Temperature Responsive Copolymers Films of Polyether and Bio-Based Polyamide Loaded with Imidazolium Ionic Liquids for Smart Packaging Applications
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
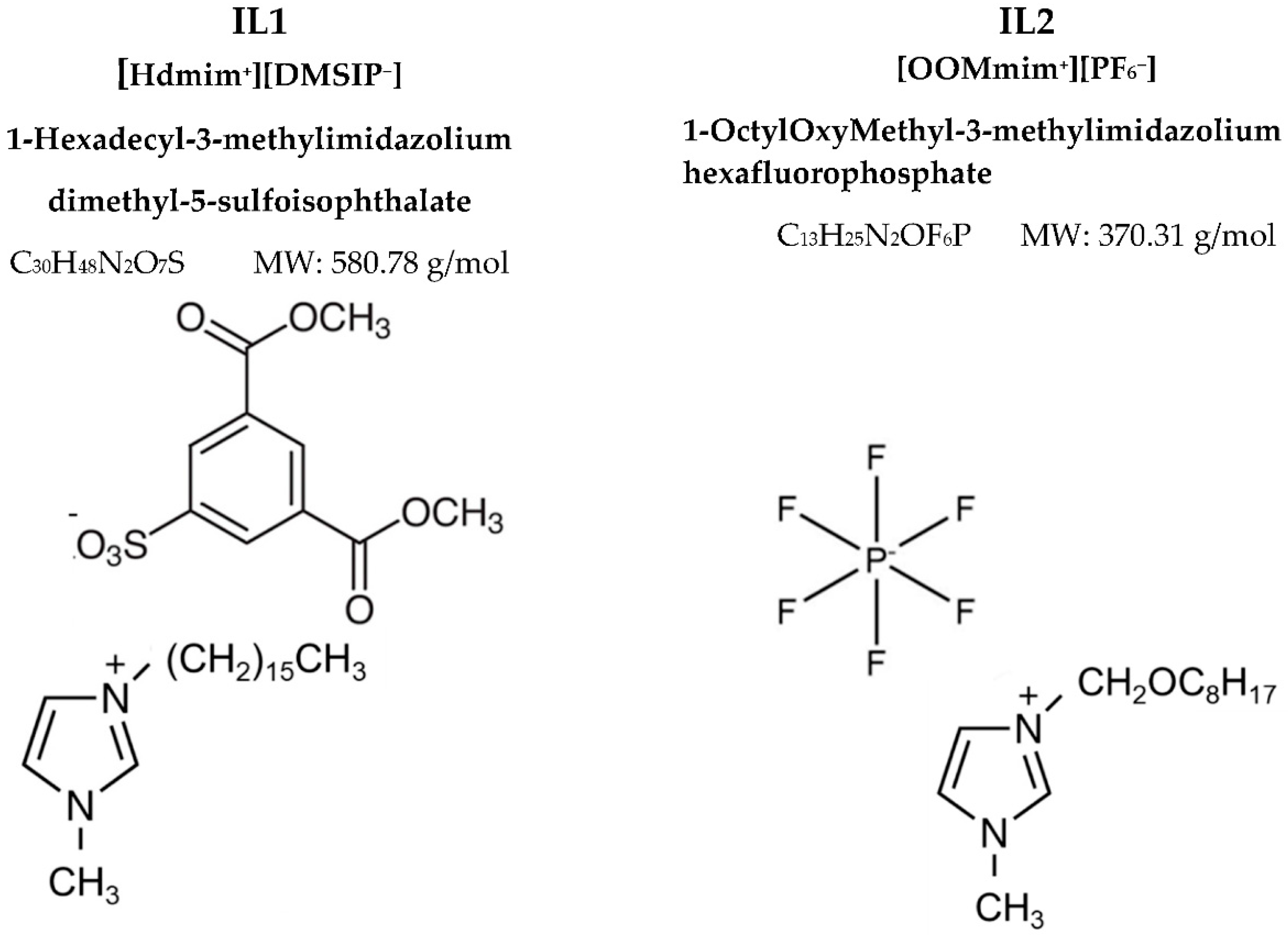
2.2.1. IL Synthesis and Characterization
2.2.2. Membrane Preparation
2.2.3. Membrane Characterization
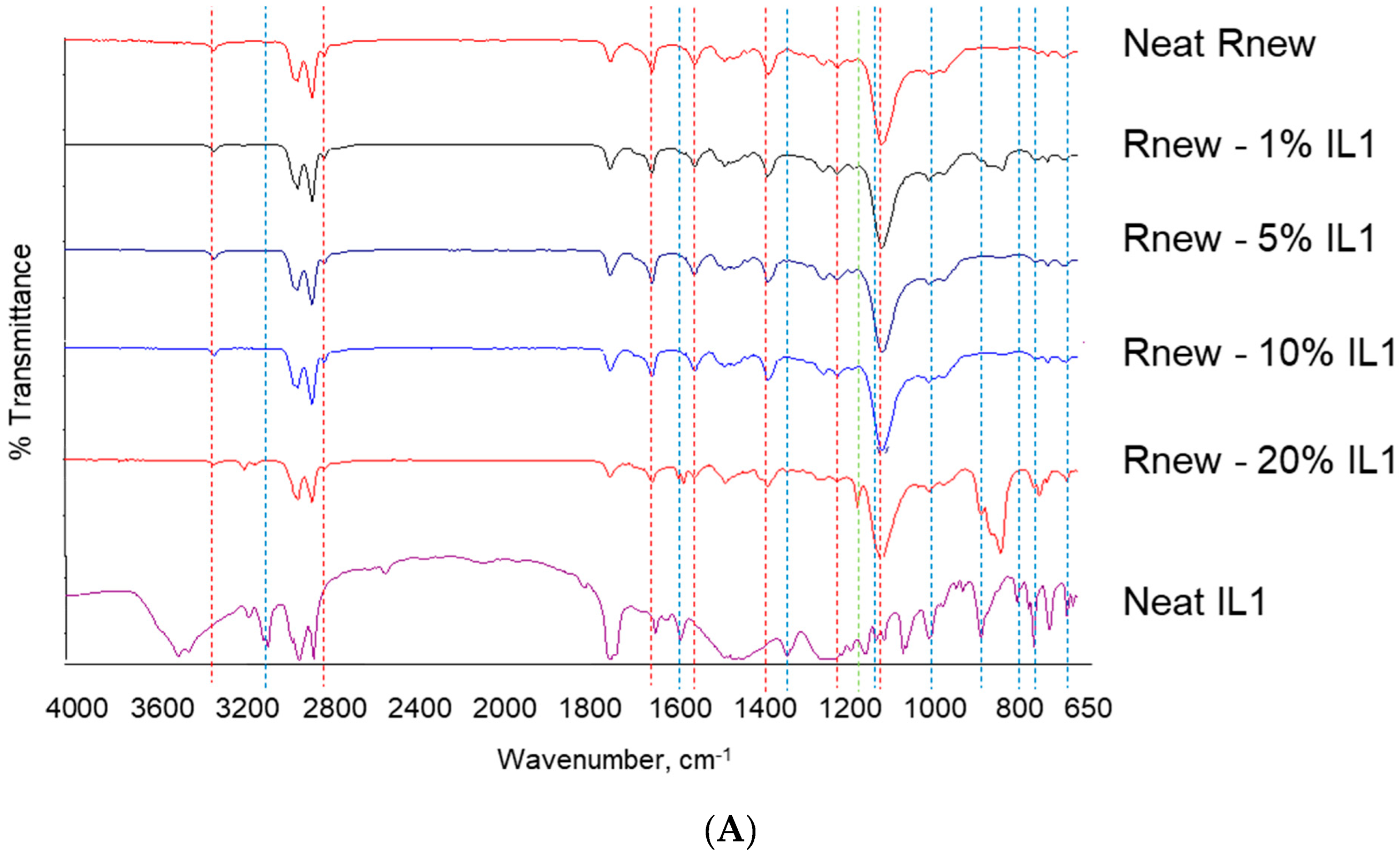
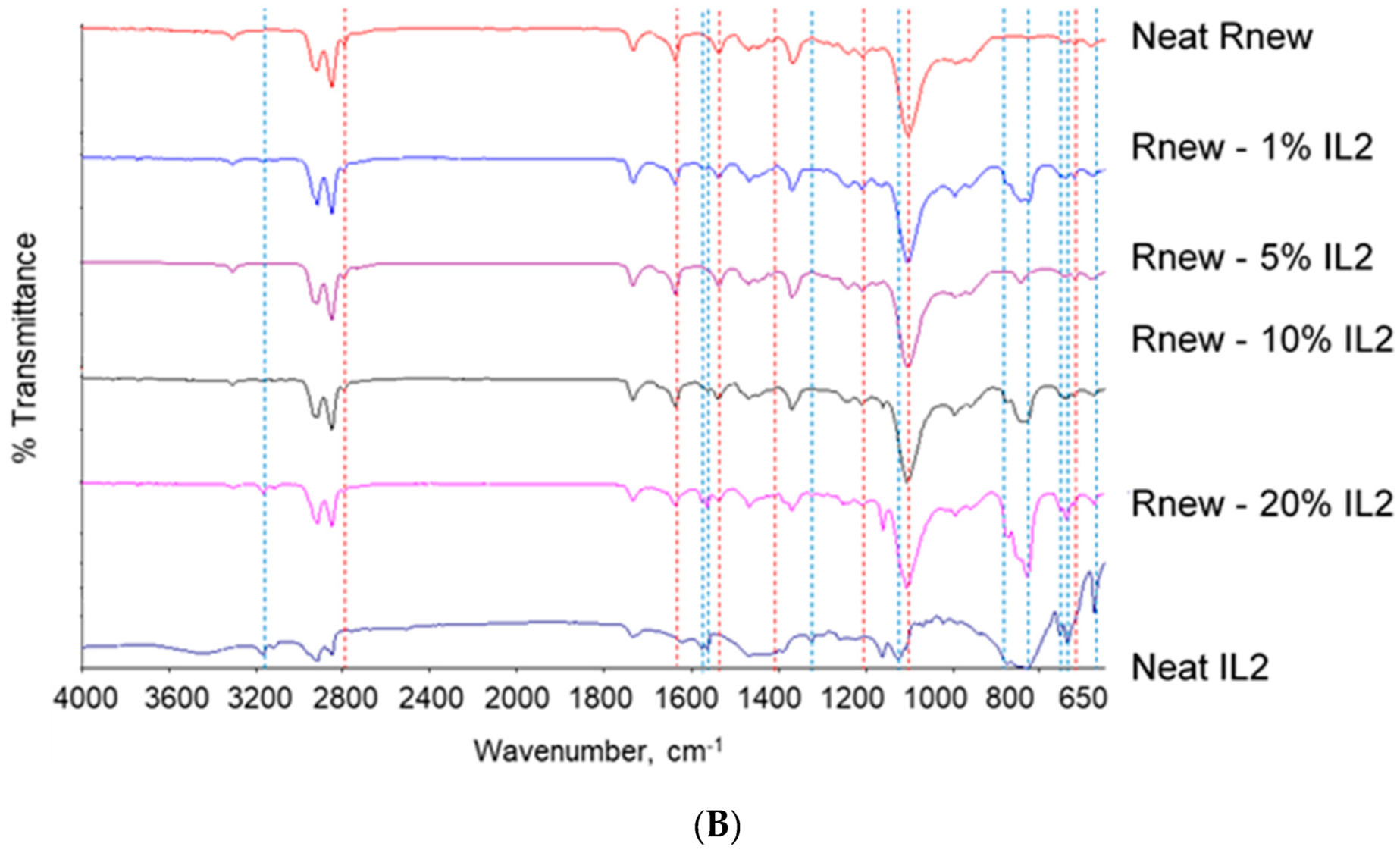
Fourier Transform Infrared (FT-IR)
Thermogravimetric Analysis (TGA)
Differential Scanning Calorimetry (DSC)
2.2.4. Gas Permeation Tests
3. Results
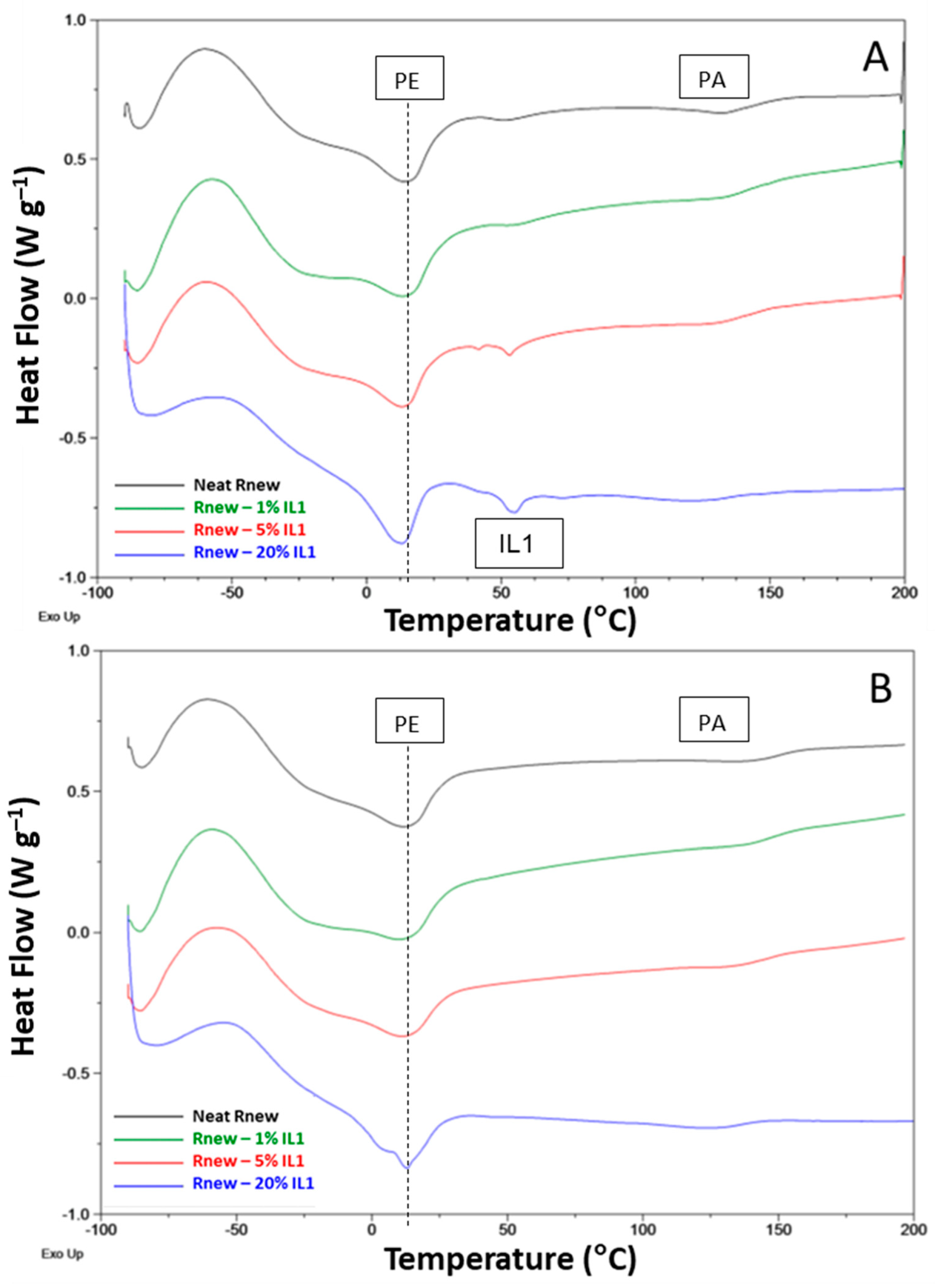
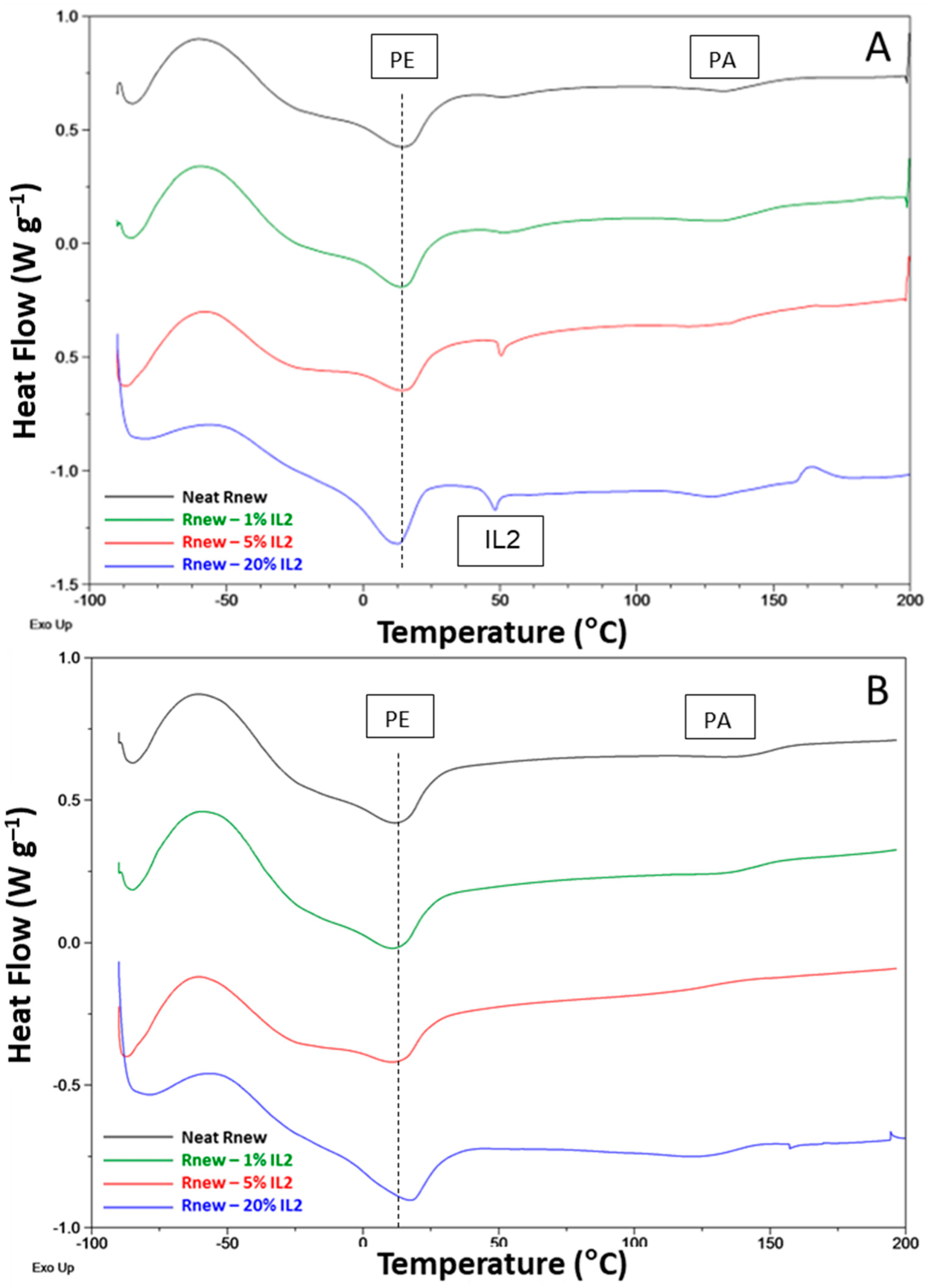
3.1. Different Behaviour of the Two ILs
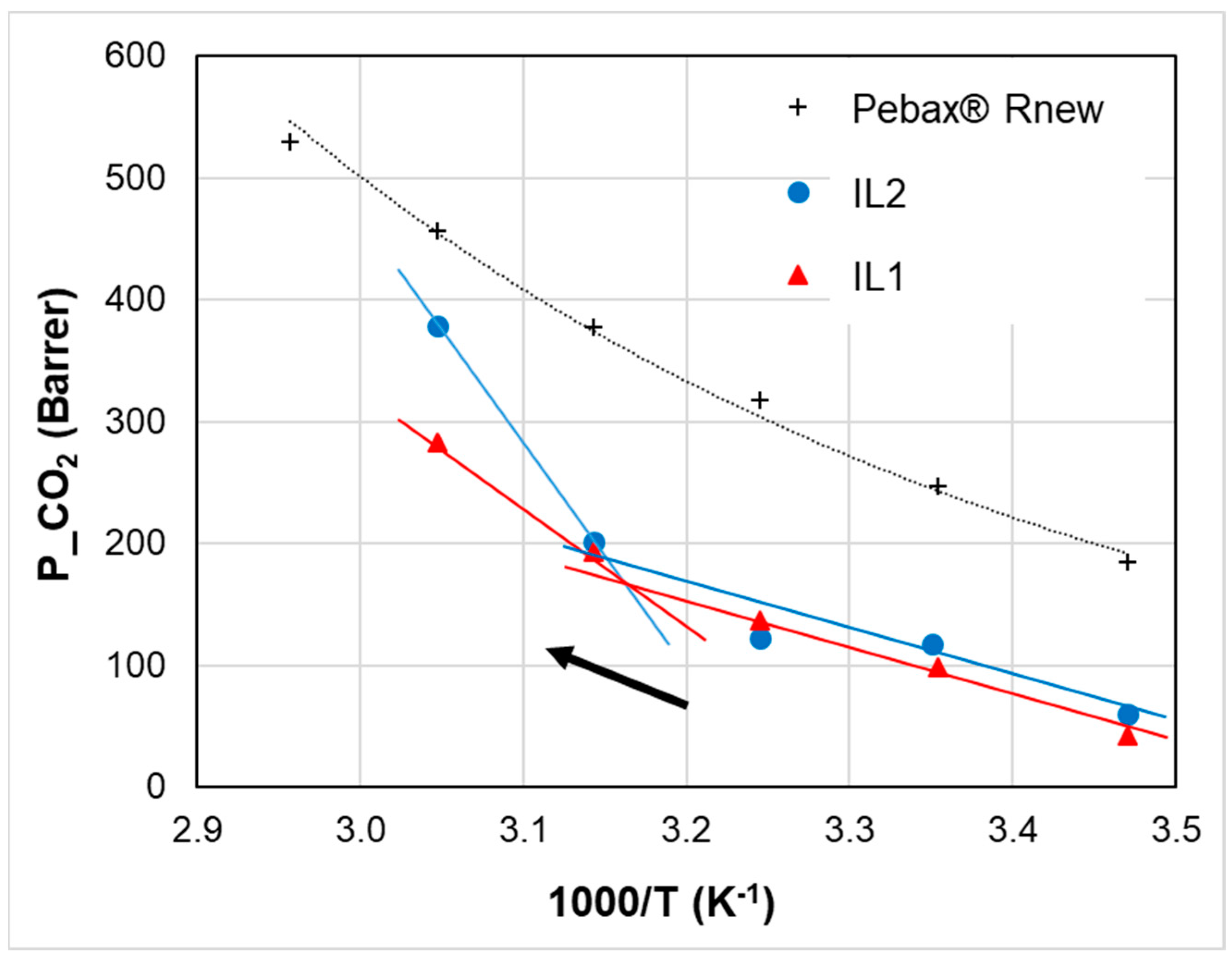
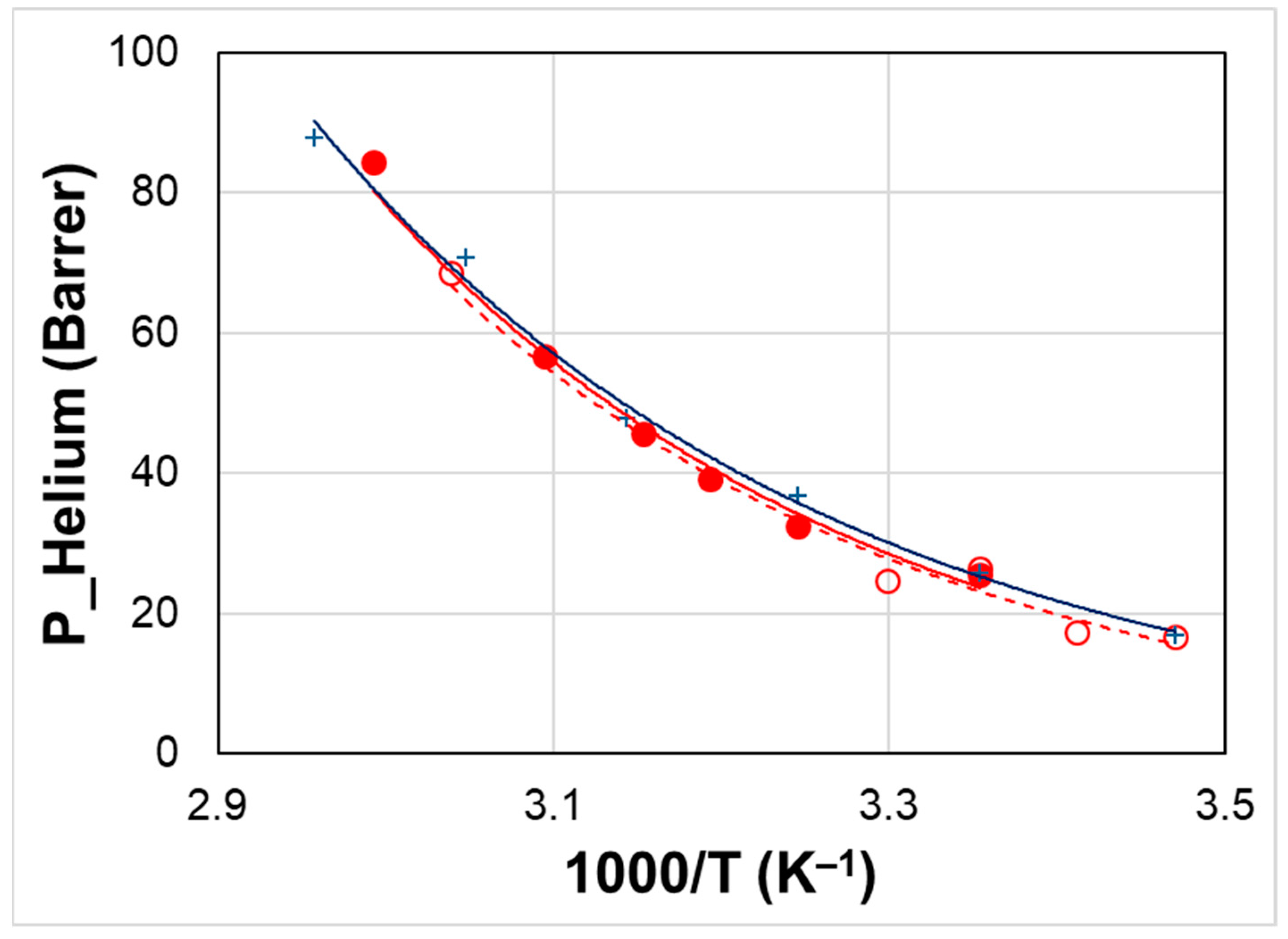
3.2. Temperature Effect
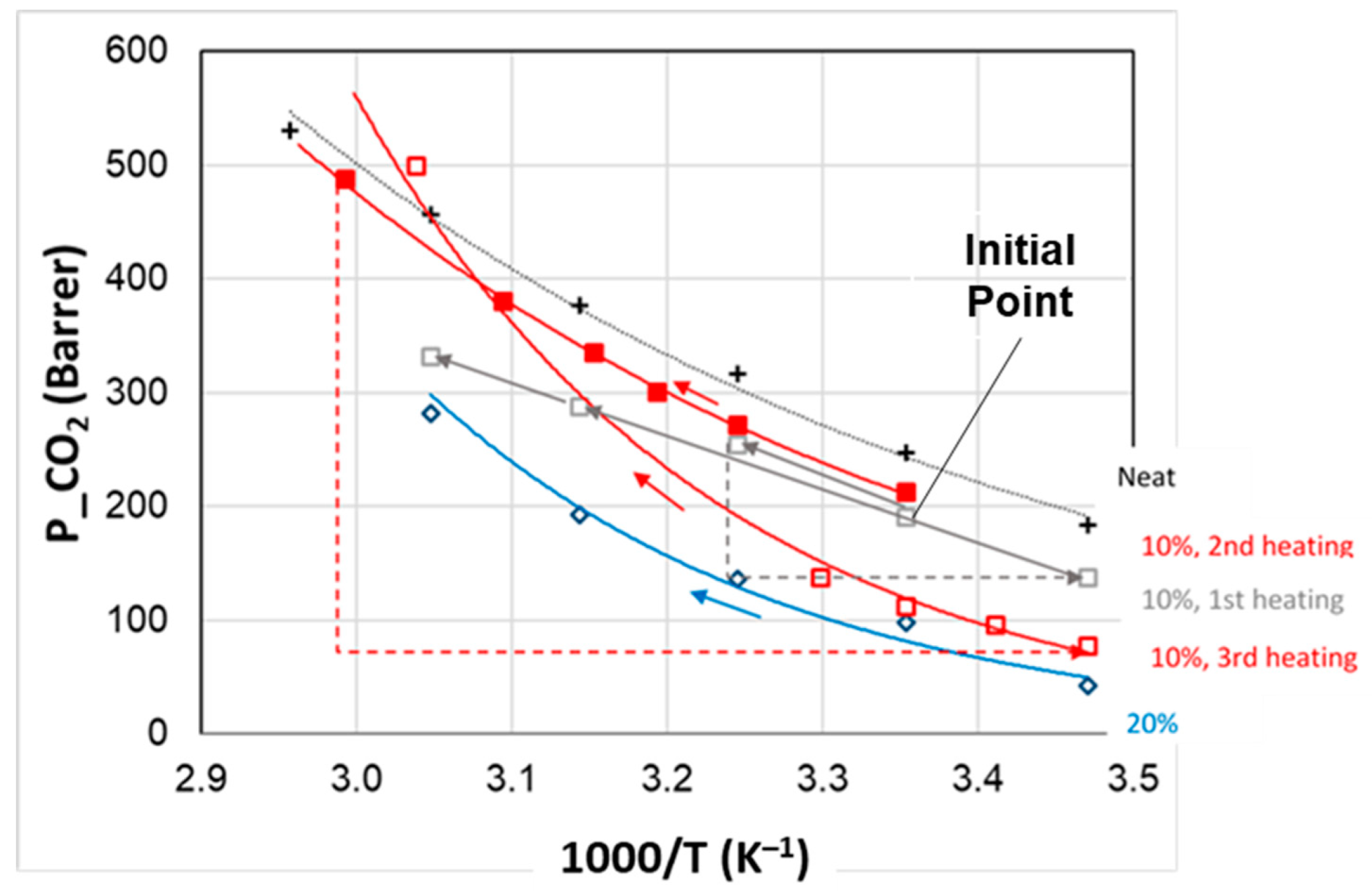
3.3. Thermal Cycling
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adibfar, A.; Hosseini, S.; Eslaminejad, M.B. Smart Polymeric Systems: A Biomedical Viewpoint. In Cell Biology and Translational Medicine, Volume 10; Advances in Experimental Medicine and Biology; Turksen, K., Ed.; Springer: Cham, Switzerland, 2020; Volume 1298. [Google Scholar]
- Hoogenboom, R. Chapter 2—Temperature-responsive polymers: Properties, synthesis and applications. In Smart Polymers and Their Applications; Elsevier: Amsterdam, The Netherlands, 2014; pp. 15–44. [Google Scholar]
- Zhu, J.; Bai, L.; Chen, B.; Fei, W. Thermodynamical properties of phase change materials based on ionic liquids. Chem. Eng. J. 2009, 147, 58–62. [Google Scholar] [CrossRef]
- Todros, S.; Natali, A.N.; Piga, M.; Giffin, G.A.; Pace, G.; Noto, V.D. Interplay between chemical structure and ageing on mechanical and electric relaxations in poly(ether-block-amide)s. Polym. Degrad. Stabil. 2013, 98, 1126–1137. [Google Scholar] [CrossRef]
- Cai, J.; Jiang, J.; Zhou, Z.; Ding, Y.; Zhang, Y.; Wang, F.; Han, C.; Guo, J.; Shao, Q.; Du, H.; et al. Toughening Poly(lactic acid) by Melt Blending with Poly(ether-block-amide) Copolymer. Sci. Adv. Mater. 2017, 9, 1683–1692. [Google Scholar] [CrossRef]
