.T.; Brandon, J.M.; Zhang, D.; Li, Q.-Z.; Dobbs, M.B.; et al. Genome-wide association studies of adolescent idiopathic scoliosis suggest candidate susceptibility genes. Hum. Mol. Genet. 2011, 20, 1456–1466. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.G.; Bamford, S.; Jubb, H.C.; Sondka, Z.; Beare, D.M.; Bindal, N.; Boutselakis, H.; Cole, C.G.; Creatore, C.; Dawson, E.; et al. COSMIC: The Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019, 47, D941–D947. [Google Scholar] [CrossRef]
- Arendt, M.L.; Melin, M.; Tonomura, N.; Koltookian, M.; Courtay-Cahen, C.; Flindall, N.; Bass, J.; Boerkamp, K.; Megquir, K.; Youell, L.; et al. Genome-Wide Association Study of Golden Retrievers Identifies Germ-Line Risk Factors Predisposing to Mast Cell Tumours. PLoS Genet. 2015, 11, e1005647. [Google Scholar] [CrossRef]
- Olsson, M.; Meadows, J.R.S.; Truvé, K.; Pielberg, G.R.; Puppo, F.; Mauceli, E.; Quilez, J.; Tonomura, N.; Zanna, G.; Docampo, M.J.; et al. A Novel Unstable Duplication Upstream of HAS2 Predisposes to a Breed-Defining Skin Phenotype and a Periodic Fever Syndrome in Chinese Shar-Pei Dogs. PLoS Genet. 2011, 7, e1001332. [Google Scholar] [CrossRef]
- Lv, Y.S.; Yao, Y.S.; Rong, L.; Lin, M.E.; Deng, B.H.; Xie, Y.; Huang, H.; Lin, T.X.; Xu, K.W.; Huang, J. Intravesical hyaluronidase causes chronic cystitis in a rat model: A potential model of bladder pain syndrome/interstitial cystitis. Int. J. Urol. 2014, 21, 601–607. [Google Scholar] [CrossRef]
- Nakaichi, M.; Takeshita, Y.; Okuda, M.; Nakamoto, Y.; Itamoto, K.; Une, S.; Sasaki, N.; Kadosawa, T.; Takahashi, T.; Taura, Y. Expression of the MDR1 Gene and P-Glycoprotein in Canine Mast Cell Tumor Cell Lines. J. Vet. Med. Sci. 2007, 69, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Spritz, R.A. Molecular Basis of Human Piebaldism. J. Investig. Dermatol. 1994, 103, 137S–140S. [Google Scholar] [CrossRef] [PubMed]
- Phung, B.; Steingrímsson, E.; Rönnstrand, L. Differential activity of c-KIT splice forms is controlled by extracellular peptide insert length. Cell Signal. 2013, 25, 2231–2238. [Google Scholar] [CrossRef]
- Morgan, E.; O’Connell, K.; Thomson, M.; Griffin, A. Canine T cell lymphoma treated with lomustine, vincristine, procarbazine, and prednisolone chemotherapy in 35 dogs. Vet. Comp. Oncol. 2018, 16, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.; Ghosh, J.; Kapur, R. Mastocytosis: A mutated KIT receptor induced myeloproliferative disorder. Oncotarget 2015, 6, 18250–18264. [Google Scholar] [CrossRef] [PubMed]
- Lennartsson, J.; Rönnstrand, L. Stem Cell Factor Receptor/c-Kit: From Basic Science to Clinical Implications. Physiol. Rev. 2012, 92, 1619–1649. [Google Scholar] [CrossRef]
- Letard, S.; Yang, Y.; Hanssens, K.; Palmérini, F.; Leventhal, P.S.; Guéry, S.; Moussy, A.; Kinet, J.-P.; Hermine, O.; Dubreuil, P. Gain-of-Function Mutations in the Extracellular Domain of KIT Are Common in Canine Mast Cell Tumors. Mol. Cancer Res. 2008, 6, 1137–1145. [Google Scholar] [CrossRef]
- Mochizuki, H.; Thomas, R.; Moroff, S.; Breen, M. Genomic profiling of canine mast cell tumors identifies DNA copy number aberrations associated with KIT mutations and high histological grade. Chromosom. Res. 2017, 25, 129–143. [Google Scholar] [CrossRef]
- Chen, P.; Marconato, L.; Sabattini, S.; Kiupel, M. Mutations in Exons 8 and 11 of c-kit Gene in Canine Subcutaneous Mast Cell Tumors and Their Association with Cell Proliferation. Vet. Sci. 2022, 9, 493. [Google Scholar] [CrossRef]
- Zhou, X.; Santos, G.S.; Zhan, Y.; Oliveira, M.M.S.; Rezaei, S.; Singh, M.; Peuget, S.; Westerberg, L.S.; Johnsen, J.I.; Selivanova, G. Mutant p53 gain of function mediates cancer immune escape that is counteracted by APR-246. Br. J. Cancer 2022, 127, 2060–2071. [Google Scholar] [CrossRef]
- Vozdova, M.; Kubickova, S.; Fictum, P.; Fröhlich, J.; Jelinek, F.; Rubes, J. Prevalence and prognostic value of c-kit and TP53 mutations in canine mast cell tumours. Vet. J. 2019, 247, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Biasoli, D.; Compston-Garnett, L.; Ricketts, S.L.; Birand, Z.; Courtay-Cahen, C.; Fineberg, E.; Arendt, M.; Boerkamp, K.; Melin, M.; Koltookian, M.; et al. A synonymous germline variant in a gene encoding a cell adhesion molecule is associated with cutaneous mast cell tumour development in Labrador and Golden Retrievers. PLoS Genet. 2019, 15, e1007967. [Google Scholar] [CrossRef]
