Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
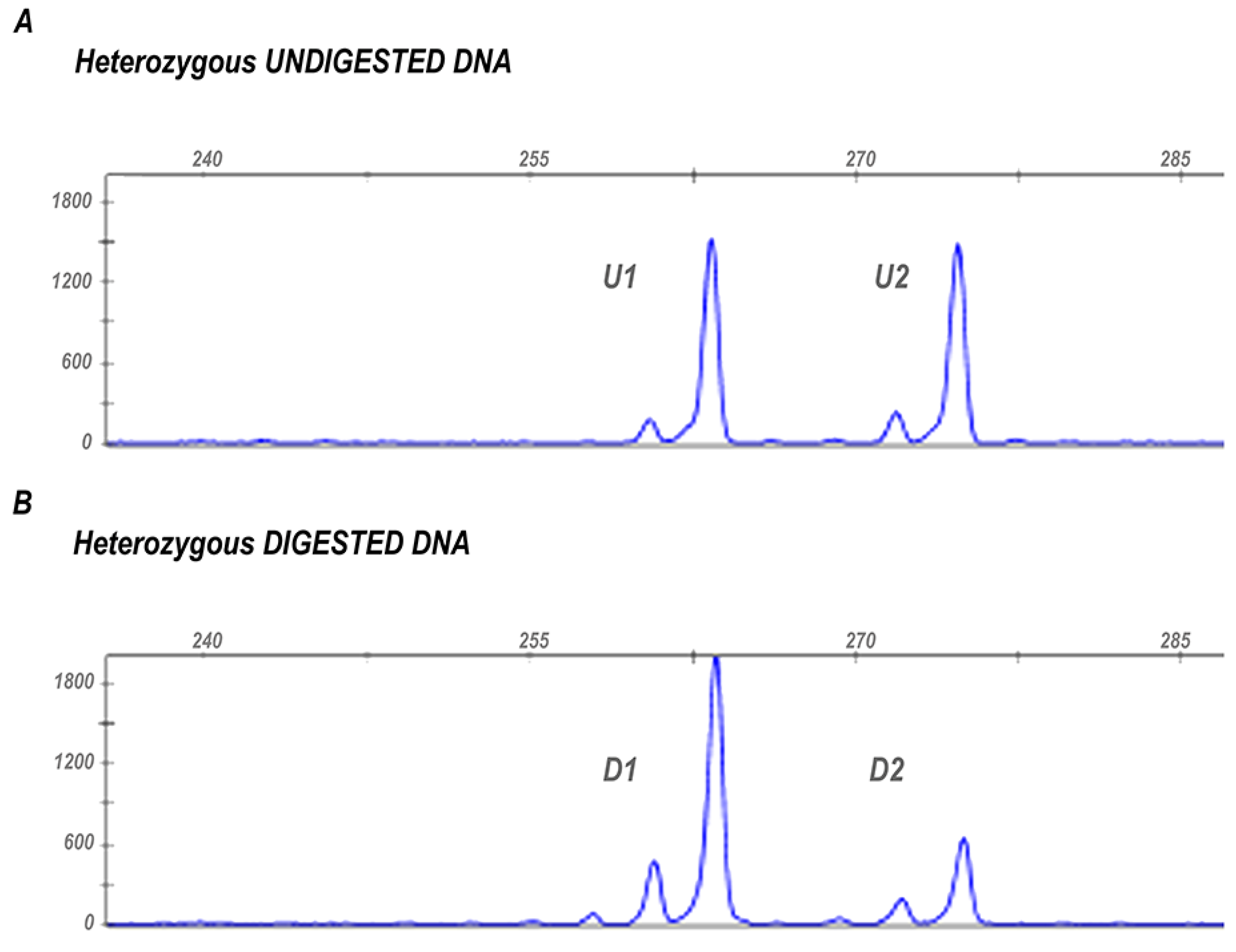
2.2. Assessment of X-Chromosome Inactivation (XCI)
2.3. Statistical Analyses
3. Results
Quantification of the Degree of XCI Skewing in MG Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Billi, A.C.; Kahlenberg, J.M.; Gudjonsson, J.E. Sex bias in autoimmunity. Curr. Opin. Rheumatol. 2019, 31, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Gilhus, N.E.; Tzartos, S.; Evoli, A.; Palace, J.; Burns, T.M.; Verschuuren, J.J.G.M. Myasthenia Gravis. Nat. Rev. Dis. Primer 2019, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Catalin, J.; Silviana, J.; Claudia, B. Clinical Presentation of Myasthenia Gravis; IntechOpen: London, UK, 2019. [Google Scholar]
- Rousseff, R.T. Diagnosis of Myasthenia Gravis. J. Clin. Med. 2021, 10, 1736. [Google Scholar] [CrossRef] [PubMed]
- Ciafaloni, E. Myasthenia Gravis and Congenital Myasthenic Syndromes. Contin. Lifelong Learn. Neurol. 2019, 25, 1767–1784. [Google Scholar] [CrossRef]
- Koneczny, I.; Herbst, R. Myasthenia Gravis: Pathogenic Effects of Autoantibodies on Neuromuscular Architecture. Cells 2019, 8, 671. [Google Scholar] [CrossRef]
- Dresser, L.; Wlodarski, R.; Rezania, K.; Soliven, B. Myasthenia Gravis: Epidemiology, Pathophysiology and Clinical Manifestations. J. Clin. Med. 2021, 10, 2235. [Google Scholar] [CrossRef]
- Gilhus, N.E.; Verschuuren, J. Myasthenia gravis: Subgroup classification and therapeutic strategies. Lancet Neurol. 2015, 14, 1023–1036. [Google Scholar] [CrossRef]
- Mao, Z.-F.; Mo, X.-A.; Qin, C.; Lai, Y.-R.; Hackett, M. Incidence of Thymoma in Myasthenia Gravis: A Systematic Review. J. Clin. Neurol. 2012, 8, 161–169. [Google Scholar] [CrossRef]
- Zagoriti, Z.; Kambouris, M.E.; Patrinos, G.P.; Tzartos, S.J.; Poulas, K. Recent Advances in Genetic Predisposition of Myasthenia Gravis. BioMed Res. Int. 2013, 2013, 404053. [Google Scholar] [CrossRef]
- Invernizzi, P.; Pasini, S.; Selmi, C.; Miozzo, M.; Podda, M. Skewing of X chromosome inactivation in autoimmunity. Autoimmunity 2008, 41, 272–277. [Google Scholar] [CrossRef]
- Ortona, E.; Pierdominici, M.; Maselli, A.; Veroni, C.; Aloisi, F.; Shoenfeld, Y. Sex-based differences in autoimmune diseases. Ann. Ist. Super. Sanita 2016, 52, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Mougeot, J.C.; Noll, B.; Mougeot, F.K.B. Sjögren’s syndrome X-chromosome dose effect: An epigenetic perspective. Oral Dis. 2018, 25, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, M.J.; Mahmoudi, M.; Ghotloo, S. Escape from X chromosome inactivation and female bias of autoimmune diseases. Mol. Med. 2020, 26, 127. [Google Scholar] [CrossRef] [PubMed]
