Predictive Value of SLCO1B1 c.521T>C Polymorphism on Observed Changes in the Treatment of 1136 Statin-Users
Abstract
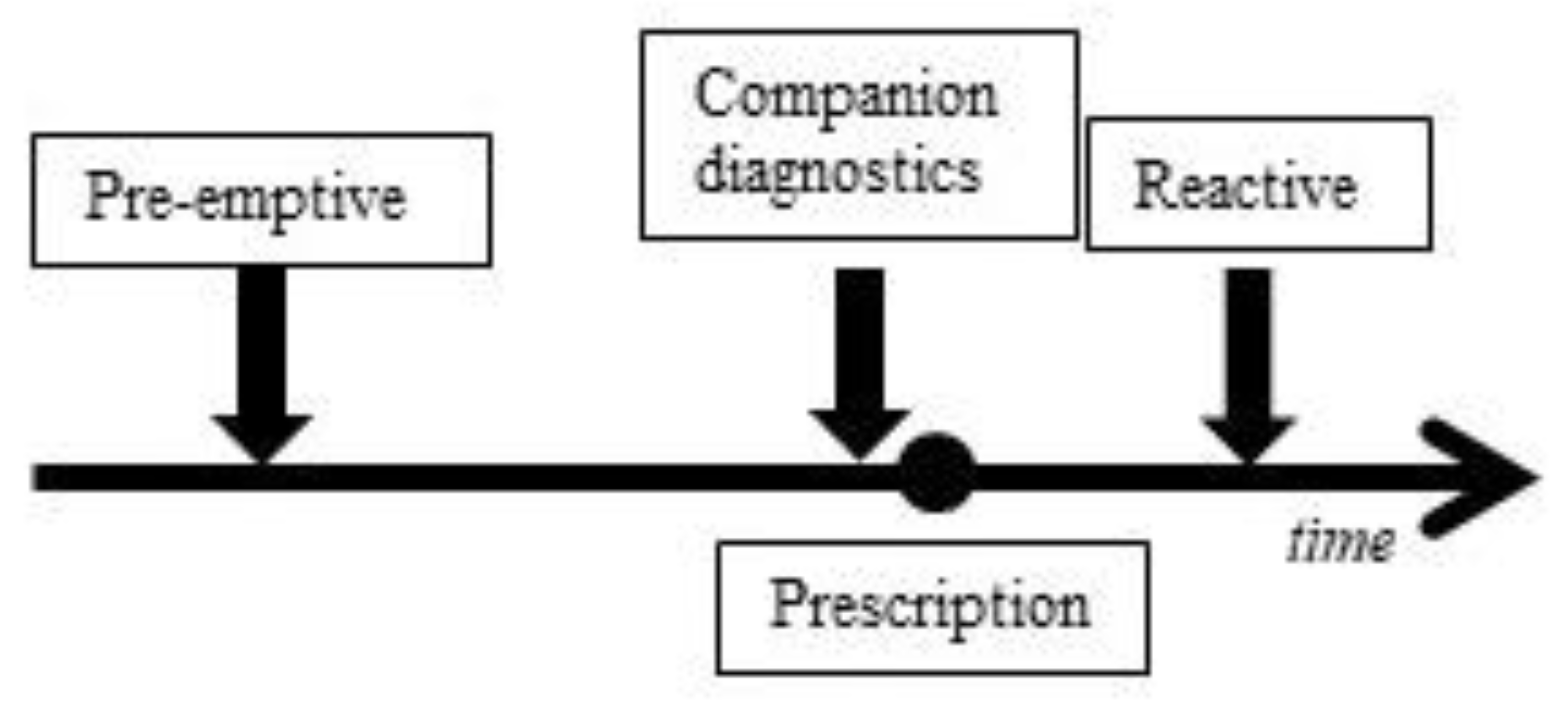
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Follow-Up
2.3. Genotyping
2.4. Outcomes
2.5. Covariables
2.5.1. Potential Confounders
2.5.2. Effect Modifier
2.6. Statistical Analysis
3. Results
3.1. Population Sample
3.2. Dose Change
3.3. Any Change in Drug Use: Discontinuation and Switching
3.4. Test Characteristics for SRM
3.5. Time to Establish Stable Dosing
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boudreau, D.M.; Yu, O.; Johnson, J. Statin use and cancer risk: A comprehensive review. Expert. Opin. Drug Saf. 2010, 9, 603–621. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.E.; Martin, S.S. Approach to Statin Use in 2016: An Update. Curr. Atheroscler. Rep. 2016, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Postmus, I.; Trompet, S.; Deshmukh, H.A.; Barnes, M.R.; Li, X.; Warren, H.R.; Chasman, D.I.; Zhou, K.; Arsenault, B.J.; Donnelly, L.A.; et al. Pharmacogenetic meta-analysis of genome-wide association studies of LDL cholesterol response to statins. Nat. Commun. 2014, 5, 5068. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J. The safety of statins in clinical practice. Lancet 2007, 370, 1781–1790. [Google Scholar] [CrossRef]
- Canestaro, W.J.; Austin, M.A.; Thummel, K.E. Genetic factors affecting statin concentrations and subsequent myopathy: A HuGENet systematic review. Genet. Med. 2014, 16, 810–819. [Google Scholar] [CrossRef]
- Ganga, H.V.; Slim, H.B.; Thompson, P.D. A systematic review of statin-induced muscle problems in clinical trials. Am. Heart J. 2014, 168, 6–15. [Google Scholar] [CrossRef]
- Hubacek, J.A.; Dlouha, D.; Adamkova, V.; Zlatohlavek, L.; Viklicky, O.; Hruba, P.; Ceska, R.; Vrablik, M. SLCO1B1 polymorphism is not associated with risk of statin-induced myalgia/myopathy in a Czech population. Med. Sci. Monit. 2015, 21, 1454–1459. [Google Scholar] [CrossRef]
- Janssen, S.P.; Smulders, Y.M.; Gerdes, V.E.; Visseren, F.L. Muscle problems due to statins: Underestimated. Ned. Tijdschr. Geneeskd. 2010, 154, A1684. [Google Scholar]
