Microscopic Examination of Polymeric Monoguanidine, Hydrochloride-Induced Cell Membrane Damage in Multidrug-Resistant Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Materials and Methods
2.1. Polymerized Monoguanidine Antibacterial Agents and Other Chemicals
2.2. Strains
2.3. Evaluation of Bactericidal Dynamics
2.4. Assessment of Outer Membrane Permeability by Beta-Lactamase Enzyme
2.5. Assessment of Membrane Permeability by Confocal Fluorescence Microscopy
2.6. Field Emission Scanning Electronic Microscopy
2.7. Transmission Electron Microscopy
3. Results
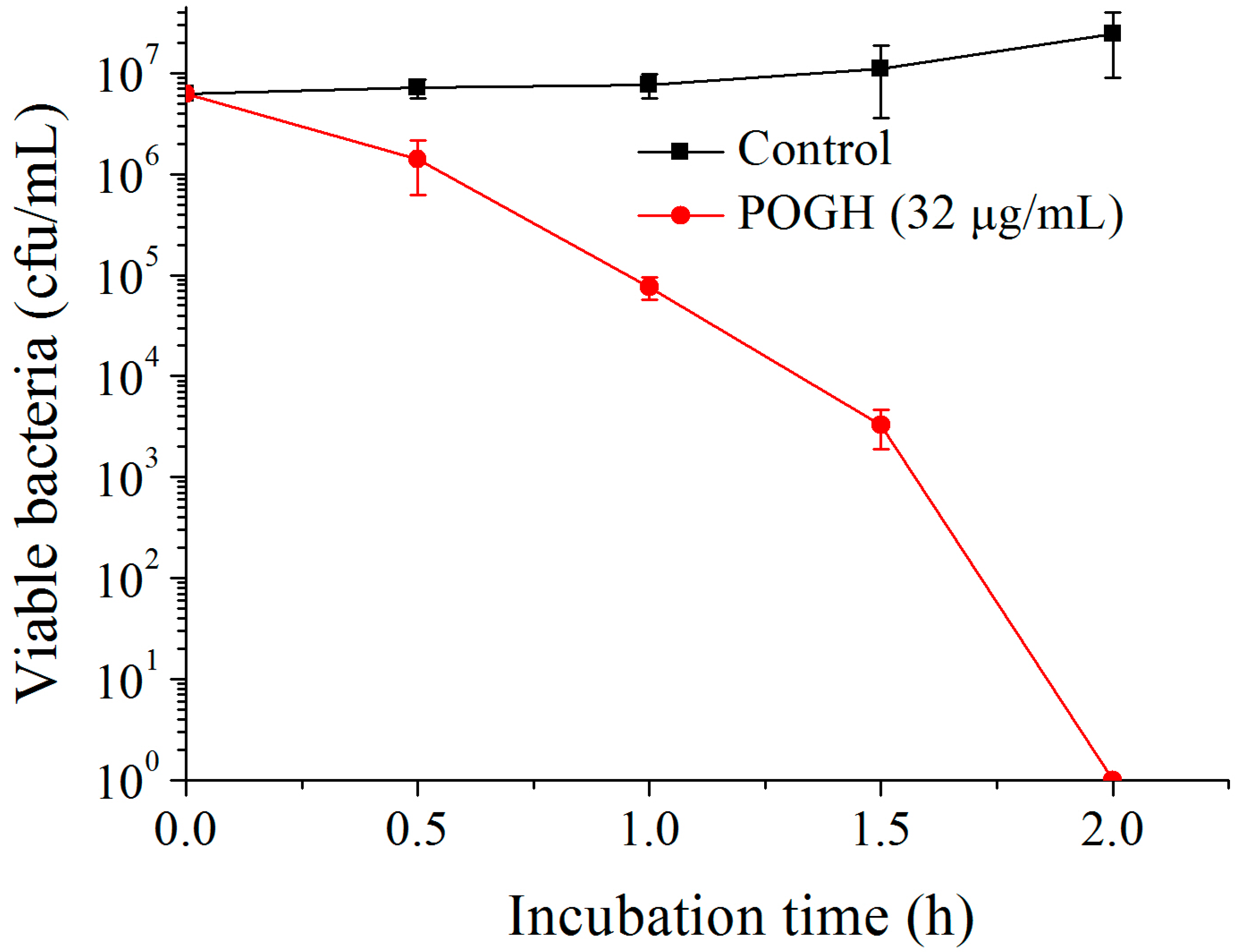
3.1. Polyoctamethylene Monoguanidine Hydrochloride (POGH) Has Bactericidal Activity against MDR-PA
3.2. POGH Alters the Permeability of the Outer Membrane of MDR-PA Cell and Induces Beta-Lactamase Release in Periplasm
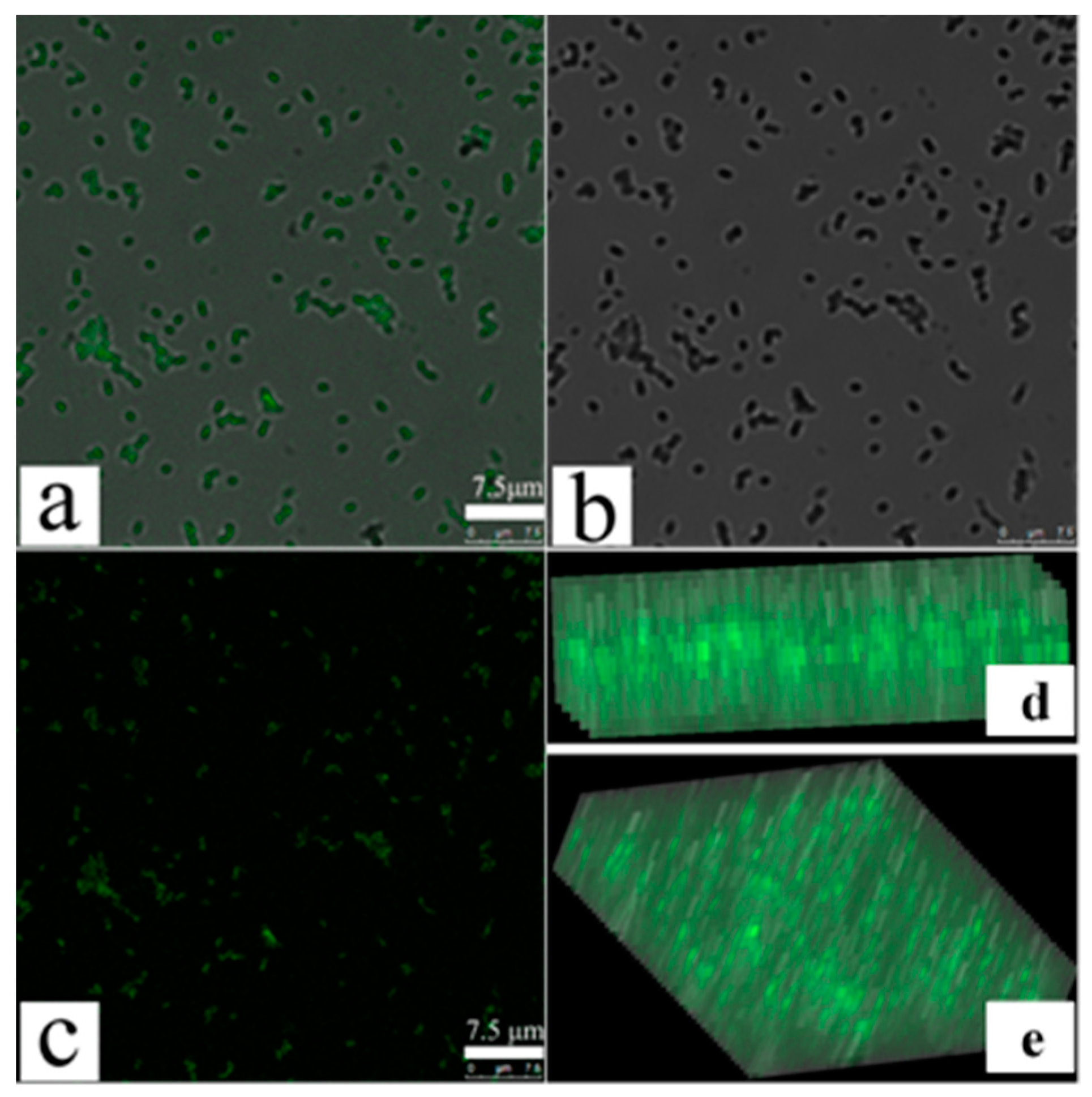
3.3. Confocal Fluorescence Microscopy Detects an Increase in MDR-PA Membrane Permeability
3.4. Scanning Electron Microscopy Shows Changes in MDR-PA Cellular Surface Morphology and Cell Membrane Damage
3.5. Transmission Electron Microscopy Shows Changes in MDR-PA Intracellular Structure and Cell Membrane Damage
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MDR-PA | multidrug resistant Pseudomonas aeruginosa |
| MRSA | methicillin-resistant Staphylococcus aureus |
| POGH | polyoctamethylene monoguanidine hydrochloride |
References
- Takahashi, H.; Caputo, G.A.; Vemparala, S.; Kuroda, K. Synthetic random copolymers as a molecular platform to mimic host-defense antimicrobial peptides. Bioconjug. Chem. 2017, 28, 1340–1350. [Google Scholar] [CrossRef]
- Kuroda, K.; Caputo, G.A. Antimicrobial polymers as synthetic mimics of host-defense peptides. Wiley Interdiscip. Rev. 2013, 5, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Palermo, E.F.; Kuroda, K. Structural determinants of antimicrobial activity in polymers which mimic host defense peptides. Appl. Microbiol. Biotechnol. 2010, 87, 1605–1615. [Google Scholar] [CrossRef]
- Muñoz-Bonilla, A.; Fernández-García, M. The roadmap of antimicrobial polymeric materials in macromolecular nanotechnology. Eur. Polym. J. 2015, 65, 46–62. [Google Scholar] [CrossRef]
