A Novel Biomimetic Approach to Repair Enamel Cracks/Carious Damages and to Reseal Dentinal Tubules by Amorphous Polyphosphate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of Ca-polyP/retinyl Acetate Microparticles
2.3. Toothpaste Test Samples
2.4. Human Mesenchymal Stem Cells
2.5. Cell Proliferation/Cell Viability Assays
2.6. Gene Expression Studies
2.7. Bacterial Studies
2.8. Tooth Samples
2.9. Cutting of the Tooth Samples
2.10. Microscopic Inspections
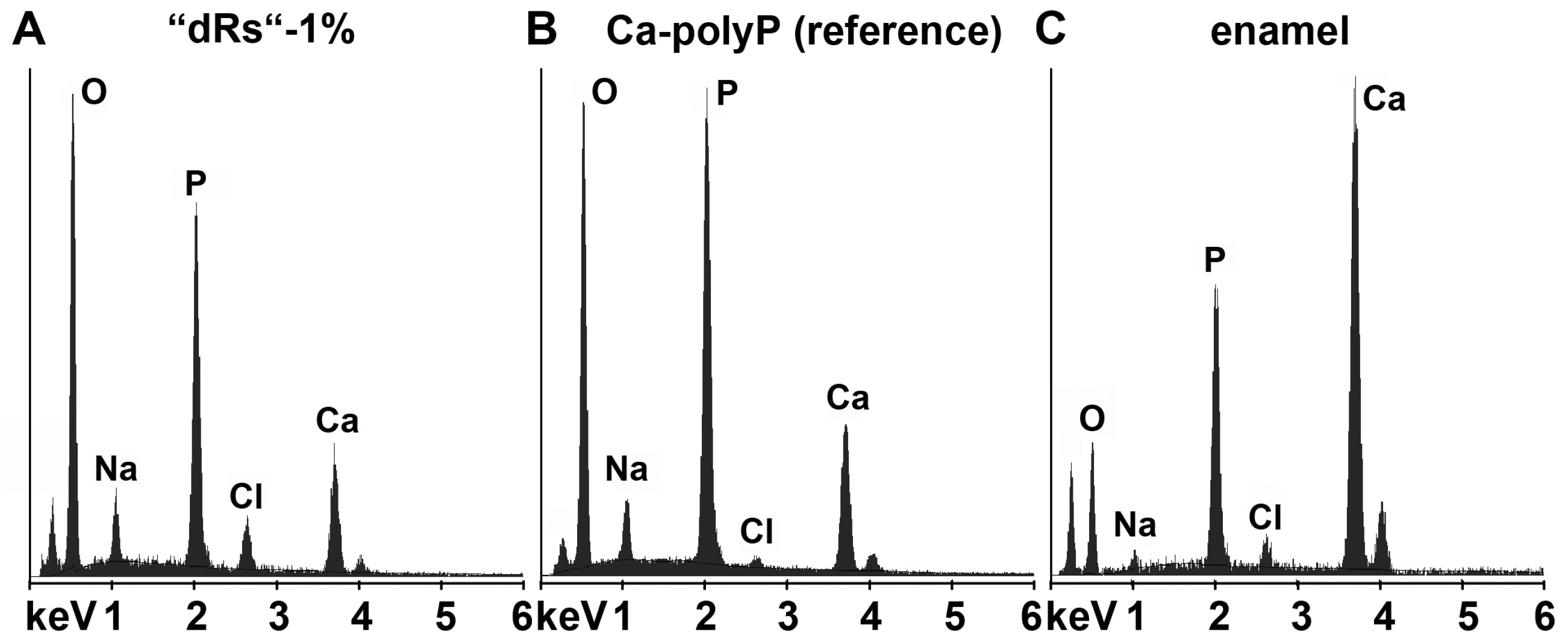
2.11. Energy Dispersive X-ray Spectroscopy
2.12. Statistical Analysis
3. Results
3.1. Fabrication of Ca-polyP/Retinol Microparticles
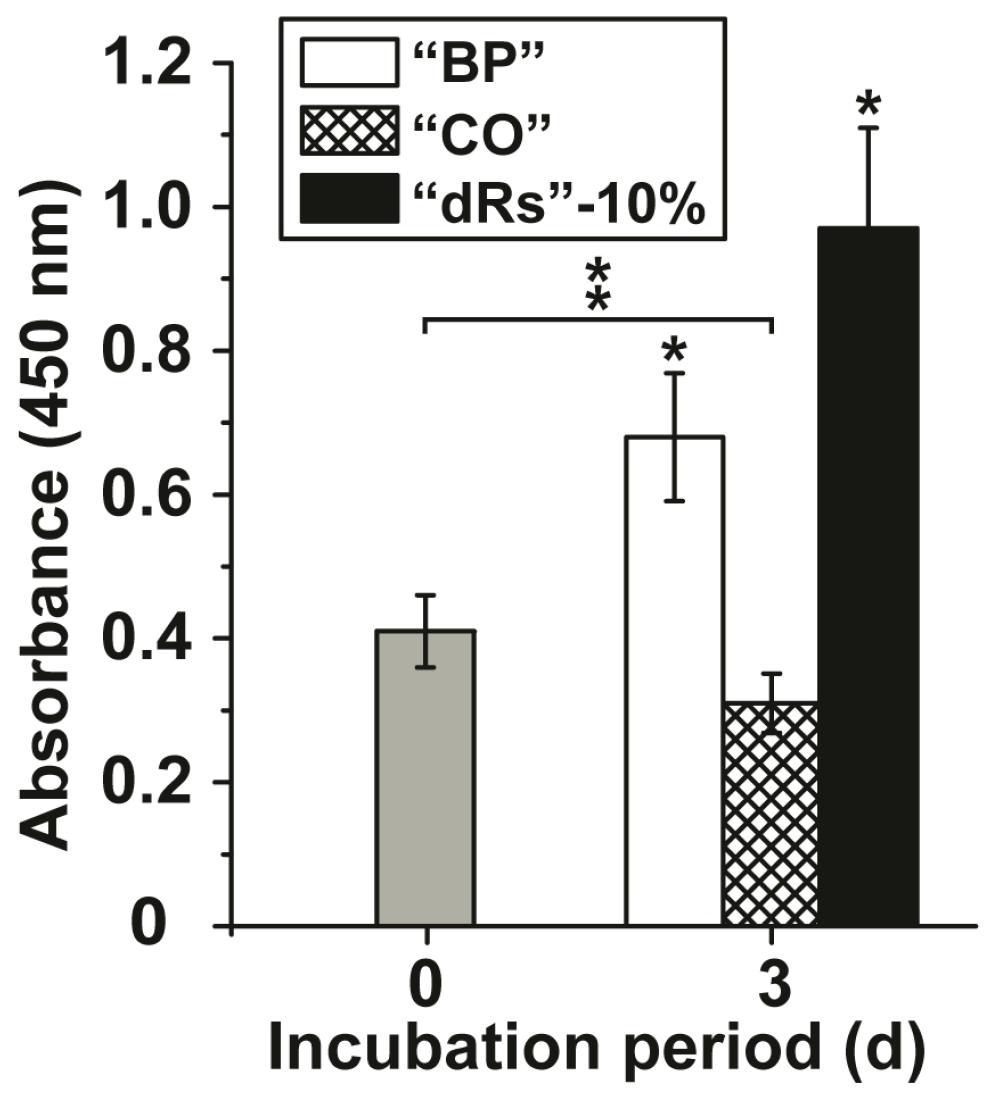
3.2. Effect of the Microparticles on Growth of MSC
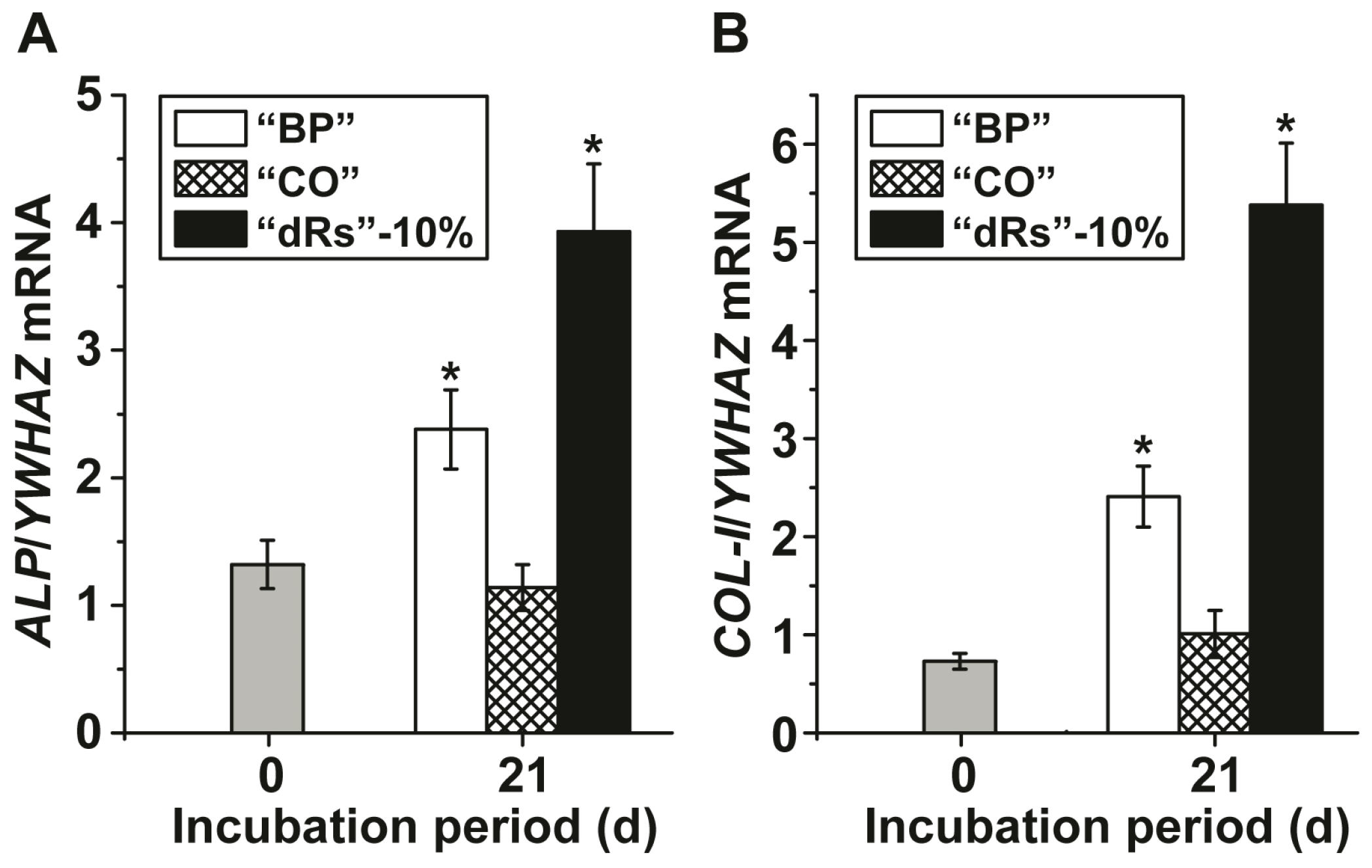
3.3. Alteration of Gene Expression in Response to the Different Paste Formulae
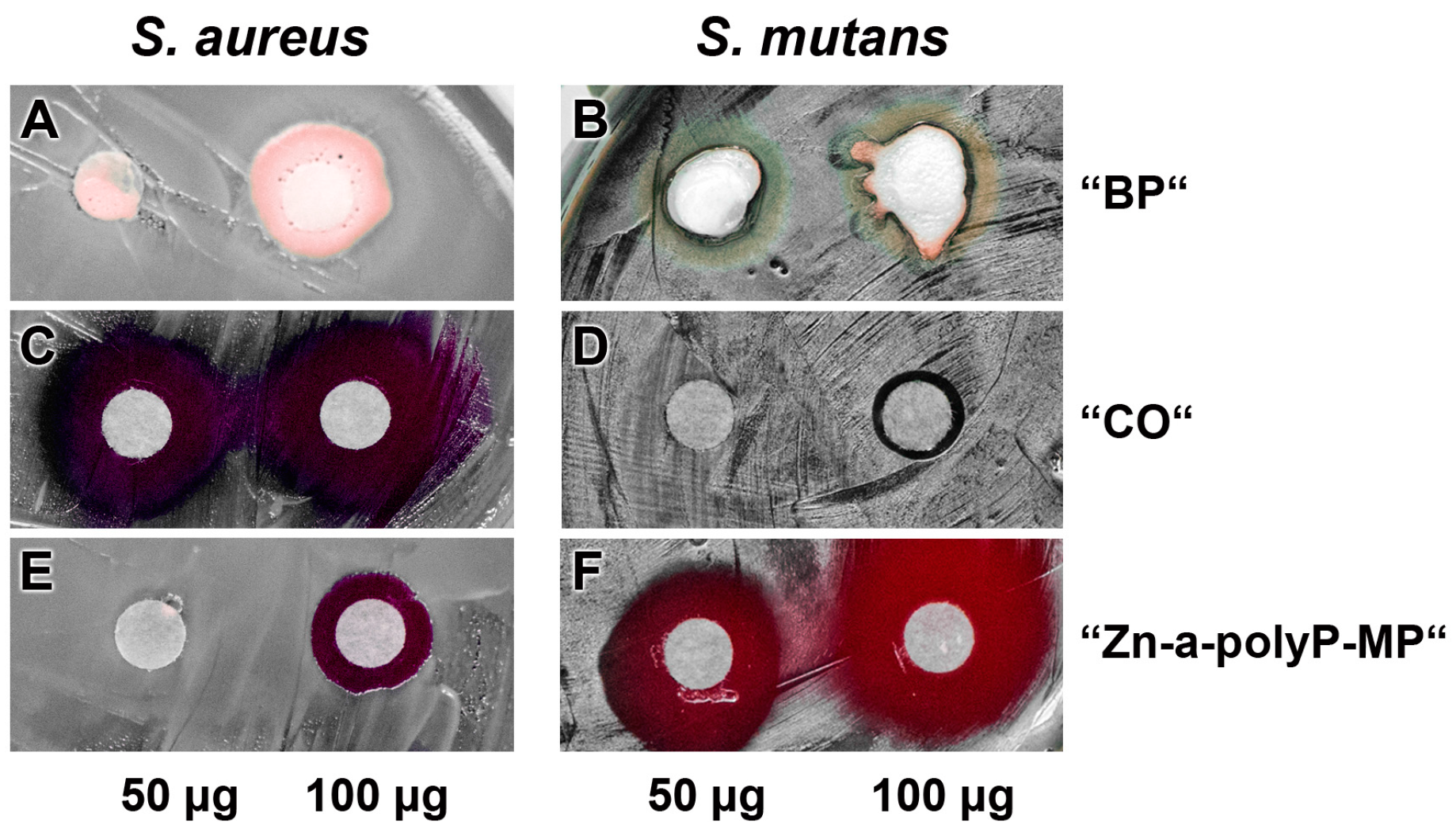
3.4. Antibacterial Activity
3.5. Occlusion of Tooth Damages
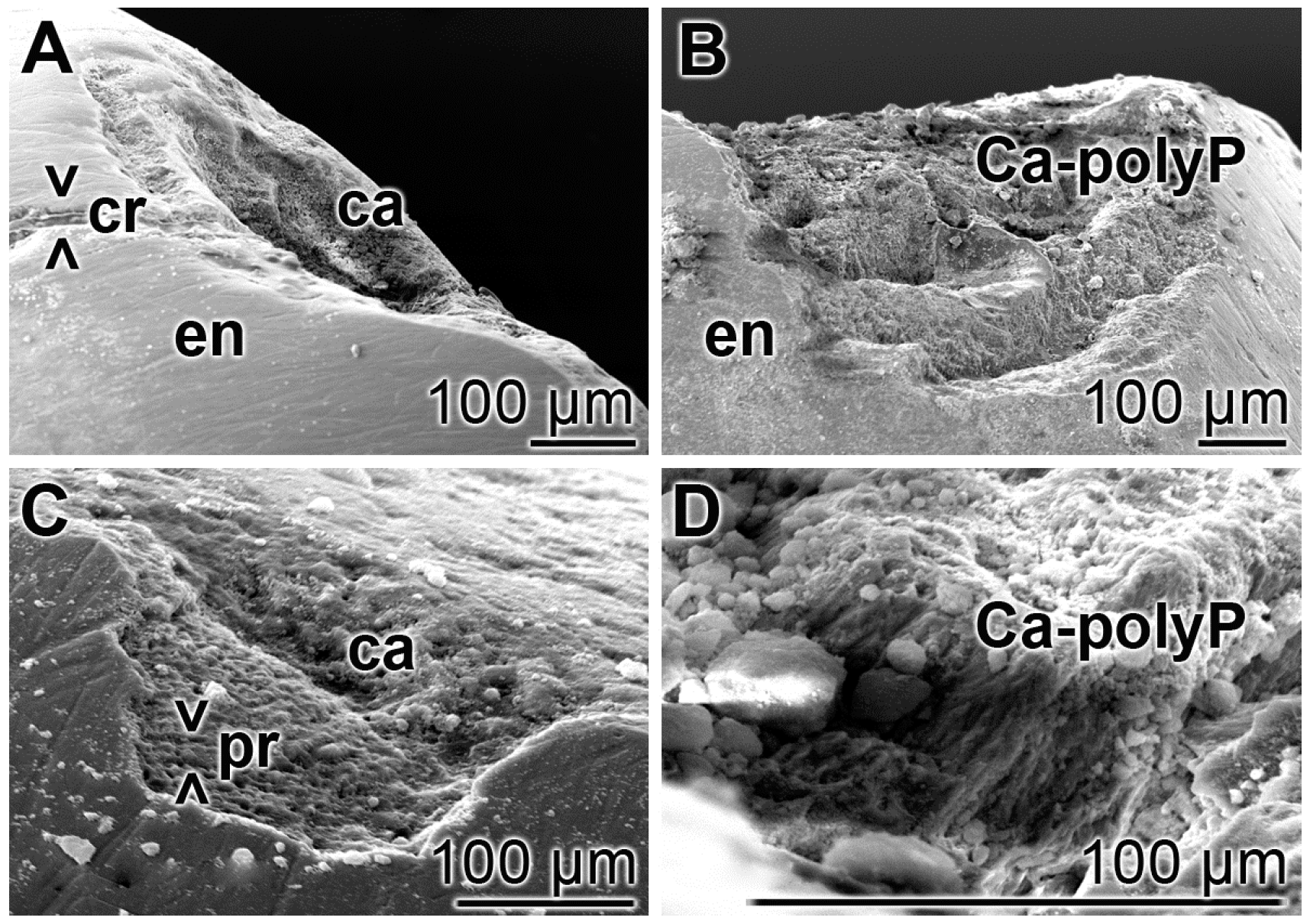
3.6. Repair of Carious Tooth Lesions in the Enamel
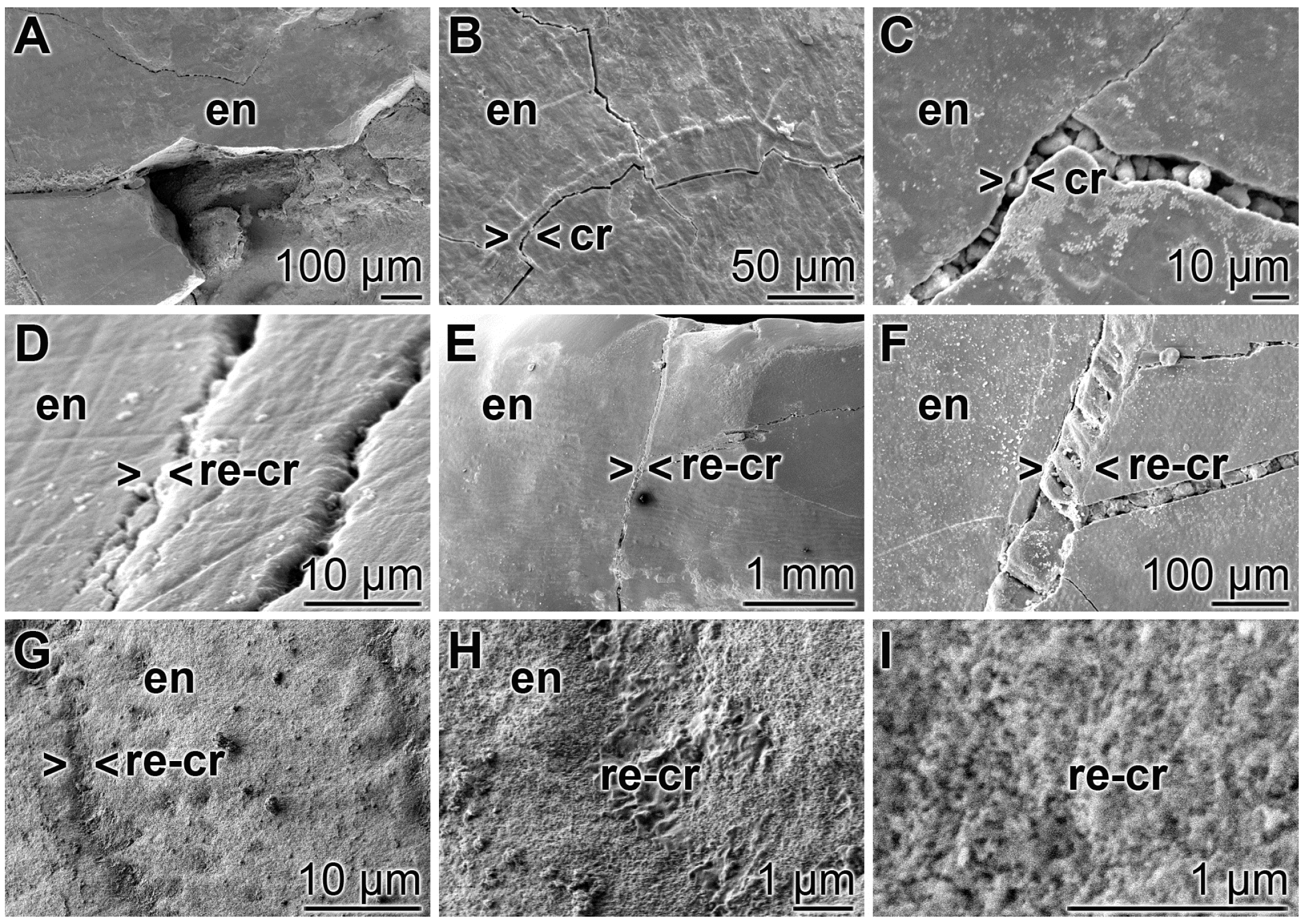
3.7. Repair of Cracks in the Enamel Region
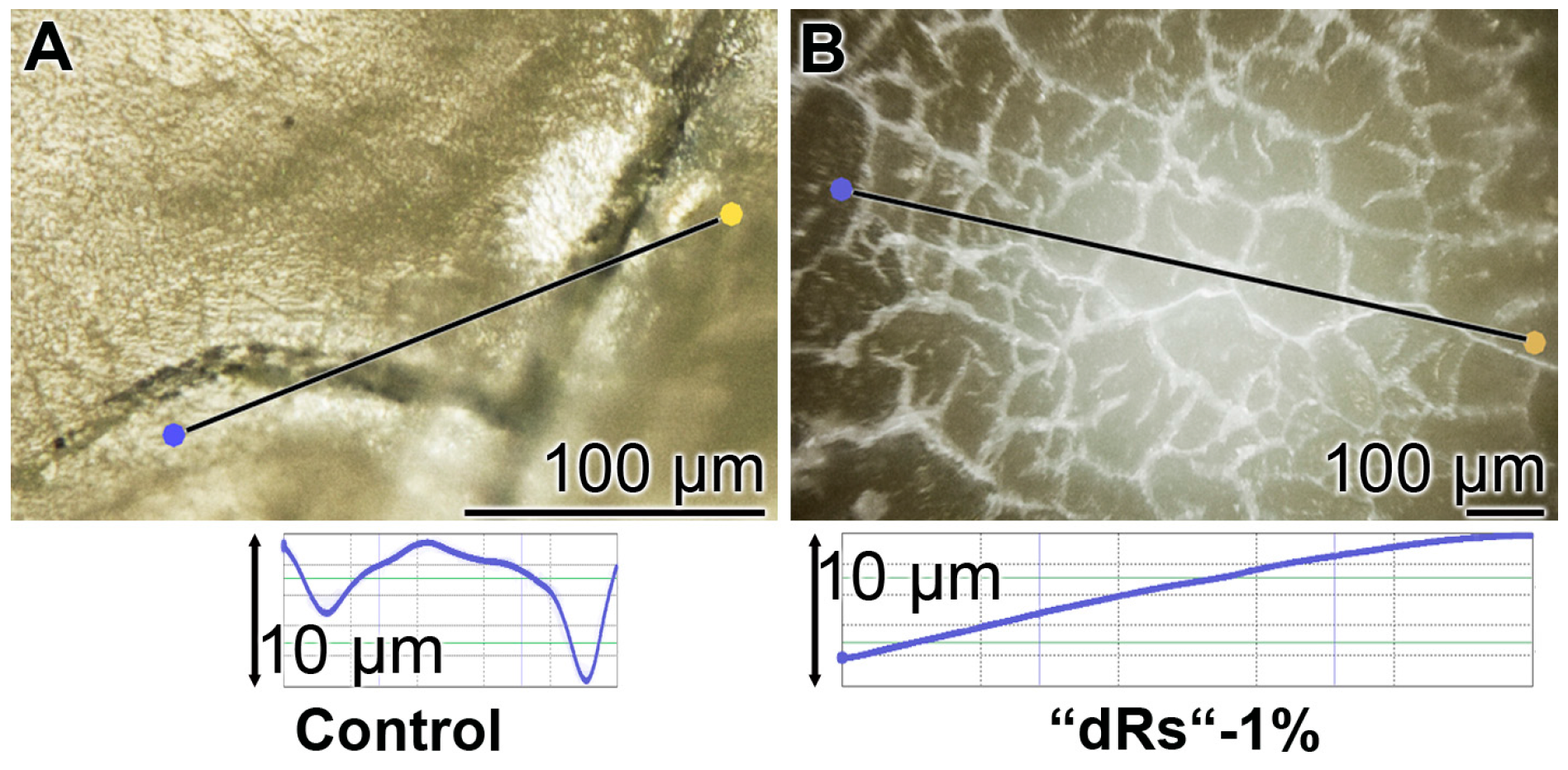
3.8. Resealing of Cracks and the Surface Roughness of the Closed Cracks
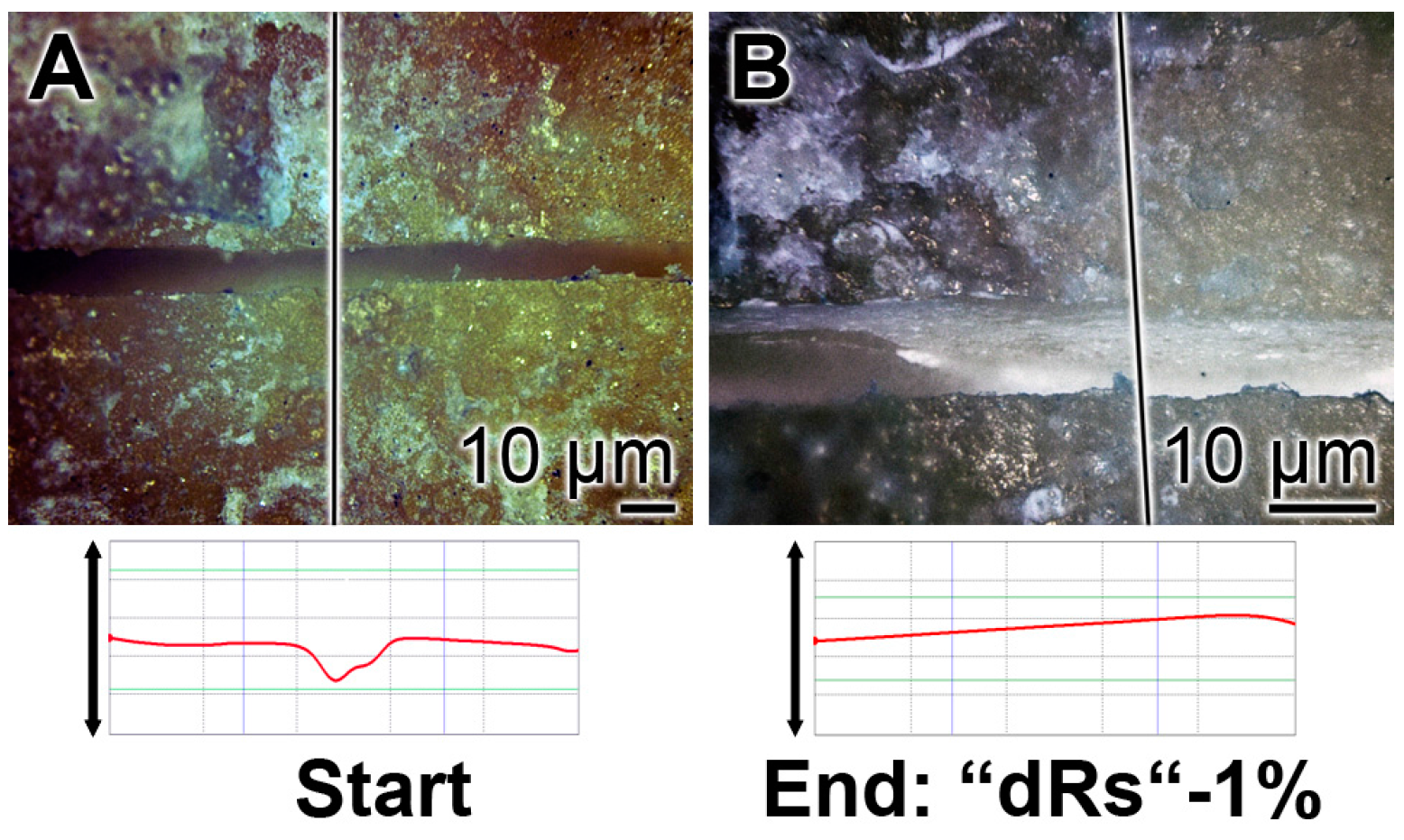
3.9. Stability of the Sealing by “dRs”-1%
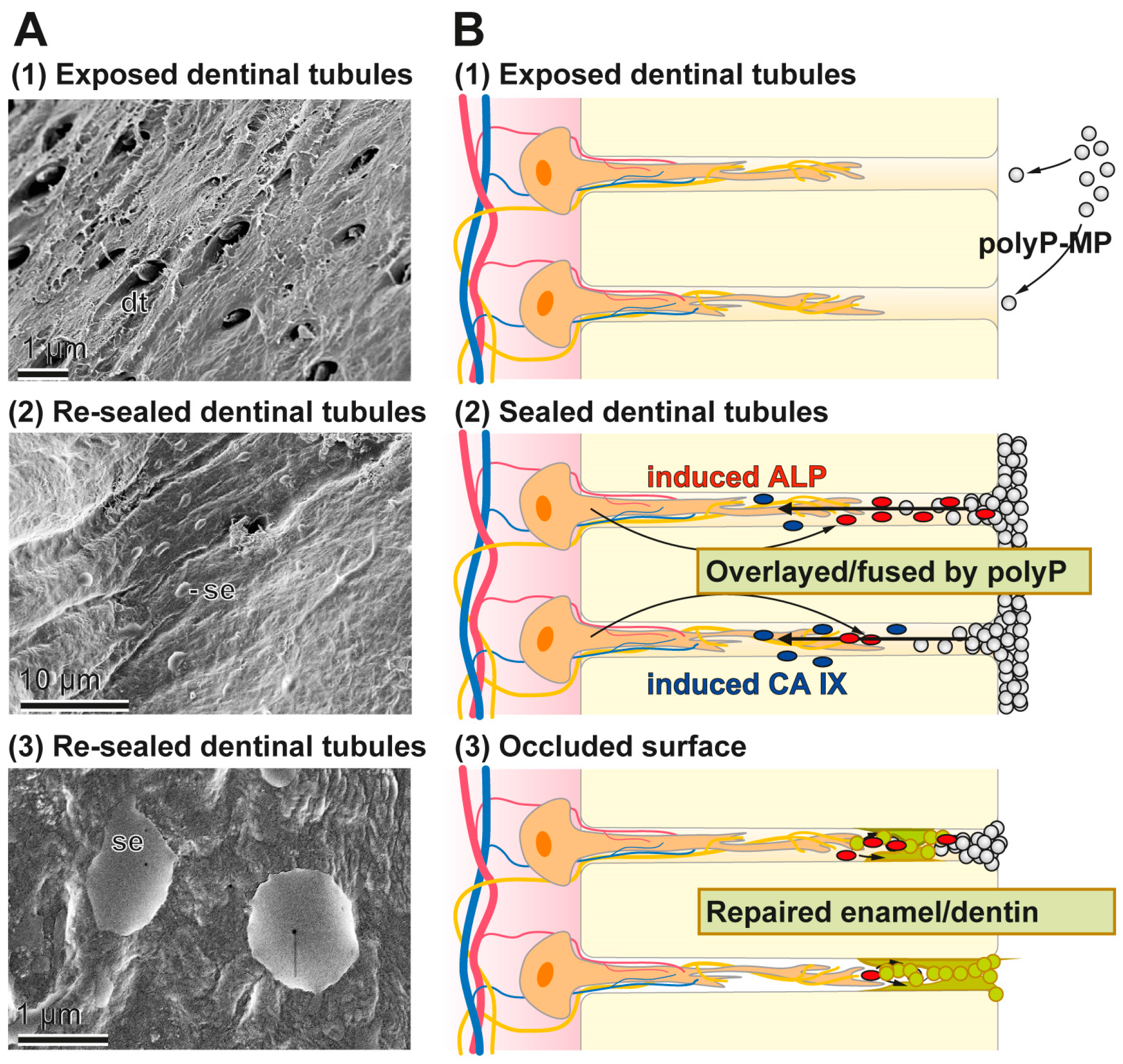
3.10. Occlusion of Exposed Dentinal Tubules
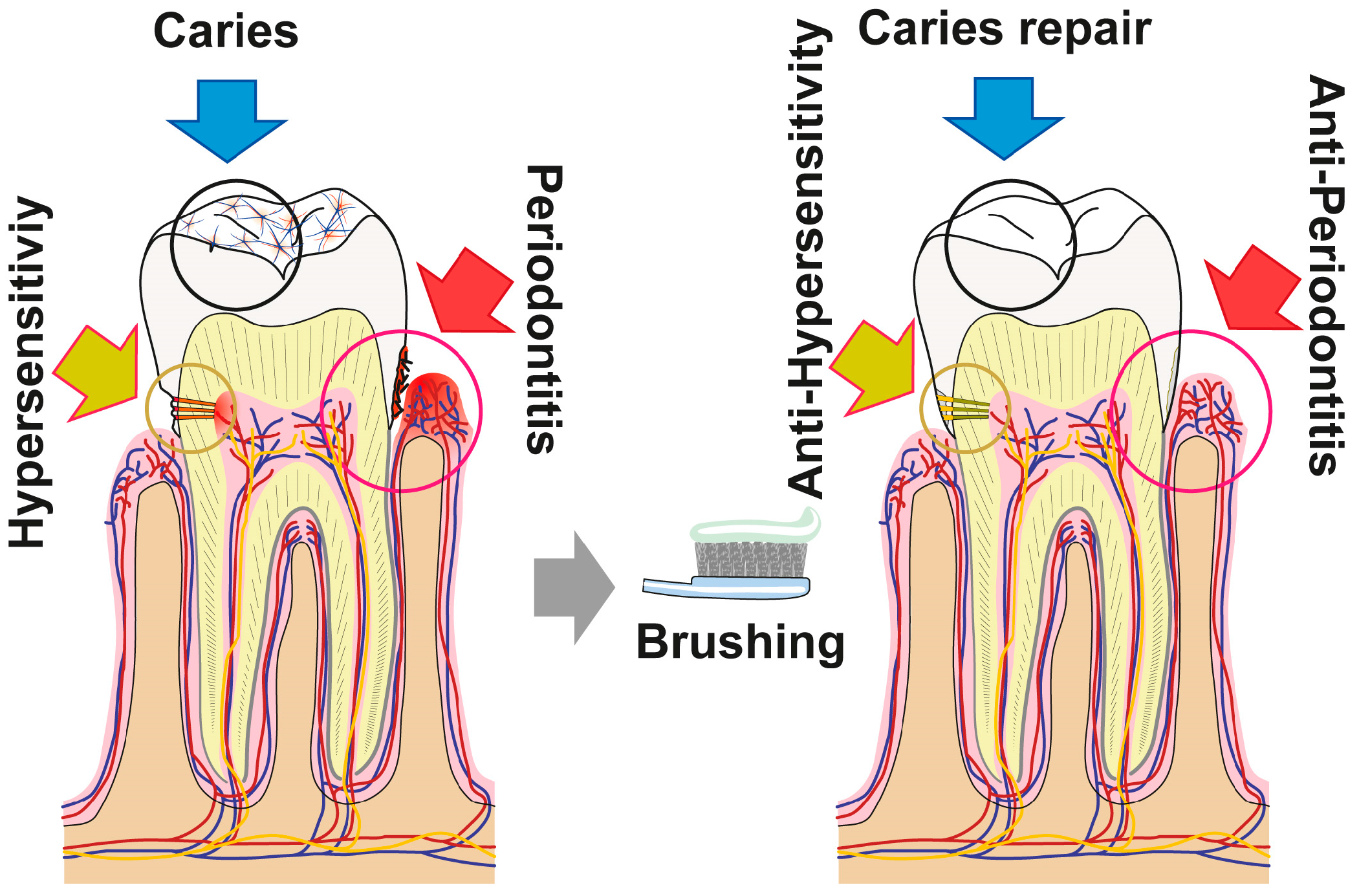
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Slootweg, P.J. Dental Pathology—A Practical Introduction; Springer: Berlin, Germany, 2007. [Google Scholar]
- Teruel, J.D.; Alcolea, A.; Hernández, A.; Ruiz, A.J. Comparison of chemical composition of enamel and dentine in human, bovine, porcine and ovine teeth. Arch. Oral Biol. 2015, 60, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Amarasiriwardena, D.; Goodman, A.H. Application of laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS) to investigate trace metal spatial distributions in human tooth enamel and dentine growth layers and pulp. Anal. Bioanal. Chem. 2004, 378, 1608–1615. [Google Scholar] [CrossRef] [PubMed]
