Magnesium Pyrazolyl-Indolyl Complexes as Catalysts for Ring-Opening Polymerization of L-Lactide
Abstract
:1. Introduction
2. Experimental Section
2.1. General Information
2.2. Preparations
2.3. Crystal Structure Data
| Parameters | 1 |
|---|---|
| Formula | C30H32MgN6O2 |
| Fw | 532.93 |
| T, K | 100 (2) |
| Crystal system | Trigonal |
| Space group | R-3c |
| a, Å | 23.6115 (3) |
| b, Å | 23.6115 (3) |
| c, Å | 26.0131 (5) |
| γ, ° | 120 |
| V, Å3 | 12,559.4 (3) |
| Z | 18 |
| ρcalc, Mg/m3 | 1.268 |
| μ (Mo Kα), mm−1 | 0.102 |
| F(000) | 5076 |
| Crystal size | 0.60 × 0.45 × 0.30 mm3 |
| Theta range for data collection | 2.91° to 29.24° |
| Reflections collected | 25,170 |
| Independent reflections | 3490 [R(int) = 0.0269] |
| No. of parameters | 177 |
| R1 a | 0.0337 |
| wR2 a | 0.0893 |
| GoF b | 1.002 |
3. Results and Discussion
3.1. Preparations of Ligand Precursors and Magnesium Complexes
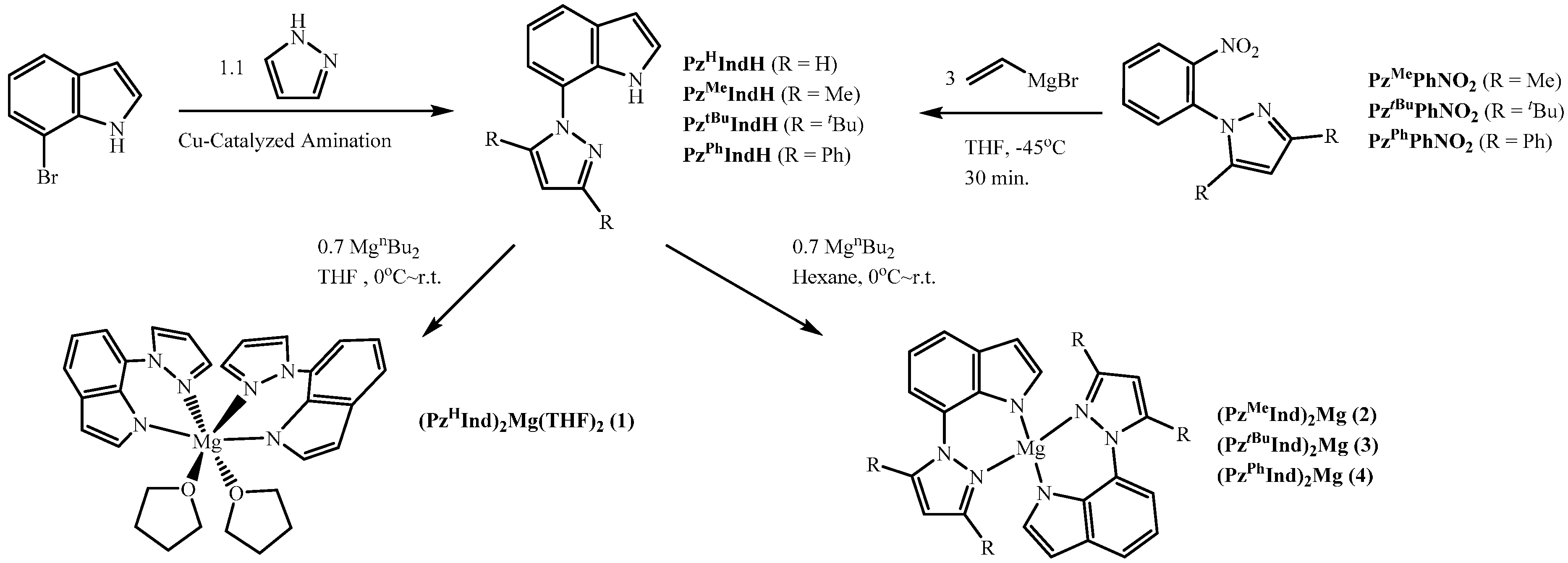
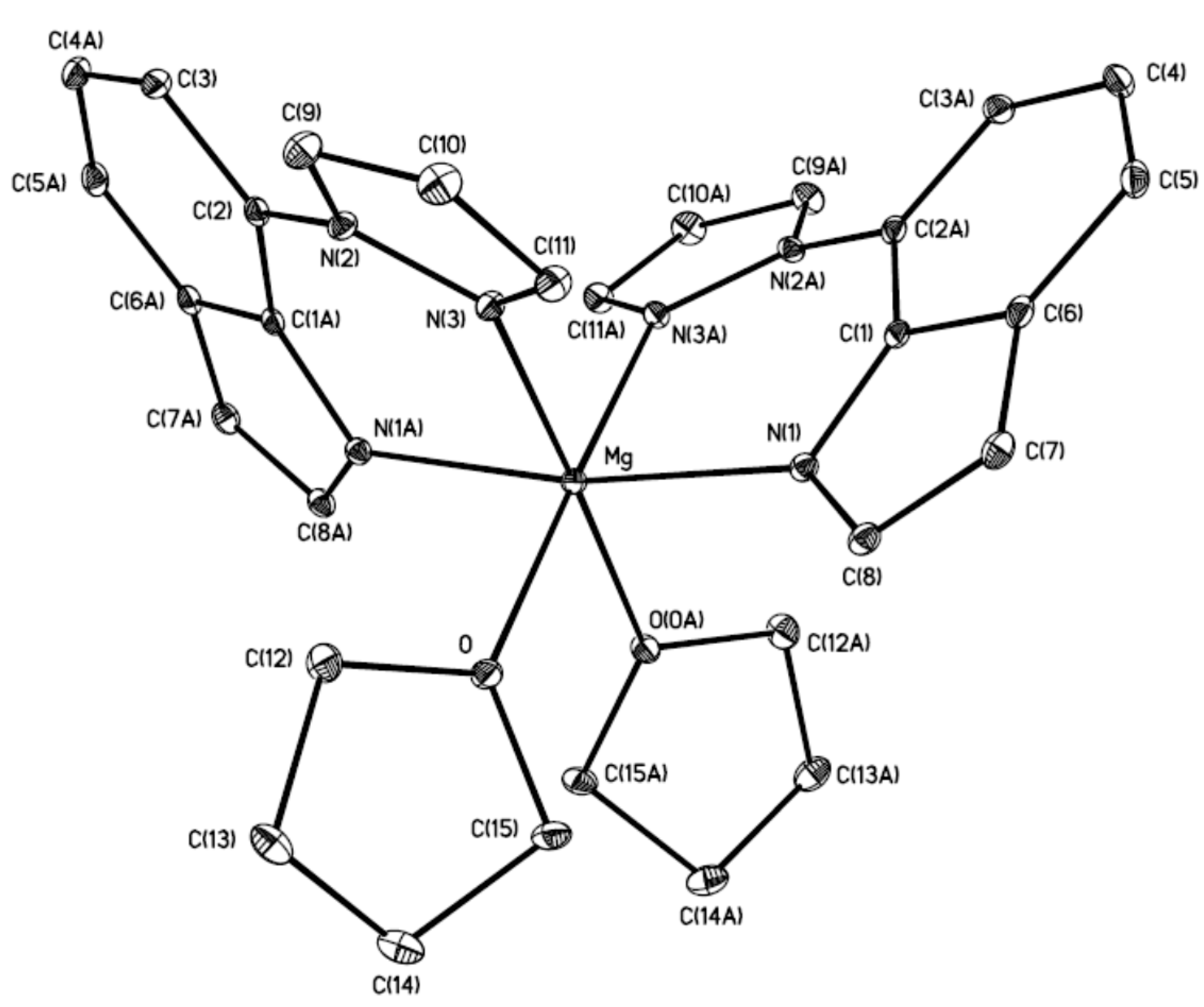
| Selected Bonds | (Å) | Selected Bond Angles | (°) |
|---|---|---|---|
| Mg–N(1) | 2.1067 (9) | N(1)–Mg–N(1A) | 169.40 (5) |
| Mg–N(3) | 2.2156 (9) | N(1)–Mg–N(3A) | 85.05 (3) |
| Mg–O | 2.1516 (8) | O–Mg–O(0A) | 87.62 (4) |
| N(3)–Mg–O(0A) | 175.46 (3) | ||
| N(1)–Mg–O(0A) | 91.36 (3) | ||
| N(3)–Mg–N(3A) | 96.38 (5) |
3.2. Polymerization Studies
| Entry | Catalyst | [LA]0:[Mg]0:[ROH] | T (°C) | t (min) | Mn (obsd) b | Mn (calcd) c | Conversion (%) d | Mw/Mn b |
|---|---|---|---|---|---|---|---|---|
| 1 | 4 | 100:1:1 | 0 | 1 | 14,000 | 10,300 | 70 | 1.20 |
| 2 e | 4 | 100:1:1 | 0 | 1 | – | – | 61 | – |
| 3 f | 4 | 100:1:1 | 0 | 1 | – | – | 6 | – |
| 4 g | 4 | 100:1:1 | 0 | 1 | – | – | 20 | – |
| 5 h | 4 | 100:1:1 | 0 | 1 | – | – | 15 | – |
| 6 | 4 | 100:1:0 | 0 | 1 | – | – | trace | – |
| 7 | 4 | 100:0:1 | 0 | 1 | – | – | trace | – |
