Winners and Losers of Atlantification: The Degree of Ocean Warming Affects the Structure of Arctic Microbial Communities
Abstract
1. Introduction
2. Materials and Methods
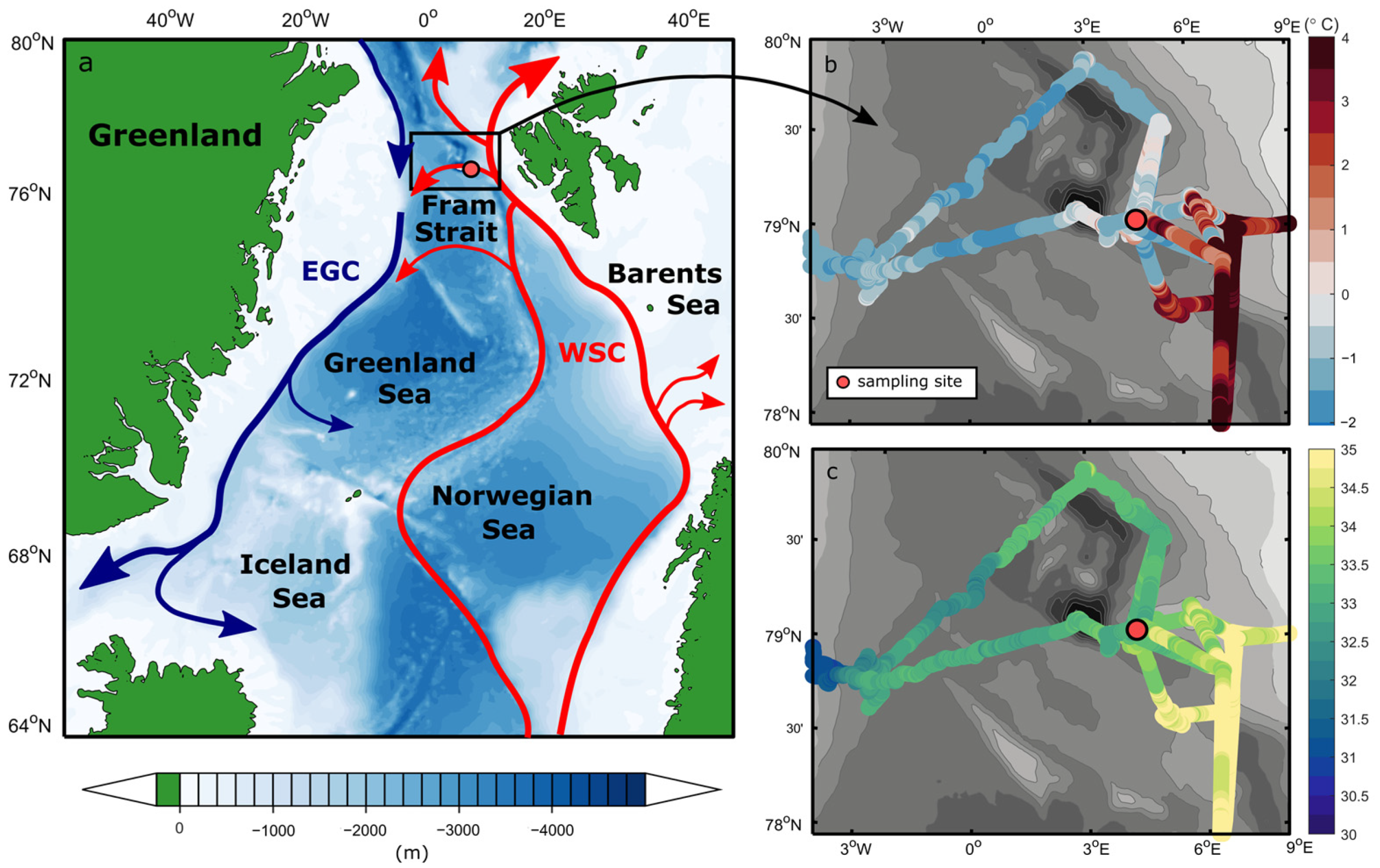
2.1. Study Site and Seawater Physical Properties
2.2. Experimental Set-Up
2.3. Biomass Parameters
2.4. Community Composition and Diversity Analyses
2.5. Data Handling & Analyses
3. Results
3.1. Physical Ocean Properties and Water Masses
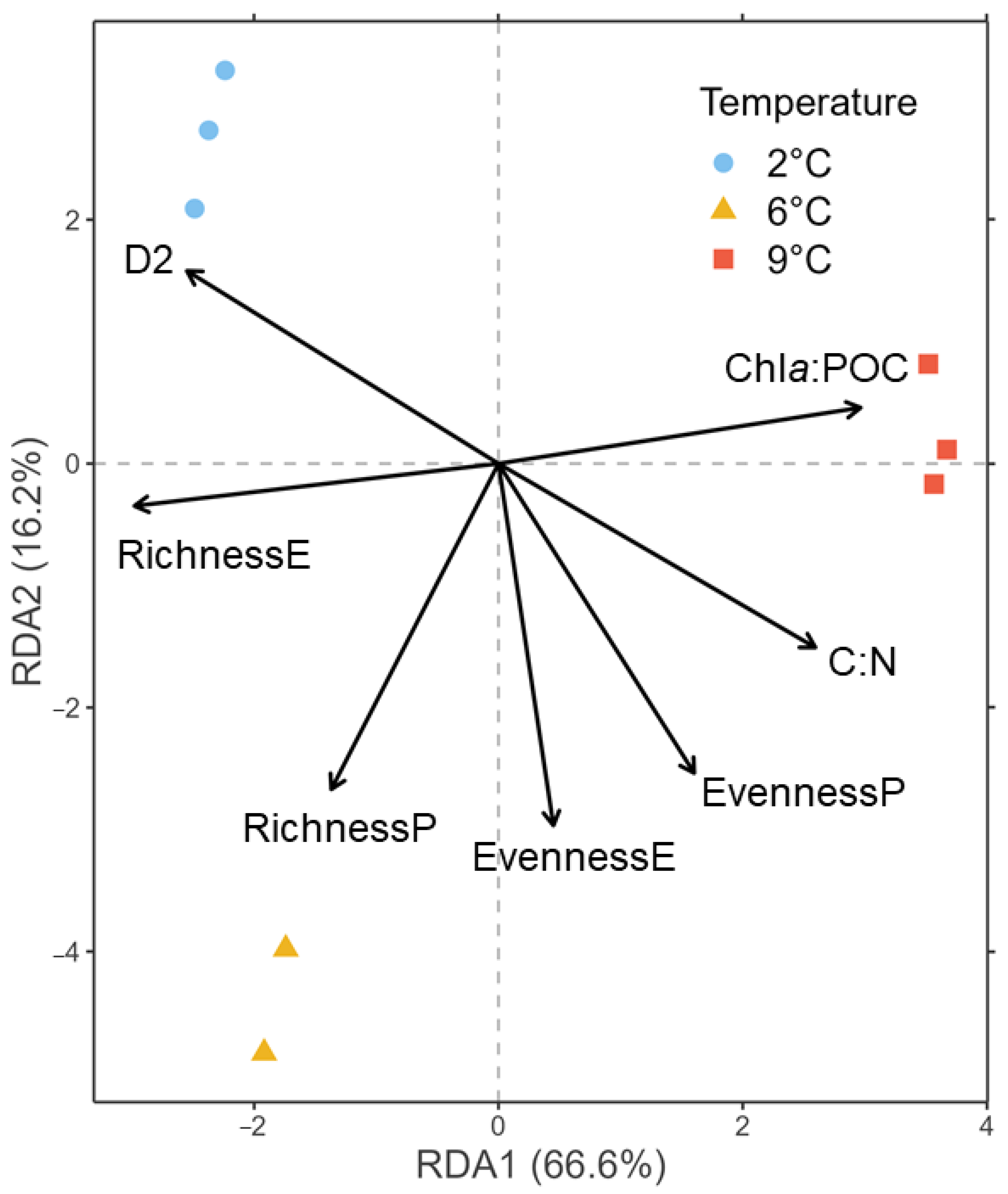
3.2. Biomass and Diversity Parameters
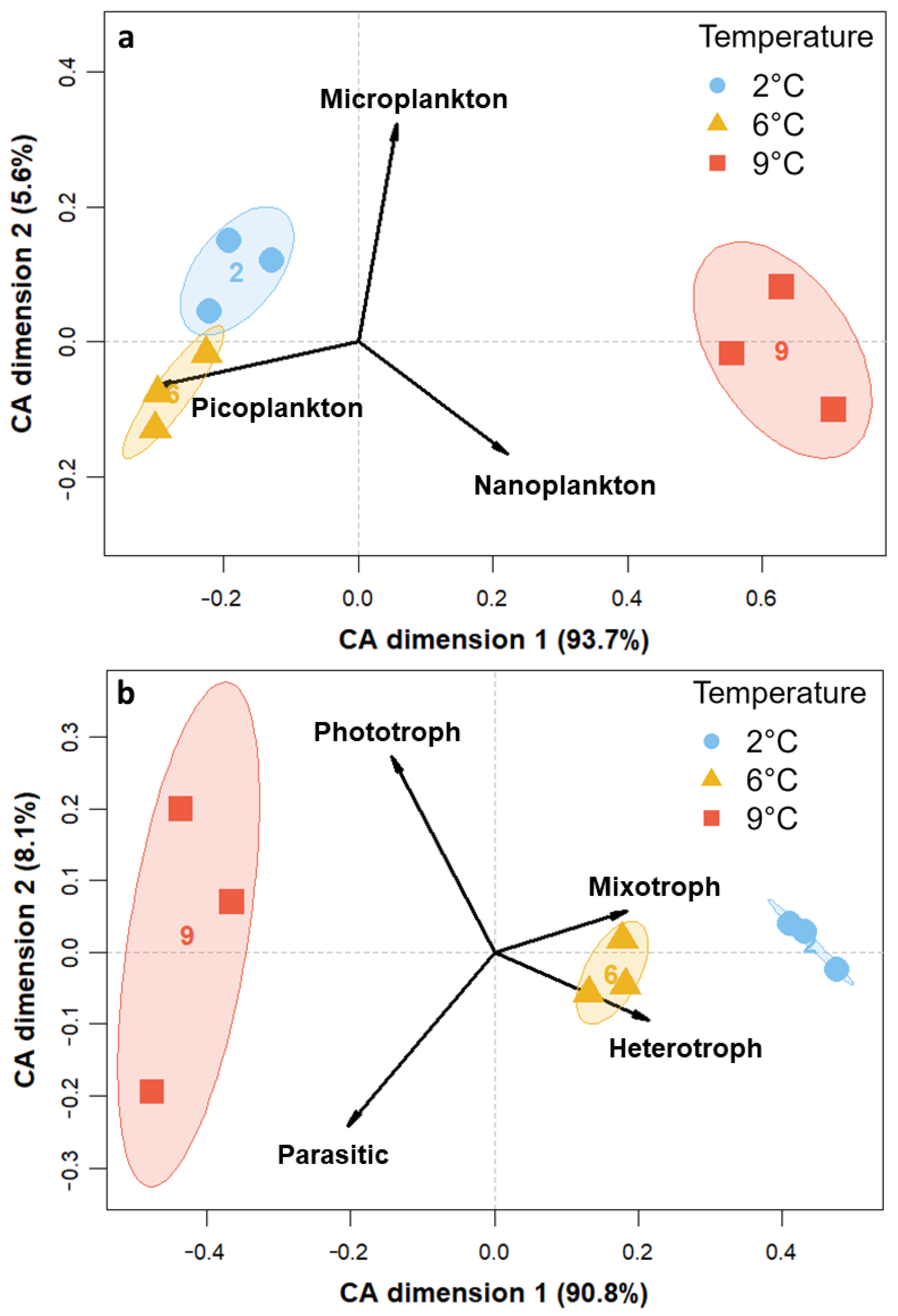
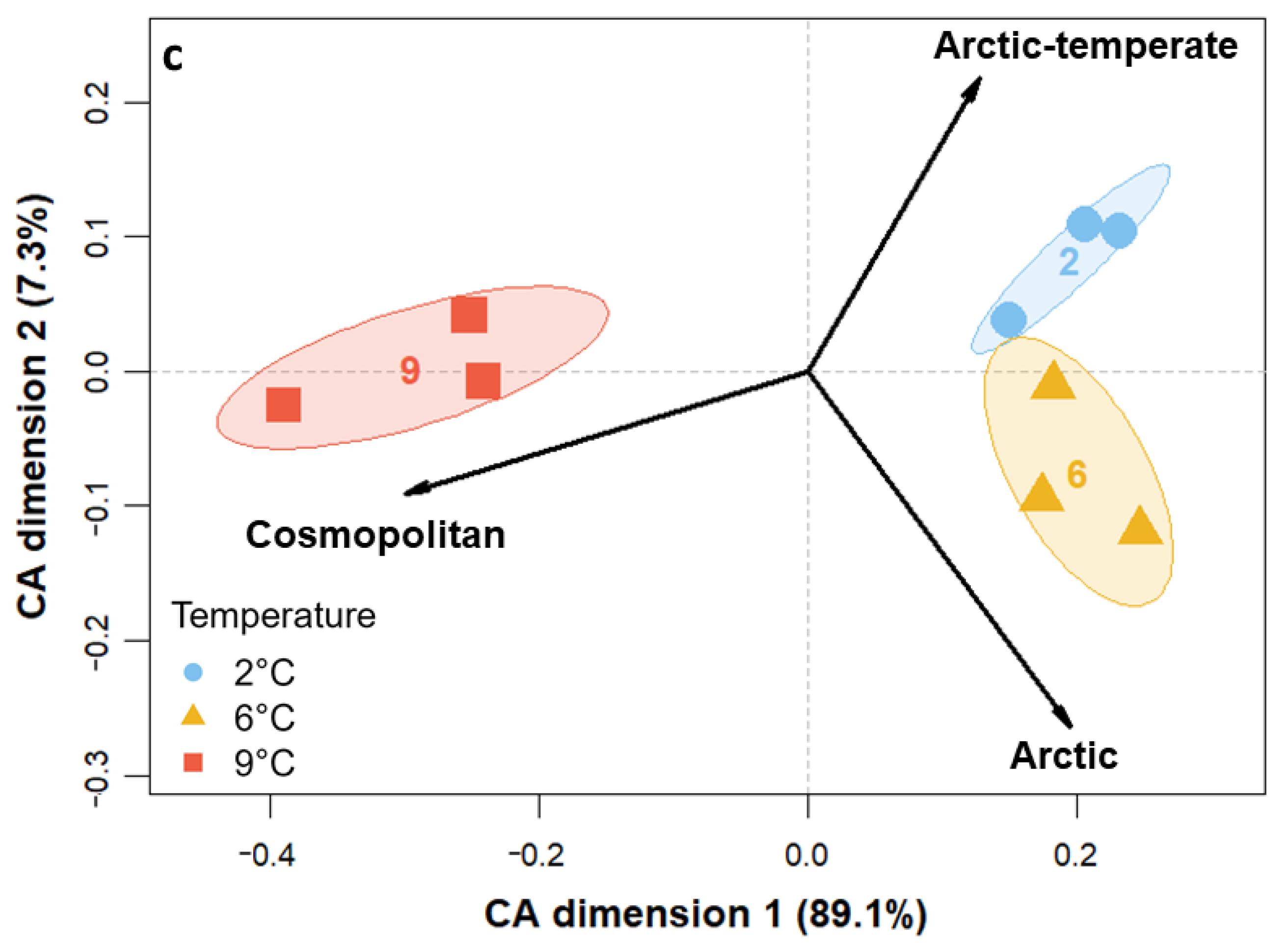
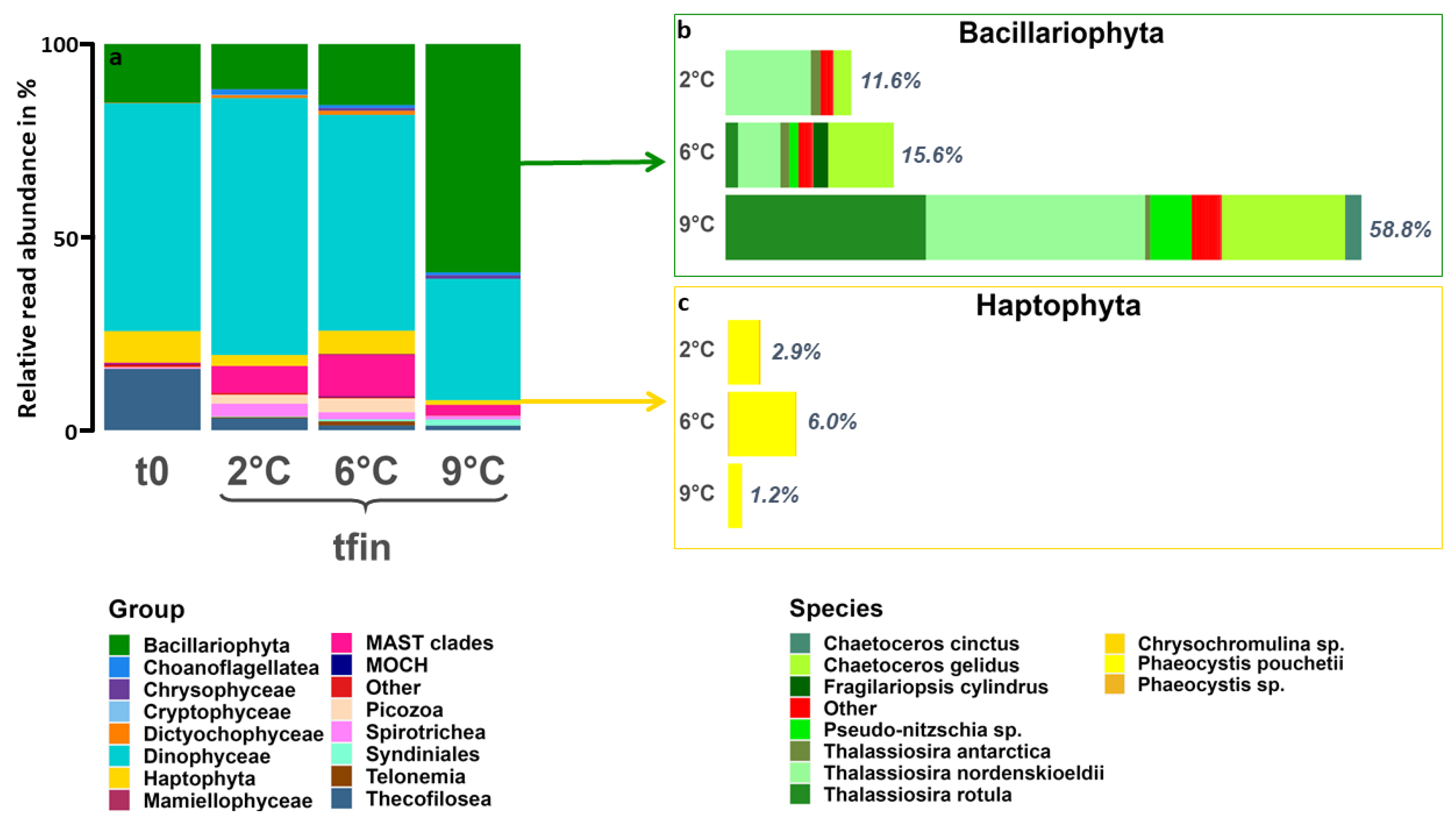
3.3. Community Composition—Eukaryotes
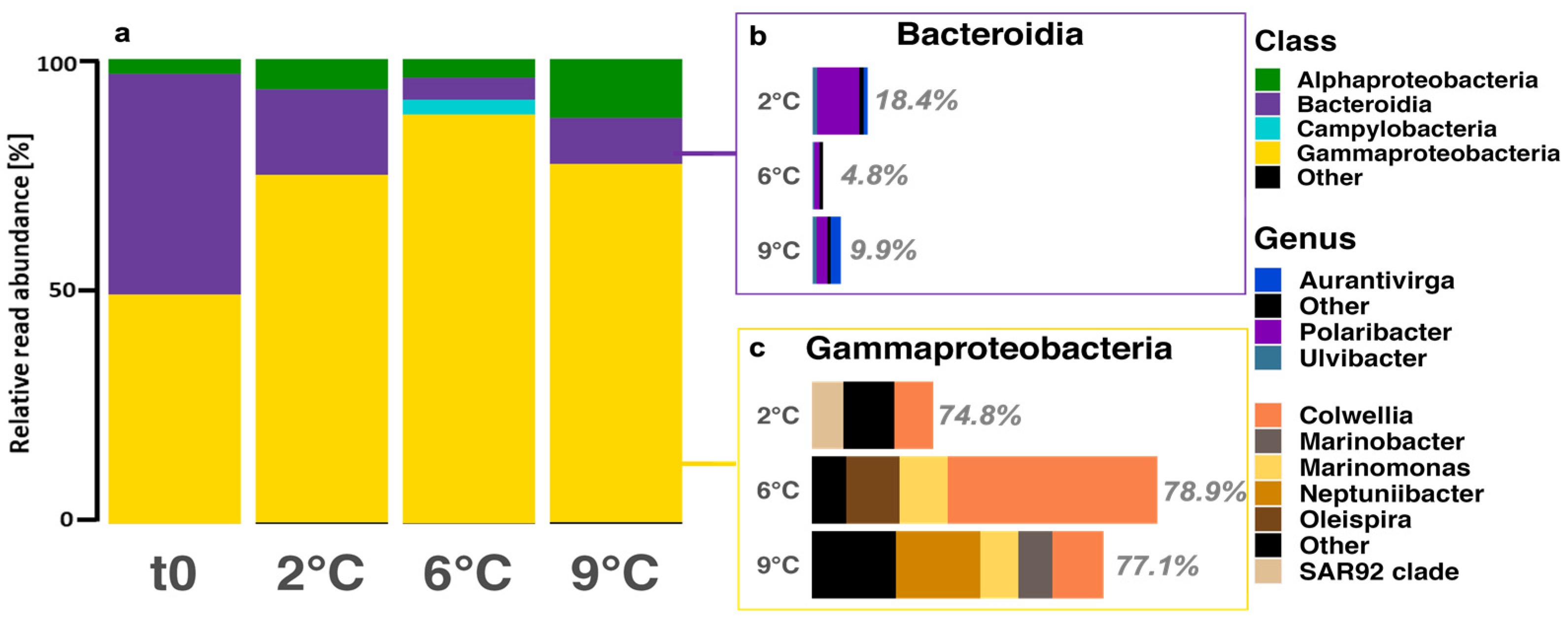
3.4. Community Composition—Bacteria
4. Discussion
4.1. Warming Induces an Increase in Photoautotrophic, Intermediate-Sized Organisms
4.2. Tipping Point for Arctic Key Eukaryotes Lies between 6 °C and 9 °C
4.3. Species-Specific Responses to Warming
4.4. Bacterial Diversity and Composition Response to Warming
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tesi, T.; Muschitiello, F.; Mollenhauer, G.; Miserocchi, S.; Langone, L.; Ceccarelli, C.; Panieri, G.; Chiggiato, J.; Nogarotto, A.; Hefter, J.; et al. Rapid Atlantification along the Fram Strait at the beginning of the 20th century. Sci. Adv. 2021, 7, eabj2946. [Google Scholar] [CrossRef]
- Polyakov, I.V.; Pnyushkov, A.V.; Alkire, M.B.; Ashik, I.M.; Baumann, T.M.; Carmack, E.C.; Goszczko, I.; Guthrie, J.; Ivanov, V.V.; Kanzow, T.; et al. Greater role for Atlantic inflows on sea-ice loss in the Eurasian Basin of the Arctic Ocean. Science 2017, 356, 285–291. [Google Scholar] [CrossRef]
- Beszczynska-Möller, A.; Fahrbach, E.; Schauer, U.; Hansen, E. Variability in Atlantic water temperature and transport at the entrance to the Arctic Ocean, 1997–2010. ICES J. Mar. Sci. 2012, 69, 852–863. [Google Scholar] [CrossRef]