- Hurdle, J.G.; O’Neill, A.J.; Chopra, I.; Lee, R.E. Targeting bacterial membrane function: An underexploited mechanism for treating persistent infections. Nat. Rev. Microbiol. 2011, 9, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Gorityala, B.K.; Guchhait, G.; Goswami, S.; Fernando, D.M.; Kumar, A.; Zhanel, G.G.; Schweizer, F. Hybrid antibiotic overcomes resistance in P. aeruginosa by enhancing outer membrane penetration and reducing efflux. J. Med. Chem. 2016, 59, 8441–8455. [Google Scholar] [CrossRef] [PubMed]
- Ouberai, M.; El Garch, F.; Bussiere, A.; Riou, M.; Alsteens, D.; Lins, L.; Baussanne, I.; Dufrene, Y.F.; Brasseur, R.; Decout, J.L.; et al. The Pseudomonas aeruginosa membranes: A target for a new amphiphilic aminoglycoside derivative? Biochim. Biophys. Acta Biomembr. 2011, 1808, 1716–1727. [Google Scholar] [CrossRef] [PubMed]
- Mingeot-Leclercq, M.P.; Decout, J.L. Bacterial lipid membranes as promising targets to fight antimicrobial resistance, molecular foundations and illustration through the renewal of aminoglycoside antibiotics and emergence of amphiphilic aminoglycosides. MedChemComm 2016, 7, 586–611. [Google Scholar] [CrossRef]
- Pollard, J.E.; Snarr, J.; Chaudhary, V.; Jennings, J.D.; Shaw, H.; Christiansen, B.; Wright, J.; Jia, W.Y.; Bishop, R.E.; Savage, P.B. In vitro evaluation of the potential for resistance development to ceragenin CSA-13. J. Antimicrob. Chemother. 2012, 67, 2665–2672. [Google Scholar] [CrossRef]
- Shireen, T.; Singh, M.; Das, T.; Mukhopadhyay, K. Differential adaptive responses of Staphylococcus aureus to in vitro selection with different antimicrobial peptides. Antimicrob. Agents Chemother. 2013, 57, 5134–5137. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Paino, M.; Muñoz-Bonilla, A.; Fernández-García, M. Antimicrobial polymers in the nano-world. Nanomaterials 2017, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric materials with antimicrobial activity. Prog. Polym. Sci. 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Villanueva, M.E.; Gonzalez, J.A.; Rodriguez-Castellon, E.; Teves, S.; Copello, G.J. Antimicrobial surface functionalization of PVC by a guanidine based antimicrobial polymer. Mater. Sci. Eng. C 2016, 67, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Moore, L.E. Cationic antiseptics: Diversity of action under a common epithet. J. Appl. Microbiol. 