| 8 | 1 | 100:1:1 | 0 | 5 | – | – | trace | – |
| 9 | 2 | 100:1:1 | 0 | 5 | 18,800 | 11,700 | 80 | 1.05 |
| 10 | 3 | 100:1:1 | 0 | 5 | 24,200 | 14,300 | 98 | 1.38 |
| 11 | 4 | 100:1:1 | 0 | 5 | 17,400 | 13,600 | 93 | 1.25 |
| 12 | 3 | 200:1:1 | 0 | 15 | 33,400 | 25,800 | 89 | 1.42 |
| 13 | 4 | 200:1:1 | 0 | 15 | 27,800 | 27,600 | 95 | 1.24 |
| 14 | 4 | 150:1:1 | 30 | 3 | 27,000 | 21,000 | 96 | 1.29 |
| 15 | 4 | 200:1:1 | 30 | 5 | 33,000 | 28,700 | 99 | 1.39 |
| 16 | 4 | 250:1:1 | 30 | 7 | 41,900 | 35,500 | 98 | 1.38 |
| 17 | 4 | 300:1:1 | 30 | 10 | 46,200 | 42,600 | 98 | 1.36 |
| 18 | 4 | 350:1:1 | 30 | 15 | 48,500 | 50,100 | 99 | 1.22 |
| 19 | 4 | 400:1:1 | 30 | 20 | 55,800 | 57,200 | 99 | 1.32 |
| 20 | 4 | 600:1:2 | 30 | 12 | 43,600 | 42,800 | 99 | 1.20 |
| 21 | 4 | 600:1:3 | 30 | 7 | 31,100 | 28,200 | 98 | 1.20 |
| 22 | 4 | 600:1:4 | 30 | 5 | 23,200 | 21,200 | 98 | 1.19 |
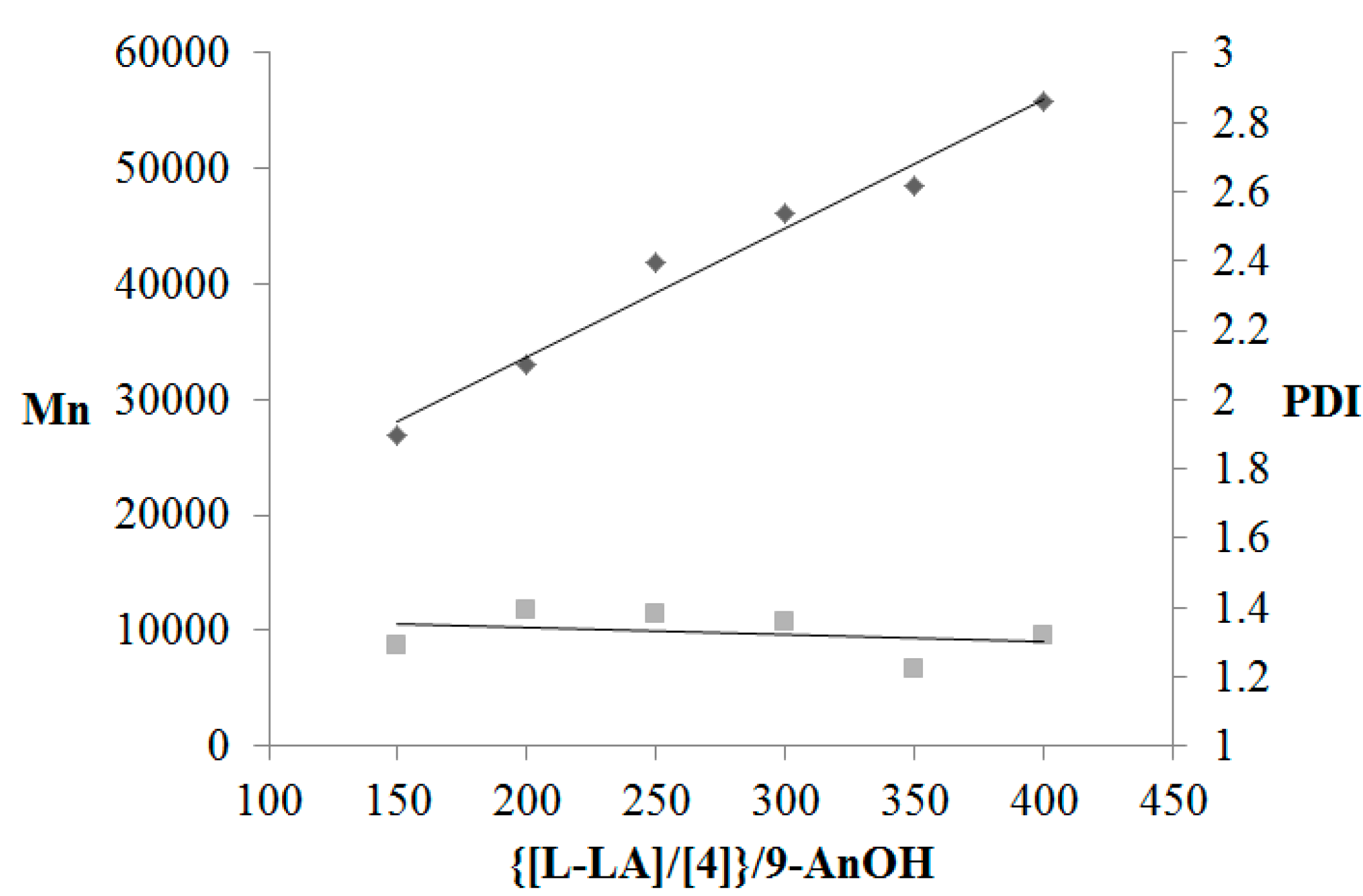
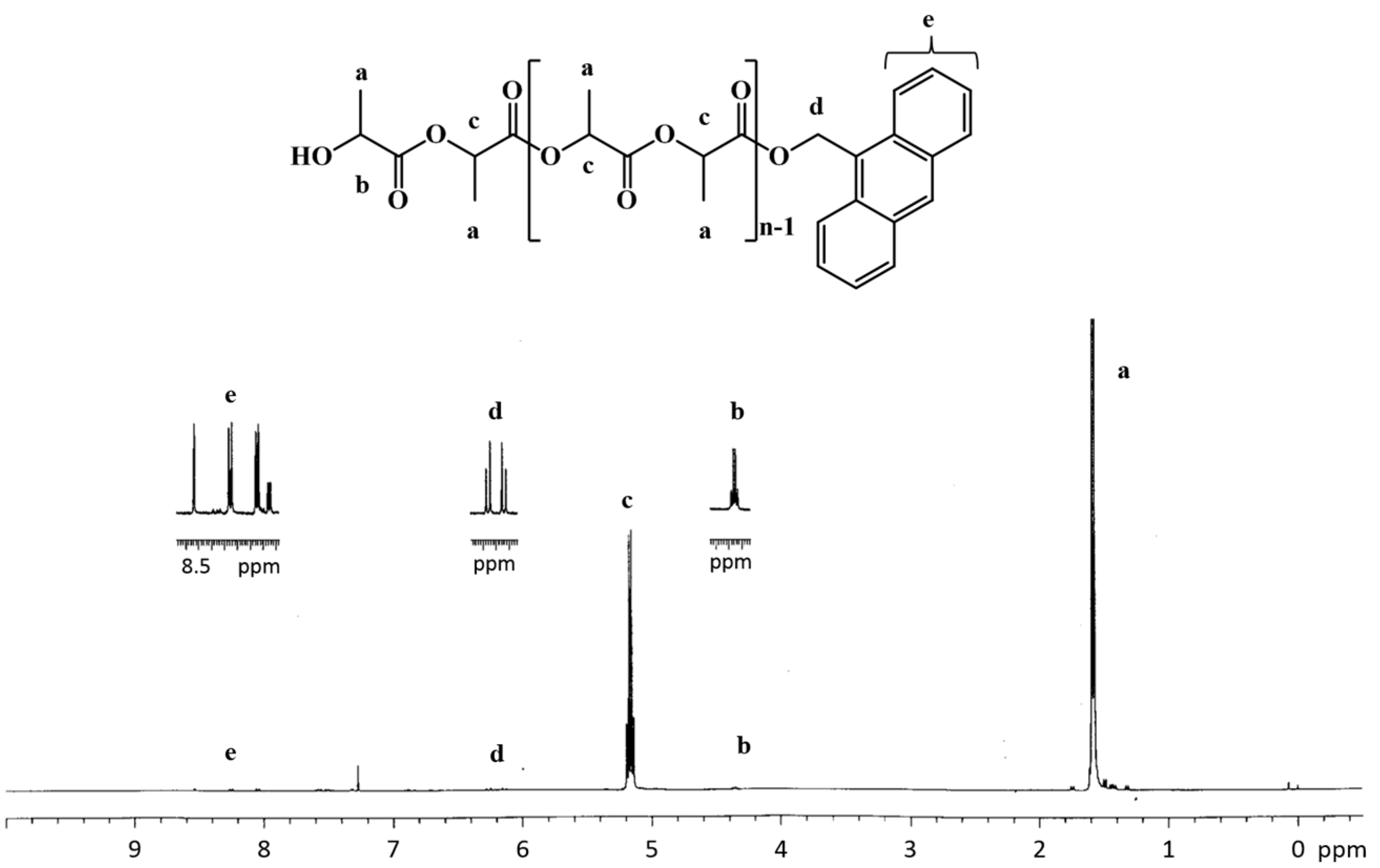
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Uhrich, K.E.; Cannizzaro, S.M.; Langer, R.S.; Shakesheff, K.M. Polymeric systems for controlled drug release. Chem. Rev. 1999, 99, 3181–3198. [Google Scholar] [CrossRef] [PubMed]
- Albertsson, A.-C.; Varma, I.K. Recent developments in ring opening polymerization of lactones for biomedical applications. Biomacromolecules 2003, 4, 1466–1486. [Google Scholar] [CrossRef] [PubMed]
- Dechy-Cabaret, O.; Martin-Vaca, B.; Bourissou, D. Controlled ring-opening polymerization of lactide and glycolide. Chem. Rev. 2004, 104, 6147–6176. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.K. Synthesis of functionalized biodegradable polyesters. Chem. Soc. Rev. 2007, 36, 1573–1580. [Google Scholar] [CrossRef] [PubMed]