- Syrett, C.M.; Anguera, M.C. When the balance is broken: X-linked gene dosage from two X chromosomes and female-biased autoimmunity. J. Leukoc. Biol. 2019, 106, 919–932. [Google Scholar] [CrossRef]
- Xing, E.; Billi, A.C.; Gudjonsson, J.E. Sex Bias and Autoimmune Diseases. J. Investig. Dermatol. 2021, 142, 857–866. [Google Scholar] [CrossRef]
- Wutz, A.; Valencia, K. Recent insights into the regulation of X-chromosome inactivation. Adv. Genom. Genet. 2015, 5, 227–238. [Google Scholar] [CrossRef][Green Version]
- Gribnau, J.; Barakat, T.S. X-Chromosome Inactivation and Its Implications for Human Disease. bioRxiv 2017, 076950. [Google Scholar] [CrossRef]
- Chitnis, S.; Monteiro, J.; Glass, D.; Apatoff, B.; Salmon, J.; Concannon, P.; Gregersen, P.K. The role of X-chromosome inactivation in female predisposition to autoimmunity. Arthritis Res. Ther. 2000, 2, 399–406. [Google Scholar] [CrossRef]
- Youness, A.; Miquel, C.-H.; Guéry, J.-C. Escape from X Chromosome Inactivation and the Female Predominance in Autoimmune Diseases. Int. J. Mol. Sci. 2021, 22, 1114. [Google Scholar] [CrossRef]
- Miozzo, M.; Selmi, C.; Gentilin, B.; Grati, F.R.; Sirchia, S.; Oertelt-Prigione, S.; Zuin, M.; Gershwin, M.E.; Podda, M.; Invernizzi, P. Preferential X chromosome loss but random inactivation characterize primary biliary cirrhosis. Hepatology 2007, 46, 456–462. [Google Scholar] [CrossRef]
- Angum, F.; Khan, T.; Kaler, J.; Siddiqui, L.; Hussain, A. The Prevalence of Autoimmune Disorders in Women: A Narrative Review. Cureus 2020, 12, e8094. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.M.; Koelsch, K.A.; Kurien, B.T.; Harley, I.T.W.; Wren, J.D.; Harley, J.B.; Scofield, R.H. Characterization of cxorf21 Provides Molecular Insight Into Female-Bias Immune Response in SLE Pathogenesis. Front. Immunol. 2019, 10, 2160. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, X.; Gibson, A.; Edberg, J.; Kimberly, R.P.; Absher, D.M. Skewed allelic expression on X chromosome associated with aberrant expression of XIST on systemic lupus erythematosus lymphocytes. Hum. Mol. Genet. 2020, 29, 2523–2534. [Google Scholar] [CrossRef] [PubMed]
- Kanaan, S.B.; Onat, O.E.; Balandraud, N.; Martin, G.V.; Nelson, J.L.; Azzouz, D.F.; Auger, I.; Arnoux, F.; Martin, M.; Roudier, J.; et al. Evaluation of X Chromosome Inactivation with Respect to HLA Genetic Susceptibility in Rheumatoid Arthritis and Systemic Sclerosis. PLoS ONE 2016, 11, e0158550. [Google Scholar] [CrossRef]
- Simmonds, M.J.; Kavvoura, F.K.; Brand, O.J.; Newby, P.R.; Jackson, L.E.; Hargreaves, C.E.; Franklyn, J.A.; Gough, S.C.L. Skewed X Chromosome Inactivation and Female Preponderance in Autoimmune Thyroid Disease: An Association Study and Meta-Analysis. J. Clin. Endocrinol. Metab. 2014, 99, E127–E131. [Google Scholar] [CrossRef] [PubMed]
