Genetics of Omega-3 Long-Chain Polyunsaturated Fatty Acid Metabolism and Meat Eating Quality in Tattykeel Australian White Lambs
Abstract
1. Introduction
2. Fatty Acids, Classifications and Functions
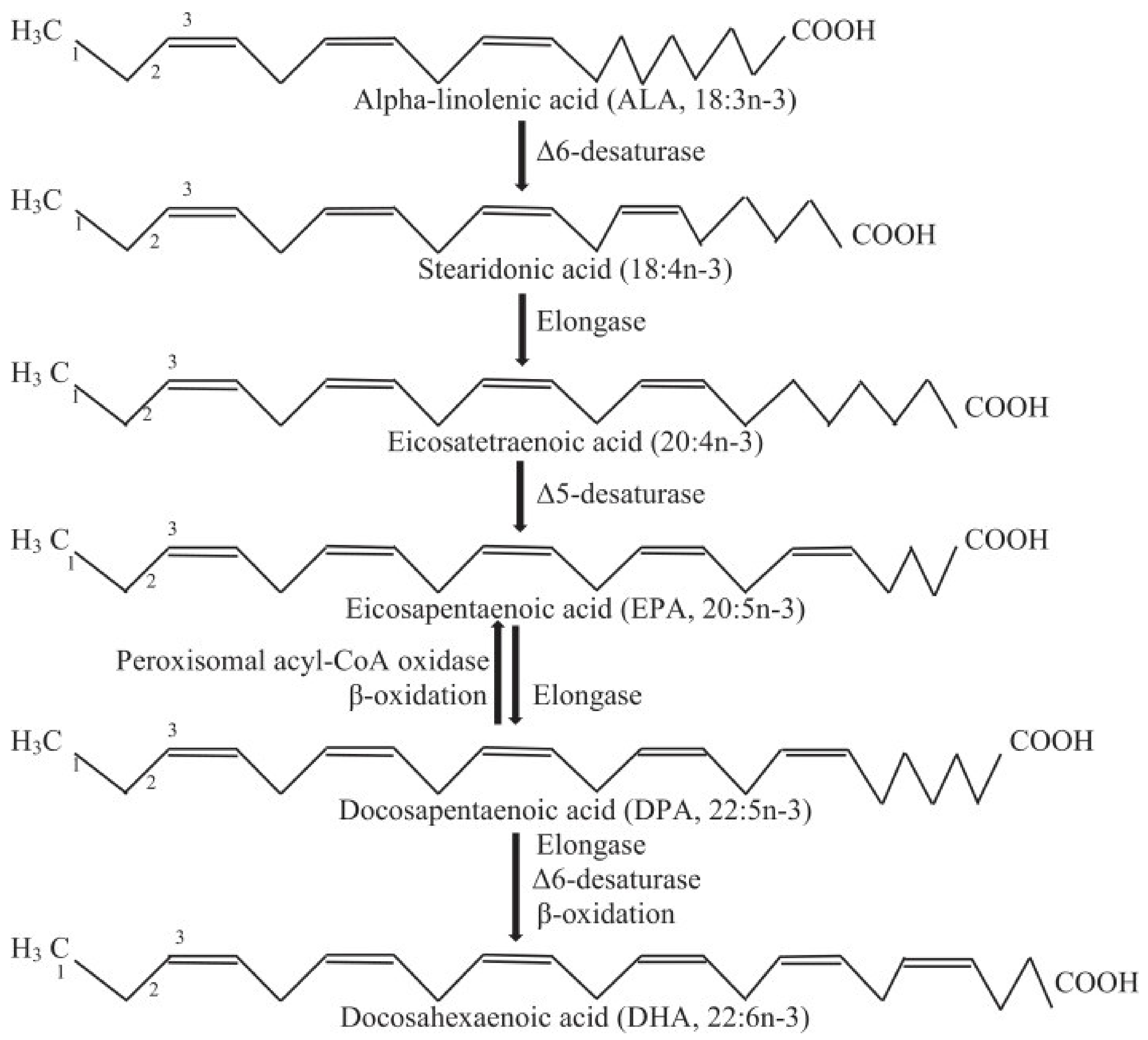
2.1. Omega-3 Long-Chain Polyunsaturated Fatty Acids
Fatty Acid Profile and Nutritional Value
2.2. Factors Affecting Fat Profile in Ruminant Muscle and Adipose Tissues
2.2.1. Biohydrogenation
2.2.2. Influence of Concentrate or Forage Finishing on Lamb Performance and Meat Quality
3. Lipogenic Genes and Associations with Genetic Selection for Meat Quality
3.1. Stearoyl-CoA Desaturase (SCD)
3.2. Fatty Acid Synthase (FASN)
3.3. Fatty Acid Binding Protein4 (FABP4)
3.4 Other Fat Related Genes
4. Meat Eating Quality
4.1. Influence of IMF on Lamb Eating Quality
4.1.1. Tenderness
4.1.2. Flavour
4.1.3. Juiciness
4.2. Fat Melting Point
5. Conclusions and Future Research
- Early selection decision tools for meat quality traits in TAW lambs are currently non-existent. Most reported selection programmes on fatty acid profile and meat quality traits in other sheep breeds are based on carcass data after the animals have been slaughtered and long gone. Pioneering studies using biopsy sampling of the Longissimus muscle in rams, ewes and lambs to directly determine n-3 LC-PUFA, IMF and FMP contents while the animals are young and alive for early selection and breeding purposes are needed.
- Published data on how parents selected for their high n-3 LC-PUFA, IMF and low FMP pass these genes to their offspring are currently non-existent in the TAW breed. Pioneer studies to estimate heritability values based on actual performance data and not estimated breeding values are recommended.
- SCD, FASN and FABP4 genes have been documented to exert some influence on carcass fat traits in other bovine and ovine breeds. No such data exist for the TAW breed. There is the need to sequence the FABP4, FASN and SCD genes to provide foundational data underpinning their roles in fatty acid metabolism unique to the TAW breed.
- In-depth feedlot growth studies are required for better understanding of the interactions between n-3 LC-PUFA oil diets, finishing performance, and carcass traits of TAW lambs to afford industry players the opportunity to utilise them for greater economic gains.
- A cost–benefit analysis of the implication of including n-3 LC-PUFA rich oil in feedlot finishing diets will be of immense industry significance to lamb producers, feed millers and meat processors.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OECD. Meat Consumption 2019. Available online: https://data.oecd.org/agroutput/meat-consumption.htm (accessed on 18 February 2020). [CrossRef]
- Rowe, J.B. The Australian sheep industry—Undergoing transformation. Anim. Prod. Sci. 2010, 50, 991–997. [Google Scholar] [CrossRef]
- MLA. Meat and Livestock Australia. Australia’s Sheep Meat Industry: Fresh Facts 2018. Available online: https://www.mla.com.au/globalassets/mla-corporate/prices--markets/documents/trends--analysis/fastfacts--maps/mla_sheep-fast-facts-2018.pdf (accessed on 4 October 2019).
- Milewski, S. Health-promoting properties of sheep products. Med. Weter. 2006, 62, 516–519. [Google Scholar]
- Nuernberg, K.; Fisher, A.; Nuernberg, G.; Ender, K.; Dannenberger, D. Meat quality and fatty acid composition of lipids in muscle and fatty tissue of Skudde lambs fed grass versus concentrate. Small Rum. Res. 2008, 74, 279–283. [Google Scholar] [CrossRef]
- FAO. Food and Agriculture Organization of the United Nations. Meat Quality 2014. Available online: http://www.fao.org/ag/againfo/themes/en/meat/quality_meat (accessed on 18 February 2020).
- MLA. Meat and Livestock Australia. Eating Quality 2020. Available online: https://www.mla.com.au/research-and-development/feeding-finishing-nutrition/eating-quality (accessed on 18 February 2020).
