Catalytic Activity of Oxidized Carbon Black and Graphene Oxide for the Crosslinking of Epoxy Resins
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation Procedures
2.2.1. Preparation of Oxidized Graphitic Materials
2.2.2. Dispersion of Graphite-Based Nanofillers and the Cure of the Epoxy Resin
2.3. Characterization Techniques
2.3.1. Elemental Analysis
2.3.2. Wide-Angle X-ray Diffraction
2.3.3. Thermogravimetric Analysis
2.3.4. Infrared Spectroscopy
2.3.5. BET Specific Surface Areas
2.3.6. Rheometry
2.3.7. Differential Scanning Calorimetry
3. Results
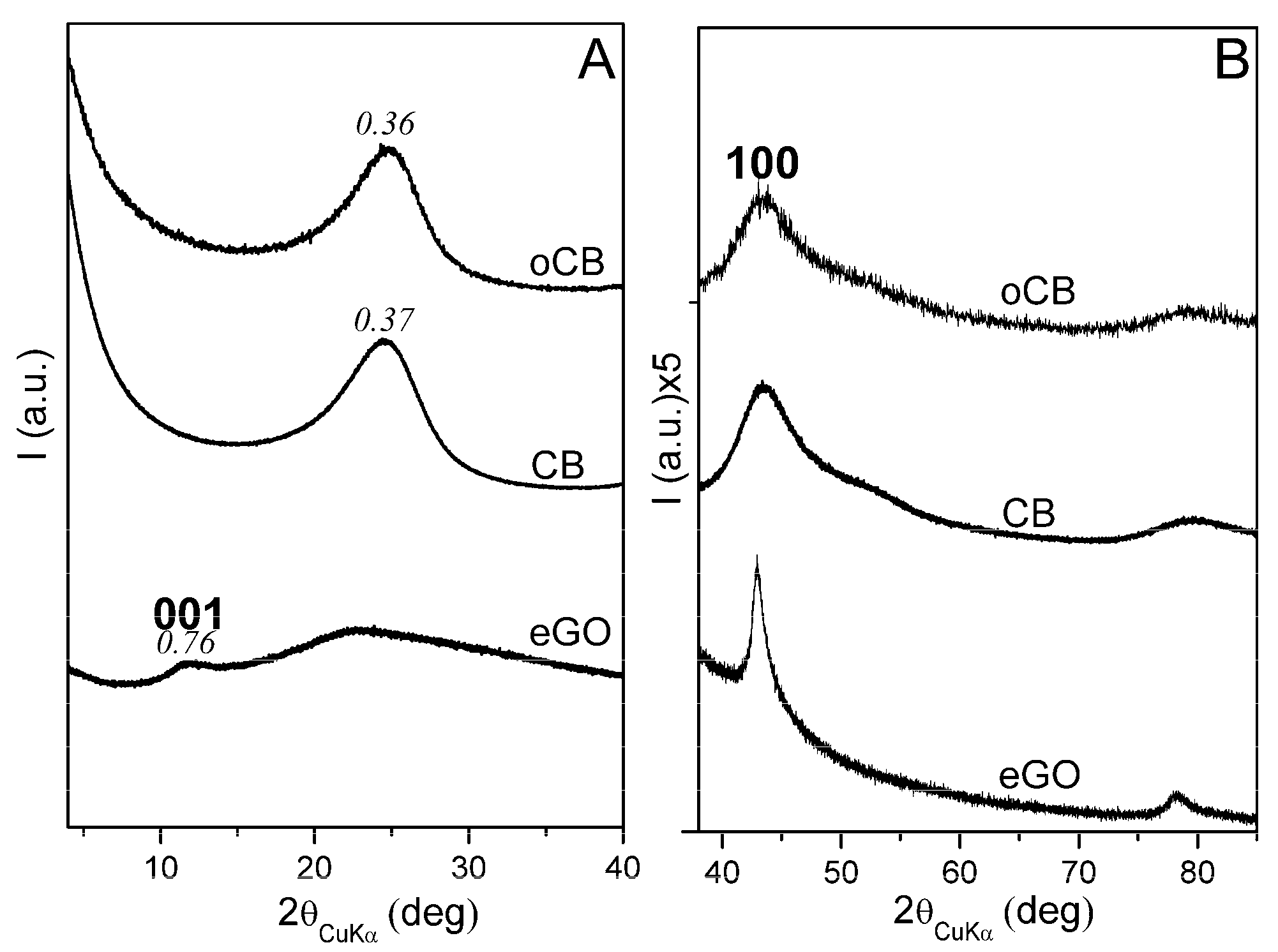
3.1. Characterization of the Carbon Nanofillers
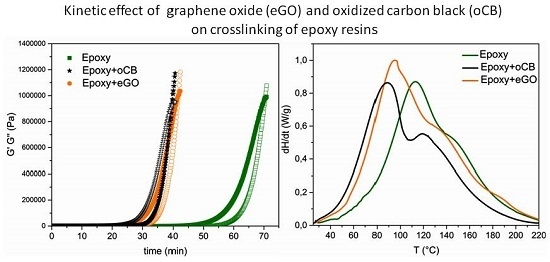
3.2. Gel Time of the Epoxy Resin Crosslinking Reaction by the Rheometer
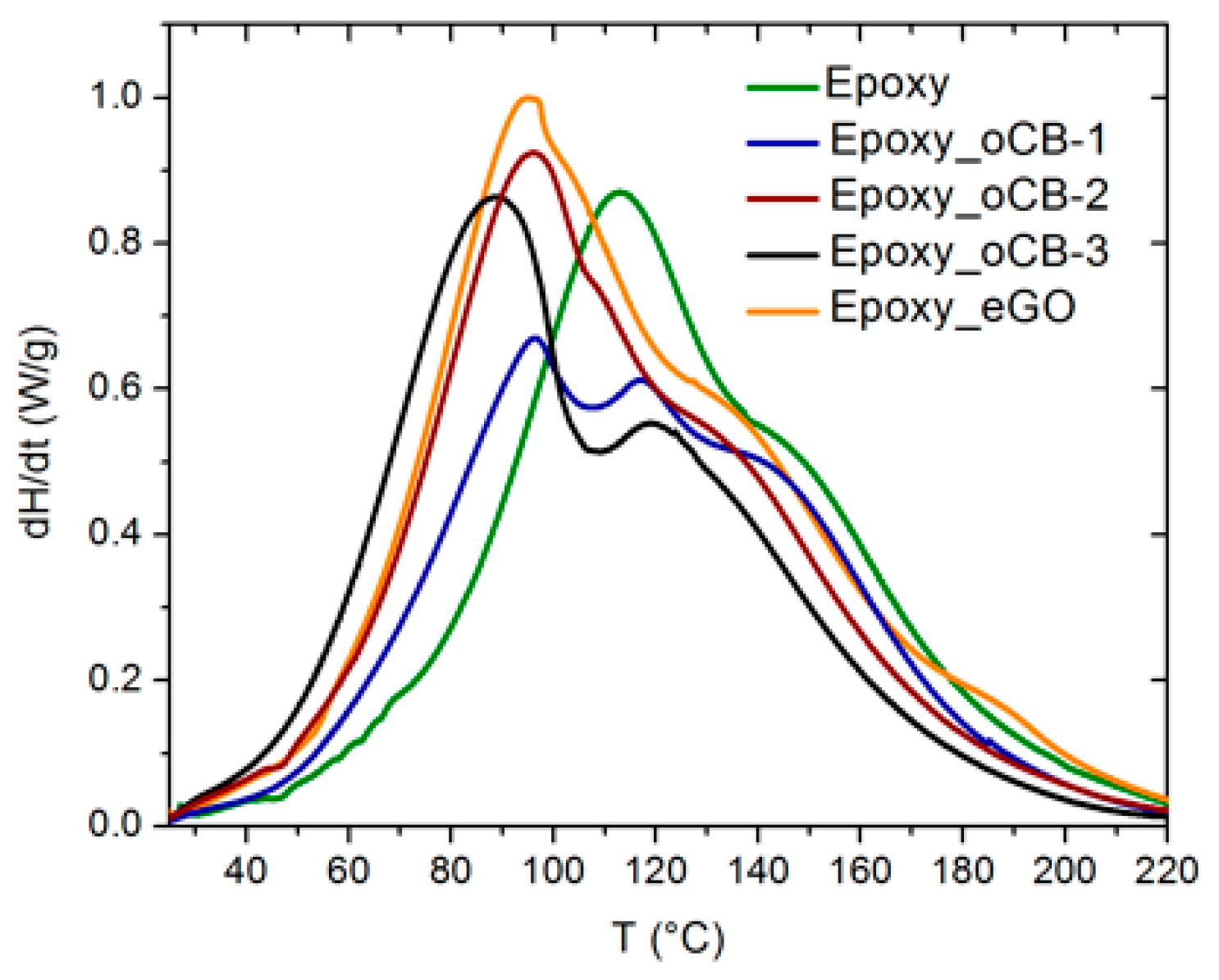
3.3. DSC Heating Scans of Unfilled and Filled Epoxy Resins
3.4. Kinetics of the Epoxy Resin Crosslinking Reaction by Isothermal DSC
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jia, H.-P.; Dreyer, D.R.; Bielawski, C.W. C-H oxidation using graphite oxide. Tetrahedron 2011, 67, 4431–4464. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Jia, H.-P.; Bielawski, C.W. Graphene oxide: A convenient carbocatalyst for facilitating oxidation and hydration reactions. Angew. Chem. Int. Ed. 2010, 49, 6813–6816. [Google Scholar] [CrossRef]
- Boukhvalov, D.V.; Dreyer, D.R.; Bielawski, C.W.; Soon, Y.-W. A computational investigation of the catalytic properties of graphene oxide: Exploring mechanisms by using DFT methods. ChemCatChem 2012, 4, 1844–1849. [Google Scholar] [CrossRef]
