Hybridization of Russian Sturgeon (Acipenser gueldenstaedtii, Brandt and Ratzeberg, 1833) and American Paddlefish (Polyodon spathula, Walbaum 1792) and Evaluation of Their Progeny
Abstract
1. Introduction
2. Materials and Methods
2.1. Reproduction and Nursing
2.2. Microsatellite Marker Analysis
2.3. Flow Cytometry and DNA Content Analysis
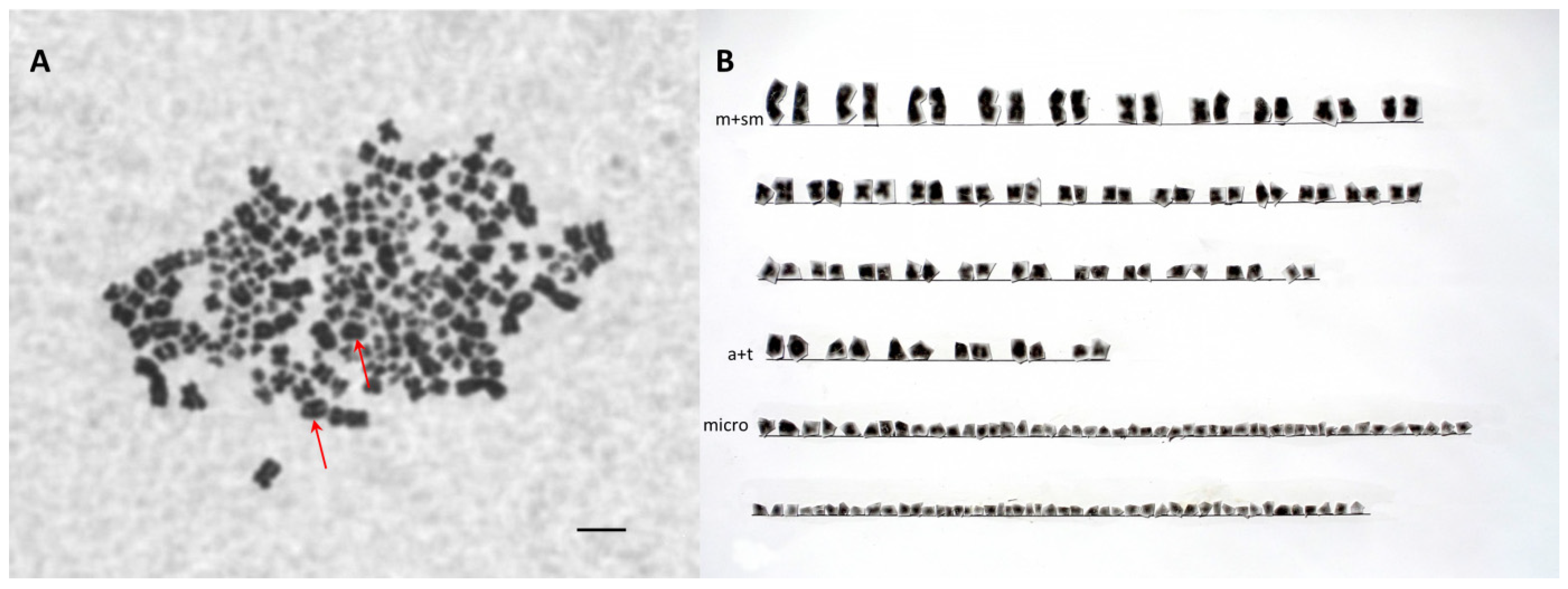
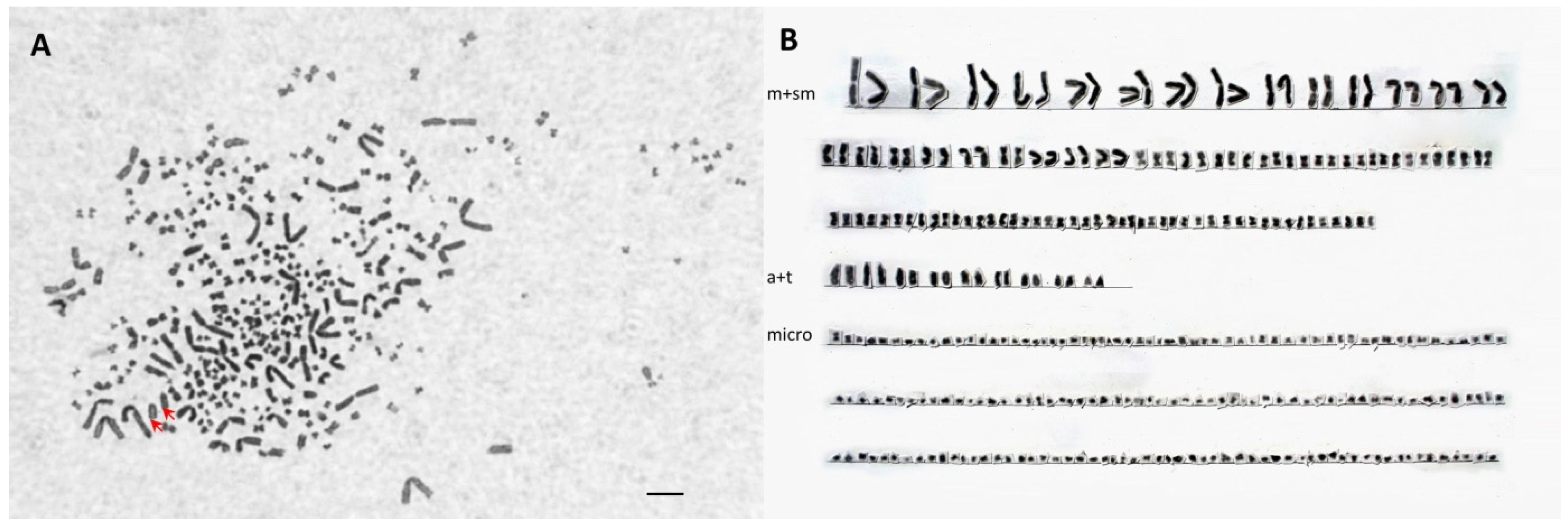
2.4. Chromosome Analysis
2.5. Morphometry
3. Results
3.1. Fertilization, Survival, Growth of Hybrids
3.2. Microsatellite Analysis
3.3. Flow Cytometry and DNA Content Analysis
3.4. Chromosome Analysis and Ploidy Level in Different Cytometry Class Groups
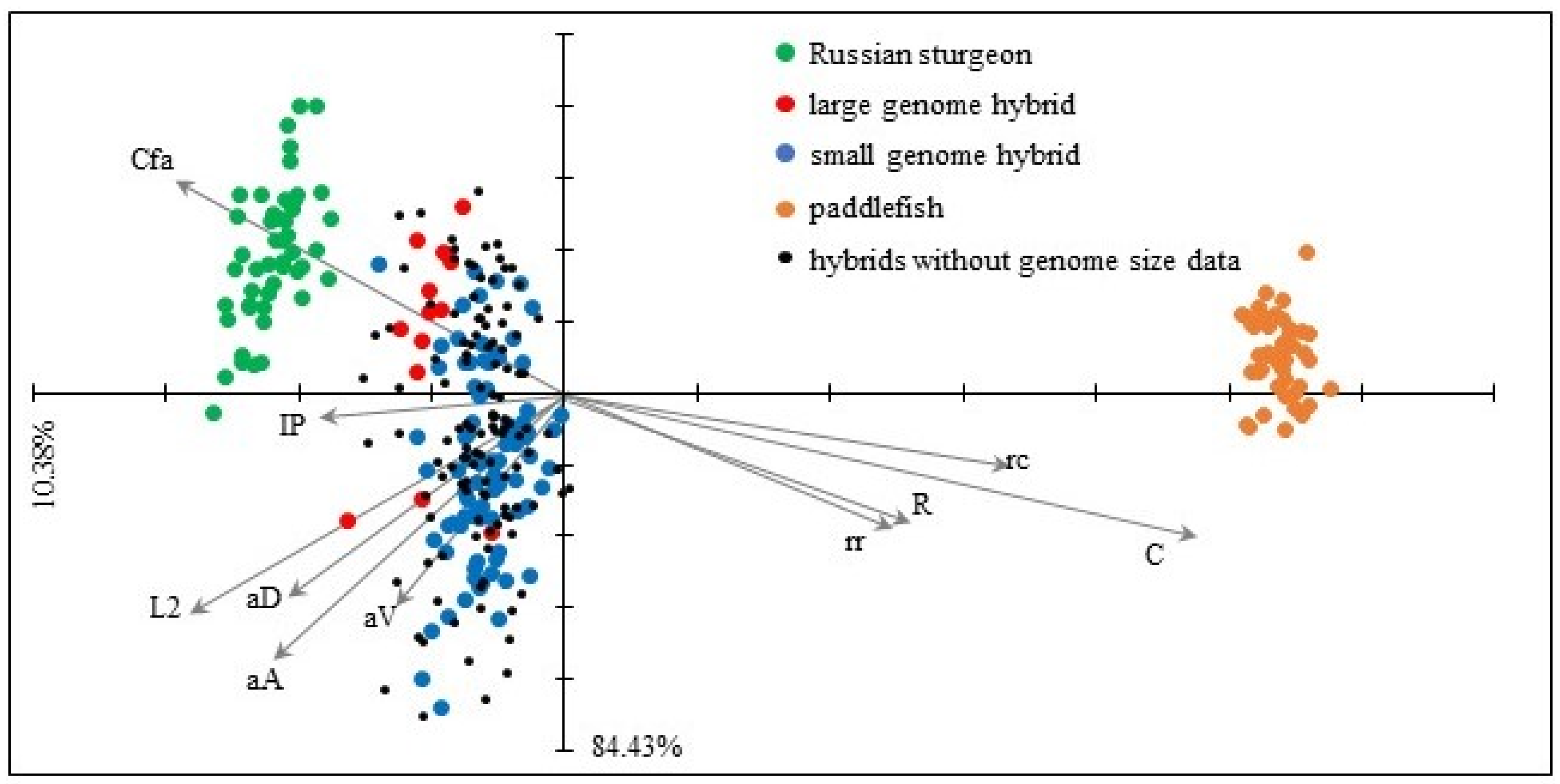
3.5. Morphometry
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leggatt, R.A.; Iwama, G.K. Occurrence of polyploidy in the fishes. Rev. Fish Boil. Fish. 2003, 13, 237–246. [Google Scholar] [CrossRef]
- Berrebi, P. Speciation of the genus Barbus in the north Mediterranean basin: Recent advances from biochemical genetics. Boil. Conserv. 1995, 72, 237–249. [Google Scholar] [CrossRef]
- Birstein, V.J.; Hanner, R.; DeSalle, R. Phylogeny of the Acipenseriformes: Cytogenetic and molecular approaches. Environ. Biol. Fishes 1997, 48, 127–155. [Google Scholar] [CrossRef]
- Sallan, L. Major issues in the origins of ray-finned fish (Actinopterygii) biodiversity. Boil. Rev. 2014, 89, 950–971. [Google Scholar] [CrossRef] [PubMed]
- Hilton, E.J.; Grande, L. Review of the fossil record of sturgeons, family Acipenseridae (Actinopterygii: Acipenseriformes), from North America. J. Paléontol. 2006, 80, 672–683. [Google Scholar] [CrossRef]
- Bemis, W.; Findeis, E.K.; Grande, L. An overview of Acipenseriformes. Environ. Biol. Fishes 1997, 48, 25–71. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, W.; Li, L.; Ma, X.; Chen, J. Genetic variation and relationships of seven sturgeon species and ten interspecific hybrids. Genet. Sel. Evol. 2013, 45, 21. [Google Scholar] [CrossRef]
- Symonová, R.; Havelka, M.; Amemiya, C.T.; Howell, W.M.; Kořínková, T.; Flajšhans, M.; Gela, D.; Ráb, P. Molecular cytogenetic differentiation of paralogs of HOX paralogs in duplicated and re-diploidized genome of the North American paddlefish (Polyodon spathula). BMC Genet. 2017, 18, 19. [Google Scholar] [CrossRef]
- Nelson, S.J.; Grande, C.T.; Wilson, V.H.M. Fishes of the World; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; pp. 118–121. [Google Scholar]
- Zhang, H.; Jarić, I.; Roberts, D.L.; He, Y.; Du, H.; Wu, J.; Wang, C.; Wei, Q. Extinction of one of the world’s largest freshwater fishes: Lessons for conserving the endangered Yangtze fauna. Sci. Total. Environ. 2020, 710, 136242. [Google Scholar] [CrossRef]
- Rajkov, J.; Shao, Z.; Berrebi, P. Evolution of Polyploidy and Functional Diploidization in Sturgeons: Microsatellite Analysis in 10 Sturgeon Species. J. Hered. 2014, 105, 521–531. [Google Scholar] [CrossRef]
