Crystallization Behaviors and Structure Transitions of Biocompatible and Biodegradable Diblock Copolymers
Abstract
:1. Introduction
2. PEO-b-PCL Diblock Copolymers
2.1. Crystallization Morphology and Ring-Banded Spherulite of PEO-b-PCL Diblock Copolymers [20]
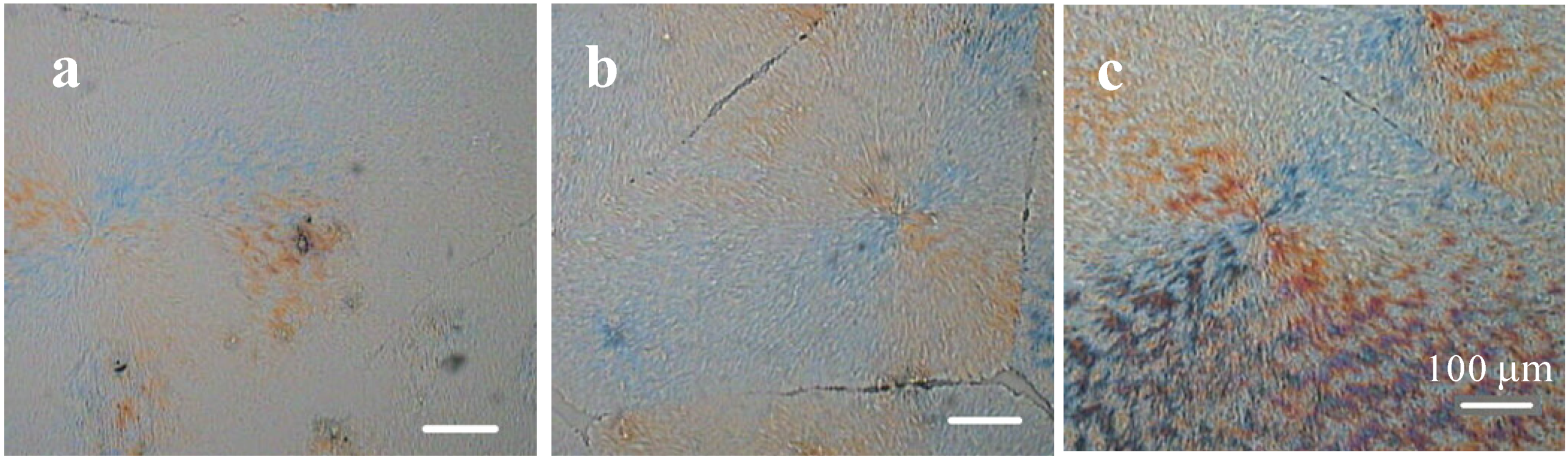

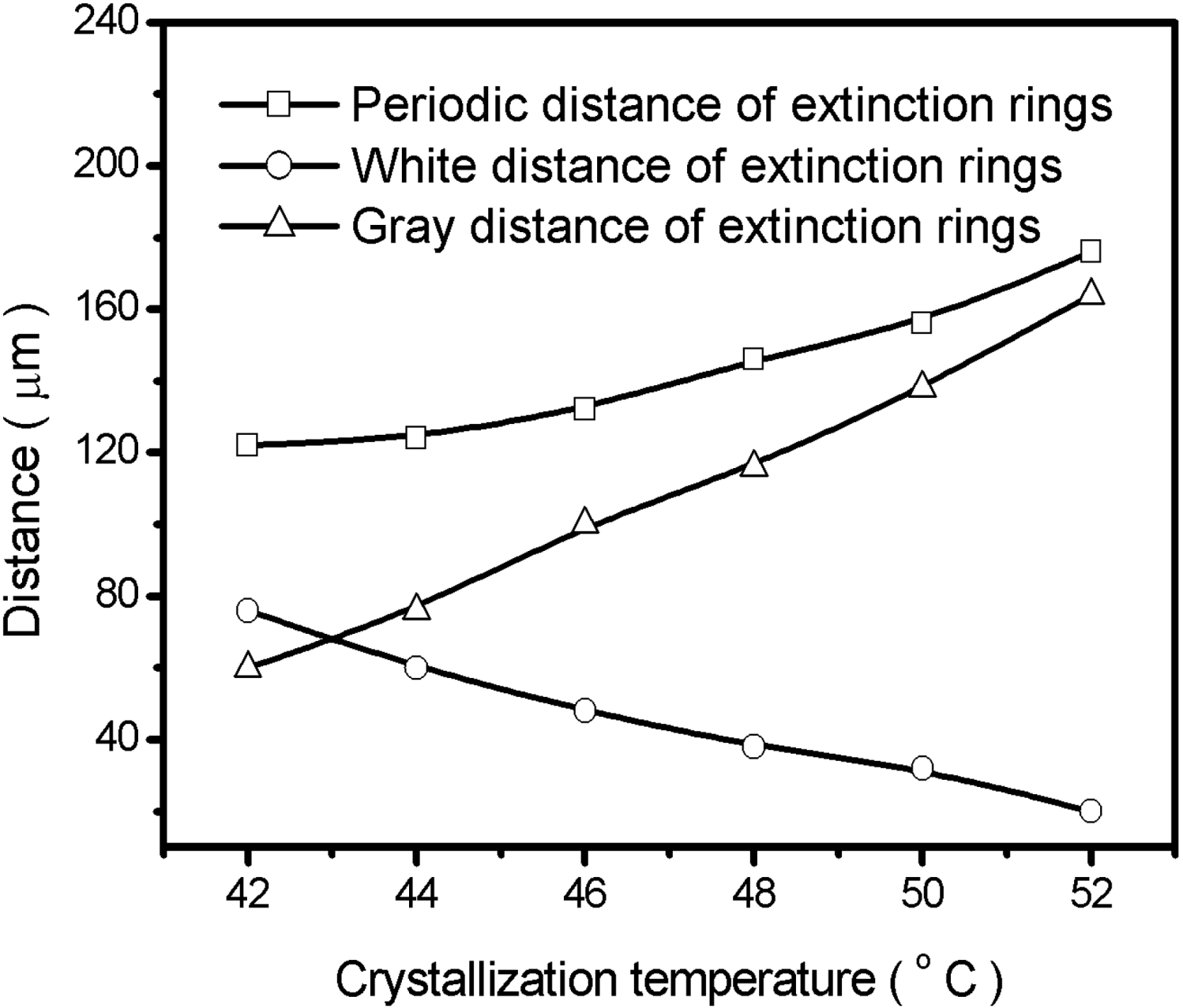
2.2. Crystallization Transitions in PEO-b-PCL Diblock Copolymers [29]


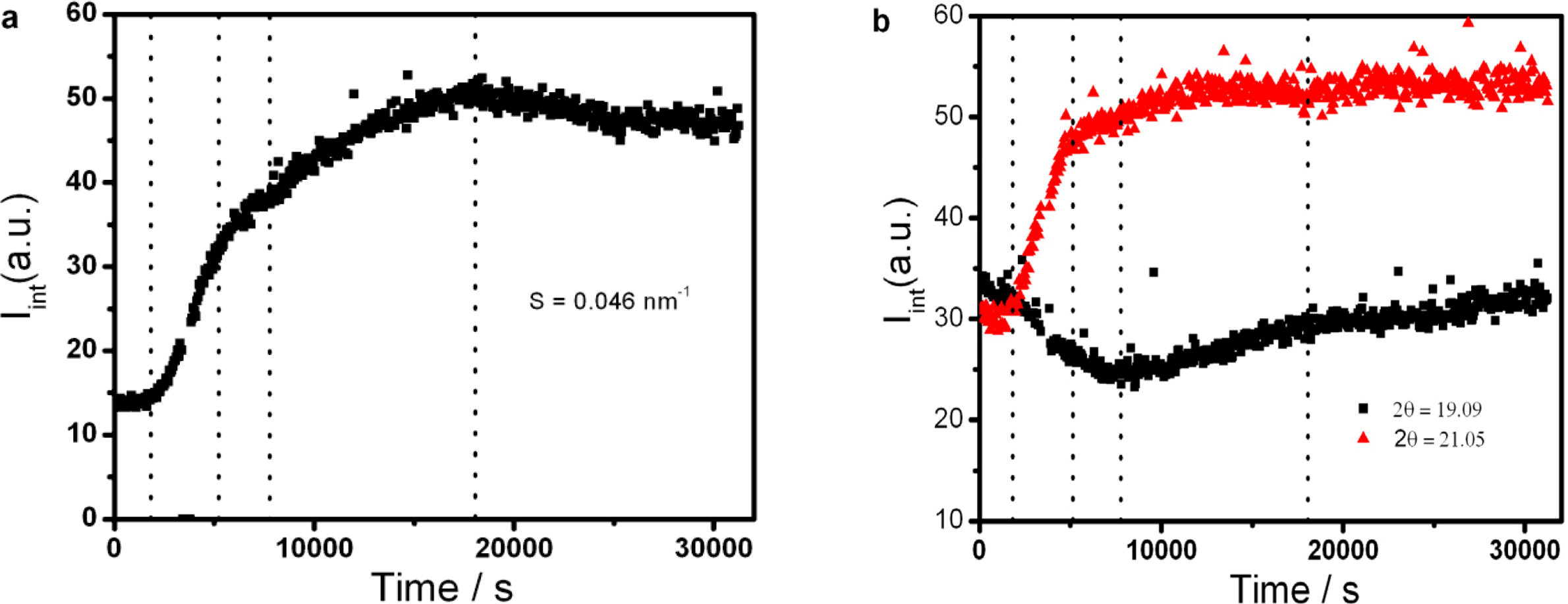
2.3. Soft Nanoconfinement on the Crystallization Behavior of PEO-b-PCL Diblock Copolymers [37]
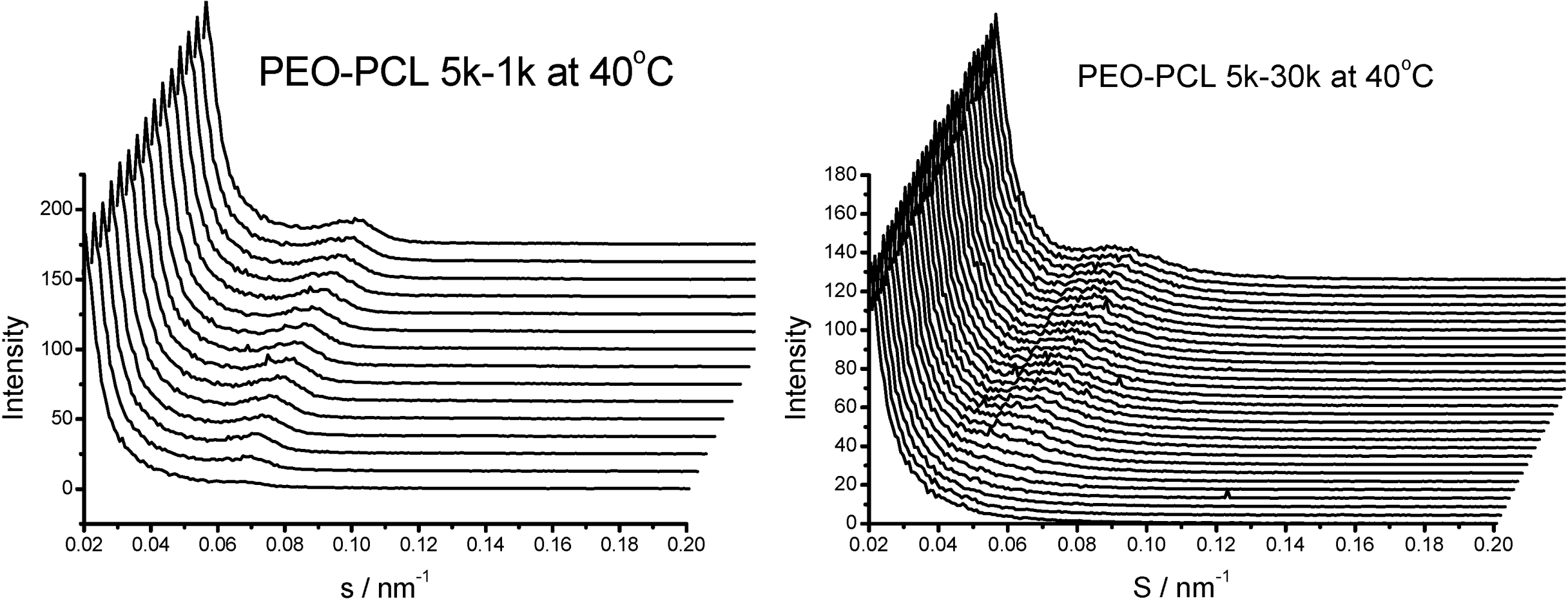
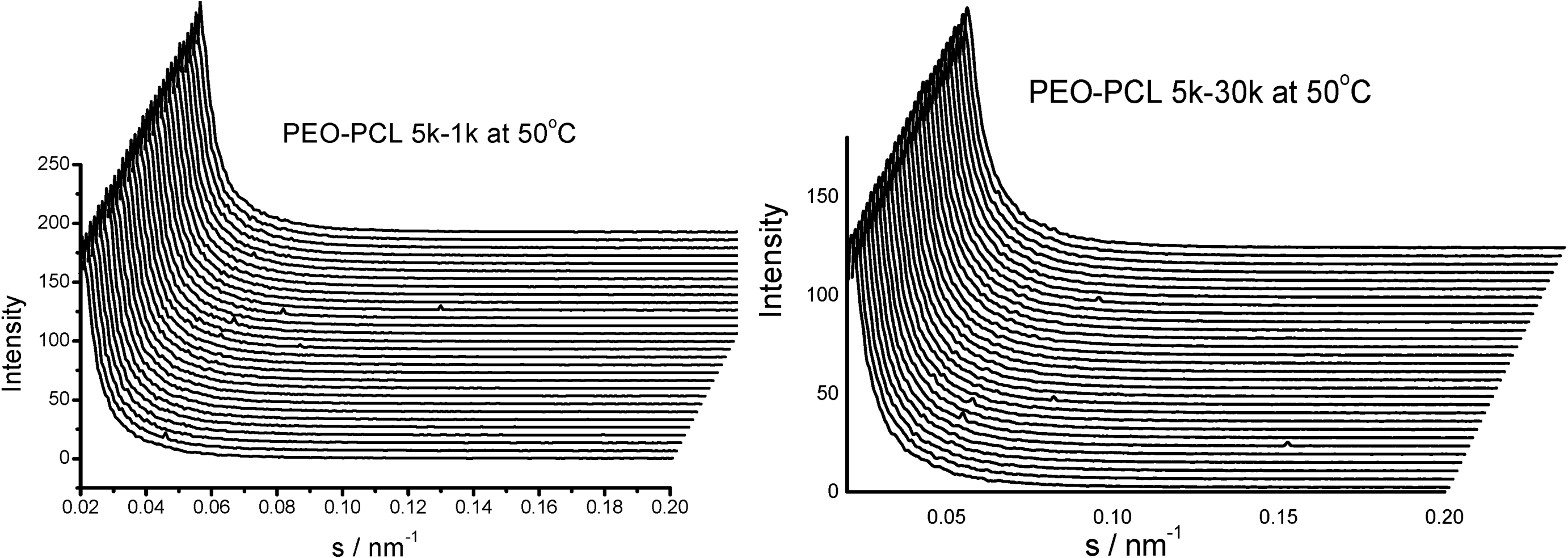
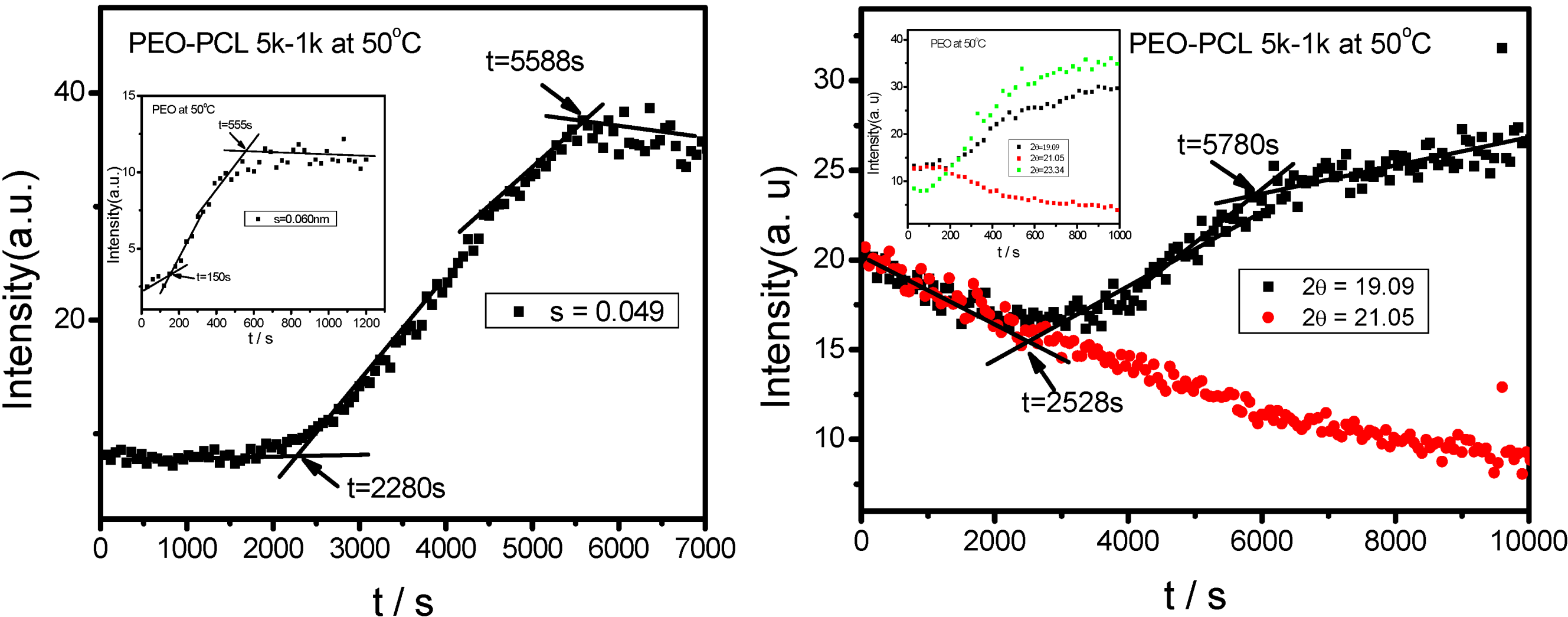


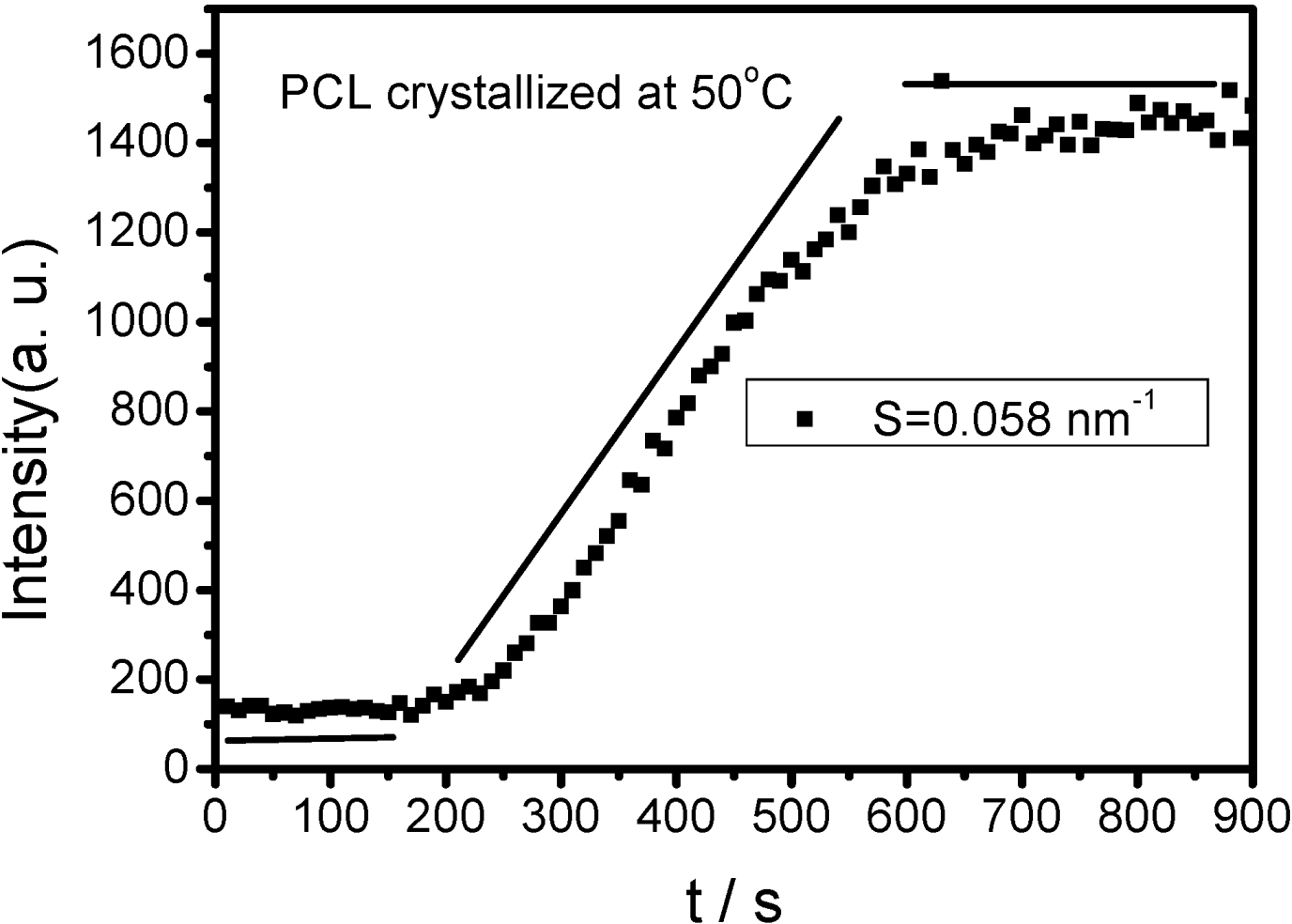
 is used to value the crystallization behavior of homopolymers and the diblock block copolymers. It is difficult to obtain QPEO at 40 °C, because the crystallization is too fast to be tracked by synchrotron SAXS/WAXS with the limited cooling rate from high temperature. QPCL = 10 at 40 °C. QPEO = 3 and QPCL = 500 at 50 °C. When Q = 1, the crystallization of the crystalline block in the diblock copolymer is not affected. The bigger Q, the more effect on the crystallization behavior can be found in the asymmetric diblock copolymers. Therefore, the PEO block has more effect on the PCL crystallization behavior in the present observed asymmetric diblock copolymers at least at 50 °C. The confinement on the crystallization behavior in the asymmetric diblock copolymers may be due to the chemical structure of the copolymers and the crystalline property of the blocks. The molecular weight of the blocks in the present investigated asymmetric diblock copolymers is different. The crystallization of PEO block in the copolymer is in the confinement of PCL block with 1 k molecular weight; however, the crystallization of PCL block in the copolymer is in the confinement of PEO block with 5 k molecular weight. On the other hand, there is a much higher molecular weight of the whole copolymer for the crystalline PCL than that for the crystalline PEO.
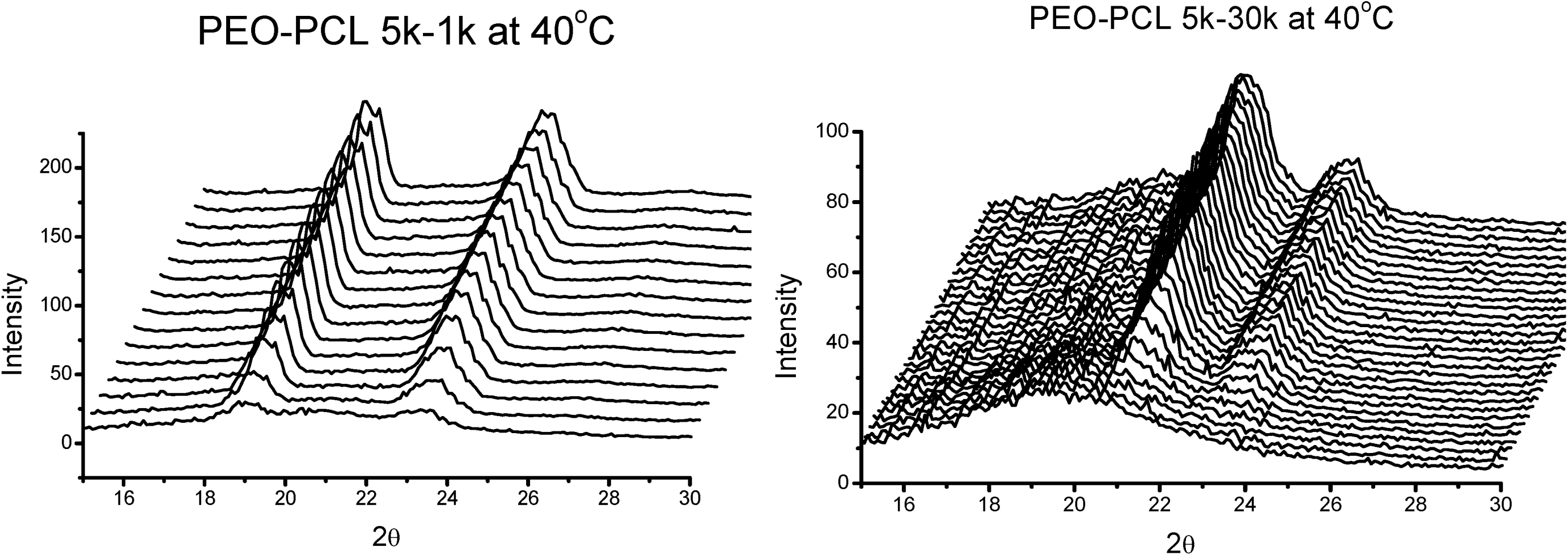