- Vrablik, M.; Zlatohlavek, L.; Stulc, T.; Adamkova, V.; Prusikova, M.; Schwarzova, L.; Hubacek, J.A.; Ceska, R. Statin-associated myopathy: From genetic predisposition to clinical management. Physiol. Res. 2014, 63 (Suppl. 3), S327–S334. [Google Scholar] [CrossRef]
- Geers, H.C.; Bouvy, M.L.; Heerdink, E.R. Estimates of statin discontinuation rates are influenced by exposure and outcome definitions. Ann. Pharmacother. 2011, 45, 576–581. [Google Scholar] [CrossRef]
- Mantel-Teeuwisse, A.; Goettsch, W.; Klungel, O.; De Boer, A.; Herings, R. Long term persistence with statin treatment in daily medical practice. Heart 2004, 90, 1065–1066. [Google Scholar] [CrossRef]
- Ramsey, L.B.; Johnson, S.G.; Caudle, K.E.; Haidar, C.E.; Voora, D.; Wilke, R.A.; Maxwell, W.D.; McLeod, H.L.; Krauss, R.M.; Roden, D.M.; et al. The clinical pharmacogenetics implementation consortium guideline for SLCO1B1 and simvastatin-induced myopathy: 2014 update. Clin. Pharmacol. Ther. 2014, 96, 423–428. [Google Scholar] [CrossRef]
- Sirtori, C.R. The pharmacology of statins. Pharmacol. Res. 2014, 88, 3–11. [Google Scholar] [CrossRef]
- Ghatak, A.; Faheem, O.; Thompson, P.D. The genetics of statin-induced myopathy. Atherosclerosis 2010, 210, 337–343. [Google Scholar] [CrossRef]
- Leusink, M.; Onland-Moret, N.C.; de Bakker, P.I.; de, B.A.; AH, M.-v.d.Z. Seventeen years of statin pharmacogenetics: A systematic review. Pharmacogenomics 2016, 17, 163–180. [Google Scholar] [CrossRef]
- Patel, J.; Superko, H.R.; Martin, S.S.; Blumenthal, R.S.; Christopher-Stine, L. Genetic and immunologic susceptibility to statin-related myopathy. Atherosclerosis 2015, 240, 260–271. [Google Scholar] [CrossRef]
- Niemi, M. Transporter pharmacogenetics and statin toxicity. Clin. Pharmacol. Ther. 2010, 87, 130–133. [Google Scholar] [CrossRef]
- KNMP. Drug Safety Monitoring. Pharmacogenetics. Atorvastatin SLCO1B1 521TC-521CC. Available online: https://kennisbank.knmp.nl/article/farmacogenetica/4057-4058.html (accessed on 26 February 2016).
