Biodegradable Covalently Crosslinked Poly[N-(2-Hydroxypropyl) Methacrylamide] Nanogels: Preparation and Physicochemical Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Monomers
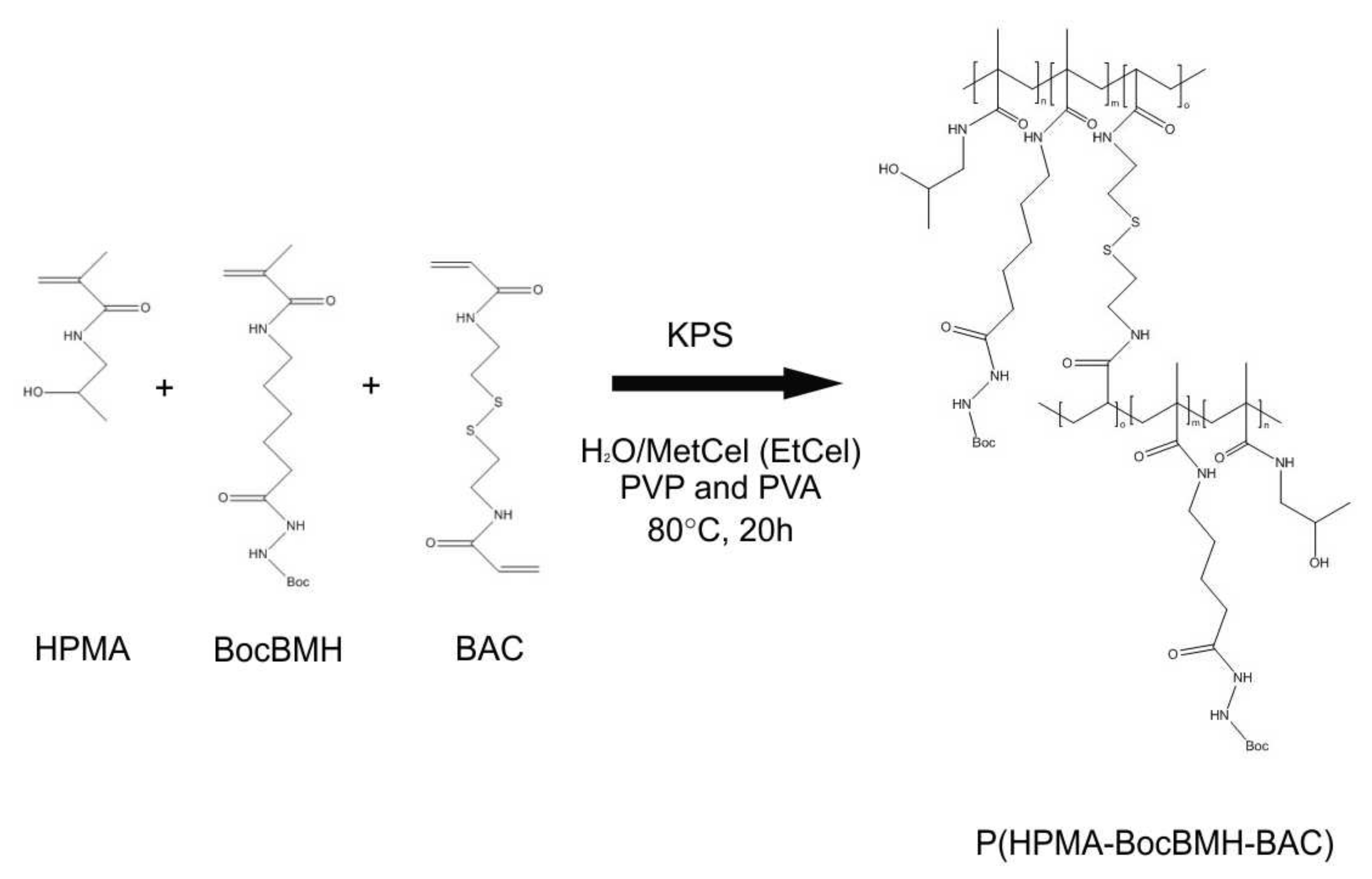
2.3. Dispersion Polymerization of N-(2-Hydroxypropyl) Methacrylamide, N-(Tert-butoxycarbonyl)-N′-(6-methacrylamidohexanoyl) Hydrazine, and N,N′-Bis(acryloyl) Cystamine
2.4. Study of Nanogel Degradation
2.5. Characterization
3. Results and Discussion
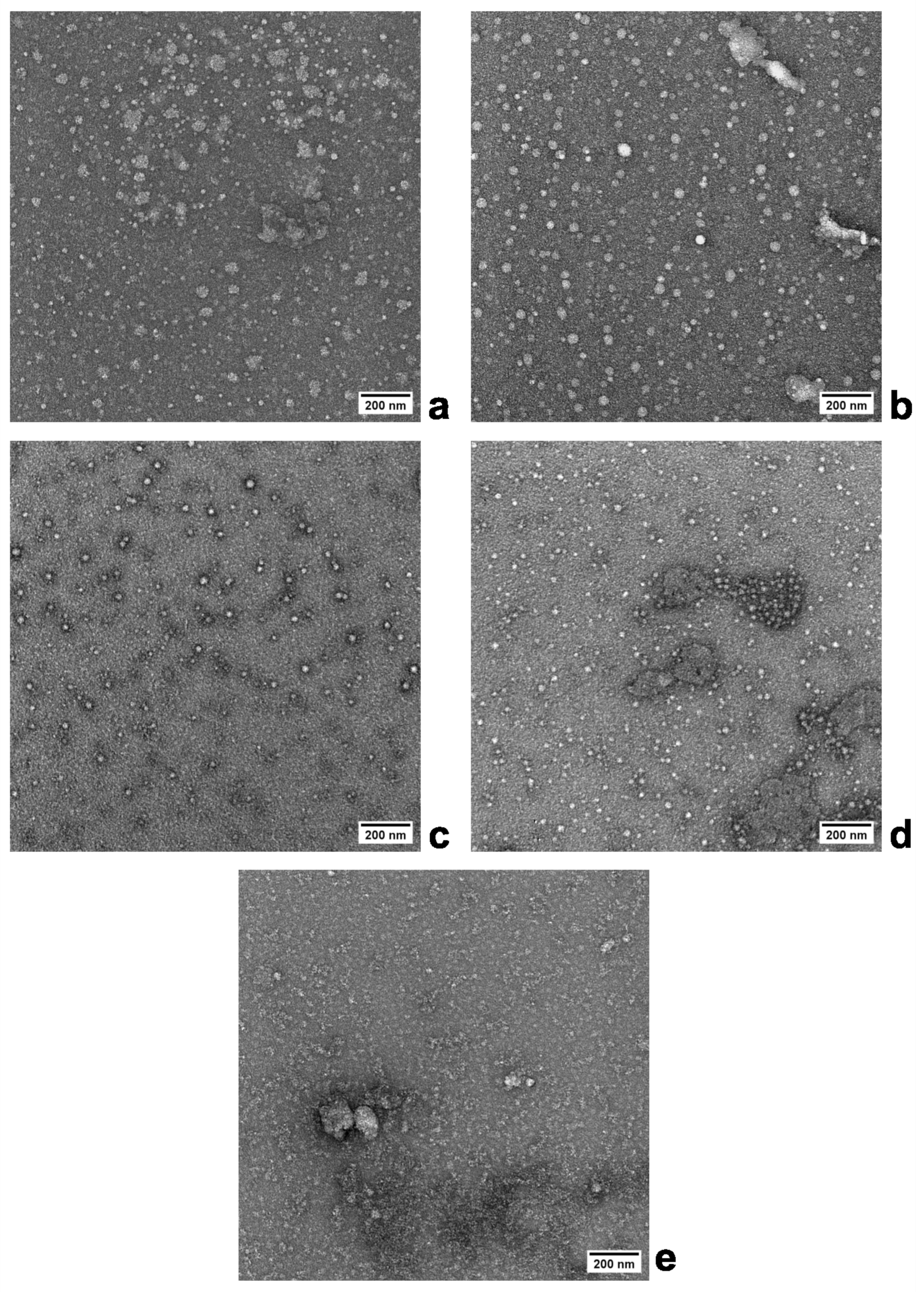
3.1. Preparation of Nanogels—Effect of the BAC Crosslinking Comonomer Concentration
3.2. Preparation of Nanogels—Effect of the Polymerization Medium Composition
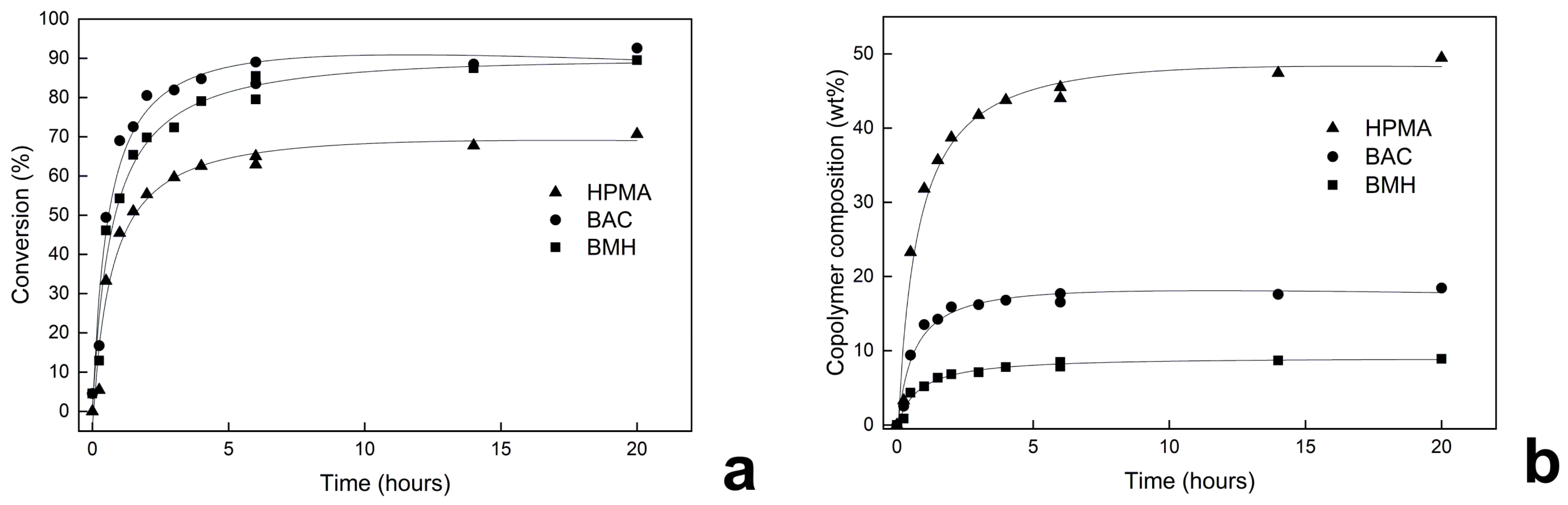
3.3. Kinetic Study of Dispersion Polymerization
3.4. Study of Nanogel Degradation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, S.; Tang, J.; Ji, F.; Lin, W.; Chen, S. Recent advances in zwitterionic hydrogels: Preparation, property, and biomedical application. Gels 2022, 8, 46. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhang, S.; Wu, F.; Li, D.; Zhang, X.; Chen, W.; Xing, B. Rational design of nanogels for overcoming the biological barriers in various administration routes. Angew. Chem. Int. Ed. 2021, 60, 14760–14778. [Google Scholar] [CrossRef] [PubMed]
- Gray, D.M.; Town, A.R.; Niezabitowska, E.; Rannard, S.P.; McDonald, T.O. Dual-responsive degradable core-shell nanogels with tuneable aggregation behavior. RSC Adv. 2022, 12, 2196–2206. [Google Scholar] [CrossRef] [PubMed]
- Hajebi, S.; Abdollahi, A.; Roghani-Mamaqani, H.; Salami-Kalajahi, M. Temperature-responsive poly(N-isopropylacrylamide) nanogels: The role of hollow cavities and different shell cross-linking densities on doxorubicin loading and release. Langmuir 2020, 36, 2683–2694. [Google Scholar] [CrossRef]
