Coating Materials to Increase the Stability of Liposomes
Abstract
1. Introduction
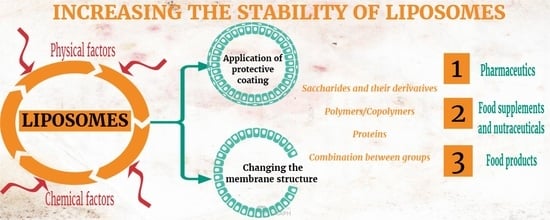
2. Physical and Chemical Factors Influencing Liposome Stability after Being Obtained
2.1. Physical Factors
2.2. Chemical Factors
3. Coating Materials for Liposomes with Applications in Drug Delivery
4. Coating Materials for Liposomes with Applications in the Food Industry
5. The Influence of Polymer Coatings on the Absorption of Liposomes
6. Patent Applications and Patents on Coated Liposomes
7. Liposome Commercial Products Available on the Market
8. Conclusions
9. Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jacob, S.; Nair, A.B.; Shah, J.; Gupta, S.; Boddu, S.H.; Sreeharsha, N. Lipid Nanoparticles as a Promising Drug Delivery Carrier for Topical Ocular Therapy—An Overview on Recent Advances. Pharmaceutics 2022, 14, 533. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Lee, J.; Jo, Y.J.; Choi, M.J. Application of Liposome Encapsulating Lactobacillus curvatus Extract in Cosmetic Emulsion Lotion. Materials 2021, 14, 7571. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Yang, J.H.; Kim, K.S.; Park, G.D. Effect of Liposome Storage of Cyanocobalamin on Its Degradation by Ascorbic Acid. Food Suppl. Biomater. Health 2021, 1, e7. [Google Scholar] [CrossRef]
- Nguyen, T.X.; Huang, L.; Liu, L.; Abdalla, A.M.E.; Gauthier, M.; Yang, G. Chitosan-coated nano-liposomes for the oral delivery of berberine hydrochloride. J. Mater. Chem. B 2014, 2, 7149–7159. [Google Scholar] [CrossRef] [PubMed]
- Marsanasco, M.; Del Valle Alonso, S. Why produce food-bioactive compounds to generate functional grade foods? In Functional Foods-Phytochemicals and Health Promoting Potential; Muhammad, S.A., Muhammad, H.A., Eds.; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Van Hoogevest, P.; Wendel, A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid. Sci. Technol. 2014, 116, 1088–1107. [Google Scholar] [CrossRef]
- Nakhaei, P.; Margiana, R.; Bokov, D.O.; Abdelbasset, W.K.; Kouhbanani, M.A.J.; Varma, R.S. Liposomes: Structure, biomedical applications, and stability parameters with emphasis on cholesterol. Front. Bioeng. Biotechnol. 2021, 9, 748. [Google Scholar] [CrossRef]
- Karunaratne, D.N.; Pamunuwa, G.K.; Nicholas, I.H.; Ariyarathna, I.R. Science of Spices and Culinary Herbs-Latest Laboratory. In Pre-Clinical, and Clinical Studies; Atta-ur-Rahman, Choudhary, M.I., Yousuf, S., Eds.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2019; Volume 1, pp. 104–147. [Google Scholar] [CrossRef]
- Subramanian, P. Lipid-based nanocarrier system for the effective delivery of nutraceuticals. Molecules 2021, 26, 5510. [Google Scholar] [CrossRef]
- Arpita, S.; Kumar, S.V. Liposomes–A Review. Int. J. Indig. Herbs Drugs 2020, 5, 1–6. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Kenwat, R.; Kurmi, B.D.; Sahu, M.K. Solid lipid nanoparticles: A review on recent perspectives and patents. Expert. Opin. Ther. Pat. 2020, 30, 179–194. [Google Scholar] [CrossRef]
- Ajeeshkumar, K.K.; Aneesh, P.A.; Raju, N.; Suseela, M.; Ravishankar, C.N.; Benjakul, S. Advancements in liposome technology: Preparation techniques and applications in food, functional foods, and bioactive delivery: A review. Compr. Rev. Food. Sci. Food Saf. 2021, 20, 1280–1306. [Google Scholar] [CrossRef]
- Lazuardi, M.; Suharjomo, S.; Chien, C.H.; He, J.L.; Sugihartuti, R.; Maslachah, L. Encapsulation of progesterone-like compounds in 10% liposome increases their concentration in rats administered an injectable dosage form of these compounds. Kafkas Univ. Veter-Fak. Derg. 2022, 28, 27–34. [Google Scholar] [CrossRef]
- de Freitas, C.F.; Calori, I.R.; Tessaro, A.L.; Caetano, W.; Hioka, N. Rapid formation of small unilamellar vesicles (suv) through low-frequency sonication: An innovative approach. Colloid. Surf. B 2019, 181, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Subramani, T.; Ganapathyswamy, H. An overview of liposomal nano-encapsulation techniques and its applications in food and nutraceutical. J. Food. Sci. Technol. 2020, 57, 3545–3555. [Google Scholar] [CrossRef] [PubMed]
- Sheoran, R.; Khokra, S.L.; Chawla, V.; Dureja, H. Recent patents, formulation techniques, classification and characterization of liposomes. Recent Pat. Nanotechnol. 2019, 13, 17–27. [Google Scholar] [CrossRef]
- Trucillo, P.; Reverchon, E. Production of PEG-coated liposomes using a continuous supercritical assisted process. J. Supercrit. Fluids 2021, 167, 105048. [Google Scholar] [CrossRef]
- Rashidinejad, A.; Jafari, S.M. Nanoencapsulation of bioactive food ingredients. In Handbook of Food Nanotechnology; Jafari, S.M., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 279–344. [Google Scholar] [CrossRef]
- Saudagar, R.B.; Saokar, S. Anti-inflammatory natural compounds from herbal and marine origin. J. Drug Deliv. Ther. 2019, 9, 669–672. Available online: https://jddtonline.info/index.php/jddt/article/view/2906 (accessed on 15 April 2022).
- Andra, V.V.S.N.L.; Bhatraju, L.V.K.P.; Ruddaraju, L.K. A comprehensive review on novel liposomal methodologies, commercial formulations, clinical trials and patents. BioNanoScience 2022, 12, 274–291. [Google Scholar] [CrossRef]
- Pamunuwa, G.K.; Karunaratne, D. Liposomal Delivery of Plant Bioactives Enhances Potency in Food Systems: A Review. J. Food Qual. 2022, 2022, 1–11. [Google Scholar] [CrossRef]
- Emami, S.; Azadmard-Damirchi, S.; Peighambardoust, S.H.; Valizadeh, H.; Hesari, J. Liposomes as carrier vehicles for functional compounds in food sector. J. Exp. Nanosci. 2016, 11, 737–759. [Google Scholar] [CrossRef]
- Mehnath, S.; Das, A.K.; Verma, S.K.; Jeyaraj, M. Biosynthesized/green-synthesized nanomaterials as potential vehicles for delivery of antibiotics/drugs. In Comprehensive Analytical Chemistry; Sandeep, K.V., Ashok, K.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; Volume 94, pp. 363–432. [Google Scholar] [CrossRef]
- Liu, P.; Chen, G.; Zhang, J. A review of liposomes as a drug delivery system: Current status of approved products, regulatory environments, and future perspectives. Molecules 2022, 27, 1372. [Google Scholar] [CrossRef]
- Pentak, D.; Ploch-Jankowska, A.; Zięba, A.; Kozik, V. The Advances and Challenges of Liposome-Assisted Drug Release in the Presence of Serum Albumin Molecules: The Influence of Surrounding pH. Materials 2022, 15, 1586. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Ahsan, H. Lipid-based formulations in cosmeceuticals and biopharmaceuticals. Biomed. Dermatol. 2020, 4, 1–10. [Google Scholar] [CrossRef]
- Zarrabi, A.; Abadi, M.A.; Khorasani, S.; Mohammadabadi, M.R.; Jamshidi, A.; Torkaman, S.; Taghavi, E.; Mozafari, M.R.; Rasti, B. Nanoliposomes and tocosomes as multifunctional nanocarriers for the encapsulation of nutraceutical and dietary molecules. Molecules 2020, 25, 638. [Google Scholar] [CrossRef]
- Padilla, C.E.; Villanueva, F.J.E. Encapsulation of food active ingredients in liposomes. J. Nutr. Health. Food. Eng. 2018, 8, 238–239. [Google Scholar] [CrossRef]
- Liu, W.; Hou, Y.; Jin, Y.; Wang, Y.; Xu, X.; Han, J. Research progress on liposomes: Application in food, digestion behavior and absorption mechanism. Trends Food Sci. Technol. 2020, 104, 177–189. [Google Scholar] [CrossRef]