- Bondar, V.; Freeman, B.D.; Pinnau, I. Gas transport properties of poly(ether-b-amide) segmented block copolymers. J. Polym. Sci. Part B Polym. Phys. 2000, 38, 2051–2062. [Google Scholar] [CrossRef]
- Bernardo, P.; Jansen, J.C.; Bazzarelli, F.; Tasselli, F.; Fuoco, A.; Friess, K.; Izák, P.; Jarmarová, V.; Kačírková, M.; Clarizia, G. Gas transport properties of PEBAX®/Room Temperature Ionic Liquid gel membranes. Sep. Purif. Tech. 2012, 97, 73–82. [Google Scholar] [CrossRef]
- Magana, S.; Gain, O.; Gouanvé, F.; Espuche, E. Influence of different alkyl-methylimidazolium tetrafluoroborate ionic liquids on the structure of Pebax® films. Consequences on thermal, mechanical, and water sorption and diffusion properties. J. Polym. Sci. Part B Polym. Phys. 2016, 54, 811–824. [Google Scholar] [CrossRef]
- Estahbanati, E.G.; Omidkhah, M.; Amooghin, A.E. Preparation and characterization of novel Ionic liquid/Pebax membranes for efficient CO2/light gases separation. J. Ind. Eng. Chem. 2017, 51, 77–89. [Google Scholar] [CrossRef]
- Dong, L.-L.; Zhang, C.-F.; Zhang, Y.-Y.; Bai, Y.-X.; Gu, J.; Sun, Y.-P.; Chen, M.-Q. Improving CO2/N2 separation performance using nonionic surfactant Tween containing polymeric gel membranes. RSC Adv. 2015, 5, 4947–4957. [Google Scholar] [CrossRef]
- Bernardo, P.; Clarizia, G. Enhancing Gas Permeation Properties of Pebax® 1657 Membranes via Polysorbate Nonionic Surfactants Doping. Polymers 2020, 12, 253. [Google Scholar] [CrossRef]
- Fu, Q.; Wong, E.H.H.; Kim, J.; Scofield, J.M.P.; Gurr, P.A.; Kentish, S.E.; Qiao, G.G. The effect of soft nanoparticles morphologies on thin film composite membrane performance. J. Mater. Chem. A 2014, 2, 17751–17756. [Google Scholar] [CrossRef]
