Connexin 43 Gene Therapy Delivered by Polymer-Modified Salmonella in Murine Tumor Models
Abstract
:1. Introduction
2. Experimental Section
2.1. Cells, Bacteria, Reagents, and Mice
2.2. Preparation of DNA-PAH-Modified Salmonella
2.3. Characterization of the DNA-PAH-Modified Salmonella
2.4. Animal Studies
2.5. Infection of Tumor Cells with Salmonella
2.6. Western Blot Analysis
2.7. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Poly(allylamine hydrochloride) (PAH)-Modified Salmonella
| Bacteria | Particle size (nm) | Zeta potential (mV) |
|---|---|---|
| Salmonella | 1068.25 ± 80.52 | −13.8 ± 1.60 |
| PAH-Salmonella | 2279.25 ± 415.58 | 6.08 ± 3.93 |
| DNA-PAH-Salmonella | 4774.00 ± 993.37 | −22 ± 6.89 |
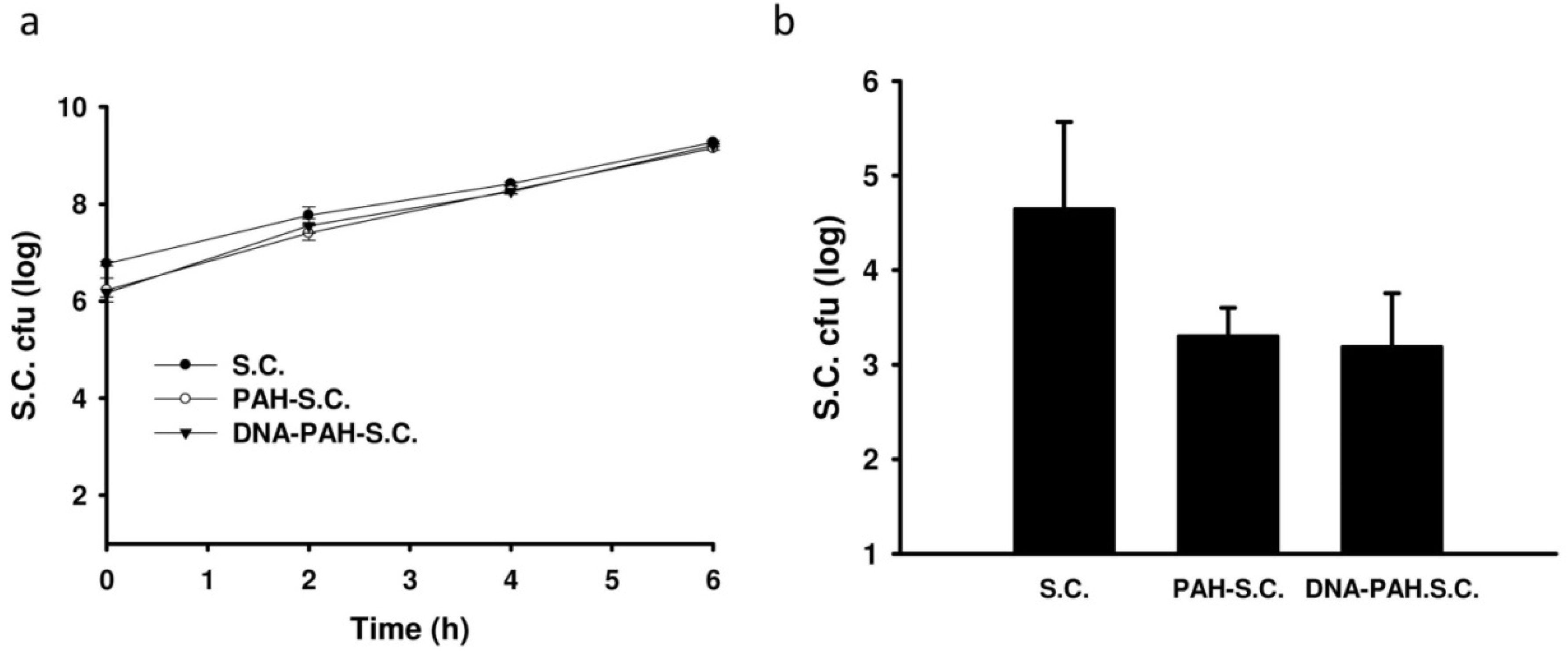
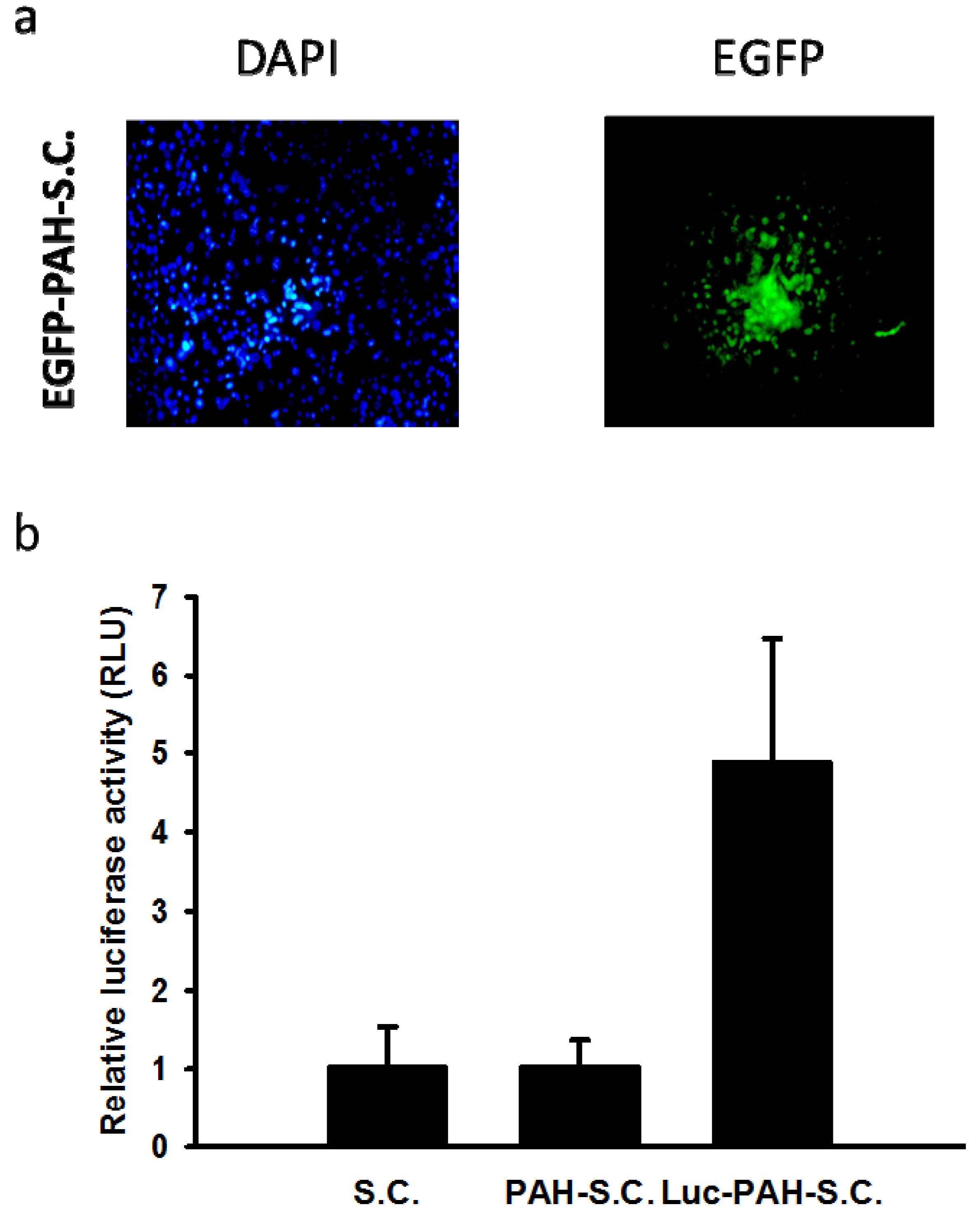
3.2. Tissue Distributions of DNA-PAH-S.C. in Mice
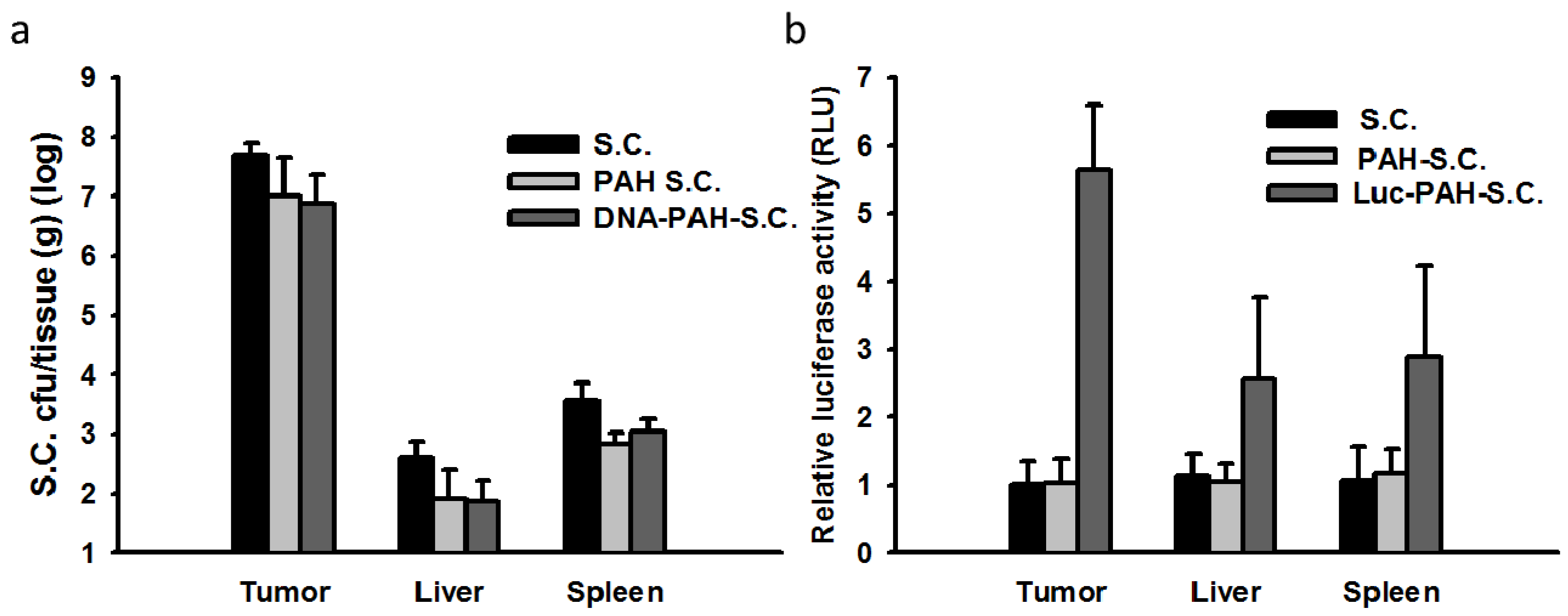
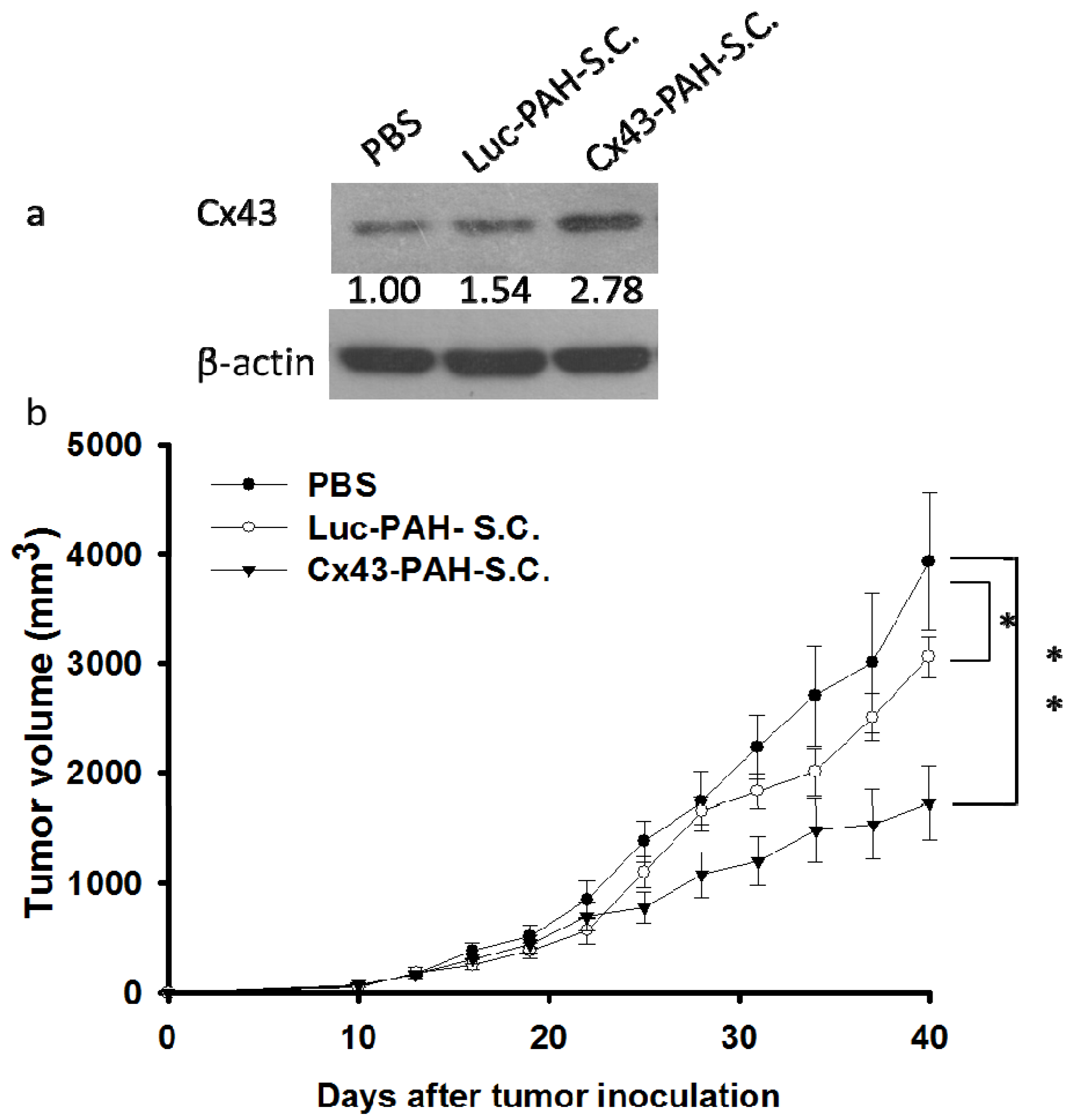
3.3. Inhibition of Tumor Growth by DNA-PAH-Modified Salmonella
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, F.; Zhang, L.; Hoffman, R.M.; Zhao, M. Vessel destruction by tumor-targeting Salmonella typhimurium A1-R is enhanced by high tumor vascularity. Cell. Cycle 2010, 9, 4518–4524. [Google Scholar] [CrossRef]