- Marnela, M.; Rudels, B.; Houssais, M.N.; Beszczynska-Möller, A.; Eriksson, P.B. Recirculation in the Fram Strait and transports of water in and north of the Fram Strait derived from CTD data. Ocean Sci. 2013, 9, 499–519. [Google Scholar] [CrossRef]
- de Steur, L.; Hansen, E.; Gerdes, R.; Karcher, M.; Fahrbach, E.; Holfort, J. Freshwater fluxes in the East Greenland Current: A decade of observations. Geophys. Res. Lett. 2009, 36, L23611. [Google Scholar] [CrossRef]
- Rantanen, M.; Karpechko, A.Y.; Lipponen, A.; Nordling, K.; Hyvärinen, O.; Ruosteenoja, K.; Vihma, T.; Laaksonen, A. The Arctic has warmed nearly four times faster than the globe since 1979. Commun. Earth Environ. 2022, 3, 168. [Google Scholar] [CrossRef]
- Previdi, M.; Smith, K.L.; Polvani, L.M. Arctic amplification of climate change: A review of underlying mechanisms. Environ. Res. Lett. 2021, 16, 093003. [Google Scholar] [CrossRef]
- Oziel, L.; Baudena, A.; Ardyna, M.; Massicotte, P.; Randelhoff, A.; Sallee, J.B.; Ingvaldsen, R.B.; Devred, E.; Babin, M. Faster Atlantic currents drive poleward expansion of temperate phytoplankton in the Arctic Ocean. Nat. Commun. 2020, 11, 1705. [Google Scholar] [CrossRef]
- Cadotte, M.W.; Arnillas, C.A.; Livingstone, S.W.; Yasui, S.-L.E. Predicting communities from functional traits. Trends Ecol. Evol. 2015, 30, 510–511. [Google Scholar] [CrossRef]
- Fernandez-Gonzalez, C.; Maranon, E. Effect of temperature on the unimodal size scaling of phytoplankton growth. Sci. Rep. 2021, 11, 953. [Google Scholar] [CrossRef]
- Boscolo-Galazzo, F.; Crichton, K.A.; Barker, S.; Pearson, P.N. Temperature dependency of metabolic rates in the upper ocean: A positive feedback to global climate change? Glob. Planet. Chang. 2018, 170, 201–212. [Google Scholar] [CrossRef]
- Anderson, S.I.; Rynearson, T.A. Variability approaching the thermal limits can drive diatom community dynamics. Limnol. Oceanogr. 2020, 65, 1961–1973. [Google Scholar] [CrossRef]