- KNMP. Drug Safety Monitoring. Pharmacogenetics. Simvastatin SLCO1B1 521TC-521CC. Available online: https://kennisbank.knmp.nl/article/farmacogenetica/4055-4056.html (accessed on 26 February 2016).
- Swen, J.; Nijenhuis, M.; de Boer, A.; Grandia, L.; Maitland-van der Zee, A.-H.; Mulder, H.; Rongen, G.; Van Schaik, R.; Schalekamp, T.; Touw, D. Pharmacogenetics: From bench to byte—An update of guidelines. Clin. Pharmacol. Ther. 2011, 89, 662–673. [Google Scholar] [CrossRef]
- Xiang, Q.; Chen, S.-Q.; Ma, L.-Y.; Hu, K.; Zhang, Z.; Mu, G.-Y.; Xie, Q.-F.; Zhang, X.-D.; Cui, Y.-M. Association between SLCO1B1 T521C polymorphism and risk of statin-induced myopathy: A meta-analysis. Pharm. J. 2018, 18, 721–729. [Google Scholar] [CrossRef]
- Link, E.; Parish, S.; Armitage, J.; Bowman, L.; Heath, S.; Matsuda, F.; Gut, I.; Lathrop, M.; Collins, R. SLCO1B1 variants and statin-induced myopathy—A genomewide study. N. Engl. J. Med. 2008, 359, 789–799. [Google Scholar] [CrossRef]
- Carr, D.F.; O’Meara, H.; Jorgensen, A.L.; Campbell, J.; Hobbs, M.; McCann, G.; van, S.T.; Pirmohamed, M. SLCO1B1 genetic variant associated with statin-induced myopathy: A proof-of-concept study using the clinical practice research datalink. Clin. Pharmacol. Ther. 2013, 94, 695–701. [Google Scholar] [CrossRef] [PubMed]
- de Keyser, C.E.; Peters, B.J.; Becker, M.L.; Visser, L.E.; Uitterlinden, A.G.; Klungel, O.H.; Verstuyft, C.; Hofman, A.; AH, M.-v.d.Z.; Stricker, B.H. The SLCO1B1 c.521T>C polymorphism is associated with dose decrease or switching during statin therapy in the Rotterdam Study. Pharm. Genom. 2014, 24, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, L.A.; Doney, A.S.; Tavendale, R.; Lang, C.C.; Pearson, E.R.; Colhoun, H.M.; McCarthy, M.I.; Hattersley, A.T.; Morris, A.D.; Palmer, C.N. Common nonsynonymous substitutions in SLCO1B1 predispose to statin intolerance in routinely treated individuals with type 2 diabetes: A go-DARTS study. Clin. Pharmacol. Ther. 2011, 89, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Voora, D.; Shah, S.H.; Spasojevic, I.; Ali, S.; Reed, C.R.; Salisbury, B.A.; Ginsburg, G.S. The SLCO1B1*5 genetic variant is associated with statin-induced side effects. J. Am. Coll. Cardiol. 2009, 54, 1609–1616. [Google Scholar] [CrossRef]
- Brunham, L.R.; Lansberg, P.J.; Zhang, L.; Miao, F.; Carter, C.; Hovingh, G.K.; Visscher, H.; Jukema, J.W.; Stalenhoef, A.F.; Ross, C.J.; et al. Differential effect of the rs4149056 variant in SLCO1B1 on myopathy associated with simvastatin and atorvastatin. Pharm. J. 2012, 12, 233–237. [Google Scholar] [CrossRef]
- Santos, P.C.; Gagliardi, A.C.; Miname, M.H.; Chacra, A.P.; Santos, R.D.; Krieger, J.E.; Pereira, A.C. SLCO1B1 haplotypes are not associated with atorvastatin-induced myalgia in Brazilian patients with familial hypercholesterolemia. Eur. J. Clin. Pharmacol. 2012, 68, 273–279. [Google Scholar] [CrossRef]