- Nagakura, C.; Hayashi, K.; Zhao, M.; Yamauchi, K.; Yamamoto, N.; Tsuchiya, H.; Tomita, K.; Bouvet, M.; Hoffman, R.M. Efficacy of a genetically-modified Salmonella typhimurium in an orthotopic human pancreatic cancer in nude mice. Anticancer Res. 2009, 29, 1873–1878. [Google Scholar]
- Yam, C.; Zhao, M.; Hayashi, K.; Ma, H.; Kishimoto, H.; McElroy, M.; Bouvet, M.; Hoffman, R.M. Monotherapy with a tumor-targeting mutant of S. typhimurium inhibits liver metastasis in a mouse model of pancreatic cancer. J. Surg. Res. 2010, 164, 248–255. [Google Scholar] [CrossRef]
- Hayashi, K.; Zhao, M.; Yamauchi, K.; Yamamoto, N.; Tsuchiya, H.; Tomita, K.; Kishimoto, H.; Bouvet, M.; Hoffman, R.M. Systemic targeting of primary bone tumor and lung metastasis of high-grade osteosarcoma in nude mice with a tumor-selective strain of Salmonella typhimurium. Cell. Cycle 2009, 8, 870–875. [Google Scholar] [CrossRef]
- Kimura, H.; Zhang, L.; Zhao, M.; Hayashi, K.; Tsuchiya, H.; Tomita, K.; Bouvet, M.; Wessels, J.; Hoffman, R.M. Targeted therapy of spinal cord glioma with a genetically modified Salmonella typhimurium. Cell. Prolif. 2010, 43, 41–48. [Google Scholar] [CrossRef]
- Lee, C.H.; Wu, C.L.; Shiau, A.L. Endostatin gene therapy delivered by Salmonella choleraesuis in murine tumor models. J. Gene Med. 2004, 6, 1382–1393. [Google Scholar] [CrossRef]