- Jia, H.-P.; Dreyer, D.R.; Bielawski, C.W. Graphite Oxide as an auto-tandem oxidation-hydration-aldol coupling catalyst. Adv. Synth. Catal. 2011, 353, 528–532. [Google Scholar] [CrossRef]
- Kumar, A.V.; Rao, K.R. Recyclable graphite oxide catalyzed Friedel–Crafts addition of indoles to α,β-unsaturated ketones. Tetrahedron Lett. 2011, 52, 5188–5191. [Google Scholar] [CrossRef]
- Verma, S.; Mungse, H.P.; Kumar, N.; Choudhary, S.; Jain, S.L.; Sain, B.; Khatri, O.P. Graphene oxide: An efficient and reusable carbocatalyst for aza-Michael addition of amines to activated alkenes. Chem. Commun. 2011, 4, 12673–12675. [Google Scholar] [CrossRef] [PubMed]
- Acocella, M.R.; Mauro, M.; Falivene, L.; Cavallo, L.; Guerra, G. Inverting the diastereoselectivity of the mukaiyama–michael addition with graphite-based catalysts. ACS Catal. 2014, 4, 492–496. [Google Scholar] [CrossRef]
- Acocella, M.R.; De Pascale, M.; Maggio, M.; Guerra, G. Graphite oxide as catalyst for diastereoselective Mukaiyama aldol reaction of 2-(trimethylsilyloxy)furan in solvent free conditions. J. Mol. Catal. A 2015, 408, 237–241. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Jarvis, K.A.; Ferriera, P.J.; Bielawski, C.W. Graphite oxide as a dehydrative polymerization catalyst: A one-step synthesis of carbon-reinforced poly(phenylene methylene) composites. Macromolecules 2011, 44, 7659–7667. [Google Scholar] [CrossRef]
- Mauro, M.; Acocella, M.R.; Esposito Corcione, C.; Maffezzoli, A.; Guerra, G. Catalytic activity of graphite-based nanofillers on cure reaction of epoxy resins. Polymer 2014, 22, 5612–5615. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Alvaro, M.; Concepcion, P.; Fornes, V.; Garcia, H. Graphene oxide as an acid catalyst for the room temperature ring opening of epoxides. Chem. Commun. 2012, 48, 5443–5445. [Google Scholar] [CrossRef] [PubMed]