- Nafisi, V.; Hägg, M.-B. Development of Nanocomposite Membranes Containing Modified Si Nanoparticles in PEBAX-2533 as a Block Copolymer and 6FDA-Durene Diamine as a Glassy Polymer. ACS Appl. Mater. Interfaces 2014, 6, 15643–15652. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Isfahani, A.P.; Muchtar, A.; Sakurai, K.; Shrestha, B.B.; Qin, D.; Yamaguchi, D.; Sivaniah, E.; Ghalei, B. Pebax/ionic liquid modified graphene oxide mixed matrix membranes for enhanced CO2 capture. J. Membr. Sci. 2018, 565, 370–379. [Google Scholar] [CrossRef]
- Li, M.; Zhang, X.; Zeng, S.; Bai, L.; Gao, H.; Deng, J.; Yang, Q.; Zhang, S. Pebax-based composite membranes with high gas transport properties enhanced by ionic liquids for CO2 separation. RSC Adv. 2017, 7, 6422–6431. [Google Scholar] [CrossRef]
- Abdul Halim, K.A.; Kennedy, J.E.; Farrell, J.B. Preparation and Characterisation of Polyamide Homopolymer and Block Copolymer Nanocomposites. Aust. J. Basic Appl. Sci. 2013, 7, 108–115. [Google Scholar]
- Cao, Y.; Mu, T. Comprehensive Investigation on the Thermal Stability of 66 Ionic Liquids by Thermogravimetric Analysis. Ind. Eng. Chem. Res. 2014, 53, 8651–8664. [Google Scholar] [CrossRef]
- Capello, C.; Fischer, U.; Hungerbühler, K. What is a green solvent? A comprehensive framework for the environmental assessment of solvents. Green Chem. 2007, 9, 927–934. [Google Scholar] [CrossRef]
- Otaegi, I.; Aranburu, N.; Guerrica-Echevarría, G. Attaining Toughness and Reduced Electrical Percolation Thresholds in Bio-Based PA410 by Combined Addition of Bio-Based Thermoplastic Elastomers and CNTs. Polymers 2021, 13, 3420. [Google Scholar] [CrossRef] [PubMed]
- Clarizia, G.; Bernardo, P.; Carroccio, S.C.; Ussia, M.; Restuccia, C.; Parafati, L.; Calarco, A.; Zampino, D. Heterogenized Imidazolium-based ionic liquids in Pebax®RNew. Thermal, Gas transport and antimicrobial properties. Polymers 2020, 12, 1419. [Google Scholar] [CrossRef] [PubMed]
- Zampino, D.; Mancuso, M.; Zaccone, R.; Ferreri, T.; Borzacchiello, A.; Zeppetelli, S.; Dattilo, S.; Ussia, M.; Ferreri, L.; Carbone, D.C.; et al. Thermo-mechanical, antimicrobial and biocompatible properties of PVC blends based on imidazolium ionic liquids. Mater. Sci. Eng. C 2021, 122, 111920. [Google Scholar] [CrossRef] [PubMed]