2005, 99, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.F.; Wang, H.; Ziaee, Z.; Chibante, F.; Zheg, A.N.; Xiao, H.N. Non-leaching antimicrobial biodegradable PBAT films through a facile and novel approach. Mater. Sci. Eng. C 2016, 58, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Ghamrawi, S.; Bouchara, J.P.; Tarasyuk, O.; Rogalsky, S.; Lyoshina, L.; Bulko, O.; Bardeau, J.F. Promising silicones modified with cationic biocides for the development of antimicrobial medical devices. Mater. Sci. Eng. C 2017, 75, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Kim, K.J.; Lee, D.G. Antifungal activity of the cationic antimicrobial polymer-polyhexamethylene guanidine hydrochloride and its mode of action. Fungal Biol. 2017, 121, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Donalisio, M.; Ranucci, E.; Cagno, V.; Civra, A.; Manfredi, A.; Cavalli, R.; Ferruti, P.; Lembo, D. Agmatine-containing poly(amidoamine)s as a novel class of antiviral macromolecules: Structural properties and in vitro evaluation of infectivity inhibition. Antimicrob. Agents Chemother. 2014, 58, 6315–6319. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, N.; Pirrone, V.; Passic, S.; Zhu, W.; Kholodovych, V.; Welsh, W.; Rando, R.F.; Labib, M.E.; Wigdahl, B.; Krebs, F.C. Specific interactions between the viral coreceptor CXCR4 and the biguanide-based compound NB325 mediate inhibition of human immunodeficiency virus type 1 infection. Antimicrob. Agents Chemother. 2009, 53, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.X.; Wei, D.F.; Lu, Y.H. Polyhexamethylene guanidine hydrochloride shows bactericidal advantages over chlorhexidine digluconate against ESKAPE bacteria. Biotechnol. Appl. Biochem. 2015, 62, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Bera, S.; Zhanel, G.G.; Schweizer, F. Antibacterial activity of guanidinylated neomycin B- and kanamycin A-derived amphiphilic lipid conjugates. J. Antimicrob. Chemother. 2010, 65, 1224–1227. [Google Scholar] [CrossRef] [PubMed]
- Grare, M.; Dibama, H.M.; Lafosse, S.; Ribon, A.; Mourer, M.; Regnouf-de-Vains, J.B.; Finance, C.; Duval, R.E. Cationic compounds with activity against multidrug-resistant bacteria: Interest of a new compound compared with two older antiseptics, hexamidine and chlorhexidine. Clin. Microbiol. Infect. 2010, 16, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.X.; Wei, D.F.; Guan, Y.; Zheng, A.N.; Zhong, J.J. Extensive in vitro activity of guanidine hydrochloride polymer analogs against antibiotics-resistant clinically isolated strains. Mater. Sci. Eng. C 2011, 31, 1836–1843. [Google Scholar] [CrossRef]