- Atkinson, D.; Ciotti, B.J.; Montagnes, D.J.S. Protists decrease in size linearly with temperature: Ca. 2.5% °C−1. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2003, 270, 2605–2611. [Google Scholar] [CrossRef]
- Morán, X.A.G.; López-Urrutia, Á.; Calvo-Díaz, A.; Li, W.K.W. Increasing importance of small phytoplankton in a warmer ocean. Glob. Chang. Biol. 2010, 16, 1137–1144. [Google Scholar] [CrossRef]
- Daufresne, M.; Lengfellner, K.; Sommer, U. Global warming benefits the small in aquatic ecosystems. Proc. Natl. Acad. Sci. USA 2009, 106, 12788–12793. [Google Scholar] [CrossRef]
- Marañón, E. Cell Size as a Key Determinant of Phytoplankton Metabolism and Community Structure. Annu. Rev. Mar. Sci. 2015, 7, 241–264. [Google Scholar] [CrossRef]
- Hillebrand, H.; Acevedo-Trejos, E.; Moorthi, S.D.; Ryabov, A.; Striebel, M.; Thomas, P.K.; Schneider, M.L. Cell size as driver and sentinel of phytoplankton community structure and functioning. Funct. Ecol. 2021, 36, 276–293. [Google Scholar] [CrossRef]
- Brown, J.H.; Gillooly, J.F.; Allen, A.P.; Savage, V.M.; West, G.B. Toward a metabolic theory of ecology. Ecology 2004, 85, 1771–1789. [Google Scholar] [CrossRef]
- Chen, B.; Landry, M.R.; Huang, B.; Liu, H. Does warming enhance the effect of microzooplankton grazing on marine phytoplankton in the ocean? Limnol. Oceanogr. 2012, 57, 519–526. [Google Scholar] [CrossRef]
- Hancke, K.; Glud, R.N. Temperature effects on respiration and photosynthesis in three diatom-dominated benthic communities. Aquat. Microb. Ecol. 2004, 37, 265–281. [Google Scholar] [CrossRef]
- Regaudie-de-Gioux, A.; Duarte, C.M. Temperature dependence of planktonic metabolism in the ocean. Glob. Biogeochem. Cycles 2012, 26, GB1015. [Google Scholar] [CrossRef]
- Yvon-Durocher, G.; Caffrey, J.M.; Cescatti, A.; Dossena, M.; Giorgio, P.D.; Gasol, J.M.; Montoya, J.M.; Pumpanen, J.; Staehr, P.A.; Trimmer, M.; et al. Reconciling the temperature dependence of respiration across timescales and ecosystem types. Nature 2012, 487, 472–476. [Google Scholar] [CrossRef]