- Kaniewska, K.; Marcisz, K.; Karbarz, M. Transport of ionic species affected by interactions with a pH-sensitive monolayer of microgel particles attached to electrode surface. J. Electroanal. Chem. 2023, 931, 117183. [Google Scholar] [CrossRef]
- Clegg, J.R.; Irani, A.S.; Ander, E.W.; Ludolph, C.M.; Venkataraman, A.K.; Zhong, J.X.; Peppas, N.A. Synthetic networks with tunable responsiveness, biodegradation, and molecular recognition for precision medicine applications. Sci. Adv. 2023, 5, eaax7946. [Google Scholar] [CrossRef] [PubMed]
- Maruf, A.; Milewska, M.; Lalik, A.; Student, S.; Wandzik, I. A simple synthesis of reduction-responsive acrylamide-type nanogels for miRNA delivery. Molecules 2023, 28, 761. [Google Scholar] [CrossRef]
- Oh, J.K.; Bencherif, S.A.; Matyjaszewski, K. Atom transfer radical polymerization in inverse miniemulsion: A versatile route toward preparation and functionalization of microgels/nanogels for targeted drug delivery applications. Polymer 2009, 50, 4407–4423. [Google Scholar] [CrossRef]
- Kawaguchi, S.; Ito, K. Dispersion Polymerization. In Polymer Particles; Okubo, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 299–328. [Google Scholar] [CrossRef]
- Uyama, H.; Kobayashi, S. Dispersion polymerization of styrene in aqueous alcohol solution: Effects of reaction parameters on the polymer particle formation. Polym. Int. 1994, 34, 339–344. [Google Scholar] [CrossRef]
- Macková, H.; Horák, D. Effects of the reaction parameters on the properties of thermosensitive poly(N-isopropylacrylamide) microspheres prepared by precipitation and dispersion polymerization. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 968–982. [Google Scholar] [CrossRef]
- Uyama, H.; Kato, H.; Kobayashi, S. Dispersion polymerization of N-vinylformamide in polar media. Preparation of monodisperse hydrophilic polymer particles. Polym. J. 1994, 26, 858–863. [Google Scholar] [CrossRef][Green Version]