- Oule, M.K.; Azinwi, R.; Bernier, A.M.; Kablan, T.; Maupertuis, A.M.; Mauler, S. Polyhexamethylene guanidine hydrochloride-based disinfectant: A novel tool to fight meticillin-resistant Staphylococcus aureus and nosocomial infections. J. Med. Microbiol. 2008, 57, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Buxbaum, A.; Kratzer, C.; Graninger, W.; Georgopoulos, A. Antimicrobial and toxicological profile of the new biocide Akacid plus®. J. Antimicrob. Chemother. 2006, 58, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Broxton, P.; Woodcock, P.M.; Heatley, F.; Gilbert, P. Interaction of some polyhexamethylene biguanides and membrane phospholipids in Escherichia coli. J. Appl. Microbiol. 1984, 57, 115–124. [Google Scholar]
- Chawner, J.A.; Gilbert, P. Interaction of the bisbiguanides chlorhexidine and alexidine with phospholipid vesicles: Evidence for separate modes of action. J. Appl. Microbiol. 1989, 66, 253–258. [Google Scholar] [CrossRef]
- Li, X.Z.; Zhang, L.; Srikumar, R.; Poole, K. β-Lactamase inhibitors are substrates for the multidrug efflux pumps of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 1998, 42, 399–403. [Google Scholar] [PubMed]
- Li, X.Z.; Poole, K. Interplay between the MexA-MexB-OprM multidrug efflux system and the outer membrane barrier in the multiple antibiotic resistance of Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2000, 45, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Lamers, R.P.; Nguyen, U.T.; Nguyen, Y.; Buensuceso, R.N.C.; Burrows, L.L. Loss of membrane-bound lytic transglycosylases increases outer membrane permeability and beta-lactam sensitivity in Pseudomonas aeruginosa. MicrobiologyOpen 2015, 4, 879–895. [Google Scholar] [CrossRef] [PubMed]
- Mangoni, M.L.; Papo, N.; Barra, D.; Simmaco, M.; Bozzi, A.; Di Giulio, A. Effects of the antimicrobial peptide temporin L on cell morphology, membrane and viability of Escherichia coli. Biochem. J. 2004, 380, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.F.; Mowery, B.P.; Lee, S.E.; Stahl, S.S.; Lehrer, R.I.; Gellman, S.H.; Epand, R.M. Dual mechanism of bacterial lethality for a cationic sequence-random copolymer that mimics host-defense antimicrobial peptides. J. Mol. Biol. 2008, 379, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.T.; Huang, M.L.; Kirshenbaum, K. Osmoprotective polymer additives attenuate the membrane pore-forming activity of