- Zhao, M.; Yang, M.; Ma, H.; Li, X.; Tan, X.; Li, S.; Yang, Z.; Hoffman, R.M. Targeted therapy with a Salmonella typhimurium leucine-arginine auxotroph cures orthotopic human breast tumors in nude mice. Cancer Res. 2006, 66, 7647–7652. [Google Scholar] [CrossRef]
- Zhao, M.; Yang, M.; Li, X.M.; Jiang, P.; Baranov, E.; Li, S.; Xu, M.; Penman, S.; Hoffman, R.M. Tumor-targeting bacterial therapy with amino acid auxotrophs of GFP-expressing Salmonella typhimurium. Proc. Natl. Acad. Sci. USA 2005, 102, 755–760. [Google Scholar]
- Harris, T.J.; Green, J.J.; Fung, P.W.; langer, R.; Anderson, D.G.; Bhatia, S.N. Tissue-specific gene delivery via nanoparticle coating. Biomaterials 2010, 31, 998–1006. [Google Scholar] [CrossRef]
- Chang, W.W.; Lai, C.H.; Chen, M.C.; Liu, C.F.; Kuan, Y.D.; Lin, S.T.; Lee, C.H. Salmonella enhance chemosensitivity in tumor through connexin 43 upregulation. Int. J. Cancer 2013, 133, 1926–1935. [Google Scholar] [CrossRef]
- Chang, W.W.; Kuan, Y.D.; Chen, M.C.; Lin, S.T.; Lee, C.H. Tracking of mouse breast cancer stem-like cells with Salmonella. Exp. Biol. Med. 2012, 237, 1189–1196. [Google Scholar]
- Lee, C.H.; Lin, Y.H.; Hsieh, J.L.; Chen, M.C.; Kuo, W.L. A polymer coating applied to Salmonella prevents the binding of Salmonella-specific antibodies. Int. J. Cancer 2013, 132, 717–725. [Google Scholar] [CrossRef]