- He, L.H.; Swain, M.V. Understanding the mechanical behaviour of human enamel from its structural and compositional characteristics. J. Mech. Behav. Biomed. Mater. 2008, 1, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.D.; Wang, Y.J.; Wei, K.; Zhang, S.H.; Shi, X.T. Self-organization of hydroxyapatite nanorods through oriented attachment. Biomaterials 2007, 28, 2275–2280. [Google Scholar] [CrossRef] [PubMed]
- Moradian-Oldak, J. Protein-mediated enamel mineralization. Front. Biosci. 2012, 17, 1996–2023. [Google Scholar] [CrossRef]
- Uskoković, V.; Kim, M.K.; Li, W.; Habelitz, S. Enzymatic processing of amelogenin during continuous crystallization of apatite. J. Mater. Res. 2008, 23, 3184–3195. [Google Scholar]
- Brunton, P.A.; Davies, R.P.; Burke, J.L.; Smith, A.; Aggeli, A.; Brookes, S.J.; Kirkham, J. Treatment of early caries lesions using biomimetic self-assembling peptides—A clinical safety trial. Br. Dent. J. 2013, 215, E6. [Google Scholar] [CrossRef] [PubMed]
- Matson, J.B.; Zha, R.H.; Stupp, S.I. Peptide self-assembly for crafting functional biological materials. Curr. Opin. Solid State Mater. Sci. 2011, 15, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Schröder, H.C.; Tolba, E.; Neufurth, M.; Diehl-Seifert, B.; Wang, X.H. Mineralization of bone-related SaOS-2 cells under physiological hypoxic conditions. FEBS J. 2016, 283, 74–87. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Neufurth, M.; Huang, J.; Wang, K.; Feng, Q.; Schröder, H.C.; Diehl-Seifert, B.; Muñoz-Espí, R.; Wang, X.H. Non-enzymatic transformation of amorphous CaCO3 into calcium phosphate mineral after exposure to sodium phosphate in vitro: Implications for in vivo hydroxyapatite bone formation. ChemBioChem 2015, 16, 1323–1332. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Schröder, H.C.; Müller, W.E.G. Enzymatically synthesized inorganic polymers as morphogenetically active bone scaffolds: Application in regenerative medicine. Int. Rev. Cell Mol. Biol. 2014, 313, 27–77. [Google Scholar] [PubMed]