- Mims, S.D.; Shelton, W.L.; Linhart, O.; Wang, C. Induced Meiotic Gynogenesis of Paddlefish Polyodon spathula. J. World Aquac. Soc. 1997, 28, 334–343. [Google Scholar] [CrossRef]
- Mims, S.D.; Shelton, W.L. Induced meiotic gynogenesis in shovelnose sturgeon. Aquac. Int. 1998, 6, 323–329. [Google Scholar] [CrossRef]
- Zou, Y.C.; Wei, Q.W.; Pan, G.B. Induction of meiotic gynogenesis in paddlefish (Polyodon spathula) and its confirmation using microsatellite markers. J. Appl. Ichthyol. 2011, 27, 496–500. [Google Scholar] [CrossRef]
- Peng, Z.; Ludwig, A.; Wang, D.; Diogo, R.; Wei, Q.; He, S. Age and biogeography of major clades in sturgeons and paddlefishes (Pisces: Acipenseriformes). Mol. Phylogenetics Evol. 2007, 42, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Grande, L.; Bemis, W. Osteology and Phylogenetic Relationships of Fossil and Recent Paddlefishes (Polyodontidae) with Comments on the Interrelationships of Acipenseriformes. J. Vertebr. Paléontol. 1991, 11, 1–121. [Google Scholar] [CrossRef]
- Vasil’Ev, V.P.; Vasil’Eva, E.D.; Shedko, S.; Novomodny, G.V. How many times has polyploidization occurred during acipenserid evolution? New data on the karyotypes of sturgeons (Acipenseridae, Actinopterygii) from the Russian Far East. J. Ichthyol. 2010, 50, 950–959. [Google Scholar] [CrossRef]
- Iegorova, V.; Psenicka, M.; Lebeda, I.; Rodina, M.; Saito, T. Polyspermy produces viable haploid/diploid mosaics in sturgeon. Boil. Reprod. 2018, 99, 695–706. [Google Scholar] [CrossRef]
- Havelka, M.; Hulak, M.; Bailie, D.A.; Prodöhl, P.; Flajšhans, M. Extensive genome duplications in sturgeons: New evidence from microsatellite data. J. Appl. Ichthyol. 2013, 29, 704–708. [Google Scholar] [CrossRef]
- Havelka, M.; Bytyutskyy, D.; Symonová, R.; Ráb, P.; Flajšhans, M. The second highest chromosome count among vertebrates is observed in cultured sturgeon and is associated with genome plasticity. Genet. Sel. Evol. 2016, 48, 12. [Google Scholar] [CrossRef]
- Štěch, L.; Linhart, O.; Shelton, W.L.; Mims, S.D. Minimally Invasive Surgical Removal of Ovulated Eggs from Paddlefish. Aquac. Int. 1999, 7, 129–133. [Google Scholar] [CrossRef]
- Rónyai, A.; Váradi, L. World Animal Science: Production of Aquatic Animals; Elsevier Science: Amsterdam, The Netherland, 1995; Volume 1, pp. 95–108, Chapter 5. [Google Scholar]
- Shelton, W.L.; Mims, S.D.; Semmens, K.J.; Cuevas-Uribe, R. Artificial propagation of Paddlefish: An overview of developments. In Paddlefish: Ecological, Aquacultural, and Regulatory Challenges of Managing a Global Resource; Schooley, J.D., Scarnecchia, D.L., Eds.; American Fisheries Society: Bethesda, MD, USA, 2019; pp. 191–210. [Google Scholar]
- Heist, E.J.; Nicholson, E.H.; Sipiorski, J.T.; Keeney, D.B. Microsatellite Markers for the Paddlefish (Polyodon spathula). Conserv. Genet. 2002, 3, 205–207. [Google Scholar] [CrossRef]