- Tong, R.; Cheng, J. Paclitaxel-initiated, controlled polymerization of lactide for the formulation of polymeric nanoparticulate delivery vehicles. Angew. Chem. Int. Ed. 2008, 47, 4830–4834. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, B.J.; Hillmyer, M.A.; Tolman, W.B. Polymerization of lactide and related cyclic esters by discrete metal complexes. J. Chem. Soc. Dalton Trans. 2001. [Google Scholar] [CrossRef]
- Wu, J.; Yu, T.-L.; Chen, C.-T.; Lin, C.-C. Recent developments in main group metal complexes catalyzed/initiated polymerization of lactides and related cyclic esters. Coord. Chem. Rev. 2006, 250, 602–626. [Google Scholar] [CrossRef]
- Platel, R.H.; Hodgson, L.M.; Williams, C.K. Biocompatible initiators for lactide polymerization. Polym. Rev. 2008, 48, 11–63. [Google Scholar] [CrossRef]
- Labet, M.; Thielemans, W. Synthesis of polycaprolactone: A review. Chem. Soc. Rev. 2009, 38, 3484–3504. [Google Scholar] [CrossRef] [PubMed]
- Wheaton, C.A.; Hayes, P.G.; Ireland, B.J. Complexes of Mg, Ca and Zn as homogeneous catalysts for lactide polymerization. Dalton Trans. 2009. [Google Scholar] [CrossRef] [PubMed]
- Stanford, M.J.; Dove, A.P. Stereocontrolled ring-opening polymerisation of lactide. Chem. Soc. Rev. 2010, 39, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Sutar, A.K.; Maharana, T.; Dutta, S.; Chen, C.-T.; Lin, C.-C. Ring-opening polymerization by lithium catalysts: An overview. Chem. Soc. Rev. 2010, 39, 1724–1746. [Google Scholar] [CrossRef] [PubMed]
- Arbaoui, A.; Redshaw, C. Metal catalysts for 3-caprolactone polymerization. Polym. Chem. 2010, 1, 801–826. [Google Scholar] [CrossRef]