- Cardoso, C.; Afonso, C.; Bandarra, N.M. Seafood lipids and cardiovascular health. Nutrire 2016, 41, 7. [Google Scholar] [CrossRef]
- Scollan, N.D.; Dannenberger, D.; Nuernberg, K.; Richardson, L.; Mackintosh, S.; Hocquette, J.F.; Moloney, A.P. Enhancing the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2014, 97, 384–394. [Google Scholar] [CrossRef]
- De Smet, S.; Vossen, E. Meat: The balance between nutrition and health: A review. Meat Sci. 2016, 120, 145–156. [Google Scholar] [CrossRef]
- Gu, Z.; Suburu, J.; Chen, H.; Chen, Q.C. Mechanisms of omega-3 polyunsaturated fatty acids in prostate cancer prevention. Biomed. Res. Int. 2013, 10, 10. [Google Scholar] [CrossRef]
- Calviello, G.; Serini, S.; Piccioni, E. n-3 polyunsaturated fatty acids and the prevention of colorectal cancer: Molecular mechanisms involved. Curr. Med. Chem. 2009, 14, 3059–3069. [Google Scholar] [CrossRef]
- Chi, S.-C.; Tuan, H.-I.; Kang, Y.-N. Effects of polyunsaturated fatty acids on nonspecific typical dry eye disease: A systematic review and meta-analysis of randomized clinical trials. Nutrients 2019, 11, 942. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, H.; Kuang, L.; Meng, H.; Zhou, X. Omega-3 fatty acids for the treatment of depressive disorders in children and adolescents: A meta-analysis of randomized placebo-controlled trials. Child Adolesc. Psychiatry Ment. Health 2019, 13, 36. [Google Scholar] [CrossRef]
- Funaki, M. Saturated fatty acids and insulin resistance. J. Med. Inv. 2009, 56, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Sripetchwandee, J.; Chattipakorn, N.; Chattipakorn, S.C. Links between obesity-induced brain insulin resistance, brain mitochondrial dysfunction, and dementia. Front. Endocrinol. 2018, 9, 496. [Google Scholar] [CrossRef] [PubMed]
- Janssen, C.I.F.; Kiliaan, A.J. Long-chain polyunsaturated fatty acids (LCPUFA) from genesis to senescence: The influence of LCPUFA on neural development, aging and neurodegeneration. Prog. Lipid Res. 2014, 53, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Sheard, P.R.; Richardson, R.I.; Hughes, S.I.; Whittington, F.M. Fat deposition, fatty acid composition and meat quality: A review. Meat Sci. 2008, 78, 343–358. [Google Scholar] [CrossRef]
- Scollan, N.D.; Hocquette, J.F.; Nuernberg, K.; Dannenberger, D.; Richardson, I.; Moloney, A. Innovations in beef production systems that enhance the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2006, 74, 17–33. [Google Scholar] [CrossRef]
- Wood, J.D.; Richardson, R.I.; Nute, G.R.; Fisher, A.V.; Campo, M.M.; Kasapidou, E.; Sheard, P.R.; Enser, M. Effects of fatty acids on meat quality: A review. Meat Sci. 2004, 66, 21–32. [Google Scholar] [CrossRef]
- Webb, E.C.; O’Neill, H.A. The animal fat paradox and meat quality. Meat Sci. 2008, 80, 28–36. [Google Scholar] [CrossRef]
- Mwangi, F.W.; Charmley, E.; Gardiner, C.P.; Malau-Aduli, B.S.; Kinobe, R.T.; Malau-Aduli, A.E.O. Diet and genetics influence beef cattle performance and meat quality characteristics. Foods 2019, 8, 648. [Google Scholar] [CrossRef]
- Ladeira, M.M.; Schoonmaker, J.P.; Gionbelli, M.P.; Dias, J.C.O.; Gionbelli, T.R.S.; Carvalho, J.R.R.; Teixeira, P.D. Nutrigenomics and beef quality: A review about lipogenesis. Int. J. Mol. Sci. 2016, 17, 918. [Google Scholar] [CrossRef]
- Calder, P.C.; Adolph, M.; Deutz, N.E.; Grau, T.; Innes, J.K.; Klek, S.; Lev, S.; Mayer, K.; Michael-Titus, A.T.; Pradelli, L.; et al. Lipids in the intensive care unit: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2018, 37, 1–18. [Google Scholar] [CrossRef]
- Kenar, J.A.; Moser, B.R.; List, G.R. Naturally occurring fatty acids: Source, chemistry and uses. In Fatty Acids: Chemistry, Synthesis and Applications, 1st ed.; Ahmad, M.U., Ed.; AOCS Press, Elsevier Inc.: Oxford, UK, 2017; pp. 23–82. [Google Scholar]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health implications of high dietary omega-6 polyunsaturated fatty acids. J. Nutr. Metab. 2012, 2012. [Google Scholar] [CrossRef]
- Leyland, B.; Boussiba, S.; Khozin-Goldberg, I. A review of diatom lipid droplets. Biology 2020, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Sakamoto, T.; Ishii, A.; Yamaji, K.; Uemoto, Y.; Sasago, N.; Kobayashi, E.; Kobayashi, N.; Matsuhashi, T.; Maruyama, S.; et al. The g.841G>C SNP of FASN gene is associated with fatty acid composition in beef cattle. Anim. Sci. J. 2015, 86, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Melton, S.L.; Black, J.M.; Davis, G.W.; Backus, W.R. Flavour and selected chemical components of ground beef from steers backgrounded on pasture and fed corn up to 140 days. J. Food Sci. 1982, 47, 699–704. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, C. Effect of ω-3 polyunsaturated fatty acid-supplemented parenteral nutrition on inflammatory and immune function in postoperative patients with gastrointestinal malignancy. A meta-analysis of randomized control trials in China. Medicine 2018, 97, e0472. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef]
- Insel, P.; Ross, D.; McMahon, K.; Bernstein, M. Nutrition, 6th ed.; Jones & Bartlett Publishers: Massachusetts, MA, USA, 2018; Volume 6, p. 1002. [Google Scholar]
- Khan, W.A.; Chun-Mei, H.; Khan, N.; Iqbal, A.; Lyu, S.W.; Shah, F. Bioengineered plants can be a useful source of omega-3 fatty acids. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef]
- Yehuda, S.; Rabinovitz, S.; Mostofsky, D.I. Essential fatty acids and the brain: From infancy to aging. Neurobiol. Aging 2005, 26, 98–102. [Google Scholar] [CrossRef]
- Sampath, H.; Ntambi, J.M. The role of fatty acid desaturases in epidermal metabolism. Dermato-endocrinology 2011, 3, 62–64. [Google Scholar] [CrossRef]
- Tao, L.; Sun, T.; Magnuson, A.D.; Qamar, T.R.; Xin Gen Lei, X.G. Defatted microalgae-mediated enrichment of n–3 polyunsaturated fatty acids in chicken muscle is not affected by dietary selenium, vitamin E, or corn oil. J. Nutr. 2018, 148, 1547–1555. [Google Scholar] [CrossRef]
- Calder, P.C. Docosahexaenoic acid. Ann. Nutr. Metab. 2016, 69, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Very long chain omega-3 (n-3) fatty acids and human health. Eur. J. Lipid Sci. Technol. 2014, 116, 1280–1300. [Google Scholar] [CrossRef]
- Mayer, K.; Seeger, W. Fish oil in critical illness. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Tevar, R.; Jho, D.H.; Babcock, T.; Helton, W.S.; Espat, N.J. Omega-3 fatty acid supplementation reduces tumor growth and vascular endothelial growth factor expression in a model of progressive non-metastasizing malignancy. JPEN J. Parenter. Enteral. Nutr. 2002, 26, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Yaqoob, P. Understanding omega-3 polyunsaturated fatty acids. Postgrad. Med. 2009, 121, 148–157. [Google Scholar] [CrossRef]
- Weylandt, K.H. Docosapentaenoic acid derived metabolites and mediators—The new world of lipid mediator medicine in a nutshell. Eur. J. Pharm. 2016, 785, 108–115. [Google Scholar] [CrossRef]
- Kaur, G.; Cameron-Smith, D.; Garg, M.; Sinclair, A.J. Docosapentaenoic acid (22:5n-3): A review of its biological effects. Prog. Lipid Res. 2011, 50, 28–34. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Muntner, P. Heart disease and stroke statistics-2017 update: A report from the American Heart Association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics 2016. CA Cancer J. Clin. 2017, 66, 7–30. [Google Scholar] [CrossRef]
- Nichols, P.D.; Kitessa, S.M.; Abeywardena, M. Commentary on a trial comparing krill oil versus fish oil. Lipids Health Dis. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Lemaitre, R.N.; King, I.B.; Song, X.; Spiegelman, D.; Sacks, F.M.; Rimm, E.B.; Siscovick, D.S. Circulating long-chain ω-3 fatty acids and incidence of congestive heart failure in older adults: The cardiovascular health study: A cohort study. Ann. Intern. Med. 2011, 155, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, I.J.; Pockley, G.; Galloway, J.H.; Greaves, M.; Preston, E. The effects of dietary omega-3 polyunsaturated fatty acids on erythrocyte membrane phospholipids, erythrocyte deformability and blood viscosity in healthy volunteers. Atherosclerosis 1985, 55, 267–281. [Google Scholar] [CrossRef]