- Puccetti, L.; Ciani, F.; Auteri, A. Genetic involvement in statins induced myopathy. Preliminary data from an observational case-control study. Atherosclerosis 2010, 211, 28–29. [Google Scholar] [CrossRef]
- Ferrari, M.; Guasti, L.; Maresca, A.; Mirabile, M.; Contini, S.; Grandi, A.M.; Marino, F.; Cosentino, M. Association between statin-induced creatine kinase elevation and genetic polymorphisms in SLCO1B1, ABCB1 and ABCG2. Eur. J. Clin. Pharmacol. 2014, 70, 539–547. [Google Scholar] [CrossRef]
- Linde, R.; Peng, L.; Desai, M.; Feldman, D. The role of vitamin D and SLCO1B1*5 gene polymorphism in statin-associated myalgias. Dermatoendocrinology 2010, 2, 77–84. [Google Scholar] [CrossRef]
- Hopewell, J.C.; Parish, S.; Offer, A.; Link, E.; Clarke, R.; Lathrop, M.; Armitage, J.; Collins, R.; Group, M.B.H.P.S.C. Impact of common genetic variation on response to simvastatin therapy among 18 705 participants in the Heart Protection Study. Eur. Heart J. 2013, 34, 982–992. [Google Scholar] [CrossRef]
- Jansen, M.E.; Rigter, T.; Rodenburg, W.; Fleur, T.; Houwink, E.; Weda, M.; Cornel, M.C. Review of the Reported Measures of Clinical Validity and Clinical Utility as Arguments for the Implementation of Pharmacogenetic Testing: A Case Study of Statin-Induced Muscle Toxicity. Front. Pharmacol. 2017, 8, 555. [Google Scholar] [CrossRef]
- Gillis, N.K.; Innocenti, F. Evidence required to demonstrate clinical utility of pharmacogenetic testing: The debate continues. Clin. Pharmacol. Ther. 2014, 96, 655–657. [Google Scholar] [CrossRef]
- Grosse, S.D.; Khoury, M.J. What is the clinical utility of genetic testing? Genet. Med. 2006, 8, 448–450. [Google Scholar] [CrossRef]
- Scott, S.A. Personalizing medicine with clinical pharmacogenetics. Genet. Med. 2011, 13, 987–995. [Google Scholar] [CrossRef]
- Dotson, W.D.; Bowen, M.S.; Kolor, K.; Khoury, M.J. Clinical utility of genetic and genomic services: Context matters. Genet. Med. 2015, 18, 672–674. [Google Scholar] [CrossRef]
- Martin, M.A.; Kroetz, D.L. Abacavir pharmacogenetics—From initial reports to standard of care. Pharmacotherapy 2013, 33, 765–775. [Google Scholar] [CrossRef]
- Deenen, M.J.; Meulendijks, D.; Cats, A.; Sechterberger, M.K.; Severens, J.L.; Boot, H.; Smits, P.H.; Rosing, H.; Mandigers, C.M.; Soesan, M. Upfront genotyping of DPYD* 2A to individualize fluoropyrimidine therapy: A safety and cost analysis. J. Clin. Oncol. 2016, 34, 227–234. [Google Scholar] [CrossRef]
- KNMP. Drug Safety Monitoring. Pharmacogenetics. Available online: https://kennisbank.knmp.nl/article/farmacogenetica/intro.html (accessed on 29 February 2016).
