1. Introduction
Polymer brushes are thin films of polymer chains covalently anchored to surfaces, and was found to enhance the lubrication properties of water with ultra-low friction [
1], such as hip joint lubrication, and polymer brushes grafted to the rubbing surface could effectively reduce the friction and wear due to the hydration layer of the brush adsorbed on the substrate [
2,
3,
4,
5]. There are generally two methods for preparing polymer brushes, one of which is “grafting to the surface” (hereinafter referred to as “grafting to”), and the other which is “grafting from the surface” (hereinafter referred to as “grafting from”).
A polymer brush is grafted on the substrate by physical adsorption [
6] or chemical bonding [
7] in the preparation method of “grafting to”. It was found that a polymer brush, such as poly (L-lysine)-g-polyethylene glycol (hereinafter referred to as PLL-g-PEG), could be continuously re-grafted on the rubbing surface even though it was continuously destroyed by shear stress during the rubbing process, forming a unique “self-healing” property for achieving low friction [
8,
9]. However, since the maximum bonding density of polymer chains decreases with the increase in polymer molecular weight, the load-bearing capacity of polymer brushes is also limited [
10]. A polymer brush is achieved by surface-initiated polymerization in the preparation method of “grafting from” [
11]. Firstly, a suitable polymerization initiator is anchored to the surface of the substrate (the initiator used can be ionic or free radical), then the monomer is diffused into the initiator to produce a polymer brush through covalent bonding. Surface-initiated atom transfer radical polymerization (SI-ATRP) is viewed as an excellent polymerization technology that has developed rapidly in recent years for its various structures on many substrates made of different materials, such as silicon, glass, etc. [
12]. It should be noted that polymer brushes cannot be regenerated once sheared from the surface due to the limit of this preparation method.
Inspired by the microstructure and lubrication mechanism of natural synovial fluid joints, scientists found that even at relatively slow friction speeds (10
−2~10
−1 m/s), an extremely thin lubricating film could be formed on the surfaces grafted by a polymer brush, thus obtaining an ultra-low friction coefficient (10
−2~10
−3). Moreover, the friction coefficient does not depend on the sliding speed [
8,
13,
14]. This interesting phenomenon has attracted great attention of scientists, and for the first time, the polymer brush was introduced in the tribology research field by Klein [
15], where it was found that the normal force between the surfaces of two compressed mica surfaces grafted by a polymer layer in toluene increased significantly when the surface moved in parallel, and the friction force decreased significantly even at low sliding speeds. Since then, a series of research studies have been conducted to study tribological properties of polymer brushes. For example, Spencer et al. [
16,
17,
18] demonstrated that an extremely low friction coefficient can be achieved when added by the PLL-g-PEG polymer into the HEPES lubricant conducted on a pin-disk friction tester, and proposed that the fast “self-healing” behavior of PLL-g-PEG is mainly attributed to the reduction in friction and wear. Zhou et al. [
19] found that the polymer brush as coating exhibited good properties in pure water and different biological media by surface-initiated polymerization (SIP), and its friction coefficient could be as low as 0.015, which could have a wide range of applications in biological implants and medical devices. Wei Qiangbing et al. [
20] successfully achieved the adjustment control of friction by preparing a responsive polyelectrolyte brush with polymethacryloyloxyethyl dimethyl ammonium chloride, which could change the good lubricating condition (μ~10
−3) to an ultra-high friction condition (μ > 1). Qu et al. [
21] found that the hairy NP polymer as an additive in PAO exhibited excellent lubricating properties, which could effectively reduce the friction and wear conducted on the PlintTE-77 friction tester. Takahara et al. [
22] firstly measured the gap between the ball and disk of a point contact test apparatus using optical interferometry technology, where the glass disk was grafted by polymer, and a fluid lubricating layer was formed assisted by a polymer brush on contact, while it was not observed for bare glass.