- Müller, W.E.G.; Tolba, E.; Schröder, H.C.; Wang, X.H. Polyphosphate: A morphogenetically active implant material serving as metabolic fuel for bone regeneration. Macromol. Biosci. 2015, 15, 1182–1197. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Schröder, H.C.; Müller, W.E.G. Polyphosphate as a metabolic fuel in Metazoa: A foundational breakthrough invention for biomedical applications. Biotechnol. J. 2016, 11, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, J.H.; Choi, S.H.; Smith, S.A. Polyphosphate: An ancient molecule that links platelets, coagulation, and inflammation. Blood 2012, 119, 5972–5979. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Tolba, E.; Schröder, H.C.; Wang, S.; Glaßer, G.; Muñoz-Espí, R.; Link, T.; Wang, X.H. A new polyphosphate calcium material with morphogenetic activity. Mater. Lett. 2015, 148, 163–166. [Google Scholar] [CrossRef]
- Müller, W.E.G.; Neufurth, M.; Tolba, E.; Wang, S.F.; Geurtsen, W.; Feng, Q.; Schröder, H.C.; Wang, X.H. A biomimetic approach to ameliorate dental hypersensitivity by amorphous polyphosphate microparticles. Dent. Mater. 2016, 32, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Tolba, E.; Schröder, H.C.; Diehl-Seifert, B.; Wang, X.H. Retinol encapsulated into amorphous Ca2+ polyphosphate nanospheres acts synergistically in MC3T3-E1 cells. Eur. J. Pharm. Biopharm. 2015, 93, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Beertsen, W.; Van den Bos, T. Alkaline phosphatase induces the deposition of calcified layers in relation to dentin: An in vitro study to mimic the formation of afibrillar acellular cementum. J. Dent. Res. 1991, 70, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, L.; Demarco, F.F.; Zhang, Z.; Araujo, F.B.; Shi, S.; Nör, J.E. Dentin-derived BMP-2 and odontoblast differentiation. J. Dent. Res. 2010, 89, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Hosseinkhani, H.; Hosseinkhani, M.; Tian, F.; Kobayashi, H.; Tabata, Y. Bone regeneration on a collagen sponge self-assembled peptide-amphiphile nanofiber hybrid scaffold. Tissue Eng. 2007, 13, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T.; Bizenjima, T.; Ishii, Y.; Imamura, K.; Suzuki, E.; Seshima, F.; Saito, A. Enhanced healing of surgical periodontal defects in rats following application of a self-assembling peptide nanofiber hydrogel. J. Clin. Periodontol. 2016, 43, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Marcy, J.A.; Kraft, A.A.; Hotchkiss, D.K.; Molins, R.A.; Olson, D.G.; Walker, H.W.; Merkenich, K. Effects of selected commercial phosphate products on the natural bacterial flora of a cooked meat system. J. Food Sci. 1988, 53, 391–393. [Google Scholar] [CrossRef]
