Sequence Characterization of Extra-Chromosomal Circular DNA Content in Multiple Blackgrass (Alopecurus myosuroides) Populations
Abstract
1. Introduction
2. Materials and Methods
2.1. Blackgrass Populations, Plant Material, and Genomic DNA Extraction
2.2. eccDNA Sequence Enrichment, Sequencing, and Analysis
3. Results
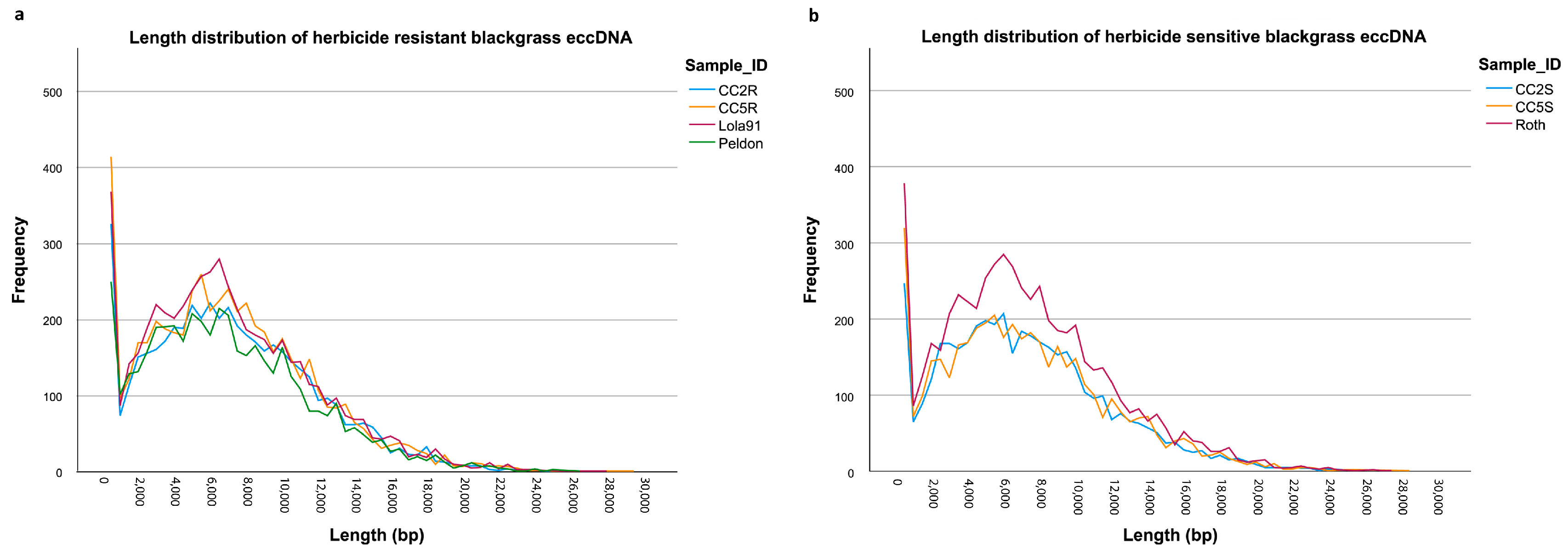
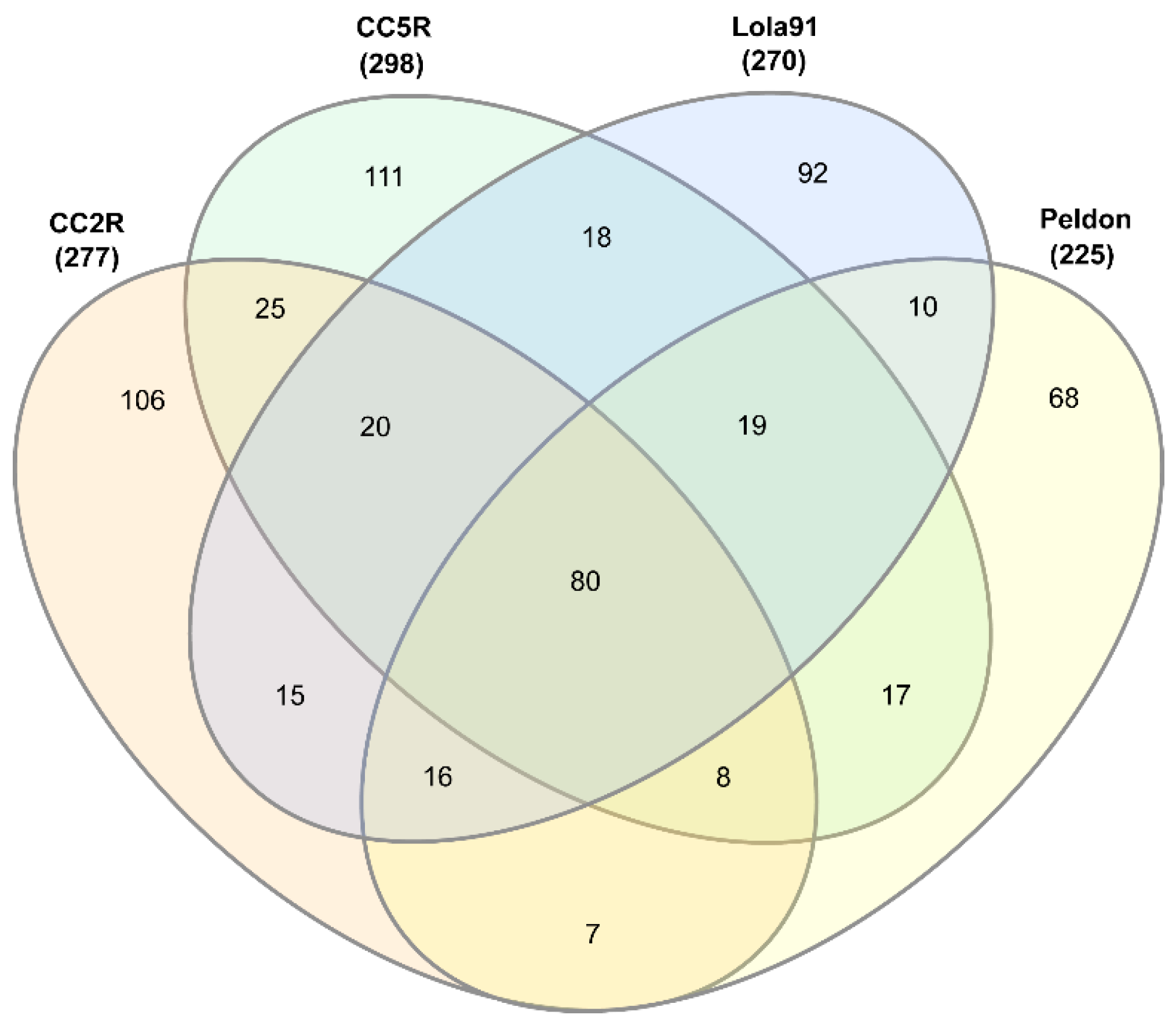
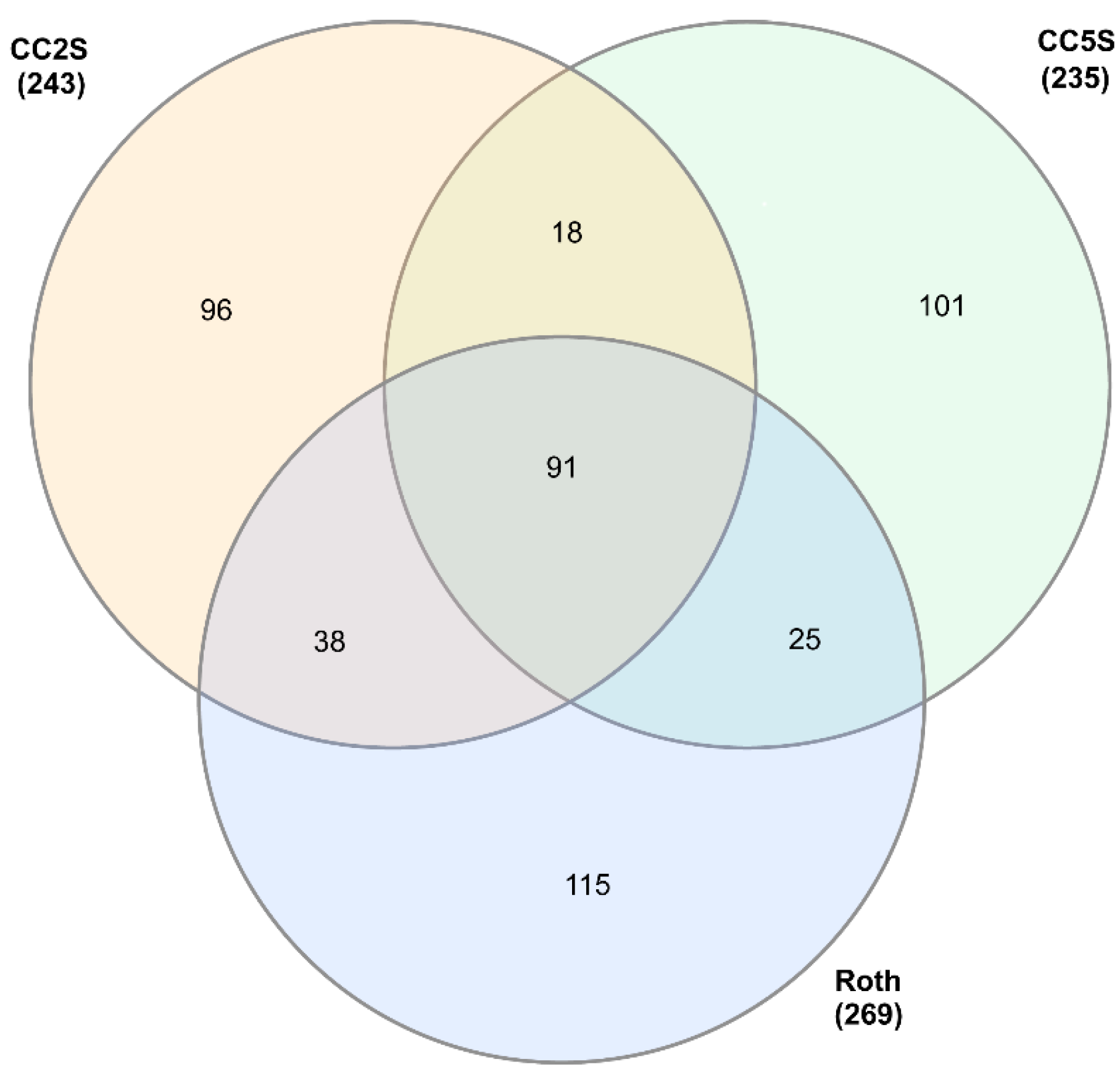
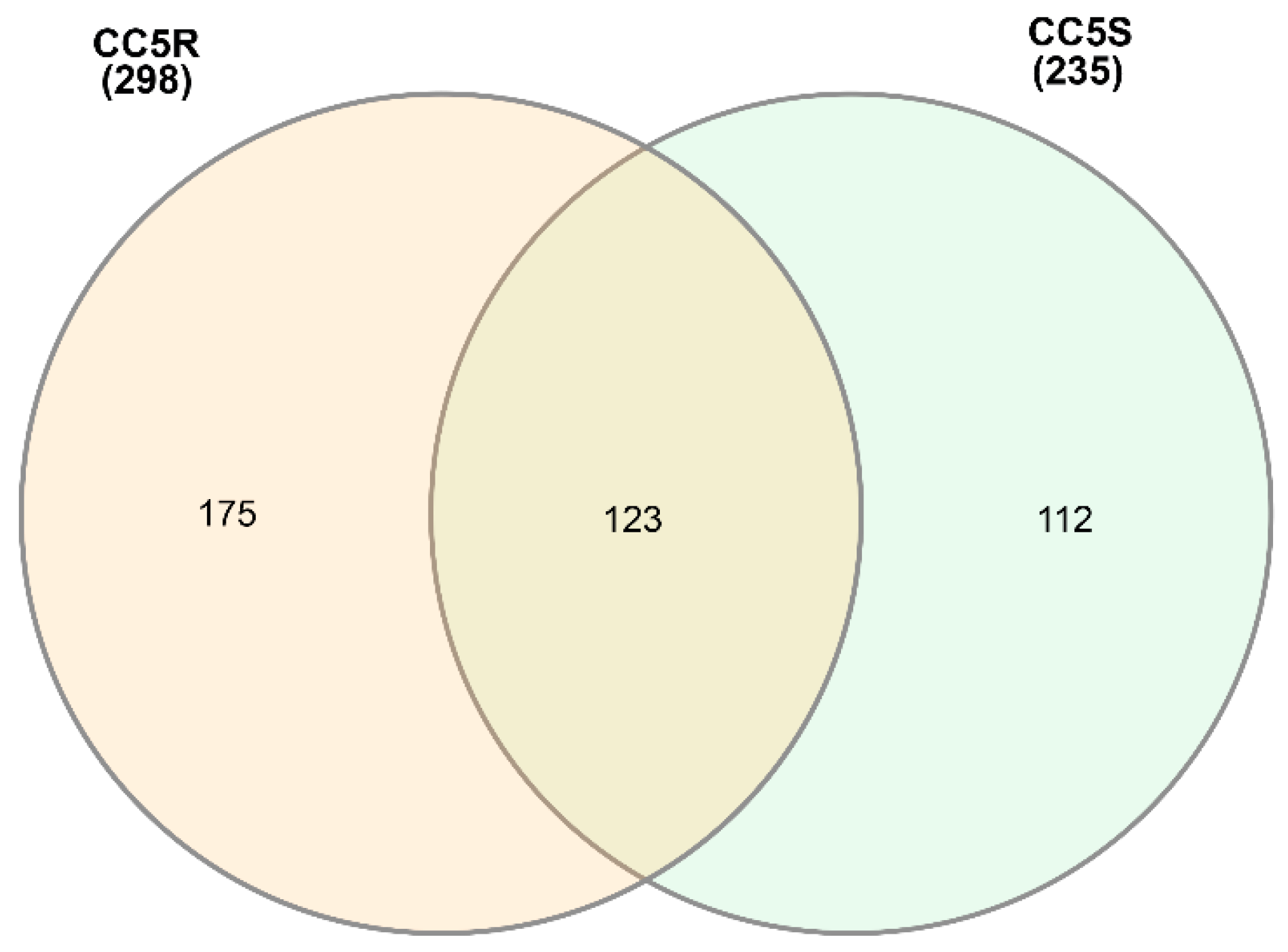
3.1. eccDNA Content and Coding Structure in Multiple Blackgrass Populations
3.2. Coding Content of eccDNAs in Herbicide-Resistant and -Sensitive Blackgrass Populations
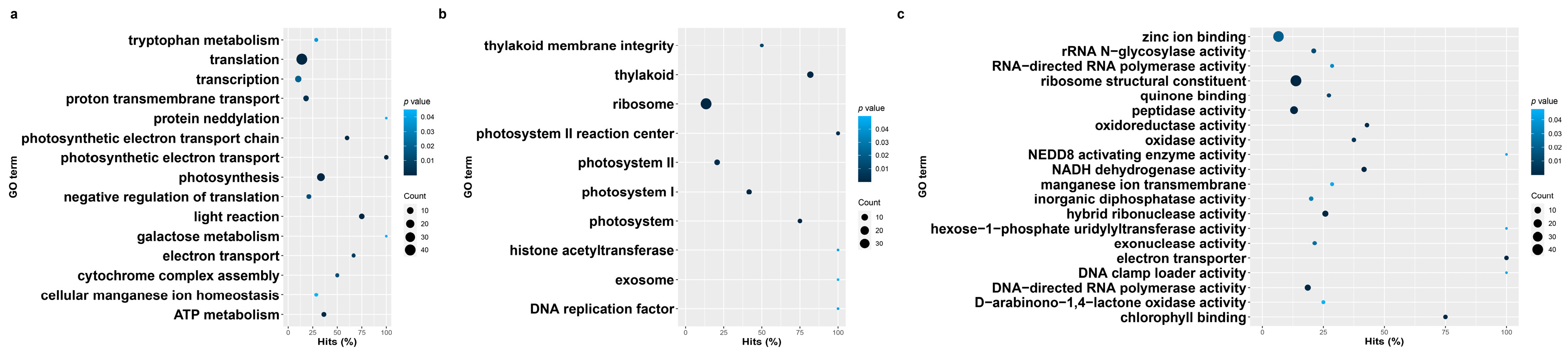
3.3. Gene Ontology Enrichment of Blackgrass eccDNA
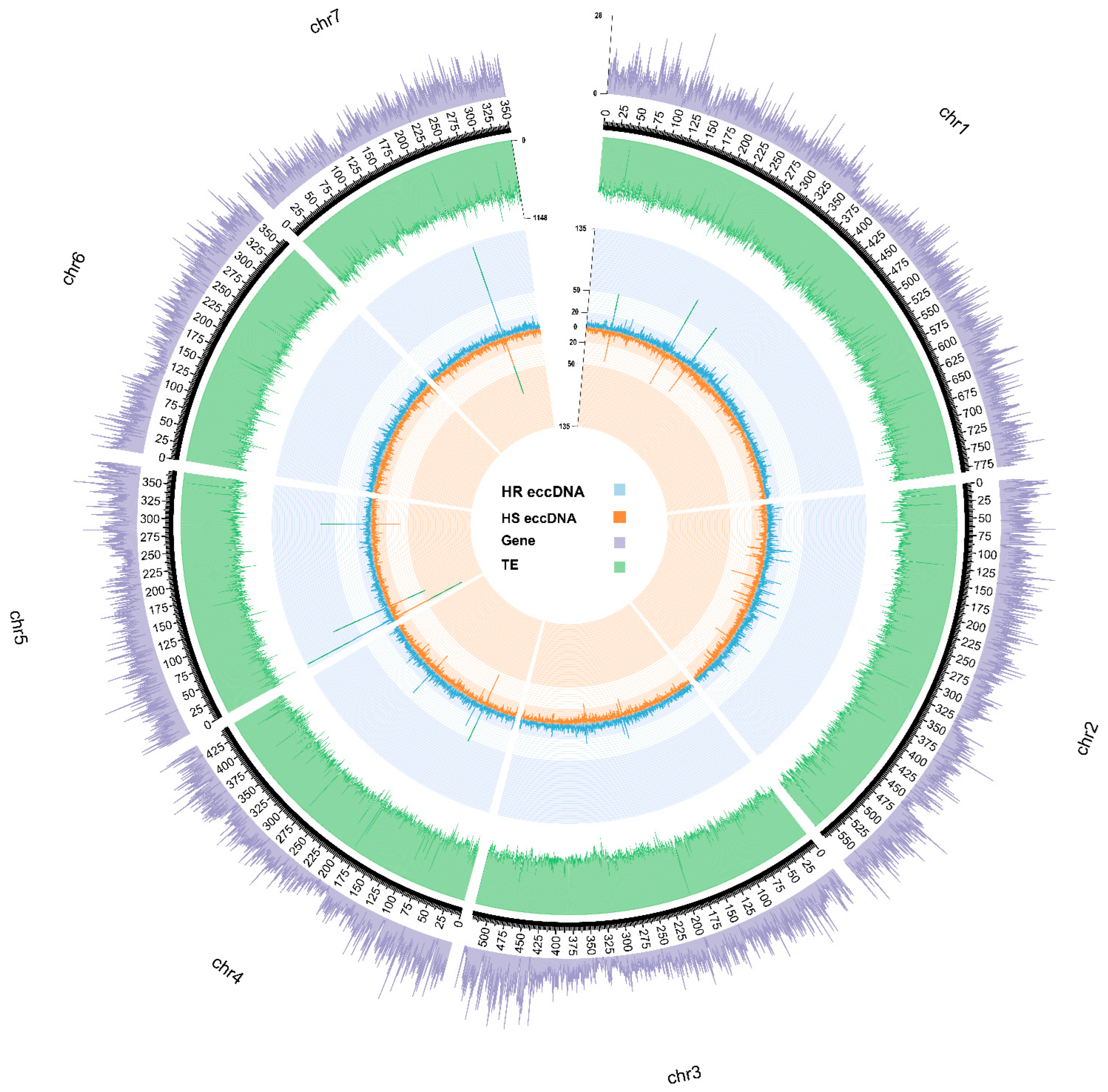
3.4. Genomic Origins of Blackgrass eccDNA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spier Camposano, H.; Molin, W.T.; Saski, C.A. Sequence characterization of eccDNA content in glyphosate sensitive and resistant Palmer amaranth from geographically distant populations. PLoS ONE 2022, 17, e0260906. [Google Scholar] [CrossRef] [PubMed]
- Gaubatz, J.W. Extrachromosomal circular DNAs and genomic sequence plasticity in eukaryotic cells. Mutat. Res. 1990, 237, 271–292. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Menut, S.; Mechali, M. Regulated formation of extrachromosomal circular DNA molecules during development in Xenopus laevis. Mol. Cell. Biol. 1999, 19, 6682–6689. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Yacobi, K.; Segal, D. Extrachromosomal circular DNA of tandemly repeated genomic sequences in Drosophila. Genome Res. 2003, 13, 1133–1145. [Google Scholar] [CrossRef]
- Moller, H.D.; Parsons, L.; Jorgensen, T.S.; Botstein, D.; Regenberg, B. Extrachromosomal circular DNA is common in yeast. Proc. Natl. Acad. Sci. USA 2015, 112, E3114–E3122. [Google Scholar] [CrossRef]