- Ishido, N.; Inoue, N.; Watanabe, M.; Hidaka, Y.; Iwatani, Y. The Relationship between Skewed X Chromosome Inactivation and the Prognosis of Graves’ and Hashimoto’s Diseases. Thyroid 2015, 25, 256–261. [Google Scholar] [CrossRef]
- Santiwatana, S.; Mahachoklertwattana, P.; Limwongse, C.; Khlairit, P.; Pongratanakul, S.; Roothumnong, E.; Prangphan, K.; Choubtum, L.; Songdej, D.; Poomthavorn, P. Skewed X chromosome inactivation in girls and female adolescents with autoimmune thyroid disease. Clin. Endocrinol. 2018, 89, 863–869. [Google Scholar] [CrossRef]
- Fontana, L.; Bedeschi, M.F.; Cagnoli, G.A.; Costanza, J.; Persico, N.; Gangi, S.; Porro, M.; Ajmone, P.F.; Colapietro, P.; Santaniello, C.; et al. (Epi)genetic profiling of extraembryonic and postnatal tissues from female monozygotic twins discordant for Beckwith–Wiedemann syndrome. Mol. Genet. Genom. Med. 2020, 8, e1386. [Google Scholar] [CrossRef]
- Salsano, E.; Tabano, S.; Sirchia, S.M.; Colapietro, P.; Castellotti, B.; Gellera, C.; Rimoldi, M.; Pensato, V.; Mariotti, C.; Pareyson, D.; et al. Preferential expression of mutant ABCD1 allele is common in adrenoleukodystrophy female carriers but unrelated to clinical symptoms. Orphanet J. Rare Dis. 2012, 7, 10. [Google Scholar] [CrossRef]
- Minks, J.; Robinson, W.P.; Brown, C.J. A skewed view of X chromosome inactivation. J. Clin. Investig. 2008, 118, 20–23. [Google Scholar] [CrossRef]
- Manoukian, S.; Verderio, P.; Tabano, S.; Colapietro, P.; Pizzamiglio, S.; Grati, F.R.; Calvello, M.; Peissel, B.; Burn, J.; Pensotti, V.; et al. X chromosome inactivation pattern in BRCA gene mutation carriers. Eur. J. Cancer Oxf. Engl. 2013, 49, 1136–1141. [Google Scholar] [CrossRef] [PubMed]
- Migliore, L.; Nicolì, V.; Stoccoro, A. Gender Specific Differences in Disease Susceptibility: The Role of Epigenetics. Biomedicines 2021, 9, 652. [Google Scholar] [CrossRef] [PubMed]
- Özçelik, T.; Uz, E.; Akyerli, C.B.; Bagislar, S.; Mustafa, A.C.; Gursoy, A.; Akarsu, N.; Toruner, G.; Kamel, N.; Gullu, S. Evidence from autoimmune thyroiditis of skewed X-chromosome inactivation in female predisposition to autoimmunity. Eur. J. Hum. Genet. 2006, 14, 791–797. [Google Scholar] [CrossRef][Green Version]
- Chabchoub, G.; Uz, E.; Maalej, A.; Mustafa, A.C.; Rebai, A.; Mnif, M.; Bahloul, Z.; Farid, N.R.; Ozcelik, T.; Ayadi, H. Analysis of skewed X-chromosome inactivation in females with rheumatoid arthritis and autoimmune thyroid diseases. Arthritis Res. Ther. 2009, 11, R106. [Google Scholar] [CrossRef] [PubMed]
- Özbalkan, Z.; Baǧışlar, S.; Kiraz, S.; Akyerli, C.B.; Özer, H.T.E.; Yavuz, S.; Birlik, A.M.; Çalgüneri, M.; Özçelik, T. Skewed X chromosome inactivation in blood cells of women with scleroderma. Arthritis Rheum. 2005, 52, 1564–1570. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, G.P. Gender bias in autoimmune diseases: X chromosome inactivation in women with multiple sclerosis. J. Neurol. Sci. 2009, 286, 43–46. [Google Scholar] [CrossRef]