- Amagai, Y.; Tanaka, A.; Matsuda, A.; Oida, K.; Jung, K.; Nishikawa, S.; Jang, H.; Ishizaka, S.; Matsuda, H. Increased Expression of the Antiapoptotic Protein MCL1 in Canine Mast Cell Tumors. J. Vet. Med. Sci. 2013, 75, 971–974. [Google Scholar] [CrossRef] [PubMed]
- Śmiech, A.; Ślaska, B.; Surdyka, M.; Grzybowska-Szatkowska, L.; Łopuszyński, W.; Różańska, D. Identification of additional mitochondrial DNA mutations in canine mast cell tumours. Acta Vet. Scand. 2015, 58, 28. [Google Scholar] [CrossRef] [PubMed]
- Destexhe, E.; Lespagnard, L.; Degeyter, M.; Heymann, R.; Coignoul, F. Immunohistochemical Identification of Myoepithelial, Epithelial, and Connective Tissue Cells in Canine Mammary Tumors. Vet. Pathol. 1993, 30, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Conrad, D.; Kehl, A.; Müller, T.; Klopfleisch, R.; Aupperle-Lellbach, H. Immunohistochemical and Molecular Genetic Analysis of Canine Digital Mast Cell Tumours. Animals 2023, 13, 1694. [Google Scholar] [CrossRef] [PubMed]
- Brocks, B.A.W.; Bertram, C.A.; Bartel, A.; Kirpensteijn, J.; Collins-Webb, A.; Catlin, C.; Thaiwong, T.; Kiupel, M. Internal Tandem Duplication of Exon 8 of c-kit Is Associated With Longer Total Survival in Canine Cutaneous Mast Cell Tumors. Vet. Pathol. 2021, 58, 315–324. [Google Scholar] [CrossRef]
- Jark, P.C.; Mundin, D.B.; de Carvalho, M.; Ferioli, R.B.; A Anai, L.; Marchi, F.A.; Rogatto, S.R.; Laufer-Amorim, R.; Tinucci-Costa, M. Genomic copy number variation associated with clinical outcome in canine cutaneous mast cell tumors. Res. Vet. Sci. 2017, 111, 26–30. [Google Scholar] [CrossRef]
- Giantin, M.; Granato, A.; Baratto, C.; Marconato, L.; Vascellari, M.; Morello, E.M.; Vercelli, A.; Mutinelli, F.; Dacasto, M. Global Gene Expression Analysis of Canine Cutaneous Mast Cell Tumor: Could Molecular Profiling Be Useful for Subtype Classification and Prognostication? PLoS ONE 2014, 9, e95481. [Google Scholar] [CrossRef]
- Shubbar, E.; Kovács, A.; Hajizadeh, S.; Parris, T.Z.; Nemes, S.; Gunnarsdóttir, K.; Einbeigi, Z.; Karlsson, P.; Helou, K. Elevated cyclin B2 expression in invasive breast carcinoma is associated with unfavorable clinical outcome. BMC Cancer 2013, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Halasi, M.; Gartel, A.L. FOX(M1) News—It Is Cancer. Mol. Cancer Ther. 2013, 12, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wan, L.; Zhong, J.; Inuzuka, H.; Liu, P.; Sarkar, F.H.; Wei, W. Cdc20: A Potential Novel Therapeutic Target for Cancer Treatment. Curr. Pharm. Des. 2013, 19, 3210–3214. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Pathak, H.B.; Zhang, H.; Zhou, Y.; Einarson, M.B.; Vathipadiekal, V.; Gunewardena, S.; Birrer, M.J.; Godwin, A.K. An RNA Interference Lethality Screen of the Human Druggable Genome to Identify Molecular Vulnerabilities in Epithelial Ovarian Cancer. PLoS ONE 2012, 7, e47086. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, A.M.; Alfaro, A.; Roman-Basaure, E.; Guardado-Estrada, M.; Palma, Í.; Serralde, C.; Medina, I.; Juárez, E.; Bermúdez, M.; Márquez, E.; et al. Mitosis Is a Source of Potential Markers for Screening and Survival and Therapeutic Targets in Cervical Cancer. PLoS ONE 2013, 8, e55975. [Google Scholar] [CrossRef]
- Yun, H.J.; Cho, Y.-H.; Moon, Y.; Park, Y.W.; Yoon, H.-K.; Kim, Y.-J.; Cho, S.-H.; Lee, Y.-I.; Kang, B.-S.; Kim, W.-J.; et al. Transcriptional targeting of gene expression in breast cancer by the promoters of protein regulator of cytokinesis 1 and ribonuclease reductase 2. Exp. Mol. Med. 2008, 40, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.-J.; Guo, J.-J.; Lv, T.-J.; Jin, H.-Y.; Ding, J.-X.; Feng, W.-W.; Zhang, Y.; Hua, K.-Q. Prognostic value of centromere protein-A expression in patients with epithelial ovarian cancer. Tumor Biol. 2013, 34, 2971–2975. [Google Scholar] [CrossRef] [PubMed]
- Garnett, M.J.; Mansfeld, J.; Godwin, C.; Matsusaka, T.; Wu, J.; Russell, P.; Pines, J.; Venkitaraman, A.R. UBE2S elongates ubiquitin chains on APC/C substrates to promote mitotic exit. Nat. Cell Biol. 2009, 11, 1363–1369. [Google Scholar] [CrossRef] [PubMed]