- Olsen, D.G.; Merkenich, K. Effect of inorganic polyphosphates on ground beef characteristics: Microbiological effects on frozen beef patties. J. Food Sci. 1987, 52, 46–49. [Google Scholar]
- Obritsch, J.A.; Ryu, D.; Lampila, L.E.; Bullerman, L.B. Antibacterial effects of long-chain polyphosphates on selected spoilage and pathogenic bacteria. J. Food Prot. 2008, 71, 1401–1405. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.H.; Park, J.H.; Lee, J.Y. Antibacterial action of polyphosphate on porphyromonas gingivalis. Antimicrob. Agents Chemother. 2011, 55, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Banas, J.A. Virulence properties of streptococcus mutans. Front. Biosci. 2004, 9, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Brighenti, F.L.; Takeshita, E.M.; Sant’ana, C.O.; Buzalaf, M.A.; Delbem, A.C. Effect of low fluoride acidic dentifrices on dental remineralization. Braz. Dent. J. 2013, 24, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Afonso, R.L.; Pessan, J.P.; Igreja, B.B.; Cantagallo, C.F.; Danelon, M.; Delbem, A.C. In situ protocol for the determination of dose-response effect of low-fluoride dentifrices on enamel remineralization. J. Appl. Oral Sci. 2013, 21, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Park, B.K.; Gonzales, E.L.; Yang, S.M.; Bang, M.; Choi, C.S.; Shin, C.Y. Effects of triclosan on neural stem cell viability and survival. Biomol. Ther. 2016, 24, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A. Transcript of FDA media briefing on the FDA’s proposed rule amending the tentative final monograph for over-the-counter consumer antiseptics. 2013. Available online: http://www.fda.gov/downloads/NewsEvents/Newsroom/MediaTranscripts/UCM378989.pdf (accessed on 14 February 2016).
- Suller, M.T.; Russell, A.D. Triclosan and antibiotic resistance in Staphylococcus aureus. J. Antimicrob. Chemother. 2000, 46, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Andujar, M.B.; Couble, P.; Couble, M.L.; Magloire, H. Differential expression of type I and type III collagen genes during tooth development. Development 1991, 111, 691–698. [Google Scholar] [PubMed]