- Ajellal, N.; Carpentier, J.-F.; Guillaume, C.; Guillaume, S.M.; Helou, M.; Poirier, V.; Sarazin, Y.; Trifonov, A. Metal-catalyzed immortal ring-opening polymerization of lactones, lactides and cyclic carbonates. Dalton Trans. 2010, 39, 8363–8376. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.-C.; Chang, J.-C.; Lee, W.-Y.; Lee, H.M.; Huang, J.-H. Synthesis and characterization of lithium and yttrium complexes containing tridentate pyrrolyl ligands. Single-crystal X-ray structures of {Li[C4H2N(CH2NMe2)2-2,5]}2 (1) and {[C4H2N(CH2NMe2)2-2,5]YCl2(μ-Cl).Li(OEt2)2}2 (2) and ring-opening polymerization of ε-caprolactone. J. Organometal. Chem. 2005, 690, 4168–4174. [Google Scholar]
- Hsieh, K.-C.; Lee, W.-Y.; Hsueh, L.-F.; Lee, H.M.; Huang, J.-H. Synthesis and characterization of zirconium and hafnium aryloxide compounds and their reactivity towards lactide and ε-caprolactone polymerization. Eur. J. Inorg. Chem. 2006, 2006, 2306–2312. [Google Scholar] [CrossRef]
- Hsieh, I-P.; Huang, C.-H.; Lee, H.M.; Kuo, P.-C.; Huang, J.-H.; Lee, H.-I.; Cheng, J.-T.; Lee, G.-H. Indium complexes incorporating bidentate substituted pyrrole ligand: Synthesis, characterization, and ring-opening polymerization of ε-caprolactone. Inorg. Chim. Acta 2006, 359, 497–504. [Google Scholar]
- Yang, Y.; Li, S.; Cui, D.; Chen, X.; Jing, X. Pyrrolide-ligated organoyttrium complexes. Synthesis, characterization, and lactide polymerization behavior. Organometallics 2007, 26, 671–678. [Google Scholar] [CrossRef]
- Zi, G.; Wang, Q.; Xiang, L.; Song, H. Lanthanide and group 4 metal complexes with new chiral biaryl-based NNO-donor ligands. Dalton Trans. 2008. [Google Scholar] [CrossRef] [PubMed]
- Broomfield, L.M.; Wright, J.A.; Bochmann, M. Synthesis, structures and reactivity of 2-phosphorylmethyl-1H-pyrrolato complexes of titanium, yttrium and zinc. Dalton Trans. 2009. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-M.; Hsiao, C.-S.; Datta, A.; Hung, C.-H.; Chang, L.-C.; Lee, T.-Y.; Huang, J.