- Peng, H.R.; Mirouze, M.; Bucher, E. Extrachromosomal circular DNA: A neglected nucleic acid molecule in plants. Curr. Opin. Plant Biol. 2022, 69, 102263. [Google Scholar] [CrossRef]
- Hotta, Y.; Bassel, A. Molecular Size and Circularity of DNA in Cells of Mammals and Higher Plants. Proc. Natl. Acad. Sci. USA 1965, 53, 356–362. [Google Scholar] [CrossRef]
- Mansisidor, A.; Molinar, T.; Srivastava, P.; Dartis, D.D.; Delgado, A.P.; Blitzblau, H.G.; Klein, H.; Hochwagen, A. Genomic Copy-Number Loss Is Rescued by Self-Limiting Production of DNA Circles. Mol. Cell 2018, 72, 583–593. [Google Scholar] [CrossRef]
- Paulsen, T.; Kumar, P.; Koseoglu, M.M.; Dutta, A. Discoveries of Extrachromosomal Circles of DNA in Normal and Tumor Cells. Trends Genet. 2018, 34, 270–278. [Google Scholar] [CrossRef]
- Kilzer, J.M.; Stracker, T.; Beitzel, B.; Meek, K.; Weitzman, M.; Bushman, F.D. Roles of host cell factors in circularization of retroviral DNA. Virology 2003, 314, 460–467. [Google Scholar] [CrossRef]
- Garfinkel, D.J.; Stefanisko, K.M.; Nyswaner, K.M.; Moore, S.P.; Oh, J.; Hughes, S.H. Retrotransposon suicide: Formation of Ty1 circles and autointegration via a central DNA flap. J. Virol. 2006, 80, 11920–11934. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Molin, W.T.; Yaguchi, A.; Blenner, M.A.; Saski, C.A. The eccDNA Replicon: A Heritable, Extra-Nuclear Vehicle that Enables Gene Amplification and Glyphosate Resistance in Amaranthus palmeri. Plant Cell 2020, 32, 2132–2140. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, V.; Luebeck, J.; Nguyen, N.D.; Bakhtiari, M.; Turner, K.M.; Schwab, R.; Carter, H.; Mischel, P.S.; Bafna, V. Exploring the landscape of focal amplifications in cancer using AmpliconArchitect. Nat. Commun. 2019, 10, 392. [Google Scholar] [CrossRef]
