Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies
Abstract
:1. Introduction
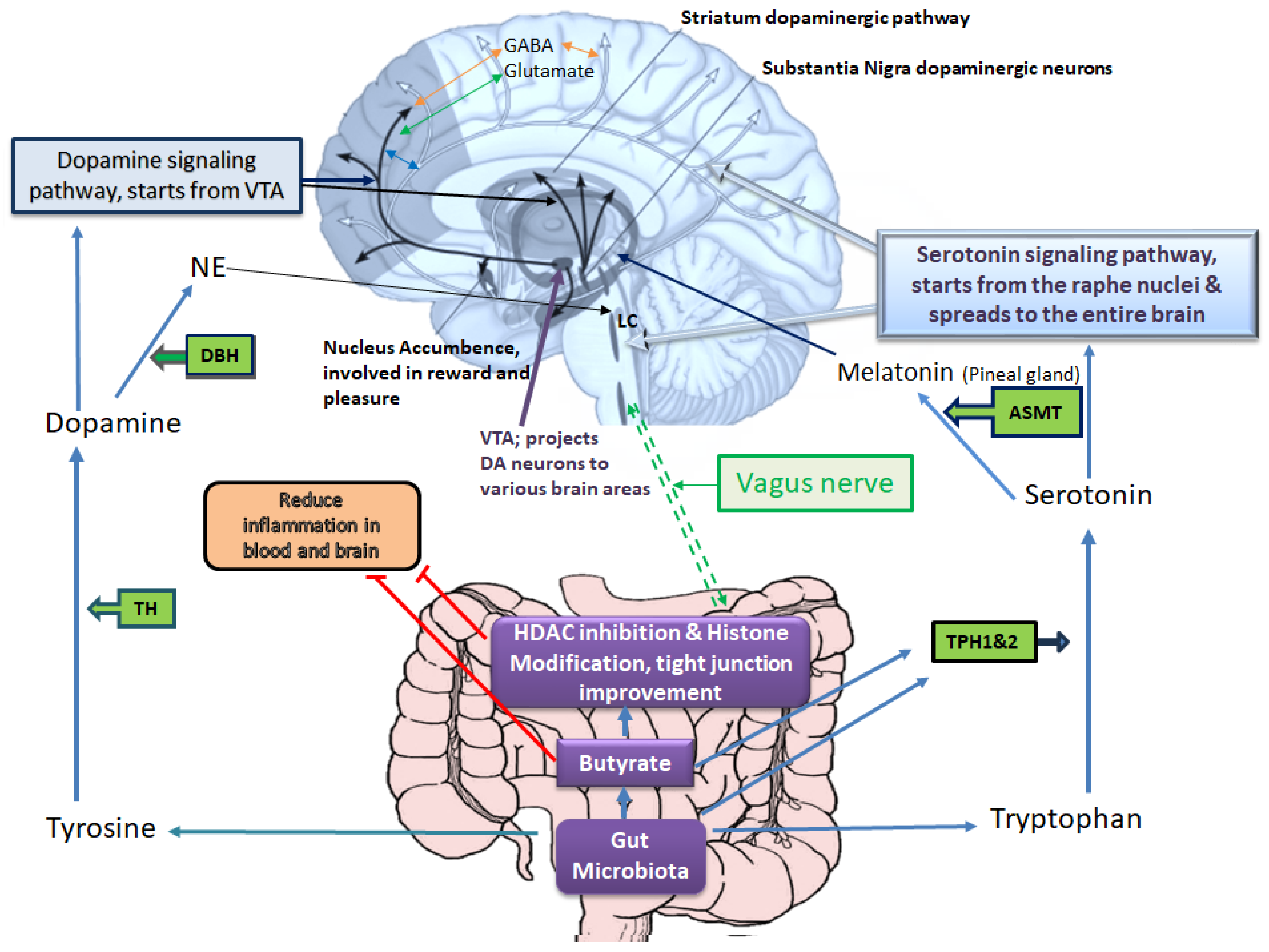
2. Roles of Leaky Gut in the Pathophysiology of Depressive Disorders via Alterations of Gut Microbiota-Derived Metabolites Affecting the Epigenetic Landscape
3. The Gut Microbiome Composition and Its Role in the Development of Depressive Disorders via Epigenetic Alterations
4. Depression and Maternal Diet and Environmental Contaminants Which Affect the Gut Microbiome and Epigenome
5. Microbiota Derived SCFAs for Depressive Disorders Therapy via Epigenetic Changes
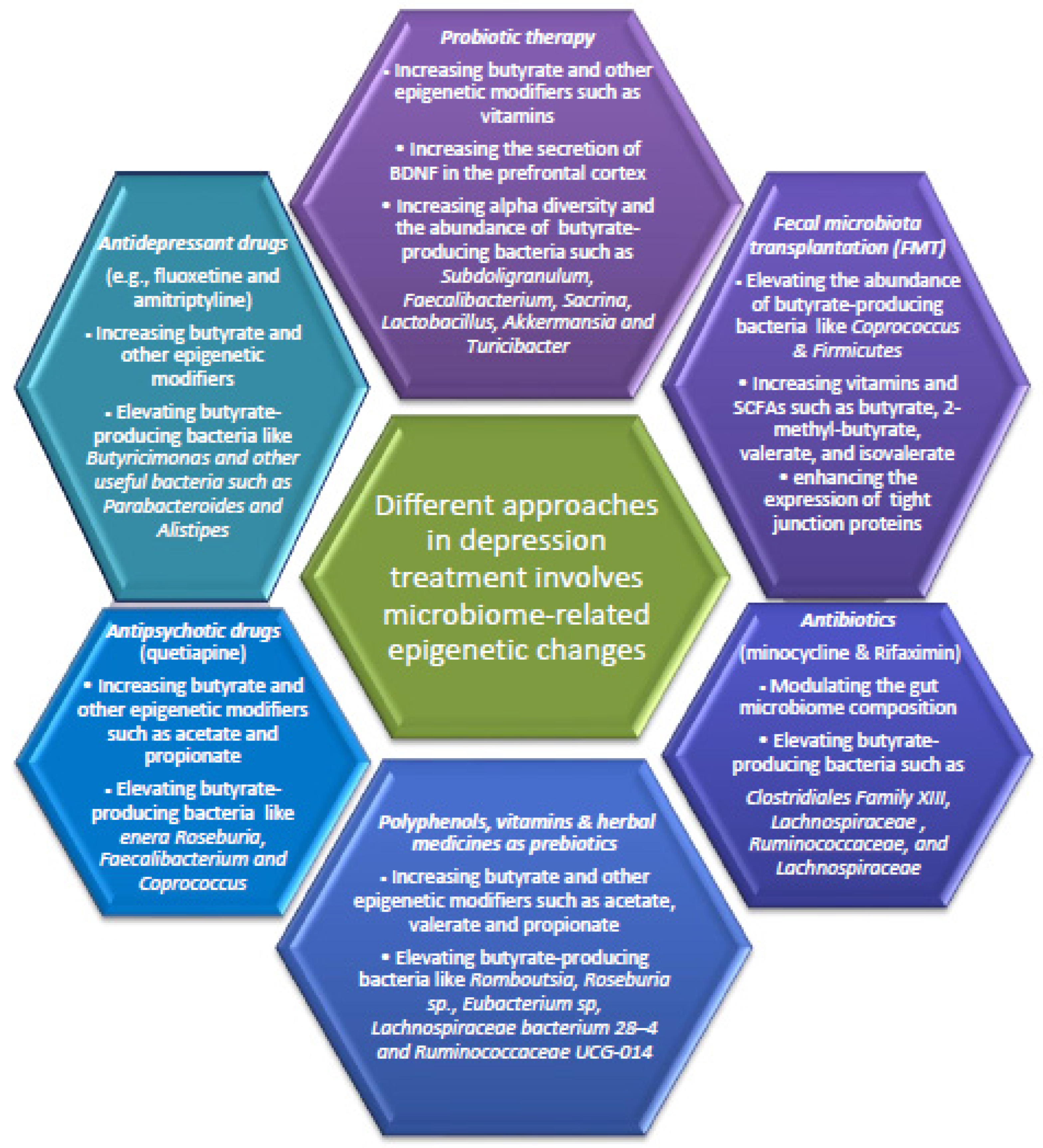
6. Fecal Microbiota Transplantation and Probiotic Therapy for Depressive Disorders through Epigenetic Changes
7. Polyphenols and Herbal Medicine as Prebiotics for Depressive Disorders via Epigenetic Changes
8. The Antipsychotic and Antidepressant Drugs also Improve Depression in Part by Modulating Gut Microbiota Associated Epigenetic Changes
9. Antibiotics for Depressive-like Symptoms by Modulating Gut Microbiota-Related Epigenetic Changes
10. Gut Microbiota-Related Vitamins Modulating Epigenetic Codes in Depressive
11. Potentials and Challenges for Translating Gut Microbiome Research to the Clinic for Treatments of Depressive Disorders
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smith, K.; De Torres, I. A world of depression. Nature 2014, 515, 180–181. [Google Scholar] [CrossRef]
- Bromet, E.; Andrade, L.H.; Hwang, I.; Sampson, N.A.; Alonso, J.; De Girolamo, G.; De Graaf, R.; Demyttenaere, K.; Hu, C.; Iwata, N. Cross-national epidemiology of DSM-IV major depressive episode. BMC Med. 2011, 9, 90. [Google Scholar] [CrossRef]
- Brigitta, B. Pathophysiology of depression and mechanisms of treatment. Dialogues Clin. Neurosci. 2002, 4, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Salari, N.; Hosseinian-Far, A.; Jalali, R.; Vaisi-Raygani, A.; Rasoulpoor, S.; Mohammadi, M.; Rasoulpoor, S.; Khaledi-Paveh, B. Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: A systematic review and meta-analysis. Glob. Health 2020, 16, 57. [Google Scholar] [CrossRef] [PubMed]
- Thase, M.E. Depression and sleep: Pathophysiology and treatment. Dialogues Clin. Neurosci. 2006, 8, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.S.; Aguilar-Gaxiola, S.; Alonso, J.; Angermeyer, M.C.; Borges, G.; Bromet, E.J.; Bruffaerts, R.; De Girolamo, G.; De Graaf, R.; Gureje, O. Use of mental health services for anxiety, mood, and substance disorders in 17 countries in the WHO world mental health surveys. Lancet 2007, 370, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.-L.; Bleich, A.; Van Praag, H.M. The monoamine hypothesis of depression: The case for serotonin. In Role of Serotonin in Psychiatric Disorders; Routledge: London, UK, 2023; pp. 91–128. [Google Scholar]
- Cosci, F.; Chouinard, G. The monoamine hypothesis of depression revisited: Could it mechanistically novel antidepressant strategies? In Neurobiology of Depression; Elsevier: Amsterdam, The Netherlands, 2019; pp. 63–73. [Google Scholar]
- Moret, C.; Briley, M. The importance of norepinephrine in depression. Neuropsychiatr. Dis. Treat. 2011, 7, 9–13. [Google Scholar]
- Duarte, P.; Cuadrado, A.; León, R. Monoamine oxidase inhibitors: From classic to new clinical approaches. In Reactive Oxygen Species: Network Pharmacology and Therapeutic Applications; Springer: Chem, Switezrland, 2021; pp. 229–259. [Google Scholar]
- Edinoff, A.N.; Akuly, H.A.; Hanna, T.A.; Ochoa, C.O.; Patti, S.J.; Ghaffar, Y.A.; Kaye, A.D.; Viswanath, O.; Urits, I.; Boyer, A.G. Selective serotonin reuptake inhibitors and adverse effects: A narrative review. Neurol. Int. 2021, 13, 387–401. [Google Scholar] [CrossRef]
- Fries, G.R.; Saldana, V.A.; Finnstein, J.; Rein, T. Molecular pathways of major depressive disorder converge on the synapse. Mol. Psychiatry 2023, 28, 284–297. [Google Scholar] [CrossRef]
- Correia, A.S.; Vale, N. Tryptophan metabolism in depression: A narrative review with a focus on serotonin and kynurenine pathways. Int. J. Mol. Sci. 2022, 23, 8493. [Google Scholar] [CrossRef]
- Fatima, M.; Ahmad, M.H.; Srivastav, S.; Rizvi, M.A.; Mondal, A.C. A selective D2 dopamine receptor agonist alleviates depression through up-regulation of tyrosine hydroxylase and increased neurogenesis in hippocampus of the prenatally stressed rats. Neurochem. Int. 2020, 136, 104730. [Google Scholar] [CrossRef] [PubMed]
- Banskota, S.; Khan, W.I. Gut-derived serotonin and its emerging roles in immune function, inflammation, metabolism and the gut–brain axis. Curr. Opin. Endocrinol. Diabetes Obes. 2022, 29, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Waclawiková, B.; El Aidy, S. Role of microbiota and tryptophan metabolites in the remote effect of intestinal inflammation on brain and depression. Pharmaceuticals 2018, 11, 63. [Google Scholar] [CrossRef]
- Mittal, R.; Debs, L.H.; Patel, A.P.; Nguyen, D.; Patel, K.; O’Connor, G.; Grati, M.h.; Mittal, J.; Yan, D.; Eshraghi, A.A. Neurotransmitters: The critical modulators regulating gut–brain axis. J. Cell. Physiol. 2017, 232, 2359–2372. [Google Scholar] [CrossRef] [PubMed]
- Lener, M.S.; Niciu, M.J.; Ballard, E.D.; Park, M.; Park, L.T.; Nugent, A.C.; Zarate, C.A., Jr. Glutamate and γ-aminobutyric acid systems in the pathophysiology of major depression and antidepressant response to ketamine. Biol. Psychiatry 2017, 81, 886–897. [Google Scholar] [CrossRef]
- Hasler, G. Pathophysiology of depression: Do we have any solid evidence of interest to clinicians? World Psychiatry 2010, 9, 155. [Google Scholar] [CrossRef]
- Duman, R.S.; Sanacora, G.; Krystal, J.H. Altered connectivity in depression: GABA and glutamate neurotransmitter deficits and reversal by novel treatments. Neuron 2019, 102, 75–90. [Google Scholar] [CrossRef]
- Lukić, I.; Ivković, S.; Mitić, M.; Adžić, M. Tryptophan metabolites in depression: Modulation by gut microbiota. Front. Behav. Neurosci. 2022, 16, 987697. [Google Scholar] [CrossRef]
- Flasbeck, V.; Hirsch, J.; Petrak, F.; Meier, J.J.; Herpertz, S.; Gatermann, S.; Juckel, G. Microbiome composition and central serotonergic activity in patients with depression and type 1 diabetes. Eur. Arch. Psychiatry Clin. Neurosci. 2023, in press. [Google Scholar] [CrossRef]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell 2015, 161, 264–276. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, C.; Zhao, D.; Wang, C.; Li, C. Dietary proteins regulate serotonin biosynthesis and catabolism by specific gut microbes. J. Agric. Food Chem. 2020, 68, 5880–5890. [Google Scholar] [CrossRef]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F., III; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015, 29, 1395. [Google Scholar] [CrossRef]
- Nankova, B.B.; Agarwal, R.; MacFabe, D.F.; La Gamma, E.F. Enteric bacterial metabolites propionic and butyric acid modulate gene expression, including CREB-dependent catecholaminergic neurotransmission, in PC12 cells-possible relevance to autism spectrum disorders. PLoS ONE 2014, 9, e103740. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.; Gao, Z.; Liu, X.; Rong, Z.; Jia, J.; Kang, K.; Guo, W.; Li, J. Intravenous administration of sodium propionate induces antidepressant or prodepressant effect in a dose dependent manner. Sci. Rep. 2020, 10, 19917. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kasper, L.H. The role of microbiome in central nervous system disorders. Brain Behav. Immun. 2014, 38, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Zhang, L.; Zhang, B.; Kong, L.; Pan, X.; Goossens, T.; Song, Z. Dietary sodium butyrate improves female broiler breeder performance and offspring immune function by enhancing maternal intestinal barrier and microbiota. Poult. Sci. 2023, 102, 102658. [Google Scholar] [CrossRef]