- McQuown, E.C.; Sloss, B.L.; Sheehan, R.J.; Rodzen, J.; Tranah, G.J.; May, B. Microsatellite Analysis of Genetic Variation in Sturgeon: New Primer Sequences forScaphirhynchusandAcipenser. Trans. Am. Fish. Soc. 2000, 129, 1380–1388. [Google Scholar] [CrossRef]
- Shimizu, M.; Kosaka, N.; Shimada, T.; Nagahata, T.; Iwasaki, H.; Nagai, H.; Shiba, T.; Emi, M. Universal Fluorescent Labeling (UFL) Method for Automated Microsatellite Analysis. DNA Res. 2002, 9, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Darzynkiewicz, Z.; Juan, G. DNA Content Measurement for DNA Ploidy and Cell Cycle Analysis. Curr. Protoc. Cytom. 1997, 1, 7.5.1–7.5.24. [Google Scholar] [CrossRef]
- Tiersch, T.R.; Chandler, R.W.; Wachtel, S.S.; Elias, S. Reference standards for flow cytometry and application in comparative studies of nuclear DNA content. Cytometry 1989, 10, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Toth, B.; Varkonyi, E.; Hidas, A.; Meleg, E.E.; Váradi, L. Genetic analysis of offspring from intra- and interspecific crosses of Carassius auratus gibelio by chromosome and RAPD analysis. J. Fish Boil. 2005, 66, 784–797. [Google Scholar] [CrossRef]
- Earley, E.M. Chromosome preparations from monolayer cell cultures. J. Tissue Cult. Methods 1975, 1, 31–35. [Google Scholar] [CrossRef]
- LeVan, A.; Fredga, K.; Sandberg, A.A. Nomenclature for centromeric position on chromosomes. Hereditas 2009, 52, 201–220. [Google Scholar] [CrossRef]
- Holčik, J. General Introduction to Fishes, Acipenseriformes. In The freshwater fishes of Europe; Aula-Verlag: Wiesbaden, Germany, 1989; Volume 1, p. 469. [Google Scholar]
- Keszka, S.; Krzykawski, S. Morphometry of juvenile Russian sturgeon (Acipenser gueldenstaedtii Brandt et Ratzeburg, 1833) from fish farms. Acta Sci. Pol. Pisc. 2008, 7, 21–36. [Google Scholar]
- Hoover, J.J.; Boysen, K.A.; Murphy, C.E.; George, S.G. Morphological Variation in Juvenile Paddlefish; American Fisheries Society: Bethesda, MD, USA, 2009; pp. 157–171. [Google Scholar]
- Elliott, N.G.; Haskard, K.; Koslow, J.A. Morphometric analysis of orange roughy (Hoplostethus atlanticus) off the continental slope of southern Australia. J. Fish Boil. 1995, 46, 202–220. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics software package for education and data analysis. Pal. Elect. 2001, 4, 1–9. [Google Scholar]
- Fontana, F.; Lanfredi, M.; Rossi, R.; Bronzi, P.; Arlati, G. Karyotypic characterization of Acipenser gueldenstaedti with C-, AgNO 3, and fluorescence banding techniques. Ital. J. Zool. 1996, 63, 113–118. [Google Scholar] [CrossRef]
- Mims, S.D.; Shelton, W.L. Paddlefish Aquaculture; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; p. 63. [Google Scholar]
- Du, K.; Stöck, M.; Kneitz, S.; Klopp, C.; Woltering, J.M.; Adolfi, M.C.; Feron, R.; Prokopov, D.; Makunin, A.; Kichigin, I.; et al. The sterlet sturgeon genome sequence and the mechanisms of segmental rediploidization. Nat. Ecol. Evol. 2020, 4, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Li, S.; Cai, W.; Yang, H.; Jiang, X. Ploidy polymorphism and morphological variation among reciprocal hybrids by Megalobrama amblycephala×Tinca tinca. Aquaculture 2007, 270, 574–579. [Google Scholar] [CrossRef]