is used to value the crystallization behavior of homopolymers and the diblock block copolymers. It is difficult to obtain QPEO at 40 °C, because the crystallization is too fast to be tracked by synchrotron SAXS/WAXS with the limited cooling rate from high temperature. QPCL = 10 at 40 °C. QPEO = 3 and QPCL = 500 at 50 °C. When Q = 1, the crystallization of the crystalline block in the diblock copolymer is not affected. The bigger Q, the more effect on the crystallization behavior can be found in the asymmetric diblock copolymers. Therefore, the PEO block has more effect on the PCL crystallization behavior in the present observed asymmetric diblock copolymers at least at 50 °C. The confinement on the crystallization behavior in the asymmetric diblock copolymers may be due to the chemical structure of the copolymers and the crystalline property of the blocks. The molecular weight of the blocks in the present investigated asymmetric diblock copolymers is different. The crystallization of PEO block in the copolymer is in the confinement of PCL block with 1 k molecular weight; however, the crystallization of PCL block in the copolymer is in the confinement of PEO block with 5 k molecular weight. On the other hand, there is a much higher molecular weight of the whole copolymer for the crystalline PCL than that for the crystalline PEO.2.4. Confined Crystalline Lamella Formation in PEO-b-PCL Diblock Copolymers [43]
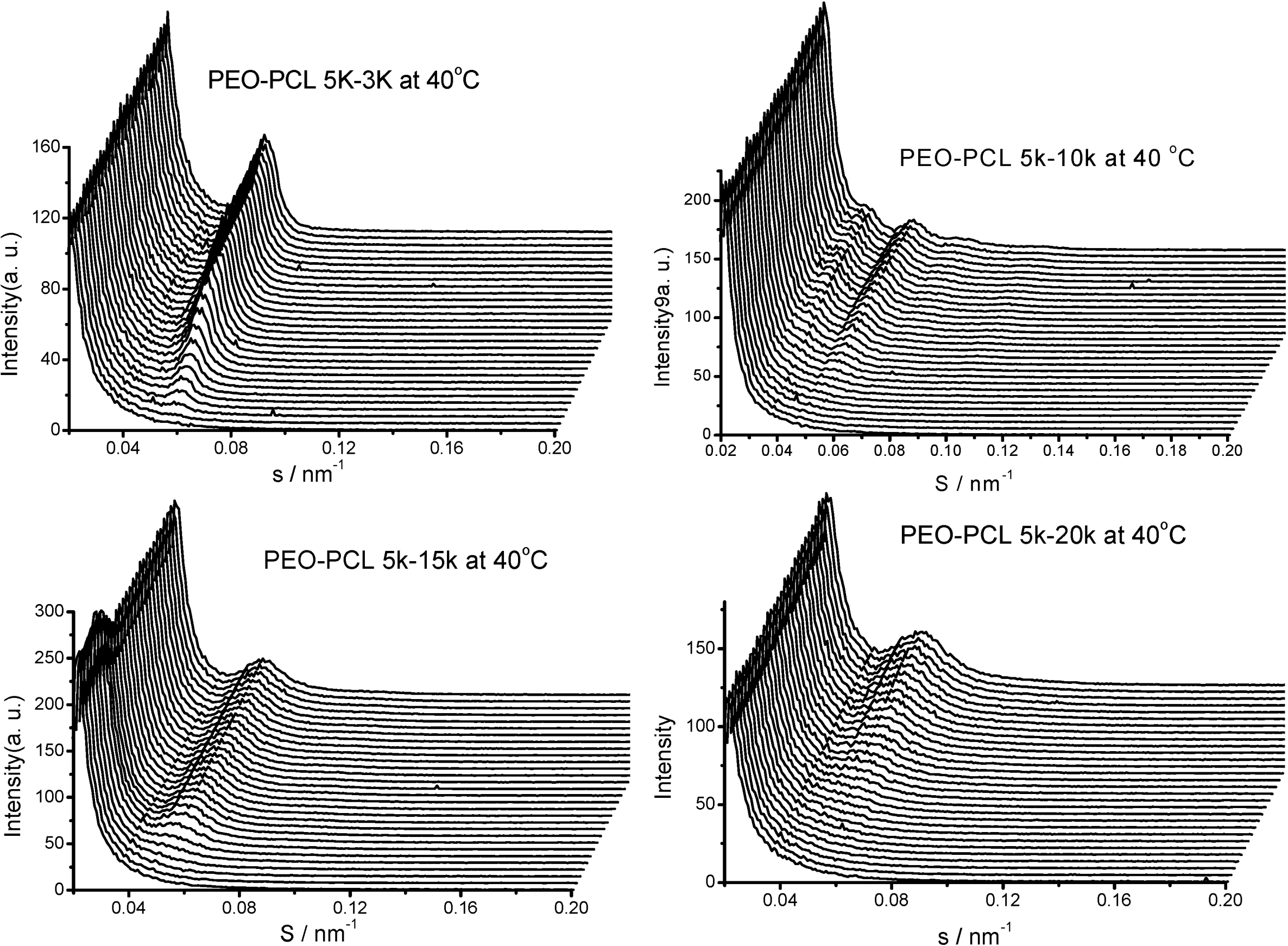
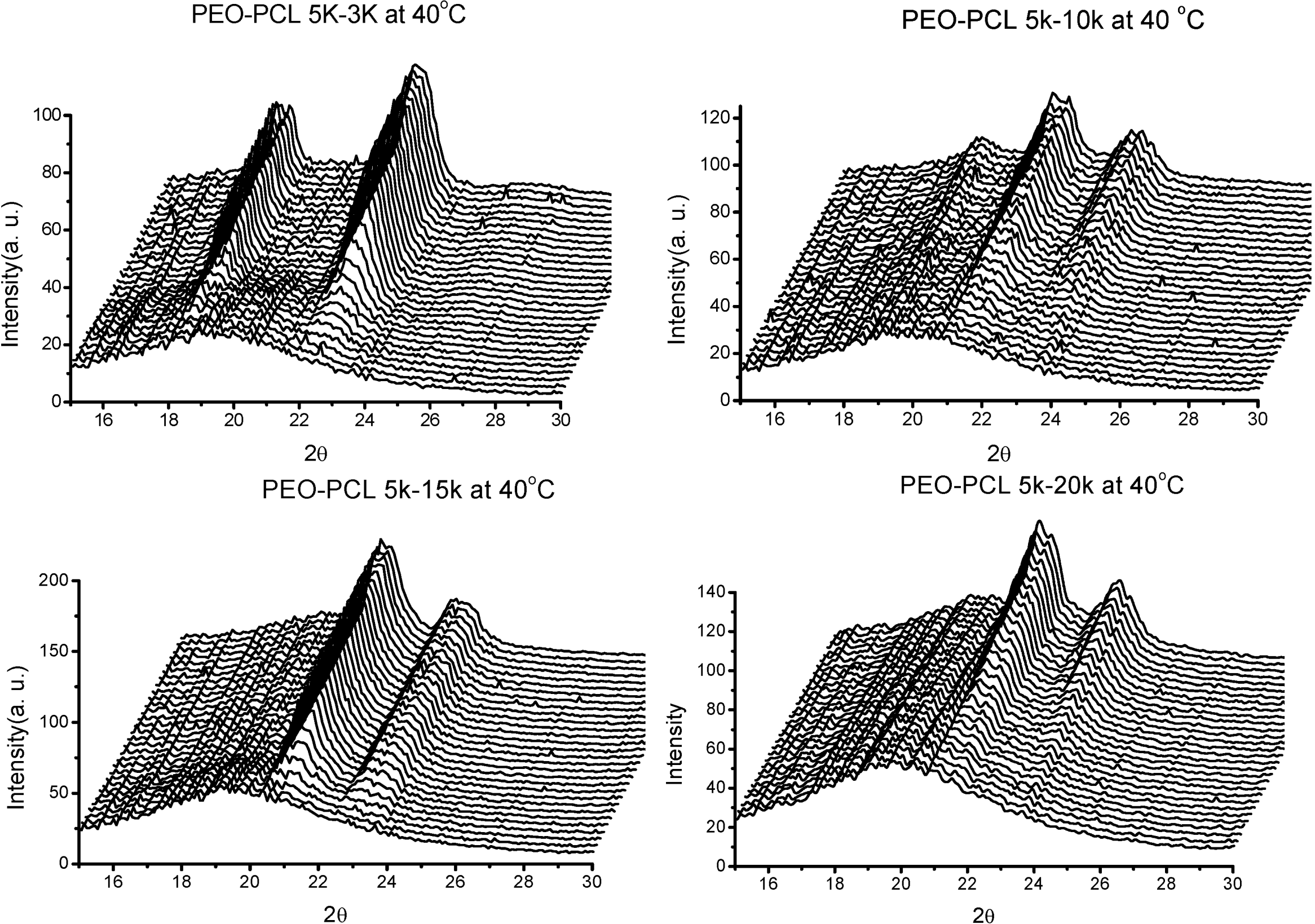

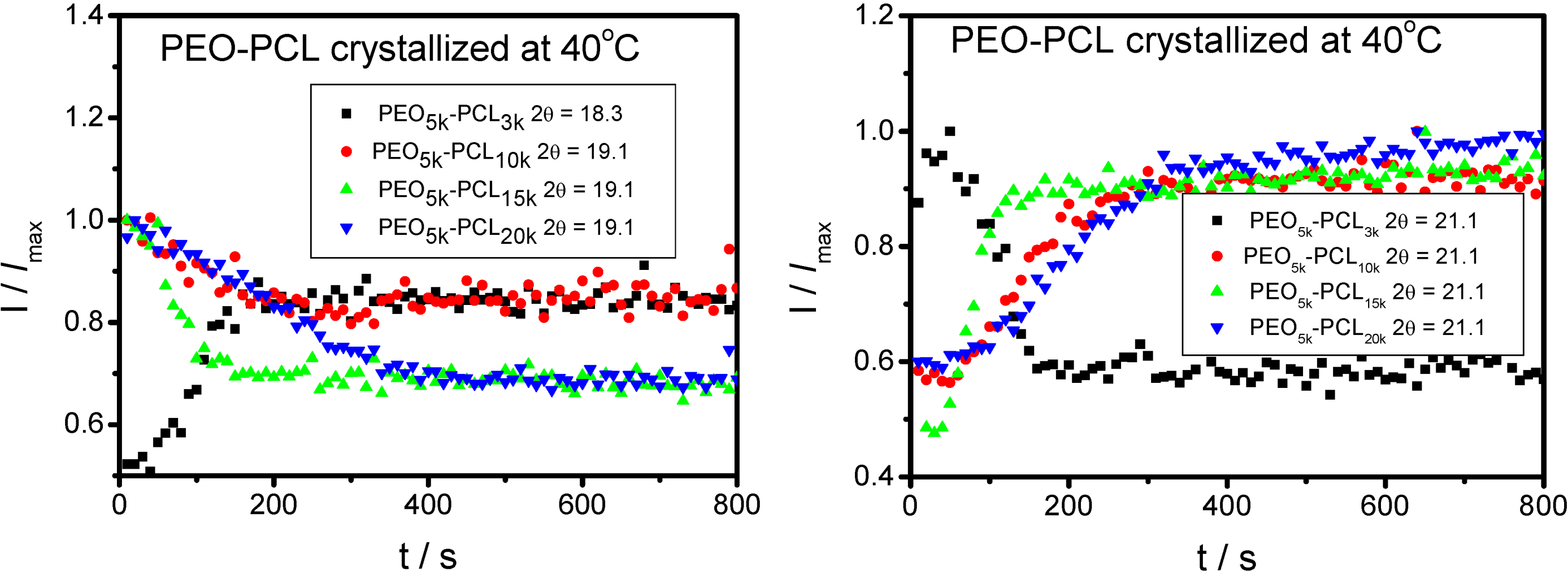

3. PEO-b-PLA Diblock Copolymers
3.1. Soft Confined Crystallization and Microphase Separation-Determined Morphologies of PEO-b-PCLA Diblock Copolymers [7]

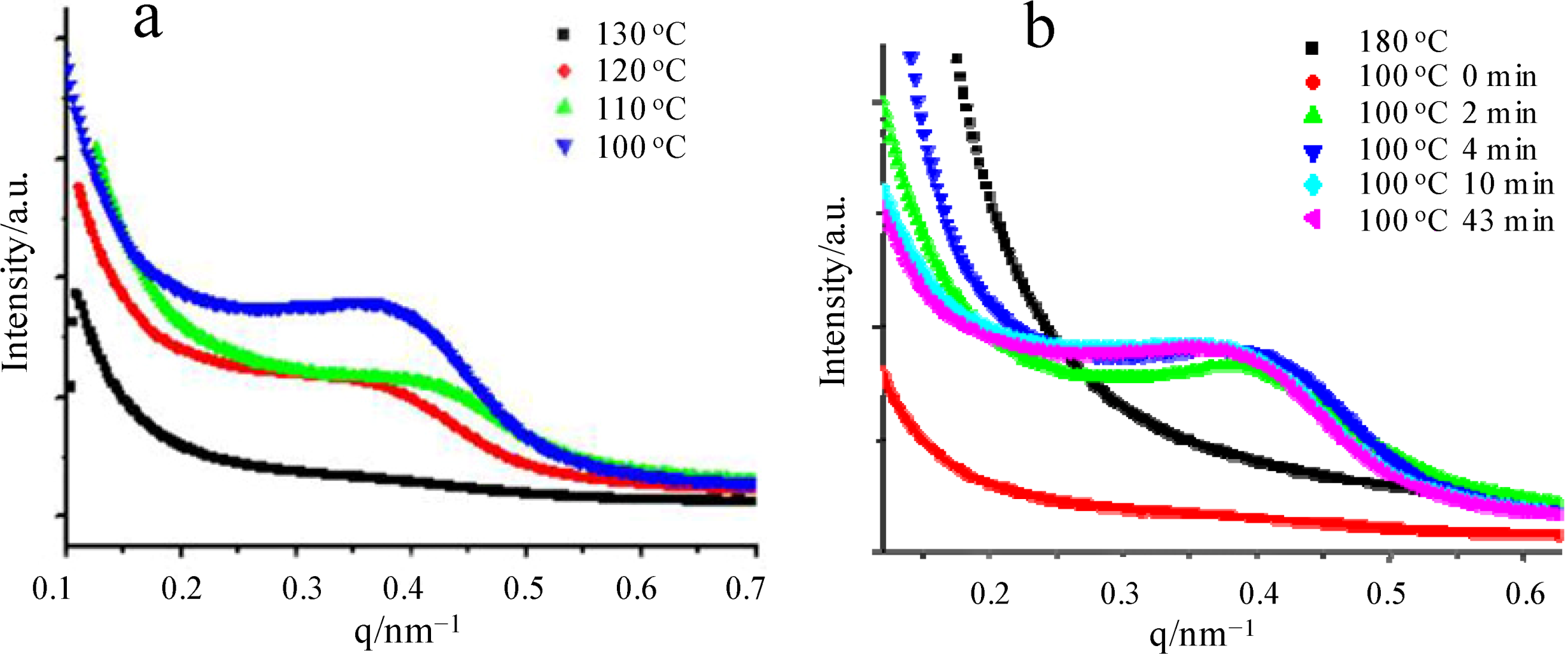
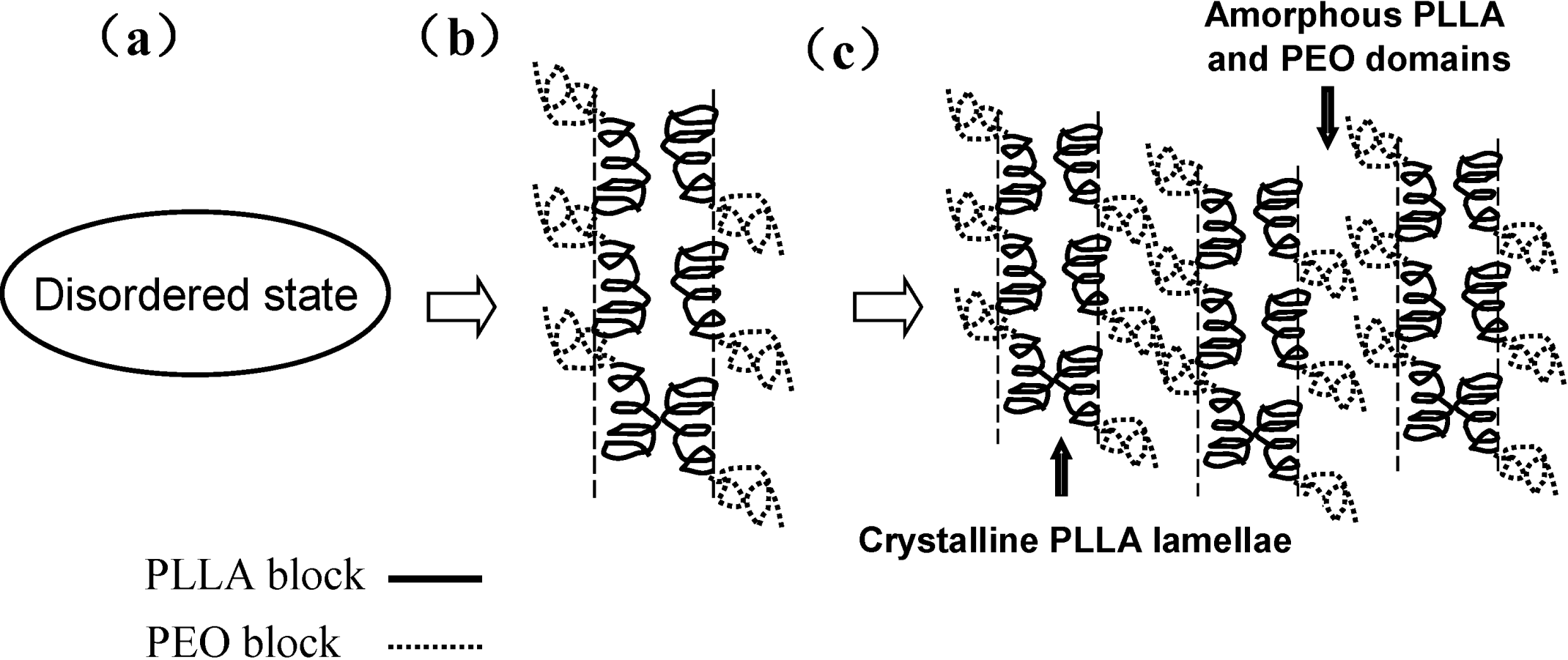
3.2. Hard Confined Crystallization and Microphase Separation-Determined Morphologies of PEO-b-PCLA Diblock Copolymers [7]

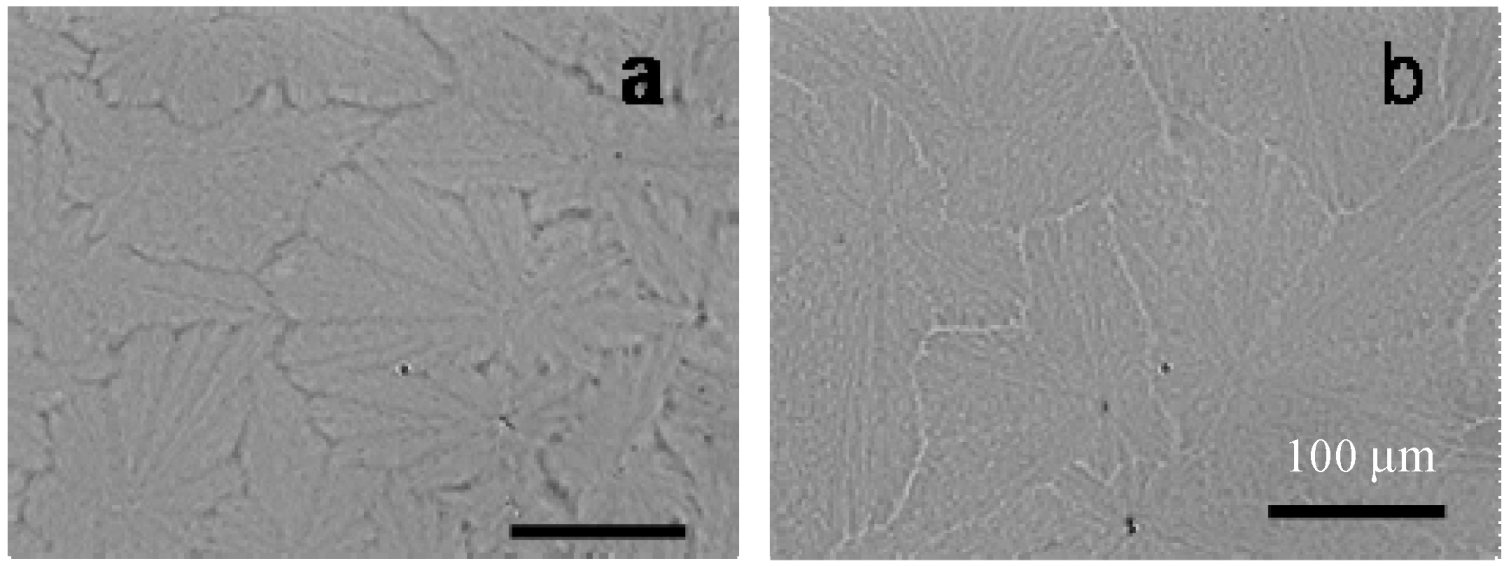
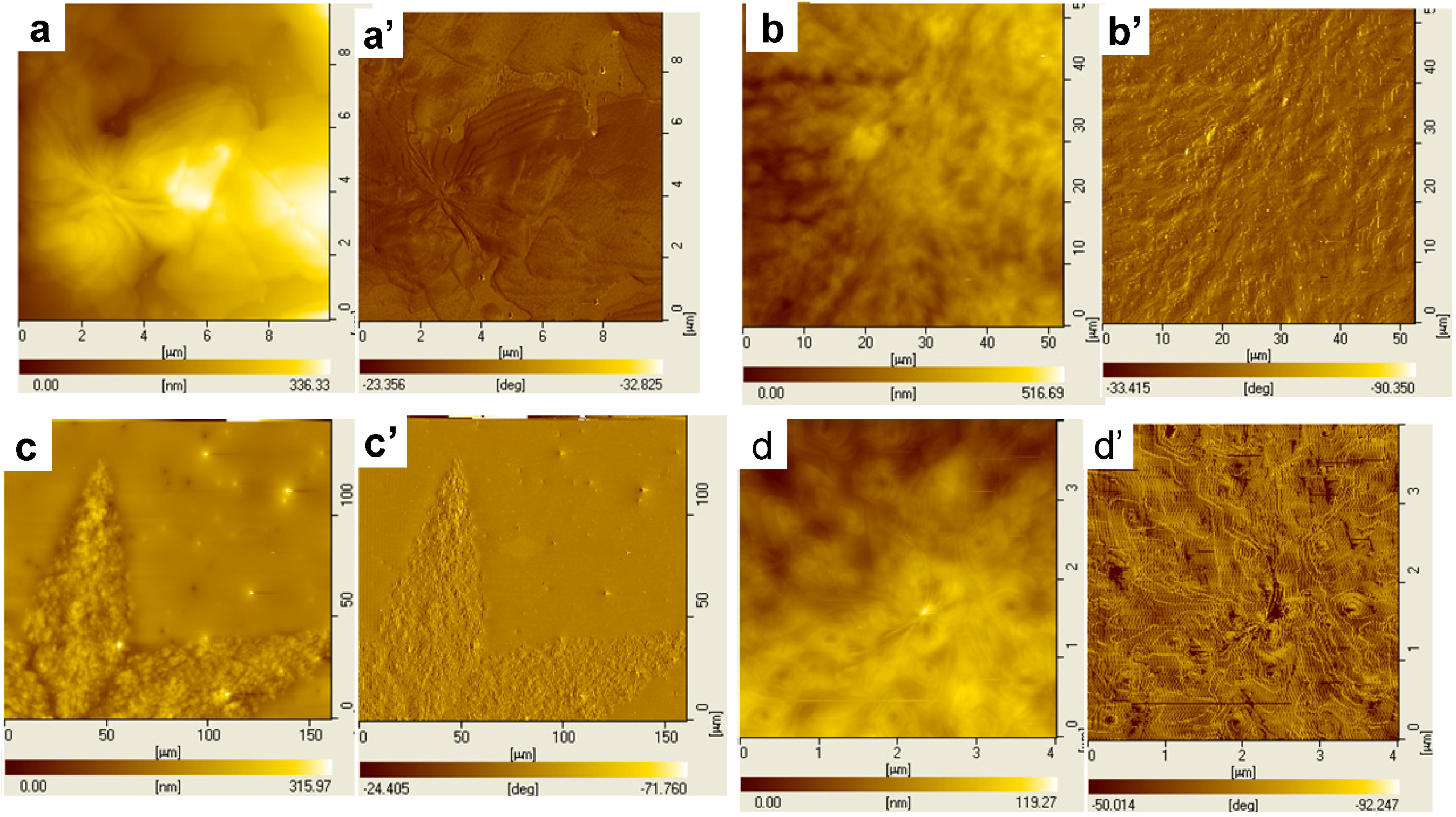
3.3. Dendritic Superstructures and Structure Transitions in PEO-b-PCL Diblock Copolymers [48]

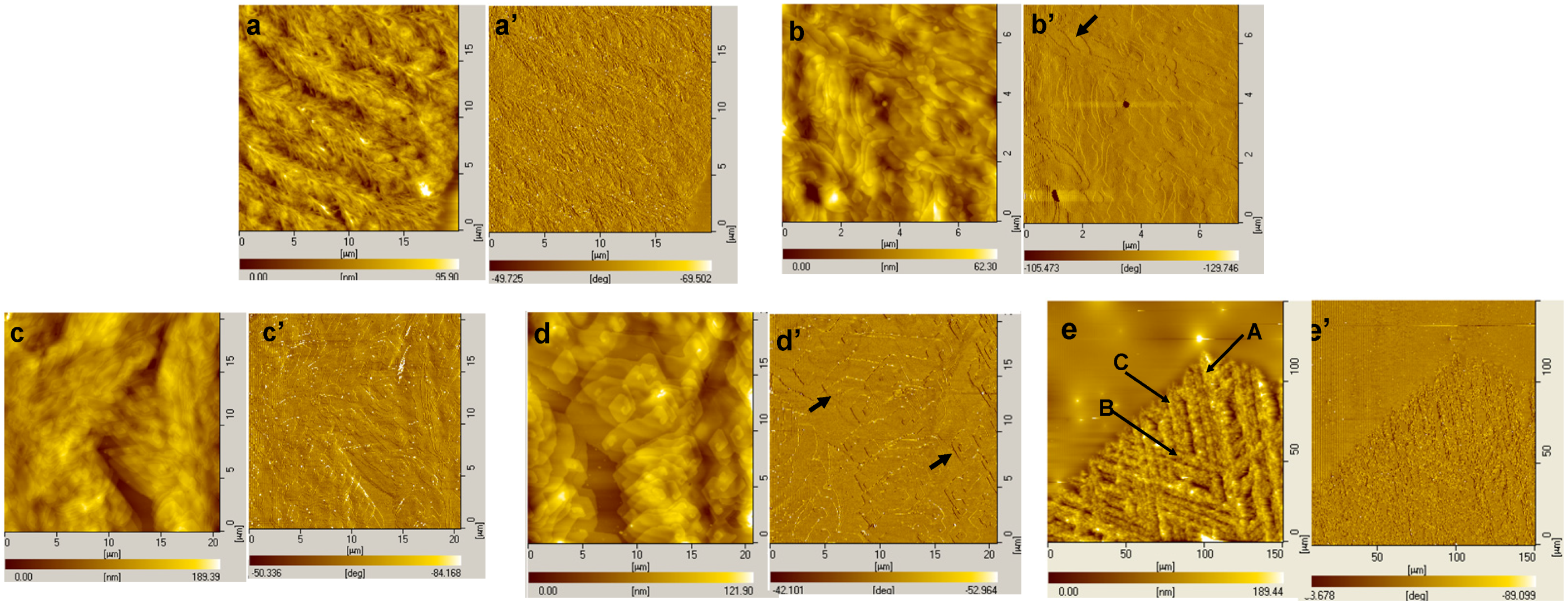