- Erny, D.; Hrabě de Angelis, A.L.; Jaitin, D.; Wieghofer, P.; Staszewski, O.; David, E.; Keren-Shaul, H.; Mahlakoiv, T.; Jakobshagen, K.; Buch, T. Host microbiota constantly control maturation and function of microglia in the CNS. Nat. Neurosci. 2015, 18, 965–977. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 2016, 164, 337–340. [Google Scholar] [CrossRef]
- Stilling, R.M.; Dinan, T.G.; Cryan, J.F. Microbial genes, brain & behaviour–epigenetic regulation of the gut–brain axis. Genes Brain Behav. 2014, 13, 69–86. [Google Scholar]
- Pearson-Leary, J.; Zhao, C.; Bittinger, K.; Eacret, D.; Luz, S.; Vigderman, A.S.; Dayanim, G.; Bhatnagar, S. The gut microbiome regulates the increases in depressive-type behaviors and in inflammatory processes in the ventral hippocampus of stress vulnerable rats. Mol. Psychiatry 2020, 25, 1068–1079. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, R.; Cheng, M.; Wang, L.; Chao, J.; Li, J.; Zheng, P.; Xie, P.; Zhang, Z.; Yao, H. Gut microbiota from NLRP3-deficient mice ameliorates depressive-like behaviors by regulating astrocyte dysfunction via circHIPK2. Microbiome 2019, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Janowska, M.; Rog, J.; Karakula-Juchnowicz, H. Disruptions within gut microbiota composition induced by improper antibiotics therapy as a probable trigger factor for development of depression—Case Reports. Ann. Agric. Environ. Med. 2021, 28, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Park, A.; Collins, J.; Blennerhassett, P.; Ghia, J.; Verdu, E.; Bercik, P.; Collins, S. Altered colonic function and microbiota profile in a mouse model of chronic depression. Neurogastroenterol. Motil. 2013, 25, 733-e575, Erratum in Neurogastroenterol. Motil. 2013, 25, 857. [Google Scholar] [CrossRef] [PubMed]
- Desbonnet, L.; Clarke, G.; Traplin, A.; O’Sullivan, O.; Crispie, F.; Moloney, R.D.; Cotter, P.D.; Dinan, T.G.; Cryan, J.F. Gut microbiota depletion from early adolescence in mice: Implications for brain and behaviour. Brain Behav. Immun. 2015, 48, 165–173. [Google Scholar] [CrossRef]
- Begum, N.; Mandhare, A.; Tryphena, K.P.; Srivastava, S.; Shaikh, M.F.; Singh, S.B.; Khatri, D.K. Epigenetics in depression and gut-brain axis: A molecular crosstalk. Front. Aging Neurosci. 2022, 14, 1048333. [Google Scholar] [CrossRef]
- Huang, C.; Yang, X.; Zeng, B.; Zeng, L.; Gong, X.; Zhou, C.; Xia, J.; Lian, B.; Qin, Y.; Yang, L. Proteomic analysis of olfactory bulb suggests CACNA1E as a promoter of CREB signaling in microbiota-induced depression. J. Proteom. 2019, 194, 132–147. [Google Scholar] [CrossRef]
- Moloney, G.M.; O’Leary, O.F.; Salvo-Romero, E.; Desbonnet, L.; Shanahan, F.; Dinan, T.G.; Clarke, G.; Cryan, J.F. Microbial regulation of hippocampal miRNA expression: Implications for transcription of kynurenine pathway enzymes. Behav. Brain Res. 2017, 334, 50–54. [Google Scholar] [CrossRef]
- Stilling, R.M.; Moloney, G.M.; Ryan, F.J.; Hoban, A.E.; Bastiaanssen, T.F.; Shanahan, F.; Clarke, G.; Claesson, M.J.; Dinan, T.G.; Cryan, J.F. Social interaction-induced activation of RNA splicing in the amygdala of microbiome-deficient mice. Elife 2018, 7, e33070. [Google Scholar] [CrossRef]
- Rudzki, L.; Maes, M. From “leaky gut” to impaired glia-neuron communication in depression. In Major Depressive Disorder: Rethinking and Understanding Recent Discoveries; Springer: Berlin/Heidelberg, Germany, 2021; pp. 129–155. [Google Scholar]
- Arrieta, M.-C.; Bistritz, L.; Meddings, J.B. Alterations in intestinal permeability. Gut 2006, 55, 1512–1520. [Google Scholar] [CrossRef]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef]
- Mass, M.; Kubera, M.; Leunis, J.-C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuroendocrinol. Lett. 2008, 29, 117–124. [Google Scholar]
- Ait-Belgnaoui, A.; Bradesi, S.; Fioramonti, J.; Theodorou, V.; Bueno, L. Acute stress-induced hypersensitivity to colonic distension depends upon increase in paracellular permeability: Role of myosin light chain kinase. Pain 2005, 113, 141–147. [Google Scholar] [CrossRef]
- Simpson, C.A.; Adler, C.; du Plessis, M.R.; Landau, E.R.; Dashper, S.G.; Reynolds, E.C.; Schwartz, O.S.; Simmons, J.G. Oral microbiome composition, but not diversity, is associated with adolescent anxiety and depression symptoms. Physiol. Behav. 2020, 226, 113126. [Google Scholar] [CrossRef] [PubMed]
- Wingfield, B.; Lapsley, C.; McDowell, A.; Miliotis, G.; McLafferty, M.; O’Neill, S.M.; Coleman, S.; McGinnity, T.M.; Bjourson, A.J.; Murray, E.K. Variations in the oral microbiome are associated with depression in young adults. Sci. Rep. 2021, 11, 15009. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.V.-A. Gut microbiome composition and diversity are related to human personality traits. Hum. Microbiome J. 2020, 15, 100069. [Google Scholar] [CrossRef]
- Maes, M.; Kubera, M.; Leunis, J.C.; Berk, M.; Geffard, M.; Bosmans, E. In depression, bacterial translocation may drive inflammatory responses, oxidative and nitrosative stress (O&NS), and autoimmune responses directed against O&NS-damaged neoepitopes. Acta Psychiatr. Scand. 2013, 127, 344–354. [Google Scholar]
- Rubas, N.C.; Maunakea, A. Medical school hotline: Immunoepigenetic-microbiome Axis: Implications for health disparities research in native Hawaiians and pacific Islanders. Hawai’i J. Health Soc. Welf. 2021, 80, 195. [Google Scholar]
- Long, D.; Liu, M.; Li, H.; Song, J.; Jiang, X.; Wang, G.; Yang, X. Dysbacteriosis induces abnormal neurogenesis via LPS in a pathway requiring NF-κB/IL-6. Pharmacol. Res. 2021, 167, 105543. [Google Scholar] [CrossRef]
- Moussaoui, N.; Jacobs, J.P.; Larauche, M.; Biraud, M.; Million, M.; Mayer, E.; Taché, Y. Chronic early-life stress in rat pups alters basal corticosterone, intestinal permeability, and fecal microbiota at weaning: Influence of sex. J. Neurogastroenterol. Motil. 2017, 23, 135–143. [Google Scholar] [CrossRef]
- Huang, Y.; Shi, X.; Li, Z.; Shen, Y.; Shi, X.; Wang, L.; Li, G.; Yuan, Y.; Wang, J.; Zhang, Y. Possible association of Firmicutes in the gut microbiota of patients with major depressive disorder. Neuropsychiatr. Dis. Treat. 2018, 3329–3337. [Google Scholar] [CrossRef] [PubMed]
- de Clercq, N.C.; Groen, A.K.; Romijn, J.A.; Nieuwdorp, M. Gut microbiota in obesity and undernutrition. Adv. Nutr. 2016, 7, 1080–1089. [Google Scholar] [CrossRef] [PubMed]
- Soret, R.; Chevalier, J.; De Coppet, P.; Poupeau, G.; Derkinderen, P.; Segain, J.P.; Neunlist, M. Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats. Gastroenterology 2010, 138, 1772–1782.e4. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, J.; He, T.; Becker, S.; Zhang, G.; Li, D.; Ma, X. Butyrate: A double-edged sword for health? Adv. Nutr. 2018, 9, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D.; Nakanishi, Y.; Uetake, C.; Kato, K.; Kato, T. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhou, J.; Qi, H.; Wei, J.; Yang, Y.; Yue, J.; Liu, X.; Zhang, Y.; Yang, R. LncRNA lncLy6C induced by microbiota metabolite butyrate promotes differentiation of Ly6Chigh to Ly6Cint/neg macrophages through lncLy6C/C/EBPβ/Nr4A1 axis. Cell Discov. 2020, 6, 87. [Google Scholar] [CrossRef]