- Uz, E.; Mustafa, C.; Topaloglu, R.; Bilginer, Y.; Dursun, A.; Kasapcopur, O.; Ozen, S.; Bakkaloglu, A.; Ozcelik, T. Increased frequency of extremely skewed X chromosome inactivation in juvenile idiopathic arthritis. Arthritis Care Res. 2009, 60, 3410–3412. [Google Scholar] [CrossRef]
- Melzer, N.; Ruck, T.; Fuhr, P.; Gold, R.; Hohlfeld, R.; Marx, A.; Melms, A.; Tackenberg, B.; Schalke, B.; Schneider-Gold, C.; et al. Clinical features, pathogenesis, and treatment of myasthenia gravis: A supplement to the Guidelines of the German Neurological Society. J. Neurol. 2016, 263, 1473–1494. [Google Scholar] [CrossRef]
- Cortés-Vicente, E.; Álvarez-Velasco, R.; Segovia, S.; Paradas, C.; Casasnovas, C.; Guerrero-Sola, A.; Pardo, J.; Ramos-Fransi, A.; Sevilla, T.; de Munain, A.L.; et al. Clinical and therapeutic features of myasthenia gravis in adults based on age at onset. Neurology 2020, 94, e1171–e1180. [Google Scholar] [CrossRef]
- Busque, L.; Mio, R.; Mattioli, J.; Brais, E.; Blais, N.; Lalonde, Y.; Maragh, M.; Gilliland, D. Nonrandom X-Inactivation Patterns in Normal Females: Lyonization Ratios Vary with Age. Blood 1996, 88, 59–65. [Google Scholar] [CrossRef]
- Hatakeyama, C.; Anderson, C.; Beever, C.; Peñaherrera, M.; Brown, C.; Robinson, W. The dynamics of X-inactivation skewing as women age. Clin. Genet. 2004, 66, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, M.; Knudsen, G.P.S.; Bathum, L.; Naumova, A.K.; Sørensen, I.A.T.; Brix, T.H.; Svendsen, A.J.; Christensen, K.; Kyvik, K.O.; Ørstavik, K.H. Twin study of genetic and aging effects on X chromosome inactivation. Eur. J. Hum. Genet. 2005, 13, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Tukiainen, T.; Villani, A.-C.; Yen, A.; Rivas, M.A.; Marshall, J.L.; Satija, R.; Aguirre, M.; Gauthier, L.; Fleharty, M.; Kirby, A.; et al. Landscape of X chromosome inactivation across human tissues. Nature 2017, 550, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Shvetsova, E.; Sofronova, A.; Monajemi, R.; Gagalova, K.; Draisma, H.H.M.; White, S.J.; Santen, G.W.E.; Chuva de Sousa Lopes, S.M.; Heijmans, B.T.; van Meurs, J.; et al. Skewed X-inactivation is common in the general female population. Eur. J. Hum. Genet. 2018, 27, 455–465. [Google Scholar] [CrossRef]
- Zito, A.; Davies, M.N.; Tsai, P.-C.; Roberts, S.; Andres-Ejarque, R.; Nardone, S.; Bell, J.T.; Wong, C.C.Y.; Small, K.S. Heritability of skewed X-inactivation in female twins is tissue-specific and associated with age. Nat. Commun. 2019, 10, 5339. [Google Scholar] [CrossRef]