- Shiau, A.L.; Shen, Y.T.; Hsieh, J.L.; Wu, C.L.; Lee, C.H. Scutellaria barbata inhibits angiogenesis through downregulation of HIF-1 α in lung tumor. Environ. Toxicol. 2014, 29, 363–370. [Google Scholar] [CrossRef]
- Chang, W.W.; Liu, J.J.; Liu, C.F.; Liu, W.S.; Lim, Y.P.; Cheng, Y.J.; Lee, C.H. An extract of Rhodobacter sphaeroides reduces cisplatin-induced nephrotoxicity in mice. Toxins 2013, 5, 2353–2365. [Google Scholar]
- ImageJ. Image Processing and Analysis in Java. Available online: http://rsbweb.nih.gov/ij/ (accessed on 6 September 2013).
- Liu, W.S.; Kuan, Y.D.; Chiu, K.H.; Wang, W.K.; Chang, F.H.; Liu, C.H.; Lee, C.H. The extract of Rhodobacter sphaeroides inhibits melanogenesis through the MEK/ERK signaling pathway. Mar. Drugs 2013, 11, 1899–1908. [Google Scholar]
- Lee, C.H.; Lin, S.T.; Liu, J.J.; Chang, W.W.; Hsieh, J.L.; Wang, W.K. Salmonella induce autophagy in melanoma by the downregulation of AKT/mTOR pathway. Gene Ther. 2014, 21, 309–316. [Google Scholar]
- Lee, C.H.; Wu, C.L.; Chen, S.H.; Shiau, A.L. Humoral immune responses inhibit the antitumor activities mediated by Salmonella enterica serovar Choleraesuis. J. Immunother. 2009, 32, 376–388. [Google Scholar]