- Yamawaki, Y.; Fuchikami, M.; Morinobu, S.; Segawa, M.; Matsumoto, T.; Yamawaki, S. Antidepressant-like effect of sodium butyrate (HDAC inhibitor) and its molecular mechanism of action in the rat hippocampus. World J. Biol. Psychiatry 2012, 13, 458–467. [Google Scholar] [CrossRef]
- Noriega, B.S.; Sanchez-Gonzalez, M.A.; Salyakina, D.; Coffman, J. Understanding the impact of omega-3 rich diet on the gut microbiota. Case Rep. Med. 2016, 2016, 6. [Google Scholar] [CrossRef]
- Wong, M.-L.; Inserra, A.; Lewis, M.; Mastronardi, C.A.; Leong, L.; Choo, J.; Kentish, S.; Xie, P.; Morrison, M.; Wesselingh, S. Inflammasome signaling affects anxiety-and depressive-like behavior and gut microbiome composition. Mol. Psychiatry 2016, 21, 797–805. [Google Scholar] [CrossRef]
- Tillmann, S.; Abildgaard, A.; Winther, G.; Wegener, G. Altered fecal microbiota composition in the Flinders sensitive line rat model of depression. Psychopharmacology 2019, 236, 1445–1457. [Google Scholar] [CrossRef]
- Yu, M.; Jia, H.; Zhou, C.; Yang, Y.; Zhao, Y.; Yang, M.; Zou, Z. Variations in gut microbiota and fecal metabolic phenotype associated with depression by 16S rRNA gene sequencing and LC/MS-based metabolomics. J. Pharm. Biomed. Anal. 2017, 138, 231–239. [Google Scholar] [CrossRef]
- Bangsgaard Bendtsen, K.M.; Krych, L.; Sørensen, D.B.; Pang, W.; Nielsen, D.S.; Josefsen, K.; Hansen, L.H.; Sørensen, S.J.; Hansen, A.K. Gut microbiota composition is correlated to grid floor induced stress and behavior in the BALB/c mouse. PLoS ONE 2012, 7, e46231. [Google Scholar] [CrossRef]
- Chevalier, G.; Siopi, E.; Guenin-Macé, L.; Pascal, M.; Laval, T.; Rifflet, A.; Boneca, I.G.; Demangel, C.; Colsch, B.; Pruvost, A. Effect of gut microbiota on depressive-like behaviors in mice is mediated by the endocannabinoid system. Nat. Commun. 2020, 11, 6363. [Google Scholar] [CrossRef]
- Zheng, P.; Zeng, B.; Zhou, C.; Liu, M.; Fang, Z.; Xu, X.; Zeng, L.; Chen, J.; Fan, S.; Du, X. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol. Psychiatry 2016, 21, 786–796. [Google Scholar] [CrossRef]
- Valles-Colomer, M.; Falony, G.; Darzi, Y.; Tigchelaar, E.F.; Wang, J.; Tito, R.Y.; Schiweck, C.; Kurilshikov, A.; Joossens, M.; Wijmenga, C. The neuroactive potential of the human gut microbiota in quality of life and depression. Nat. Microbiol. 2019, 4, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Radjabzadeh, D.; Bosch, J.A.; Uitterlinden, A.G.; Zwinderman, A.H.; Ikram, M.A.; van Meurs, J.B.; Luik, A.I.; Nieuwdorp, M.; Lok, A.; van Duijn, C.M. Gut microbiome-wide association study of depressive symptoms. Nat. Commun. 2022, 13, 7128. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.T.; Rowan-Nash, A.D.; Sheehan, A.E.; Walsh, R.F.; Sanzari, C.M.; Korry, B.J.; Belenky, P. Reductions in anti-inflammatory gut bacteria are associated with depression in a sample of young adults. Brain Behav. Immun. 2020, 88, 308–324. [Google Scholar] [CrossRef] [PubMed]
- Ising, M.; Maccarrone, G.; Brückl, T.; Scheuer, S.; Hennings, J.; Holsboer, F.; Turck, C.W.; Uhr, M.; Lucae, S. FKBP5 gene expression predicts antidepressant treatment outcome in depression. Int. J. Mol. Sci. 2019, 20, 485. [Google Scholar] [CrossRef]
- Chen, H.-M.; Chung, Y.-C.E.; Chen, H.-C.; Liu, Y.-W.; Chen, I.-M.; Lu, M.-L.; Hsiao, F.S.-H.; Chen, C.-H.; Huang, M.-C.; Shih, W.-L. Exploration of the relationship between gut microbiota and fecal microRNAs in patients with major depressive disorder. Sci. Rep. 2022, 12, 20977. [Google Scholar] [CrossRef] [PubMed]
- Buchenauer, L.; Haange, S.-B.; Bauer, M.; Rolle-Kampczyk, U.E.; Wagner, M.; Stucke, J.; Elter, E.; Fink, B.; Vass, M.; von Bergen, M. Maternal exposure of mice to glyphosate induces depression-and anxiety-like behavior in the offspring via alterations of the gut-brain axis. Sci. Total Environ. 2023, 905, 167034. [Google Scholar] [CrossRef]
- Chen, F.; Chen, Z.; Chen, M.; Chen, G.; Huang, Q.; Yang, X.; Yin, H.; Chen, L.; Zhang, W.; Lin, H. Reduced stress-associated FKBP5 DNA methylation together with gut microbiota dysbiosis is linked with the progression of obese PCOS patients. npj Biofilms Microbiomes 2021, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Becerra, C.Y.; Wells, R.K.; Kunihiro, B.P.; Lee, R.H.; Umeda, L.; Allan, N.P.; Rubas, N.C.; McCracken, T.A.; Nunokawa, C.K.; Lee, M.-H. Examining the immunoepigenetic-gut microbiome axis in the context of self-esteem among Native Hawaiians and other Pacific Islanders. Front. Genet. 2023, 14, 1125217. [Google Scholar] [CrossRef] [PubMed]
- Xie, A.; Ensink, E.; Li, P.; Gordevičius, J.; Marshall, L.L.; George, S.; Pospisilik, J.A.; Aho, V.T.; Houser, M.C.; Pereira, P.A. Bacterial butyrate in Parkinson’s disease is linked to epigenetic changes and depressive symptoms. Mov. Disord. 2022, 37, 1644–1653. [Google Scholar] [CrossRef] [PubMed]
- Aitbali, Y.; Ba-M’hamed, S.; Elhidar, N.; Nafis, A.; Soraa, N.; Bennis, M. Glyphosate based-herbicide exposure affects gut microbiota, anxiety and depression-like behaviors in mice. Neurotoxicology Teratol. 2018, 67, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, S.; Le Roy, T.; Furgiuele, S.; Coste, V.; Bindels, L.B.; Leyrolle, Q.; Neyrinck, A.M.; Quoilin, C.; Amadieu, C.; Petit, G. Gut microbiota-induced changes in β-hydroxybutyrate metabolism are linked to altered sociability and depression in alcohol use disorder. Cell Rep. 2020, 33, 108238. [Google Scholar] [CrossRef] [PubMed]
- McGaughey, K.D.; Yilmaz-Swenson, T.; Elsayed, N.M.; Cruz, D.A.; Rodriguiz, R.M.; Kritzer, M.D.; Peterchev, A.V.; Roach, J.; Wetsel, W.C.; Williamson, D.E. Relative abundance of Akkermansia spp. and other bacterial phylotypes correlates with anxiety-and depressive-like behavior following social defeat in mice. Sci. Rep. 2019, 9, 3281. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, H.; Rao, X.; Liu, L.; Zheng, P.; Li, W.; Zhou, W.; Chai, T.; Ji, P.; Song, J. Proteomic profiling of lysine acetylation indicates mitochondrial dysfunction in the hippocampus of gut microbiota-absent mice. Front. Mol. Neurosci. 2021, 14, 594332. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wang, H.; Chen, X.; Zhang, Y.; Li, W.; Rao, X.; Liu, Y.; Zhao, L.; Pu, J.; Gui, S. Integrative analysis of long non-coding RNAs, messenger RNAs, and MicroRNAs indicates the neurodevelopmental dysfunction in the Hippocampus of gut microbiota-dysbiosis mice. Front. Mol. Neurosci. 2022, 14, 745437. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, J.; Gao, Z.; Zhang, Y.; Gu, J. Gut microbiota-induced microRNA-206-3p increases anxiety-like behaviors by inhibiting expression of Cited2 and STK39. Microb. Pathog. 2023, 176, 106008. [Google Scholar] [CrossRef]
- Ma, L.; Wang, L.; Chang, L.; Shan, J.; Qu, Y.; Wang, X.; Fujita, Y.; Hashimoto, K. A role of microRNA-149 in the prefrontal cortex for prophylactic actions of (R)-ketamine in inflammation model. Neuropharmacology 2022, 219, 109250. [Google Scholar] [CrossRef]