- Mengel-From, J.; Lindahl-Jacobsen, R.; Nygaard, M.; Soerensen, M.; Ørstavik, K.H.; Hertz, J.M.; Andersen-Ranberg, K.; Tan, Q.; Christensen, K. Skewness of X-chromosome inactivation increases with age and varies across birth cohorts in elderly Danish women. Sci. Rep. 2021, 11, 4326. [Google Scholar] [CrossRef]
- Champion, K.M.; Gilbert, J.; Asimakopoulos, F.A.; Hinshelwood, S.; Green, A. Clonal haemopoiesis in normal elderly women: Implications for the myeloproliferative disorders and myelodysplastic syndromes. Br. J. Haematol. 1997, 97, 920–926. [Google Scholar] [CrossRef]
- Dunford, A.; Weinstock, D.M.; Savova, V.; Schumacher, S.E.; Cleary, J.P.; Yoda, A.; Sullivan, T.J.; Hess, J.M.; Gimelbrant, A.A.; Beroukhim, R.; et al. Tumor-suppressor genes that escape from X-inactivation contribute to cancer sex bias. Nat. Genet. 2016, 49, 10–16. [Google Scholar] [CrossRef]
- Parsons, B.L. Multiclonal tumor origin: Evidence and implications. Mutat. Res. Mutat. Res. 2018, 777, 1–18. [Google Scholar] [CrossRef]
- Li, G.; Su, Q.; Liu, G.-Q.; Gong, L.; Zhang, W.; Zhu, S.-J.; Zhang, H.-L.; Feng, Y.-M. Skewed X chromosome inactivation of blood cells is associated with early development of lung cancer in females. Oncol. Rep. 2006, 16, 859–864. [Google Scholar] [CrossRef]
- Li, G.; Jin, T.; Liang, H.; Tu, Y.; Zhang, W.; Gong, L.; Su, Q.; Gao, G. Skewed X-chromosome inactivation in patients with esophageal carcinoma. Diagn. Pathol. 2013, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Pageau, G.J.; Hall, L.L.; Ganesan, S.; Livingston, D.M.; Lawrence, J.B. The disappearing Barr body in breast and ovarian cancers. Nat. Cancer 2007, 7, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Lee, H.J.; Kim, J.; Lee, J.J.; Maeng, L.-S. Dysregulation of X Chromosome Inactivation in High Grade Ovarian Serous Adenocarcinoma. PLoS ONE 2015, 10, e0118927. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Larson, N.B.; Fogarty, Z.; Larson, M.C.; Kalli, K.R.; Lawrenson, K.; Gayther, S.; Fridley, B.L.; Goode, E.L.; Winham, S.J. An integrative approach to assess X-chromosome inactivation using allele-specific expression with applications to epithelial ovarian cancer. Genet. Epidemiol. 2017, 41, 898–914. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.L.; Moysich, K.; Kohli, S.; Lele, S.; Odunsi, K.; Eng, K.H. Transmission of X-linked Ovarian Cancer: Characterization and Implications. Diagnostics 2020, 10, 90. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, M.; Langerød, A.; Knudsen, G.P.; Weber, B.L.; Børresen-Dale, A.-L.; Ørstavik, K.H. High frequency of skewed X inactivation in young breast cancer patients. J. Med Genet. 2002, 39, 30–33. [Google Scholar] [CrossRef][Green Version]