- Clarizia, G.; Bernardo, P.; Gorrasi, G.; Zampino, D.; Carroccio, S.C. Influence of the preparation method and photo-oxidation treatment on the thermal and gas transport properties of dense films based on a poly(ether-block-amide) copolymer. Materials 2018, 11, 1326. [Google Scholar] [CrossRef] [PubMed]
- Crank, J. The Mathematics of Diffusion, 2nd ed.; Clarendon Press: Oxford, UK, 1975. [Google Scholar]
- Wijmans, J.G.; Baker, R.W. The solution-diffusion model: A review. J. Membr. Sci. 1995, 107, 1–21. [Google Scholar] [CrossRef]
- Beiragh, H.H.; Omidkhah, M.; Abedini, R.; Khosravi, T.; Pakseresht, S. Synthesis and characterization of poly (ether-block-amide) mixed matrix membranes incorporated by nanoporous ZSM-5 particles for CO2/CH4 separation. Asia-Pac. J. Chem. Eng. 2016, 11, 522–532. [Google Scholar] [CrossRef]
- Nayebi, R.; Tarigh, G.D.; Shemirani, F. Porous ionic liquid polymer: A reusable adsorbent with broad operating pH range for speciation of nitrate and nitrite. Sci. Rep. 2019, 9, 11130. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, T.; Ranga Rao, G. Synthesis and characterization of hybrid molecular material prepared by ionic liquid and silicotungstic acid. Mater. Chem. Phys. 2008, 112, 853–857. [Google Scholar] [CrossRef]
- Zheng, D.; Dong, L.; Huang, W.; Wu, X.; Nie, N. A review of imidazolium ionic liquids research and development towards working pair of absorption cycle. Renew. Sustain. Energy Rev. 2014, 37, 47–68. [Google Scholar] [CrossRef]
- Zoppi, R.A.; de Castro, C.R.; Yoshida, I.V.P.; Nunes, S.P. Hybrids of SiO2 and poly(amide 6-b-ethylene oxide). Polymer 1997, 38, 5705–5712. [Google Scholar] [CrossRef]
- Villanueva, M.; Coronas, A.; García, J.; Salgado, J. Thermal Stability of Ionic Liquids for Their Application as New Absorbents. Ind. Eng. Chem. Res. 2013, 52, 15718–15727. [Google Scholar] [CrossRef]