- Subramanyam, G.B.; Parrish, D.B. Colorimetric reagents for determining vitamin A in feeds and foods. J. Assoc. Off. Anal. Chem. 1976, 59, 1125–1130. [Google Scholar] [PubMed]
- Kulakovskaya, T.; Pavlov, E.; Dedkova, E.N. Inorganic Polyphosphates in Eukaryotic Cells; Springer International Publishing: Cham, Switzerland, 2016; pp. 3–14. [Google Scholar]
- Wiens, M.; Wang, X.H.; Schloßmacher, U.; Lieberwirth, I.; Glasser, G.; Ushijima, H.; Schröder, H.C.; Müller, W.E.G. Osteogenic potential of bio-silica on human osteoblast-like (SaOS-2) cells. Calcif. Tissue Int. 2010, 87, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.G.; Wang, X.H.; Diehl-Seifert, B.; Kropf, K.; Schloßmacher, U.; Lieberwirth, I.; Glasser, G.; Wiens, M.; Schröder, H.C. Inorganic polymeric phosphate/polyphosphate as an inducer of alkaline phosphatase and a modulator of intracellular Ca2+ level in osteoblasts (SaOS-2 cells) in vitro. Acta Biomater. 2011, 7, 2661–2671. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Berreur, M.; Blanchard, F.; Chevalier, C.; Guisle-Marsollier, I.; Masson, M.; Rédini, F.; Heymann, D. Receptor activator of nuclear factor-kappaB ligand (RANKL) directly modulates the gene expression profile of RANK-positive Saos-2 human osteosarcoma cells. Oncol. Rep. 2007, 18, 1365–1371. [Google Scholar] [PubMed]
- Wiens, M.; Wang, X.H.; Schröder, H.C.; Kolb, U.; Schloßmacher, U.; Ushijima, H.; Müller, W.E.G. The role of biosilica in the osteoprotegerin/RANKL ratio in human osteoblastlike cells. Biomaterials 2010, 31, 7716–7725. [Google Scholar] [CrossRef] [PubMed]
- Gaillot, O.; Wetsch, M.; Fortineau, N.; Berche, P. Evaluation of CHROMagar staph. aureus, a new chromogenic medium, for isolation and presumptive identification of staphylococcus aureus from human clinical specimens. J. Clin. Microbiol. 2000, 38, 1587–1591. [Google Scholar] [PubMed]
- Müller, W.E.G.; Wang, X.H.; Guo, Y.W.; Schröder, H.C. Potentiation of the cytotoxic activity of copper by polyphosphate on biofilm-producing bacteria: A bioinspired approach. Mar. Drugs 2012, 10, 2369–2387. [Google Scholar] [CrossRef] [PubMed]