- Zhao, H.; Zou, H.; Lu, W. Ploidy polymorphism and morphological variation among reciprocal hybrids of Pseudosciaena crocea (♀) × Miichthys miiuy (♂). Aquac. Res. 2015, 47, 3390–3398. [Google Scholar] [CrossRef]
- Beck, M.; Biggers, C.J. Ploidy of Hybrids between Grass Carp and Bighead Carp Determined by Morphological Analysis. Trans. Am. Fish. Soc. 1983, 112, 808–811. [Google Scholar] [CrossRef]
- Cassani, J.R.; Caton, W.E.; Clark, B. Morphological comparisons of diploid and triploid hybrid grass carp, Ctenopharyngodon idella ♀ × Hypophthalmichthys nobilis ♂. J. Fish Boil. 1984, 25, 269–278. [Google Scholar] [CrossRef]
- Rohner, N.; Bercsényi, M.; Orbán, L.; Kolanczyk, M.E.; Linke, D.; Brand, M.; Nusslein-Volhard, C.; Harris, M.P. Duplication of fgfr1 Permits Fgf Signaling to Serve as a Target for Selection during Domestication. Curr. Boil. 2009, 19, 1642–1647. [Google Scholar] [CrossRef] [PubMed]
- Force, A.; Lynch, M.; Pickett, F.B.; Amores, A.; Yan, Y.L.; Postlethwait, J. Preservation of duplicate genes by complementary, degenerative mutations. Genetics 1999, 151, 1531–1545. [Google Scholar]
- Váradi, L.; Hidas, A.; Varkonyi, E.; Horvath, L. Interesting phenomena in hybridization of carp (Cyprinus carpio) and rosy barb (Barbus conchonius). Aquaculture 1995, 129, 211–214. [Google Scholar] [CrossRef]
- Arefjev, V.A.; Nikolaev, A.I. Cytological Analysis of the Reciprocal Hybrids between Low- and High-Chromosome Acipenserids, the Great Sturgeon, Huso huso(L.), and the Russian Sturgeon, Acipenser gueldenstaedtii Brandt. Cytologia 1991, 56, 495–502. [Google Scholar] [CrossRef]
- Schreier, A.; May, B.; Gille, D. Incidence of spontaneous autopolyploidy in cultured populations of white sturgeon, Acipenser transmontanus. Aquaculture 2013, 416, 141–145. [Google Scholar] [CrossRef]
- Fopp-Bayat, D.; Fopp-Bayat, R. Spontaneous gynogenesis in Siberian sturgeon Acipenser baeri Brandt. Aquac. Res. 2007, 38, 776–779. [Google Scholar] [CrossRef]
- Havelka, M.; Hulak, M.; Rodina, M.; Flajšhans, M. First evidence of autotriploidization in sterlet (Acipenser ruthenus). J. Appl. Genet. 2013, 54, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Gille, D.A.; Famula, T.R.; May, B.P.; Schreier, A. Evidence for a maternal origin of spontaneous autopolyploidy in cultured white sturgeon (Acipenser transmontanus). Aquaulture 2015, 435, 467–474. [Google Scholar] [CrossRef]
- Leal, M.J.; Van Eenennaam, J.P.; Schreier, A.; Todgham, A.E. Triploidy in white sturgeon (Acipenser transmontanus): Effects of acute stress and warm acclimation on physiological performance. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2019, 229, 10–17. [Google Scholar] [CrossRef]