- Verschuren, W.; Blokstra, A.; Picavet, H.; Smit, H. Cohort profile: The Doetinchem cohort study. Int. J. Epidemiol. 2008, 37, 1236–1241. [Google Scholar] [CrossRef]
- Picavet, H.S.J.; Blokstra, A.; Spijkerman, A.M.; Verschuren, W.M. Cohort Profile Update: The Doetinchem Cohort Study 1987–2017: Lifestyle, health and chronic diseases in a life course and ageing perspective. Int. J. Epidemiol. 2017, 46, 1751–1751g. [Google Scholar] [CrossRef]
- Bouwman, F.G.; Boer, J.M.; Imholz, S.; Wang, P.; Verschuren, W.M.; Dollé, M.E.; Mariman, E.C.M. Gender-specific genetic associations of polymorphisms in ACE, AKR1C2, FTO and MMP2 with weight gain over a 10-year period. Genes Nutr. 2014, 9, 434. [Google Scholar] [CrossRef]
- Klop, C.; Welsing, P.M.; Elders, P.J.; Overbeek, J.A.; Souverein, P.C.; Burden, A.M.; van Onzenoort, H.A.; Leufkens, H.G.; Bijlsma, J.W.; de, V.F. Long-term persistence with anti-osteoporosis drugs after fracture. Osteoporos. Int. 2015, 26, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudpour, S.H.; Asselbergs, F.W.; de Keyser, C.E.; Souverein, P.C.; Hofman, A.; Stricker, B.H.; de, B.A.; AH, M.-v.d.Z. Change in prescription pattern as a potential marker for adverse drug reactions of angiotensin converting enzyme inhibitors. Int. J. Clin. Pharm. 2015, 37, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- NHG. NHG Guideline: Cardiovascular Risk Management. Available online: https://www.nhg.org/standaarden/volledig/cardiovasculair-risicomanagement (accessed on 2 December 2016).
- Mosshammer, D.; Schaeffeler, E.; Schwab, M.; Morike, K. Mechanisms and assessment of statin-related muscular adverse effects. Br. J. Clin. Pharmacol. 2014, 78, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Stroes, E.S.; Thompson, P.D.; Corsini, A.; Vladutiu, G.D.; Raal, F.J.; Ray, K.K.; Roden, M.; Stein, E.; Tokgozoglu, L.; Nordestgaard, B.G.; et al. Statin-associated muscle symptoms: Impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur. Heart J. 2015, 36, 1012–1022. [Google Scholar] [CrossRef]
- Peyser, B.; Perry, E.P.; Singh, K.; Gill, R.D.; Mehan, M.R.; Haga, S.B.; Musty, M.D.; Milazzo, N.A.; Savard, D.; Li, Y.-J. Effects of delivering SLCO1B1 pharmacogenetic information in randomized trial and observational settings. Circ. Genom. Precis. Med. 2018, 11, e002228. [Google Scholar] [CrossRef]
- De Jong, H.J.; Damoiseaux, J.G.; Vandebriel, R.J.; Souverein, P.C.; Gremmer, E.R.; Wolfs, M.; Klungel, O.H.; Van, L.H.; Cohen Tervaert, J.W.; Verschuren, W.M. Statin use and markers of immunity in the Doetinchem cohort study. PLoS ONE 2013, 8, e77587. [Google Scholar] [CrossRef]