- Shalu, S.K.; Chaurasia, R.K.; Singh, S. Chandra, thermal stability, complexing behavior, and ionic transport of polymeric gel membranes based on polymer PVdF-HFP and ionic liquid, [BMIM][BF4]. J. Phys. Chem. B 2013, 117, 897–906. [Google Scholar] [CrossRef]
- Ahmadi, A.; Haghbakhsh, R.; Raeissi, S.; Hemmati, V. A simple group contribution correlation for the prediction of ionic liquid heat capacities at different temperatures. Fluid Phase Equilibria 2015, 403, 95–103. [Google Scholar] [CrossRef]
- Farahani, N.; Gharagheizi, F.; Mirkhani, S.A.; Tumba, K. A simple correlation for prediction of heat capacities of ionic liquids. Fluid Phase Equilibria 2013, 337, 73–82. [Google Scholar] [CrossRef]
- Ioelovich, M. Relationships between Cohesive Energy, Glass Transition Temperature and Melting Point for Various Polymers. Sci. Isr.—Technol. Advant. 2021, 23, 139–146. [Google Scholar]
- Bernardo, P.; Clarizia, G. A review of the recent progress in the development of Nanocomposites based on poly(ether-block-amide) copolymers as membranes for CO2 separation. Polymers 2022, 14, 10. [Google Scholar]
- Barbi, V.; Funari, S.S.; Gehrke, R.; Scharnagl, N.; Stribeck, N. SAXS and the Gas Transport in Polyether-block-polyamide Copolymer Membranes. Macromolecules 2003, 36, 749–758. [Google Scholar] [CrossRef]
- Azizi, N.; Mohammadi, T.; Behbahani, R.M. Comparison of permeability performance of PEBAX-1074/TiO2, PEBAX-1074/SiO2 and PEBAX-1074/Al2O3 nanocomposite membranes for CO2/CH4 separation. Chem. Eng. Res. Des. 2017, 117, 177–189. [Google Scholar] [CrossRef]
- Holloczki, O.; Kelemen, Z.; Kçnczçl, L.; Szieberth, D.; Nyulaszi, L.; Stark, A.; Kirchner, B. Significant Cation Effects in Carbon Dioxide–Ionic Liquid Systems. Chem. Phys. Chem. 2013, 14, 315–320. [Google Scholar] [CrossRef]
- Xiong, Y.-B.; Wang, H.; Wang, Y.-J.; Wang, R.-M. Novel imidazolium-based poly(ionic liquid)s: Preparation, characterization, and absorption of CO2. Polym. Adv. Technol. 2012, 23, 835–840. [Google Scholar] [CrossRef]
