How Can Ozone and Relative Humidity Affect Artists’ Alkyd Paints? A FT-IR and Py-GC/MS Systematic Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Weathering Experiments
2.3. Attenuated Total Reflection Fourier-Transform Infrared Spectroscopy (ATR-FTIR)
2.4. Thermally Assisted Hydrolysis and Methylation (THM)–Single Shot Pyrolysis–Gas Chromatography/Mass Spectrometry (TMH–SS-Py–GC/MS)
2.4.1. Sample Treatment, Qualitative and Quantitative Analysis
2.4.2. Apparatus and Methodology
3. Results
3.1. Pure Alkyd Resin
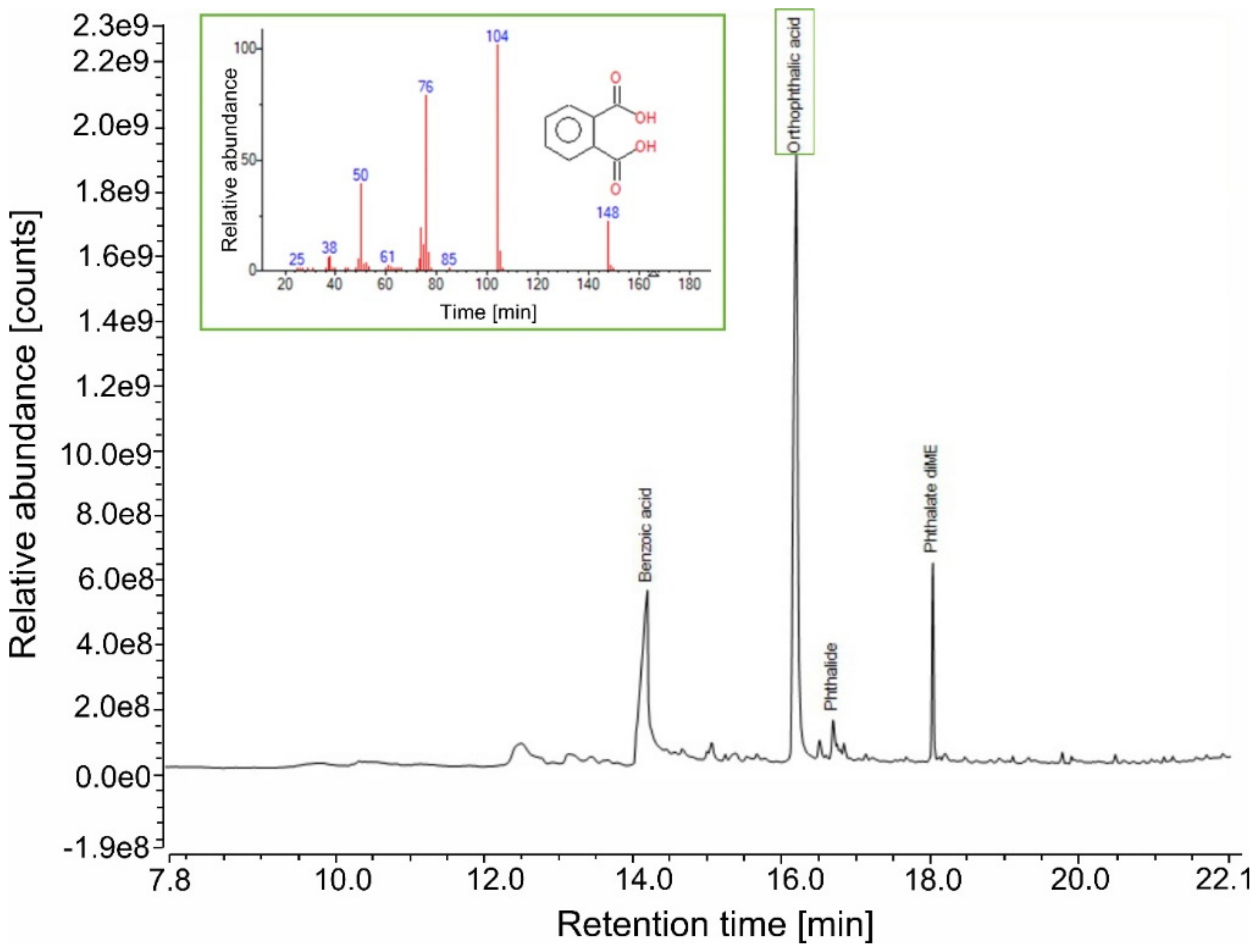
- Thanks to the detection of phthalic acid (as dimethyl ester), the polybasic acid used in alkyd manufacture was identified as orthophthalic acid (PA). To confirm its presence and exclude other polybasic acidic structures (such as phthalic anhydride, which is the most common type of dibasic acid used in alkyd paints), thermal desorption (TD) of Alk_ref was performed (see TD in Appendix A, Figure A1). This result differed from similar studies carried out on the same type of alkyd resin Medium 4 by Lukas in 2013, where the polybasic acid was found to be phthalic anhydride [6]. However, it is also known that industrial formulations may vary in composition/raw materials for their preparation, so this new variation is not entirely unexpected; in 2016 Anghelone et al. [9] revealed the presence of orthophthalic-based resin.