Since the polymer brush has been successfully applied in urinary catheters as coating due to its good lubrication performance, it has greatly eased the pain of the patients. Additionally, it can be view as potential green lubrication for its reduction in friction and wear. Regrettably, the water lubrication mechanism of polymer brushes has not been understood so far, and consequently, their wide application has been limited, but it is agreed that the hydration effect of polymer brushes is the key factor. The tribological properties, such as friction and wear, have been intensively investigated, while few studies have been conducted to study the film formation and lubrication mechanism of polymer brushes considering hydrodynamics. Based on this, this study comprehensively studied the effects of polymer brushes (PLL-g-PEG and PSPMA) grafted by different preparation methods on the macro-tribological properties under an aqueous lubrication environment by measuring friction. Then, we investigated their lubrication behavior through in situ observation, and the film thickness induced by the polymer brush was measured on a laboratory set for film thickness measurement apparatus based on interference technology. The film-forming mechanism of aqueous lubrication was further revealed, which provided necessary technical support for guiding the design of polymer lubricating materials, and which was important to further explore the lubrication mechanism of the polymer brush in an aqueous lubricant and develop green lubrication.
2. Materials and Methods
2.1. Test Materials of PLL-g-PEG Brushes
The materials contained 30% hydrogen peroxide, 98% concentrated sulfuric acid, sodium hydroxide, N-(2-hydroxyethyl) piperazine-N-(-2-ethanesulfonic acid) solvent (HEPES solvent, purchased from Shanghai Yuanye Biotechnology Co., Ltd., Shanghai, China), and poly(L-Lysine)-g-poly(ethylene glycol) (PLL(20)-g(3.6)-PEG(5) solvent (purchased from Creative PEGworks Company, Durham, NC, USA)). The specific copolymer used, PLL(20)-g(3.6)-PEG(5), has a PLL molecular weight of 20 kDa, a PEG side chain molecular weight of 5 kDa, and a lysine to PEG side chain graft ratio of 3.6. The chemical reagents used in the experiment were all of analytical grade.
2.2. Test Materials of PSPMA Brushes
The materials: Dopamine hydrochloride (98%), tris(hydroxymethyl)aminomethane (THAM, 99.8%), and potassium 3-sulfonic acid propyl methacrylate (SPMA, 96%) (purchased from Aladdin Reagent (Shanghai) Co., Ltd., Shanghai, China); 2-Bromo-2-methylpropionyl bromide (98%) (purchased from Beijing Bailingwei Technology Co., Ltd., Beijing, China); cuprous bromide (CuBr) and sodium tetraborate (Na2B4O7) (purchased from Sinopharm Chemical Reagent Co., Ltd., Shanghai, China); and methanol, ethanol (99%), dichloromethane, 2,2’-bipyridine (bipy), copper chloride (CuCl2), and magnesium sulfate (MgSO4) (purchased from Tianjin Bodi Chemical Co., Ltd., Tianjin, China). The reagents were analytically pure and had not undergone further processing before use. The deionized water used was self-made in the laboratory.
2.3. Preparation of Polymer Brush PLL-g-PEG
First, 0.238 g of the HEPES solvent was dissolved in about 90 mL deionized water, diluted to 100 mL with deionized water, and the pH was adjusted to 7.4 with 1 mol/L NaOH to obtain a HEPES solution with a concentration of 10 mM. Then, we weighed the PLL-g-PEG solvent and dissolved it in a 10 mM HEPES solution, and prepared a HEPES aqueous solution with a concentration of 0.25 mg/mL of PLL-g-PEG according to the required amount of the experiment. The lubricants used in the test were the HEPES solution and the HEPES aqueous solution of PLL-g-PEG with a concentration of 0.25 mg/mL. The molecular structure of -PEG is shown in
Figure 1a.