4. Summary and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hamley, I. Interfaces Crystallization Viscoelasticity; Springer: Berlin, Germany, 1999; p. 113. [Google Scholar]
- Müller, A.J.; Balsamo, V.; Arnal, M.L. Block Copolymers II; Springer: Berlin, Germany, 2005; p. 1. [Google Scholar]
- Muthukumar, M.; Ober, C.; Thomas, E.L. Competing interactions and levels of ordering in self-organizing polymeric materials. Science 1997, 277, 1225–1232. [Google Scholar] [CrossRef]
- Ryan, A.J.; Hamley, I.W.; Bras, W.; Bates, F.S. Structure development in semicrystalline diblock copolymers crystallizing from the ordered melt. Macromolecules 1995, 28, 3860–3868. [Google Scholar] [CrossRef]
- Rangarajan, P.; Register, R.A.; Fetters, L.J.; Bras, W.; Naylor, S.; Ryan, A.J. Crystallization of a Weakly Segregated Polyolefin Diblock Copolymer. Macromolecules 1995, 28, 4932–4938. [Google Scholar] [CrossRef]
- Sun, J.; Hong, Z.; Yang, L.; Tang, Z.; Chen, X.; Jing, X. Study on crystalline morphology of poly(l-lactide)-poly(ethylene glycol) diblock copolymer. Polymer 2004, 45, 5969–5977. [Google Scholar] [CrossRef]
- Huang, S.; Jiang, S.; An, L.; Chen, X. Crystallization and morphology of poly(ethylene oxide-b-lactide) crystalline–crystalline diblock copolymers. J. Polym. Sci. B Polym. Phys. 2008, 46, 1400–1411. [Google Scholar] [CrossRef]
- Södergård, A.; Stolt, M. Properties of lactic acid based polymers and their correlation with composition. Prog. Polym. Sci. 2002, 27, 1123–1163. [Google Scholar] [CrossRef]
- Uhrich, K.E.; Cannizzaro, S.M.; Langer, R.S.; Shakesheff, K.M. Polymeric systems for controlled drug release. Chem. Rev. 1999, 99, 3181–3198. [Google Scholar] [CrossRef]
- Bates, F.S.; Fredrickson, G.H. Block copolymer thermodynamics: Theory and experiment. Ann. Rev. Phys. Chem. 1990, 41, 525–557. [Google Scholar] [CrossRef]
- Zhu, L.; Cheng, S.Z.; Calhoun, B.H.; Ge, Q.; Quirk, R.P.; Thomas, E.L.; Hsiao, B.S.; Yeh, F.; Lotz, B. Crystallization temperature-dependent crystal orientations within nanoscale confined lamellae of a self-assembled crystalline–amorphous diblock copolymer. J. Am. Chem. Soc. 2000, 122, 5957–5967. [Google Scholar] [CrossRef]
- Müller, A.J.; Arnal, M.L.; Balsamo, V. Progress in Understanding of Polymer Crystallization; Springer: Berlin, Germany, 2007; p. 229. [Google Scholar]