- Lose, F.; Duffy, D.L.; Kay, G.F.; Kedda, M.A.; Spurdle, A.B.; Kathleen Cuningham Foundation Consortium for Research into Familial Breast Cancer; Australian Ovarian Cancer Study Management Group. Skewed X Chromosome Inactivation and Breast and Ovarian Cancer Status: Evidence for X-Linked Modifiers of BRCA1. J. Natl. Cancer Inst. 2008, 100, 1519–1529. [Google Scholar] [CrossRef]
- Kristiansen, M.; Helland, Å.; Kristensen, G.B.; Olsen, A.O.; Lønning, P.E.; Børresen-Dale, A.-L.; Ørstavik, K.H. X chromosome inactivation in cervical cancer patients. Cancer Genet. Cytogenet. 2003, 146, 73–76. [Google Scholar] [CrossRef]
- Li, G.; Zhang, Z.; Jin, T.; Liang, H.; Tu, Y.; Gong, L.; Chen, Z.; Gao, G. High frequency of the X-chromosome inactivation in young female patients with high-grade glioma. Diagn. Pathol. 2013, 8, 101. [Google Scholar] [CrossRef]
- Dragin, N.; Bismuth, J.; Cizeron-Clairac, G.; Biferi, M.G.; Berthault, C.; Serraf, A.; Nottin, R.; Klatzmann, D.; Cumano, A.; Barkats, M.; et al. Estrogen-mediated downregulation of AIRE influences sexual dimorphism in autoimmune diseases. J. Clin. Investig. 2016, 126, 1525–1537. [Google Scholar] [CrossRef]
- Invernizzi, P.; Miozzo, M.; Selmi, C.; Persani, L.; Battezzati, P.M.; Zuin, M.; Lucchi, S.; Meroni, P.L.; Marasini, B.; Zeni, S.; et al. X Chromosome Monosomy: A Common Mechanism for Autoimmune Diseases. J. Immunol. 2005, 175, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.W. Intragenomic Conflict and Immune Tolerance: Do Selfish X-Linked Alleles Drive Skewed X Chromosome Inactivation? Genome Biol. Evol. 2017, 10, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Avidan, N.; Le Panse, R.; Harbo, H.F.; Bernasconi, P.; Poulas, K.; Ginzburg, E.; Cavalcante, P.; Colleoni, L.; Baggi, F.; Antozzi, C.; et al. VAV 1 and BAFF, via NF κB pathway, are genetic risk factors for myasthenia gravis. Ann. Clin. Transl. Neurol. 2014, 1, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Bo, C.; Wang, J.; Zhang, H.; Cao, Y.; Lu, X.; Wang, T.; Wang, Y.; Li, S.; Kong, X.; Sun, X.; et al. Global pathway view analysis of microRNA clusters in myasthenia gravis. Mol. Med. Rep. 2019, 19, 2350–2360. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.L.; Prchal, J.T. X-linked clonality testing: Interpretation and limitations. Blood 2007, 110, 1411–1419. [Google Scholar] [CrossRef]
- Allen, R.C.; Zoghbi, H.Y.; Moseley, A.B.; Rosenblatt, H.M.; Belmont, J.W. Methylation of HpaII and HhaI sites near the polymorphic CAG repeat in the human androgen-receptor gene correlates with X chromosome inactivation. Am. J. Hum. Genet. 1992, 51, 1229–1239. [Google Scholar]
- Zhang, X.; Li, Y.; Ma, L.; Zhang, G.; Liu, M.; Wang, C.-Y.; Zheng, Y.; Li, R. A new sex-specific underlying mechanism for female schizophrenia: Accelerated skewed X chromosome inactivation. Biol. Sex Differ. 2020, 11, 39. [Google Scholar] [CrossRef]