- Takahashi, K.; Uyama, H.; Kobayashi, S. Preparation of reactive monodisperse particles in the micron range by dispersion polymerization of glycidyl methacrylate. Polym. J. 1998, 30, 684–686. [Google Scholar] [CrossRef]
- Horák, D. Effect of reaction parameters on the particle size in the dispersion polymerization of 2-hydroxyethyl methacrylate. J. Polym. Sci. Part A Polym. Chem. 1999, 37, 3785–3792. [Google Scholar] [CrossRef]
- Chytil, P.; Kostka, L.; Etrych, T. HPMA copolymer-based nanomedicines in controlled drug delivery. J. Pers. Med. 2021, 11, 115. [Google Scholar] [CrossRef]
- Talelli, M.; Rijcken, C.J.F.; van Nostrum, C.F.; Storm, G.; Hennink, W.E. Micelles based on HPMA copolymers. Adv. Drug Deliv. Rev. 2010, 62, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Zhou, M.; Huang, Y.; Zhong, J.; Zhou, Z.; Luo, K. Dual-sensitive and biodegradable core-crosslinked HPMA copolymer–doxorubicin conjugate-based nanoparticles for cancer therapy. Polym. Chem. 2017, 8, 2370–2380. [Google Scholar] [CrossRef]
- Phan, H.; Cavanagh, R.; Jacob, P.; Destouches, D.; Vacherot, F.; Brugnoli, B.; Howdle, S.; Taresco, V.; Couturaud, B. Synthesis of multifunctional polymersomes prepared by polymerization-induced self-assembly. Polymers 2023, 15, 3070. [Google Scholar] [CrossRef]
- Šálek, P.; Zbořilová, D.; Pavlova, E.; Kočková, O.; Konefal, R.; Morávková, Z.; Janoušková, O. Fluorescent poly[N-(2-hydroxypropyl) methacrylamide] nanogel by dispersion polymerization as a contrast agent for live-cell imaging. J. Appl. Polym. Sci. 2023, 140, e54331. [Google Scholar] [CrossRef]
- Wutzel, H.; Richter, F.H.; Li, Y.; Sheiko, S.S.; Klok, H.-A. Poly[N-(2-hydroxypropyl)methacrylamide] nanogels by RAFT polymerization in inverse emulsion. Polym. Chem. 2014, 5, 1711–1719. [Google Scholar] [CrossRef]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Chytil, P.; Etrych, T.; Kříž, J.; Subr, V.; Ulbrich, K. N-(2-Hydroxypropyl)methacrylamide-based polymer conjugates with pH-controlled activation of doxorubicin for cell-specific or passive tumour targeting. Synthesis by RAFT polymerisation and physicochemical characterisation. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2010, 41, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Ulbrich, K.; Etrych, T.; Chytil, P.; Jelínková, M.; Říhová, B. Antibody-targeted polymer-doxorubicin conjugates with pH-controlled activation. J. Drug Target. 2004, 12, 477–489. [Google Scholar] [CrossRef]
- Etrych, T.; Mrkvan, T.; Chytil, P.; Koňák, Č.; Říhová, B.; Ulbrich, K. N-(2-hydroxypropyl) methacrylamide-based polymer conjugates with pH-controlled activation of doxorubicin. I. New synthesis, physicochemical characterization and preliminary biological evaluation. J. Appl. Polym. Sci. 2008, 109, 3050–3061. [Google Scholar] [CrossRef]
- Abbrent, S.; Mahun, A.; Smrčková, M.D.; Kobera, L.; Konefał, R.; Černoch, P.; Dušek, K.; Brus, J. Copolymer chain formation of 2-oxazolines by in situ 1H-NMR spectroscopy: Dependence of sequential composition on substituent structure and monomer ratios. RSC Adv. 2021, 11, 10468–10478. [Google Scholar] [CrossRef]
- Gottlieb, H.E.; Kotlyar, V.; Nudelman, A. NMR chemical shifts of common laboratory solvents as trace impurities. J. Org. Chem. 1997, 62, 7512–7515. [Google Scholar] [CrossRef]
- Filipe, V.; Hawe, A.; Jiskoot, W. Critical evaluation of Nanoparticle tracking analysis (NTA) by nanosight for the measurement of nanoparticles and protein aggregates. Pharm. Res. 2010, 27, 796–810. [Google Scholar] [CrossRef] [PubMed]
- Šálek, P.; Filipová, M.; Horák, D.; Proks, V.; Janoušková, O. Enhanced solid phase extraction of DNA using hydrophilic monodisperse poly(methacrylic acid-co-ethylene dimethacrylate) microparticles. Mol. Biol. Rep. 2019, 46, 3063–3072. [Google Scholar] [CrossRef]
- Cors, M.; Wrede, O.; Wiehemeier, L.; Feoktystov, A.; Cousin, F.; Hellweg, T.; Oberdisse, J. Spatial distribution of core monomers in acrylamide-based core-shell microgels with linear swelling behavior. Sci. Rep. 2019, 9, 13812. [Google Scholar] [CrossRef]