- Mehta, D.; Cornet, L.; Hirsch-Hoffmann, M.; Zaidi, S.S.; Vanderschuren, H. Full-length sequencing of circular DNA viruses and extrachromosomal circular DNA using CIDER-Seq. Nat. Protoc. 2020, 15, 1673–1689. [Google Scholar] [CrossRef] [PubMed]
- Moller, H.D. Circle-Seq: Isolation and Sequencing of Chromosome-Derived Circular DNA Elements in Cells. Methods Mol. Biol. 2020, 2119, 165–181. [Google Scholar] [PubMed]
- Turner, K.M.; Deshpande, V.; Beyter, D.; Koga, T.; Rusert, J.; Lee, C.; Li, B.; Arden, K.; Ren, B.; Nathanson, D.A.; et al. Extrachromosomal oncogene amplification drives tumour evolution and genetic heterogeneity. Nature 2017, 543, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Turner, K.M.; Nguyen, N.; Raviram, R.; Erb, M.; Santini, J.; Luebeck, J.; Rajkumar, U.; Diao, Y.; Li, B.; et al. Circular ecDNA promotes accessible chromatin and high oncogene expression. Nature 2019, 575, 699–703. [Google Scholar] [CrossRef]
- Shibata, Y.; Kumar, P.; Layer, R.; Willcox, S.; Gagan, J.R.; Griffith, J.D.; Dutta, A. Extrachromosomal microDNAs and chromosomal microdeletions in normal tissues. Science 2012, 336, 82–86. [Google Scholar] [CrossRef]
- Koo, D.H.; Molin, W.T.; Saski, C.A.; Jiang, J.; Putta, K.; Jugulam, M.; Friebe, B.; Gill, B.S. Extrachromosomal circular DNA-based amplification and transmission of herbicide resistance in crop weed Amaranthus palmeri. Proc. Natl. Acad. Sci. USA 2018, 115, 3332–3337. [Google Scholar] [CrossRef]
- Molin, W.T.; Yaguchi, A.; Blenner, M.; Saski, C.A. Autonomous replication sequences from the Amaranthus palmeri eccDNA replicon enable replication in yeast. BMC Res. Notes 2020, 13, 330. [Google Scholar] [CrossRef]