antimicrobial peptoids. Biopolymers 2015, 103, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Schneider, V.A.F.; Coorens, M.; Ordonez, S.R.; Tjeerdsma-van Bokhoven, J.L.M.; Posthuma, G.; van Dijk, A.; Haagsman, H.P.; Veldhuizen, E.J.A. Imaging the antimicrobial mechanism(s) of cathelicidin-2. Sci. Rep. 2016, 6, 32948–32959. [Google Scholar] [CrossRef] [PubMed]
- Zorko, M.; Jerala, R. Alexidine and chlorhexidine bind to lipopolysaccharide and lipoteichoic acid and prevent cell activation by antibiotics. J. Antimicrob. Chemother. 2008, 62, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Chawner, J.A.; Gilbert, P. Adsorption of alexidine and chlorhexidine to Escherichia coli and membrane components. Int. J. Pharm. 1989, 55, 209–215. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [PubMed]
- Chindera, K.; Mahato, M.; Sharma, A.K.; Horsley, H.; Kloc-Muniak, K.; Kamaruzzaman, N.F. The antimicrobial polymer PHMB enters cells and selectively condenses bacterial chromosomes. Sci. Rep. 2016, 6, 23121–23134. [Google Scholar] [CrossRef] [PubMed]
- Girardello, R.; Visconde, M.; Cayo, R.; de Figueiredo, R.; Mori, M.A.D.; Lincopan, N. Diversity of polymyxin resistance mechanisms among Acinetobacter baumannii clinical isolates. Diagn. Microbiol. Infect. Dis. 2017, 87, 37–44. [Google Scholar] [CrossRef] [PubMed]




© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, X.; Meng, L.; Zhang, N.; Zhou, Z. Microscopic Examination of Polymeric Monoguanidine, Hydrochloride-Induced Cell Membrane Damage in Multidrug-Resistant Pseudomonas aeruginosa. Polymers 2017, 9, 398. https://doi.org/10.3390/polym9090398
Cao X, Meng L, Zhang N, Zhou Z. Microscopic Examination of Polymeric Monoguanidine, Hydrochloride-Induced Cell Membrane Damage in Multidrug-Resistant Pseudomonas aeruginosa. Polymers. 2017; 9(9):398. https://doi.org/10.3390/polym9090398
Chicago/Turabian StyleCao, Xun, Lu Meng, Niya Zhang, and Zhongxin Zhou. 2017. "Microscopic Examination of Polymeric Monoguanidine, Hydrochloride-Induced Cell Membrane Damage in Multidrug-Resistant Pseudomonas aeruginosa" Polymers 9, no. 9: 398. https://doi.org/10.3390/polym9090398
APA StyleCao, X., Meng, L., Zhang, N., & Zhou, Z. (2017). Microscopic Examination of Polymeric Monoguanidine, Hydrochloride-Induced Cell Membrane Damage in Multidrug-Resistant Pseudomonas aeruginosa. Polymers, 9(9), 398. https://doi.org/10.3390/polym9090398