- The polyol was identified as pentaerythritol (PE), a five-carbon tetraol commonly used since the 1960s in the resin formulation. Upon TMAH derivatization and THM–SS-Py–GC/MS analysis, PE dissociated into di-, tri-, and tetra-PE methyl esters. Compared to previous publications on the same commercial resin [9], the % of polyol in Alk_ref film was higher. Lukas alkyd was found to contain between 8 and 21% in fresh and aged paints by Wei et al. [6], whereas in the current study, the PE% was 29% (see Table 4). As seen above, this difference was not mainly due to analytical methodologies but likely to a different blend within the commercial formulation. Moreover, other studies reported very dissimilar contents of PE (ranging from 23% to 0%) according to the blend considered and the presence of organic and inorganic pigments [31].
- The detection of saturated monocarboxylic acids (palmitic and stearic acids being the most abundant), dicarboxylic saturated acids (suberic, azelaic, sebacic acids), and unsaturated fatty acids (oleic and linoleic acids) allowed the presence of a vegetable drying oil to be identified. According to the calculated molar ratio between palmitic and stearic acids (P/S = 1.6), the oil component could be linseed oil. Nevertheless, as mentioned before, other kinds of drying or semi-drying oils might be present. The amount of oil (summing the most abundant fatty acids) of the Alk_ref was about 30%, as reported in Table 4.
- One of the most significant peaks was related to benzoic acid (BA), an aromatic monobasic acid generally present in Py–GC/MS alkyd data. Considering the relatively high % of BA (27%, Table 4), it is more likely present as a stopping agent in the commercial formulation more than a pyrolysis product of the polybasic acid.