- Rangarajan, P.; Register, R.A.; Adamson, D.H.; Fetters, L.J.; Bras, W.; Naylor, S.; Ryan, A.J. Dynamics of structure formation in crystallizable block copolymers. Macromolecules 1995, 28, 1422–1428. [Google Scholar] [CrossRef]
- Nojima, S.; Kato, K.; Yamamoto, S.; Ashida, T. Crystallization of block copolymers. 1. Small-angle X-ray scattering study of a .epsilon.-caprolactone-butadiene diblock copolymer. Macromolecules 1992, 25, 2237–2242. [Google Scholar] [CrossRef]
- Reiter, G.; Castelein, G.; Sommer, J.-U.; Röttele, A.; Thurn-Albrecht, T. Direct visualization of random crystallization and melting in arrays of nanometer-size polymer crystals. Phys. Rev. Lett. 2001, 87. [Google Scholar] [CrossRef]
- Hamley, I.; Castelletto, V.; Castillo, R.V.; Müller, A.J.; Martin, C.; Pollet, E.; Dubois, P. Crystallization in poly(l-lactide)-b-poly(ε-caprolactone) double crystalline diblock copolymers: A study using X-ray scattering, differential scanning calorimetry, and polarized optical microscopy. Macromolecules 2005, 38, 463–472. [Google Scholar] [CrossRef]
- Nojima, S.; Toei, M.; Hara, S.; Tanimoto, S.; Sasaki, S. Size dependence of crystallization within spherical microdomain structures. Polymer 2002, 43, 4087–4090. [Google Scholar] [CrossRef]
- Joncheray, T.J.; Denoncourt, K.M.; Meier, M.A.; Schubert, U.S.; Duran, R.S. Two-dimensional self-assembly of linear poly(ethylene oxide)-b-poly(ε-caprolactone) copolymers at the air–water interface. Langmuir 2007, 23, 2423–2429. [Google Scholar] [CrossRef]
- Naolou, T.; Busse, K.; Lechner, B.-D.; Kressler, J. The behavior of poly (ε-caprolactone) and poly (ethylene oxide)-b-poly (ε-caprolactone) grafted to a poly (glycerol adipate) backbone at the air/water interface. Colloid Polym. Sci. 2014, 292, 1199–1208. [Google Scholar] [CrossRef]
- Jiang, S.; He, C.; An, L.; Chen, X.; Jiang, B. Crystallization and ring-banded spherulite morphology of poly(ethylene oxide)-block-poly(ε-caprolactone) diblock copolymer. Macromol. Chem. Phys. 2004, 205, 2229–2234. [Google Scholar] [CrossRef]