-H. Monomeric, dimeric, and trimeric calcium compounds containing substituted pyrrolyl and ketiminate ligands: synthesis and structural characterization. Inorg. Chem. 2009, 48, 8004–8011. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Velders, A.H.; Dijkstra, P.J.; Zhong, Z.; Chen, X.; Feijen, J. Polymerization of lactide using achiral bis(pyrrolidene) schiff base aluminum complexes. Macromolecules 2009, 42, 1058–1066. [Google Scholar] [CrossRef]
- Katiyar, V.; Nanavati, H. Ring-opening polymerization of l-lactide using N-heterocyclic molecules: Mechanistic, kinetics and DFT studies. Polym. Chem. 2010, 1, 1491–1500. [Google Scholar] [CrossRef]
- Qiao, S.; Ma, W.-A.; Wang, Z.-X. Synthesis and characterization of aluminum and zinc complexes supported by pyrrole-based ligands and catalysis of the aluminum complexes toward the ring-opening polymerization of ε-caprolactone. J. Organometal. Chem. 2011, 696, 2746–2753. [Google Scholar] [CrossRef]
- Huang, W.-Y.; Chuang, S.-J.; Chunag, N.-T.; Hsiao, C.-S.; Datta, A.; Chen, S.-J.; Hu, C.-H.; Huang, J.-H.; Lee, T.-Y.; Lin, C.-H. Aluminium complexes containing bidentate and symmetrical tridentate pincer type pyrrolyl ligands: Synthesis, reactions and ring opening polymerization. Dalton Trans. 2011, 40, 7423–7433. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, L.-F.; Chuang, N.-T.; Lee, C.-Y.; Datta, A.; Huang, J.-H.; Lee, T.-Y. Magnesium complexes containing η1- and η3-pyrrolyl or ketiminato ligands: Synthesis, structural Investigation and ε-caprolactone ring-opening polymerisation. Eur. J. Inorg. Chem. 2011, 2011, 5530–5537. [Google Scholar] [CrossRef]
- Li, G.; Lamberti, M.; Mazzeo, M.; Pappalardo, D.; Roviello, G.; Pellecchia, C. Anilidopyridyl-pyrrolide and anilidopyridyl-indolide group 3 metal complexes: highly active initiators for the ring-opening polymerization of rac-lactide. Organometallics 2012, 31, 1180–1188. [Google Scholar] [CrossRef]