- Liu, L.; Wang, H.; Yu, Y.; Zeng, B.; Rao, X.; Chen, J.; Zhou, C.; Zheng, P.; Pu, J.; Yang, L. Microbial regulation of a lincRNA–miRNA–mRNA network in the mouse hippocampus. Epigenomics 2020, 12, 1377–1387. [Google Scholar] [CrossRef]
- Han, K.; Ji, L.; Wang, C.; Shao, Y.; Chen, C.; Liu, L.; Feng, M.; Yang, F.; Wu, X.; Li, X. The host genetics affects gut microbiome diversity in Chinese depressed patients. Front. Genet. 2023, 13, 976814. [Google Scholar] [CrossRef]
- Liu, X.; Li, X.; Xia, B.; Jin, X.; Zou, Q.; Zeng, Z.; Zhao, W.; Yan, S.; Li, L.; Yuan, S. High-fiber diet mitigates maternal obesity-induced cognitive and social dysfunction in the offspring via gut-brain axis. Cell Metab. 2021, 33, 923–938.e6. [Google Scholar] [CrossRef]
- Bruce-Keller, A.J.; Fernandez-Kim, S.-O.; Townsend, R.L.; Kruger, C.; Carmouche, R.; Newman, S.; Salbaum, J.M.; Berthoud, H.-R. Maternal obese-type gut microbiota differentially impact cognition, anxiety and compulsive behavior in male and female offspring in mice. PLoS ONE 2017, 12, e0175577. [Google Scholar] [CrossRef]
- Radford-Smith, D.E.; Probert, F.; Burnet, P.W.; Anthony, D.C. Modifying the maternal microbiota alters the gut–brain metabolome and prevents emotional dysfunction in the adult offspring of obese dams. Proc. Natl. Acad. Sci. USA 2022, 119, e2108581119. [Google Scholar] [CrossRef]
- Hebert, J.C.; Radford-Smith, D.E.; Probert, F.; Ilott, N.; Chan, K.W.; Anthony, D.C.; Burnet, P.W. Mom’s diet matters: Maternal prebiotic intake in mice reduces anxiety and alters brain gene expression and the fecal microbiome in offspring. Brain Behav. Immun. 2021, 91, 230–244. [Google Scholar] [CrossRef]
- Yu, L.; Zhong, X.; He, Y.; Shi, Y. Butyrate, but not propionate, reverses maternal diet-induced neurocognitive deficits in offspring. Pharmacol. Res. 2020, 160, 105082. [Google Scholar] [CrossRef]
- Liu, Z.; Li, L.; Ma, S.; Ye, J.; Zhang, H.; Li, Y.; Sair, A.T.; Pan, J.; Liu, X.; Li, X. High-dietary fiber intake alleviates antenatal obesity-induced postpartum depression: Roles of gut microbiota and microbial metabolite short-chain fatty acid involved. J. Agric. Food Chem. 2020, 68, 13697–13710. [Google Scholar] [CrossRef]
- Dickerson, A.S.; Wu, A.C.; Liew, Z.; Weisskopf, M. A scoping review of non-occupational exposures to environmental pollutants and adult depression, anxiety, and suicide. Curr. Environ. Health Rep. 2020, 7, 256–271. [Google Scholar] [CrossRef] [PubMed]
- Giambò, F.; Costa, C.; Teodoro, M.; Fenga, C. Role-playing between environmental pollutants and human gut microbiota: A complex bidirectional interaction. Front. Med. 2022, 9, 810397. [Google Scholar] [CrossRef] [PubMed]
- Guardia-Escote, L.; Basaure, P.; Biosca-Brull, J.; Cabré, M.; Blanco, J.; Pérez-Fernández, C.; Sánchez-Santed, F.; Domingo, J.L.; Colomina, M.T. APOE genotype and postnatal chlorpyrifos exposure modulate gut microbiota and cerebral short-chain fatty acids in preweaning mice. Food Chem. Toxicol. 2020, 135, 110872. [Google Scholar] [CrossRef]
- Perez-Fernandez, C.; Morales-Navas, M.; Guardia-Escote, L.; Garrido-Cárdenas, J.A.; Colomina, M.T.; Giménez, E.; Sánchez-Santed, F. Long-term effects of low doses of Chlorpyrifos exposure at the preweaning developmental stage: A locomotor, pharmacological, brain gene expression and gut microbiome analysis. Food Chem. Toxicol. 2020, 135, 110865. [Google Scholar] [CrossRef]
- Martinez, A.; Al-Ahmad, A.J. Effects of glyphosate and aminomethylphosphonic acid on an isogeneic model of the human blood-brain barrier. Toxicol. Lett. 2019, 304, 39–49. [Google Scholar] [CrossRef]
- Winstone, J.K.; Pathak, K.V.; Winslow, W.; Piras, I.S.; White, J.; Sharma, R.; Huentelman, M.J.; Pirrotte, P.; Velazquez, R. Glyphosate infiltrates the brain and increases pro-inflammatory cytokine TNFα: Implications for neurodegenerative disorders. J. Neuroinflammation 2022, 19, 193. [Google Scholar] [CrossRef] [PubMed]
- Dechartres, J.; Pawluski, J.L.; Gueguen, M.M.; Jablaoui, A.; Maguin, E.; Rhimi, M.; Charlier, T.D. Glyphosate and glyphosate-based herbicide exposure during the peripartum period affects maternal brain plasticity, maternal behaviour and microbiome. J. Neuroendocrinol. 2019, 31, e12731. [Google Scholar] [CrossRef] [PubMed]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- Kasubuchi, M.; Hasegawa, S.; Hiramatsu, T.; Ichimura, A.; Kimura, I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients 2015, 7, 2839–2849. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011, 13, 517–526. [Google Scholar] [CrossRef]
- Krautkramer, K.A.; Kreznar, J.H.; Romano, K.A.; Vivas, E.I.; Barrett-Wilt, G.A.; Rabaglia, M.E.; Keller, M.P.; Attie, A.D.; Rey, F.E.; Denu, J.M. Diet-microbiota interactions mediate global epigenetic programming in multiple host tissues. Mol. Cell 2016, 64, 982–992. [Google Scholar] [CrossRef]
- Kimura, I.; Inoue, D.; Maeda, T.; Hara, T.; Ichimura, A.; Miyauchi, S.; Kobayashi, M.; Hirasawa, A.; Tsujimoto, G. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41). Proc. Natl. Acad. Sci. USA 2011, 108, 8030–8035. [Google Scholar] [CrossRef]
- Fellows, R.; Denizot, J.; Stellato, C.; Cuomo, A.; Jain, P.; Stoyanova, E.; Balázsi, S.; Hajnády, Z.; Liebert, A.; Kazakevych, J. Microbiota derived short chain fatty acids promote histone crotonylation in the colon through histone deacetylases. Nat. Commun. 2018, 9, 105. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.L.; Rosenberger, T.A. Acetate supplementation increases brain histone acetylation and inhibits histone deacetylase activity and expression. Mol. Cell. Biochem. 2011, 352, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Soliman, M.L.; Smith, M.D.; Houdek, H.M.; Rosenberger, T.A. Acetate supplementation modulates brain histone acetylation and decreases interleukin-1β expression in a rat model of neuroinflammation. J. Neuroinflammation 2012, 9, 51. [Google Scholar] [CrossRef]
- Guo, W.; Zhang, Z.; Li, L.; Liang, X.; Wu, Y.; Wang, X.; Ma, H.; Cheng, J.; Zhang, A.; Tang, P. Gut microbiota induces DNA methylation via SCFAs predisposing obesity-prone individuals to diabetes. Pharmacol. Res. 2022, 182, 106355. [Google Scholar] [CrossRef]
- Resende, W.R.; Valvassori, S.S.; Reus, G.Z.; Varela, R.B.; Arent, C.O.; Ribeiro, K.F.; Bavaresco, D.V.; Andersen, M.L.; Zugno, A.I.; Quevedo, J. Effects of sodium butyrate in animal models of mania and depression: Implications as a new mood stabilizer. Behav. Pharmacol. 2013, 24, 569–579. [Google Scholar] [CrossRef]
- Yamawaki, Y.; Yoshioka, N.; Nozaki, K.; Ito, H.; Oda, K.; Harada, K.; Shirawachi, S.; Asano, S.; Aizawa, H.; Yamawaki, S. Sodium butyrate abolishes lipopolysaccharide-induced depression-like behaviors and hippocampal microglial activation in mice. Brain Res. 2018, 1680, 13–38. [Google Scholar] [CrossRef]
- Valvassori, S.S.; Resende, W.R.; Budni, J.; Dal-Pont, G.C.; Bavaresco, D.V.; Reus, G.Z.; Carvalho, A.F.; Goncalves, C.L.; Furlanetto, C.B.; Streck, E.L. Sodium butyrate, a histone deacetylase inhibitor, reverses behavioral and mitochondrial alterations in animal models of depression induced by early-or late-life stress. Curr. Neurovascular Res. 2015, 12, 312–320. [Google Scholar] [CrossRef]