- Keith, H.D. Banding in spherulites: two recurring topics. Polymer 2001, 42, 09987–09993. [Google Scholar] [CrossRef]
- Keith, H.D.; Padden, F.J., Jr. Banding in polyethylene and other spherulites. Macromolecules 1996, 29, 7776–7786. [Google Scholar] [CrossRef]
- Keith, H.D.; Padden, F.J., Jr.; Russell, T.P. Morphological changes in polyesters and polyamides induced by blending with small concentrations of polymer diluents. Macromolecules 1989, 22, 666–675. [Google Scholar] [CrossRef]
- Keith, H.D.; Padden, F.J., Jr. Twisting orientation and the role of transient states in polymer crystallization. Polymer 1984, 25, 28–42. [Google Scholar] [CrossRef]
- Saracovan, I.; Keith, H.D.; Manley, R.S.J.; Brown, G.R. Banding in spherulites of polymers having uncompensated main-chain chirality. Macromolecules 1999, 32, 8918–8922. [Google Scholar] [CrossRef]
- Wang, Z.G.; Wang, X.; Yu, D.; Jiang, B. The formation of ring-banded spherulites of poly(ɛ-caprolactone) in its miscible mixtures with poly(styrene-co-acrylonitrile). Polymer 1997, 38, 5897–5901. [Google Scholar] [CrossRef]
- Lotz, B.; Thierry, A. Spherulite morphology of form III isotactic poly(l-butene). Macromolecules 2003, 36, 286–290. [Google Scholar] [CrossRef]
- Bogdanov, B.; Vidts, A.; Schacht, E.; Berghmans, H. Isothermal crystallization of poly(ε-caprolactone–ethylene glycol) block copolymers. Macromolecules 1999, 32, 726–731. [Google Scholar] [CrossRef]
- Jiang, S.; He, C.; Men, Y.; Chen, X.; An, L.; Funari, S.S.; Chan, C.-M. Study of temperature dependence of crystallisation transitions of a symmetric PEO-PCL diblock copolymer using simultaneous SAXS and WAXS measurements with synchrotron radiation. Eur. Phys. J. E 2008, 27, 357–364. [Google Scholar] [CrossRef]
- Nojima, S.; Ito, K.; Ikeda, H. Composition dependence of crystallized lamellar morphology formed in crystalline–crystalline diblock copolymers. Polymer 2007, 48, 3607–3611. [Google Scholar] [CrossRef]
- Nojima, S.; Kiji, T.; Ohguma, Y. Characteristic melting behavior of double crystalline poly(ε-caprolactone)-block-polyethylene copolymers. Macromolecules 2007, 40, 7566–7572. [Google Scholar] [CrossRef]