- Gibis, M.; Zeeb, B.; Weiss, J. Formation, characterization, and stability of encapsulated hibiscus extract in multilayered liposomes. Food Hydrocoll. 2014, 38, 28–39. [Google Scholar] [CrossRef]
- Lee, D.E.; Lew, M.G.; Woodbury, D.J. Vesicle fusion to planar membranes is enhanced by cholesterol and low temperature. Chem. Phys. Lipids 2013, 166, 45–54. [Google Scholar] [CrossRef]
- Sepúlveda, C.T.; Alemán, A.; Zapata, J.E.; Montero, M.P.; Gómez-Guillén, M.C. Characterization and storage stability of spray dried soy-rapeseed lecithin/trehalose liposomes loaded with a tilapia viscera hydrolysate. Innov. Food Sci. Emerg. Technol. 2021, 71, 102708. [Google Scholar] [CrossRef]
- Boafo, G.F.; Magar, K.T.; Ekpo, M.D.; Qian, W.; Tan, S.; Chen, C. The Role of Cryoprotective Agents in Liposome Stabilization and Preservation. Int. J. Mol. Sci. 2022, 23, 12487. [Google Scholar] [CrossRef]
- Liu, W.; Ye, A.; Han, F.; Han, J. Advances and challenges in liposome digestion: Surface interaction, biological fate, and GIT modeling. Adv. Colloid Interface Sci. 2019, 263, 52–67. [Google Scholar] [CrossRef]
- Sydykov, B.; Oldenhof, H.; Sieme, H.; Wolkers, W.F. Storage stability of liposomes stored at elevated subzero temperatures in DMSO/sucrose mixtures. PLoS ONE 2018, 13, e0199867. [Google Scholar] [CrossRef] [PubMed]
- Ioele, G.; Grande, F.; De Luca, M.; Occhiuzzi, M.A.; Garofalo, A.; Ragno, G. Photodegradation of anti-inflammatory drugs: Stability tests and lipid nanocarriers for their photoprotection. Molecules 2021, 26, 5989. [Google Scholar] [CrossRef] [PubMed]
- Mohan, A.; Rajendran, S.R.; He, Q.S.; Bazinet, L.; Udenigwe, C.C. Encapsulation of food protein hydrolysates and peptides: A review. RSC Adv. 2015, 5, 79270–79278. [Google Scholar] [CrossRef]
- Petrović, S.; Tačić, A.; Savić, S.; Nikolić, V.; Nikolić, L.; Savić, S. Sulfanilamide in solution and liposome vesicles; in vitro release and UV-stability studies. Saudi Pharm. J. 2017, 25, 1194–1200. [Google Scholar] [CrossRef]
- Huang, J.; Wang, Q.; Chu, L.; Xia, Q. Liposome-chitosan hydrogel bead delivery system for the encapsulation of linseed oil and quercetin: Preparation and in vitro characterization studies. LWT Food Sci. Technol. 2020, 117, 108615. [Google Scholar] [CrossRef]
- Lin, X.; Li, B.; Wen, J.; Wu, J.; Tang, D.; Yu, Y.; Xu, Y.; Xu, B. Storage Stability and In Vitro Bioaccessibility of Liposomal Betacyanins from Red Pitaya (Hylocereus polyrhizus). Molecules 2022, 27, 1193. [Google Scholar] [CrossRef]
- Pentak, D. In vitro spectroscopic study of piperine-encapsulated nanosize liposomes. Eur. Biophys. J. 2016, 45, 175–186. [Google Scholar] [CrossRef]
- Ghareaghajlou, N.; Hallaj-Nezhadi, S.; Ghasempour, Z. Nano-liposomal system based on lyophilization of monophase solution technique for encapsulating anthocyanin-rich extract from red cabbage. Dye. Pigment. 2022, 202, 110263. [Google Scholar] [CrossRef]
- Guldike, B.; Gibi, M.; Boyacioglu, D.; Capanoglu, E.; Weiss, J. Physical and chemical stability of anthocyanin-rich black carrot extract-loaded liposomes during storage. Food Res. Int. 2018, 108, 491–497. [Google Scholar] [CrossRef]
- Xia, H.; Tang, Y.; Huang, R.; Liang, J.; Ma, S.; Chen, D.; Feng, Y.; Lei, Y.; Zhang, Q.; Yang, Y.; et al. Nanoliposome Use to Improve the Stability of Phenylethyl Resorcinol and Serve as a Skin Penetration Enhancer for Skin Whitening. Coatings 2022, 12, 362. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, K.; Liu, Q.; Liu, X.; Mou, B.; Lai, O.M.; Tan, C.P.; Cheong, L.Z. Selective antibacterial activities and storage stability of curcumin-loaded nanoliposomes prepared from bovine milk phospholipid and cholesterol. Food Chem. 2022, 367, 130700. [Google Scholar] [CrossRef] [PubMed]
- Almurshedi, A.S.; Radwan, M.; Omar, S.; Alaiya, A.A.; Badran, M.M.; Elsaghire, H.; Saleem, Y.I.; Hutcheon, G.A. A novel pH-sensitive liposome to trigger delivery of afatinib to cancer cells: Impact on lung cancer therapy. J. Mol. Liq. 2018, 259, 154–166. [Google Scholar] [CrossRef]
- Tai, K.; Rappolt, M.; Mao, L.; Gao, Y.; Li, X.; Yuan, F. The stabilization and release performances of curcumin-loaded liposomes coated by high and low molecular weight chitosan. Food Hydrocoll. 2020, 99, 105355. [Google Scholar] [CrossRef]
- Alkhatib, D.; Zelai, N. Preparation, characterization and stability of silver sulfadiazine nanoliposomes. Trop. J. Pharm. Res. 2021, 20, 665–671. [Google Scholar] [CrossRef]
- Lombardo, D.; Kiselev, M.A. Methods of Liposomes Preparation: Formation and Control Factors of Versatile Nanocarriers for Biomedical and Nanomedicine Application. Pharmaceutics 2022, 14, 543. [Google Scholar] [CrossRef]
- Rems, L.; Viano, M.; Kasimova, M.A.; Miklavčič, D.; Tarek, M. The contribution of lipid peroxidation to membrane permeability in electropermeabilization: A molecular dynamics study. Bioelectrochemistry 2019, 125, 46–57. [Google Scholar] [CrossRef]
- Tan, C.; Wang, J.; Sun, B. Biopolymer-liposome hybrid systems for controlled delivery of bioactive compounds: Recent advances. Biotechnol. Adv. 2021, 48, 107727. [Google Scholar] [CrossRef]
- Shi, N.Q.; Qi, X.R. Preparation of Drug Liposomes by Reverse-Phase Evaporation. In Liposome-Based Drug Delivery Systems; Lu, W.L., Qi, X.R., Eds.; Biomaterial Engineering; Springer: Berlin, Germany, 2021; pp. 37–46. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y.Y.; Chang, Q.; Shu, Q.; Shen, N.; Wang, H.; Xie, Y.; Deng, X. Pressure-Controlled Encapsulation of Graphene Quantum Dots into Liposomes by the Reverse-Phase Evaporation Method. Langmuir 2021, 37, 14096–14104. [Google Scholar] [CrossRef]
- Delma, K.L.; Lechanteur, A.; Evrard, B.; Semdé, R.; Piel, G. Sterilization methods of liposomes: Drawbacks of conventional methods and perspectives. Int. J. Pharm. 2021, 597, 120271. [Google Scholar] [CrossRef]
- Qi, N.; Tang, X.; Lin, X.; Gu, P.; Cai, C.; Xu, H.; Zhang, Y. Sterilization stability of vesicular phospholipid gels loaded with cytarabine for brain implant. Int. J. Pharm. 2012, 427, 234–241. [Google Scholar] [CrossRef]
- Sakar, F.; Özer, A.Y.; Erdogan, S.; Ekizoglu, M.; Kart, D.; Özalp, M.; Colak, S.; Zencir, Y. Nano drug delivery systems and gamma radiation sterilization. Pharm. Dev. Technol. 2017, 22, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Shao, P.; Wang, P.; Niu, B.; Kang, J. Environmental stress stability of pectin-stabilized resveratrol liposomes with different degree of esterification. Int. J. Biol. Macromol. 2018, 119, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.U.; Omran, Z.; Anton, H.; Mély, Y.; Akram, S.; Vandamme, T.F.; Anton, N. Development of doxorubicin hydrochloride loaded pH-sensitive liposomes: Investigation on the impact of chemical nature of lipids and liposome composition on pH-sensitivity. Eur. J. Pharm. Biopharm. 2018, 133, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.R.; Wei, X.Q.; Zhang, S.; Fu, N.; Lin, Y.F.; Cai, X.X.; Peng, Q. Effects of micro-environmental pH of liposome on chemical stability of loaded drug. Nanoscale Res. Lett. 2017, 12, 1–8. [Google Scholar] [CrossRef]