- Stewart, A. SLCO1B1 Polymorphisms and Statin-Induced Myopathy. PLoS Curr. 2013, 5. [Google Scholar] [CrossRef]
- Vassy, J.L.; Chun, S.; Advani, S.; Ludin, S.A.; Smith, J.G.; Alligood, E.C. Impact of SLCO 1B1 Pharmacogenetic Testing on Patient and Healthcare Outcomes: A Systematic Review. Clin. Pharmacol. Ther. 2018, 106, 360–373. [Google Scholar] [CrossRef]
- de Wert, G.; Dondorp, W.; Clarke, A.; Dequeker, E.M.; Cordier, C.; Deans, Z.; van El, C.G.; Fellmann, F.; Hastings, R.; Hentze, S. Opportunistic genomic screening. Recommendations of the European Society of Human Genetics. Eur. J. Hum. Genet. 2021, 29, 365–377. [Google Scholar] [CrossRef]
- van der Wouden, C.H.; Bank, P.C.; Özokcu, K.; Swen, J.J.; Guchelaar, H.-J. Pharmacist-initiated pre-emptive pharmacogenetic panel testing with clinical decision support in primary care: Record of PGx results and real-world impact. Genes 2019, 10, 416. [Google Scholar] [CrossRef]
- Brunham, L.R.; Baker, S.; Mammen, A.; Mancini, G.J.; Rosenson, R.S. Role of genetics in the prediction of statin-associated muscle symptoms and optimization of statin use and adherence. Cardiovasc. Res. 2018, 114, 1073–1081. [Google Scholar] [CrossRef]
- Lanting, P.; Drenth, E.; Boven, L.; van Hoek, A.; Hijlkema, A.; Poot, E.; van der Vries, G.; Schoevers, R.; Horwitz, E.; Gans, R. Practical Barriers and Facilitators Experienced by Patients, Pharmacists and Physicians to the Implementation of Pharmacogenomic Screening in Dutch Outpatient Hospital Care—An Explorative Pilot Study. J. Pers. Med. 2020, 10, 293. [Google Scholar] [CrossRef]
- Rigter, T.; Jansen, M.E.; Groot, J.M.d.; Janssen, S.W.; Rodenburg, W.; Cornel, M.C. Implementation of pharmacogenetics in primary care: A multi-stakeholder perspective. Front. Genet. 2020, 11, 10. [Google Scholar] [CrossRef]
| SLCO1B1 Genotype | TT (n = 789) | TC (n = 316) | CC (n = 31) | Total |
|---|---|---|---|---|
| Sex | ||||
| Male, % | 51.1 | 51.3 | 38.7 | 50.8 |
| Female, % | 48.9 | 48.7 | 61.3 | 49.2 |
| Age (years), mean (±SD) | 62.5 (9.1) | 62.8 (9.1) | 61.8 (9.4) | 62.6 (9.1) |
| BMI (kg/m2), mean (±SD) | 27.8 (4.5) | 27.4 (4.5) | 28.3 (4.4) | 27.7 (4.6) |
| Diastolic blood pressure (mmHg), mean (±SD) | 83.5 (10.9) | 83.6 (10.4) | 82.5 (11.0) | 83.5 (10.7) |
| Systolic blood pressure (mmHg), mean (±SD) | 136.9 (18.4) | 136.0 (18.0) | 136.5 (21.0) | 136.5 (18.4) |
| Total cholesterol (mmol/L), mean (±SD) | 5.9 (1.2) | 5.9 (1.2) | 6.2 (1.3) | 5.9 (1.2) |
| Smoking | ||||
| Current, % | 27.9 | 25.2 | 32.3 | 27.3 |
| Past, % | 45.9 | 45.5 | 48.4 | 45.8 |
| Never, % | 26.2 | 29.3 | 19.4 | 26.9 |
| Statin dispensed | ||||
| Simvastatin, % | 83.8 | 76.6 | 80.6 | 81.7 |
| Atorvastatin, % | 16.2 | 23.4 | 19.4 | 18.3 |