- Chen, B. Assessing the accuracy of the “two-point” dilution technique. Limnol. Oceanogr. Methods 2015, 13, 521–526. [Google Scholar] [CrossRef]
- Liu, K.; Chen, B.; Zhang, S.; Sato, M.; Shi, Z.; Liu, H. Marine phytoplankton in subtropical coastal waters showing lower thermal sensitivity than microzooplankton. Limnol. Oceanogr. 2018, 64, 1103–1119. [Google Scholar] [CrossRef]
- Anderson, S.I.; Barton, A.D.; Clayton, S.; Dutkiewicz, S.; Rynearson, T.A. Marine phytoplankton functional types exhibit diverse responses to thermal change. Nat. Commun. 2021, 12, 6413. [Google Scholar] [CrossRef]
- Chen, B. Thermal diversity affects community responses to warming. Ecol. Model. 2022, 464, 109846. [Google Scholar] [CrossRef]
- Boyd, P.W.; Rynearson, T.A.; Armstrong, E.A.; Fu, F.; Hayashi, K.; Hu, Z.; Hutchins, D.A.; Kudela, R.M.; Litchman, E.; Mulholland, M.R.; et al. Marine phytoplankton temperature versus growth responses from polar to tropical waters--outcome of a scientific community-wide study. PLoS ONE 2013, 8, e63091. [Google Scholar] [CrossRef]
- Huertas, I.E.; Rouco, M.; López-Rodas, V.; Costas, E. Warming will affect phytoplankton differently: Evidence through a mechanistic approach. Proc. R. Soc. B Biol. Sci. 2011, 278, 3534–3543. [Google Scholar] [CrossRef]
- Thomas, M.K.; Kremer, C.T.; Klausmeier, C.A.; Litchman, E. A Global Pattern of Thermal Adaptation in Marine Phytoplankton. Science 2012, 338, 1085–1088. [Google Scholar] [CrossRef]
- Greco, M.; Werner, K.; Zamelczyk, K.; Rasmussen, T.L.; Kucera, M. Decadal trend of plankton community change and habitat shoaling in the Arctic gateway recorded by planktonic foraminifera. Glob. Chang. Biol. 2022, 28, 1798–1808. [Google Scholar] [CrossRef]