- Acocella, M.R.; D’Urso, L.; Maggio, M.; Guerra, G. Green regio- and enantioselective aminolysis catalyzed by graphite and graphene oxide under solvent-free conditions. ChemCatChem 2016, 8, 1915–1920. [Google Scholar] [CrossRef]
- Roy, B.; Ghosh, S.; Ghosh, P.; Basu, B. Graphene oxide (GO) or reduced graphene oxide (rGO): Efficient catalysts for one-pot metal-free synthesis of quinoxalines from 2-nitroaniline. Tetrahedron Lett. 2015, 56, 6762–6767. [Google Scholar] [CrossRef]
- Basu, B.; Kundu, S.; Sengupt, D. Graphene oxide as a carbocatalyst: The first example of a one-pot sequential dehydration–hydrothiolation of secondary aryl alcohols. RSC Adv. 2013, 3, 22130. [Google Scholar] [CrossRef]
- Navalon, S.; Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Carbocatalysis by graphene-based materials. Chem. Rev. 2014, 14, 6179–6212. [Google Scholar] [CrossRef] [PubMed]
- Dhakshinamoorthy, A.; Primo, A.; Concepcion, P.; Alvaro, M.; Garcia, H. Doped graphene as a metal-free carbocatalyst for the selective aerobic oxidation of benzylic hydrocarbons, cyclooctane and styrene. Chem. Eur. J. 2013, 19, 7547–7554. [Google Scholar] [CrossRef] [PubMed]
- Primo, A.; Neatu, F.; Florea, M.; Parvulescu, V.; Garcia, H. Graphenes in the absence of metals as carbocatalysts for selective acetylene hydrogenation and alkene hydrogenation. Nat. Commun. 2014, 5, 5291. [Google Scholar]