- Watanabe, Y.; Tatsuno, I. Omega-3 polyunsaturated fatty acids for cardiovascular diseases: Present, past and future. Expert Rev. Clin. Pharmacol. 2017, 10, 865–873. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Abeywardena, M.Y.; Head, R.J. Long chain n−3 polyunsaturated fatty acids and blood vessel function. Cardiovasc. Res. 2001, 52, 361–371. [Google Scholar] [CrossRef]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef]
- Koletzko, B.; Lien, E.; Agostoni, C.; Bohles, H.; Campoy, C.; Cetin, I.; Decsi, T.; Dudenhausen, J.W.; Dupont, C. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: Review of current knowledge and consensus recommendations. J. Perinat. Med. 2008, 36, 5–14. [Google Scholar] [CrossRef]
- Astorg, P. Dietary n-6 and n-3 polyunsaturated fatty acids and prostate cancer risk: A review of epidemiological and experimental evidence. Cancer Causes Control 2004, 15, 367–386. [Google Scholar] [CrossRef]
- Leitzmann, M.; Stampfer, M.; Michaud, D.; Augustsson, K.; Colditz, G.; Willett, W.; Giovannucci, E. Dietary intake of n-3 and n-6 fatty acids and the risk of prostate cancer. Am. J. Clin. Nutr. 2004, 80, 204–216. [Google Scholar] [CrossRef]
- Welch, A.A.; Shakya-Shrestha, S.; Lentjes, M.A.H.; Wareham, N.J.; Khaw, K.T. Dietary intake and status of n-3 polyunsaturated fatty acids in a population of fish-eating and non-fish-eating meat-eaters, vegetarians, and vegans and the precursor-product ratio of α-linolenic acid to long-chain n-3 polyunsaturated fatty acids: Results from the EPIC-Norfolk cohort. Am. J. Clin. Nutr. 2010, 92, 1040–1051. [Google Scholar] [PubMed]
- Gould, J.F.; Smithers, L.G.; Makrides, M. The effect of maternal omega-3 (n-3) LCPUFA supplementation during pregnancy on early childhood cognitive and visual development: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. New evidence that omega-3 fatty acids have a role in primary prevention of coronary heart disease. J. Public Health Emerg. 2017, 1, 35. [Google Scholar] [CrossRef]
- Certik, M.; Shimizu, S. Biosynthesis and regulation of microbial polyunsaturated fatty acid production. J. Biosci. Bioeng. 1999, 87, 1–14. [Google Scholar] [CrossRef]
- Jacobs, D.R.; Ruzzin, J.; Lee, D.H. Environmental pollutants: Downgrading the fish food stock affects chronic disease risk. J. Intern. Med. 2014, 276, 240–242. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty acid composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Mori, T.A. Omega-3 fatty acids and cardiovascular disease: Epidemiology and effects on cardiometabolic risk factors. Food Funct. 2014, 5, 2004–2019. [Google Scholar] [CrossRef]
- Brenna, J.T.; Salem, N.; Sinclair, A.J.; Cunnane, S.C. Alpha-linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot. Essent. Fatty Acids 2009, 80, 85–91. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Taylor, D.S.; Yu-Poth, S.; Huth, P.; Moriarty, K.; Fishell, V.; Hargrove, R.L.; Zhao, G.X.; Etherton, T.D. Polyunsaturated fatty acids in the food chain in the United States. Am. J. Clin. Nutr. 2000, 71, 179S–188S. [Google Scholar] [CrossRef]
- Lum, K.K.; Kim, J.; Lei, X.G. Dual potential of microalgae as a sustainable biofuel feedstock and animal feed. J. Anim. Sci. Biotechnol. 2013, 4. [Google Scholar] [CrossRef]
- Gregory, M.K.; Geier, M.S.; Gibson, R.A.; James, M.J. Functional characterization of the chicken fatty acid elongases. J. Nutr. 2013, 143, 12–16. [Google Scholar] [CrossRef]
- Yazdi, P.G. A review of the biologic and pharmacologic role of docosapentaenoic acid n-3. F1000Research 2013, 2, 256. [Google Scholar] [CrossRef][Green Version]
- Sun, Q.; Ma, J.; Campos, H.; Rexrode, K.M.; Albert, C.M.; Mozaffarian, D.; Hu, F.B. Blood concentrations of individual long-chain n–3 fatty acids and risk of nonfatal myocardial infarction. Am. J. Clin. Nutr. 2008, 88, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Reinders, I.; Virtanen, J.K.; Brouwer, I.A.; Tuomainen, T.P. Association of serum n-3 polyunsaturated fatty acids with C-reactive protein in men. Eur. J. Clin. Nutr. 2002, 66, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Rissanen, T.; Voutilainen, S.; Nyyssonen, K.; Lakka, T.A.; Salonen, J.T. Fish oil–derived fatty acids, docosahexaenoic acid and docosapentaenoic acid, and the risk of acute coronary events: The Kuopio ischaemic heart disease risk factor study. Circulation 2000, 102, 2677–2679. [Google Scholar] [CrossRef]
- Aase, K.; Ernkvist, M.; Ebarasi, L.; Jakobsson, L.; Majumdar, A.; Yi, C.; Birot, O.; Ming, Y.; Kvanta, A.; Edholm, D.; et al. Angiomotin regulates endothelial cell migration during embryonic angiogenesis. Genes Dev. 2007, 21, 2055–2068. [Google Scholar] [CrossRef] [PubMed]
- Phang, M.; Garg, M.L.; Sinclair, A.J. Inhibition of platelet aggregation by omega-3 polyunsaturated fatty acids is gender specific—Redefining platelet response to fish oils. Prostagland. Leukot. Essen. Fatty Acids. 2009, 81, 35–40. [Google Scholar] [CrossRef]
- Augustsson, K.; Michaud, D.S.; Rimm, E.B.; Leitzman, M.F.; Stampfer, M.J.; Walter, C.W.; Giovannucci, E. A prospective study of intake of fish and marine fatty acids and prostate cancer. Cancer Epidemiol. Biomark. Prev. 2003, 12, 64–67. [Google Scholar]
- Kanayasu-Toyoda, T.; Morita, I.; Murota, S.I. Docosapentaenoic acid (22:5, n-3), an elongation metabolite of eicosapentaenoic acid (20:5, n-3), is a potent stimulator of endothelial cell migration on pretreatment in vitro. Prostagland. Leukot. Essen. Fatty Acids. 1996, 54, 319–325. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Y.; Liang, Y.; Tian, X.; Peng, C.; Ma, K.Y.; Liu, J.; Huang, Y.; Chen, Z.Y. DPA n-3, DPA n-6 and DHA improve lipoprotein profiles and aortic function in hamsters fed a high cholesterol diet. Atherosclerosis 2012, 221, 397–404. [Google Scholar] [CrossRef]
- Malau-Aduli, A.E.O.; Holman, B.W.B. Molecular genetics-nutrition interactions in ruminant fatty acid metabolism and meat quality. In Molecular and Quantitative Animal Genetics, 1st ed.; Khatib, H., Ed.; John Wiley & Sons Inc.: New York, NY, USA, 2015; pp. 197–214. [Google Scholar]
- Warren, H.E.; Scollan, N.D.; Enser, M.; Hughes, S.I.; Richardson, R.I.; Wood, J.D. Effects of breed and a concentrate or grass silage diet on beef quality in cattle of 3 ages. I: Animal performance, carcass quality and muscle fatty acid composition. Meat Sci. 2008, 78, 256–269. [Google Scholar] [CrossRef] [PubMed]
- National Health and Medical Research Council, Australian Government Department of Health and Ageing. Nutrient Reference Values for Australia and New Zealand. Canberra: National Health and Medical Research Council. 2006. Available online: https://www.nhmrc.gov.au/sites/default/files/images/nutrient-refererence-dietary-intakes.pdf (accessed on 25 May 2020).
- Le, H.V.; Nguyen, D.V.; Nguyen, Q.V.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Fatty acid profiles of muscle, liver, heart and kidney of Australian prime lambs fed different polyunsaturated fatty acids enriched pellets in a feedlot system. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Ponnampalam, E.N.; Bunter, K.L.; Pearce, K.M.; Mortimer, S.I.; Pethick, D.W.; Ball, A.J.; Hopkins, D.L. Sources of variation of health claimable long chain omega-3 fatty acids in meat from Australian lamb slaughtered at similar weights. Meat Sci. 2014, 96, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation of European Union. Statement on a conceptual framework for the risk assessment of certain food additives re-evaluated under Commission Regulation (EU) No 257/20102014. European Food Safety Authority Panel on Food additives and Nutrient Sources added to Food (ANS). EFSA J. 2014, 6, 3697. [Google Scholar]
- World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases; Technical Report Series 797; WHO: Geneva, Switzerland, 1990. [Google Scholar]
- Simopoulos, A.P. Evolutionary aspects of diet: The omega-6/omega-3 ratio and the brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Expt. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef]
- Corsinovi, L.; Biasi, F.; Poli, G.; Leonarduzzi, G.; Isaia, G. Dietary lipids and their oxidized products in Alzheimer’s disease. Mol. Nutr. Food Res. 2011, 55, S161–S172. [Google Scholar] [CrossRef]
- Pighin, D.; Pazos, A.; Chamorro, V.; Paschetta, F.; Cunzolo, S.; Godoy, F.; Messina, V.; Pordomingo, A.; Grigioni, G. A contribution of beef to human health: A review of the role of the animal production systems. Sci. World J. 2016. [CrossRef]