- Cadena, C.; Anthony, J.L.; Shah, J.K.; Morrow, T.I.; Brennecke, J.F.; Maginn, E.J. Why is CO2 So Soluble in Imidazolium-Based Ionic Liquids? J. Am. Chem. Soc. 2004, 126, 5300–5308. [Google Scholar] [CrossRef]
- Pilato, L.A.; Litz, L.M.; Hargitay, B.; Osoborne, R.C.; Farnham, A.G.; Kawakami, J.H.; Fritze, P.; McGrath, J. Polymers for permselective membrane gas separations. Polym Prepr. 1975, 16, 41–46. [Google Scholar]
- Wolf, C.; Angellier-Coussy, H.; Gontard, N.; Doghieri, F.; Guillard, V. How the shape of fillers affects the barrier properties of polymer/non-porous particles nanocomposites: A review. J. Membr. Sci. 2018, 556, 393–418. [Google Scholar] [CrossRef]
- Elamin, K.; Shojaatalhosseini, M.; Danyliv, O.; Martinelli, A.; Swenson, J. Conduction mechanism in polymeric membranes based on PEO or PVdF-HFP and containing a piperidinium ionic liquid. Electrochim. Acta 2019, 299, 979–986. [Google Scholar] [CrossRef]
- Supasitmongkola, S.; Styring, P. High CO2 solubility in ionic liquids and a tetraalkylammonium-based poly(ionic liquid). Energy Environ. Sci. 2010, 3, 1961–1972. [Google Scholar] [CrossRef]
- Castiglione, F.; Simonutti, R.; Mauri, M.; Mele, A. Cage-Like Local Structure of Ionic Liquids Revealed by a 129Xe Chemical Shift. J. Phys. Chem. Lett. 2013, 4, 1608–1612. [Google Scholar] [CrossRef] [PubMed]
- Saielli, G.; Bagno, A.; Castiglione, F.; Simonutti, R.; Mauri, M.; Mele, A. Understanding Cage Effects in Imidazolium Ionic Liquids by 129Xe NMR: MD Simulations and Relativistic DFT Calculations. J. Phys. Chem. B 2014, 118, 13963–13968. [Google Scholar] [CrossRef]
- Klähn, M.; Seduraman, A. What Determines CO2 Solubility in Ionic Liquids? A Molecular Simulation Study. J. Phys. Chem. B 2015, 119, 10066–10078. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.A.A.; Neves, C.M.S.S.; Freire, M.G.; Russina, O.; Triolo, A.; Coutinho, J.A.P.; Santos, L.M.N.B.F. Alkylimidazolium Based Ionic Liquids: Impact of Cation Symmetry on Their Nanoscale Structural Organization. J. Phys. Chem. B 2013, 117, 10889–10897. [Google Scholar] [CrossRef] [PubMed]