- Cohen, S.; Segal, D. Extrachromosomal circular DNA in eukaryotes: Possible involvement in the plasticity of tandem repeats. Cytogenet. Genome Res. 2009, 124, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.P.; Grant, P.A. The SAGA continues: Expanding the cellular role of a transcriptional co-activator complex. Oncogene 2007, 26, 5329–5340. [Google Scholar] [CrossRef] [PubMed]
- Herrup, K.; Yang, Y. Cell cycle regulation in the postmitotic neuron: Oxymoron or new biology? Nat. Rev. Neurosci. 2007, 8, 368–378. [Google Scholar] [CrossRef]
- Molin, W.T.; Wright, A.A.; Lawton-Rauh, A.; Saski, C.A. The unique genomic landscape surrounding the EPSPS gene in glyphosate resistant Amaranthus palmeri: A repetitive path to resistance. BMC Genom. 2017, 18, 91. [Google Scholar] [CrossRef] [PubMed]
- Moller, H.D.; Mohiyuddin, M.; Prada-Luengo, I.; Sailani, M.R.; Halling, J.F.; Plomgaard, P.; Maretty, L.; Hansen, A.J.; Snyder, M.P.; Pilegaard, H.; et al. Circular DNA elements of chromosomal origin are common in healthy human somatic tissue. Nat. Commun. 2018, 9, 1069. [Google Scholar] [CrossRef]
- Ain, Q.; Schmeer, C.; Wengerodt, D.; Witte, O.W.; Kretz, A. Extrachromosomal Circular DNA: Current Knowledge and Implications for CNS Aging and Neurodegeneration. Int. J. Mol. Sci. 2020, 21, 2477. [Google Scholar] [CrossRef] [PubMed]
- Tomaska, L.; Nosek, J.; Kramara, J.; Griffith, J.D. Telomeric circles: Universal players in telomere maintenance? Nat. Struct. Mol. Biol. 2009, 16, 1010–1015. [Google Scholar] [CrossRef]
- Mazzucco, G.; Huda, A.; Galli, M.; Piccini, D.; Giannattasio, M.; Pessina, F.; Doksani, Y. Telomere damage induces internal loops that generate telomeric circles. Nat. Commun. 2020, 11, 5297. [Google Scholar] [CrossRef]
- Hull, R.M.; King, M.; Pizza, G.; Krueger, F.; Vergara, X.; Houseley, J. Transcription-induced formation of extrachromosomal DNA during yeast ageing. PLoS Biol. 2019, 17, e3000471. [Google Scholar] [CrossRef]