- Henderson, G.; Cox, F.; Ganesh, S.; Jonker, A.; Young, W.; Global, R.C.C.; Janssen, P.H. Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Morgavi, D.; Forano, E.; Martin, C.; Newbold, C. Microbial ecosystem and methanogenesis in ruminants. Animal 2010, 4, 1024–1036. [Google Scholar] [CrossRef] [PubMed]
- Doreau, M.; Meynadier, A.; Fievez, V.; Ferlay, A. Ruminal Metabolism of Fatty Acids: Modulation of Polyunsaturated, Conjugated and Trans fatty Acids in Meat and Milk In Handbook of Lipids in Human Function: Fatty Acids; Watson, F.F., de Meester, F., Eds.; Academic Press and AOC Press: San Diego, CA, USA, 2016; pp. 521–542. [Google Scholar]
- Demeyer, D.I.; Mertens, B.; De Smet, S.; Ulens, M. Mechanisms linking colorectal cancer to the consumption of processed red meat: A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, 2747–2766. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.C.; Wallace, R.J.; Moate, P.J.; Mosley, E.E. Recent advances in biohydrogenation of unsaturated fatty acids within the rumen microbial ecosystem. J. Anim. Sci. 2008, 86, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Beam, T.M.; Jenkins, T.C.; Moate, P.J.; Kohn, K.A.; Palmquist, D.L. Effects of amount and source of fat on the rates of lipolysis and biohydrogenation of fatty acids in ruminal contents. J. Dairy Sci. 2000, 83, 2564–2573. [Google Scholar] [CrossRef]
- Buccioni, A.; Decandia, M.; Minieri, S.; Molle, G.; Cabiddu, A. Lipid metabolism in the rumen: New insights on lipolysis and biohydrogenation with an emphasis on the role of endogenous plant factors. Anim. Feed Sci. Tech. 2012, 174, 1–25. [Google Scholar] [CrossRef]
- Van Nevel, C.J.; Demeyer, D.I. Influence of pH on lipolysis and biohydrogenation of soybean oil by rumen contents in vitro. Reprod. Nutr. Dev. 1996, 36, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Dewanckele, L.; Vlaeminck, B.; Hernandez-Sanabria, E.; Ruiz-Gonzalez, A.; Dubruyne, S.; Jeyanathan, J.; Frievez, V. Rumen biohydrogenation and microbial community changes upon early life supplementation of 22:6n-3 enriched microalgae to goats. Front. Microbiol. 2018, 9, 573. [Google Scholar] [CrossRef]
- Edwards, H.D.; Shelver, W.L.; Choi, S.; Nisbet, D.J.; Krueger, N.A.; Anderson, R.C.; Smith, S.B. Immunogenic inhibition of prominent ruminal bacteria as a means to reduce lipolysis and biohydrogenation activity in vitro. Food Chem. 2017, 218, 372–377. [Google Scholar] [CrossRef]
- Palmquist, D.L.; Lock, A.L.; Shingfield, K.J.; Bauman, D.E. Biosynthesis of conjugated linoleic acid in ruminants and humans. Adv. Food Nutr. Res. 2005, 50, 179–217. [Google Scholar]
- Li, D.; Wang, J.Q.; Bu, D.P. Ruminal microbe of hydrogenation of trans- vaccenic acid to stearic acid in vitro. BMC Res. Notes 2012, 5. [Google Scholar] [CrossRef]
- Ferlay, A.; Bernard, L.; Meynadier, A.; Malpuech-Brugère, C. Production of trans and conjugated fatty acids in dairy ruminants and their putative effects on human health: A review. Biochimie 2017, 141, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Bickerstaffe, D.; Noakes, D.E.; Annison, E.F. Quantitative aspects of fatty acid biohydrogenation, absorption and transfer into milk fat in the lactating goat, with special reference to the cis- and trans-isomers of octadecenoate and linoleate. Biochem. J. 1972, 130, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Griinari, J.M.; Bauman, D.E. Biosynthesis of conjugated linoleic acid and its incorporation into meat and milk in ruminants. In Advances in Conjugated Linoleic Acid Research; AOCS Press: Urbana, IL, USA, 1999; pp. 180–220. [Google Scholar]
- Demeyer, D.I. Quantitative aspects of microbial metabolism in the rumen and hindgut. In Rumen Microbial Metabolism and Ruminant Digestion, Institut Nationale de la Recherche Agronomique; Jouruay, J.-P., Ed.; INRA: Paris, France, 1991; Volume 7, pp. 217–237. [Google Scholar]
- Moss, A.R.; Jouany, J.-P.; Newbold, J. Methane production by ruminants: Its contribution to global warming. Ann. Zootech. 2000, 49, 231–253. [Google Scholar] [CrossRef]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; de Los Reyes-Gavilán, C.G.; Salazar, N. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed]
- Fruet, A.P.B.; Stefanello, F.S.; Trombetta, F.; De Souza, A.N.M.; Junior, A.G.R.; Tonetto, C.J.; Flores, J.L.C.; Scheibler, R.B.; Bianchi, R.M.; Pacheco, P.S.; et al. Growth performance and carcass traits of steers finished on three different systems including legume–grass pasture and grain diets. Animal 2019, 13, 1552–1562. [Google Scholar] [CrossRef]
- Redfearn, D.D.; Venuto, B.C.; Pitman, W.D.; Alison, M.W.; Ward, J.D. Cultivar and environmental effects on annual ryegrass forage yield, yield distribution, and nutritive value. Crop Sci. 2002, 42, 2049–2054. [Google Scholar] [CrossRef]
- Buxton, D.; Hornstein, J.S.; Wedin, W.F.; Marten, G.C. Forage quality in stratified canopies of alfalfa, birdsfoot trefoil and red clover. Crop Sci. 1985, 25, 273–279. [Google Scholar] [CrossRef]
- Sleugh, B.; Moore, K.J.; George, J.R.; Brummer, E.C. Binary legume-grass mixtures improve forage yield, quality, and seasonal distribution. Agron. J. 2000, 92, 24–29. [Google Scholar] [CrossRef]
- Fruet, A.P.B.; Stefanello, F.S.; Júnior, A.G.R.; De Souza, A.N.M.; Tonetto, C.J.; Nörnberg, J.L. Whole grains in the finishing of culled ewes in pasture or feedlot: Performance, carcass characteristics and meat quality. Meat Sci. 2016, 113, 97–103. [Google Scholar] [CrossRef]
- Aldai, N.; Dugan, M.E.R.; Kramer, J.K.G.; Martinez, A.; Lopez-Campos, O.; Mantecon, A.R.; Osoro, K. Length of concentrate finishing affects the fatty acid composition of grass-fed and genetically lean beef: An emphasis on trans-18:1 and conjugated linoleic acid profiles. Animal 2011, 5, 1643–1652. [Google Scholar] [CrossRef]
- Demirel, G.; Ozpinar, H.; Nazli, B.; Keser, O. Fatty acids of lamb meat from two breeds fed different forage: Concentrate ratio. Meat Sci. 2006, 72, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.D.; Kerth, C.R.; Branden, K.; Rankins, D.L.; Kriese-Anderson, L.A.; Prevatt, J.W. Finishing steers on winter annual ryegrass (Lolium multiflorum Lam.) with varied levels of corn supplementation I: Effects on animal performance, carcass traits, and forage quality. J. Anim. Sci. 2009, 87, 2690–2699. [Google Scholar] [CrossRef] [PubMed]
- Duckett, S.K.; Neel, J.P.S.; Lewis, R.M.; Fontenot, J.P.; Clapham, W.N. Effects of forage species or concentrate finishing on animal performance, carcass and meat quality. J. Anim. Sci. 2013, 91, 1454–1467. [Google Scholar] [CrossRef] [PubMed]
- Realini, C.; Duckett, S.K.; Brito, G.; Dalla Rizza, M.; de Mattos, D. Effect of pasture vs. concentrate feeding with or without antioxidants on carcass characteristics, fatty acid composition, and quality of Uruguayan beef. Meat Sci. 2004, 66, 567–577. [Google Scholar] [CrossRef]
- Raes, K.; De Smet, S.; Demeyer, D. Effect of dietary fatty acids on incorporation of long chain polyunsaturated fatty acids and conjugated linoleic acid in lamb, beef and pork meat: A review. Anim. Feed Sci. Tech. 2004, 113, 199–221. [Google Scholar] [CrossRef]
- Arelovich, H.M.; Marinissen, J.; Gardner, B.A.; Martinez, M.F.; Bravo, R.D. Effects of oats grain supplements on performance, rumen parameters and composition of beef from cattle grazing oats pasture. Anim. Prod. Sci. 2017, 57, 665–674. [Google Scholar] [CrossRef]
- Rovadoscki, G.A.; Pertile, S.F.N.; Alvarenga, A.B.; Cesar, A.S.M.; Pértille, F.; Petrini, J.; Franzo, V.; Soares, W.V.B.; Morota, G.; Spangler, M.L.; et al. Estimates of genomic heritability and genome-wide association study for fatty acids profile in Santa Inês sheep. BMC Genom. 2018, 19, 375. [Google Scholar] [CrossRef]
- Tiezzi, F.; Parker-Gaddis, K.L.; Cole, J.B.; Clay, J.S.; Maltecca, C. A genome-wide association study for clinical mastitis in first parity US Holstein cows using single-step approach and genomic matrix re-weighting procedure. PLoS ONE 2015, 10, e114919. [Google Scholar] [CrossRef]
- Goddard, M.E.; Hayes, B.J. Genomic selection. J. Anim. Breed. Genet. 2007, 124, 323–330. [Google Scholar] [CrossRef]
- Bolormaa, S.; Pryce, J.E.; Kemper, K.; Savin, K.; Hayes, B.J.; Barendse, W.; Zhang, Y.; Reich, C.M.; Mason, B.A.; Bunch, R.J.; et al. Accuracy of prediction of genomic breeding values for residual feed intake and carcass and meat quality traits in Bos taurus, Bos indicus, and composite beef cattle. J. Anim. Sci. 2013, 91, 3088–3104. [Google Scholar] [CrossRef]