- Nojima, S.; Akutsu, Y.; Akaba, M.; Tanimoto, S. Crystallization behavior of poly(ε-caprolactone) blocks starting from polyethylene lamellar morphology in poly(ε-caprolactone)-block-polyethylene copolymers. Polymer 2005, 46, 4060–4067. [Google Scholar] [CrossRef]
- Meng, F.; Hiemstra, C.; Engbers, G.H.M.; Feijen, J. Biodegradable polymersomes. Macromolecules 2003, 36, 3004–3006. [Google Scholar] [CrossRef]
- Cerrai, P.; Tricoli, M.; Andruzzi, F.; Paci, M.; Paci, M. Polyether-polyester block copolymers by non-catalysed polymerization of ɛ-caprolactone with poly(ethylene glycol). Polymer 1989, 30, 338–343. [Google Scholar] [CrossRef]
- Perret, R.; Skoulios, A. Synthèse et caractérisation de copolymères séquencés polyoxyéthylène/poly-ε-caprolactone. Makromol. Chem. 1972, 156, 143–156. (In Germany) [Google Scholar] [CrossRef]
- Wang, Z.-G.; Hsiao, B.S.; Sirota, E.B.; Agarwal, P.; Srinivas, S. Probing the early stages of melt crystallization in polypropylene by simultaneous small- and wide-angle X-ray scattering and laser light scattering. Macromolecules 2000, 33, 978–989. [Google Scholar] [CrossRef]
- Xue, F.; Chen, X.; An, L.; Funari, S.S.; Jiang, S. Soft nanoconfinement effects on the crystallization behavior of asymmetric poly(ethylene oxide)-block-poly(ε-caprolactone) diblock copolymers. Polym. Int. 2012, 61, 909–917. [Google Scholar] [CrossRef]
- Hamley, I.W. Solid state structure of block copolymers. In The Physics of Block Copolymers; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Harkless, C.R.; Singh, M.A.; Nagler, S.E.; Stephenson, G.B.; Jordan-Sweet, J.L. Small-angle X-ray-scattering study of ordering kinetics in a block copolymer. Phys. Rev. Lett. 1990, 64. [Google Scholar] [CrossRef]
- Oono, Y.; Bahiana, M. ⅔ -Power law for copolymer lamellar thickness implies a ⅓ -power law for spinodal decomposition. Phys. Rev. Lett. 1988, 61. [Google Scholar] [CrossRef]
- Hashimoto, T.; Kowsaka, K.; Shibayama, M.; Suehiro, S. Time-resolved small-angle X-ray scattering studies on the kinetics of the order-disorder transition of block polymers. 1. Experimental technique. Macromolecules 1986, 19, 750–754. [Google Scholar] [CrossRef]