- Ashrafizadeh, M.; Tam, K.C.; Javadi, A.; Abdollahi, M.; Sadeghnejad, S.; Bahramian, A. Synthesis and physicochemical properties of dual-responsive acrylic acid/butyl acrylate cross-linked nanogel systems. J. Colloid Interface Sci. 2019, 556, 313–323. [Google Scholar] [CrossRef]
- Ponomareva, E.; Tadgell, B.; Hildebrandt, M.; Krüsmann, M.; Prévost, S.; Mulvaney, P.; Karg, M. The fuzzy sphere morphology is responsible for the increase in light scattering during the shrinkage of thermoresponsive microgels. Soft Matter. 2022, 18, 807–825. [Google Scholar] [CrossRef]
- Town, A.; Niezabitowska, E.; Kavanagh, J.; Barrow, M.; Kearns, V.R.; García-Tuñón, E.; McDonald, T.O. Understanding the phase and morphological behavior of dispersions of synergistic dual-stimuli-responsive poly(N-isopropylacrylamide) nanogels. J. Phys. Chem. B 2019, 123, 6303–6313. [Google Scholar] [CrossRef]
- Virtanen, O.L.J.; Richtering, W. Kinetics and particle size control in non-stirred precipitation polymerization of N-isopropylacrylamide. Colloid Polym. Sci. 2014, 292, 1743–1756. [Google Scholar] [CrossRef]
- Onita, K.; Onishi, M.; Omura, T.; Wakiya, T.; Suzuki, T.; Minami, H. Preparation of monodisperse bio-based polymer particles via dispersion polymerization. Langmuir 2022, 38, 7341–7345. [Google Scholar] [CrossRef]
- Niezabitowska, E.; Town, A.R.; Sabagh, B.; Moctezuma, M.D.M.; Kearns, V.R.; Spain, S.G.; Rannard, S.P.; Mcdonald, T.O. Insights into the internal structures of nanogels using a versatile Asymmetric-flow field-flow fractionation method. Nanoscale Adv. 2020, 2, 4713–4721. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, L.; Yang, Y.; Xu, X.; Huang, Y. Tumor targeting by pH-sensitive, biodegradable, cross-linked N-(2-hydroxypropyl) methacrylamide copolymer micelles. Biomaterials 2014, 35, 6622–6635. [Google Scholar] [CrossRef]