- Qiu, J.; Liu, R.; Ma, Y.; Li, Y.; Chen, Z.; He, H.; Chen, J.; Tong, L.; Huang, C.; You, Q. Lipopolysaccharide-induced depression-like behaviors is ameliorated by sodium butyrate via inhibiting neuroinflammation and oxido-nitrosative stress. Pharmacology 2020, 105, 550–560. [Google Scholar] [CrossRef]
- Han, A.; Sung, Y.-B.; Chung, S.-Y.; Kwon, M.-S. Possible additional antidepressant-like mechanism of sodium butyrate: Targeting the hippocampus. Neuropharmacology 2014, 81, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wang, F.; Hong, G.; Pang, M.; Xu, H.; Li, H.; Tian, F.; Fang, R.; Yao, Y.; Liu, J. Antidepressant-like effects of sodium butyrate and its possible mechanisms of action in mice exposed to chronic unpredictable mild stress. Neurosci. Lett. 2016, 618, 159–166. [Google Scholar] [CrossRef]
- Cristiano, C.; Cuozzo, M.; Coretti, L.; Liguori, F.; Cimmino, F.; Turco, L.; Avagliano, C.; Aviello, G.; Mollica, M.; Lembo, F. Oral sodium butyrate supplementation ameliorates paclitaxel-induced behavioral and intestinal dysfunction. Biomed. Pharmacother. 2022, 153, 113528. [Google Scholar] [CrossRef]
- Jangra, A.; Sriram, C.S.; Dwivedi, S.; Gurjar, S.S.; Hussain, M.I.; Borah, P.; Lahkar, M. Sodium phenylbutyrate and edaravone abrogate chronic restraint stress-induced behavioral deficits: Implication of oxido-nitrosative, endoplasmic reticulum stress cascade, and neuroinflammation. Cell. Mol. Neurobiol. 2017, 37, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hou, L.; Wang, C.; Jia, X.; Qin, X.; Wu, C. Short term intrarectal administration of sodium propionate induces antidepressant-like effects in rats exposed to chronic unpredictable mild stress. Front. Psychiatry 2018, 9, 454. [Google Scholar] [CrossRef]
- Huang, W.; Hu, W.; Cai, L.; Zeng, G.; Fang, W.; Dai, X.; Ye, Q.; Chen, X.; Zhang, J. Acetate supplementation produces antidepressant-like effect via enhanced histone acetylation. J. Affect. Disord. 2021, 281, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Wolugbom, J.A., Jr.; Areloegbe, S.E.; Olaniyi, K.S. Protective Role of Acetate Against Depressive-Like Behaviour Associated with Letrozole-Induced PCOS Rat Model: Involvement of HDAC2 and DNA Methylation. Mol. Neurobiol. 2023, 60, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Eiseman, B.; Silen, W.; GS, B.; AJ, K. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery 1958, 44, 854–859. [Google Scholar] [PubMed]
- Bakker, G.J.; Nieuwdorp, M. Fecal microbiota transplantation: Therapeutic potential for a multitude of diseases beyond Clostridium difficile. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Wortelboer, K.; Nieuwdorp, M.; Herrema, H. Fecal microbiota transplantation beyond Clostridioides difficile infections. EBioMedicine 2019, 44, 716–729. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Guo, Q.; Wen, Z.; Tan, S.; Chen, J.; Lin, L.; Chen, P.; He, J.; Wen, J.; Chen, Y. The multiple effects of fecal microbiota transplantation on diarrhea-predominant irritable bowel syndrome (IBS-D) patients with anxiety and depression behaviors. Microb. Cell Factories 2021, 20, 233. [Google Scholar] [CrossRef]
- Knudsen, J.K.; Michaelsen, T.Y.; Bundgaard-Nielsen, C.; Nielsen, R.E.; Hjerrild, S.; Leutscher, P.; Wegener, G.; Sørensen, S. Faecal microbiota transplantation from patients with depression or healthy individuals into rats modulates mood-related behaviour. Sci. Rep. 2021, 11, 21869. [Google Scholar] [CrossRef]
- Kelly, J.R.; Borre, Y.; O’Brien, C.; Patterson, E.; El Aidy, S.; Deane, J.; Kennedy, P.J.; Beers, S.; Scott, K.; Moloney, G. Transferring the blues: Depression-associated gut microbiota induces neurobehavioural changes in the rat. J. Psychiatr. Res. 2016, 82, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Churchward, M.A.; Michaud, E.R.; Mullish, B.H.; Blanco, J.M.; Perez, I.G.; Marchesi, J.R.; Xu, H.; Kao, D.; Todd, K.G. Short-chain fatty and carboxylic acid changes associated with fecal microbiota transplant communally influence microglial inflammation. Heliyon 2023, 9, e16908. [Google Scholar] [CrossRef] [PubMed]
- Doll, J.P.; Vázquez-Castellanos, J.F.; Schaub, A.-C.; Schweinfurth, N.; Kettelhack, C.; Schneider, E.; Yamanbaeva, G.; Mählmann, L.; Brand, S.; Beglinger, C. Fecal microbiota transplantation (FMT) as an adjunctive therapy for depression—Case report. Front. Psychiatry 2022, 13, 815422. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.; Xie, R.; Lin, L.; Jiang, J.; Du, L.; Zeng, X.; Li, G.; Wang, C.; Qiao, Y. Fecal microbiota transplantation ameliorates gut microbiota imbalance and intestinal barrier damage in rats with stress-induced depressive-like behavior. Eur. J. Neurosci. 2021, 53, 3598–3611. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Bhawal, S.; Kapila, S.; Kapila, R. Probiotic lactobacilli mediate their immunoregulatory functions in intestinal cells via modulation of H3 histone acetylation. J. Appl. Microbiol. 2023, 134, lxac045. [Google Scholar] [CrossRef]
- Messaoudi, M.; Lalonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.-F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef]
- Xu, N.; Fan, W.; Zhou, X.; Liu, Y.; Ma, P.; Qi, S.; Gu, B. Probiotics decrease depressive behaviors induced by constipation via activating the AKT signaling pathway. Metab. Brain Dis. 2018, 33, 1625–1633. [Google Scholar] [CrossRef]
- Guo, H.; Liu, X.; Chen, T.; Wang, X.; Zhang, X. Akkermansia muciniphila Improves Depressive-like Symptoms by Modulating the Level of 5-HT Neurotransmitters in the Gut and Brain of Mice. Mol. Neurobiol. 2023, in press. [Google Scholar] [CrossRef]
- Wang, J.; Ji, H.; Wang, S.; Liu, H.; Zhang, W.; Zhang, D.; Wang, Y. Probiotic Lactobacillus plantarum promotes intestinal barrier function by strengthening the epithelium and modulating gut microbiota. Front. Microbiol. 2018, 9, 1953. [Google Scholar] [CrossRef]
- Sun, J.; Wang, F.; Hu, X.; Yang, C.; Xu, H.; Yao, Y.; Liu, J. Clostridium butyricum attenuates chronic unpredictable mild stress-induced depressive-like behavior in mice via the gut-brain axis. J. Agric. Food Chem. 2018, 66, 8415–8421. [Google Scholar] [CrossRef]
- Chen, T.; Wang, R.; Duan, Z.; Yuan, X.; Ding, Y.; Feng, Z.; Bu, F.; Liu, L.; Wang, Q.; Zhou, J. Akkermansia muciniphila protects against psychological disorder-induced gut microbiota-mediated colonic mucosal barrier damage and aggravation of colitis. Front. Cell. Infect. Microbiol. 2021, 11, 723856. [Google Scholar] [CrossRef] [PubMed]
- Tian, P.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Bifidobacterium with the role of 5-hydroxytryptophan synthesis regulation alleviates the symptom of depression and related microbiota dysbiosis. J. Nutr. Biochem. 2019, 66, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Tian, P.; Zou, R.; Song, L.; Zhang, X.; Jiang, B.; Wang, G.; Lee, Y.-k.; Zhao, J.; Zhang, H.; Chen, W. Ingestion of Bifidobacterium longum subspecies infantis strain CCFM687 regulated emotional behavior and the central BDNF pathway in chronic stress-induced depressive mice through reshaping the gut microbiota. Food Funct. 2019, 10, 7588–7598. [Google Scholar] [CrossRef]