- Dhinakaran, D.I.; Lipton, A.P. Antimicrobial potential of the marine sponge Sigmadocia pumila from the south eastern region of India. World J. Fish Mar. Sci. 2012, 4, 344–348. [Google Scholar]
- Valgas, C.; Machado de Souza, S.; Smânia, E.F.A.; Smânia, A., Jr. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 2007, 38, 369–380. [Google Scholar] [CrossRef]
- Christy, J.E.; Jenothiny, S.; Pathmanathan, M.K.; Jeyadevan, J.P. Antibacterial activity of sequentially extracted organic solvent extracts of fruits, flowers and leaves of Lawsonia inermis L. from Jaffna. Asian Pac. J. Trop. Biomed. 2012, 2, 798–802. [Google Scholar]
- Penumatsa, N.V.; Sharanesha, R.B. Bleaching of fluorosis stains using sodium hypochlorite. J. Pharm. Bioallied Sci. 2015, 7, S766–S768. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Shen, X.; Liu, J.; Hou, Y.; Zhu, M.; Huang, J.; Xu, P. Effects of dentifrice containing hydroxyapatite on dentinal tubule occlusion and aqueous hexavalent chromium cations sorption: A preliminary study. PLoS ONE 2012, 7, e45283. [Google Scholar] [CrossRef] [PubMed]
- Petrie, A.; Watson, P. Statistics for Veterinary and Animal Science; Wiley-Blackwell: Oxford, UK, 2013; pp. 85–99. [Google Scholar]
- Qiu, C.; Xiao, X.; Liu, R. Biomimetic synthesis of spherical nano-hydroxyapatite in the presence of polyethylene glycol. Ceram. Int. 2008, 34, 1747–1751. [Google Scholar] [CrossRef]
- Chun, K.J.; Choi, H.H.; Lee, J.Y. Comparison of mechanical property and role between enamel and dentin in the human teeth. J. Dent. Biomech. 2014, 5, 1758736014520809. [Google Scholar] [CrossRef] [PubMed]
- Poorni, S.; Kumar, R.A.; Shankar, P.; Indira, R.; Ramachandran, S. Effect of 10% sodium ascorbate on the calcium: Phosphorus ratio of enamel bleached with 35% hydrogen peroxide: An in vitro quantitative energy-dispersive X-ray analysis. Contemp. Clin. Dent. 2010, 1, 223–226. [Google Scholar] [PubMed]
- Kakei, M.; Nakahara, H.; Kumegawa, M.; Mishima, H.; Kozawa, Y. High-resolution electron microscopy of the crystallites of fossil enamels obtained from various geological ages. J. Dent. Res. 2001, 80, 1560–1564. [Google Scholar] [CrossRef] [PubMed]