- Franzé, S.; Selmin, F.; Samaritani, E.; Minghetti, P.; Cilurzo, F. Lyophilization of liposomal formulations: Still necessary, still challenging. Pharmaceutics 2018, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Schnitzer, E.; Pinchuk, I.; Bor, A.; Leikin-Frenkel, A.; Lichtenberg, D. Oxidation of liposomal cholesterol and its effect on phospholipid peroxidation. Chem. Phys. Lipids. 2007, 146, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.; Voig, M.; Simo, J.; Danner, A.K.; Frey, H.; Mailänder, V.; Helm, M.; Morsbach, S.; Landfester, K. Functionalization of liposomes with hydrophilic polymers results in macrophage uptake independent of the protein corona. Biomacromolecules 2019, 20, 2989–2999. [Google Scholar] [CrossRef]
- Li, X.; Tang, C.; Salama, M.; Xia, M.; Huang, X.; Sheng, L.; Cai, Z. Encapsulation efficiency and oral delivery stability of chitosan–liposome-encapsulated immunoglobulin Y. J. Food. Sci. 2022, 87, 1708–1720. [Google Scholar] [CrossRef]
- Assadpour, S.; Akhtari, J.; Shiran, M.R. Pharmacokinetics study of chitosan-coated liposomes containing sumatriptan in the treatment of migraine. Casp. J. Intern. Med. 2022, 13, 90. [Google Scholar] [CrossRef]
- Kari, N.; Shishir, M.R.I.; Li, Y.; Zineb, O.Y.; Mo, J.; Tangpong, J.; Chen, W. Pelargonidin-3-O-Glucoside Encapsulated Pectin-Chitosan-Nanoliposomes Recovers Palmitic Acid-Induced Hepatocytes Injury. Antioxidants 2022, 11, 623. [Google Scholar] [CrossRef]
- Katual, M.K.; Gogna, S.; Singh, G. Advancements in Treatment of Laceration by Chitosan Coated Flexible Liposomes of Mupirocin: A current prospective. Sea 2022, 4, 5. [Google Scholar]
- Salehi, S.; Nourbakhsh, M.S.; Yousefpour, M.; Rajabzadeh, G.; Sahab-Negah, S. Chitosan-coated niosome as an efficient curcumin carrier to cross the blood–brain barrier: An animal study. J. Lipos. Res. 2022, 32, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Tamaddon, L.; Mohamadi, N.; Bavarsad, N. Preparation and Characterization of Mucoadhesive Loratadine Nanoliposomes for Intranasal Administration. Turk. J. Pharm. Sci. 2021, 18, 492. [Google Scholar] [CrossRef] [PubMed]
- Lopes, N.A.; Mertins, O.; Pinilla, C.M.B.; Brandelli, A. Nisin induces lamellar to cubic liquid-crystalline transition in pectin and polygalacturonic acid liposomes. Food Hydrocoll. 2021, 112, 106320. [Google Scholar] [CrossRef]
- Lopes, N.A.; Pinilla, C.M.B.; Brandelli, A. Antimicrobial activity of lysozyme-nisin co-encapsulated in liposomes coated with polysaccharides. Food Hydrocoll. 2019, 93, 1–9. [Google Scholar] [CrossRef]
- Lopes, N.A.; Pinilla, C.M.B.; Brandelli, A. Pectin and polygalacturonic acid-coated liposomes as novel delivery system for nisin: Preparation, characterization and release behavior. Food Hydrocoll. 2017, 70, 1–7. [Google Scholar] [CrossRef]
- Ghaleshahi, A.Z.; Rajabzadeh, G. The influence of sodium alginate and genipin on physico-chemical properties and stability of WPI coated liposomes. Food Res. Int. 2020, 130, 108966. [Google Scholar] [CrossRef]
- Ghaleshahi, A.Z.; Rajabzadeh, G.; Ezzatpanah, H. Influence of Sodium Alginate and Genipin on Stability of Chitosome Containing Perilla Oil in Model and Real Drink. Eur. J. Lipid. Sci. Technol. 2020, 122, 1900358. [Google Scholar] [CrossRef]
- Trucillo, P.; Cardea, S.; Baldino, L.; Reverchon, E. Production of liposomes loaded alginate aerogels using two supercritical CO2 assisted techniques. J. CO2 Util. 2020, 39, 101161. [Google Scholar] [CrossRef]
- Maestrelli, F.; Mura, P.; González-Rodríguez, M.L.; Cózar-Bernal, M.J.; Rabasco, A.M.; Mannelli, L.D.C.; Ghelardini, C. Calcium alginate microspheres containing metformin hydrochloride niosomes and chitosomes aimed for oral therapy of type 2 diabetes mellitus. Int. J. Pharm. 2017, 530, 430–439. [Google Scholar] [CrossRef]
- Gottesmann, M.; Goycoolea, F.M.; Steinbacher, T.; Menogni, T.; Hensel, A. Smart drug delivery against Helicobacter pylori: Pectin-coated, mucoadhesive liposomes with antiadhesive activity and antibiotic cargo. Appl. Microbiol. 2020, 104, 5943–5957. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.T.T.; Tran, V.H.; Lam, T.T. Encapsulation of tagitinin C in liposomes coated by Tithonia diversifolia pectin. J. Microencapsul. 2019, 36, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Iacob, A.T.; Lupascu, F.G.; Apotrosoaei, M.; Vasinc, I.M.; Tauser, R.G.; Lupascu, D.; Giusca, S.E.; Caruntu, I.-D.; Profire, L. Recent biomedical approaches for chitosan-based materials as drug delivery nanocarriers. Pharmaceutics 2021, 13, 587. [Google Scholar] [CrossRef] [PubMed]
- Pu, C.; Tang, W.; Li, X.; Li, M.; Sun, Q. Stability enhancement efficiency of surface decoration on curcumin-loaded liposomes: Comparison of guar gum and its cationic counterpart. Food Hydrocoll. 2019, 87, 29–37. [Google Scholar] [CrossRef]
- Barba, A.A.; Bochicchio, S.; Bertoncin, P.; Lamberti, G.; Dalmoro, A. Coating of nanolipid structures by a novel simil-microfluidic technique: Experimental and theoretical approaches. Coatings 2019, 9, 491. [Google Scholar] [CrossRef]
- Kaminski, G.A.; Sierakowski, M.R.; Pontarolo, R.; Dos Santos, L.A.; de Freitas, R.A. Layer-by-layer polysaccharide-coated liposomes for sustained delivery of epidermal growth factor. Carbohyd. Polym. 2016, 140, 129–135. [Google Scholar] [CrossRef]
- Amjadi, S.; Almasi, H.; Hamishehkar, H.; Khaledabad, M.A.; Lim, L.T. Coating of betanin and carvone Co-loaded nanoliposomes with synthesized cationic inulin: A strategy for enhancing the stability and bioavailability. Food. Chem. 2022, 373, 131403. [Google Scholar] [CrossRef]
- Joseph, A.; Kumar, D.; Balakrishnan, A.; Shanmughan, P.; Maliakel, B.; Krishnakumar, I.M. Surface-engineered liposomal particles of calcium ascorbate with fenugreek galactomannan enhanced the oral bioavailability of ascorbic acid: A randomized, double-blinded, 3-sequence, crossover study. Rsc. Adv. 2021, 11, 38161–38171. [Google Scholar] [CrossRef]
- Kari, O.K.; Tavakol, S.; Parkkila, P.; Baan, S.; Savolaine, R.; Ruoslaht, T.; Johansson, N.G.; Ndika, J.; Alenius, H.; Viitala, T.; et al. Light-Activated Liposomes Coated with Hyaluronic Acid as a Potential Drug Delivery System. Pharmaceutics 2020, 12, 763. [Google Scholar] [CrossRef]
- Menon, P.; Teo, Y.Y.; Misra, M. Effect of diethylaminoethyl-dextran coated liposomes on the rheological properties of carbopol gel. Appl. Rheol. 2018, 28, 1–6. [Google Scholar] [CrossRef]
- Refai, H.; Hassan, D.; Abdelmonem, R. Development and characterization of polymer-coated liposomes for vaginal delivery of sildenafil citrate. Drug. Deliv. 2017, 24, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Farooq, A.; Iqbal, A.; Rana, N.F.; Fatima, M.; Maryam, T.; Batool, F.; Rehman, Z.; Menaa, F.; Azhar, S.; Nawaz, A.; et al. A Novel Sprague-Dawley Rat Model Presents Improved NASH/NAFLD Symptoms with PEG Coated Vitexin Liposomes. Int. J. Mol. Sci. 2022, 23, 3131. [Google Scholar] [CrossRef] [PubMed]
- Nunes, S.S.; Fernandes, R.S.; Cavalcante, C.H.; da Costa César, I.; Leite, E.A.; Lopes, S.C.A.; Ferretti, A.; Rubello, D.; Townsend, D.M.; de Oliveira, M.C.; et al. Influence of PEG coating on the biodistribution and tumor accumulation of pH-sensitive liposomes. Drug. Deliv. Transl. Res. 2019, 9, 123–130. [Google Scholar] [CrossRef]
- Haseena, M. Scholar: National School of Leadership, 8. 2019. Available online: https://jconsortium.com/index.php/scholar/article/view/23 (accessed on 15 April 2022).