The surface of the substrate was ultrasonically cleaned with petroleum ether, absolute ethanol, and deionized water, dried with nitrogen, then immersed in a Pirahan (H
2SO
4:H
2O
2 = 70:30,
V/
V) solution at 90 °C for 30 min. After repeated rinsing with a large amount of deionized water, it was dried with nitrogen again, then irradiated with ultraviolet for 20 min by a vacuum ultra-ultraviolet surface treatment device, thereby obtaining the surface of the silicon substrate rich in hydroxylation on the surface. The hydroxylated surface was fully immersed in the aqueous solution prepared above. Since the polymer backbone PLL has a positive charge (NH
3+) in a neutral environment, it attracted each other on the hydroxylated surface with a negative charge (OH
−) and was grafted to the substrate surface. The hydroxylated surface was fully immersed in the aqueous solution prepared above. Since the polymer backbone PLL has a positive charge (NH
3+) in a neutral environment, it attracted each other on the hydroxylated surface with a negative charge (OH
−) and was grafted onto the substrate surface. The polymer side chain PEG is easy to bind a large number of water molecules, forming a nano-thick polymer brush in a neutral environment to form a hydration layer with a good lubricating effect. During the experiment, the soaking time was 30 min to ensure sufficient grafting of the polymer brush on the surface of the substrate. The schematic diagram of the adsorption of polymer PLL-g-PEG to the surface of hydroxylated substrates in an aqueous environment is shown in
Figure 1b.
2.4. Preparation of Polymer Brush PSPMA
Using the SI-ARTP technology to graft the polymer brush PSPMA on the surface of the steel ball and the glass disk was mainly achieved through the assembly of the surface initiator and the initiation of polymerization. The preparation method is shown in
Figure 1c. Before grafting, the dopamine concentrate (DOPAMA) needed to be prepared first. For the specific synthesis process, please refer to the method reported in reference [
23]. The substrates were the surfaces of steel balls and glass disks. Before preparation, the steel balls and glass disks were immersed in petroleum ether for ultrasonic treatment for 30 min. Then, they were rinsed with ethanol, rinsed with deionized water, and finally blow-dried with nitrogen to remove surface impurities.
A certain amount of the prepared dopamine concentrate was added to ethanol and stirred at room temperature for 10 min. The steel ball was completely immersed in this solution for the assembly of the substrate surface initiator, and stored in the dark for 12 h. Then, we removed the steel ball and rinsed the surface of the substrate with a large amount of ethanol. A certain amount of monomer 3-sulfonic acid propyl methacrylate potassium salt (SPMA) was added to the mixed solution of methanol and water with a volume ratio of 1:2, and nitrogen protection was introduced and stirred at room temperature for 30 min. We added catalyst 2,2′−bipyridine (bipy) and cuprous bromide (CuBr) to it in turn, and continued stirring for 20 min under nitrogen protection until a homogeneous reddish-brown monomer solution was formed. We placed the clean steel balls or glass disks that had been assembled with the initiators into the monomer solution, removed them after 4 h of surface-initiated polymerization under nitrogen protection, then rinsed the substrate surface with a large amount of deionized water. Lastly, we blow-dried with nitrogen for use.
Due to the large size of the grafted surface, the preparation method of the glass disk surface was slightly complicated, and the assembly of the surface initiator needed to be completed in two steps. In the first step, an appropriate amount of tris buffer solution with a concentration of 10 mmol/L was prepared, and a dopamine hydrochloride solution with a concentration of 0.5 mg/mL was prepared with it. The glass plate was fully soaked in the solution for 48 h, then the glass plate was removed and rinsed with deionized water. In the second step, the glass plate was soaked in a mixed solution of 2-bromo-2-methylpropionyl bromide and dichloromethane with a volume ratio of 1:200 for 12 h, then the glass plate was removed and the surface was rinsed with ethanol. The monomer solution used in the preparation of surface-initiated polymerization was similar to the steel ball method. The difference was that the glass surface to be grafted needed to be placed on a polished zinc plate for polymerization reaction. A tiny gap was left, and the prepared monomer solution was dropped into the gap between the zinc plate and the glass plate, after which the monomer solution filled the entire gap by capillary force, and the glass plate was removed after 3 h of polymerization. The substrate surface was rinsed with plenty of deionized water, and finally blow-dried with nitrogen for use.