- Manimaran, V.; Nivetha, R.P.; Tamilanban, T.; Narayanan, J.; Vetriselvan, S.; Fuloria, N.K.; Chinni, S.V.; Sekar, M.; Fuloria, S.; Wong, L.S.; et al. Nanogels as novel drug nanocarriers for CNS drug delivery. Front. Mol. Biosci. 2023, 10, 1232109. [Google Scholar] [CrossRef] [PubMed]
- Duracher, D.; Elaïssari, A.; Pichot, C. Preparation of poly(N-isopropylmethacrylamide) latexes kinetic studies and characterization. J. Polym. Sci. Part A Polym. Chem. 1999, 37, 1823–1837. [Google Scholar] [CrossRef]
- Hazot, P.; Chapel, J.P.; Pichot, C.; Elaissari, A.; Delair, T. Preparation of poly(N-ethyl methacrylamide) particles via an emulsion/precipitation process: The role of the crosslinker. J. Polym. Sci. Part A Polym. Chem. 2002, 40, 1808–1817. [Google Scholar] [CrossRef]
- Liu, P.; Pearce, C.M.; Anastasiadi, R.M.; Resmini, M.; Castilla, A.M. Covalently crosslinked nanogels: An NMR study of the effect of monomer reactivity on composition and structure. Polymers 2019, 11, 353. [Google Scholar] [CrossRef] [PubMed]
- Etrych, T.; Kovář, L.; Šubr, V.; Braunová, A.; Pechar, M.; Chytil, P.; Říhova, B.; Ulbrich, K. High-molecular-weight polymers containing biodegradable disulfide bonds: Synthesis and in vitro verification of intracellular degradation. J. Bioact. Compat. Polym. 2010, 25, 5–26. [Google Scholar] [CrossRef]






| Sample | Solvent (wt%) | BAC (wt%) | Yield (%) | TEM | DLS | A4F | ρ | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Dn (nm) | Dw (nm) | Đ | Dh (nm) | PDI | Dg (nm) | MR (%) | |||||
| NG1 ** | H2O/MetCel (80/20) | 5 | 49 | 28 | 52 | 1.86 | 84.5 | 0.376 | 26.2 | 71.4 | 0.31 |
| NG2 ** | H2O/MetCel (80/20) | 10 | 54 | 21 | 23 | 1.10 | 41.8 | 0.509 | 15.8 | 76 | 0.38 |
| NG3 ** | H2O/MetCel (80/20) | 15 | 52 | 13 | 15 | 1.15 | 79.1 | 0.472 | 37.8 | 60.9 | 0.48 |
| NG4 ** | H2O/MetCel (80/20) | 20 | 55 | 22 | 25 | 1.14 | 71.4 | 0.942 | 24 | 95.6 | 0.34 |