- Nazeer, N.; Panicker, J.T.; Rajalekshmi, S.M.; Shaiju, S.D.A. A Review on Surface Modified Sterically Stabilized Liposomes. Int. J. Innov. Sci. Res. Technol. 2019, 4, 795–801. [Google Scholar]
- Hermal, F.; Frisch, B.; Specht, A.; Bourel-Bonnet, L.; Heurtault, B. Development and characterization of layer-by-layer coated liposomes with poly (L-lysine) and poly (L-glutamic acid) to increase their resistance in biological media. Int. J. Pharm. 2020, 586, 119568. [Google Scholar] [CrossRef] [PubMed]
- Martí Coma-Cros, E.; Biosca, A.; Lantero, E.; Manca, M.L.; Caddeo, C.; Gutiérrez, L.; Ramírez, M.; Borgheti-Cardoso, L.N.; Manconi, M.; Fernàndez-Busquets, X. Antimalarial activity of orally administered curcumin incorporated in Eudragit®-containing liposomes. Int J. Mol. Sci. 2018, 19, 1361. [Google Scholar] [CrossRef]
- Pan, L.; Li, H.; Hou, L.; Chang, Z.; Li, Y.; Li, X. Gastrointestinal digestive fate of whey protein isolate coated liposomes loading astaxanthin: Lipolysis, release, and bioaccessibility. Food Biosci. 2022, 45, 101464. [Google Scholar] [CrossRef]
- Pan, L.; Zhang, X.; Fan, X.; Li, H.; Xu, B.; Li, X. Whey protein isolate coated liposomes as novel carrier systems for astaxanthin. Eur. J. Lipid. Sci. Technol. 2020, 122, 1900325. [Google Scholar] [CrossRef]
- Yi, X.; Zheng, Q.; Pan, M.H.; Chiou, Y.S.; Li, Z.; Li, L.; Chen, Y.; Hu, J.; Duan, S.; Wei, S.; et al. Liposomal vesicles-protein interaction: Influences of iron liposomes on emulsifying properties of whey protein. Food Hydrocoll. 2019, 89, 602–612. [Google Scholar] [CrossRef]
- Taguchi, K.; Okamoto, Y.; Matsumoto, K.; Otagiri, M.; Chuang, V.T.G. When albumin meets liposomes: A feasible drug carrier for biomedical applications. Pharmaceuticals 2021, 14, 296. [Google Scholar] [CrossRef]
- Li, M.; Du, C.; Guo, N.; Teng, Y.; Meng, X.; Sun, H.; Li, S.; Yu, P.; Galons, H. Composition design and medical application of liposomes. Eur. J. Med. Chem. 2019, 164, 640–653. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Nakhaei, E.; Kawano, T.; Murata, M.; Kishimura, A.; Mori, T.; Katayama, Y. Ligand-mediated coating of liposomes with human serum albumin. Langmuir 2018, 34, 2324–2331. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.H.; Lee, M.K.; Lim, S.J. Enhanced stability of indocyanine green by encapsulation in zein-phosphatidylcholine hybrid nanoparticles for use in the phototherapy of cancer. Pharmaceutics 2021, 13, 305. [Google Scholar] [CrossRef]
- Dong, Y.; Dong, P.; Huang, D.; Mei, L.; Xia, Y.; Wang, Z.; Pan, X.; Li, G.; Wu, C. Fabrication and characterization of silk fibroin-coated liposomes for ocular drug delivery. Eur. J. Pharm. Biopharm. 2015, 91, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Battogtokh, G.; Joo, Y.; Abuzar, S.M.; Park, H.; Hwang, S.J. Gelatin Coating for the Improvement of Stability and Cell Uptake of Hydrophobic Drug-Containing Liposomes. Molecules 2022, 27, 1041. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Ansari, B.; Gharsallaoui, A. Polyelectrolytes-stabilized liposomes for efficient encapsulation of Lactobacillus rhamnosus and improvement of its survivability under adverse conditions. Food Chem. 2022, 372, 131358. [Google Scholar] [CrossRef]
- Barrera, Y.A.B.; Husteden, C.; Alherz, J.; Fuhrmann, B.; Wölk, C.; Groth, T. Extracellular matrix-inspired surface coatings functionalized with dexamethasone-loaded liposomes to induce osteo-and chondrogenic differentiation of multipotent stem cells. Mater. Sci. Eng. C 2021, 131, 112516. [Google Scholar] [CrossRef]
- Dutta, S.; Moses, J.A.; Anandharamakrishnan, C. Biomedical and food applications of biopolymer-based liposome. In Biopolymer-Based Formulations; Kunal, P., Indranil, B., Preetam, S., Doman, K., Win-Ping, D., Navneet, K.D., Kaustav, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 167–192. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Soofi, M.; Rezaei, M. Enhanced physicochemical stability of ω-3 PUFAs concentrates-loaded nanoliposomes decorated by chitosan/gelatin blend coatings. Food. Chem. 2021, 345, 128865. [Google Scholar] [CrossRef]
- Mosafer, J.; Sabbaghi, A.H.; Badiee, A.; Dehghan, S.; Tafaghodi, M. Preparation, characterization and in vivo evaluation of alginate-coated chitosan and trimethylchitosan nanoparticles loaded with PR8 influenza virus for nasal immunization. Asian J. Pharm. Sci. 2019, 14, 216–221. [Google Scholar] [CrossRef]
- Parchen, G.P.; Jacumazo, J.; Koop, H.S.; Biscaia, S.M.P.; Trindade, E.S.; Silveira, J.L.M.; de Freitas, R.A. Modulation of epidermal growth factor release by biopolymer-coated liposomes. J. Pharm. Sci. 2020, 109, 2294–2301. [Google Scholar] [CrossRef]
- Liu, W.; Tian, M.; Kong, Y.; Lu, J.; Li, N.; Han, J. Multilayered vitamin C nanoliposomes by self-assembly of alginate and chitosan: Long-term stability and feasibility application in mandarin juice. LWT Food Sci. Technol. 2017, 75, 608–615. [Google Scholar] [CrossRef]
- Karim, N.; Shishir, M.R.I.; Chen, W. Surface decoration of neohesperidin-loaded nanoliposome using chitosan and pectin for improving stability and controlled release. Int. J. Biol. Macromol. 2020, 164, 2903–2914. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, L.N.D.M.; de Paula, E.; Rossi, D.A.; Monteiro, G.P.; Júnior, E.C.V.; Silva, R.R.; Fonseca, B.B. Hybrid pectin-liposome formulation against multi-resistant bacterial strains. Pharmaceutics 2020, 12, 769. [Google Scholar] [CrossRef]
- Raj, V.; Raorane, C.J.; Lee, J.H.; Lee, J. Appraisal of chitosan-gum Arabic-coated bipobiopolymericocarriers for efficient dye removal and eradication of the plant pathogen Botrytis cinerea. ACS Appl. Mater. Inter. 2021, 13, 47354–47370. [Google Scholar] [CrossRef]
- Islam, N.; Ferro, V. Recent advances in chitosan-based nanoparticulate pulmonary drug delivery. Nanoscale 2016, 8, 14341–14358. [Google Scholar] [CrossRef]
- Mehdizadeh, A.; Shahidi, S.A.; Shariatifar, N.; Shiran, M.; Ghorbani-HasanSaraei, A. Evaluation of chitosan-zein coating containing free and nano-encapsulated Pulicaria gnaphalodes (Vent.) Boiss. extract on quality attributes of rainbow trout. J. Aquat. Food. Prod. Technol. 2021, 30, 62–75. [Google Scholar] [CrossRef]
- Deygen, I.M.; Seidl, C.; Kölmel, D.K.; Bednarek, C.; Heissler, S.; Kudryashova, E.V.; Bräse, S.; Schepers, U. Novel prodrug of doxorubicin modified by stearoylspermine encapsulated into PEG-chitosan-stabilized liposomes. Langmuir 2016, 32, 10861–10869. [Google Scholar] [CrossRef]
- Gomaa, A.I.; Martinent, C.; Hammami, R.; Fliss, I.; Subirade, M. Dual coating of liposomes as encapsulating matrix of antimicrobial peptides: Development and characterization. Front. Chem. 2017, 5, 103. [Google Scholar] [CrossRef] [PubMed]
- Rezvani, M.; Manca, M.L.; Muntoni, A.; De Gioannis, G.; Pedraz, J.L.; Gutierrez, G.; Manconi, M. From process effluents to intestinal health promotion: Developing biopolymer-whey liposomes loaded with gingerol to heal intestinal wounds and neutralize oxidative stress. Int. J. Pharm. 2022, 613, 121389. [Google Scholar] [CrossRef] [PubMed]
- Luo, R.; Lin, M.; Zhang, C.; Shi, J.; Zhang, S.; Chen, Q.; Gao, F. Genipin-crosslinked human serum albumin coating using a tannic acid layer for enhanced oral administration of curcumin in the treatment of ulcerative colitis. Food Chem. 2020, 330, 127241. [Google Scholar] [CrossRef]
- Zamani-Ghaleshahi, A.; Rajabzadeh, G.; Ezzatpanah, H.; Ghavami, M. Biopolymer coated nanoliposome as enhanced carrier system of perilla oil. Food. Biophys. 2020, 15, 273–287. [Google Scholar] [CrossRef]
- Shah, V.; Jobanputra, A.; Saxena, B.; Nivsarkar, M. Development and Characterization of Saturated Fatty Acid-Engineered, Silica-Coated Lipid Vesicular System for Effective Oral Delivery of Alfa-Choriogonadotropin. AAPS PharmSciTech 2021, 22, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pham, X.H.; Park, S.M.; Ham, K.M.; Kyeong, S.; Son, B.S.; Kim, J.; Hahm, E.; Kim, Y.H.; Bock, S.; Kim, W.; et al. Synthesis and application of silica-coated quantum dots in biomedicine. Int. J. Mol. Sci. 2021, 22, 10116. [Google Scholar] [CrossRef]
- Bewernitz, M.A.; Lovett, A.C.; Gower, L.B. Liquid–Solid Core-Shell Microcapsules of Calcium Carbonate Coated Emulsions and Liposomes. Appl. Sci. 2020, 10, 8551. [Google Scholar] [CrossRef]
- Wu, S.; Jiang, M.; Mao, H.; Zhao, N.; He, D.; Chen, Q.; Liu, D.; Zhang, W.; Song, X.M. A sensitive cholesterol electrochemical biosensor based on biomimetic cerasome and graphene quantum dots. Anal. Bioanal. Chem. 2022, 414, 3593–3603. [Google Scholar] [CrossRef]