- Van Raden, P.M. Efficient methods to compute genomic predictions. J. Dairy Sci. 2008, 91, 4414–4423. [Google Scholar] [CrossRef]
- Meuwissen, T.H.; Hayes, B.J.; Goddard, M.E. Prediction of total genetic value using genome-wide dense marker maps. Genetics 2001, 157, 1819–1829. [Google Scholar] [PubMed]
- Casas, E.; White, S.N.; Wheeler, T.L.; Shackelford, S.D.; Koohmaraie, M.; Riley, D.G.; Chase, C.C.; Johnson, D.D.; Smith, T.P.L. Effects of calpastatin and mu-calpain markers in beef cattle on tenderness traits. J. Anim. Sci. 2006, 84, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Hayes, B.J.; Bowman, P.J.; Chamberlain, A.C.; Goddard, M.E. Genomic selection in dairy cattle: Progress and challenges. J. Dairy Sci. 2009, 92, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Maharani, D.; Jung, Y.; Jung, W.Y.; Jo, C.; Ryoo, S.H.; Lee, S.H.; Yeon, S.H.; Lee, J.H. Association of five candidate genes with fatty acid composition in Korean cattle. Mol. Biol. Rep. 2012, 39, 6113–6121. [Google Scholar] [CrossRef]
- Bhuiyan, M.S.A.; Yu, S.L.; Jeon, J.T.; Yoon, D.; Cho, Y.M.; Park, E.W.; Kim, N.K.; Kim, K.S.; Lee, J.H. DNA polymorphisms in SREBF1 and FASN genes affect fatty acid composition in Korean cattle (Hanwoo). Asian-Aust. J. Anim. Sci. 2009, 22, 765–773. [Google Scholar] [CrossRef]
- Esteves, C.; Livramento, K.G.; Paiva, L.V.; Peconick, A.P.; Garcia, I.F.F.; Garbossa, C.A.P.; Faria, P.B. The polymorphisms of genes associated with the profile of fatty acids of sheep. Arq. Bras. Med. Vet. Zootec. 2019, 71, 303–313. [Google Scholar] [CrossRef]
- Gu, M.; Cosenza, G.; Iannaccone, M.; Macciotta, N.P.P.; Guo, Y.; Di Stasio, L.; Pauciullo, A. The single nucleotide polymorphism g.133A>C in the stearoyl CoA desaturase gene (SCD) promoter affects gene expression and quali-quantitative properties of river buffalo milk. J. Dairy Sci. 2019, 102, 442–451. [Google Scholar] [CrossRef]
- Paton, C.M.; Ntambi, J.M. Biochemical and physiological function of stearoyl-CoA desaturase. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E28–E37. [Google Scholar] [CrossRef]
- Mannen, H. Identification and utilization of genes associated with beef qualities. Anim. Sci. J. 2011, 82, 1–7. [Google Scholar] [CrossRef]
- Milanesi, E.; Nicoloso, L.; Crepaldi, P. Stearoyl CoA desaturase (SCD) gene polymorphisms in Italian cattle breeds. J. Anim. Breed. Genet. 2008, 125, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Lengi, A.J.; Corl, B.A. Identification and characterization of a novel bovine stearoyl-coA desaturase isoform with homology to human SCD5. Lipids 2007, 42, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Ntambi, J.M. Regulation of stearoyl-CoA desaturase genes: Role in cellular metabolism and preadipocyte differentiation. Biochem. Biophys. Res. Commun. 1999, 266, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B.; Gill, C.A.; Lunt, D.K.; Brooks, M.A. Regulation of fat and fatty acid composition in beef cattle. Asian-Aust. J. Anim. Sci. 2009, 22, 1225–1233. [Google Scholar] [CrossRef]
- Bauman, D.E.; Mather, I.H.; Wall, R.J.; Lock, A.L. Major advances associated with the biosynthesis of milk. J. Dairy Sci. 2006, 89, 1235–1243. [Google Scholar] [CrossRef]
- Guillou, H.; Zadravec, D.; Martin, P.G.P.; Jacobsson, A. The key roles of elongases and desaturases in mammalian fatty acid metabolism: Insights from transgenic mice. Prog. Lipid Res. 2010, 49, 186–199. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef]
- Reynolds, C.M.; Roche, H.M. Conjugated linoleic acid and inflammatory cell signaling. Prost. Leukot. Essent. Fatty Acids 2010, 82, 199–204. [Google Scholar] [CrossRef]
- Calvo, J.H.; González-Calvo, L.; Dervishi, E.; Blanco, M.; Iguácel, L.P.; Sarto, P.; Pérez-Campo, F.M.; Serrano, M.; Bolado-Carrancio, A.; Rodríguez-Rey, J.C.; et al. A functional variant in the stearoyl-CoA desaturase (SCD) gene promoter affects gene expression in ovine muscle. Livest. Sci. 2019, 219, 62–70. [Google Scholar] [CrossRef]
- Carta, A.; Casu, S.; Usai, M.; Addis, M.; Fiori, M.; Fraghi, A.; Miari, S.; Mura, L.; Piredda, G.; Schibler, L. Investigating the genetic component of fatty acid content in sheep milk. Small Rum. Res. 2008, 79, 22–28. [Google Scholar] [CrossRef]
- Armstrong, E.; Ciappesoni, G.; Iriarte, W.; Da Silva, C.; Macedo, F.; Navajas, E.A.; Brito, G.; San Julián, R.; Gimeno, D.; Postiglioni, A. Novel genetic polymorphisms associated with carcass traits in grazing Texel sheep. Meat Sci. 2018, 145, 202–208. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, M.; Gutiérrez-Gil, B.; García-Gámez, E.; Arranz, J.J. Genetic variability of the stearoyl-CoA desaturase gene in sheep. Mol. Cell. Probes 2009, 23, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; Ntambi, J.M. Role of stearoyl-coenzyme A desaturase in lipid metabolism. Prostaglandins Leukot. Essent. Fatty Acids 2003, 68, 113–121. [Google Scholar] [CrossRef]
- Dervishi, E.; Serrano, C.; Joy, M.; Serrano, M.; Rodellar, C.; Calvo, J.H. Effect of the feeding system on the fatty acid composition, expression of the Δ9 -desaturase, Peroxisome Proliferator-Activated Receptor Alpha, Gamma, and Sterol Regulatory Element Binding Protein 1 genes in the semitendinous muscle of light lambs of the Rasa Aragonesa breed. BMC Vet. Res. 2010, 6, 6–40. [Google Scholar]
- Roy, R.; Gautier, M.; Hayes, H.; Laurent, P.; Osta, R. Assignment of the fatty acid synthase (FASN) gene to bovine chromosome 19 (19q22) by in situ hybridization and confirmation by somatic cell hybrid mapping. Cytogenet. Cell. Genet. 2001, 93, 141–142. [Google Scholar] [CrossRef] [PubMed]
- Chirala, S.S.; Chang, H.; Matzuk, M.; Abu-Elheiga, L.; Mao, J.; Mahson, K.; Finegold, M.; Wakil, S.J. Fatty acid synthesis is essential in embryonic development: Fatty acid synthase null mutants and most of the heterozygotes die in utero. Proc. Natl. Acad. Sci. USA 2003, 100, 6358–6363. [Google Scholar] [CrossRef]
- Zhang, S.; Knight, T.J.; Reecy, J.M.; Beitz, D.C. DNA polymorphisms in bovine fatty acid synthase are associated with beef fatty acid composition. Anim. Genet. 2008, 39, 62–70. [Google Scholar] [CrossRef]
- Morris, C.A.; Cullen, N.G.; Glass, B.G.; Hyndman, D.L.; Manley, T.R.; Hickey, S.M.; McEwan, J.S.; Pitchford, W.S.; Bottema, C.D.K.; Lee, M.A.H. Fatty acid synthase effects on bovine adipose fat and milk fat. Mamm. Genom. 2007, 18, 64–74. [Google Scholar] [CrossRef]
- Roy, R.; Ordovas, L.; Zaragoza, P.; Romero, A.; Moreno, C.; Altarriba, J.; Rodellar, C. Association of polymorphisms in the bovine FASN gene with milk fat content. Anim. Genet. 2006, 37, 215–218. [Google Scholar] [CrossRef]
- Mohammed, M.E.A.; Abeer, A.; Elsamani, F.; Elsheikh, O.M.; Hodow, A.; Haji, O.K. Simulation of the fatty acid synthase complex mechanism of action. Chem. Bulg. J. Sci. Edu. 2013, 22, 405–412. [Google Scholar]
- Chakravarty, B.; Gu, Z.; Chirala, S.S.; Wakil, S.J.; Quiocho, F.A. Human fatty acid synthase: Structure and substrate selectivity of the thioesterase domain. Proc. Natl. Acad. Sci. USA 2004, 101, 15567–15572. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.-Y.; Lee, Y.-S.; La, B.-M.; Yeo, J.S.; Chung, E.; Kim, Y.; Lee, C. Fatty acid composition of beef is associated with exonic nucleotide variants of the gene encoding FASN. Mol. Biol. Rep. 2012, 39, 4083–4090. [Google Scholar] [CrossRef] [PubMed]
- Sanz, A.; Serrano, C.; Ranera, B.; Dervishi, E.; Zaragoza, P.; Calvo, J.H.; Rodellar, C. Novel polymorphisms in the 5′UTR of FASN, GPAM, MC4R and PLIN1 ovine candidate genes: Relationship with gene expression and diet. Small Rum. Res. 2015, 123, 70–74. [Google Scholar] [CrossRef]
- Wang, B.; Yang, L.; Luo, Y.; Su, R.; Su, L.; Zhao, L.; Jin, Y.E. Effects of feeding regimens on meat quality, fatty acid composition and metabolism as related to gene expression in Chinese Sunit sheep. Small Rum. Res. 2018, 169, 127–133. [Google Scholar] [CrossRef]
- Ekegbu, U.J.; Burrows, L.; Amirpour-Hamed, N.; Zhou, H.; Hicford, J.G.H. Gene polymorphisms in PROP1 associated with growth traits in sheep. Gene 2019, 683, 41–46. [Google Scholar] [CrossRef]
- Kucharski, M.; Kaczor, U. Fatty Acid binding protein 4 (FABP4) and the body lipid balance. Folia Biologica 2017, 65, 181–186. [Google Scholar] [CrossRef]