- Acocella, M.R.; Mauro, M.; Guerra, G. Regio- and Enantioselective friedel–crafts reactions of indoles to epoxides catalyzed by graphene oxide: A green approach. ChemSusChem 2014, 7, 3279–3283. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Chen, C.; Xue, Y.; Wu, J.; Wang, J.; Fan, W. Graphene oxide: An efficient acid catalyst for alcoholysis and esterification reactions. ChemCatChem 2014, 6, 3080–3083. [Google Scholar] [CrossRef]
- Acocella, M.R.; Esposito Corcione, C.; Giuri, A.; Maggio, M.; Maffezzoli, A.; Guerra, G. Graphene oxide as a catalyst for ring opening reactions in amine crosslinking of epoxy resins. RSC Adv. 2016, 6, 23858–23865. [Google Scholar] [CrossRef]
- Sangermano, M.; Tagliaferro, A.; Foix, D.; Castellino, M.; Celasco, E. In situ reduction of graphene oxide in an epoxy resin thermally cured with amine. Macromol. Mater. Eng. 2014, 299, 757–763. [Google Scholar] [CrossRef]
- Carton, A.; Stevenson, W.T.K.; Wang, S. Interfacial reactions in carbon-epoxy composites. Br. Polym. J. 1987, 19, 459. [Google Scholar]
- Nakahara, M.; Takada, T.; Kumagai, H.; Sanada, Y. Surface chemistry of carbon black through curing process of epoxy resin. Carbon 1995, 11, 1537–1544. [Google Scholar] [CrossRef]
- Maggio, M.; Acocella, M.R.; Guerra, G. Intercalation compounds of carbon black. RSC Adv. 2016, 6, 105565–105572. [Google Scholar] [CrossRef]
- Opalicki, M.; Kenny, J.M.; Nicolais, L. Cure kinetics of neat and carbon-fiber-reinforced TGDDM/DDS epoxy systems. J. Appl. Polym. Sci. 1996, 61, 1025–1037. [Google Scholar] [CrossRef]