- Hasan, M.; Messaoud, G.B.; Michaux, F.; Tamayol, A.; Kahn, C.J.; Belhaj, N.; Linder, M.; Arab-Tehrany, E. Chitosan-coated liposomes encapsulating curcumin: Study of lipid–polysaccharide interactions and nanovesicle behavior. Rsc. Adv. 2016, 6, 45290–45304. [Google Scholar] [CrossRef]
- Mikušová, V.; Mikuš, P. Advances in chitosan-based nanoparticles for drug delivery. Int. J. Mol. Sci. 2021, 22, 9652. [Google Scholar] [CrossRef]
- Jøraholmen, M.W.; Bhargava, A.; Julin, K.; Johannessen, M.; Škalko-Basnet, N. The antimicrobial properties of chitosan can be tailored by formulation. Mar. Drugs 2020, 18, 96. [Google Scholar] [CrossRef]
- Ramezanzade, L.; Hosseini, S.F.; Akbari-Adergani, B.; Yaghmur, A. Cross-linked chitosan-coated liposomes for encapsulation of fish-derived peptide. LWT Food Sci. Technol. 2021, 150, 112057. [Google Scholar] [CrossRef]
- Seyedabadi, M.M.; Rostami, H.; Jafari, S.M.; Fathi, M. Development and characterization of chitosan-coated nanoliposomes for encapsulation of caffeine. Food Biosci. 2021, 40, 100857. [Google Scholar] [CrossRef]
- Ran, L.; Chi, Y.; Huang, Y.; He, Q.; Ren, Y. Synergistic antioxidant effect of glutathione and edible phenolic acids and improvement of the activity protection by coencapsulation into chitosan-coated liposomes. LWT Food Sci. Technol. 2020, 127, 109409. [Google Scholar] [CrossRef]
- Hao, J.; Guo, B.; Yu, S.; Zhang, W.; Zhang, D.; Wang, J.; Wang, Y. Encapsulation of the flavonoid quercetin with chitosan-coated nano-liposomes. LWT Food Sci. Technol. 2017, 85, 37–44. [Google Scholar] [CrossRef]
- Akgün, D.; Gültekin-Özgüven, M.; Yücetepe, A.; Altin, G.; Gibis, M.; Weiss, J.; Özçelik, B. Stirred-type yoghurt incorporated with sour cherry extract in chitosan-coated liposomes. Food Hydrocoll. 2020, 101, 105532. [Google Scholar] [CrossRef]
- Kumar, S.; Deepak, V.; Kumari, M.; Dutta, P.K. Antibacterial activity of diisocyanate-modified chitosan for biomedical applications. Int. J. Biol. Macromol. 2016, 84, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar, S.; Chandran, S.V.; Selvamurugan, N.; Nazeer, R.A. Biodistribution and pharmacokinetics of thiolated chitosan nanoparticles for oral delivery of insulin in vivo. Int. J. Biol. Macromol. 2020, 150, 281–288. [Google Scholar] [CrossRef]
- Tsai, L.C.; Chen, C.H.; Lin, C.W.; Ho, Y.C.; Mi, F.L. Development of multifunctional nanoparticles self-assembled from trimethyl chitosan and fucoidan for enhanced oral delivery of insulin. Int. J. Biol. Macromol. 2019, 126, 141–150. [Google Scholar] [CrossRef]
- Sharma, M.; Sharma, R.; Jain, D.K.; Saraf, A. Enhancement of oral bioavailability of poorly water-soluble carvedilol by chitosan nanoparticles: Optimization and pharmacokinetic study. Int. J. Biol. Macromol. 2019, 135, 246–260. [Google Scholar] [CrossRef]
- Chaves, M.A.; Filho, P.L.O.; Jange, C.G.; Sinigaglia-Coimbra, R.; Oliveira, C.L.P.; Pinho, S.C. Structural characterization of multilamellar liposomes coencapsulating curcumin and vitamin D3. Colloids Surf. A Physicochem. Eng. Asp. 2018, 549, 112–121. [Google Scholar] [CrossRef]
- Qiu, C.; Zhao, M.; Decker, E.A.; McClements, D.J.; Qiu, C.; Zhao, M.; Decker, E.A.; McClements, D.J. Influence of anionic dietary fibers (xanthan gum and pectin) on oxidative stability and lipid digestibility of wheat protein-stabilized fish oil-in-water emulsion. Food Res. Int. 2015, 74, 131–139. [Google Scholar] [CrossRef]
- Grijalvo, S.; Mayr, J.; Eritja, R.; Díaz, D.D. Biodegradable liposome-encapsulated hydrogels for biomedical applications: A marriage of convenience. Biomater. Sci. 2016, 4, 555–574. [Google Scholar] [CrossRef]
- De Leo, V.; Milano, F.; Agostiano, A.; Catucci, L. Recent advancements in polymer/liposome assembly for drug delivery: From surface modifications to hybrid vesicles. Polymers 2021, 13, 1027. [Google Scholar] [CrossRef] [PubMed]
- Sahatsapan, N.; Pamornpathomkul, B.; Rojanarata, T.; Ngawhirunpat, T.; Poonkhum, R.; Opanasopit, P.; Patrojanasophon, P. Feasibility of mucoadhesive chitosan maleimide-coated liposomes for improved buccal delivery of a protein drug. J. Drug Deliv. Sci. Tec. 2022, 69, 103173. [Google Scholar] [CrossRef]
- Cao, Y.; Dong, X.; Chen, X. Polymer-Modified Liposomes for Drug Delivery: From Fundamentals to Applications. Pharmaceutics 2022, 14, 778. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P.; Narula, J.; Khaw, B.A.; Trubetskoy, V.S. PEG-modified liposomes for gamma-and magnetic resonance imaging. In Stealth Liposomes; Lasic, D.D., Martin, F.J., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 247–260. [Google Scholar]
- Mastrotto, F.; Brazzale, C.; Bellato, F.; De Martin, S.; Grange, G.; Mahmoudzadeh, M.; Caliceti, P. In vitro and in vivo behavior of liposomes decorated with PEGs with different chemical features. Mol. Pharm. 2019, 17, 472–487. [Google Scholar] [CrossRef]
- Abbina, S.; Parambath, A. PEGylation and its alternatives: A summary. In Engineering of Biomaterials for Drug Delivery Systems: Beyond Polyethylene Glycol; Parambath, A., Ed.; Woodhead Publishing Series in Biomaterials; Woodhead: Duxford, UK, 2018; pp. 363–376. [Google Scholar] [CrossRef]
- Hatakeyama, H.; Akita, H.; Harashima, H. The polyethyleneglycol dilemma: Advantage and disadvantage of PEGylation of liposomes for systemic genes and nucleic acids delivery to tumors. Biol. Pharm. Bull. 2013, 36, 892–899. [Google Scholar] [CrossRef]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly (ethylene glycol) in drug delivery: Pros and cons as well as potential alternatives. Angew. Chem. Int. Ed. 2010, 49, 6288–6308. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Sun, M.; Zhang, J.; Bi, Y. Coating liposomes with ring-like PEG: The synthesis and stealth effect of cholesterol–PEG–cholesterol. Mater. Adv. 2022, 3, 2417–2424. [Google Scholar] [CrossRef]
- Sadzuka, Y.; Nakade, A.; Hirama, R.; Miyagishima, A.; Nozawa, Y.; Hirota, S.; Sonobe, T. Effects of mixed polyethyleneglycol modification on fixed aqueous layer thickness and antitumor activity of doxorubicin containing liposome. Int. J. Pharm. 2002, 238, 171–180. [Google Scholar] [CrossRef]
- Lila, A.S.A.; Nawata, K.; Shimizu, T.; Ishida, T.; Kiwada, H. Use of polyglycerol (PG), instead of polyethylene glycol (PEG), prevents induction of the accelerated blood clearance phenomenon against long-circulating liposomes upon repeated administration. Int. J. Pharm. 2013, 456, 235–242. [Google Scholar] [CrossRef]
- Lin, W.; Kampf, N.; Goldberg, R.; Driver, M.J. Poly-phosphocholinated liposomes form stable superlubrication vectors. Langmuir 2019, 35, 6048–6054. [Google Scholar] [CrossRef]
- de Morais, F.A.P.; Gonçalves, R.S.; Braga, G.; Calori, I.R.; Pereira, P.C.S.; Batistela, V.R.; Caetano, W.; Hioka, N. Stable dipalmitoylphosphatidylcholine liposomes coated with an F127 copolymer for hypericin loading and delivery. ACS Appl. Nano. Mater. 2020, 3, 4530–4541. [Google Scholar] [CrossRef]
- Lane, R.S.; Haller, F.M.; Chavaroche, A.A.; Almond, A.; DeAngelis, P.L. Heparosan-coated liposomes for drug delivery. Glycobiology 2017, 27, 1062–1074. [Google Scholar] [CrossRef] [PubMed]
- Gaber, M.; Medhat, W.; Hany, M.; Saher, N.; Fang, J.Y.; Elzoghby, A. Protein-lipid nanohybrids as emerging platforms for drug and gene delivery: Challenges and outcomes. J. Control. Release 2017, 254, 75–91. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Moosa, B.A.; Croissant, J.G.; Khashab, N.M. Electrostatic Assembly/Disassembly of Nanoscaled Colloidosomes for Light-Triggered Cargo Release. Angew. Chem. Int. Ed. 2015, 127, 6908–6912. [Google Scholar] [CrossRef]
- Román-Aguirre, M.; Leyva-Porras, C.; Cruz-Alcantar, P.; Aguilar-Elguézabal, A.; Saavedra-Leos, M.Z. Comparison of Polysaccharides as Coatings for Quercetin-Loaded Liposomes (QLL) and Their Effect as Antioxidants on Radical Scavenging Activity. Polymers 2020, 12, 2793. [Google Scholar] [CrossRef]
- Mohammadi, A.; Jafari, S.M.; Mahoonak, A.S.; Ghorbani, M. Liposomal/nanoliposomal encapsulation of food-relevant enzymes and their application in the food industry. Food Bioprocess. Technol. 2021, 14, 23–38. [Google Scholar] [CrossRef]
- Šeremet, D.; Vugrinec, K.; Petrović, P.; Butorac, A.; Kuzmić, S.; Cebin, A.V.; Mandura, A.; Lovrić, M.; Pjanović, R.; Komes, D. Formulation and characterization of liposomal encapsulated systems of bioactive ingredients from traditional plant mountain germander (Teucrium montanum L.) for incorporation into coffee drinks. Food Chem. 2022, 370, 131257. [Google Scholar] [CrossRef]