- Latruffe, N.; Vamecq, J. Peroxisome proliferators and peroxisome proliferator activated receptors (PPAR) as regulators of lipid metabolism. Biochimie 1997, 79, 81–94. [Google Scholar] [CrossRef]
- Li, L.; Jiang, J.; Wang, L.; Zhong, T.; Chen, B.; Zhan, S.; Zhang, H.; Du, L. Expression patterns of peroxisome proliferator activated receptor gamma 1 versus gamma 2 and their association with intramuscular fat in goat tissues. Gene 2013, 528, 195–200. [Google Scholar] [CrossRef]
- Yan, W.; Zhou, H.; Luo, Y.Z.; Hu, J.; Hickford, J.G. Allelic variation in ovine fatty acid-binding protein (FABP4) gene. Mol. Biol. Rep. 2012, 39, 10621–10625. [Google Scholar] [CrossRef]
- Yan, W.; Zhou, H.; Hu, J.; Luo, Y.; Hickford, J.G.H. Variation in the FABP4 gene affects carcass and growth traits in sheep. Meat Sci. 2018, 145, 334–339. [Google Scholar] [CrossRef]
- Dervishi, E.; Serrano, E.; Joy, M.; Serrano, M.; Rodellar, C.; Calvo, J.H. The effect of feeding system in the expression of genes related with fat metabolism in semitendinous muscle in sheep. Meat Sci. 2011, 89, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Barendse, W.; Bunch, R.J.; Thomas, M.B.; Harrison, B.E. A splice site single nucleotide polymorphism of the fatty acid binding protein 4 gene appears to be associated with intramuscular fat deposition in longissimus muscle in Australian cattle. Anim. Genet. 2009, 40, 770–773. [Google Scholar] [CrossRef] [PubMed]
- Hoashi, S.; Hinenoya, T.; Tanaka, A.; Ohsaki, H.; Sasazaki, S.; Taniguchi, M.; Oyama, K.; Mukai, F.; Mannen, H. Association between fatty acid compositions and genotypes of FABP4 and LXR-alpha in Japanese Black cattle. BMC Genet. 2008, 9, 84–90. [Google Scholar] [CrossRef]
- Ardicli, S.; Samli, H.; Alpay, F.; Dincel, D.; Soyudal, B.; Balci, F. Association of single nucleotide polymorphisms in the FABP4 gene with carcass characteristics and meat quality in Holstein bulls. Ann. Anim. Sci. 2017, 17, 117–130. [Google Scholar] [CrossRef]
- Dujková, R.; Ranganathan, Y.; Dufek, A.; Macák, J.; Bezdíˇcek, J. Polymorphic effects of FABP4 and SCD genes on intramuscular fatty acid profiles in longissimus muscle from two cattle breeds. Acta Vet. BRNO 2015, 84, 327–336. [Google Scholar] [CrossRef]
- Cho, S.; Park, T.S.; Yoon, D.H.; Cheong, H.S.; Namgoong, S.; Park, B.L.; Lee, H.W.; Han, C.S.; Kim, E.M.; Cheong, I.C.; et al. Identification of genetic polymorphisms in FABP3 and FABP4 and putative association with back fat thickness in Korean native cattle. BMB Rep. 2008, 41, 29–34. [Google Scholar] [CrossRef]
- Bahnamiri, H.Z.; Zali, A.; Ganjkhanlou, M.; Sadaghi, M.; Sharbabak, H.M. Regulation of lipid metabolism in adipose depots of fat-tailed and thin-tailed lambs during negative and positive energy balances. Gene 2018, 641, 203–211. [Google Scholar] [CrossRef]
- Backhtiarizadeh, M.R.; Moradi-Shahrbabak, M.; Ebrahimie, E. Underlying functional genomics of fat deposition in adipose tissue. Gene 2013, 521, 122–128. [Google Scholar] [CrossRef]
- Storch, J.; Corsico, B. The emerging functions and mechanisms of mammalian Fatty Acid-Binding Proteins. Ann. Rev. Nutr. 2008, 28, 73–95. [Google Scholar] [CrossRef]
- Rempel, L.A.; Casas, E.; Shackelford, S.D.; Wheeler, T.L. Relationship of polymorphisms within metabolic genes and carcass traits in crossbred beef cattle. J. Anim. Sci. 2012, 90, 1311–1316. [Google Scholar] [CrossRef]
- Shin, S.C.; Chung, E.R. Association of SNP marker in the leptin gene with carcass and meat quality traits in Korean cattle. Asian-Aust. J. Anim. Sci. 2006, 20, 1–6. [Google Scholar] [CrossRef]
- Thaller, G.; Kühn, C.; Winter, A.; Ewald, G.; Bellmann, O.; Wegner, J.; Zühlke, H.; Fries, R. DGAT1, a new positional and functional candidate gene for intramuscular fat deposition in cattle. Anim. Genet. 2003, 34, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Barendse, W.; Bunch, R.J.; Harrison, B.E.; Thomas, M.B. The growth hormone 1 GH1: C.457C & gtG mutation is associated with intramuscular and rump fat distribution in a large sample of Australian feedlot cattle. Anim. Genet. 2006, 37, 211–214. [Google Scholar] [PubMed]
- Gao, Y.; Zhang, R.; Hu, X.; Li, N. Application of genomic technologies to the improvement of meat quality of farm animals. Meat Sci. 2007, 77, 36–45. [Google Scholar] [CrossRef]
- Greguła-Kania, M.; Gruszecki, T.M.; Junkuszew, A.; Juszczuk-Kubiak, E.; Florek, M. Association of CAST gene polymorphism with carcass value and meat quality in two synthetic lines of sheep. Meat Sci. 2019, 154, 69–74. [Google Scholar] [CrossRef]
- Grochowska, E.; Borys, B.; Lisiak, D.; Mroczkowski, S. Genotypic and allelic effects of the myostatin gene (MSTN) on carcass, meat quality, and biometric traits in Colored Polish Merino sheep. Meat Sci. 2019, 151, 4–17. [Google Scholar] [CrossRef]
- Fan, Y.; Ren, C.; Meng, F.; Deng, K.; Zhang, G.; Wang, F. Effects of algae supplementation in high-energy dietary on fatty acid composition and the expression of genes involved in lipid metabolism in Hu sheep managed under intensive finishing system. Meat Sci. 2019, 157. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, H.; Forrest, R.H.J.; Hu, J.; Li, S.; Luo, Y.; Hickford, J.G.H. Variation in the ovine MYF5 gene and its effect on carcass lean meat yield in New Zealand Romney sheep. Meat Sci. 2017, 131, 146–151. [Google Scholar] [CrossRef]
- Freking, B.A.; Murphy, S.K.; Wylie, A.A.; Rhodes, S.J.; Keele, J.W.; Leymaster, K.A.; Jirtle, R.L.; Smith, T.P. Identification of the single base change causing the callipyge muscle hypertrophy phenotype, the only known example of polar overdominance in mammals. Genome Res. 2002, 12, 1496–1506. [Google Scholar] [CrossRef]
- Clop, A.; Marcq, F.; Takeda, H.; Pirottin, D.; Tordoir, X.; Bibe, B.; Bouix, J.; Caiment, F.; Elsen, J.M.; Eychenne, F.; et al. A mutation creating a potential illegitimate microRNA target site in the myostatin gene affects muscularity in sheep. Nat. Genet. 2006, 38, 813–818. [Google Scholar] [CrossRef]
- Malau-Aduli, A.E.O.; Bignell, C.W.; McCulloch, R.; Kijas, J.W.; Nichols, P.D. Genetic association of delta-six fatty acid desaturase single nucleotide polymorphic molecular marker and muscle long chain omega-3 fatty acids in Australian lamb. In Global Challenges to Production, Processing and Consumption of Meat, Proceedings of the 57th International Congress of Meat Science and Technology, 7–12 August 2011; De Smet, S., Ed.; University of Ghent: Ghent, Belgium; Volume 57, p. 126. [Google Scholar]
- Knight, M.I.; Daetwyler, H.D.; Hayes, B.J.; Hayden, M.J.; Ball, A.J.; Pethick, D.W.; McDonagh, M.B. An independent validation association study of carcass quality, shear force, intramuscular fat percentage and omega-3 polyunsaturated fatty acid content with gene markers in Australian lamb. Meat Sci. 2014, 96, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, A.S.H.; Pires, V.M.R.; Fontes, C.M.D.A.; Prates, J.A.M. Expression of genes controlling fat deposition in two genetically diverse beef cattle breeds fed high or low silage diets. BMC Vet. Res. 2013, 9, 118. [Google Scholar] [CrossRef] [PubMed]
- Anton, I.; Kovacs, K.; Hollo, G.; Farkas, V.; Lehel, L.; Hajda, Z.; Zsolnai, A. Effect of leptin, DGAT1 and TG gene polymorphisms on the intramuscular fat of Angus cattle in Hungary. Livest. Sci. 2011, 135, 300–303. [Google Scholar] [CrossRef]
- Mullen, A.M.; Stapleton, P.C.; Corcoran, D.; Hamill, R.M.; White, A. Understanding meat quality through the application of genomic and proteomic approaches. Meat Sci. 2006, 74, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Fujii, J.; Otsu, K.; Zorzato, F.; de Leon, S.; Khanna, V.K.; Weiler, J.E.; O’Brien, P.J.; MacLennan, D.H. Identification of a mutation in porcine ryanodine receptor associated with malignant hyperthermia. Science 1991, 253, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Larsen, N.; Short, T.; Plastow, G.; Rothschild, M.F. A missense variant of the porcine melanocortin-4 receptor (MC4R) gene is associated with fatness, growth, and feed intake traits. Mamm. Genome 2000, 11, 131–135. [Google Scholar] [CrossRef]
- Milan, D.; Jeon, J.T.; Looft, C.; Amarger, V.; Robic, A.; Thelander, M.; Rogel-Gaillard, C.; Paul, S.; Iannuccelli, N.; Rask, L.; et al. A mutation in PRKAG3 associated with excess glycogen content in pig skeletal muscle. Science 2000, 288, 1248–1251. [Google Scholar] [CrossRef]