- Van Eenennaam, J.P.; Fiske, A.J.; Leal, M.J.; Cooley-Rieders, C.; Todgham, A.E.; Conte, F.S.; Schreier, A. Mechanical shock during egg de-adhesion and post-ovulatory ageing contribute to spontaneous autopolyploidy in white sturgeon culture (Acipenser transmontanus). Aquaculture 2020, 515, 734530. [Google Scholar]
- Dorota, F.-B.; Pawel, W. Spontaneous and induced gynogenesis in sterlet Acipenser ruthenus Brandt. Caryologia 2007, 60, 315–318. [Google Scholar] [CrossRef][Green Version]
- Havelka, M.; Arai, K. Hybridization and Polyploidization in Sturgeon. In Sex Control in Aquaculture; Wiley: Hoboken, NJ, USA, 2018; pp. 669–687. [Google Scholar]
- Bronzi, P.; Chebanov, M.; Michaels, J.T.; Wei, Q.; Rosenthal, H.; Gessner, J. Sturgeon meat and caviar production: Global update 2017. J. Appl. Ichthyol. 2019, 35, 257–266. [Google Scholar] [CrossRef]
- Varga, M.; Berzi-Nagy, L.; Csukas, B.; Gyalog, G. Long-term dynamic simulation of environmental impacts on ecosystem-based pond aquaculture. Environ. Model. Softw. 2020. [Google Scholar] [CrossRef]

| Codes of Parents | Progeny with Expected Ploidy | Progeny with Spontaneous Alloploidy | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Female | Male | n | Range (pg) | Mean (pg) | Ploidy | n | Range (pg) | Mean (pg) | Ploidy |
| 6F15 | 2E75 | 11 | 6.96–8.1 | 7.55 ± 0.39 | 3n (SH) | 1 | 10.7 | - | 5n (LH) |
| 1748. | 2E75 | 9 | 7.19–8.42 | 7.59 ± 0.33 | 3n (SH) | 3 | 11.78–12.71 | 12.09 ± 0.44 | 5n (LH) |
| Ag03 | 596B | 22 | 4.39–7.79 | 6.56 ± 0.69 | 3n (SH) | 1 | 10.36 | - | 5n (LH) |
| Ag03 | 551F | 24 | 6.53–8.84 | 7.62 ± 0.55 | 3n (SH) | 1 | 13.17 | - | 5n (LH) |
| Ag03 | 6517 | 17 | 5.93–8.16 | 7.05 ± 0.51 | 3n (SH) | 8 | 10.87–12.87 | 11.48 ± 0.68 | 5n (LH) |
| Locus | Inheritance | Primer Sequences (5′-3′) | Annealing Temperature (°C) | Repeat Motif | |
|---|---|---|---|---|---|
| P. spathula | A. gueldenstaedtii | ||||
| Psp-28 | Disomic | Tetrasomic | F: Tail-CAAAGGCATCCCCTACCAC | 56 | GA |
| R: GCTGGACAAAAAGTATGGAGTGC | |||||
| Psp-29 | Tetrasomic | Disomic | F: Tail-GGGGTCTAATAAAATCCACCGTTC | 56 | GCAC |
| R: TTGCCTTGTGCTCTGTGTTCC | |||||
| Psp-32 | Disomic | Tetrasomic | F: Tail-AATGACTCAGTTGTGTGCTGC | 60 | GT |
| R: AAGTGTAGGGGAATCTCACCAG | |||||
| Spl-101 | Disomic | Tetrasomic | F: Tail-CCCTCCACTGGAAATTTGAC | 52 | TCTA |
| R: GCAATCAACAAG GTCTCTTTCA | |||||
| Tail (17bp) | - | - | ATTACCGCGGCTGCTGG | - | - |
| Russian Sturgeon | Hybrids | American Paddlefish | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Large Genome | Small Genome | Without Genome Data | ||||||||||||||
| Morphometric Characters | ||||||||||||||||
| TL | total length | 328.10 | (11.63) | - | 350.27 | (31.54) | - | 308.52 | (24.77) | - | 323.79 | (25.31) | - | 239.45 | (10.44) | - |
| L2 | standard length | 242.48 | (11.46) | a | 257.93 | (30.84) | b | 235.34 | (23.98) | a | 245.02 | (24.19) | ab | 196.94 | (10.82) | c |
| aA | preanal distance | 201.78 | (11.97) | a | 221.80 | (31.08) | a | 200.96 | (24.31) | b | 208.93 | (24.01) | b | 169.04 | (10.75) | c |