- Amjadi, S.; Almasi, H.; Hamishehkar, H.; Khaledabad, M.A.; Lim, L.T. Cationic inulin as a new surface decoration hydrocolloid for improving the stability of liposomal nanocarriers. Colloid. Surface. B 2022, 213, 112401. [Google Scholar] [CrossRef]
- Gibis, M.; Rahn, N.; Weiss, J. Physical and oxidative stability of uncoated and chitosan-coated liposomes containing grape seed extract. Pharmaceutics 2013, 5, 421–433. [Google Scholar] [CrossRef]
- Chen, S.; Ma, X.; Han, Y.; Wei, Y.; Guo, Q.; Yang, S.; Zhang, Y.; Liao, W.; Gao, Y. Effect of chitosan molecular weight on zein-chitosan nanocomplexes: Formation, characterization, and the delivery of quercetagetin. Int. J. Biol. Macromol. 2020, 164, 2215–2223. [Google Scholar] [CrossRef]
- Lin, L.; Zhu, Y.; Cui, H. Inactivation of Escherichia coli O157: H7 treated by poly-L-lysine-coated bacteriophages liposomes in pork. J. Food. Safety. 2018, 38, e12535. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, H.; Duan, L.R.; Li, H.; Yang, Q.; Tu, H.H.; Cao, W.; Wang, S.W. Preparation, characterization and evaluation of bufalin liposomes coated with citrus pectin. Colloids Surf. A Physicochem. Eng. Asp. 2014, 444, 54–62. [Google Scholar] [CrossRef]
- Feng, S.; Sun, Y.; Wang, P.; Sun, P.; Ritzoulis, C.; Shao, P. Co-encapsulation of resveratrol and epigallocatechin gallate in low methoxyl pectin-coated liposomes with great stability in orange juice. Int. J. Food. Sci. Technol. 2020, 55, 1872–1880. [Google Scholar] [CrossRef]
- Belhaj, N.; Arab-Tehrany, E.; Loing, E.; Bézivin, C. Skin delivery of hydrophilic molecules from liposomes and polysaccharide-coated liposomes. Int. J. Cosmet. Sci. 2017, 39, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Fu, D.; Zhang, X.; Chai, J.; Tian, S.; Han, J. Development and validation of a new artificial gastric digestive system. J. Food Res. Int. 2019, 122, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Lu, Y.; Qi, J. Oral delivery of liposomes. Ther. Deliv. 2015, 6, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Zaeim, D.; Mulet-Cabero, A.I.; Read, S.A.; Liu, W.; Han, J.; Wilde, P.J. Effect of oil droplet size on the gastric digestion of milk protein emulsions using a semi-dynamic gastric model. Food Hydrocoll. 2022, 124, 107278. [Google Scholar] [CrossRef]
- Infantes-Garcia, M.R.; Verkempinck, S.H.E.; Gonzalez-Fuentes, P.G.; Hendrickx, M.E.; Grauwet, T. Lipolysis products formation during in vitro gastric digestion is affected by the emulsion interfacial composition. Food Hydrocoll. 2021, 110, 106163. [Google Scholar] [CrossRef]
- Liu, W.; Jin, Y.; Wilde, P.J.; Hou, Y.; Wang, Y.; Han, J. Mechanisms, physiology, and recent research progress of gastric emptying. Crit. Rev. Food Sci. Nutr. 2021, 61, 2742–2755. [Google Scholar] [CrossRef]
- Salhi, A.; Carriere, F.; Grundy, M.M.L.; Aloulou, A. Enzymes involved in lipid digestion. In Bioaccessibility and Digestibility of Lipids from Food; Grundy, M.M.L., Wilde, P.J., Eds.; Springer International Publishing: Cham, Switzerland, 2003; pp. 3–28. [Google Scholar] [CrossRef]
- Liu, W.; Kong, Y.; Ye, A.; Shen, P.; Dong, L.; Xu, X.; Han, J. Preparation, formation mechanism and in vitro dynamic digestion behavior of quercetin-loaded liposomes in hydrogels. Food Hydrocoll. 2020, 104, 105743. [Google Scholar] [CrossRef]
- Macierzanka, A.; Torcello-Gómez, A.; Jungnickel, C.; Maldonado-Valderrama, J. Bile salts in digestion and transport of lipids. Adv. Colloid Interface Sci. 2019, 274, 102045. [Google Scholar] [CrossRef]
- Cuomo, F.; Cofelice, M.; Venditti, F.; Ceglie, A.; Miguel, M.; Lindman, B.; Lopez, F. In-vitro digestion of curcumin loaded chitosan-coated liposomes. Colloids Surf. B Biointerfaces 2018, 168, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, J.; Xia, W. Hypocholesterolaemic effects of different chitosan samples in vitro and in vivo. Food. Chem. 2008, 107, 419–425. [Google Scholar] [CrossRef]
- Garg, U.; Chauhan, S.; Nagaich, U.; Jain, N. Current advances in chitosan nanoparticles-based drug delivery and targeting. Adv. Pharm. Bull. 2019, 9, 195. [Google Scholar] [CrossRef] [PubMed]
- Han, H.K.; Shin, H.J.; Ha, D.H. Improved oral bioavailability of alendronate via the mucoadhesive liposomal delivery system. Eur. J. Pharm. Sci. 2012, 46, 500–507. [Google Scholar] [CrossRef]
- Huang, A.; Makhlof, A.; Ping, Q.; Tozuka, Y.; Takeuchi, H. N-trimethyl chitosan-modified liposomes as carriers for oral delivery of salmon calcitonin. Drug. Deliv. 2011, 18, 562–569. [Google Scholar] [CrossRef]
- Park, S.N.; Jo, N.R.; Jeon, S.H. Chitosan-coated liposomes for enhanced skin permeation of resveratrol. J. Ind. Eng. Chem. 2014, 20, 1481–1485. [Google Scholar] [CrossRef]
- Ducat, E.; Evrard, B.; Peulen, O.; Piel, G. Cellular uptake of liposomes monitored by confocal microscopy and flow cytometry. J. Drug. Deliv. Sci. Tec. 2011, 21, 469–477. [Google Scholar] [CrossRef]
- Wang, J.; Byrne, J.D.; Napier, M.E.; DeSimone, J.M. More effective nanomedicines through particle design. Small 2011, 7, 1919–1931. [Google Scholar] [CrossRef]
- Nejdl, L.; Kudr, J.; Moulick, A.; Hegerova, D.; Ruttkay-Nedecky, B.; Gumulec, J.; Cihalova, K.; Smerkova, K.; Dostalova, S.; Krizkova, S.; et al. Platinum nanoparticles induce damage to DNA and inhibit DNA replication. PLoS ONE 2017, 12, e0180798. [Google Scholar] [CrossRef]
- Rautio, J.; Kumpulainen, H.; Heimbach, T.; Oliyai, R.; Oh, D.; Järvinen, T.; Savolainen, J. Prodrugs: Design and clinical applications. Nat. Rev. Drug. Discov. 2008, 7, 255–270. [Google Scholar] [CrossRef] [PubMed]
- Nagy, N.A.; Castenmiller, C.; Vigario, F.L.; Sparrius, R.; van Capel, T.M.; de Haas, A.M.; de Jong, E.C. Uptake kinetics of liposomal formulations of differing charge influences development of in vivo dendritic cell immunotherapy. J. Pharm. Sci. 2022, 111, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Andar, A.U.; Hood, R.R.; Vreeland, W.N.; DeVoe, D.L.; Swaan, P.W. Microfluidic preparation of liposomes to determine particle size influence on cellular uptake mechanisms. Pharm. Res. 2014, 31, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, M.; Azadi, A.; Rafiei, P.; Ashrafi, H. A pharmacokinetic overview of nanotechnology-based drug delivery systems: An ADME-oriented approach. Crit. Rev. ™ Ther. Drug Carr. Syst. 2013, 30, 435–467. [Google Scholar] [CrossRef]
- Li, M.; Al-Jamal, K.T.; Kostarelos, K.; Reineke, J. Physiologically based pharmacokinetic modeling of nanoparticles. ACS Nano 2010, 4, 6303–6317. [Google Scholar] [CrossRef]
- Oberdörster, G. Safety assessment for nanotechnology and nanomedicine: Concepts of nanotoxicology. J. Intern. Med. 2010, 267, 89–105. [Google Scholar] [CrossRef]
- Yang, R.S.; Chang, L.W.; Yang, C.S.; Lin, P. Pharmacokinetics and physiologically-based pharmacokinetic modeling of nanoparticles. J. Nanosci. Nanotechnol. 2010, 10, 8482–8490. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Long-circulating and target-specific nanoparticles: Theory to practice. Pharmacol. Rev. 2001, 53, 283–318. [Google Scholar]
- Soppimath, K.S.; Aminabhavi, T.M.; Kulkarni, A.R.; Rudzinski, W.E. Biodegradable polymeric nanoparticles as drug delivery devices. J. Control. Release 2001, 70, 1–20. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal formulations in clinical use: An updated review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef]
- Shen, Z.; Fisher, A.; Liu, W.K.; Li, Y. PEGylated “stealth” nanoparticles and liposomes. In Engineering of Biomaterials for Drug Delivery Systems; Parambath, A., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 1–26. [Google Scholar] [CrossRef]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Kobarfard, F.; Jave, Z.; Rajabi, S.; Sharifi-Rad, J. Multivesicular liposome (Depofoam) in human diseases. Iran. J. Pharm. Res. 2020, 19, 9. [Google Scholar] [CrossRef] [PubMed]
- Motamarry, A.; Asemani, D.; Haemmerich, D. Thermosensitive liposomes. In Liposome; IntechOpen Limited: London, UK, 2017; pp. 187–212. [Google Scholar] [CrossRef]
- Barenholz, Y.C. Doxil®—The first FDA-approved nano-drug: Lessons learned. J. Control. Release 2012, 160, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. U.S. Method of Making Liposomes, Liposome Compositions Made by the Methods, and Methods of Using the Same. U.S. Patent 10016389, 10 July 2018. [Google Scholar]
- Lipocelltech™–Next Generation Liposomes, The Netherlands. Available online: https://lipocelltech.com/#production (accessed on 15 April 2022).