- Peng, K.-F.; Chen, Y.; Chen, C.-T. Synthesis and catalytic application of magnesium complexes bearing pendant indolyl ligands. Dalton Trans. 2015, 44, 9610–9619. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cui, D.; Hou, Z.; Li, W.; Li, Y. Highly cis-1,4-selective living polymerization of 1,3-conjugated dienes and copolymerization with ε-caprolactone by bis(phosphino)carbazolide rare-earth-metal complexes. Organometallics 2011, 30, 760–767. [Google Scholar] [CrossRef]
- Cacchi, S.; Fabrizi, G. Synthesis and functionalization of indoles through palladium-catalyzed reactions. Chem. Rev. 2005, 105, 2873–2920. [Google Scholar] [CrossRef] [PubMed]
- Dalpozzo, R.; Bartoli, G. Bartoli indole synthesis. Curr. Org. Chem. 2005, 9, 163–178. [Google Scholar] [CrossRef]
- Humphrey, G.R.; Kuethe, J.T. Practical methodologies for the synthesis of indoles. Chem. Rev. 2006, 106, 2875–2911. [Google Scholar] [CrossRef] [PubMed]
- Cacchi, S.; Fabrizi, G.; Goggiamani, A. Copper catalysis in the construction of indole and benzo[b]furan rings. Org. Biomol. Chem. 2011, 9, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Inman, M.; Moody, C.J. Indole synthesis—Something old, something new. Chem. Sci. 2013, 4, 29–41. [Google Scholar] [CrossRef]
- Sánchez-Barba, L.F.; Garcés, A.; Fajardo, M.; Alonso-Moreno, C.; Fernández-Baeza, J.; Otero, A.; Antiñolo, A.; Tejeda, J.; Lara-Sánchez, A.; López-Solera, M.I. Well-defined alkyl heteroscorpionate magnesium complexes as excellent initiators for the ROP of cyclic esters. Organometallics 2007, 26, 6403–6411. [Google Scholar] [CrossRef]