| Start dose simvastatin (mg) | ||||
| 10 mg, % | 11.0 | 8.8 | 12.0 | 10.4 |
| 20 mg, % | 37.7 | 45.8 | 52.0 | 40.2 |
| 40 mg, % | 51.3 | 45.4 | 36.0 | 49.3 |
| Start dose atorvastatin (mg) | ||||
| 5 mg, % | 0.3 | 0.0 | 0.0 | 0.2 |
| 10 mg, % | 57.0 | 54.1 | 66.7 | 56.3 |
| 20 mg, % | 33.6 | 36.5 | 33.3 | 34.6 |
| 40 mg, % | 6.3 | 8.1 | 0.0 | 6.7 |
| 80 mg, % | 1.6 | 1.4 | 0.0 | 1.4 |
| Simvastatin | Atorvastatin | ||||
|---|---|---|---|---|---|
| Dose Change | Event (%) | Crude OR (95% CI) | Adjusted OR a (95% CI) | Crude OR (95% CI) | Adjusted OR b (95% CI) |
| TT | 30 (4.1) | ||||
| TC/CC | 9 (3.0) | 0.50 (0.19–1.31) | 0.42 (0.15–1.16) | 1.72 (0.42–7.11) | 1.97 (0.43–9.04) |
| Total | 39 (3.4) | ||||
| Change (discontinuation or switch) | Event (%) | Crude OR (95% CI) | Adjusted OR a (95% CI) | Crude OR (95% CI) | Adjusted OR a (95% CI) |
| TT | 529 (67.1) | ||||
| TC/CC | 223 (64.3) | 0.93 (0.69–1.25) | 0.80 (0.58–1.11) | 0.58 (0.31–1.09) | 0.57 (0.27–1.18) |
| Total | 752 (66.2) | ||||
| Time to establish stable dosing regimen | Median (IQR), days | Crude OR (95% CI) | Adjusted OR a (95% CI) | Crude OR (95% CI) | Adjusted OR a (95% CI) |
| TT | 90 (97) | ||||
| TC/CC | 89 (108) | 1.02 (0.87–1.21) | 1.06 (0.89–1.26) | 0.84 (0.61–1.14) | 0.82 (0.57–1.16) |
| Total | 90 (97) | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jansen, M.E.; Rigter, T.; Fleur, T.M.C.; Souverein, P.C.; Verschuren, W.M.M.; Vijverberg, S.J.; Swen, J.J.; Rodenburg, W.; Cornel, M.C. Predictive Value of SLCO1B1 c.521T>C Polymorphism on Observed Changes in the Treatment of 1136 Statin-Users. Genes 2023, 14, 456. https://doi.org/10.3390/genes14020456
Jansen ME, Rigter T, Fleur TMC, Souverein PC, Verschuren WMM, Vijverberg SJ, Swen JJ, Rodenburg W, Cornel MC. Predictive Value of SLCO1B1 c.521T>C Polymorphism on Observed Changes in the Treatment of 1136 Statin-Users. Genes. 2023; 14(2):456. https://doi.org/10.3390/genes14020456
Chicago/Turabian StyleJansen, Marleen E., Tessel Rigter, Thom M. C. Fleur, Patrick C. Souverein, W. M. Monique Verschuren, Susanne J. Vijverberg, Jesse J. Swen, Wendy Rodenburg, and Martina C. Cornel. 2023. "Predictive Value of SLCO1B1 c.521T>C Polymorphism on Observed Changes in the Treatment of 1136 Statin-Users" Genes 14, no. 2: 456. https://doi.org/10.3390/genes14020456
APA StyleJansen, M. E., Rigter, T., Fleur, T. M. C., Souverein, P. C., Verschuren, W. M. M., Vijverberg, S. J., Swen, J. J., Rodenburg, W., & Cornel, M. C. (2023). Predictive Value of SLCO1B1 c.521T>C Polymorphism on Observed Changes in the Treatment of 1136 Statin-Users. Genes, 14(2), 456. https://doi.org/10.3390/genes14020456