- Gresham, D.; Usaite, R.; Germann, S.M.; Lisby, M.; Botstein, D.; Regenberg, B. Adaptation to diverse nitrogen-limited environments by deletion or extrachromosomal element formation of the GAP1 locus. Proc. Natl. Acad. Sci. USA 2010, 107, 18551–18556. [Google Scholar] [CrossRef]
- Lanciano, S.; Carpentier, M.-C.; Llauro, C.; Jobet, E.; Robakowska-Hyzorek, D.; Lasserre, E.; Ghesquière, A.; Panaud, O.; Mirouze, M. Sequencing the extrachromosomal circular mobilome reveals retrotransposon activity in plants. PLoS Genet. 2017, 13, e1006630. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.; Wang, L.; Cho, J. PopRice extrachromosomal DNA sponges ABSCISIC ACID-INSENSITIVE 5 in rice seed-to-seedling transition. Plant Physiol. 2023, 192, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Mbodj, A.; Soundiramourtty, A.; Llauro, C.; Ghesquière, A.; Ingouff, M.; Keith Slotkin, R.; Pontvianne, F.; Catoni, M.; Mirouze, M. Extrachromosomal circular DNA and structural variants highlight genome instability in Arabidopsis epigenetic mutants. Nat. Commun. 2023, 14, 5236. [Google Scholar] [CrossRef] [PubMed]
- Moss, S.R.; Perryman, S.A.M.; Tatnell, L.V. Managing herbicide-resistant blackgrass (Alopecurus myosuroides): Theory and practice. Weed Technol. 2007, 21, 300–309. [Google Scholar] [CrossRef]
- Hicks, H.L.; Comont, D.; Coutts, S.R.; Crook, L.; Hull, R.; Norris, K.; Neve, P.; Childs, D.Z.; Freckleton, R.P. The factors driving evolved herbicide resistance at a national scale. Nat. Ecol. Evol. 2018, 2, 529–536. [Google Scholar] [CrossRef]
- Heap, I. The International Herbicide-Resistant Weed Database. 2023. Available online: https://www.weedscience.org/Home.aspx (accessed on 28 September 2023).