- Crispin, E.G. Re-Oiled, and Hyper-Oiled Lecithin Carrier Vehicles. U.S. Patent 20220071198A1, 4 December 2017. [Google Scholar]
- Chiu, G.N.; Abraham, S.A.; Ickenstein, L.M.; Karlsson, G.; Edwards, K.; Bally, M.B. Encapsulation of doxorubicin into thermosensitive liposomes via complexation with the transition metal manganese. J. Control. Release 2005, 104, 271–288. [Google Scholar] [CrossRef] [PubMed]
| Factors Range/Value | Stability Measurements | Testing Time | Active Compounds Encapsulated in Liposomes | Bibliography |
|---|---|---|---|---|
| Temperature | ||||
| 4 °C, 25 °C, 37 °C | 75.54%, 67.57%, 20.28% retention rates | 21 days | Betacyanin | [40] |
| 20–47 °C | 3.47–4.33% release | 20 days | Piperine | [41] |
| −150 °C, −80 °C, −25 °C (Liquid nitrogen) | 76–90%, 75–88%, 37% retention | 90 days | Carboxyfluorescein | [35] |
| 4 °C, 25 °C | 14.25%, 40.04% loss in nanoliposomes | 21 days | Red cabbage anthocyanins | [42] |
| Room temperature | 30–90% degradation of extract | 21 days | Black carrot extract | [43] |
| 4 °C, 25 °C | 96.81%, 8.82% encapsulation efficiency | 90 days | Phenylethyl resorcinol | [44] |
| 4 °C, 50 °C | 89.43% retention rate | 30 days | Curcumin | [45] |
| 4 °C, 25 °C | 90%, 80% encapsulation efficiency | 30 days | Afatinib | [46] |
| 4 °C | 95% change rate of vesicle size | 21 days | Curcumin | [47] |
| Light | ||||
| UV-A (12.86 W/m2), UV-B (15 W/m2), UV-C (14.29 W/m2) irradiation | Instability to UV irradiation: UV-A < UV-B < UV-C; 25.841%, 32.881%, 35.678% degradation for small unilamellar vesicles liposomes | 4 h | Fulfonamides, sulfamethoxypyridazine, sulfachloropyridazine | [38] |
| UV light irradiance (0.35 W/m2) | 88% retention | 6 h | Curcumin | [47] |
| Natural sunlight | 75.21% retention rate | 180 days | Quercetin | [39] |
| Direct sunlight; sun UV irradiation through window glass; outdoors under the shade | Completely degraded | 24 days | Silver sulfadiazine | [48] |
| UVA irradiation | 100–36.5% degradation | 48 h | Phenylethyl resorcinol | [44] |
| Fluorescent lamp (20 Watt) or dark | 49.44% improved storage stability | 30 days | Curcumin | [45] |
| D65/ID65 emission Standard (artificial daylight fluorescent lamp, indoor indirect daylight) | Not available | 30 days | Anti-inflammatory drugs | [36] |
| pH Value | Stability Measurements | Testing Time | Active Compounds Encapsulated in Liposomes | Bibliography |
|---|---|---|---|---|
| 3, 4.3, 5, 7 | 50%, 80%, 80%, 80 retention rates | 21 days | Betacyanin | [48] |
| 5.5, 6.0, 6.4, 7.4 | Not available | 30 min | Doxorubicin hydrochloride | [58] |
| 5.5, 7.4 | Cationic liposomes 63.6%, nontargeting liposomes 28.1%, pH-sensitive liposomes 35.6%—release rate in neutral pH 7.4 | 4 h for acidic conditions, 24 h for alkaline conditions | Afatinib | [46] |
| 2.5, 5.0, 7.4 | 55%, 43%, 34% encapsulation efficiency post-incubation | 6 h | Curcumin | [59] |
| 4.5, 7.4, 10.5 | Completely degraded at day 18 acidic, neutral conditions, degraded at day 30 alkaline conditions | 30 days | Silver sulfadiazine | [48] |
| 5, 6, 7, 8, 9 | >80% acidic conditions retention rate, <50% neutral and alkaline conditions retention rate | 30 days | Curcumin | [45] |
| Coating Material | Type of Encapsulated Drugs | The Advantage of the Coating Material Used | Bibliography |
|---|---|---|---|
| Saccharides and their derivatives | |||
| Chitosan | Immunoglobulin/mupirocin/Curcumin/sumatriptan/mucoadhesive loratadine/pelargonidin-3 -O-glucoside |
| [63,64,65,66,67,68] |
| Poly-galacturonic acid | Nisin |
| [69,70,71] |
| Sodium alginate | Calcitonin/Perilla Oil |
| [72,73] |
| Calcium alginate | Oxaliplatin/Acid Folic/Ampicillin/Metformin |
| [74,75] |
| Pectin | Resveratrol/Amoxicillin/Tagitinine C/Phlorizin |
| [57,76,77] |
| Starch | Fasudil |
| [78] |
| Guar gum | Hydrophobic bioactive compounds/Vitamin D3 |
| [79,80] |
| Xanthan gum | Dioctadecyl dimethylammonium bromide |
| [81] |
| Cationic inulin | Betaine/carvone |
| [82] |
| Galactomannan | Ascorbic acid |
| [83] |
| Hyaluronic acid | Antitumoral drug delivery/anti-melanoma agents (dacarbazine and eugenol) |
| [84] |
| Diethylaminomethyl-dextran | Drug |
| [85] |
| Hydroxypropyl methylcellulose | Sildenafil |
| [86] |
| Polymer/Copolymer | |||
| Polyethylene glycol PEG | Vitexin |
| [17,87,88] |
| Poly (hydroxyethyl-l-asparagine) | Antitumoral drugs |
| [89,90] |
| Poly(L-lysine) and poly (L-glutamic acid) | Recommended for drug formulations |
| [91] |
| Eudragit EPO | Curcumin |
| [92] |
| Proteins | |||
| Whey protein | Astaxanthin, iron |
| [93,94,95] |
| Albumin | Vancomycin/paclitaxel/ellagic acid |
| [96,97,98] |
| Zein | Indocyanine green |
| [99] |
| Silk fibroin (SF) | Ibuprofen |
| [100] |
| Gelatine | Arginyl-glycyl-aspartic acid/aspartic acid/Lactobacilli rhamnoses/Amphotericin B |
| [101,102] |
| Collagen | Dexamethasone |
| [103] |
| Combinations between groups of materials | |||
| Chitosan–gelatine | ω-3 PUFA/BSA |
| [104,105] |
| Chitosan–sodium alginate | Inactivated PR8 Influenza virus/cationic liposomes/resveratrol |
| [106,107,108] |
| Chitosan–pectin | Neohesperidin/norfloxacin |
| [109,110] |
| Chitosan–arabic gum | 5I-1H-indole (5ID) |
| [111] |
| Chitosan–xanthan gum | Pulmonary drugs |
| [112] |
| Chitosan–zein | Pulicaria gnaphadoles (Vent) Boiss |
| [113] |
| PEG–chitosan | Doxorubicin/Stearoyl spermine |
| [114] |
| Pectin–whey protein | Negatively and positively charged liposomes |
| [115] |
| Pectin–polygalacturonic acid | Nisin |
| [71] |
| Whey protein, xanthan gum, tragacanth, arabic gum, and sodium alginate | Gingerol |
| [116] |
| Other | |||
| Genipin (glycoside, cross-linker) | Flaxseed oil/perilla oil/tannic acid |
| [51,72,117,118] |
| Silica | Epirubicin-hydrochloride/Alfa-choriogonadotropin |
| [119,120] |
| Calciumcarbonate | Drug |
| [121] |
| Ceramic | Cholesterol |
| [122] |
| Coating Material | Type of Encapsulated Food-Grade Active Compounds | The Advantage of the Coating Material Used | Bibliography |
|---|---|---|---|
| Saccharides and their derivatives | |||
| Chitosan | Fish-derived peptide/glutathione/caffeic acid/flavonoids/quercetin/sour cherry extract/Morus nigra waste extract/caffeine |
| [126,127,128,129,130] |
| Sodium alginate | Vitamin C |
| [83] |
| Calcium alginate | Enzymes |
| [155] |
| Pectin | Echinacosides and verbascoside |
| [156] |
| Inulin | Without active compound |
| [157] |
| Lactose | Quercetin |
| [154] |
| Proteins | |||
| Whey protein | Astaxanthin |
| [68,69] |
| Combinations between groups of materials | |||
| Chitosan–sodium alginate | Polyelectrolyte |
| [83] |
| Chitosan–pectin | Hibiscus extract |
| [158] |
| Chitosan–zein | Pulicaria gnaphalodes (Vent) extract/quercetagetin |
| [113,159] |
| Pectin–whey protein | Antimicrobial peptides |
| [90] |
| Others | |||
| Poly (L-lysine) | Bacteriophage/bioactive peptides |
| [160] |
| Drug | Formulation/Coating Technology | Used for | Expected Results after the Clinical Trial | Phase | Clinicaltrials.gov Identifier |
|---|---|---|---|---|---|
| Bupivacaine | Liposomal bupivacaine/DepoFoam technology | Opioid use pain, postoperative colectomy colorectal surgery | A substantially longer duration of action than normal bupivacaine (96 h versus 8–9 h, respectively) | IV | NCT03638635 |
| 2B3-101- (glutathione (GSH) pegylated liposomal doxorubicin hydrochloride formulation) | PEG coating | Meningeal carcinomatosis | Coating liposomes with PEG guarantees that they circulate for a longer period of time in plasma | II | NCT01818713 |
| Anti-EGFR immunoliposomes loaded with doxorubicin | Attached are monoclonal antibodies or antibody fragments to the surface of liposomes (immunoliposomes, antibody-linked nanoparticles) | Solid tumors | Immunoliposomes coated with antibodies bind more selectively to antigens expressed on target cells, and they are internalized more efficiently. Drug resistance can be overcome by such delivery systems | I | NCT01702129 |
| Liposomal astaxanthin | Not specified | Bioavailability of astaxanthin formulations | Human crossover pharmacokinetic study pathways of astaxanthin in the bloodstream | Not applicable | NCT02397811 |
| Doxil | Not specified | Breast cancer | Assess the effectiveness of using heat therapy in addition to the chemotherapy drug Doxil to treat recurrent breast cancer that has spread to the chest wall after mastectomy | II | NCT02192021 |
| Patent Application/Patent, ID, Title, Year | Material Coating | Effects on Stability |
|---|---|---|