- Mauro, M.; Cipolletti, V.; Galimberti, M.; Longo, P.; Guerra, G. Chemically reduced graphite oxide with improved shape anisotropy. J. Phys. Chem. C 2012, 116, 24809–24813. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Esposito, C.; Maffezzoli, A. Glass transition in thermosetting clay-nanocomposite polyurethanes. Thermochim. Acta 2009, 485, 43–48. [Google Scholar]
- Laza, J.M.; Julian, C.A.; Larrauri, E.; Rodriguez, M.; Leon, L.M. Thermal scanning rheometer analysis of curing kinetic of an epoxy resin: An amine as curing agent. Polymer 1999, 40, 35–45. [Google Scholar] [CrossRef]
- Henning Winter, H.; Chambon, F. Analysis of linear viscoelasticity of a crosslinking polymer at the gel point. J. Rheol. 1986, 30, 367. [Google Scholar] [CrossRef]
- Flory, P.J. Principles of Polymer Chemistry; Cornell University Press: Ithaca, NY, USA, 1953. [Google Scholar]
- Wei, Z.; Yang, Y.; Hou, Y.; Liu, Y.; He, X.; Deng, S. A new approach towards acid catalysts with high reactivity based on graphene nanosheets. ChemCatChem 2014, 6, 2354–2363. [Google Scholar] [CrossRef]
- He, D.; Kou, Z.; Xiong, Y.; Cheng, K.; Chen, X.; Pan, M.; Mu, S. Simultaneous sulfonation and reduction of graphene oxide as highly efficient supports for metal nanocatalysts. Carbon 2014, 66, 312–319. [Google Scholar] [CrossRef]
- Maffezzoli, A.; Trivisano, A.; Opalicki, M.; Mijovic, J.; Kenny, J.M. Correlation between dielectric and chemorheological properties during cure of epoxy-based composites. J. Mater. Sci. 1994, 29, 800–808. [Google Scholar] [CrossRef]
- Guerrero-Ruiz, A.; Badenes, P.; Rodrı́guez-Ramos, I. Study of some factors affecting the Ru and Pt dispersions over high surface area graphite-supported catalysts. Appl. Catal. A Gen. 1998, 173, 313–321. [Google Scholar] [CrossRef]