- Varah, A.; Ahodo, K.; Coutts, S.R.; Hicks, H.L.; Comont, D.; Crook, L.; Hull, R.; Neve, P.; Childs, D.Z.; Freckleton, R.P.; et al. The costs of human-induced evolution in an agricultural system. Nat. Sustain. 2020, 3, 63–71. [Google Scholar] [CrossRef]
- Comont, D.; Lowe, C.; Hull, R.; Crook, L.; Hicks, H.L.; Onkokesung, N.; Beffa, R.; Childs, D.Z.; Edwards, R.; Freckleton, R.P.; et al. Evolution of generalist resistance to herbicide mixtures reveals a trade-off in resistance management. Nat. Commun. 2020, 11, 3086. [Google Scholar] [CrossRef]
- Cai, L.; Comont, D.; MacGregor, D.; Lowe, C.; Beffa, R.; Neve, P.; Saski, C. The blackgrass genome reveals patterns of non-parallel evolution of polygenic herbicide resistance. New Phytol. 2023, 237, 1891–1907. [Google Scholar] [CrossRef]
- Mellado-Sánchez, M.; McDiarmid, F.; Cardoso, V.; Kanyuka, K.; MacGregor, D.R. Virus-mediated transient expression techniques enable gene function studies in black-grass. Plant Physiol. 2020, 183, 455–459. [Google Scholar] [CrossRef]
- Li, Z.; Parris, S.; Saski, C.A. A simple plant high-molecular-weight DNA extraction method suitable for single-molecule technologies. Plant Methods 2020, 16, 38. [Google Scholar] [CrossRef]
- Li, W.; Godzik, A. Cd-hit: A fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 2006, 22, 1658–1659. [Google Scholar] [CrossRef] [PubMed]
- RepeatMasker Open-4.0. Available online: http://www.repeatmasker.org (accessed on 11 January 2023).
- Cantarel, B.L.; Korf, I.; Robb, S.M.; Parra, G.; Ross, E.; Moore, B.; Holt, C.; Sanchez Alvarado, A.; Yandell, M. MAKER: An easy-to-use annotation pipeline designed for emerging model organism genomes. Genome Res. 2008, 18, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Young, M.D.; Wakefield, M.J.; Smyth, G.K.; Oshlack, A. Gene ontology analysis for RNA-seq: Accounting for selection bias. Genome Biol. 2010, 11, R14. [Google Scholar] [CrossRef]
- Villanueva, R.A.M.; Chen, Z.J. ggplot2: Elegant Graphics for Data Analysis, 2nd edition. Meas. Interdiscip. Res. Perspect. 2019, 17, 160–167. [Google Scholar] [CrossRef]
- Chan, P.P.; Lowe, T.M. GtRNAdb: A database of transfer RNA genes detected in genomic sequence. Nucleic Acids Res. 2009, 37, D93–D97. [Google Scholar] [CrossRef] [PubMed]
- Li, H. Minimap2: Pairwise alignment for nucleotide sequences. Bioinformatics 2018, 34, 3094–3100. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, A.R. BEDTools: The Swiss-Army Tool for Genome Feature Analysis. Curr. Protoc. Bioinform. 2014, 47, 11–12. [Google Scholar] [CrossRef] [PubMed]
- Tetard-Jones, C.; Sabbadin, F.; Moss, S.; Hull, R.; Neve, P.; Edwards, R. Changes in the proteome of the problem weed blackgrass correlating with multiple-herbicide resistance. Plant J. 2018, 94, 709–720. [Google Scholar] [CrossRef]
- Cummins, I.; Cole, D.J.; Edwards, R. A role for glutathione transferases functioning as glutathione peroxidases in resistance to multiple herbicides in black-grass. Plant J. 1999, 18, 285–292. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Li, J.; Zhou, X. Extrachromosomal circular DNA (eccDNA): An emerging star in cancer. Biomark. Res. 2022, 10, 53. [Google Scholar] [CrossRef]
- Fan, W.; Wang, L.; Chu, J.; Li, H.; Kim, E.Y.; Cho, J. Tracing Mobile DNAs: From Molecular to Population Scales. Front. Plant Sci. 2022, 13, 837378. [Google Scholar] [CrossRef] [PubMed]
- Conde, A.; Chaves, M.M.; Geros, H. Membrane transport, sensing and signaling in plant adaptation to environmental stress. Plant Cell Physiol. 2011, 52, 1583–1602. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, B.; Yuan, F. The Role of Transmembrane Proteins in Plant Growth, Development, and Stress Responses. Int. J. Mol. Sci. 2022, 23, 13627. [Google Scholar] [CrossRef]
- Pu, X.J.; Lv, X.; Tan, T.H.; Fu, F.Q.; Qin, G.W.; Lin, H.H. Roles of mitochondrial energy dissipation systems in plant development and acclimation to stress. Ann. Bot. 2015, 116, 583–600. [Google Scholar] [CrossRef]
- Barreto, P.; Koltun, A.; Nonato, J.; Yassitepe, J.; Maia, I.G.; Arruda, P. Metabolism and Signaling of Plant Mitochondria in Adaptation to Environmental Stresses. Int. J. Mol. Sci. 2022, 23, 11176. [Google Scholar] [CrossRef]