| aD | predorsal distance | 191.62 | (11.43) | ab | 204.93 | (30.73) | b | 187.47 | (23.37) | a | 194.07 | (23.84) | ab | 160.18 | (10.54) | c |
| aV | preventral distance | 161.32 | (11.82) | ab | 173.13 | (31.08) | b | 158.17 | (23.72) | a | 164.31 | (23.21) | a | 145.78 | (10.14) | c |
| Cfa | length of upper caudal lobe | 92.50 | (13.33) | a | 94.15 | (33.65) | b | 76.81 | (28.11) | b | 83.61 | (30.08) | b | 43.43 | (9.58) | c |
| Cfb | length of lower caudal lobe | 34.12 | (13.85) | a | 43.48 | (36.16) | b | 37.46 | (27.31) | b | 39.96 | (27.15) | b | 36.31 | (10.56) | c |
| C | head length | 61.26 | (8.81) | a | 81.97 | (27.5) | b | 84.93 | (21.28) | c | 86.48 | (18.79) | c | 147.43 | (9.37) | d |
| R | preorbital length | 26.56 | (10.29) | a | 38.30 | (26.6) | b | 44.62 | (19.51) | c | 44.41 | (18.59) | c | 78.99 | (9.76) | d |
| IP | length of pectoral fin | 41.85 | (10.95) | a | 44.01 | (26.08) | ab | 37.90 | (20.29) | b | 40.46 | (21.19) | b | 18.73 | (12.37) | d |
| IV | length of ventral fin | 22.60 | (13.81) | a | 28.01 | (33.02) | b | 23.78 | (25.21) | b | 25.51 | (25.57) | b | 14.47 | (12.56) | c |
| IA | length of anal fin | 26.81 | (15.39) | a | 30.56 | (34.13) | ab | 27.43 | (25.46) | b | 29.60 | (25.02) | b | 22.92 | (13.64) | c |
| ID | length of dorsal fin | 25.27 | (15.31) | a | 28.53 | (33.62) | ab | 25.91 | (24.09) | b | 27.06 | (24.07) | b | 23.53 | (10.76) | c |
| HC | head depth | 28.66 | (11.64) | a | 28.46 | (28.9) | b | 26.70 | (19.94) | b | 27.74 | (22.64) | b | 17.43 | (10.82) | c |
| h | caudal peduncle depth | 11.03 | (13.96) | a | 10.70 | (33.36) | b | 9.87 | (27.18) | b | 10.53 | (28.77) | b | 6.56 | (12.88) | c |
| iO | interorbital distance | 21.54 | (9.77) | a | 23.58 | (24.7) | a | 24.15 | (18.07) | b | 24.70 | (18.57) | b | 18.76 | (8.25) | c |
| rr | snout length | 28.46 | (9.5) | a | 40.80 | (25.95) | b | 46.21 | (21.66) | c | 46.04 | (19.29) | c | 79.02 | (9.63) | d |
| rc | snout tip–barbel base distance | 12.26 | (11.71) | a | 19.52 | (27.38) | b | 26.25 | (20.22) | c | 25.91 | (20.37) | c | 65.84 | (9.44) | d |
| SRr | head width on mouth | 27.70 | (10.47) | a | 27.63 | (23.9) | b | 27.52 | (19.04) | ab | 27.77 | (19.85) | ab | 20.86 | (10.59) | c |
| SRC | head width on barbel base | 18.12 | (11.82) | a | 17.90 | (21.81) | a | 19.01 | (19.42) | b | 19.30 | (21.61) | b | 17.10 | (8.5) | c |
| SO | mouth width | 13.66 | (12.58) | a | 16.32 | (28.87) | b | 17.31 | (22.31) | c | 17.59 | (21.09) | c | 16.64 | (11.3) | d |
| O | eye diameter | 6.20 | (8.93) | a | 6.79 | (13.48) | a | 6.29 | (14.14) | a | 6.38 | (14.85) | a | 3.80 | (11.07) | b |
| sL | scute length | 10.76 | (11.04) | a | 9.81 | (11.99) | a | 3.71 | (103.77) | b | 4.77 | (62.9) | b | - | - | - |
| Meristic parameters | ||||||||||||||||
| sDL | number of dorsal scutes | 13.06 | (12.71) | a | 11.8 | (40.79) | a | 3.9 | (125.53) | b | 5.37 | (98.16) | b | - | - | - |