- Hashimoto, T.; Kowsaka, K.; Shibayama, M.; Kawai, H. Time-resolved small-angle x-ray scattering studies on the kinetics of the order-disorder transition of block polymers. 2. Concentration and temperature dependence. Macromolecules 1986, 19, 754–762. [Google Scholar] [CrossRef]
- Xue, F.-F.; Chen, X.-S.; An, L.-J.; Funari, S.S. Confined lamella formation in crystalline-crystalline poly(ethylene oxide)-b-poly(ε-caprolactone) diblock copolymers. Chin. J. Polym. Sci. 2013, 31, 1260–1270. [Google Scholar] [CrossRef]
- Yasuniwa, M.; Tsubakihara, S.; Iura, K.; Ono, Y.; Dan, Y.; Takahashi, K. Crystallization behavior of poly(l-lactic acid). Polymer 2006, 47, 7554–7563. [Google Scholar] [CrossRef]
- Xu, J.; Guo, B.-H.; Zhou, J.-J.; Li, L.; Wu, J.; Kowalczuk, M. Observation of banded spherulites in pure poly(l-lactide) and its miscible blends with amorphous polymers. Polymer 2005, 46, 9176–9185. [Google Scholar] [CrossRef]
- Maillard, D.; Prud’ Homme, R.E. Crystallization of ultrathin films of polylactides: From chain chirality to lamella curvature and twisting. Macromolecules 2008, 41, 1705–1712. [Google Scholar] [CrossRef]
- Tsuji, H.; Tezuka, Y.; Saha, S.K.; Suzuki, M.; Itsuno, S. Spherulite growth of l-lactide copolymers: Effects of tacticity and comonomers. Polymer 2005, 46, 4917–4927. [Google Scholar] [CrossRef]
- Huang, S.; Jiang, S.; Chen, X.; An, L. Dendritic superstructures and structure transitions of asymmetric poly(l-lactide-b-ethylene oxide) diblock copolymer thin films. Langmuir 2009, 25, 13125–13132. [Google Scholar] [CrossRef]
- Zhang, J.; Tashiro, K.; Tsuji, H.; Domb, A.J. Disorder-to-order phase transition and multiple melting behavior of poly(l-lactide) investigated by simultaneous measurements of WAXD and DSC. Macromolecules 2008, 41, 1352–1357. [Google Scholar] [CrossRef]
- Cho, T.-Y.; Strobl, G. Temperature dependent variations in the lamellar structure of poly(l-lactide). Polymer 2006, 47, 1036–1043. [Google Scholar] [CrossRef]
- Zhang, J.; Duan, Y.; Sato, H.; Tsuji, H.; Noda, I.; Yan, S.; Ozaki, Y. Crystal modifications and thermal behavior of poly(l-lactic acid) revealed by infrared spectroscopy. Macromolecules 2005, 38, 8012–8021. [Google Scholar] [CrossRef]