- Gerbens, F.; Jansen, A.; van Erp, A.J.; Harders, F.; Meuwissen, T.H.; Rettenberger, G.; Veerkamp, J.H.; Te Pas, M.F. The adipocyte fatty acid-binding protein locus: Characterization and association with intramuscular fat content in pigs. Mamm. Genome 1998, 9, 1022–1026. [Google Scholar] [CrossRef]
- Gerbens, F.; van Erp, A.J.; Harders, F.L.; Verburg, F.J.; Meuwissen, T.H.; Veerkamp, J.H.; Te Pas, M.F.W. Effect of genetic variants of the heart fatty acid-binding protein gene on intramuscular fat and performance traits in pigs. J. Anim. Sci. 1999, 77, 846–852. [Google Scholar] [CrossRef]
- Ciobanu, D.C.; Bastiaansen, J.W.; Lonergan, S.M.; Thomsen, H.; Dekkers, J.C.; Plastow, G.S.; Rothschild, M.F. New alleles in calpastatin gene are associated with meat quality traits in pigs. J. Anim. Sci. 2004, 82, 2829–2839. [Google Scholar] [CrossRef]
- Van Laere, A.S.; Nguyen, M.; Braunschweig, M.; Nezer, C.; Collette, C.; Moreau, L.; Archibald, A.L.; Haley, C.S.; Buys, N.; Tally, M.; et al. A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 2003, 425, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.G.; Li, N.; Deng, X.M.; Lian, Z.X.; Li, H.; Wu, C.X. Single nucleotide polymorphism analysis on chicken extracellular fatty acid binding protein gene and its associations with fattiness trait. Sci. China 2001, 44, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Li, H.; Li, N.; Leng, L.; Wang, Y. Tissue expression and association with fatness traits of liver fatty acid-binding protein gene in chicken. Poultry Sci. 2006, 85, 1890–1895. [Google Scholar] [CrossRef] [PubMed]
- Pannier, L.; Gardner, G.E.; O’Reilly, R.A.; Pethick, D.W. Factors affecting lamb eating quality and the potential for their integration into an MSA sheepmeat grading model. Meat Sci. 2018, 144, 43–52. [Google Scholar] [CrossRef]
- Lambe, N.R.; McLean, K.A.; Gordon, J.; Evans, D.; Clelland, N.; Bunger, L. Prediction of intramuscular fat content using CT scanning of packaged lamb cuts and relationships with meat eating quality. Meat Sci. 2017, 123, 112–119. [Google Scholar] [CrossRef]
- Flakemore, A.; McEvoy, P.D.; Balogun, R.O.; Malau-Aduli, B.S.; Nichols, P.D.; Malau-Aduli, A.E.O. Degummed crude canola oil supplementation affects fat depot melting points in purebred and first-cross Merino sheep. Anim. Vet. Sci. 2014, 2, 75–80. [Google Scholar] [CrossRef]
- Calnan, H.B.; Jacob, R.H.; Pethick, D.W.; Gardner, G.E. Selection for intramuscular fat and lean meat yield will improve the bloomed colour of Australian lamb loin meat. Meat Sci. 2017, 131, 187–195. [Google Scholar] [CrossRef]
- Hocquette, J.F.; Richardson, R.I.; Prache, S.; Medale, F.; Duffy, G.; Scollan, N.D. The future trends for research on quality and safety of animal products. Ital. J. Anim. Sci. 2005, 4, 49–72. [Google Scholar] [CrossRef]
- Indurain, G.; Carr, T.R.; Goñi, M.V.; Insausti, K.; Beriain, M.J. The relationship of carcass measurements to carcass composition and intramuscular fat in Spanish beef. Meat Sci. 2009, 82, 155–161. [Google Scholar] [CrossRef]
- Joo, S.T.; Kim, G.D.; Hwang, Y.H.; Ryu, Y.C. Control of fresh meat quality through manipulation of muscle fiber characteristics. Meat Sci. 2013, 95, 828–836. [Google Scholar] [CrossRef]
- Park, G.B.; Moon, S.S.; Ko, Y.D.; Ha, J.K.; Lee, J.G.; Chang, H.H.; Joo, S.T. Influence of slaughter weight and sex on yield and quality grades of Hanwoo (Korean native cattle) carcasses. J. Anim. Sci. 2002, 80, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Holman, B.W.B.; Malau-Aduli, A.E.O. Spirulina as a livestock supplement and animal feed. J. Anim. Physiol. Anim. Nutr. 2013, 97, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.B.; Kawachi, H.; Choi, C.B.; Choi, C.W.; Wu, G.; Sawyer, J.E. Cellular regulation of bovine intramuscular adipose tissue development and composition. J. Anim. Sci. 2009, 87, E72–E82. [Google Scholar] [CrossRef]
- Hausman, G.J.; Dodson, M.V.; Ajuwon, K.; Azain, M.; Barnes, K.M.; Guan, L.L.; Jiang, Z.; Poulos, S.P.; Sainz, E.D.; Smith, S.; et al. Board-Invited Review: The biology and regulation of preadipocyte and adipocytes in meat animals, J. Anim. Sci. 2009, 87, 1218–1246. [Google Scholar] [CrossRef] [PubMed]
- Hocquette, J.F.; Mainsant, P.; Daudin, J.D.; Cassar-Malek, I.; Rémond, D.; Doreau, M.; Sans, P.; Bauchart, D.; Agabriel, J.; Verbecke, W.; et al. Will meat be produced in vitro in the future? INRA Prod. Anim. 2013, 26, 363–374. [Google Scholar] [CrossRef]
- Wyness, L. Nutritional aspects of red meat in the diet. In Nutrition and Climate Change: Major Issues Confronting the Meat Industry, Wood, J.D., Rowlings, C., Eds.; Nottingham University Press: Nottingham, UK, 2013; pp. 1–22. [Google Scholar]
- Wall, K.R.; Kerth, C.R.; Miller, R.K.; Alvarado, C. Grilling temperature effects on tenderness, juiciness, flavor and volatile aroma compounds of aged ribeye, strip loin, and top sirloin steaks. Meat Sci. 2019, 150, 141–148. [Google Scholar] [CrossRef]
- Koohmaraie, R.A.; Whipple, G.; Crouse, L. Acceleration of postmortem tenderization in lamb and Brahaman-cross beef carcasses through infusion of calcium chloride. J. Anim Sci. 1990, 68, 1278–1283. [Google Scholar]
- Koohmaraie, M.; Kent, M.P.; Shackelford, S.D.; Veiseth, E.; Wheeler, T.M. Meat tenderness and muscle growth: Is there any relationship? Meat Sci. 2002, 62, 345–352. [Google Scholar] [CrossRef]
- Cohen-Zinder, M.; Orlov, A.; Trofimyuk, O.; Agmon, R.; Kabiya, R.; Shor-Shimoni, E.; Wagner, E.K.; Hussey, K.; Leibovich, H.; Miron, J.; et al. Dietary supplementation of Moringa oleifera silage increases meat tenderness of Assaf lambs. Small Rum. Res. 2017, 151, 110–116. [Google Scholar] [CrossRef]
- Muchenje, V.; Dzama, K.; Chimonyo, M.; Strydom, P.E.; Raats, J.G. Relationship between pre-slaughter responsiveness and beef quality in three cattle breeds. Meat Sci. 2009, 81, 653–657. [Google Scholar] [CrossRef]
- Razminowicz, R.H.; Kreuzer, M.; Scheeder, M.R.L. Quality of retail beef from two grass-based production systems in comparison with conventional beef. Meat Sci. 2006, 73, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Perlo, F.; Bonato, P.; Teira, G.; Tisocco, O.; Vicentin, J.; Pueyo, J.; Mansilla, A. Meat quality of lambs produced in the Mesopotamia region of Argentina finished on different diets. Meat Sci. 2008, 79, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Sañudo, C.; Alfonso, M.; Sanchez, A.; Berge, P.; Dransfield, E.; Zygoyiannis, D.; Stamataris, C.; Thorkelsson, G.; Valdimarsdottir, T.; Piasentier, E.; et al. Meat texture of lambs from different European production systems. Aust. J. Agric. Res. 2003, 54, 551–560. [Google Scholar] [CrossRef]
- De Brito, G.F.; Ponnampalam, E.R.; Hopkins, D.L. The effect of extensive feeding systems on growth rate, carcass traits, and meat quality of finishing lambs. Compr. Rev. Food Sci. Food Saf. 2017, 16, 23–38. [Google Scholar] [CrossRef]
- Montossi, F.; Font-i-Furnols, M.; del Campo, M.; San Julián, R.; Brito, G.; Sañudo, C. Sustainable sheep production and consumer preference trends: Compatibilities, contradictions, and unresolved dilemmas. Meat Sci. 2013, 95, 772–789. [Google Scholar] [CrossRef]
- Yang, C.; Liu, J.; Wu, X.; Bao, P.; Long, R.; Guo, X.; Ding, X.; Yan, P. The response of gene expression associated with lipid metabolism, fat deposition and fatty acid profile in the longissimus dorsi muscle of Gannan yaks to different energy levels of diets. PLoS ONE 2017, 12, e187604. [Google Scholar] [CrossRef]
- Jurie, C.; Cassarmalek, I.; Bonnet, M.; Leroux, C.; Bauchart, D.; Boulesteix, P.; Pethick, D.W.; Hocquette, J.F. Adipocyte fatty acid-binding protein and mitochondrial enzyme activities in muscles as relevant indicators of marbling in cattle. J. Anim Sci. 2007, 85, 2660–2669. [Google Scholar] [CrossRef]
- Oliveira, D.M.; Chalfun-Junior, A.; Chizzotti, M.L.; Barreto, H.G.; Coelho, T.C.; Paiva, L.V.; Coelho, C.P.; Teixeira, P.D.; Schoonmaker, J.P.; Ladeira, M.M. Expression of genes involved in lipid metabolism in the muscle of beef cattle fed soybean or rumen-protected fat, with or without monensin supplementation. J. Anim Sci. 2014, 92, 5426–5436. [Google Scholar] [CrossRef] [PubMed]
- Lusk, J.L.; Fox, J.A.; Schroeder, T.C.; Mintert, J.; Koohmaraie, M. In-store valuation of steak tenderness. Am. J. Agric. Econ. 2001, 83, 539–550. [Google Scholar] [CrossRef]