- Baig, R.M.; Mahjabeen, I.; Sabir, M.; Masood, N.; Ali, K.; Malik, F.A.; Kayani, M.A. Mutational spectrum of Gelsolin and its down regulation is associated with breast cancer. Dis. Markers 2013, 34, 71–80. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Liu, L.; Chen, J.; Hu, Q.; Shen, S.; Zhou, Y.; Chen, S.; Xue, C.; Cui, G.; et al. Upregulation of FEN1 Is Associated with the Tumor Progression and Prognosis of Hepatocellular Carcinoma. Dis. Markers 2020, 2020, 2514090. [Google Scholar] [CrossRef]
- Pulz, L.H.; Barra, C.N.; Alexandre, P.A.; Huete, G.C.; Cadrobbi, K.G.; Nishiya, A.T.; de Freitas, S.H.; Fukumasu, H.; Strefezzi, R.F. Identification of two molecular subtypes in canine mast cell tumours through gene expression profiling. PLoS ONE 2019, 14, e0217343. [Google Scholar] [CrossRef] [PubMed]
- Kagan, H.M.; Li, W. Lysyl oxidase: Properties, specificity, and biological roles inside and outside of the cell. J. Cell. Biochem. 2003, 88, 660–672. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 2006, 6, 392–401. [Google Scholar] [CrossRef]
- Gabbiani, G. The myofibroblast in wound healing and fibrocontractive diseases. J. Pathol. 2003, 200, 500–503. [Google Scholar] [CrossRef]
- Valastyan, S.; Weinberg, R.A. Tumor Metastasis: Molecular Insights and Evolving Paradigms. Cell 2011, 147, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Daves, M.H.; Hilsenbeck, S.G.; Lau, C.C.; Man, T.-K. Meta-analysis of multiple microarray datasets reveals a common gene signature of metastasis in solid tumors. BMC Med. Genom. 2011, 4, 56. [Google Scholar] [CrossRef]
- Blacklock, K.B.; Birand, Z.; Biasoli, D.; Fineberg, E.; Murphy, S.; Flack, D.; Bass, J.; Di Palma, S.; Blackwood, L.; McKay, J.; et al. Identification of molecular genetic contributants to canine cutaneous mast cell tumour metastasis by global gene expression analysis. PLoS ONE 2018, 13, e0208026. [Google Scholar] [CrossRef]
- Hansen, C.G.; Bright, N.A.; Howard, G.; Nichols, B.J. SDPR induces membrane curvature and functions in the formation of caveolae. Nature 2009, 11, 807–814. [Google Scholar] [CrossRef]
- Leone, A.; Flatow, U.; VanHoutte, K.; Steeg, P.S. Transfection of human nm23-H1 into the human MDA-MB-435 breast carcinoma cell line: Effects on tumor metastatic potential, colonization and enzymatic activity. Oncogene 1993, 8, 2325–2333. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8395676 (accessed on 30 October 2023).
- Montagner, M.; Enzo, E.; Forcato, M.; Zanconato, F.; Parenti, A.; Rampazzo, E.; Basso, G.; Leo, G.; Rosato, A.; Bicciato, S.; et al. SHARP1 suppresses breast cancer metastasis by promoting degradation of hypoxia-inducible factors. Nature 2012, 487, 380–384. [Google Scholar] [CrossRef]
- Chen, D.; Sun, Y.; Wei, Y.; Zhang, P.; Rezaeian, A.H.; Teruya-Feldstein, J.; Gupta, S.; Liang, H.; Lin, H.-K.; Hung, M.-C.; et al. LIFR is a breast cancer metastasis suppressor upstream of the Hippo-YAP pathway and a prognostic marker. Nat. Med. 2012, 18, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Fenger, J.M.; Bear, M.D.; Volinia, S.; Lin, T.-Y.; Harrington, B.K.; A London, C.; Kisseberth, W.C. Overexpression of miR-9 in mast cells is associated with invasive behavior and spontaneous metastasis. BMC Cancer 2014, 14, 84. [Google Scholar] [CrossRef] [PubMed]
- Miyake, M.; Nakano, K.; Ieki, Y.; Adachi, M.; Huang, C.L.; Itoi, S.; Koh, T.; Taki, T. Motility related protein 1 (MRP-1/CD9) expression: Inverse correlation with metastases in breast cancer. Cancer Res. 1995, 55, 4127–4131. Available online: http://www.ncbi.nlm.nih.gov/pubmed/7664290 (accessed on 30 October 2023). [PubMed]
- Mouneimne, G.; Hansen, S.D.; Selfors, L.M.; Petrak, L.; Hickey, M.M.; Gallegos, L.L.; Simpson, K.J.; Lim, J.; Gertler, F.B.; Hartwig, J.H.; et al. Differential Remodeling of Actin Cytoskeleton Architecture by Profilin Isoforms Leads to Distinct Effects on Cell Migration and Invasion. Cancer Cell 2012, 22, 615–630. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Ströbel, P.; Marx, A.; Hofmann, I. Plakophilin-associated RNA-binding proteins in prostate cancer and their implications in tumor progression and metastasis. Virchows Arch. 2013, 463, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Akl, M.R.; Nagpal, P.; Ayoub, N.M.; Prabhu, S.A.; Gliksman, M.; Tai, B.; Hatipoglu, A.; Goy, A.; Suh, K.S. Molecular and clinical profiles of syndecan-1 in solid and hematological cancer for prognosis and precision medicine. Oncotarget 2015, 6, 28693–28715. [Google Scholar] [CrossRef]