- Soltwedel, T. The Expedition PS126 of the Research Vessel POLARSTERN to the Fram Strait in 2021; 1866–3192; Alfred-Wegener-Institut, Helmholtz-Zentrum für Polar-und Meeresforschung: Bremerhaven, Germany, 2021; pp. 1–131. [Google Scholar]
- Hoppmann, M.; Tippenhauer, S.; Soltwedel, T. Continuous thermosalinograph oceanography along RV POLARSTERN cruise track PS126. Alfred Wegener Institute, Helmholtz Centre for Polar and Marine Research, Bremerhaven. Pangaea 2022. [Google Scholar] [CrossRef]
- Löder, M.G.J.; Aberle, N.; Klaas, C.; Kraberg, A.C.; Wiltshire, K.H. Conserving original in situ diversity in microzooplankton grazing set-ups. Mar. Biodivers. Rec. 2010, 3, E28. [Google Scholar] [CrossRef]
- Soltwedel, T.; Bauerfeind, E.; Bergmann, M.; Bracher, A.; Budaeva, N.; Busch, K.; Cherkasheva, A.; Fahl, K.; Grzelak, K.; Hasemann, C.; et al. Natural variability or anthropogenically-induced variation? Insights from 15 years of multidisciplinary observations at the arctic marine LTER site HAUSGARTEN. Ecol. Indic. 2016, 65, 89–102. [Google Scholar] [CrossRef]
- Nöthig, E.-M.; Ramondenc, S.; Haas, A.; Hehemann, L.; Walter, A.; Bracher, A.; Lalande, C.; Metfies, K.; Peeken, I.; Bauerfeind, E.; et al. Summertime Chlorophyll a and Particulate Organic Carbon Standing Stocks in Surface Waters of the Fram Strait and the Arctic Ocean (1991–2015). Front. Mar. Sci. 2020, 7, 350. [Google Scholar] [CrossRef]
- Kaiser, P.; Hagen, W.; von Appen, W.-J.; Niehoff, B.; Hildebrandt, N.; Auel, H. Effects of a Submesoscale Oceanographic Filament on Zooplankton Dynamics in the Arctic Marginal Ice Zone. Front. Mar. Sci. 2021, 8, 625395. [Google Scholar] [CrossRef]
- Calbet, A.; Saiz, E. How much is enough for nutrients in microzooplankton dilution grazing experiments? J. Plankton Res. 2018, 40, 109–117. [Google Scholar] [CrossRef]
- Pierrot, D.; Wallace, D.W.R.; Lewis, E.R.; Pierrot, D.; Wallace, R.; Wallace, D.W.R.; Wallace, W.E. MS Excel Program Developed for CO2 System Calculations; US Department of Energy: Oak Ridge, TN, USA, 2011.