- Elmasry, G.; Barbin, D.F.; Sun, D.; Allen, P. Meat quality evaluation by hyperspectral imaging technique: An overview. Crit. Rev. Food Sci. Nutr. 2002, 52, 689–711. [Google Scholar] [CrossRef]
- Ali, M.S.; Yang, H.S.; Jeong, J.Y.; Moon, S.H.; Hwang, Y.H.; Park, G.B.; Joo, S.T. Effects of chilling temperature of carcass on breast meat quality of duck. Poult. Sci. 2008, 87, 1860–1867. [Google Scholar] [CrossRef] [PubMed]
- Starkey, C.P.; Geesink, G.H.; Collins, D.; Oddy, V.H.; Hopkins, D.L. Do sarcomere length, collagen content, pH, intramuscular fat and desmin degradation explain variation in the tenderness of three ovine muscles? Meat Sci. 2016, 113, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Rhee, M.S.; Wheeler, T.L.; Shackelford, S.D.; Koohmaraie, M. Variation in palatability and biochemical traits within and among eleven beef muscles. J. Anim. Sci. 2004, 534–550. [Google Scholar] [CrossRef] [PubMed]
- Guzek, D.; Głąska, D.; Pogorzelski, G.; Kozań, K.; Pietras, J.; Konarska, M.; Sakowska, A.; Głąski, K.; Pogorzelska, E.; Barszczewski, J.; et al. Variation of meat quality parameters due to conformation and fat class in Limousin bulls slaughtered at 25 to 27 months of age. Asian-Aust. J. Anim. Sci. 2013, 26, 716–722. [Google Scholar] [CrossRef]
- Arshad, M.S.; Sohaib, M.; Ahmad, R.S.; Nadeem, M.T.; Imran, A.; Arshad, M.U.; Kwon, J.H.; Amjad, Z. Ruminant meat flavour influenced by different factors with special reference to fatty acids. Lipids Hlth. Dis. 2018, 17, 1–13. [Google Scholar]
- Khan, M.I.; Jang, S.; Nam, K.C.; Jo, C. Postmortem aging of beef with a special reference to dry aging. Korean J. Food Sci. Anim. Resour. 2016, 36, 159–169. [Google Scholar] [CrossRef]
- Calkins, C.R.; Hodgen, J.M. A fresh look at meat flavour. Meat Sci. 2007, 77, 63–80. [Google Scholar] [CrossRef]
- Mottram, D.S. Flavour formation in meat and meat products: A review. Food Chem. 1998, 62, 415–424. [Google Scholar] [CrossRef]
- Mottram, D.S. Meat flavour. In Understanding Natural Flavors; Piggott, J.R., Paterson, A., Eds.; Springer: Boston, MA, USA, 1994; pp. 140–163. [Google Scholar]
- Jamora, J.J.; Rhee, K.S. Flavour of lamb and mutton. In Quality Attributes of Muscle Foods; Xiong, Y.L., Chi-Tang, H., Shahidi, F., Eds.; Springer: Boston, MA, USA, 1999. [Google Scholar]
- Muir, P.D.; Deaker, J.M.; Bown, M.D. Effects of forage- and grain-based feeding systems on beef quality: A review. New Zeal. J. Agr. Res. 1998, 41, 623–635. [Google Scholar] [CrossRef]
- McMillin, K.W.; Hoffman, L.C. Improving the quality of meat from ratites. In Improving the Sensory and Nutritional Quality of Fresh Meat; Kerry, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 418–446. ISBN 9781845693435. [Google Scholar]
- de Lima, D.M.; de Carvalho, F.F.R.; da Silva, F.J.S.; Rangel, A.H.; Novaes, L.P.; Difante, G.D.S. Intrinsic factors affecting sheep meat quality: A review. Rev. Colomb. Cienc. Pecu. 2016, 29, 3–15. [Google Scholar] [CrossRef]
- Cloete, J.J.E.; Hoffman, L.C.; Cloete, S.W.P. A comparison between slaughter traits and meat quality of various sheep breeds: Wool, dual-purpose and mutton. Meat Sci. 2012, 91, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Hocquette, J.F.; Gondret, F.; Baéza, E.; Medale, F.; Jurie, C.; Pethick, D.W. Intramuscular fat content in meat-producing animals: Development, genetic and nutritional control, and identification of putative markers. Animal 2010, 4, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Jeremiah, L.E.; Gibson, L.L.; Aalhus, J.L.; Dugan, M.E.R. Assessment of palatability attributes of the major beef muscles. Meat Sci. 2003, 65, 949–958. [Google Scholar] [CrossRef]
- Aaslyng, M.D.; Oksama, M.; Olsen, E.V.; Bejerholm, C.; Baltzer, M.; Andersen, G.; Bredie, W.L.P.; Byrne, D.V.; Gabrielsen, G. The impact of sensory quality of pork on consumer preference. Meat Sci. 2007, 76, 61–73. [Google Scholar] [CrossRef]
- Cho, S.H.; Kim, J.; Park, B.Y.; Seong, P.N.; Kang, G.H.; Kim, J.H.; Jung, S.G.; Im, S.K.; Kim, D.H. Assessment of meat quality properties and development of a palatability prediction model for Korean Hanwoo steer beef. Meat Sci. 2010, 86, 236–242. [Google Scholar] [CrossRef]
- Yilmaz, M.T.; Karakaya, M.; Aktas, N. Composition and thermal properties of cattle fats. Eur. J. Lipid Sci. Technol. 2010, 112, 410–416. [Google Scholar] [CrossRef]
- Knothe, G.; Dunn, R.O. A comprehensive evaluation of the melting points of fatty acids and esters determined by differential scanning calorimetry. J. Am. Oil Chem. Soc. 2009, 86, 843–856. [Google Scholar] [CrossRef]
- Pitchford, W.S.; Deland, M.P.B.; Siebert, B.D.; Malau-Aduli, A.E.O.; Bottema, C.D.K. Genetic variation in fatness and fatty acid composition of crossbred cattle. J. Anim. Sci. 2002, 80, 2825–2832. [Google Scholar] [CrossRef]
- Woods, W.B.; Fearon, A.M. Dietary sources of unsaturated fatty acids for animals and their transfer into meat, milk and eggs: A review. Livest. Sci. 2009, 126, 153–159. [Google Scholar] [CrossRef]
- Yang, A.; Larsen, T.W.; Smith, S.B.; Tume, R.K. Delta (9) desaturase activity in bovine subcutaneous adipose tissue of different fatty acid composition. Lipids 1999, 34, 971–978. [Google Scholar] [CrossRef]
| Trait | Mean ± SE | Range |
|---|---|---|
| Fat melting point (°C) | 34.08 ± 1.4 | 28.0–39.0 |
| Intramuscular fat (%) | 4.4 ± 0.2 | 3.4–8.2 |
| Hot standard carcass weight (kg) | 24.6 ± 2.7 | 19.5–30.7 |
| Dressing percentage | 50.2 ± 2.2 | 47.0–54.4 |
| Fat score | 4.7 ± 0.6 | 4–5 |
| GR fat depth (mm) | 16.4 ± 3.5 | 10–24 |
| Tenderness (N) | 32.3 ± 5.1 | 20.0–38.5 |
| pH | 5.63 ± 0.11 | 5.53–6.83 |
| Overall consumer liking (9-point scale) | 8.2 ± 0.9 | 7.9–8.5 |
| Omega-3 long chain PUFA (mg/100g) | ||
| EPA (20:5n-3) | 24.3 ± 5.2 | 17.8–44.8 |
| DHA (22:6n-3) | 8.3 ± 2.7 | 3.4–12.1 |
| DPA (22:5n-3) | 25.2 ± 8.0 | 14.0–80.3 |
| EPA + DHA | 32.6 ± 7.0 | 33.6–69.9 |
| EPA + DHA + DPA | 57.9 ± 13.6 | 49.1–132.5 |
| Animal | Candidate Genes | Traits | References |
|---|---|---|---|
| Sheep | CAST MSTN FADS2, ELOVL2, SCD, CPT1α, SREBF-1 FABP4 MYF5 Callipyge | Carcass Carcass, meat quality Fatty acids Carcass yield Leg and loin yield Muscular hypertrophy | [177] [178] [179] [161,162] [180] [181] |
| GDF8 FAD CAST FASN, FABP4, DGAT1, SCD | Muscular hypertrophy Omega-3 long-chain PUFA Tenderness Fat metabolism | [182] [183] [184] [146,163] | |
| Cattle Pig | FABP4, SCD, PPAG, ACACA, LPL CAST Leptin/Thyroglobulin Myostatin DGAT1 HAL MC4R RN, PRKAG3 | Fatty acid profile Meat tenderness Marbling Growth and profile IMF/marbling Meat quality/stress Growth and fatness Meat quality | [185] [184] [186] [187] [174] [188] [189] [190] |
| AFABP/FABP4 | IMF | [191] | |
| HFABP/FABP3 | IMF | [192] | |
| CAST | Tenderness | [193] | |
| IGF2 | Growth and fatness | [194] | |
| Chicken | EX-FABP | Fatness | [195] |
| L-FABP | Fatness | [196] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pewan, S.B.; Otto, J.R.; Huerlimann, R.; Budd, A.M.; Mwangi, F.W.; Edmunds, R.C.; Holman, B.W.B.; Henry, M.L.E.; Kinobe, R.T.; Adegboye, O.A.; et al. Genetics of Omega-3 Long-Chain Polyunsaturated Fatty Acid Metabolism and Meat Eating Quality in Tattykeel Australian White Lambs. Genes 2020, 11, 587. https://doi.org/10.3390/genes11050587
Pewan SB, Otto JR, Huerlimann R, Budd AM, Mwangi FW, Edmunds RC, Holman BWB, Henry MLE, Kinobe RT, Adegboye OA, et al. Genetics of Omega-3 Long-Chain Polyunsaturated Fatty Acid Metabolism and Meat Eating Quality in Tattykeel Australian White Lambs. Genes. 2020; 11(5):587. https://doi.org/10.3390/genes11050587
Chicago/Turabian StylePewan, Shedrach Benjamin, John Roger Otto, Roger Huerlimann, Alyssa Maree Budd, Felista Waithira Mwangi, Richard Crawford Edmunds, Benjamin William Behrens Holman, Michelle Lauren Elizabeth Henry, Robert Tumwesigye Kinobe, Oyelola Abdulwasiu Adegboye, and et al. 2020. "Genetics of Omega-3 Long-Chain Polyunsaturated Fatty Acid Metabolism and Meat Eating Quality in Tattykeel Australian White Lambs" Genes 11, no. 5: 587. https://doi.org/10.3390/genes11050587
APA StylePewan, S. B., Otto, J. R., Huerlimann, R., Budd, A. M., Mwangi, F. W., Edmunds, R. C., Holman, B. W. B., Henry, M. L. E., Kinobe, R. T., Adegboye, O. A., & Malau-Aduli, A. E. O. (2020). Genetics of Omega-3 Long-Chain Polyunsaturated Fatty Acid Metabolism and Meat Eating Quality in Tattykeel Australian White Lambs. Genes, 11(5), 587. https://doi.org/10.3390/genes11050587