- Dandekar, M.P.; Palepu, M.S.K.; Satti, S.; Jaiswal, Y.; Singh, A.A.; Dash, S.P.; Gajula, S.N.R.; Sonti, R. Multi-strain probiotic formulation reverses maternal separation and chronic unpredictable mild stress-generated anxiety-and depression-like phenotypes by modulating gut microbiome–brain activity in rats. ACS Chem. Neurosci. 2022, 13, 1948–1965. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Sun, Q.; Zheng, H.; Zhang, Y.; Wang, Y.; Liu, S.; Duan, L. Roseburia hominis Alleviates Neuroinflammation via Short-Chain Fatty Acids through Histone Deacetylase Inhibition. Mol. Nutr. Food Res. 2022, 66, 2200164. [Google Scholar] [CrossRef]
- Tian, P.; Zou, R.; Wang, L.; Chen, Y.; Qian, X.; Zhao, J.; Zhang, H.; Qian, L.; Wang, Q.; Wang, G. Multi-Probiotics ameliorate Major depressive disorder and accompanying gastrointestinal syndromes via serotonergic system regulation. J. Adv. Res. 2023, 45, 117–125. [Google Scholar] [CrossRef]
- Tan, J.; Li, X.; Zhu, Y.; Sullivan, M.A.; Deng, B.; Zhai, X.; Lu, Y. Antidepressant shugan jieyu capsule alters gut microbiota and intestinal microbiome function in rats with chronic unpredictable mild stress-induced depression. Front. Pharmacol. 2022, 13, 828595. [Google Scholar] [CrossRef]
- Lin, S.; Li, Q.; Jiang, S.; Xu, Z.; Jiang, Y.; Liu, L.; Jiang, J.; Tong, Y.; Wang, P. Crocetin ameliorates chronic restraint stress-induced depression-like behaviors in mice by regulating MEK/ERK pathways and gut microbiota. J. Ethnopharmacol. 2021, 268, 113608. [Google Scholar] [CrossRef]
- Liu, J.; Fang, Y.; Cui, L.; Wang, Z.; Luo, Y.; Gao, C.; Ge, W.; Huang, T.; Wen, J.; Zhou, T. Butyrate emerges as a crucial effector of Zhi-Zi-Chi decoctions to ameliorate depression via multiple pathways of brain-gut axis. Biomed. Pharmacother. 2022, 149, 112861. [Google Scholar] [CrossRef]
- Xiong, L.; Mao, M.; Shu, Q. A preliminary study on the diversity of butyrate-producing bacteria in response to the treatment of depression with Xiaoyaosan. Lett. Appl. Microbiol. 2022, 75, 844–856. [Google Scholar] [CrossRef]
- Xiong, L.; Wu, Y.; Shu, Q.; Xiong, W. The pharmacological mechanism of Xiaoyaosan polysaccharide reveals improvement of CUMS-induced depression-like behavior by carbon source-triggered butyrate-producing bacteria. J. Appl. Microbiol. 2023, 134, lxad052. [Google Scholar] [CrossRef] [PubMed]
- Donoso, F.; Egerton, S.; Bastiaanssen, T.F.; Fitzgerald, P.; Gite, S.; Fouhy, F.; Ross, R.P.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Polyphenols selectively reverse early-life stress-induced behavioural, neurochemical and microbiota changes in the rat. Psychoneuroendocrinology 2020, 116, 104673. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Nian, T.; Liao, Z.; Xiao, F.; Wu, B.; Bi, K.; He, B.; Jia, Y. Antidepressant effects of a polysaccharide from okra (Abelmoschus esculentus (L.) Moench) by anti-inflammation and rebalancing the gut microbiota. Int. J. Biol. Macromol. 2020, 144, 427–440. [Google Scholar] [CrossRef]
- Qu, W.; Liu, S.; Zhang, W.; Zhu, H.; Tao, Q.; Wang, H.; Yan, H. Impact of traditional Chinese medicine treatment on chronic unpredictable mild stress-induced depression-like behaviors: Intestinal microbiota and gut microbiome function. Food Funct. 2019, 10, 5886–5897. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Kan, Q.; Zhao, L.; Ye, G.; He, X.; Tang, H.; Shi, F.; Zou, Y.; Liang, X.; Song, X. Prophylactic effect of Tongxieyaofang polysaccharide on depressive behavior in adolescent male mice with chronic unpredictable stress through the microbiome-gut-brain axis. Biomed. Pharmacother. 2023, 161, 114525. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Li, A.; Huang, T.; Lai, J.; Li, J.; Sublette, M.E.; Lu, H.; Lu, Q.; Du, Y.; Hu, Z. Gut microbiota changes in patients with bipolar depression. Adv. Sci. 2019, 6, 1900752. [Google Scholar] [CrossRef] [PubMed]
- Cussotto, S.; Strain, C.R.; Fouhy, F.; Strain, R.G.; Peterson, V.L.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Differential effects of psychotropic drugs on microbiome composition and gastrointestinal function. Psychopharmacology 2019, 236, 1671–1685. [Google Scholar] [CrossRef]
- Duan, J.; Huang, Y.; Tan, X.; Chai, T.; Wu, J.; Zhang, H.; Li, Y.; Hu, X.; Zheng, P.; Ji, P. Characterization of gut microbiome in mice model of depression with divergent response to escitalopram treatment. Transl. Psychiatry 2021, 11, 303. [Google Scholar] [CrossRef]
- Zhang, W.; Qu, W.; Wang, H.; Yan, H. Antidepressants fluoxetine and amitriptyline induce alterations in intestinal microbiota and gut microbiome function in rats exposed to chronic unpredictable mild stress. Transl. Psychiatry 2021, 11, 131. [Google Scholar] [CrossRef]
- Getachew, B.; Aubee, J.I.; Schottenfeld, R.S.; Csoka, A.B.; Thompson, K.M.; Tizabi, Y. Ketamine interactions with gut-microbiota in rats: Relevance to its antidepressant and anti-inflammatory properties. BMC Microbiol. 2018, 18, 222. [Google Scholar] [CrossRef]
- Ponziani, F.R.; Scaldaferri, F.; Petito, V.; Paroni Sterbini, F.; Pecere, S.; Lopetuso, L.R.; Palladini, A.; Gerardi, V.; Masucci, L.; Pompili, M. The role of antibiotics in gut microbiota modulation: The eubiotic effects of rifaximin. Dig. Dis. 2016, 34, 269–278. [Google Scholar] [CrossRef]
- Meng, C.; Feng, S.; Hao, Z.; Dong, C.; Liu, H. Antibiotics exposure attenuates chronic unpredictable mild stress-induced anxiety-like and depression-like behavior. Psychoneuroendocrinology 2022, 136, 105620. [Google Scholar] [CrossRef] [PubMed]
- Schmidtner, A.K.; Slattery, D.A.; Gläsner, J.; Hiergeist, A.; Gryksa, K.; Malik, V.A.; Hellmann-Regen, J.; Heuser, I.; Baghai, T.C.; Gessner, A. Minocycline alters behavior, microglia and the gut microbiome in a trait-anxiety-dependent manner. Transl. Psychiatry 2019, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Luo, L.; Sun, T.; Yang, L.; Cheng, L.-F.; Wang, Y.; Liu, Q.-Q.; Liu, A.; Liu, H.-Y.; Zhao, M.-G. Chronic minocycline treatment exerts antidepressant effect, inhibits neuroinflammation, and modulates gut microbiota in mice. Psychopharmacology 2020, 237, 3201–3213. [Google Scholar] [CrossRef]
- Li, H.; Xiang, Y.; Zhu, Z.; Wang, W.; Jiang, Z.; Zhao, M.; Cheng, S.; Pan, F.; Liu, D.; Ho, R.C. Rifaximin-mediated gut microbiota regulation modulates the function of microglia and protects against CUMS-induced depression-like behaviors in adolescent rat. J. Neuroinflammation 2021, 18, 254. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Apostolopoulos, V. The effects of vitamin B in depression. Curr. Med. Chem. 2016, 23, 4317–4337. [Google Scholar] [CrossRef] [PubMed]
- Uebanso, T.; Shimohata, T.; Mawatari, K.; Takahashi, A. Functional roles of B-vitamins in the gut and gut microbiome. Mol. Nutr. Food Res. 2020, 64, 2000426. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The effects of vitamin B on the immune/cytokine network and their involvement in depression. Maturitas 2017, 96, 58–71. [Google Scholar] [CrossRef]
- Xu, P.; Pang, D.; Zhou, J.; Li, S.; Chen, D.; Yu, B. Behavioral changes and brain epigenetic alterations induced by maternal deficiencies of B vitamins in a mouse model. Psychopharmacology 2021, 238, 1213–1222. [Google Scholar] [CrossRef]
- Blad, C.C.; Tang, C.; Offermanns, S. G protein-coupled receptors for energy metabolites as new therapeutic targets. Nat. Rev. Drug Discov. 2012, 11, 603–619. [Google Scholar] [CrossRef]