| sLL | number of lateral scutes | 37.72 | (11.56) | a | 42.6 | (10.38) | b | 45.01 | (20.83) | bc | 47.18 | (15.87) | c | - | - | - |
| sVL | number of ventral scutes | 10.14 | (13.22) | a | 7.93 | (34.18) | b | 5.01 | (45.21) | c | 5.55 | (39.21) | c | - | - | - |
| nB | number of barbel | 4 | (0) | - | 3.47 | (28.57) | - | 2.76 | (40.26) | - | 2.87 | (36.24) | - | 2 | (0) | - |
| PIT ID | Genome Type | Psp-28 | Psp-29 | Psp-32 | Spl_101 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 6F15 | RS1 | 220 | 222 | 201 | 134 | 162 | 164 | 166 | 294 | 322 | 330 | 334 | ||||||||
| 174B | RS2 | 220 | 201 | 162 | 164 | 166 | 294 | 304 | 330 | |||||||||||
| Ag04 | RS3 | 220 | 201 | 162 | 164 | 166 | 294 | 304 | 330 | |||||||||||
| 2E7B | P1 | 260 | 211 | 225 | 196 | 274 | ||||||||||||||
| 596B | P2 | 260 | 267 | 211 | 225 | 196 | 274 | |||||||||||||
| 6517 | P3 | 260 | 267 | 211 | 218 | 225 | 196 | 274 | ||||||||||||
| D50F80 | SH | 220 | 260 | 201 | 211 | 225 | 134 | 164 | 196 | 330 | 334 | 274 | ||||||||
| D4BE1D | SH | 220 | 260 | 201 | 211 | 225 | 134 | 164 | 196 | 322 | 330 | 274 | ||||||||
| D4B65A | SH | 220 | 260 | 201 | 211 | 225 | 134 | 164 | 196 | 322 | 330 | 274 | ||||||||
| D509E7 | SH | 220 | 260 | 201 | 211 | 225 | 134 | 164 | 196 | 322 | 330 | 274 | ||||||||
| D4B071 | SH | 220 | 260 | 201 | 211 | 225 | 134 | 162 | 196 | 294 | 322 | 274 | ||||||||
| D4AE5E | SH | 220 | 260 | 201 | 211 | 225 | 162 | 166 | 196 | 294 | 330 | 274 | ||||||||
| D49E04 | SH | 220 | 222 | 260 | 201 | 211 | 225 | 162 | 166 | 196 | 294 | 322 | 274 | |||||||
| D4BA3F | SH | 220 | 222 | 260 | 201 | 211 | 225 | 162 | 166 | 196 | 322 | 330 | 274 | |||||||
| D4A57A | SH | 220 | 222 | 260 | 201 | 211 | 225 | 162 | 166 | 196 | 322 | 334 | 274 | |||||||
| D4C792 | SH | 220 | 260 | 201 | 211 | 225 | 164 | 166 | 196 | 294 | 304 | 274 | ||||||||
| D49F88 | SH | 220 | 260 | 201 | 211 | 225 | 164 | 166 | 196 | 304 | 274 | |||||||||
| D4C16A | SH | 220 | 267 | 201 | 218 | 225 | 164 | 166 | 196 | 294 | 304 | 274 | ||||||||
| D4C4AF | LH | 220 | 222 | 260 | 201 | 211 | 225 | 134 | 162 | 164 | 166 | 196 | 294 | 322 | 274 | |||||
| D4D979 | LH | 220 | 260 | 201 | 211 | 225 | 134 | 164 | 196 | 294 | 304 | 330 | 274 | |||||||
| D4C8FF | LH | 220 | 260 | 201 | 211 | 225 | 162 | 164 | 166 | 196 | 294 | 304 | 330 | 274 | ||||||
| D4B8EB | LH | 220 | 260 | 201 | 211 | 225 | 162 | 164 | 166 | 196 | 304 | 330 | 274 | |||||||
| D4B4DF | LH | 220 | 260 | 201 | 211 | 225 | 162 | 164 | 166 | 196 | 294 | 304 | 330 | 274 | ||||||
| D50340 | LH | 220 | 260 | 201 | 211 | 225 | 162 | 166 | 196 | 294 | 304 | 330 | 274 | |||||||
| D522C1 | LH | 220 | 267 | 201 | 211 | 225 | 162 | 164 | 166 | 196 | 294 | 304 | 330 | 274 | ||||||
| PIT ID of Individuals | DNA Content (pg) | Chromosome Number | Remarks |
|---|---|---|---|
| D4AF3A | 4.39 ± 0.47 | 156 ± 4 (3n) | interspecies triploid hybrid (SH/AP) |