- Mehrbach, C.; Culberson, C.H.; Hawley, J.E.; Pytkowicx, R.M. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol. Oceanogr. 1973, 18, 897–907. [Google Scholar] [CrossRef]
- Dickson, A.G.; Millero, F.J. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep Sea Res. Part A. Oceanogr. Res. Pap. 1987, 34, 1733–1743. [Google Scholar] [CrossRef]
- Armstrong, F.A.J.; Stearns, C.R.; Strickland, J.D.H. The measurement of upwelling and subsequent biological process by means of the Technicon Autoanalyzer® and associated equipment. Deep Sea Res. Oceanogr. Abstr. 1967, 14, 381–389. [Google Scholar] [CrossRef]
- Eberlein, K.; Kattner, G. Automatic method for the determination of ortho-phosphate and total dissolved phosphorus in the marine environment. Fresenius’ Z. Für Anal. Chem. 1987, 326, 354–357. [Google Scholar] [CrossRef]
- Grasshoff, K.; Kremling, K.; Ehrhardt, M. Methods of Seawater Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Koroleff, F. Direct determination of ammonia in natural waters as indophenol blue. Inf. Tech. Methods Seawater Anal. 1970, 19–22. [Google Scholar]
- Arar, E.J.; Collins, G.B. Method 445.0 In Vitro Determination of Chlorophyll a and Pheophytin ain Marine and Freshwater Algae by Fluorescence; U.S. Environmental Protection Agency: Washington, DC, USA, 1997.
- Bradley, I.M.; Pinto, A.J.; Guest, J.S. Design and evaluation of illumina MiSeq-compatible, 18S rRNA gene-specific primers for improved characterization of mixed phototrophic communities. Appl. Environ. Microbiol. 2016, 82, 5878–5891. [Google Scholar] [CrossRef] [PubMed]
- Fadeev, E.; Salter, I.; Schourup-Kristensen, V.; Nöthig, E.-M.; Metfies, K.; Engel, A.; Piontek, J.; Boetius, A.; Bienhold, C. Microbial Communities in the East and West Fram Strait During Sea Ice Melting Season. Front. Mar. Sci. 2018, 5, 429. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Guillou, L.; Bachar, D.; Audic, S.; Bass, D.; Berney, C.; Bittner, L.; Boutte, C.; Burgaud, G.; de Vargas, C.; Decelle, J.; et al. The Protist Ribosomal Reference database (PR2): A catalog of unicellular eukaryote small sub-unit rRNA sequences with curated taxonomy. Nucleic Acids Res. 2013, 41, D597–D604. [Google Scholar] [CrossRef]
- Beule, L.; Karlovsky, P. Improved normalization of species count data in ecology by scaling with ranked subsampling (SRS): Application to microbial communities. PeerJ 2020, 8, e9593. [Google Scholar] [CrossRef]
- Sisk-Hackworth, L.; Kelley, S.T. An application of compositional data analysis to multiomic time-series data. NAR Genom. Bioinform. 2020, 2, lqaa079. [Google Scholar] [CrossRef]
- Martín-Fernández, J.-A.; Hron, K.; Templ, M.; Filzmoser, P.; Palarea-Albaladejo, J. Bayesian-multiplicative treatment of count zeros in compositional data sets. Stat. Model. 2015, 15, 134–158. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: https://www.R-project.org/ (accessed on 19 January 2023).
- RStudioTeam. RStudio: Integrated Development Environment for R. RStudio; PBC: Boston, MA, USA, 2022; Available online: http://www.rstudio.com/ (accessed on 19 January 2023).
- Ahyong, S.; Boyko, C.B.; Bailly, N.; Bernot, J.; Bieler, R.; Brandão, S.N.; Daly, M.; De Grave, S.; Gofas, S.; Hernandez, F.; et al. World Register of Marine Species (WoRMS). 2022. Available online: https://www.marinespecies.org/ (accessed on 25 February 2023).
- Hörstmann, C.; Buttigieg, P.L.; John, U.; Raes, E.J.; Wolf-Gladrow, D.; Bracher, A.; Waite, A.M. Microbial diversity through an oceanographic lens: Refining the concept of ocean provinces through trophic-level analysis and productivity-specific length scales. Environ. Microbiol. 2022, 24, 404–419. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.K.; Anestis, K.; Mansour, J.; Anschutz, A.A.; Gypens, N.; Hansen, P.J.; John, U.; Klemm, K.; Martin, J.L.; Medic, N.; et al. A dataset on trophic modes of aquatic protists. Biodivers. Data J. 2020, 8, e56648. [Google Scholar] [CrossRef] [PubMed]
- Šupraha, L.; Klemm, K.; Gran-Stadniczeñko, S.; Hörstmann, C.; Vaulot, D.; Edvardsen, B.; John, U. Diversity and biogeography of planktonic diatoms in Svalbard fjords: The role of dispersal and Arctic endemism in phytoplankton community structuring. Elem. Sci. Anthr. 2022, 10, 00117. [Google Scholar] [CrossRef]
- Props, R.; Monsieurs, P.; Mysara, M.; Clement, L.; Boon, N. Measuring the biodiversity of microbial communities by flow cytometry. Methods Ecol. Evol. 2016, 7, 1376–1385. [Google Scholar] [CrossRef]
- Fontana, S.; Thomas, M.K.; Moldoveanu, M.; Spaak, P.; Pomati, F. Individual-level trait diversity predicts phytoplankton community properties better than species richness or evenness. ISME J. 2018, 12, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.; Listmann, L.; Santelia, M.E.; Schaum, C.E. Functional redundancy in natural pico-phytoplankton communities depends on temperature and biogeography. Biol. Lett. 2020, 16, 20200330. [Google Scholar] [CrossRef]
- Greenacre, M. Compositional Data Analysis. Annu. Rev. Stat. Its Appl. 2021, 8, 271–299. [Google Scholar] [CrossRef]
- von Appen, W.-J.; Wekerle, C.; Hehemann, L.; Schourup-Kristensen, V.; Konrad, C.; Iversen, M.H. Observations of a Submesoscale Cyclonic Filament in the Marginal Ice Zone. Geophys. Res. Lett. 2018, 45, 6141–6149. [Google Scholar] [CrossRef]
- Tanioka, T.; Matsumoto, K. A meta-analysis on environmental drivers of marine phytoplankton C: N: P. Biogeosciences 2020, 17, 2939–2954. [Google Scholar] [CrossRef]
- Thompson, P.A.; Guo, M.-x.; Harrison, P.J. Effects of variation in temperature. I. On the biochemical composition of eight species of marine phytoplankton. J. Phycol. 1992, 28, 481–488. [Google Scholar] [CrossRef]