- Schofield, A.D.; Barros, M.L.; Cushion, M.G.; Schwarz, A.D.; Mountford, P. Sodium, magnesium and zinc complexes of mono(phenolate) heteroscorpionate ligands. Dalton Trans. 2009. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, M.H.; Gallucci, J.C.; Yaman, G. Synthesis and coordination chemistry of TpC*MI complexes where M = Mg, Ca, Sr, Ba and Zn and TpC* = tris[3-(2-methoxy-1,1-dimethyl)pyrazolyl]hydroborate. Dalton Trans. 2009. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-T.; Hung, C.-C.; Chang, Y.-J.; Peng, K.-F.; Chen, M.-T. Magnesium and zinc complexes containing pendant pyrazolyl-phenolate ligands as catalysts for ring opening polymerisation of cyclic esters. J. Organometal. Chem. 2013, 738, 1–9. [Google Scholar] [CrossRef]
- Dobbs, A. Total synthesis of indoles from tricholoma species via bartoli/heteroaryl radical methodologies. J. Org. Chem. 2001, 66, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Subramanyam, U.; Puranik, V.G.; Mohandas, T.P.; Sarkar, A. Pyrazole-tethered heteroditopic ligands and their transition metal complexes: Synthesis, structure, and reactivity. Eur. J. Inorg. Chem. 2005, 2005, 1254–1263. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXTL-97, Program for Refinement of Crystal Structures; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Cacchi, S.; Fabrizi, G. Update 1 of: Synthesis and functionalization of indoles through palladium-catalyzed reactions. Chem. Rev. 2011, 111, 215–283. [Google Scholar] [CrossRef] [PubMed]
- Taber, D.F.; Tirunahari, P.K. Indole synthesis: A review and proposed classification. Tetrahedron 2011, 67, 7195–7210. [Google Scholar] [CrossRef] [PubMed]
- Antilla, J.C.; Baskin, J.M.; Barder, T.E.; Buchwald, S.L. Copper-diamine-catalyzed N-arylation of pyrroles, pyrazoles, indazoles, imidazoles, and triazoles. J. Org. Chem. 2004, 69, 5578–5587. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.R.D.; Kamenz, B.L.; Hayes, P.G. Bis(pyrazolyl)carbazole as a versatile ligand for supporting lutetium alkyl and hydride complexes. Organometallics 2014, 33, 3005–3011. [Google Scholar] [CrossRef]