- Phukan, U.J.; Jeena, G.S.; Shukla, R.K. WRKY Transcription Factors: Molecular Regulation and Stress Responses in Plants. Front. Plant Sci. 2016, 7, 760. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Xu, Z.; Chen, M.; Yu, D. Functions of WRKYs in plant growth and development. Trends Plant Sci. 2023, 28, 630–645. [Google Scholar] [CrossRef]
- Li, J.; Han, G.; Sun, C.; Sui, N. Research advances of MYB transcription factors in plant stress resistance and breeding. Plant Signal. Behav. 2019, 14, 1613131. [Google Scholar] [CrossRef]
- Yang, L.D.; Jia, R.B.; Ge, T.X.; Ge, S.F.; Zhuang, A.; Chai, P.W.; Fan, X.Q. Extrachromosomal circular DNA: Biogenesis, structure, functions and diseases. Signal Transduct. Target. Ther. 2022, 7, 342. [Google Scholar] [CrossRef]
- Cao, X.; Wang, S.; Ge, L.; Zhang, W.; Huang, J.; Sun, W. Corrigendum: Extrachromosomal Circular DNA: Category, Biogenesis, Recognition, and Functions. Front. Vet. Sci. 2021, 8, 784611. [Google Scholar] [CrossRef]
- Wang, M.; Chen, X.; Yu, F.; Ding, H.; Zhang, Y.; Wang, K. Extrachromosomal Circular DNAs: Origin, formation and emerging function in Cancer. Int. J. Biol. Sci. 2021, 17, 1010–1025. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D.G.; Wang, Q.; Klaubert, S.R.; Green, K.; Wu, C.H.; Harcum, S.W.; Saski, C.A. Microevolutionary dynamics of eccDNA in Chinese hamster ovary cells grown in fed-batch cultures under control and lactate-stressed conditions. Sci. Rep. 2023, 13, 1200. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D.G.; Uy, L.; Fu, W.; Klaubert, S.R.; Harcum, S.W.; Saski, C.A. Dynamics of Amino Acid Metabolism, Gene Expression, and Circulomics in a Recombinant Chinese Hamster Ovary Cell Line Adapted to Moderate and High Levels of Extracellular Lactate. Genes 2023, 14, 1576. [Google Scholar] [CrossRef]
- Joubert, P.M.; Krasileva, K.V. The extrachromosomal circular DNAs of the rice blast pathogen Magnaporthe oryzae contain a wide variety of LTR retrotransposons, genes, and effectors. BMC Biol. 2022, 20, 260. [Google Scholar] [CrossRef] [PubMed]
- Sen, M.K.; Hamouzova, K.; Kosnarova, P.; Roy, A.; Soukup, J. Herbicide resistance in grass weeds: Epigenetic regulation matters too. Front. Plant Sci. 2022, 13, 1040958. [Google Scholar] [CrossRef] [PubMed]



| Sample ID | # eccDNA | Mean Length | Length Range | # eccDNA with Gene | # eccDNA with tRNA | % eccDNA with CDS |
|---|---|---|---|---|---|---|
| CC2R | 4812 | 7058 | 31–28,980 | 1031 | 53 | 21.43 |
| CC2S | 4233 | 6994 | 60–25,087 | 1010 | 77 | 23.86 |
| CC5R | 5288 | 6946 | 51–29,081 | 1145 | 74 | 21.65 |
| CC5S | 4332 | 7002 | 54–28,260 | 958 | 45 | 22.11 |
| Peldon | 4443 | 6868 | 36–26,368 | 962 | 59 | 21.65 |
| Lola91 | 5426 | 6918 | 49–27,814 | 1153 | 70 | 21.25 |
| Roth | 5663 | 7040 | 51–27,090 | 1291 | 70 | 22.8 |
| Pfam Accession | Annotation | # eccDNA |
|---|---|---|
| PF04195 | Putative gypsy type transposon | 416 |
| PF00361 | Proton-conducting membrane transporter | 348 |
| PF00146 | NADH dehydrogenase | 148 |
| PF00223 | Photosystem I psaA/psaB protein | 139 |
| PF00499 | NADH-ubiquinone/plastoquinone oxidoreductase chain 6 | 133 |
| PF00420 | NADH-ubiquinone/plastoquinone oxidoreductase chain 4L | 112 |
| PF13237 | 4Fe-4S dicluster domain | 103 |
| PF00124 | Photosynthetic reaction center protein | 93 |
| PF01578 | Cytochrome C assembly protein | 91 |
| PF00346 | Respiratory-chain NADH dehydrogenase, 49 Kd subunit | 87 |
| PF00662 | NADH-Ubiquinone oxidoreductase (complex I), chain 5 N-terminus | 81 |
| PF01010 | NADH-dehyrogenase subunit F, TMs, (complex I) C-terminus | 78 |
| PF00421 | Photosystem II protein | 48 |
| PF00623 | RNA polymerase Rpb1, domain 2 | 42 |
| PF01535 | PPR repeat | 41 |
| PF00006 | ATP synthase α/β family, nucleotide-binding domain | 38 |
| PF13456 | Reverse transcriptase-like | 36 |
| PF00276 | Ribosomal protein L23 | 32 |
| PF13041 | PPR repeat family | 31 |
| PF01824 | MatK/TrnK amino terminal region | 30 |
| Pfam Accession | Annotation | # eccDNA |
|---|---|---|
| PF04195 | Putative gypsy type transposon | 361 |
| PF00361 | Proton-conducting membrane transporter | 208 |
| PF00223 | Photosystem I psaA/psaB protein | 107 |
| PF00146 | NADH dehydrogenase | 91 |
| PF00499 | NADH-ubiquinone/plastoquinone oxidoreductase chain 6 | 81 |
| PF13237 | 4Fe-4S dicluster domain | 65 |
| PF00124 | Photosynthetic reaction center protein | 63 |
| PF00346 | Respiratory-chain NADH dehydrogenase, 49 Kd subunit | 62 |