- Petchey, O.L.; McPhearson, P.T.; Casey, T.M.; Morin, P.J. Environmental warming alters food-web structure and ecosystem function. Nature 1999, 402, 69–72. [Google Scholar] [CrossRef]
- Schaum, C.E.; Team, S.R.; ffrench-Constant, R.; Lowe, C.; Ólafsson, J.S.; Padfield, D.; Yvon-Durocher, G. Temperature-driven selection on metabolic traits increases the strength of an algal–grazer interaction in naturally warmed streams. Glob. Chang. Biol. 2018, 24, 1793–1803. [Google Scholar] [CrossRef] [PubMed]
- Gibert, J.P. Temperature directly and indirectly influences food web structure. Sci. Rep. 2019, 9, 5312. [Google Scholar] [CrossRef]
- Marañón, E.; Cermeno, P.; Lopez-Sandoval, D.C.; Rodriguez-Ramos, T.; Sobrino, C.; Huete-Ortega, M.; Blanco, J.M.; Rodriguez, J. Unimodal size scaling of phytoplankton growth and the size dependence of nutrient uptake and use. Ecol. Lett. 2013, 16, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Li, W.K.; McLaughlin, F.A.; Lovejoy, C.; Carmack, E.C. Smallest algae thrive as the Arctic Ocean freshens. Science 2009, 326, 539. [Google Scholar] [CrossRef]
- Neukermans, G.; Oziel, L.; Babin, M. Increased intrusion of warming Atlantic water leads to rapid expansion of temperate phytoplankton in the Arctic. Glob. Chang. Biol. 2018, 24, 2545–2553. [Google Scholar] [CrossRef]
- Vernet, M.; Richardson, T.L.; Metfies, K.; Eva-Maria, N.; Peeken, I. Models of plankton community changes during a warm water anomaly in Arctic waters show altered trophic pathways with minimal changes in carbon export. Front. Mar. Sci. 2017, 4, 160. [Google Scholar] [CrossRef]
- Wang, X.; Tang, K.W.; Wang, Y.; Smith, W.O. Temperature effects on growth, colony development and carbon partitioning in three Phaeocystis species. Aquat. Biol. 2010, 9, 239–249. [Google Scholar] [CrossRef]
- Zhang, Y.; Klapper, R.; Lohbeck, K.T.; Bach, L.T.; Schulz, K.G.; Reusch, T.B.H.; Riebesell, U. Between- and within-population variations in thermal reaction norms of the coccolithophore Emiliania huxleyi. Limnol. Oceanogr. 2014, 59, 1570–1580. [Google Scholar] [CrossRef]
- Benedetti, F.; Vogt, M.; Elizondo, U.H.; Righetti, D.; Zimmermann, N.E.; Gruber, N. Major restructuring of marine plankton assemblages under global warming. Nat. Commun. 2021, 12, 5226. [Google Scholar] [CrossRef]
- Bestion, E.; Barton, S.; Garcia, F.C.; Warfield, R.; Yvon-Durocher, G. Abrupt declines in marine phytoplankton production driven by warming and biodiversity loss in a microcosm experiment. Ecol. Lett. 2020, 23, 457–466. [Google Scholar] [CrossRef]
- Coello-Camba, A.; Agustí, S.; Vaqué, D.; Holding, J.; Arrieta, J.M.; Wassmann, P.; Duarte, C.M. Experimental Assessment of Temperature Thresholds for Arctic Phytoplankton Communities. Estuaries Coasts 2015, 38, 873–885. [Google Scholar] [CrossRef]
- Coello-Camba, A.; Agustí, S. Thermal Thresholds of Phytoplankton Growth in Polar Waters and Their Consequences for a Warming Polar Ocean. Front. Mar. Sci. 2017, 4, 168. [Google Scholar] [CrossRef]
- Stawiarski, B.; Buitenhuis, E.T.; Le Quéré, C. The Physiological Response of Picophytoplankton to Temperature and Its Model Representation. Front. Mar. Sci. 2016, 3, 164. [Google Scholar] [CrossRef]
- Bishop, I.W.; Anderson, S.I.; Collins, S.; Rynearson, T.A. Thermal trait variation may buffer Southern Ocean phytoplankton from anthropogenic warming. Glob. Chang. Biol. 2022, 28, 5755–5767. [Google Scholar] [CrossRef] [PubMed]
- Schiffrine, N.; Tremblay, J.É.; Babin, M. Interactive effects of temperature and nitrogen source on the elemental stoichiometry of a polar diatom. Limnol. Oceanogr. 2022, 67, 2750–2762. [Google Scholar] [CrossRef]
- Godhe, A.; Rynearson, T. The role of intraspecific variation in the ecological and evolutionary success of diatoms in changing environments. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160399. [Google Scholar] [CrossRef]
- Nöthig, E.-M.; Bracher, A.; Engel, A.; Metfies, K.; Niehoff, B.; Peeken, I.; Bauerfeind, E.; Cherkasheva, A.; Gäbler-Schwarz, S.; Hardge, K.; et al. Summertime plankton ecology in Fram Strait—A compilation of long- and short-term observations. Polar Res. 2015, 34, 23349. [Google Scholar] [CrossRef]
- Szeligowska, M.; Trudnowska, E.; Boehnke, R.; Dąbrowska, A.M.; Wiktor, J.M.; Sagan, S.; Błachowiak-Samołyk, K. Spatial Patterns of Particles and Plankton in the Warming Arctic Fjord (Isfjorden, West Spitsbergen) in Seven Consecutive Mid-Summers (2013–2019). Front. Mar. Sci. 2020, 7, 584. [Google Scholar] [CrossRef]
- Metfies, K.; von Appen, W.-J.; Kilias, E.; Nicolaus, A.; Nöthig, E.-M. Biogeography and Photosynthetic Biomass of Arctic Marine Pico-Eukaroytes during Summer of the Record Sea Ice Minimum 2012. PLoS ONE 2016, 11, e0148512. [Google Scholar] [CrossRef]
- Lampe, V.; Nöthig, E.-M.; Schartau, M. Spatio-Temporal Variations in Community Size Structure of Arctic Protist Plankton in the Fram Strait. Front. Mar. Sci. 2021, 7, 579880. [Google Scholar] [CrossRef]
- Worden, A.Z.; Lee, J.H.; Mock, T.; Rouzé, P.; Simmons, M.P.; Aerts, A.L.; Allen, A.E.; Cuvelier, M.L.; Derelle, E.; Everett, M.V.; et al. Green evolution and dynamic adaptations revealed by genomes of the marine picoeukaryotes Micromonas. Science 2009, 324, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Balzano, S.; Marie, D.; Gourvil, P.; Vaulot, D. Composition of the summer photosynthetic pico and nanoplankton communities in the Beaufort Sea assessed by T-RFLP and sequences of the 18S rRNA gene from flow cytometry sorted samples. ISME J. 2012, 6, 1480–1498. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, C.J.M.; Flintrop, C.M.; Rost, B. The Arctic picoeukaryote Micromonas pusilla benefits synergistically from warming and ocean acidification. Biogeosciences 2018, 15, 4353–4365. [Google Scholar] [CrossRef]