| D4C16A | 7.49 ± 0.35 | 170 ± 2 (3n) | interspecies triploid hybrid (SH/AP) |
| D4B091 | 7.57 ± 0.39 | 170 ± 6 (3n) | interspecies triploid hybrid (SH/AP), paternal haploid cell lineage (1n = 66) |
| D4AE5E | 8.42 ± 0.71 | 160 ± 4 (3n) | interspecies triploid hybrid (SH/AP) |
| D52B1B | 6.85 ± 0.78 | 176 ± 8 (3n) | interspecies triploid hybrid (SH/AP), paternal haploid cell lineage (1n = 66–68) |
| D4D979 | 10.87 ± 0.65 | 308 ± 4 (5n) | pentaploid hybrid (LH/AAP), dicentric chromosome was found, paternal haploid cell lineage (1n = 68) |
| D4C9E4 | 12.36 ± 0.84 | 302 ± 4 (5n) | pentaploid hybrid (LH/AAP) |
| D522C1 | 12.87 ± 1.58 | 306 ± 4 (5n) | pentaploid hybrid (LH/AAP) |
| Polyodon spathula | 3.90 | 120 (2n) | Symonová et al. [8] |
| Acipenser gueldenstaedtii | 7.86–7.88 | 258 ± 4 (4n) | Fontana et al. [37] |
| Species/PIT ID | Number of Metacentric Plus Submetacentric Chromosomes | Number of Middle-Sized Acrocentric Chromosomes | No. of Large Acrocentric Chromosomes | Number of Microchromosomes | Total Chromosome Number |
|---|---|---|---|---|---|
| Polyodon spatula | 44 | 4 | 72 | 120 (PP) | |
| Acipenser gueldenstaedtii | 92 | - | 155 | 247 (AA) | |
| D52B1B | 70 | 10 | 2 | 94 | 176 (SH-AP) |
| D4D979 | 118 | 16 | 2 | 174 | 310 (LH-AAP) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Káldy, J.; Mozsár, A.; Fazekas, G.; Farkas, M.; Fazekas, D.L.; Fazekas, G.L.; Goda, K.; Gyöngy, Z.; Kovács, B.; Semmens, K.; et al. Hybridization of Russian Sturgeon (Acipenser gueldenstaedtii, Brandt and Ratzeberg, 1833) and American Paddlefish (Polyodon spathula, Walbaum 1792) and Evaluation of Their Progeny. Genes 2020, 11, 753. https://doi.org/10.3390/genes11070753
Káldy J, Mozsár A, Fazekas G, Farkas M, Fazekas DL, Fazekas GL, Goda K, Gyöngy Z, Kovács B, Semmens K, et al. Hybridization of Russian Sturgeon (Acipenser gueldenstaedtii, Brandt and Ratzeberg, 1833) and American Paddlefish (Polyodon spathula, Walbaum 1792) and Evaluation of Their Progeny. Genes. 2020; 11(7):753. https://doi.org/10.3390/genes11070753
Chicago/Turabian StyleKáldy, Jenő, Attila Mozsár, Gyöngyvér Fazekas, Móni Farkas, Dorottya Lilla Fazekas, Georgina Lea Fazekas, Katalin Goda, Zsuzsanna Gyöngy, Balázs Kovács, Kenneth Semmens, and et al. 2020. "Hybridization of Russian Sturgeon (Acipenser gueldenstaedtii, Brandt and Ratzeberg, 1833) and American Paddlefish (Polyodon spathula, Walbaum 1792) and Evaluation of Their Progeny" Genes 11, no. 7: 753. https://doi.org/10.3390/genes11070753
APA StyleKáldy, J., Mozsár, A., Fazekas, G., Farkas, M., Fazekas, D. L., Fazekas, G. L., Goda, K., Gyöngy, Z., Kovács, B., Semmens, K., Bercsényi, M., Molnár, M., & Patakiné Várkonyi, E. (2020). Hybridization of Russian Sturgeon (Acipenser gueldenstaedtii, Brandt and Ratzeberg, 1833) and American Paddlefish (Polyodon spathula, Walbaum 1792) and Evaluation of Their Progeny. Genes, 11(7), 753. https://doi.org/10.3390/genes11070753