- Roux, K.J.; Amici, S.A.; Fletcher, B.S.; Notterpek, L. Modulation of Epithelial Morphology, Monolayer Permeability, and Cell Migration by Growth Arrest Specific 3/Peripheral Myelin Protein 22. Mol. Biol. Cell 2005, 16, 1142–1151. [Google Scholar] [CrossRef]
- Brameier, M.; Herwig, A.; Reinhardt, R.; Walter, L.; Gruber, J. Human box C/D snoRNAs with miRNA like functions: Expanding the range of regulatory RNAs. Nucleic Acids Res. 2011, 39, 675–686. [Google Scholar] [CrossRef]
- Mileshkin, L.R.; Moore, K.N.; Barnes, E.H.; Gebski, V.; Narayan, K.; King, M.T.; Bradshaw, N.; Lee, Y.C.; Diamante, K.; Fyles, A.W.; et al. Adjuvant chemotherapy following chemoradiotherapy as primary treatment for locally advanced cervical cancer versus chemoradiotherapy alone (OUTBACK): An international, open-label, randomised, phase 3 trial. Lancet Oncol. 2023, 24, 468–482. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Cheng, Y.; Zhou, X.; Lee, K.H.; Nakagawa, K.; Niho, S.; Tsuji, F.; Linke, R.; Rosell, R.; Corral, J.; et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): A randomised, open-label, phase 3 trial. Lancet Oncol. 2017, 18, 1454–1466. [Google Scholar] [CrossRef]
- Habanjar, O.; Diab-Assaf, M.; Caldefie-Chezet, F.; Delort, L. The Impact of Obesity, Adipose Tissue, and Tumor Microenvironment on Macrophage Polarization and Metastasis. Biology 2022, 11, 339. [Google Scholar] [CrossRef] [PubMed]
- Alegría-Torres, J.A.; Baccarelli, A.; Bollati, V.; Huang, Q.; A Vucic, E.; Brown, C.J.; Lam, W.L.; A Lewis, K.; O Tollefsbol, T.; Bartosch, C.; et al. Epigenetics lifestyle. Epigenomics 2011, 3, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Simó-Riudalbas, L.; Esteller, M. Cancer genomics identifies disrupted epigenetic genes. Hum. Genet. 2014, 133, 713–725. [Google Scholar] [CrossRef]
- Kabekkodu, S.P.; Shukla, V.; Varghese, V.K.; Souza, J.D.; Chakrabarty, S.; Satyamoorthy, K. Clustered miRNAs and their role in biological functions and diseases. Biol. Rev. 2018, 93, 1955–1986. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Garofalo, M.; Crescenti, D.; Rizzi, N.; Brunialti, E.; Vingiani, A.; Belotti, P.; Sposito, C.; Franzè, S.; Cilurzo, F.; et al. Transplantation of autologous extracellular vesicles for cancer-specific targeting. Theranostics 2021, 11, 2034–2047. [Google Scholar] [CrossRef] [PubMed]
- Jahan, S.; Mukherjee, S.; Ali, S.; Bhardwaj, U.; Choudhary, R.K.; Balakrishnan, S.; Naseem, A.; Mir, S.A.; Banawas, S.; Alaidarous, M.; et al. Pioneer Role of Extracellular Vesicles as Modulators of Cancer Initiation in Progression, Drug Therapy, and Vaccine Prospects. Cells 2022, 11, 490. [Google Scholar] [CrossRef]
- LeBleu, V.S.; Kalluri, R. Exosomes as a Multicomponent Biomarker Platform in Cancer. Trends Cancer 2020, 6, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Lobb, R.J.; Lima, L.G.; Möller, A. Exosomes: Key mediators of metastasis and pre-metastatic niche formation. Semin. Cell Dev. Biol. 2017, 67, 3–10. [Google Scholar] [CrossRef]
- Lucchetti, D.; Tenore, C.R.; Colella, F.; Sgambato, A. Extracellular Vesicles and Cancer: A Focus on Metabolism, Cytokines, and Immunity. Cancers 2020, 12, 171. [Google Scholar] [CrossRef]
- Ekström, K.; Valadi, H.; Sjöstrand, M.; Malmhäll, C.; Bossios, A.; Eldh, M.; Lötvall, J. Characterization of mRNA and microRNA in human mast cell-derived exosomes and their transfer to other mast cells and blood CD34 progenitor cells. J. Extracell. Vesicles 2012, 1, 18389. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, C.; Zamarian, V.; Stefanello, D.; Ferrari, R.; Auletta, L.; Milanesi, S.; Mauri, S.; Grieco, V.; Ceciliani, F.; Lecchi, C. Plasma small extracellular vesicles from dogs affected by cutaneous mast cell tumors deliver high levels of miR-21-5p. Front. Vet. Sci. 2023, 9, 1083174. [Google Scholar] [CrossRef]
- He, B.; Zhao, Z.; Cai, Q.; Zhang, Y.; Zhang, P.; Shi, S.; Xie, H.; Peng, X.; Yin, W.; Tao, Y.; et al. miRNA-based biomarkers, therapies, and resistance in Cancer. Int. J. Biol. Sci. 2020, 16, 2628–2647. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, P.; Crepps, M.P.; Stahr, N.A.; Kretzschmar, W.P.; Harris, H.C.; Prasad, N.; Levy, S.E.; Smith, B.F. Identification of canine circulating miRNAs as tumor biospecific markers using Next-Generation Sequencing and Q-RT-PCR. Biochem. Biophys. Rep. 2021, 28, 101106. [Google Scholar] [CrossRef]