- Benner, I.; Irwin, A.J.; Finkel, Z.V. Capacity of the common Arctic picoeukaryote Micromonas to adapt to a warming ocean. Limnol. Oceanogr. Lett. 2020, 5, 221–227. [Google Scholar] [CrossRef]
- Lovejoy, C.; Vincent, W.F.; Bonilla, S.; Roy, S.; Martineau, M.-J.; Terrado, R.; Potvin, M.; Massana, R.; Pedrós-Alió, C. Distribution, phylogeny, and growth of cold-adapted picoprasinophytes in Arctic Seas. J. Phycol. 2007, 43, 78–89. [Google Scholar] [CrossRef]
- Berge, J.; Renaud, P.E.; Darnis, G.; Cottier, F.; Last, K.; Gabrielsen, T.M.; Johnsen, G.; Seuthe, L.; Weslawski, J.M.; Leu, E.; et al. In the dark: A review of ecosystem processes during the Arctic polar night. Prog. Oceanogr. 2015, 139, 258–271. [Google Scholar] [CrossRef]
- Bachy, C.; Sudek, L.; Choi, C.J.; Eckmann, C.A.; Nöthig, E.-M.; Metfies, K.; Worden, A.Z. Phytoplankton Surveys in the Arctic Fram Strait Demonstrate the Tiny Eukaryotic Alga Micromonas and Other Picoprasinophytes Contribute to Deep Sea Export. Microorganisms 2022, 10, 961. [Google Scholar] [CrossRef]
- Aalto, N.J.; Schweitzer, H.D.; Krsmanovic, S.; Campbell, K.; Bernstein, H.C. Diversity and Selection of Surface Marine Microbiomes in the Atlantic-Influenced Arctic. Front. Microbiol. 2022, 13, 892634. [Google Scholar] [CrossRef]
- Piontek, J.; Sperling, M.; Nöthig, E.; Engel, A. Regulation of bacterioplankton activity in Fram Strait (Arctic Ocean) during early summer: The role of organic matter supply and temperature. J. Mar. Syst. 2014, 132, 83–94. [Google Scholar] [CrossRef]
- Kirchman, D.L. The ecology of Cytophaga–Flavobacteria in aquatic environments. FEMS Microbioliol. Ecol. 2002, 39, 91–100. [Google Scholar] [CrossRef]
- Salazar, G.; Cornejo-Castillo, F.; Benítez-Barrios, V.; Fraile-Nuez, E.; Álvarez-Salgado, X.A.; Duarte, C.M.; Gasol, J.M.; Acinas, S.G. Global diversity and biogeography of deep-sea pelagic prokaryotes. ISME J. 2016, 10, 596–608. [Google Scholar] [CrossRef]
- Buchan, A.; LeCleir, G.R.; Gulvik, C.A.; González, J.M. Master recyclers: Features and functions of bacteria associated with phytoplankton blooms. Nat. Rev. Microbiol. 2014, 12, 686–698. [Google Scholar] [CrossRef]
- Sipler, R.E.; Kellogg, C.T.E.; Connelly, T.L.; Roberts, Q.N.; Yager, P.L.; Bronk, D.A. Microbial Community Response to Terrestrially Derived Dissolved Organic Matter in the Coastal Arctic. Front. Microbiol. 2022, 13, 1018. [Google Scholar] [CrossRef] [PubMed]
- Underwood, G.J.C.; Michel, C.; Meisterhans, G.; Niemi, A.; Belzile, C.; Witt, M.; Dumbrell, A.J.; Koch, B.P. Organic matter from Arctic sea-ice loss alters bacterial community structure and function. Nat. Clim. Chang. 2019, 9, 170–176. [Google Scholar] [CrossRef]
- von Jackowski, A.; Becker, K.W.; Wietz, M.; Bienhold, C.; Zancker, B.; Nothig, E.M.; Engel, A. Variations of microbial communities and substrate regimes in the eastern Fram Strait between summer and fall. Environ. Microbioly 2022, 24, 4124–4136. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Blain, S.; Crispi, O.; Rembauville, M.; Obernosterer, I. Seasonal dynamics of prokaryotes and their associations with diatoms in the Southern Ocean as revealed by an autonomous sampler. Environ. Microbiol. 2020, 22, 3968–3984. [Google Scholar] [CrossRef] [PubMed]
- Gärdes, A.; Kaeppel, E.; Shehzad, A.; Seebah, S.; Teeling, H.; Yarza, P.; Glöckner, F.O.; Grossart, H.P.; Ullrich, M.S. Complete genome sequence of Marinobacter adhaerens type strain (HP15), a diatom-interacting marine microorganism. Stand. Genom. Sci. 2010, 3, 97–107. [Google Scholar] [CrossRef]
- Fontanez, K.M.; Eppley, J.M.; Samo, T.J.; Karl, D.M.; DeLong, E.F. Microbial community structure and function on sinking particles in the North Pacific Subtropical Gyre. Front. Microbiol. 2015, 6, 469. [Google Scholar] [CrossRef]
- Pérez-Hernández, M.D.; Pickart, R.S.; Pavlov, V.; Våge, K.; Ingvaldsen, R.; Sundfjord, A.; Renner, A.H.H.; Torres, D.J.; and Erofeeva, S.Y. The Atlantic Water boundary current north of Svalbard in late summer. J. Geophys. Res. Oceans 2017, 122, 2269–2290. [Google Scholar] [CrossRef]
- Hoppmann, M.M. CTD Raw Data Files from POLARSTERN Cruise PS126. 2022. Available online: https://adc.met.no/metsis/metadata/PANGAEA_940754 (accessed on 19 January 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahme, A.; Von Jackowski, A.; McPherson, R.A.; Wolf, K.K.E.; Hoppmann, M.; Neuhaus, S.; John, U. Winners and Losers of Atlantification: The Degree of Ocean Warming Affects the Structure of Arctic Microbial Communities. Genes 2023, 14, 623. https://doi.org/10.3390/genes14030623
Ahme A, Von Jackowski A, McPherson RA, Wolf KKE, Hoppmann M, Neuhaus S, John U. Winners and Losers of Atlantification: The Degree of Ocean Warming Affects the Structure of Arctic Microbial Communities. Genes. 2023; 14(3):623. https://doi.org/10.3390/genes14030623
Chicago/Turabian StyleAhme, Antonia, Anabel Von Jackowski, Rebecca A. McPherson, Klara K. E. Wolf, Mario Hoppmann, Stefan Neuhaus, and Uwe John. 2023. "Winners and Losers of Atlantification: The Degree of Ocean Warming Affects the Structure of Arctic Microbial Communities" Genes 14, no. 3: 623. https://doi.org/10.3390/genes14030623
APA StyleAhme, A., Von Jackowski, A., McPherson, R. A., Wolf, K. K. E., Hoppmann, M., Neuhaus, S., & John, U. (2023). Winners and Losers of Atlantification: The Degree of Ocean Warming Affects the Structure of Arctic Microbial Communities. Genes, 14(3), 623. https://doi.org/10.3390/genes14030623