- Several additives were detected, in particular phthalate-based compounds (such as nonyl phthalate, octyl phthalate, furfuryl hexyl phthalate) commonly added to the polymeric mixtures as plasticizers. A paint stabilizer, a UV-light absorbing piperidine compound, namely 4-methyl-2-piperidone, was identified at 6.751 min [47]. At 10.774 min, the presence of butanal dimethylhydrazone is related to its use as a paint drier [48]. Figure 2 illustrates the proposed polycondensation reaction, starting from the main compounds identified in PY–GC/MS analysis.
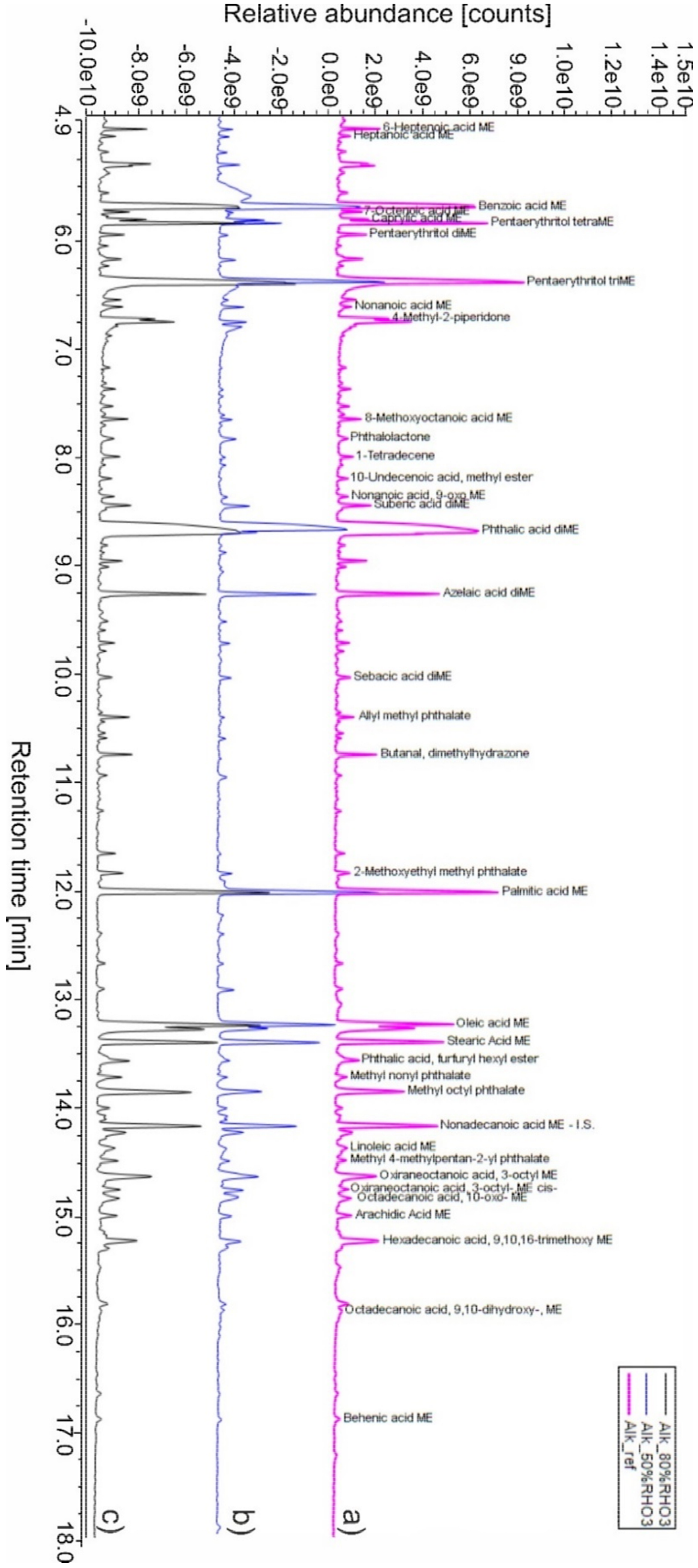
3.2. Alkyd Paints
- The addition of pigments favoured the maturity of the oil; with a specific decrease in oleic acid unsaturations, the O/S values tended to decrease compared to the pure alkyd resin. This observation was particularly evident for PG18, where the O/S value was 0.97. In other cases, such as PB29, the O/S value remained similar to the pure resin.
- Dicarboxylic fatty acids (tertiary oxidation products in the oxidative polymerization of drying oils) increased, and consequently so did the values of the molar ratios A/P and D/P, as well as %D. The %D was higher in the cases of PG50, PY37, and PW6 paints. As a result, PhA/A values slightly decreased. On the contrary, PhA/PE, BA/PE, and polyol/oil (PE/oil) ratios were quite similar to the value of the pure resin.
- In paints containing PY37, PG50, and PB29, immediately after the phthalic acid RT, a peak was detected that was not present in pure alkyd and not even in other pigments. It had a structure similar to lactones that was not identified with certainty, but that could be related to the catalysing effect of these pigments during drying and curing (Figure S1).