- Broen, A.J.C.; Wolvers-Tettero, I.L.M.; Bon, L.G.-V.; Vonk, M.C.; Coenen, M.J.H.; Lafyatis, R.; Radstake, T.R.D.J.; Langerak, A.W. Skewed X chromosomal inactivation impacts T regulatory cell function in systemic sclerosis. Ann. Rheum. Dis. 2010, 69, 2213–2216. [Google Scholar] [CrossRef]

| Patients | ||||
| No of Patients | Age (Mean ± SD) | Age of Onset | Osserman Classification | Thymic Histology |
| Informative n = 121 | 47.90 ± 12.15 | <45 years n = 74 | I n = 4 | No thymoma n = 20 |
| ≥45 years n = 45 | II n = 96 | Hyperplasia n = 18 | ||
| n.a. n = 2 | III n = 13 | Thymoma n = 82 | ||
| IV n = 6 | n.a. n = 1 | |||
| n.a. n = 2 | ||||
| Controls | ||||
| No of Controls | Age (Mean ± SD) | |||
| Informative n = 130 | 45.82 ± 11.75 | |||
| Random XCI < 75% | Moderate XCI ≥ 75% | Severe XCI ≥ 90% | |
|---|---|---|---|
| MG patients | 64 | 28 | 29 |
| Control population | 108 | 18 | 4 |
| Random XCI (XCI < 75%) | Skewed XCI (XCI ≥ 75%) | p-Value | |
|---|---|---|---|
| MG patients | 64 | 57 | <0.00001 (*) |
| Controls | 108 | 22 | |
| MG aged < 45 years | 35 | 27 | 0.53 |
| MG aged ≥ 45 years | 29 | 30 | |
| Controls aged < 45 years | 65 | 4 | 0.0007 (*) |
| Controls aged ≥ 45 years | 43 | 18 | |
| MG aged < 45 years | 35 | 27 | <0.00001 (*) |
| Controls aged < 45 years | 65 | 4 | |
| MG aged ≥ 45 years | 29 | 30 | 0.03 (*) |
| Controls aged ≥ 45 years | 43 | 18 | |
| Age of onset of MG | |||
| <45 years | 42 | 32 | 0.77 |
| ≥45 years | 21 | 24 | |
| Osserman stages | |||
| I | 3 | 1 | 0.63 |
| II | 51 | 45 | |
| III | 7 | 6 | |
| IV | 2 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolì, V.; Tabano, S.M.; Colapietro, P.; Maestri, M.; Ricciardi, R.; Stoccoro, A.; Fontana, L.; Guida, M.; Miozzo, M.; Coppedè, F.; et al. Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis. Genes 2022, 13, 696. https://doi.org/10.3390/genes13040696
Nicolì V, Tabano SM, Colapietro P, Maestri M, Ricciardi R, Stoccoro A, Fontana L, Guida M, Miozzo M, Coppedè F, et al. Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis. Genes. 2022; 13(4):696. https://doi.org/10.3390/genes13040696
Chicago/Turabian StyleNicolì, Vanessa, Silvia Maria Tabano, Patrizia Colapietro, Michelangelo Maestri, Roberta Ricciardi, Andrea Stoccoro, Laura Fontana, Melania Guida, Monica Miozzo, Fabio Coppedè, and et al. 2022. "Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis" Genes 13, no. 4: 696. https://doi.org/10.3390/genes13040696
APA StyleNicolì, V., Tabano, S. M., Colapietro, P., Maestri, M., Ricciardi, R., Stoccoro, A., Fontana, L., Guida, M., Miozzo, M., Coppedè, F., & Migliore, L. (2022). Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis. Genes, 13(4), 696. https://doi.org/10.3390/genes13040696