2.5. Test Rigs and Measurement Methods
Figure 1d is a schematic diagram of the friction tester consisting of a steel ball and 3 glass plates fixed on Anton Paar’s Physica MCR rheometer (Anton Paar Co. Ltd., Graz, Austria). The ball measurement fixture included a connecting shaft, a test ball, and a fixing device, and the 3 glass plates were fixed on a specially designed spring system to ensure that the applied force of the three plates was uniform. The diameter of the ball was 12.7 mm, the size of the plate was 15 mm × 6 mm × 3 mm (L × W × H), and they were made of GCr15 steel and K9 glass, respectively. Glass plates (Ra = 0.006 μm) were purchased from Wuxi Delmon Technology Co., Ltd., and the steel ball (Ra = 0.040 μm) was purchased from Qingdao Meike Precision Machinery Co., Ltd. Before the friction test, sufficient lubricant of about 4 mL was added to the contact area to fully submerge the ball and 3 plates. For polymer brush PLL-g-PEG, the steel ball and glass plates needed to be immersed in the mixture of HEPES and PLL-g-PEG with 0.25 mg/mL concentration followed by a wait of 30 min to ensure that their surface was adequately grafted by polymer brush PLL-g-PEG, for it was found that the contact angle did not change after 30 min during the subsequent contact angle measurement (seen in
Figure S4), which suggested that the PEG polymer brush grafted may have reached saturation and was completely grafted on the substrates. For polymer brush PSPMA, the ball and plates also needed to be completely soaked in deionized water for 5 min to ensure that the PSPMA brush grafted on the substrates could fully swollen and stretch, which was consistent with the change in contact angle with time in
Figure 2b.
The loads applied to the ball were 0.5 N and 3.5 N (corresponding to the maximum Hertz contact stress, PHertz was 217 MPa and 416 MPa), and all friction coefficients were measured starting from a velocity Ue of 1.4 m/s and successively decreasing to 0.003 m/s, which could basically cover the range from fluid dynamics to the boundary lubrication regime. The ball–plate friction pairs had to be replaced with fresh pairs for each test. Each experiment was repeated 3 times and the average value was taken.
The laboratory set for observing the contact area in situ and measuring film thickness by the optical interference method is shown in
Figure 1e. The diameter of the glass disk was 150 mm, which was made of K9 glass coated with approximately 20 nm of chromium as well as 200 nm of silica to increase its reflectivity and purchased from Shanghai Weipu Optoelectronics Technology Co., Ltd., Ra = 0.005 μm; the diameter of the steel ball was 25.4 mm and it was made of GCr15 (purchased from NSK Company, Tokyo, Japan, Ra = 0.043 μm). Two lasers, with wavelengths of 655 nm and 532 nm, were shone through the glass disk onto the contact area at the same time. The interference fringes produced due to optical phase differences of reflected light from the glass disk and the steel ball were captured via a CCD camera. Maps of lubricant film thickness in the contact area, with a thickness resolution of 1 nm, were obtained [
24].
Before the test, the ball–disk friction pair was ultrasonically cleaned with petroleum ether for more than 30 min, then the petroleum ether was cleaned with absolute ethanol, dried with nitrogen, and fixed in the specified position of the testing machine for the test.
The JC2000C1B contact angle meter was used to characterize the change in the wettability of the substrate (steel or glass) due to the grafted polymer brush, which was purchased from Shanghai Zhongchen Digital Technology Equipment Co., Ltd.
The experimental temperature was controlled at 22 ± 1 °C, and the humidity was kept at RH50 ± 2%.