- Bell, N.H. Dynamics of bone metabolism. Annu. Rev. Med. 1967, 18, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Czernin, J.; Satyamurthy, N.; Schiepers, C. Molecular mechanisms of bone 18F-NaF deposition. J. Nucl. Med. 2010, 51, 1826–1829. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, E.F. Cellular mechanisms of bone remodeling. Rev. Endocr. Metab. Disord. 2010, 11, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Kini, U.; Nandeesh, B.N. Physiology of bone formation, remodeling and metabolism. In Radionuclide and Hybrid Bone Imaging; Fogelman, I., Gnanasegaran, G., van der Wall, H., Eds.; Springer: Berlin, Germany, 2012; pp. 29–57. [Google Scholar]
- Arnett, T.R. Extracellular pH regulates bone cell function. J. Nutr. 2008, 138, S415–S418. [Google Scholar]
- Riihonen, R.; Nielsen, S.; Väänänen, H.K.; Laitala-Leinonen, T.; Kwon, T.H. Degradation of hydroxyapatite in vivo and in vitro requires osteoclastic sodium-bicarbonate co-transporter NBCn1. Matrix Biol. 2010, 29, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Schröder, H.C.; Müller, W.E.G. Enzyme-based biosilica and biocalcite: Biomaterials for the future in regenerative medicine. Trends Biotechnol. 2014, 32, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Combes, C.; Rey, C. Amorphous calcium phosphates: Synthesis, properties and uses in biomaterials. Acta Biomaterialia 2010, 6, 3362–3378. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.B.; Parkinson, C.R.; Jeffery, P.; Davies, M.; Macdonald, E.L.; Seong, J.; West, N.X. A randomised clinical trial investigating calcium sodium phosphosilicate as a dentine mineralising agent in the oral environment. J. Dent. 2015, 43, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Fritz, H.; Kennedy, D.; Fergusson, D.; Fernandes, R.; Doucette, S.; Cooley, K.; Seely, A.; Sagar, S.; Wong, R.; Seely, D. Vitamin A and retinoid derivatives for lung cancer: A systematic review and meta analysis. PLoS ONE 2011, 6, e21107. [Google Scholar] [CrossRef] [PubMed]
- Aframian, D.J.; Davidowitz, T.; Benoliel, R. The distribution of oral mucosal pH values in healthy saliva secretors. Oral Dis. 2006, 12, 420–423. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, Y.; Sun, W.B.; Zhang, H. Amorphous calcium phosphate and its application in dentistry. Chem. Cent. J. 2011, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Regös, J.; Zak, O.; Solf, R.; Vischer, W.A.; Weirich, E.G. Antimicrobial spectrum of triclosan, a broad-spectrum antimicrobial agent for topical application. II. Comparison with some other antimicrobial agents. Dermatologica 1979, 158, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Aas, J.A.; Paster, B.J.; Stokes, L.N.; Olsen, I.; Dewhirst, F.E. Defining the normal bacterial flora of the oral cavity. J. Clin. Microbiol. 2005, 43, 5721–5732. [Google Scholar] [CrossRef] [PubMed]
- Dann, A.B.; Hontela, A. Triclosan: Environmental exposure, toxicity and mechanisms of action. J. Appl. Toxicol. 2011, 31, 285–311. [Google Scholar] [CrossRef] [PubMed]
- Chibinski, A.C.; Gomes, J.R.; Camargo, K.; Reis, A.; Wambier, D.S. Bone sialoprotein, matrix metalloproteinases and type I collagen expression after sealing infected caries dentin in primary teeth. Caries Res. 2014, 48, 312–319. [Google Scholar] [CrossRef] [PubMed]






© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Müller, W.E.G.; Ackermann, M.; Neufurth, M.; Tolba, E.; Wang, S.; Feng, Q.; Schröder, H.C.; Wang, X. A Novel Biomimetic Approach to Repair Enamel Cracks/Carious Damages and to Reseal Dentinal Tubules by Amorphous Polyphosphate. Polymers 2017, 9, 120. https://doi.org/10.3390/polym9040120
Müller WEG, Ackermann M, Neufurth M, Tolba E, Wang S, Feng Q, Schröder HC, Wang X. A Novel Biomimetic Approach to Repair Enamel Cracks/Carious Damages and to Reseal Dentinal Tubules by Amorphous Polyphosphate. Polymers. 2017; 9(4):120. https://doi.org/10.3390/polym9040120
Chicago/Turabian StyleMüller, Werner E.G., Maximilian Ackermann, Meik Neufurth, Emad Tolba, Shunfeng Wang, Qingling Feng, Heinz C. Schröder, and Xiaohong Wang. 2017. "A Novel Biomimetic Approach to Repair Enamel Cracks/Carious Damages and to Reseal Dentinal Tubules by Amorphous Polyphosphate" Polymers 9, no. 4: 120. https://doi.org/10.3390/polym9040120
APA StyleMüller, W. E. G., Ackermann, M., Neufurth, M., Tolba, E., Wang, S., Feng, Q., Schröder, H. C., & Wang, X. (2017). A Novel Biomimetic Approach to Repair Enamel Cracks/Carious Damages and to Reseal Dentinal Tubules by Amorphous Polyphosphate. Polymers, 9(4), 120. https://doi.org/10.3390/polym9040120