- Singh, N.; Gurav, A.; Sivaprakasam, S.; Brady, E.; Padia, R.; Shi, H.; Thangaraju, M.; Prasad, P.D.; Manicassamy, S.; Munn, D.H. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 2014, 40, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Wadie, W.; Mohamed, S.S.; Abd El-Haleim, E.A.; Khayyal, M.T. Niacin modulates depressive-like behavior in experimental colitis through GPR109A-dependent mechanisms. Life Sci. 2023, 330, 122004. [Google Scholar] [CrossRef] [PubMed]
- Zhen, R.; Liu, C.; Wei, C.; Luo, Y.; Hu, X.; Liu, G.; Yi, H.; Huang, Y. Effect of different dosages of sodium butyrate and niacin on growth, faecal microbiota and Vitamin B metabolism in weaned piglets. J. Appl. Microbiol. 2022, 132, 4466–4475. [Google Scholar] [CrossRef]
- Trautmann, C.; Bock, A.; Urbach, A.; Hübner, C.A.; Engmann, O. Acute vitamin B12 supplementation evokes antidepressant response and alters Ntrk-2. Neuropharmacology 2020, 171, 108112. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Sinha, J.K.; Khandelwal, N.; Chakravarty, S.; Kumar, A.; Raghunath, M. Increased stress and altered expression of histone modifying enzymes in brain are associated with aberrant behaviour in vitamin B12 deficient female mice. Nutr. Neurosci. 2020, 23, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Bai, Y.; Yang, Y.; Wu, X.; Li, R. A comparison of production performance, egg quality, and cecal microbiota in laying hens receiving graded levels of vitamin B12. Front. Vet. Sci. 2021, 8, 712183. [Google Scholar] [CrossRef]
- Singh, P.; Rawat, A.; Alwakeel, M.; Sharif, E.; Al Khodor, S. The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals. Sci. Rep. 2020, 10, 21641. [Google Scholar] [CrossRef] [PubMed]
- Gominak, S. Vitamin D deficiency changes the intestinal microbiome reducing B vitamin production in the gut. The resulting lack of pantothenic acid adversely affects the immune system, producing a “pro-inflammatory” state associated with atherosclerosis and autoimmunity. Med. Hypotheses 2016, 94, 103–107. [Google Scholar]
- Tuo, L.-J.; Song, X.-Y.; Zhu, Y.-Y.; He, H.-N.; Song, Y.-P.; Chen, D.-Z.; Zheng, X.-M.; Zhang, H.; Xu, D.-X. Gestational folic acid supplement prevents vitamin D deficiency-induced depression-like behavior by reversing cortical DNA hypomethylation in adult offspring. J. Steroid Biochem. Mol. Biol. 2023, 231, 106313. [Google Scholar] [CrossRef]
- McCann, A.; Jeffery, I.B.; Ouliass, B.; Ferland, G.; Fu, X.; Booth, S.L.; Tran, T.T.; O’Toole, P.W.; O’Connor, E.M. Exploratory analysis of covariation of microbiota-derived vitamin K and cognition in older adults. Am. J. Clin. Nutr. 2019, 110, 1404–1415. [Google Scholar] [CrossRef]
- Kiely, A.; Ferland, G.; Ouliass, B.; O’Toole, P.; Purtill, H.; O’Connor, E. Vitamin K status and inflammation are associated with cognition in older Irish adults. Nutr. Neurosci. 2020, 23, 591–599. [Google Scholar] [CrossRef] [PubMed]
- McCoubrey, L.E.; Elbadawi, M.; Basit, A.W. Current clinical translation of microbiome medicines. Trends Pharmacol. Sci. 2022, 43, 281–292. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Palleja, A.; Mikkelsen, K.H.; Forslund, S.K.; Kashani, A.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Liang, S.; Feng, Q.; Zhang, C. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nat. Microbiol. 2018, 3, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Browne, H.P.; Forster, S.C.; Anonye, B.O.; Kumar, N.; Neville, B.A.; Stares, M.D.; Goulding, D.; Lawley, T.D. Culturing of ‘unculturable’ human microbiota reveals novel taxa and extensive sporulation. Nature 2016, 533, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Goodman, A.L.; Kallstrom, G.; Faith, J.J.; Reyes, A.; Moore, A.; Dantas, G.; Gordon, J.I. Extensive personal human gut microbiota culture collections characterized and manipulated in gnotobiotic mice. Proc. Natl. Acad. Sci. USA 2011, 108, 6252–6257. [Google Scholar] [CrossRef] [PubMed]
- Park, D.I.; Hisamatsu, T.; Chen, M.; Ng, S.C.; Ooi, C.J.; Wei, S.C.; Banerjee, R.; Hilmi, I.N.; Jeen, Y.T.; Han, D.S. Asian Organization for Crohn’s and Colitis and Asia Pacific Association of Gastroenterology consensus on tuberculosis infection in patients with inflammatory bowel disease receiving anti-tumor necrosis factor treatment. Part 2: Management. J. Gastroenterol. Hepatol. 2018, 33, 30–36. [Google Scholar] [CrossRef]
- Deschasaux, M.; Bouter, K.E.; Prodan, A.; Levin, E.; Groen, A.K.; Herrema, H.; Tremaroli, V.; Bakker, G.J.; Attaye, I.; Pinto-Sietsma, S.-J. Depicting the composition of gut microbiota in a population with varied ethnic origins but shared geography. Nat. Med. 2018, 24, 1526–1531. [Google Scholar] [CrossRef]
- Serrano, M.G.; Parikh, H.I.; Brooks, J.P.; Edwards, D.J.; Arodz, T.J.; Edupuganti, L.; Huang, B.; Girerd, P.H.; Bokhari, Y.A.; Bradley, S.P. Racioethnic diversity in the dynamics of the vaginal microbiome during pregnancy. Nat. Med. 2019, 25, 1001–1011. [Google Scholar] [CrossRef]
- Fatt, C.R.C.; Trivedi, M.H. Microbes and mental health: Translating preclinical findings to the clinic. Neuropsychopharmacology 2023, 49, 345–346. [Google Scholar] [CrossRef]
- Lynch, S.V.; Ng, S.C.; Shanahan, F.; Tilg, H. Translating the gut microbiome: Ready for the clinic? Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wang, H.; Zhang, H.; Chen, X.; Zhang, Y.; Wu, J.; Zhao, L.; Wang, D.; Pu, J.; Ji, P. Toward a Deeper Understanding of Gut Microbiome in Depression: The Promise of Clinical Applicability. Adv. Sci. 2022, 9, 2203707. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.C.; Vuong, H.E.; Luna, C.D.; Pronovost, G.N.; Aleksandrova, A.A.; Riley, N.G.; Vavilina, A.; McGinn, J.; Rendon, T.; Forrest, L.R. Intestinal serotonin and fluoxetine exposure modulate bacterial colonization in the gut. Nat. Microbiol. 2019, 4, 2064–2073. [Google Scholar] [CrossRef] [PubMed]
- Ianiro, G.; Tilg, H.; Gasbarrini, A. Antibiotics as deep modulators of gut microbiota: Between good and evil. Gut 2016, 65, 1906–1915. [Google Scholar] [CrossRef]
| Type of the Study | Key Findings | Epigenetic Alteration | Ref. |
|---|---|---|---|
| Experimental study in mice after maternal glyphosate exposure | DNA hypermethylation of several genes and increased abundance of Alistipes and Blautia (bacteria involved in tryptophan metabolism) and decreased abundance of Akkermansia in offspring after maternal glyphosate exposure | DNA methylation | [74] |
| Clinical study in obese patients with Polycystic ovary syndrome (POCS) | Association between reduced FKBP5 DNA methylation and stress in obese PCOS patients, particularly with higher depression score; reduced bacterial diversity; higher abundance of Bacteroides and Megamonas; reduced abundance of Bacteroidetes and Proteobacteria | DNA methylation | [75] |
| Clinical study in individuals with low self-esteem, an indicator of depressive | Differences in the immune-epigenetic-gut microbiome axis and DNA methylation at immune-metabolic genes in monocytes and deficiencies in regulatory activity of adiponectin and, thereby, downstream targets of inflammation and gut dysbiosis | DNA methylation | [76] |
| Clinical study, depressive symptoms in Parkinson’s Disease (PD) | Reducing counts of genera Roseburia, Romboutsia, and Prevotella relevant to depressive symptoms in PD patients and lower fecal butyrate levels association with DNA methylation alteration in leucocytes and neurons | DNA methylation/Histone acetylation | [77] |