4. Conclusions
(1) Two polymer brushes were prepared on the surfaces of rubbing pairs (ball-on-3 plates and ball-on-disk) by preparation methods of “grafted to” and “grafted from”, referred to as PLL-g-PEG and PSPMA, respectively, and their macro-tribology properties including friction and lubrication film thickness were compared in an aqueous lubrication environment, where it was found that PLL-g-PEG had better performance in reducing friction, while PSPMA had better lubrication performance with high load capacity.
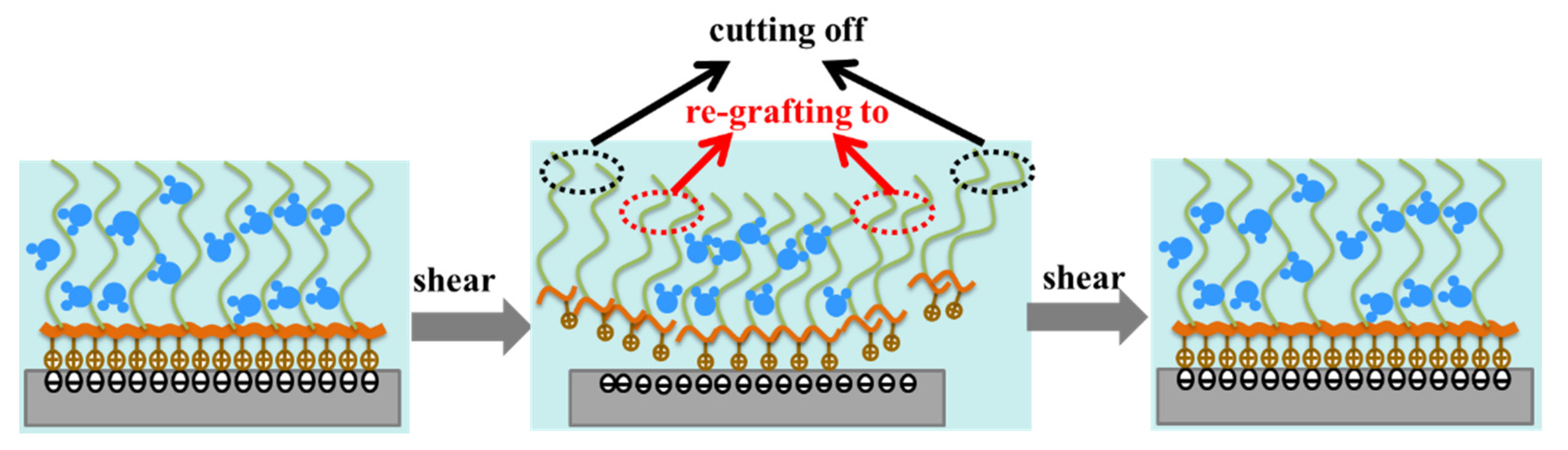
(2) It was confirmed that PLL-g-PEG has the unique property of rapid “self-healing”, which means that the polymer brush was continuously destroyed due to high shear stress and comprehensive stress during the rubbing process, while simultaneously, it could be rapidly re-grafted to the rubbing surface under the action of electrostatic force, and a dynamic equilibrium of adsorption–desorption–resorption was reached after a short period of time. It was this “self-healing” behavior that effectively reduced friction.
(3) Optical measurement results presented two different lubrication film formation mechanisms that strongly depended on the entrainment speed: it was considered to be in the thin-film lubrication regime at lower speeds (<16 mm/s), where film thickness was independent of velocity and was stable at 30~40 nm induced by the hydration effect of the polymer brush rather than the hydrodynamic effect; when velocity was greater than 32 mm/s, the film thickness and speed increased almost linearly in a logarithmic coordinate system, and the polymer brush exhibited surprising lubrication enhancement resulting from synergistic effect of the hydration effect and hydrodynamic effect.