- Ableser, M.J.; Penuela, S.; Lee, J.; Shao, Q.; Laird, D.W. Connexin43 reduces melanoma growth within a keratinocyte microenvironment and during tumorigenesis in vivo. J. Biol. Chem. 2014, 289, 1592–1603. [Google Scholar]
- Cheng, Y.J.; Chang, M.Y.; Chang, W.W.; Wang, W.K.; Liu, C.F.; Lin, S.T.; Lee, C.H. Resveratrol enhances chemosensitivity in mouse melanoma model through connexin 43 upregulation. Environ. Toxicol. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, W.-K.; Kuan, Y.-D.; Kuo, C.-Y.; Lee, C.-H. Connexin 43 Gene Therapy Delivered by Polymer-Modified Salmonella in Murine Tumor Models. Polymers 2014, 6, 1119-1128. https://doi.org/10.3390/polym6041119
Wang W-K, Kuan Y-D, Kuo C-Y, Lee C-H. Connexin 43 Gene Therapy Delivered by Polymer-Modified Salmonella in Murine Tumor Models. Polymers. 2014; 6(4):1119-1128. https://doi.org/10.3390/polym6041119
Chicago/Turabian StyleWang, Wei-Kuang, Yu-Diao Kuan, Chun-Yu Kuo, and Che-Hsin Lee. 2014. "Connexin 43 Gene Therapy Delivered by Polymer-Modified Salmonella in Murine Tumor Models" Polymers 6, no. 4: 1119-1128. https://doi.org/10.3390/polym6041119
APA StyleWang, W.-K., Kuan, Y.-D., Kuo, C.-Y., & Lee, C.-H. (2014). Connexin 43 Gene Therapy Delivered by Polymer-Modified Salmonella in Murine Tumor Models. Polymers, 6(4), 1119-1128. https://doi.org/10.3390/polym6041119