| PF00662 | NADH-Ubiquinone oxidoreductase (complex I), chain 5 N-terminus | 56 |
| PF01578 | Cytochrome C assembly protein | 55 |
| PF01010 | NADH-dehyrogenase subunit F, TMs, (complex I) C-terminus | 51 |
| PF00420 | NADH-ubiquinone/plastoquinone oxidoreductase chain 4L | 43 |
| PF13456 | Reverse transcriptase-like | 36 |
| PF00421 | Photosystem II protein | 35 |
| PF01535 | PPR repeat | 35 |
| PF00006 | ATP synthase α/β family, nucleotide-binding domain | 34 |
| PF00069 | Protein kinase domain | 32 |
| PF00646 | F-box domain | 24 |
| PF02874 | ATP synthase α/β family, β-barrel domain | 24 |
| PF03040 | CemA family | 24 |
| Category | eccDNA Gene ID | Homologous Candidate Gene | Identity (%) | E-Value | Bit Score |
|---|---|---|---|---|---|
| Resistant | CC2R_00001032 | GSTF1 | 95.434 | 4.95 × 10−162 | 436 |
| CC5R_00001033 | GSTF1 | 31.25 | 1.07 × 10−14 | 64.7 | |
| CC5R_00001372 | GSTF1 | 55 | 2.22 × 10−91 | 257 | |
| Lola91_00001333 | GSTU2 | 61.364 | 1.70 × 10−42 | 128 | |
| Lola91_00001340 | GSTU2 | 38.009 | 1.69 × 10−42 | 133 | |
| Peldon_00000910 | OPR1 | 59.6 | 7.35 × 10−108 | 306 | |
| Peldon_00000911 | OPR1 | 64.646 | 6.10 × 10−44 | 137 | |
| Sensitive | CC2S_00001307 | GSTU2 | 65.741 | 8.99 × 10−53 | 157 |
| CC5S_00001012 | GSTF2 | 61.176 | 1.37 × 10−33 | 105 | |
| Roth_00001016 | GSTU2 | 31.429 | 2.42 × 10−24 | 85.1 |
| Chr | Start (Mb) | End (Mb) | # Herbicide-Resistant eccDNA | # Herbicide-Sensitive eccDNA | Genes Located | Functional Annotation | |
|---|---|---|---|---|---|---|---|
| 1 | 251.0 | 251.5 | 83 | 48 | ALOMY1G03512 | ||
| ALOMY1G03513 | PF00646 | F-box domain | |||||
| ALOMY1G03514 | PF02485 | Core-2/I-Branching enzyme | |||||
| ALOMY1G03515 | |||||||
| ALOMY1G03516 | PF01288 | 7,8-dihydro-6-hydroxymethylpterin-pyrophosphokinase (HPPK) | |||||
| ALOMY1G03517 | PF00809 | Pterin binding enzyme | |||||
| 5 | 8.5 | 9.0 | 133 | 104 | ALOMY5G31542 | PF01015 | Ribosomal S3Ae family |
| ALOMY5G31543 | PF03641 | Possible lysine decarboxylase | |||||
| ALOMY5G31544 | |||||||
| ALOMY5G31545 | PF00294 | pfkB family carbohydrate kinase | |||||
| ALOMY5G31546 | |||||||
| ALOMY5G31547 | |||||||
| ALOMY5G31548 | |||||||
| ALOMY5G31549 | |||||||
| ALOMY5G31550 | |||||||
| ALOMY5G31551 | PF00069 | Protein kinase domain | |||||
| ALOMY5G31552 | PF00069 | Protein kinase domain | |||||
| ALOMY5G31553 | PF00069 | Protein kinase domain | |||||
| ALOMY5G31554 | |||||||
| ALOMY5G31555 | |||||||
| 5 | 45.0 | 45.5 | 82 | 55 | ALOMY5G32133 | PF06747 | CHCH domain |
| ALOMY5G32134 | |||||||
| ALOMY5G32135 | |||||||
| ALOMY5G32136 | |||||||
| ALOMY5G32137 | |||||||
| ALOMY5G32138 | PF00179 | Ubiquitin-conjugating enzyme | |||||
| ALOMY5G32139 | PF00829 | Ribosomal prokaryotic L21 protein | |||||
| ALOMY5G32140 | |||||||
| ALOMY5G32141 | |||||||
| 7 | 243.5 | 244.0 | 130 | 80 | ALOMY7G40250 | PF02493 | MORN repeat |
| ALOMY7G40251 | |||||||
| ALOMY7G40252 | |||||||
| ALOMY7G40253 | PF00023 | Ankyrin repeat | |||||
| ALOMY7G40254 | PF01694 | Rhomboid family | |||||
| ALOMY7G40255 | |||||||
| ALOMY7G40256 | PF12937 | F-box-like | |||||
| ALOMY7G40257 | PF00194 | Eukaryotic-type carbonic anhydrase | |||||
| ALOMY7G40258 | |||||||
| ALOMY7G40259 | PF00153 | Mitochondrial carrier protein | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, W.; MacGregor, D.R.; Comont, D.; Saski, C.A. Sequence Characterization of Extra-Chromosomal Circular DNA Content in Multiple Blackgrass (Alopecurus myosuroides) Populations. Genes 2023, 14, 1905. https://doi.org/10.3390/genes14101905
Fu W, MacGregor DR, Comont D, Saski CA. Sequence Characterization of Extra-Chromosomal Circular DNA Content in Multiple Blackgrass (Alopecurus myosuroides) Populations. Genes. 2023; 14(10):1905. https://doi.org/10.3390/genes14101905
Chicago/Turabian StyleFu, Wangfang, Dana R. MacGregor, David Comont, and Christopher A. Saski. 2023. "Sequence Characterization of Extra-Chromosomal Circular DNA Content in Multiple Blackgrass (Alopecurus myosuroides) Populations" Genes 14, no. 10: 1905. https://doi.org/10.3390/genes14101905
APA StyleFu, W., MacGregor, D. R., Comont, D., & Saski, C. A. (2023). Sequence Characterization of Extra-Chromosomal Circular DNA Content in Multiple Blackgrass (Alopecurus myosuroides) Populations. Genes, 14(10), 1905. https://doi.org/10.3390/genes14101905