- Di Lorenzo, M.L. Determination of spherulite growth rates of poly(l-lactic acid) using combined isothermal and non-isothermal procedures. Polymer 2001, 42, 9441–9446. [Google Scholar] [CrossRef]
- Abe, H.; Kikkawa, Y.; Inoue, Y.; Doi, Y. Morphological and kinetic analyses of regime transition for poly[(S)-lactide] crystal growth. Biomacromolecules 2001, 2, 1007–1014. [Google Scholar] [CrossRef]
- Mijovic, J.; Sy, J.-W. Molecular dynamics during crystallization of poly(l-lactic acid) as studied by broad-band dielectric relaxation spectroscopy. Macromolecules 2002, 35, 6370–6376. [Google Scholar] [CrossRef]
- Yang, J.; Zhao, T.; Zhou, Y.; Liu, L.; Li, G.; Zhou, E.; Chen, X. Single crystals of the poly(l-lactide) block and the poly(ethylene glycol) block in poly(l-lactide)–poly(ethylene glycol) diblock copolymer. Macromolecules 2007, 40, 2791–2797. [Google Scholar] [CrossRef]
- Kikkawa, Y.; Abe, H.; Iwata, T.; Inoue, Y.; Doi, Y. In situ observation of crystal growth for poly[(S)-lactide] by temperature-controlled atomic force microscopy. Biomacromolecules 2001, 2, 940–945. [Google Scholar] [CrossRef]
- Fujita, M.; Doi, Y. Annealing and melting behavior of poly(l-lactic acid) single crystals as revealed by in situ atomic force microscopy. Biomacromolecules 2003, 4, 1301–1307. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xue, F.; Jiang, S. Crystallization Behaviors and Structure Transitions of Biocompatible and Biodegradable Diblock Copolymers. Polymers 2014, 6, 2116-2145. https://doi.org/10.3390/polym6082116
Xue F, Jiang S. Crystallization Behaviors and Structure Transitions of Biocompatible and Biodegradable Diblock Copolymers. Polymers. 2014; 6(8):2116-2145. https://doi.org/10.3390/polym6082116
Chicago/Turabian StyleXue, Feifei, and Shichun Jiang. 2014. "Crystallization Behaviors and Structure Transitions of Biocompatible and Biodegradable Diblock Copolymers" Polymers 6, no. 8: 2116-2145. https://doi.org/10.3390/polym6082116
APA StyleXue, F., & Jiang, S. (2014). Crystallization Behaviors and Structure Transitions of Biocompatible and Biodegradable Diblock Copolymers. Polymers, 6(8), 2116-2145. https://doi.org/10.3390/polym6082116