- Jain, M.; Ingole, S.D.; Deshmukh, R.S.; Bharucha, S.V.; Nagvekar, A.S.; Gaikwad, R.V.; Kharde, S.D. CEA, CA 15-3, and miRNA expression as potential biomarkers in canine mammary tumors. Chromosom. Res. 2021, 29, 175–188. [Google Scholar] [CrossRef]
- Mayoral, R.J.; Pipkin, M.E.; Pachkov, M.; van Nimwegen, E.; Rao, A.; Monticelli, S. MicroRNA-221–222 Regulate the Cell Cycle in Mast Cells. J. Immunol. 2009, 182, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Mayoral, R.J.; Deho, L.; Rusca, N.; Bartonicek, N.; Saini, H.K.; Enright, A.J.; Monticelli, S. MiR-221 Influences Effector Functions and Actin Cytoskeleton in Mast Cells. PLoS ONE 2011, 6, e26133. [Google Scholar] [CrossRef] [PubMed]
- Zamarian, V.; Ferrari, R.; Stefanello, D.; Ceciliani, F.; Grieco, V.; Minozzi, G.; Chiti, L.E.; Arigoni, M.; Calogero, R.; Lecchi, C. miRNA profiles of canine cutaneous mast cell tumours with early nodal metastasis and evaluation as potential biomarkers. Sci. Rep. 2020, 10, 18918. [Google Scholar] [CrossRef]
- Zamarian, V.; Stefanello, D.; Ferrari, R.; Chiti, L.E.; Grieco, V.; DallaCosta, E.; Ceciliani, F.; Lecchi, C. Salivary miR-21 is a potential biomarker for canine mast cell tumors. Vet. Pathol. 2023, 60, 47–51. [Google Scholar] [CrossRef]
- Capodanno, A.; Boldrini, L.; Proietti, A.; Alì, G.; Pelliccioni, S.; Niccoli, C.; D’Incecco, A.; Cappuzzo, F.; Chella, A.; Lucchi, M.; et al. Let-7g and miR-21 expression in non-small cell lung cancer: Correlation with clinicopathological and molecular features. Int. J. Oncol. 2013, 43, 765–774. [Google Scholar] [CrossRef]
- Lee, Y.-N.; Brandal, S.; Noel, P.; Wentzel, E.; Mendell, J.T.; McDevitt, M.A.; Kapur, R.; Carter, M.; Metcalfe, D.D.; Takemoto, C.M. KIT signaling regulates MITF expression through miRNAs in normal and malignant mast cell proliferation. Blood 2011, 117, 3629–3640. [Google Scholar] [CrossRef] [PubMed]
- Garrett, L. Canine mast cell tumors: Diagnosis, treatment, and prognosis. Vet. Med. Res. Rep. 2014, 5, 49–58. [Google Scholar] [CrossRef]
- Séguin, B.; Leibman, N.F.; Bregazzi, V.S.; Ogilvie, G.K.; Powers, B.E.; Dernell, W.S.; Fettman, M.J.; Withrow, S.J. Clinical outcome of dogs with grade-II mast cell tumors treated with surgery alone: 55 cases (1996–1999). J. Am. Vet. Med. Assoc. 2001, 218, 1120–1123. [Google Scholar] [CrossRef]
- Dores, C.; Milovancev, M.; Russell, D. Comparison of histologic margin status in low-grade cutaneous and subcutaneous canine mast cell tumours examined by radial and tangential sections. Vet. Comp. Oncol. 2018, 16, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Gajewska, M.; Kwiecień, I.; Rutkowska, E.; Rzepecki, P.; Sułek, K. AgNOR patterns and configurations in adult acute leukemia patients. Central Eur. J. Immunol. 2022, 47, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Marconato, L.; Zorzan, E.; Giantin, M.; Di Palma, S.; Cancedda, S.; Dacasto, M. Concordance of c-kit Mutational Status in Matched Primary and Metastatic Cutaneous Canine Mast Cell Tumors at Baseline. J. Vet. Intern. Med. 2014, 28, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Amagai, Y.; Tanaka, A.; Matsuda, A.; Jung, K.; Oida, K.; Nishikawa, S.; Jang, H.; Matsuda, H. Heterogeneity of internal tandem duplications in the c-kit of dogs with multiple mast cell tumours. J. Small Anim. Pract. 2013, 54, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Bloom, F. Effect of Cortisone on Mast Cell Tumors (Mastocytoma) of the Dog. Exp. Biol. Med. 1952, 79, 651–654. [Google Scholar] [CrossRef]
- McCaw, D.L.; Miller, M.A.; Ogilvie, G.K.; Withrow, S.J.; Brewer, W.G.; Klein, M.K.; Bell, F.W.; Anderson, S.K. Response of Canine Mast Cell Tumors to Treatment With Oral Prednisone. J. Vet. Intern. Med. 1994, 8, 406–408. [Google Scholar] [CrossRef]
- Hahn, K.A.; Oglivie, G.; Rusk, T.; Devauchelle, P.; Leblanc, A.; Legendre, A.; Powers, B.; Leventhal, P.S.; Kinet, J.-P.; Palmerini, F.; et al. Masitinib is Safe and Effective for the Treatment of Canine Mast Cell Tumors. J. Veter. Intern. Med. 2008, 22, 1301–1309. [Google Scholar] [CrossRef]
- Rassnick, K.M.; Moore, A.S.; Williams, L.E.; London, C.A.; Kintzer, P.P.; Engler, S.J.; Cotter, S.M. Treatment of Canine Mast Cell Tumors with CCNU (Lomustine). J. Vet. Intern. Med. 1999, 13, 601. [Google Scholar] [CrossRef] [PubMed]