- Han, C.D. Developments in Polymer Characterisation-1, 1st ed.; Dawkins, J.V., Ed.; Applied Science: London, UK, 1978; pp. 469–470. [Google Scholar]
- Enns, J.B.; Gillhamm, J.K. Time-temperature-transformation (TTT) cure diagram: Modeling the cure behavior of thermosets. J. Appl. Polym. Sci. 1983, 28, 2567–2591. [Google Scholar] [CrossRef]
- Maffezzoli, A.; Terzi, R. Thermal analysis of visible-light-activated dental composites. Thermochim. Acta 1995, 269, 319–335. [Google Scholar] [CrossRef]
- Ryan, M.E.; Dutta, A. Kinetics of epoxy cure: A rapid technique for kinetic parameter estimation. Polymer 1979, 20, 203–206. [Google Scholar] [CrossRef]
- Kamal, M.R.; Sourour, S. Kinetics and thermal characterization of thermoset cure. Polym. Eng. Sci. 1973, 13, 59–64. [Google Scholar] [CrossRef]
- Cole, K.C. A new approach to modeling the cure kinetics of epoxy/amine thermosetting resins: Mathematical development. Macromolecules 1991, 24, 3093–3097. [Google Scholar] [CrossRef]
- Cole, K.C.; Hechler, J.J.; Noel, D. A new approach to modeling the cure kinetics of epoxy/amine thermosetting resins. 2. Application to a typical system based on bis[4-(diglycidylamino)phenyl]methane and bis(4-aminophenyl) sulfone. Macromolecules 1991, 24, 3098–3110. [Google Scholar] [CrossRef]
- Kenny, J.M.; Apicella, A.; Nicolais, L. A model for the thermal and chemorheological behavior of thermosets. I: Processing of epoxy-based composites. Polym. Eng. Sci. 1989, 29, 973–983. [Google Scholar] [CrossRef]