- Barrer, R.M.; Rideal, E.K. Permeation, Diffusion and Solution of Gases in Organic Polymers. Trans. Faraday Soc. 1939, 35, 628–656. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Campusano, R.A.; Rojas, R.E. Glass transition temperature of Ionic Liquids using molecular descriptors and artificial neural networks. Comptes Rendus Chim. 2017, 20, 573–584. [Google Scholar] [CrossRef]
- Harris, M.A.; Kinsey, T.; Wagle, D.V.; Baker, G.A.; Sangoro, J. Evidence of a liquid-liquid transition in a glass-forming ionic liquid. Proc. Natl. Acad. Sci. USA 2021, 118, e2020878118. [Google Scholar] [CrossRef]

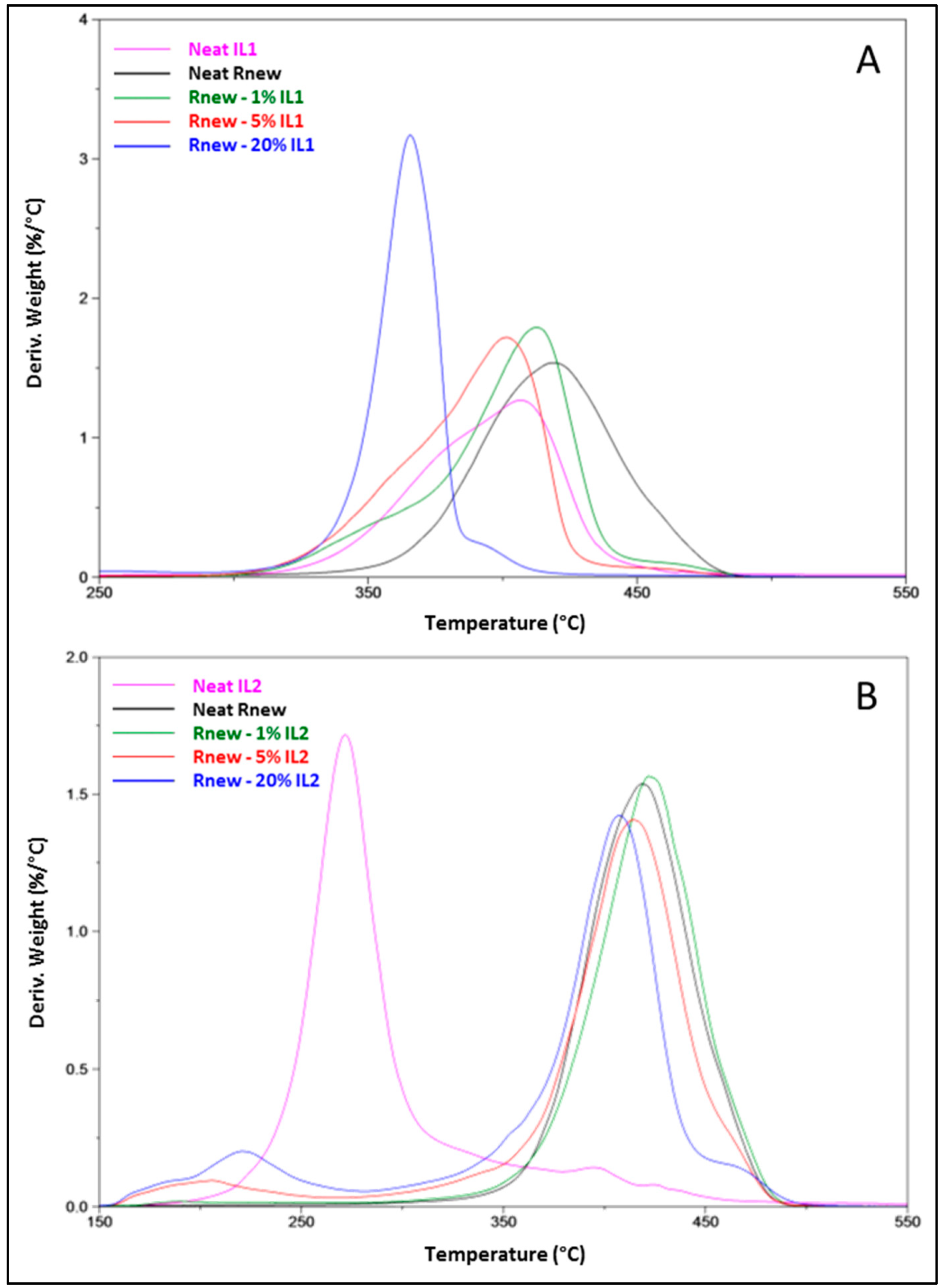





| Sample | TΔm = 5% (°C) a | TΔm = 50% (°C) b | Td1 (°C) c | % R d |
|---|---|---|---|---|
| Neat Rnew | 366 | 416 | 418 | 0.0 |
| Rnew–1% IL1 | 337 | 403 | 413 | 0.2 |
| Rnew–5% IL1 | 333 | 389 | 402 | 0.1 |
| Rnew–20% IL1 | 300 | 364 | 366 | 3.6 |
| Rnew–1% IL2 | 354 | 420 | 422 | 0.2 |
| Rnew–5% IL2 | 222 | 409 | 415 | 0.0 |
| Rnew–20% IL2 | 211 | 398 | 408 | 1.3 |
| Sample | Permeability (Barrer) | Selectivity (–) | ||||||
|---|---|---|---|---|---|---|---|---|
| CO2 | O2 | N2 | He | H2 | CO2/N2 | H2/N2 | O2/N2 | |
| Neat Rnew | 247 | 23.6 | 8.9 | 25.7 | 45.4 | 27.9 | 5.12 | 2.66 |
| Rnew–1% IL2 | 232 | 22.4 | 8.6 | 25.7 | - | 27.0 | - | 2.60 |
| Rnew–5% IL2 | 226 | 21.6 | 8.1 | 23.8 | 38.1 | 28.0 | 4.73 | 2.61 |
| Rnew–10% IL2 | 139 | 13.3 | 5.2 | 18.4 | 25.9 | 27.0 | 5.01 | 2.57 |
| Rnew–20% IL2 | 117 | 11.5 | 4.1 | 17.4 | 25.1 | 28.3 | 6.14 | 2.77 |
| Rnew–1% IL1 | 217 | 21.4 | 8.5 | 23.7 | - | 25.6 | - | 2.53 |
| Rnew–5% IL1 | 195 | 19.2 | 7.2 | 24.1 | 40.3 | 27.2 | 5.63 | 2.68 |
| Rnew–10% IL1 | 191 | 18.4 | 6.86 | 23.4 | 36.2 | 27.8 | 5.28 | 2.68 |
| Rnew–20% IL1 | 98.4 | 10.4 | 4.4 | 13.4 | 23.2 | 22.3 | 5.25 | 2.35 |
| Grade | Composition | PE | PA | Permeability (Barrer) | Ref. | |
|---|---|---|---|---|---|---|
| wt% | wt% | CO2 | T | |||
| Neat Rnew | PTMO/PA11 | 79–83 | 17–21 | 247 | 25 °C | [This work] |