- Chun, R. Cancer Chemotherapy. In Withrow & MacEwen’s Small Animal Clinical Oncology; Elsevier: Amsterdam, The Netherlands, 2007; pp. 163–192. [Google Scholar]
- Hernández, I.B.; Kromhout, J.Z.; Teske, E.; Hennink, W.E.; van Nimwegen, S.A.; Oliveira, S. Molecular targets for anticancer therapies in companion animals and humans: What can we learn from each other? Theranostics 2021, 11, 3882–3897. [Google Scholar] [CrossRef]
- London, C.; Mathie, T.; Stingle, N.; Clifford, C.; Haney, S.; Klein, M.K.; Beaver, L.; Vickery, K.; Vail, D.M.; Hershey, B.; et al. Preliminary evidence for biologic activity of toceranib phosphate (Palladia®) in solid tumours. Vet. Comp. Oncol. 2012, 10, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Papaetis, G.S.; Syrigos, K.N. Sunitinib. BioDrugs 2009, 23, 377–389. [Google Scholar] [CrossRef]
- Tozon, N.; Tratar, U.L.; Znidar, K.; Sersa, G.; Teissie, J.; Cemazar, M. Operating Procedures of the Electrochemotherapy for Treatment of Tumor in Dogs and Cats. J. Vis. Exp. 2016, 116, e54760. [Google Scholar] [CrossRef]
- Mir, L.M.; Gehl, J.; Sersa, G.; Collins, C.G.; Garbay, J.-R.; Billard, V.; Geertsen, P.F.; Rudolf, Z.; O’sullivan, G.C.; Marty, M. Standard operating procedures of the electrochemotherapy: Instructions for the use of bleomycin or cisplatin administered either systemically or locally and electric pulses delivered by the CliniporatorTM by means of invasive or non-invasive electrodes. Eur. J. Cancer Suppl. 2006, 4, 14–25. [Google Scholar] [CrossRef]
- Frimberger, A.; Moore, A.; LaRue, S.; Gliatto, J.; Bengtson, A. Radiotherapy of incompletely resected, moderately differentiated mast cell tumors in the dog: 37 cases (1989–1993). J. Am. Anim. Hosp. Assoc. 1997, 33, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Haythornthwaite, B. What prognostic information does flow cytometry provide in canine B-cell lymphoma? Vet. Evid. 2022, 7. [Google Scholar] [CrossRef]
- Mason, S.L.; Pittaway, C.; Gil, B.P.; Russak, O.; Westlake, K.; Berlato, D.; Benoit, J.; Morris, J.; Dobson, J.M. Outcomes of adjunctive radiation therapy for the treatment of mast cell tumors in dogs and assessment of toxicity: A multicenter observational study of 300 dogs. J. Vet. Intern. Med. 2021, 35, 2853–2864. [Google Scholar] [CrossRef]
- Mellanby, R.J.; Herrtage, M.E.; Chantry, J.; Dobson, J.M. Sarcoma development after radiotherapy in two dogs. Vet. Comp. Oncol. 2003, 1, 113–119. [Google Scholar] [CrossRef]
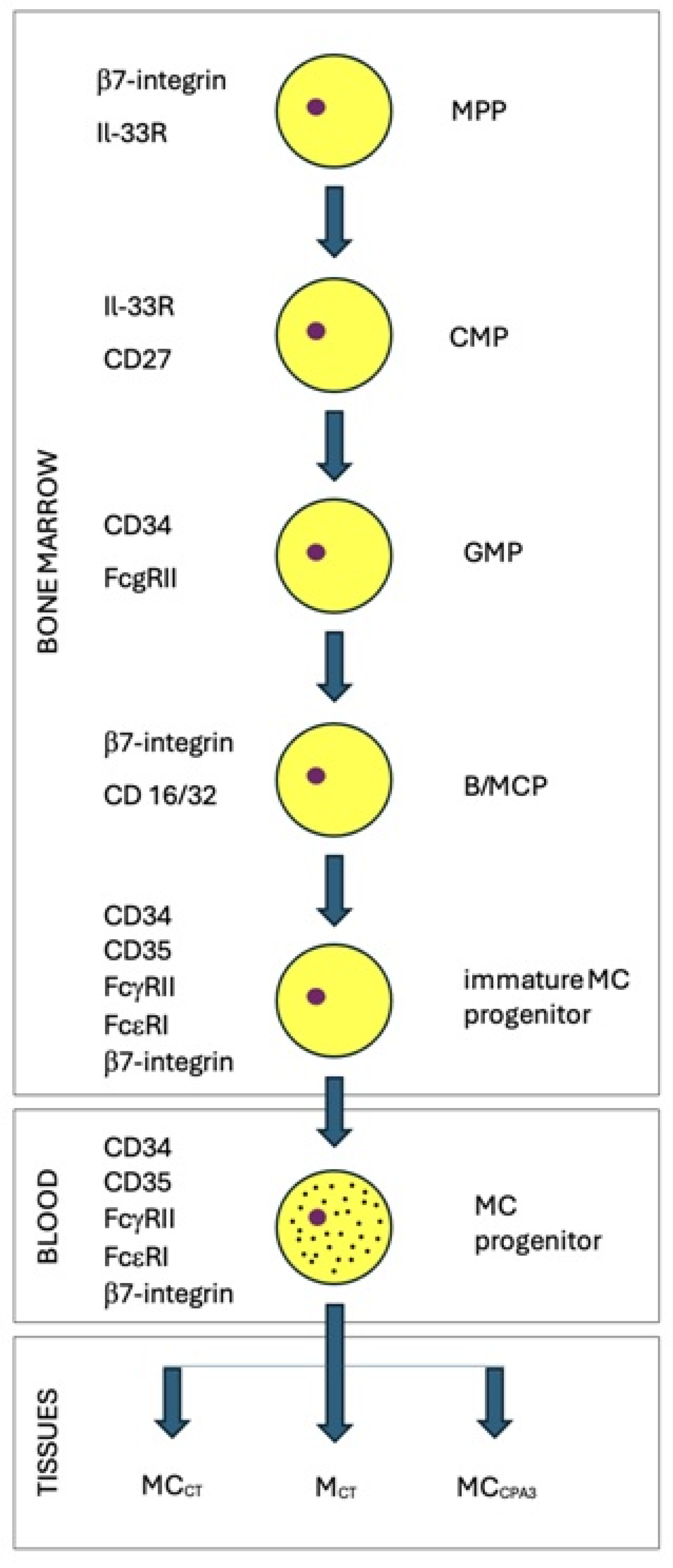
| Marker | Type | Protein Function |
|---|---|---|
| CD16 (FcγRIII, FCGR3A) | Migration | Receptor |
| CD32 (FcγRII, FCGR2) | Migration | Receptor |
| CD34 | Migration, Tissue specific | Adhesion molecule |
| CD63 | Activation | Receptor |
| ENPP3 (CD203c) | Activation | Receptor |
| FCER1 (IgE receptor, FCεRI) | Maturation | Receptor |
| ITGA4 (integrin α4, CD49d) | Migration | Receptor |
| ITGB7 (integrin β7) | Maturation, Tissue specific | Receptor |
| KIT (CD117, c-Kit) | General | Receptor |
| VCAM1 (CD106) | Migration, Tissue specific | Receptor |
| Protein | Function |
|---|---|
| CD30/Ki-1 antigen | A marker of Hodgkin’s disease and anaplastic lymphoma kinase (ALK) cells [183]. It has been shown that CD30 is expressed in human mastocytosis cells. CD30 expression in canine mastocytosis is inhibited by Il-4, which inhibits MCT cell proliferation [184]. |
| Ki67 protein (encoded by MKI67 gene) | The Ki67 protein is a general marker of cancer cells. Its synthesis is limited to G2 and M cell cycle phases [185]. The Ki67 protein has two isoforms (320 and 359 kDa), both of which bind and stabilize mitotic chromosomes [186]. Ki67 is an important factor for cancer development [187]. |
| WWOX protein (oxidoreductase with a WW domain) | WWOX is a tumor suppressor protein (46 kDa) in dogs [188] and mice [189]. The WWOX protein is involved in DNA repair after ionizing radiation [190]. In mastocytoma cells, the level of WWOX is always strongly reduced [188]. |
| Histamine H1 receptor (HR1) antagonists | HR1 antagonists (loratidine, terfenadine) inhibit the proliferation and reduce the viability of mastocytoma cells. Desloraphidine, rupatadine, and cyproheptadine are particularly effective (in higher concentrations) [191]. |