| Ceftazidime combined powder injection and preparation method and product specification thereof, CN111840232A, 2020 | The ceftazidime and chitosan nanoparticles are each coated with vesicles, and the liposomes are then combined to create liposome-mixed nanoparticles | The product purity is high, the selection range is expanded, the side effects are minimal, and the safety is high. The product obtained by the preparation process is of stable quality and good pharmacological effect. |
| Treatment of age-related macular degeneration, US2020262903A1, 2020 | The nanoparticles are coated with a drug targeting the vascular endothelial growth factor receptor (VEGFR), such as anti-VEGFR antibodies, anti-VEGFR aptamers, anti-VEGFR binding peptides | The stability of the nanocomposite at elevated temperatures indicates the successful support of GOF for liposomes |
| A biodegradable nano-theranostic composite and process of preparation thereof, US2020237667A1, 2020 | Graphene oxide (GO) was deposited in the form of a thin film on both the inner and outer surfaces of the liposomes | The stability problem was solved by reinforcing the very fragile lipid membrane-based liposome wall with a dense inclusion of GO. This makes the wall very stable, even at a pH as low as 5 for several hours and at temperature as high as 50 ᵒC |
| DNA brick-assisted liposome sorting US20210267894A1, 2021 | Liposomes were coated with DNA | The stability of a liposome was improved and more functionalization was possible when a DNA coating was applied. DNA coatings have been useful because nucleases can easily remove them, and they are inert to most biochemical reagents. |
| Liposomes encapsulating anticancer drugs and use thereof in the treatment of malignant tumors US20050100590A1, 2005 | Liposomes were coated with a lipopeptide consisting of a lipid fragment, an active oligopeptide, and an oligopeptide spacer between the other two fragments | The addition of a negatively charged phospholipid favors the stability of the liposome solution and prevents the spontaneous aggregation of the liposomes |
| Liposome-based mucus-penetrating particles for mucosal delivery US20170281541A1, 2017 | Liposomes were coated with PEG | PEG was used to increase the stability and solubility of liposomes with drugs, reduce toxicity, and prolong the half-life |
| Method of producing immunoliposomes and compositions thereof US20090232730A1, 2009 | Liposomes were coated with hyaluronan/hyaluronic acid or other glycosaminoglycans | Hyaluronan/hyaluronic acid provided protection against lyophilization and reconstitution so that only nanoscale liposomes covalently coated with hyaluronan/hyaluronic acid were structurally preserved |
| Liposomal formulations for delivery of nucleic acids US10583084, 2020 | Liposomes were coated with a glycosaminoglycan (hyaluronic acid) | The coating materials improved the condensation and stability of the liposomes |
| Class of Active Compounds | Active Compounds Encapsulated | Commercial Name(Company/Country) | Route of Administration |
|---|---|---|---|
| Pharmaceutics | |||
| Protease inhibitor | Amprenavir | Agenerase® (GlaxoSmithKline/United Kingdom) | Oral product |
| Protease inhibitor | Ritonavir | Norvir (Abbott laboratories/United States of America) | Oral product |
| Protease inhibitor | Saquinavir | Fortovase® (Hoffmann-La Roche lnc/Switzerland) | Oral product |
| Antitumor antibiotic | Doxorubicin | Doxil (Sequus Pharmaceuticals, Inc.,/United States of America), Evacet, Lipo-Dox, DC99® (Liposome Company NJ/United States of America) | Injectable product |
| Antitumor antibiotic | Daunorubicin | DaounoXome™ (NeXstar Pharmceuticals, Inc., Co/United States of America) | Injectable product |
| Antitumor antibiotic | Irinotecan | OnivydeTM (PharmaEngine/Taiwan) | Injectable product |
| Anti-inflammatory drug | Ibuprofen | Ibunex (Phoenix Pharma Pvt. Ltd./India), Solufen® (Sanofi- Aventis/France) | Oral product |
| Antihypertensive | Atorvastatin | Lipirex® (Sanofi-Aventis/France) | Oral product |
| Immunosuppressive | Cyclosporine | Neoral® (Novartis/Switzerland) | Injectable product |
| Local anesthetic | Lidocaine | ELA-Max (Ferndale Pharmaceuticals Ltd./United States of America) | Oral product |
| Antifungal | Amphotericin B | Abhope (Abbott laboratories/United States of America), Ambilon (Celon Pharma Ltd./Poland), Abelcet (Liposome Company NJ/United States of America), AmBisome (Astellas Pharma Inc.,/United States of America, NeXstar Pharmceuticals, Inc., Co/United States of America), Amphocil (Sequus Pharmaceuticals, Inc.,/United States of America), Myocet® (GP-Pharm/Spain), Amphonex (Bharat serums & vaccines ltd/India) | Injectable product |
| Photosensitizing agents | Verteporfin | Visudyne® (Bausch & Lomb Incorporated/United States of America) | Injectable product |
| Antimetabolite antineoplastic agent | Cytarabine | DepoCyt® (Pacira Pharmaceuticals, Inc/United States of America) | Injectable product |
| Chemotherapy drug | Cisplatin | Lipoplatin® (Regulon, Inc/Greece) | Injectable product |
| Opioid | Morphine sulfate | DepoDur® (Skyepharma Production SAS/France) | Injectable product |
| Antibiotic | Amikacin | MiKasome® (NeXstar Pharmceuticals, Inc., Co/United States of America) | Injectable product |
| Dietary/Food supplements and nutraceuticals | |||
| Vitamins and minerals | Vitamin C | Liposomal Vitamin C (Hypernatura®/Romania, WeightWorld/United Kingdom, NutriFlair/United States of America, Curesupport/Netherlands), Altrient C (LivOn Labs/United States of America), Vitamin C Liposomal (Actinovo Actinovo/Germany, Laboratoire Biocyte/France) | Oral products |
| Vitamin D3 | Liposomal Vitamin D3 (Lipolife/United Kingdom, California Gold Nutrition/United States of America, Dr Mercola/United States of America), Mega-Liposomal Vitamin D3 (Aurora Nutrascience/Canada), Vitamin D3 Liposomal (Laboratoire Biocyte/France) | ||
| Zinc | UltraZin®liposomal zinc (Laboratoire Biocyte/France), Liposomal zinc (AbelaPharm/Serbia) | ||
| Minerals | Cal/Mag/Zinc Liposomal (Laboratoire Biocyte/France) | ||
| Multivitamins and minerals | Lipozomal Vegan D3 K2 Magneziu (Hypernatura/Romania), Liposomal multivitamin (GymBeam/Germany), VENTUS liposomal Omega 3, Omega 6 (Life Spirit/United States of America), LVC5 (Lipolife/United Kingdom) | ||
| Proteins | Collagen | Collagen liposomal (Actinovo/Germany, Healthydrops/North America), Collagen Zooki (YourZooki/United States of America) | |
| Glutathione | Liposomal Glutathione (Hypernatura, Lipolife), Glutathione Liposomal (ActiNovo/Germany, Laboratoire Biocyte/France), Altrient Glutathione (LivOn Labs/United States of America) | ||
| Polyphenols | Resveratrol | Liposomal Resveratrol (CureSupport, Lipolife/United Kingdom, Actinovo/Germany, Healthydrops/North America,), LLR1 Liposomal Resveratrol (Lipolife/United Kingdom) | |
| Quercetin | Quercetin Liposomal (Actinovo/Germany), Liposomal Bio Quercetin (DesBio PAO/Germany) | ||
| Antioxidant mix | Liposomal NMN (Codeage/United States of America), HistX (Lipolife/United Kingdom) | ||
| Curcumin | Micelle Liposomal Turmeric (Purality Health®/United States of America), Curcumin Lipozomal (Actinovo/Germany, Somavita®/United States of America), Liposomal curcumin (Hypernatura®/Romania, Lipolife/United Kingdom, Healthy Drops/United States of America), Liposomal Curcumin C3 LIPOSOL (SABINSA/United States of America) | ||
| Others | γ-Aminobutyric acid, melatonin, magnesium | Lipozomal Sleep Formula (Hypernatura®/Romania) | |
| MSM, glucosamine, Boswellia | Lipozomal Joint Formula (Hypernatura®/Romania) | ||
| Food products | |||
| Functional teas | Plant sterols, EGCG, choline, potassium | Cholesterol Aid (Bio-Up Mimetic Technologies Codeage/United States of America) | Oral product |
| Omega-3, CoQ10, choline, EGCG | Cardio Vitality (Bio-Up Mimetic Technologies Codeage/United States of America) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasarin, D.; Ghizdareanu, A.-I.; Enascuta, C.E.; Matei, C.B.; Bilbie, C.; Paraschiv-Palada, L.; Veres, P.-A. Coating Materials to Increase the Stability of Liposomes. Polymers 2023, 15, 782. https://doi.org/10.3390/polym15030782
Pasarin D, Ghizdareanu A-I, Enascuta CE, Matei CB, Bilbie C, Paraschiv-Palada L, Veres P-A. Coating Materials to Increase the Stability of Liposomes. Polymers. 2023; 15(3):782. https://doi.org/10.3390/polym15030782
Chicago/Turabian StylePasarin, Diana, Andra-Ionela Ghizdareanu, Cristina Emanuela Enascuta, Catalin Bogdan Matei, Catalin Bilbie, Luciana Paraschiv-Palada, and Petronela-Andreea Veres. 2023. "Coating Materials to Increase the Stability of Liposomes" Polymers 15, no. 3: 782. https://doi.org/10.3390/polym15030782
APA StylePasarin, D., Ghizdareanu, A.-I., Enascuta, C. E., Matei, C. B., Bilbie, C., Paraschiv-Palada, L., & Veres, P.-A. (2023). Coating Materials to Increase the Stability of Liposomes. Polymers, 15(3), 782. https://doi.org/10.3390/polym15030782