- Trivisano, A.; Maffezzoli, A.; Kenny, J.M.; Nicolais, L. Mathematical modeling of the pultrusion of epoxy based composites. Adv. Polym. Technol. 1990, 4, 251–264. [Google Scholar] [CrossRef]
- Xie, H.; Liu, B.; Yuan, Z.; Shen, J.; Cheng, R. Cure kinetics of carbon nanotube/tetrafunctional epoxy nanocomposites by isothermal differential scanning calorimetry. J. Polym. Sci. Part B 2004, 42, 3701–3712. [Google Scholar] [CrossRef]


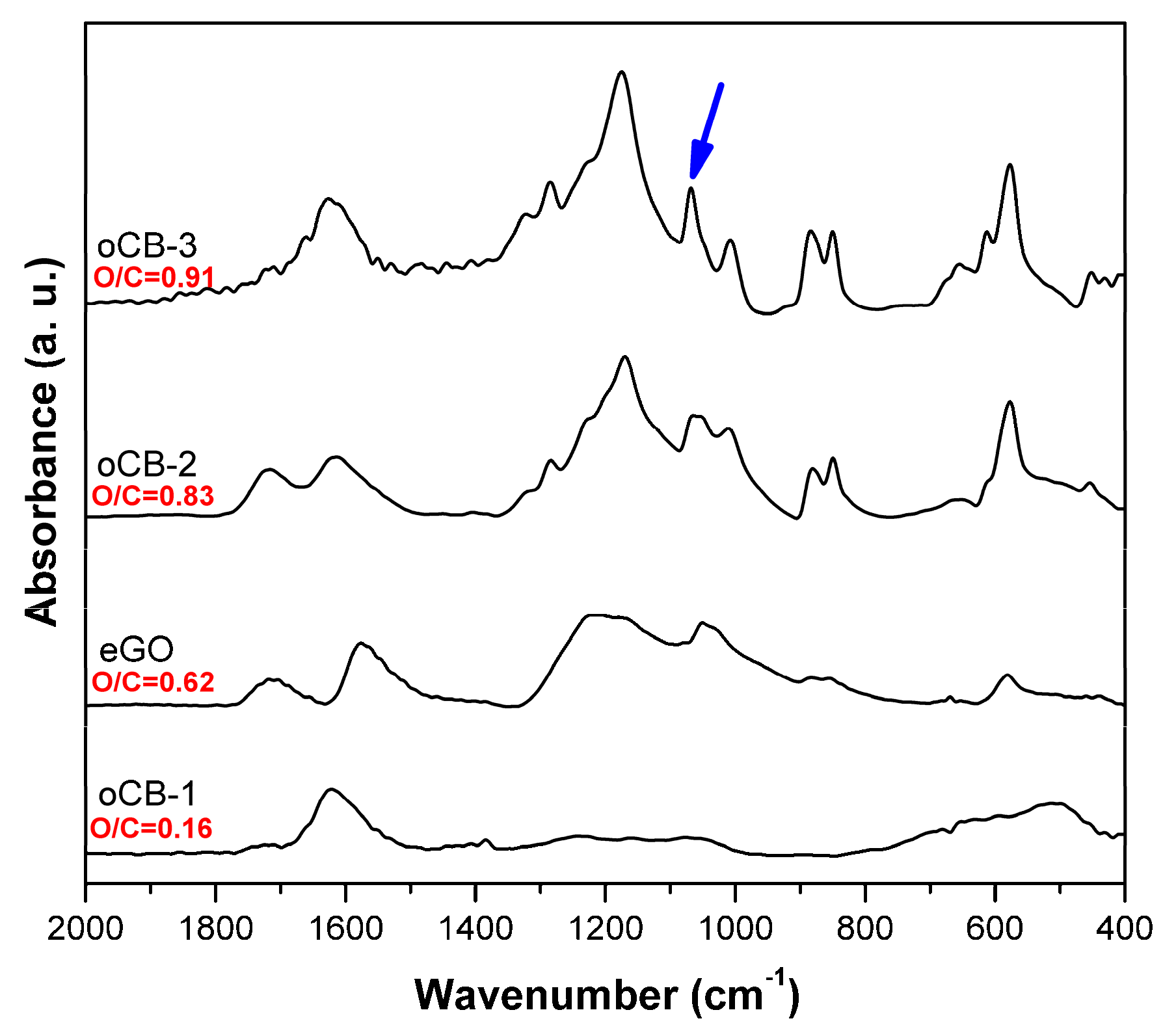



| Nanofiller | C (wt %) | H (wt %) | O (wt %) | S (wt %) | O/C |
|---|---|---|---|---|---|
| oCB-1 | 85.9 | 0.2 | 13.75 | <0.2 | 0.16 |
| oCB-2 | 50.3 | 2.3 | 41.7 | 5.4 | 0.83 |
| oCB-3 | 40.8 | 1.73 | 37.42 | 9.9 | 0.91 |
| eGO | 59.4 | 0.6 | 37.1 | 2.6 | 0.62 |
| Sample | tgel [min] |
|---|---|
| Epoxy | 70.5 ± 5.6 |
| Epoxy oCB-1 | 62.9 ± 4.4 |
| Epoxy oCB-2 | 47.9 ± 3.6 |
| Epoxy oCB-3 | 39.7 ± 2.0 |
| Epoxy_eGO | 41.7 ± 2.1 |
| Sample | ∆HU (J/g) | Tpeak °C | Amine catalyzed fraction (%) |
|---|---|---|---|
| Epoxy | 313.9 ± 1.1 | 113.7 ± 0.9 | 0 |
| Epoxy + oCB-1 (O/C = 0.16) | 327.5 ± 2.2 | 96.5 ± 0.8 | 12 |
| Epoxy + oCB-2 (O/C = 0.83) | 383.8 ± 1.1 | 96.2 ± 0.7 | 34 |
| Epoxy + oCB-3 (O/C = 0.91) | 352.5 ± 1.7 | 88.7 ± 1.1 | 56 |
| Epoxy + eGO (O/C = 0.62) | 374.4 ± 2.3 | 95.0 ± 0.7 | 29 |
| Sample | ∆H (J/g) | tpeak (s) | ∆Hresidual (J/g) | Tg (°C) |
|---|---|---|---|---|
| Epoxy | 158.0 ± 2.0 | 508 ± 1 | 74.6 ± 1.1 | 54.7 |
| Epoxy + oCB-1 | 306.0 ± 18.0 | 504 ± 19 | 18.1 ± 0.9 | 66.3 |
| Epoxy + oCB-2 | 297.7 ± 5.7 | 393 ± 6 | 62.6 ± 1.3 | 59.4 |
| Epoxy + oCB-3 | 337.0 ± 27.0 | 249 ± 63 | 13.4 ± 0.4 | 64.6 |
| Epoxy + eGO | 337.0 ± 2.0 | 306 ± 1 | 26.2 ± 0.8 | 63.2 |