| Proliferating cell nuclear antigen (PCNA) | PCNA is involved in DNA replication and DNA repair. PCNA has a PCNA-interacting protein box (PIP box) motif and forms a trimer, which slides along the DNA chain [192]. In MCT, an increased expression of PCNA is a poor prognostic factor [193]. |
| Integrins | MCT cells bind collagen, fibronectin, and intercellular matrix laminin via extracellular matrix (ECM) β1 and α1-6 integrins [194]. In particular, VLA5 integrins are the activators of MCT [195]. Changes in ECM are generated by cancer-associated fibroblast (CAF) cells, and the structure of ECM in mastocytoma is similar to the structure of ECM in the stroma of a healing wound [196]. |
| Hsp32 (heme oxygenase 1) | Hsp32 is synthesized by human cancerous mastocytes and dog MTCs [197]. Hsp32 protein is a cell survival factor. Its inhibitors include pegylated zinc protoporphyrin (PEG-ZnPP) and midostaurin, which inhibit tumor growth and induce apoptosis in human SM cells [198]. |
| Hsp90 | The protein is a chaperone, and its levels are elevated in tumors [199]. Hsp90 is also required for cancer cell survival [200]. Geldanamycin and its derivatives are the inhibitors of Hsp90 [201]. At low concentrations, Ganetespib (STA9090) induces the apoptosis of mastocytoma cells [202]. |
| p53 | The protein is expressed in mastocytoma cells, with the highest expression seen in Patnaik’s grade I and the lowest in grade II. |
| MCL1 | The anti-apoptotic protein MCL1 of the BCL2 family is present in MCT cells (independent of KIT mutations) [203]. Downregulation of MCL1 protein expression in tumors increases their susceptibility to chemotherapy. In hematopoietic U937 cells, downregulation of MCL1 via antisense strategy causes apoptosis [204]. |
| Programmed cell death ligand 1 (PD-L1) | The PD-L1 protein ligand of the programmed cell death 1 (PD-1) immunoinhibitory receptor is expressed in 66% of cases of mastocytoma and other canine cancers [205]. This is a poor prognostic factor. The PD-1 receptor causes T-cell infiltration of tumors, while PD-L1 inhibits T-cell function. Anti-PD-L1 antibodies reactivate T cells and increase IFN-γ production (also in human pancreatic cancer) [206]. The blocking of PD-L1 by antibodies can cause tumor regression [207]. |
| Down syndrome cell adhesion molecule (DSCAM) | Cell surface protein DSCAM is important for nervous system development. Mutations in humans are also associated with Down syndrome, Hirschsprung’s disease, and idiopathic scoliosis [208]. The DSCAM gene of Labrador dogs is located on chromosome 31; its mutations increase the risk of MCT up to 1.66 times. Alterations in the DSCAM gene have been described in 40 types of different cancers [209]. |
| Proteins involved in hyaluronic acid metabolism | Hyaluronic acid metabolism genes (in golden retrievers) contain nucleotide polymorphisms on chromosome 14 (for HYAL4, HYALP1, and SPAM1 genes) and on chromosome 20 (for IPK1, HYAL1-3, and GNAI2 genes) [210]. Defects in hyaluronate metabolism in the skin are responsible for the risk of MCT in Shar-Pei dogs [211]. Hyaluronate breakdown products cause mast cell activation and migration [212]. |
| Multi-drug resistance protein 1 (MDR1) | The MDR1 gene encodes a glycoprotein, which is part of the pump responsible for removing foreign substances from the cell. It also removes exogenously administered drugs previously taken up by the cell. Inhibition of MDR1 gene expression may be useful in chemotherapy [213]. |
| Gene/DNA Segment | Genetic Variant/Type of Mutation |
|---|---|
| KIT | Germline mutations in the KIT gene are observed infrequently, rather involving cancers other than MCT. Only KIT gene ITD mutations in exon 11 can be germline in MCT [221]. |
| TP53 | TP53 mutations are present in 14.6% of MCTs [222]. |
| GNB1 | In dogs, mutations in GNB1 have been found in cutaneous and subcutaneous MCTs, with a trend toward positive prognosis [223]. |
| DSCAM | Genetic variant rs850678541 inhibiting DSCAM protein synthesis [224]. |
| Single-nucleotide polymorphisms (SNPs) | SNPs in the HYAL1-4, SPAM1, and GNAI2 genes play a role in mast cell tumor development [210]. |
| MCL1 | Overexpression of the myeloid target leukemia (MCL1) gene is observed in many cancers, including in mastocytoma. It encodes a labile BCL2 family protein located in the mitochondria [225]. |