| 2533 | PTMO/PA12 | 80 | 20 | 276 a 241 b | 30 °C | [36] |
| 3533 | PTMO/PA12 | 70 | 30 | 204 a 256 b | 30 °C | [36] |
| 1657 | PEO/PA12 | 60 | 40 | 66.5 | 25 °C | [11] |
| 4011 | PEO/PA6 | 57 | 43 | 109 a 71.4 b | 30 °C | [36] |
| 1074 | PEO/PA12 | 55 | 45 | 94 a 106 b | 30 °C | [36] |
| 110.7 | 25 °C | [37] | ||||
| 4033 | PTMO/PA12 | 53 | 47 | 81 a 95 b | 30 °C | [36] |
| Sample | Activation Energy for Permeability (kJ mol−1) | ||||
|---|---|---|---|---|---|
| CO2 | O2 | N2 | H2 | ||
| Neat Rnew | Ep | 17.0 | 28.0 | 32.7 | 26.7 |
| Rnew–10% IL2 | Ep1 | 22.8 | 34.6 | 42.3 | 27.3 |
| Ep2 | 38.0 | 50.4 | 53.6 | 41.4 | |
| Rnew–20% IL2 | Ep1 | 26.0 | 37.2 | 42.3 | 34.1 |
| Ep2 | 47.6 | 55.3 | 62.2 | 45.3 | |
| Rnew–10% IL1 | Ep1 | 18.8 | 29.0 | 37.1 | 19.1 |
| Ep2 | 19.9 | 33.9 | 40.6 | 30.2 | |
| Rnew–20% IL1 | Ep1 | 43.3 | 49.7 | 59.6 | 35.6 |
| Ep2 | 30.5 | 42.9 | 46.6 | 46.3 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zampino, D.C.; Clarizia, G.; Bernardo, P. Temperature Responsive Copolymers Films of Polyether and Bio-Based Polyamide Loaded with Imidazolium Ionic Liquids for Smart Packaging Applications. Polymers 2023, 15, 1147. https://doi.org/10.3390/polym15051147
Zampino DC, Clarizia G, Bernardo P. Temperature Responsive Copolymers Films of Polyether and Bio-Based Polyamide Loaded with Imidazolium Ionic Liquids for Smart Packaging Applications. Polymers. 2023; 15(5):1147. https://doi.org/10.3390/polym15051147
Chicago/Turabian StyleZampino, Daniela C., Gabriele Clarizia, and Paola Bernardo. 2023. "Temperature Responsive Copolymers Films of Polyether and Bio-Based Polyamide Loaded with Imidazolium Ionic Liquids for Smart Packaging Applications" Polymers 15, no. 5: 1147. https://doi.org/10.3390/polym15051147
APA StyleZampino, D. C., Clarizia, G., & Bernardo, P. (2023). Temperature Responsive Copolymers Films of Polyether and Bio-Based Polyamide Loaded with Imidazolium Ionic Liquids for Smart Packaging Applications. Polymers, 15(5), 1147. https://doi.org/10.3390/polym15051147