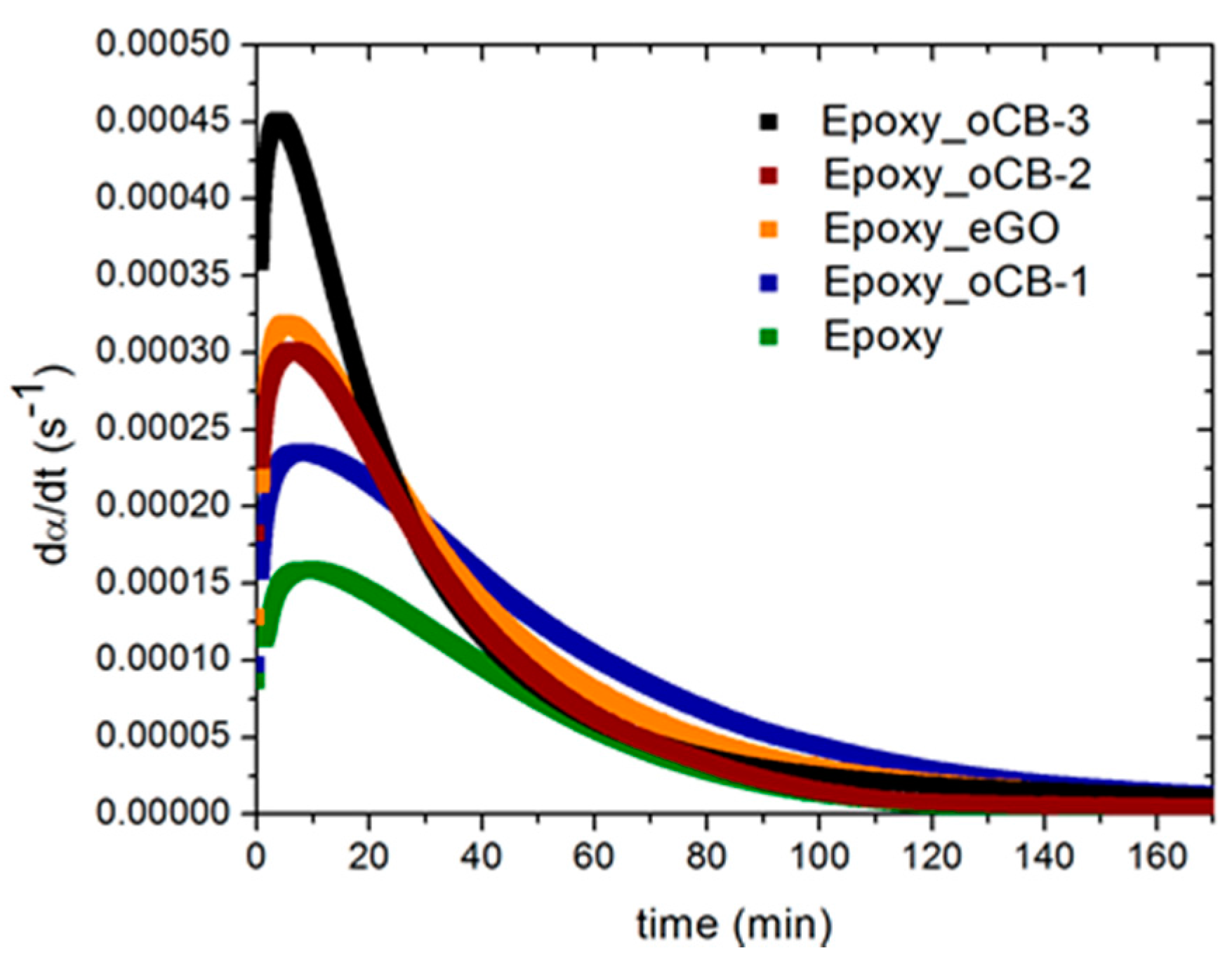
| Sample | (dα/dt)peak (s−1) | αmax | m | n | k1 (s−1) × 106 | k2 (s−1) × 106 |
|---|---|---|---|---|---|---|
| Epoxy | 1.57 × 10−4 | 0.51 | 0.51 | 1.06 | 204 | 650 |
| Epoxy + oCB-1 | 2.33 × 10−4 | 0.94 | 0.21 | 1.07 | 740 | 340 |
| Epoxy + OCB-2 | 3.02 × 10−4 | 0.78 | 0.39 | 1.22 | 230 | 630 |
| Epoxy + oCB-3 | 4.49 × 10−4 | 0.96 | 0.30 | 1.58 | 230 | 670 |
| Epoxy + eGO | 3.18 × 10−4 | 0.90 | 0.20 | 1.22 | 990 | 490 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acocella, M.R.; Esposito Corcione, C.; Giuri, A.; Maggio, M.; Guerra, G.; Maffezzoli, A. Catalytic Activity of Oxidized Carbon Black and Graphene Oxide for the Crosslinking of Epoxy Resins. Polymers 2017, 9, 133. https://doi.org/10.3390/polym9040133
Acocella MR, Esposito Corcione C, Giuri A, Maggio M, Guerra G, Maffezzoli A. Catalytic Activity of Oxidized Carbon Black and Graphene Oxide for the Crosslinking of Epoxy Resins. Polymers. 2017; 9(4):133. https://doi.org/10.3390/polym9040133
Chicago/Turabian StyleAcocella, Maria Rosaria, Carola Esposito Corcione, Antonella Giuri, Mario Maggio, Gaetano Guerra, and Alfonso Maffezzoli. 2017. "Catalytic Activity of Oxidized Carbon Black and Graphene Oxide for the Crosslinking of Epoxy Resins" Polymers 9, no. 4: 133. https://doi.org/10.3390/polym9040133
APA StyleAcocella, M. R., Esposito Corcione, C., Giuri, A., Maggio, M., Guerra, G., & Maffezzoli, A. (2017). Catalytic Activity of Oxidized Carbon Black and Graphene Oxide for the Crosslinking of Epoxy Resins. Polymers, 9(4), 133. https://doi.org/10.3390/polym9040133