| Mitochondrial D-loop sequence | D-loop mutations in mitochondrial DNA are present in 47% of dogs with MCT and are usually homoplasmic. Six haplotypes of mitochondrial DNA sequences have been described in MCT cells [226]. |
| Gene | Product |
|---|---|
| CCNB | Cyclin B is important for the transition from the G2 phase to mitosis. CCNB is an oncogene, which is important in the process of metastasis [232]. |
| FOXM1 | Encodes a transcription factor with a forkhead domain. FOXM1 has high expression in proliferating tumor cells [233]. |
| CDC20 | Encodes a karyokinetic spindle protein. CDC20 is an oncogene, which can initiate apoptosis [234]. |
| CDCA8 | Encodes a regulator of mitosis in the centromeric CPC complex. |
| NUF2 | Encodes a protein of the NDC80 complex in the kinetochore. Silencing its expression results in apoptosis [235]. |
| NUSAP1 | Encodes a karyokinetic spindle protein, which determines the survival of cancer cells [236]. |
| PRC1 | Encodes a protein regulator of cytokinesis, which is present in the G2 and M phases. Overexpressed in neurons [237]. |
| CENPP | Encodes a centromere protein, plays a role in kinetochore function and during mitosis [238]. |
| UBE2S | Encodes ubiquitin-conjugating enzyme and plays a role in mitosis [239]. |
| GSN | Encodes an anti-oncogene, which plays a role in apoptosis. The only gene with lower expression in MCT and in many other cancers [240]. |
| FEN1 | Encodes an endonuclease, which plays a role in DNA synthesis and is an anti-oncogene [241]. |
| Genes | Product |
|---|---|
| Keratin genes | Genes encoding keratin, mainly KRT1, KRT5, and KRT15 genes [248]. |
| SDPR | Serum deprivation response protein. SDPR is a metastasis suppressor gene [249]. |
| NME1 | Co-transcription of this gene and the neighboring downstream gene (NME2) generates naturally occurring transcripts (NME1-NME2), which encode a fusion protein comprising sequences sharing identity with each individual gene product [250]. |
| SHARP1 | bHLH transcription factor [251]. |
| LIFR | Leukemia inhibitory factor receptor [252]. |
| PERP | TP53 apoptosis effector. It presents low expression in mouse MCTs overexpressing miR-9 [253]. |
| SBSN and SFN | SBSN (suprabasin) and SFN (stratipin). |
| PSORS1C2 | Epithelial cell protein. |
| CLEC3B | C-type lectin domain family 3 member B. It is an ECM biomarker protein for metastasis. |
| EGR1 | Early growth response 1 gene. Its product blocks heparanase, which increases metastasis. |
| CD9 | Encodes a four-transmembrane protein, which blocks cell motility [254]. |
| BRAF and ADFN | The BRAF gene encodes serine/threonine kinase. The ADFN gene encodes multi-domain protein involved in signaling and the organization of cell junctions during embryogenesis. BRAF and ADFN deficiency increases cell motility. |
| EVL, ARHGEF10, NF2 | Their products activate stress fiber formation [255]. |
| SCRIB | Encodes scribble planar cell polarity protein. |
| PKP1 and DSP | Desmosome proteins plakophilin 1 (PKP1) and desmoplakin (DSP). Their absence promotes metastasis [256]. |
| SDC1 | Encodes a protein, which connects the cytoskeleton to the ECM [257]. |
| PMP22 | Encodes peripheral myelin protein 22. Its overexpression decreases cell motility [258]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zmorzynski, S.; Kimicka-Szajwaj, A.; Szajwaj, A.; Czerwik-Marcinkowska, J.; Wojcierowski, J. Genetic Changes in Mastocytes and Their Significance in Mast Cell Tumor Prognosis and Treatment. Genes 2024, 15, 137. https://doi.org/10.3390/genes15010137
Zmorzynski S, Kimicka-Szajwaj A, Szajwaj A, Czerwik-Marcinkowska J, Wojcierowski J. Genetic Changes in Mastocytes and Their Significance in Mast Cell Tumor Prognosis and Treatment. Genes. 2024; 15(1):137. https://doi.org/10.3390/genes15010137
Chicago/Turabian StyleZmorzynski, Szymon, Aleksandra Kimicka-Szajwaj, Angelika Szajwaj, Joanna Czerwik-Marcinkowska, and Jacek Wojcierowski. 2024. "Genetic Changes in Mastocytes and Their Significance in Mast Cell Tumor Prognosis and Treatment" Genes 15, no. 1: 137. https://doi.org/10.3390/genes15010137
APA StyleZmorzynski, S., Kimicka-Szajwaj, A., Szajwaj, A., Czerwik-Marcinkowska, J., & Wojcierowski, J. (2024). Genetic Changes in Mastocytes and Their Significance in Mast Cell Tumor Prognosis and Treatment. Genes, 15(1), 137. https://doi.org/10.3390/genes15010137