| Experimental study in mice after subchronic and chronic exposure to glyphosate-based herbicide | Decreasing abundance of butyrate-producing bacteria (Firmicutes and Lactobacillus) and other bacteria such as Corynebacterium and Bacteroidetes and, thereby, increasing anxiety and depressive behaviors | Histone acetylation | [78] |
| Transplanting the gut microbiota from patients with alcohol use disorder to mice | Significant alterations in the microbiome composition and lower hepatic synthesis of β-hydroxybutyrate (BHB) and hence increasing depressive-disorders-like behavior | Histone acetylation | [79] |
| Experimental study in mice, chronic social defeat | Reducing the frequency of G-protein-coupled receptors owing to significant reductions in the diversity and abundances of numerous bacterial genera involved in the production of butyrate and propionate (e.g., Akkermansia spp.) and hence increasing depressive disorders like behaviors | Histone acetylation | [80] |
| Experimental study, gut microbiota-absent mice | Gut microbiota dysbiosis and association between lysine acetylation alterations and mitochondrial dysfunction in the brain (hippocampus) and identifying 986 lysine acetylation sites in 543 proteins relevant to MDD | Lysine acetylation | [81] |
| Clinical study, MDD vs. the control group | Significant differences in Bacteroides and Dialister abundance and expression of six fecal miRNAs (miR-1246, miR-579-3p, miR-1276, miR-1976, miR-3144-3p, miR-4488) in cases vs. controls | microRNAs (miRNAs) | [73] |
| Clinical study in patients with depressive disorders vs. controls | Significant alterations in the composition of fecal microbiomes Involvement of two microbial-regulated lncRNA–miRNA–mRNA ceRNA regulatory networks in depressive disorders-related neurodevelopment in patients | miRNAs | [82] |
| Experimental study in mice examining the effects of gut microbiota on miR-206-3p | Increasing degeneration of mitochondria and synapses in the hippocampus and hence enhancing depressive disorders/anxiety-like behaviors by gut microbiota-induced microRNA-206-3p in mouse brain tissues | miRNAs | [83] |
| Experimental study, LPS-induced depressive-like behavior in mice | Regulating gene expression of Nfatc4 and miR-149 (or miR-7688-5p) in the PFC by gut microbiota and blocking LPS-induced depressive-disorders-like behavior by inhibition of miR-149 using antagomiR-149 | miRNAs | [84] |
| Experimental study in the hippocampus of germ-free mice | Disruption of RNA transcription and post-transcriptional regulation of a lincRNA–miRNA–mRNA network including 12 lincRNAs, six miRNAs, and 47 mRNAs during gut microbiota dysbiosis and hence generating depressive- and anxiety-like behaviors | miRNAs | [85] |
| Clinical study in Chinese patients with depressive disorders | gut microbiome diversity regulates microRNA expression in the brain and hence influences SERPINA5, a microbe-associated gene, and other spatially close genes involved in accelerating or delaying the development of depressive disorders | microRNA | [86] |
| Experiment | SCFAs Use | Key Findings | Ref |
|---|---|---|---|
| Chronic mild stress or maternal deprivation for the induction of depressive-like behaviors in adult Wistar rat | Sodium butyrate | Reversing the depressive-like behaviors following treatment with sodium butyrate | [109] |
| Lipopolysaccharide-induced depressive-disorders-like behaviors in mice | Sodium butyrate | Reducing the LPS-induced depressive state via Iba1 hippocampal expression changes and microglia activation by HDAC inhibitors | [110] |
| Maternal deprivation or chronic mild stress in adult Wistar rat | Sodium butyrate | Reversing mitochondrial alterations in the striatum of rats and depressive-like behaviors | [111] |
| Lipopolysaccharide-induced depressive-disorders-like behaviors in C57BL6/J mice | Sodium butyrate | Suppressing LPS-induced enhancement of pro-inflammatory cytokines like tumor necrosis factor-α, IL-1β, and IL-6 in the prefrontal cortex and hippocampus and inhibiting oxido-nitrosative stress | [112] |
| Induced depressive-disorders-like behaviors using chronic restraint stress in mice | Sodium butyrate | Normalizing acetylation of histone H3, HDAC2, and BDNF expression levels reduced by stress in the hippocampus | [113] |
| Chronic unpredictable mild stress (CUMS)-induced depressive-disorders-like behaviors in mice | Sodium butyrate | Restoring CUMS-induced BBB impairments by enhancing the expression of Occludin and ZO-1 proteins in the hippocampus, elevating serotonin concentration, and BDNF level | [114] |
| Forced swim and tail suspension induced immobility and depressive-disorders-like behaviors in rats | Sodium butyrate | Antidepressant-like effect of sodium butyrate through increases in histone H4 acetylation at the promoter of the transthyretin gene | [61] |
| Depressive- and anxiety-like behaviors induced by paclitaxel (PTX) in mice | Sodium butyrate | Alleviating depressive- and anxiety-like behaviors induced by PTX via restoring PTX-induced altered gut barrier integrity, microbiota composition, and food intake | [115] |
| Chronic restraint stress model in mice | Sodium phenyl butyrate | Reducing depressive-like behaviors by inhibiting oxido-nitrosative stress, neuro-inflammation, endoplasmic reticulum stress cascade, and restoring BDNF | [116] |
| CUMS in adult Sprague–Dawley rats | Sodium propionate | Inducing antidepressant effect by low- dose propionate (2 mg/kg/day) | [27] |
| CUMS in adult Sprague–Dawley rats | Sodium propionate | Inducing antidepressant effects via differential rescue of neurotransmitters in the prefrontal cortex after short-term intrarectal administration of propionate | [117] |
| Chronic social failure stress (CSDS) in mice | Acetate | Improving depressive-disorders-like behaviors by reducing the transcription of HDAC2, HDAC5, HDAC7, HDAC8, and increasing the transcription of HAT and P300, elevating the content of Ac-CoA in the nucleus, and subsequently promoting histone H3 and H4 acetylation | [118] |
| PCOS-associated depressive disorders in female Wistar rats | Acetate | Reducing the expression of HDAC2 and DNA methyltransferase in the prefrontal cortex and hippocampal tissues; inhibiting inflammation and oxidative stress; reducing depressive-like behaviors after acetate administration | [119] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nohesara, S.; Abdolmaleky, H.M.; Zhou, J.-R.; Thiagalingam, S. Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies. Genes 2023, 14, 2217. https://doi.org/10.3390/genes14122217
Nohesara S, Abdolmaleky HM, Zhou J-R, Thiagalingam S. Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies. Genes. 2023; 14(12):2217. https://doi.org/10.3390/genes14122217
Chicago/Turabian StyleNohesara, Shabnam, Hamid Mostafavi Abdolmaleky, Jin-Rong Zhou, and Sam Thiagalingam. 2023. "Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies" Genes 14, no. 12: 2217. https://doi.org/10.3390/genes14122217
APA StyleNohesara, S., Abdolmaleky, H. M., Zhou, J.-R., & Thiagalingam, S. (2023). Microbiota-Induced Epigenetic Alterations in Depressive Disorders Are Targets for Nutritional and Probiotic Therapies. Genes, 14(12), 2217. https://doi.org/10.3390/genes14122217