- Pedrares, A.S.; Teng, W.; Ruhlandt-Senge, K. Syntheses and structures of magnesium pyridine thiolates—Model compounds for magnesium binding in photosystem I. Chem. Eur. J. 2003, 9, 2019–2024. [Google Scholar] [CrossRef] [PubMed]
- Bott, R.K.J.; Schormann, M.; Hughes, D.L.; Lancaster, S.J.; Bochmann, M. Synthesis and structures of ferrocenyl-substituted salicylaldiminato complexes of magnesium, titanium and zirconium. Polyhedron 2006, 25, 387–396. [Google Scholar] [CrossRef]
- Li, C.-Y.; Su, J.-K.; Yu, C.-J.; Tai, Y.-E.; Lin, C.-H.; Ko, B.-T. Synthesis and structural characterization of magnesium complexes bearing benzotriazole phenoxide ligands: Photoluminescent properties and catalytic studies for ring-opening polymerization of l-lactide. Inorg. Chem. Commun. 2012, 20, 60–65. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-T.; Lin, D.-H.; Peng, K.-F. Magnesium Pyrazolyl-Indolyl Complexes as Catalysts for Ring-Opening Polymerization of L-Lactide. Polymers 2015, 7, 1954-1964. https://doi.org/10.3390/polym7101492
Chen C-T, Lin D-H, Peng K-F. Magnesium Pyrazolyl-Indolyl Complexes as Catalysts for Ring-Opening Polymerization of L-Lactide. Polymers. 2015; 7(10):1954-1964. https://doi.org/10.3390/polym7101492
Chicago/Turabian StyleChen, Chi-Tien, Deng-Hao Lin, and Kuo-Fu Peng. 2015. "Magnesium Pyrazolyl-Indolyl Complexes as Catalysts for Ring-Opening Polymerization of L-Lactide" Polymers 7, no. 10: 1954-1964. https://doi.org/10.3390/polym7101492
APA StyleChen, C.-T., Lin, D.-H., & Peng, K.-F. (2015). Magnesium Pyrazolyl-Indolyl Complexes as Catalysts for Ring-Opening Polymerization of L-Lactide. Polymers, 7(10), 1954-1964. https://doi.org/10.3390/polym7101492