| NG5 ** | H2O/MetCel (80/20) | 25 | 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
| NG6 ** | H2O/MetCel (75/25) | 20 | 54 | n/a | n/a | n/a | 50.6 | 0.663 | 23.8 | 77.5 | 0.47 |
| NG7 ** | H2O/MetCel (85/15) | 20 | 52 | n/a | n/a | n/a | 46.5 | 0.842 | 21.2 | 83.5 | 0.46 |
| NG8 ** | H2O/EtCel (75/25) | 20 | 54 | 20 | 37 | 1.85 | 35.7 | 0.526 | 22 | 89.3 | 0.62 |
| NG9 ** | H2O/EtCel (80/20) | 20 | 51 | 23 | 30 | 1.30 | 34.6 | 0.561 | 18.2 | 71.4 | 0.53 |
| NG10 ** | H2O/EtCel (85/15) | 20 | 54 | 36 | 45 | 1.37 | 34 | 0.654 | 24 | 86 | 0.71 |
| NG11 *** | H2O/MetCel (80/20) | 20 | 48 | 30 | 35 | 1.16 | 38 | 0.494 | 16.6 | 39.1 | 0.44 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kousalová, J.; Šálek, P.; Pavlova, E.; Konefał, R.; Kobera, L.; Brus, J.; Kočková, O.; Etrych, T. Biodegradable Covalently Crosslinked Poly[N-(2-Hydroxypropyl) Methacrylamide] Nanogels: Preparation and Physicochemical Properties. Polymers 2024, 16, 263. https://doi.org/10.3390/polym16020263
Kousalová J, Šálek P, Pavlova E, Konefał R, Kobera L, Brus J, Kočková O, Etrych T. Biodegradable Covalently Crosslinked Poly[N-(2-Hydroxypropyl) Methacrylamide] Nanogels: Preparation and Physicochemical Properties. Polymers. 2024; 16(2):263. https://doi.org/10.3390/polym16020263
Chicago/Turabian StyleKousalová, Jana, Petr Šálek, Ewa Pavlova, Rafał Konefał, Libor Kobera, Jiří Brus, Olga Kočková, and Tomáš Etrych. 2024. "Biodegradable Covalently Crosslinked Poly[N-(2-Hydroxypropyl) Methacrylamide] Nanogels: Preparation and Physicochemical Properties" Polymers 16, no. 2: 263. https://doi.org/10.3390/polym16020263
APA StyleKousalová, J., Šálek, P., Pavlova, E., Konefał, R., Kobera, L., Brus, J., Kočková, O., & Etrych, T. (2024). Biodegradable Covalently Crosslinked Poly[N-(2-Hydroxypropyl) Methacrylamide] Nanogels: Preparation and Physicochemical Properties. Polymers, 16(2), 263. https://doi.org/10.3390/polym16020263