3.3. General Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Borgioli, L. I Leganti nell’Arte Contemporanea; Nardini Editore: Firenze, Italy, 2020. [Google Scholar]
- Learner, T.; Smithen, P.; Schilling, J.W.; Krueger, M.R. Modern Paints Uncovered, Proceedings from the Modern Paints Uncovered Symposium; Getty Publications: Los Angeles, CA, USA, 2007. [Google Scholar]
- Ploeger, R.; Chiantore, O. Characterization and Stability Issues of Artists’ Alkyd Paints. Smithson Contrib. Mus. Conserv. 2012, 3, 89–95. [Google Scholar]
- Learner, T. The Analysis of Synthetic Paints by Pyrolysis-Gas Chromatography-Mass Spectrometry. Stud. Conserv. 2001, 46, 225–241. [Google Scholar]
- Bartolozzi, G.; Marchiafava, V.; Mirabello, V.; Peruzzini, M.; Picollo, M. Chemical curing in alkyd paints: An evaluation via FT-IR and NMR spectroscopies. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 118, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Pintus, V.; Schreiner, M. A comparison study of alkyd resin used in art works by Py-GC/MS and GC/MS: The influence of aging. J. Anal. Appl. Pyrol. 2013, 104, 441–447. [Google Scholar] [CrossRef]
- Perrin, F.X.; Irigoyen, M.; Aragon, E.; Vernet, J.L. Artificial aging of acrylurethane and alkyd paints: A micro-ATR spectroscopic study. Polym. Degrad. Stab. 2000, 70, 469–475. [Google Scholar] [CrossRef]
- Pintus, V.; Wei, S.; Schreiner, M. Accelerated UV ageing studies of acrylic, alkyd, and polyvinyl acetate paints: Influence of inorganic pigments. Microchem. J. 2016, 124, 949–961. [Google Scholar] [CrossRef]
- Anghelone, M.; Jembrih-Simbürger, D.; Schreiner, M. Influence of phthalocyanine pigments on the photo-degradation of alkyd artists’ paints under different conditions of artificial solar radiation. Polym. Degrad. Stab. 2016, 134, 157–168. [Google Scholar] [CrossRef]
- Pagnin, L.; Calvini, R.; Wiesinger, R.; Schreiner, M. SO2− and NOx− initiated atmospheric degradation of polymeric films: Morphological and chemical changes, influence of relative humidity and inorganic pigments. Microchem. J. 2021, 164, 106087. [Google Scholar] [CrossRef]
- Seinfeld, J.H. Urban Air Pollution: State of the Science. Science 1989, 243, 745–752. [Google Scholar] [CrossRef]
- Wiesinger, R.; Schreiner, M.; Kleber, C. Investigations of the interactions of CO2, O3 and UV light with silver surfaces by in situ IRRAS/QCM and ex situ TOF-SIMS. Appl. Surf. Sci. 2010, 256, 2735–2741. [Google Scholar] [CrossRef]
- Cass, G.R.; Druzik, J.R.; Grosjean, D.; Nazaroff, W.W.; Whitmore, P.M.; Wittman, C.L. Protection of Works of Art from Atmospheric Ozone; The Getty Conservation Institute: Los Angeles, CA, USA, 1989. [Google Scholar]
- Camuffo, D.; Fernicola, V.; Bertolin, C. Basic Environmental Mechanisms Affecting Cultural Heritage; Nardini Editore: Firenze, Italy, 2010. [Google Scholar]
- De Santis, F.; Di Palo, V.; Allegrini, I. Determination of some atmospheric pollutants inside a museum: Relationship with the concentration outside. Sci. Total Environ. 1992, 127, 211–223. [Google Scholar] [CrossRef]
- La Nasa, J.; Degano, I.; Modugno, F.; Colombini, M.P. Alkyd paints in art: Characterization using integrated mass spectrometry. Anal. Chim. Acta 2013, 797, 64–80. [Google Scholar] [CrossRef] [PubMed]
- Learner, T.J.S. Analysis of Modern Paints; The Getty Conservation Institute: Los Angeles, CA, USA, 2005. [Google Scholar]
- Kaal, J.; Castro González, M.G.; Martínez Cortizas, A.; Prieto Martínez, M.P. Use of Thermally Assisted Hydrolysis and Methylation (THM-GC-MS) to Unravel Influence of Pottery Production and Post-Depositional Processes on the Molecular Composition of Organic Matter in Sherds from a Complex Coastal Settlement. Separations 2021, 8, 140. [Google Scholar] [CrossRef]
- Carlesi, S.; Bartolozzi, G.; Cucci, C.; Marchiafava, V.; Picollo, M.; La Nasa, J.; Di Girolamo, F.; Dilillo, M.; Modugno, F.; Degano, I.; et al. Discovering “the Italian Flag” by Fernando Melani (1907–1985). Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 168, 52–59. [Google Scholar] [CrossRef]
- Osete-Cortina, L.; Doménech-Carbó, M.T. Analytical characterization of diterpenoid resins present in pictorial varnishes using pyrolysis-gas chromatography-mass spectrometry with on line trimethylsilylation. J. Chromatogr. A 2005, 1065, 265–278. [Google Scholar] [CrossRef]
- Doménech-Carbó, M.T.; Bitossi, G.; Osete-Cortina, L.; Yusá-Marco, D.J. Study of ageing of ketone resins used as picture varnishes by pyrolysis-silylation-gas chromatography-mass spectrometry. J. Anal. Appl. Pyrol. 2009, 85, 470–479. [Google Scholar] [CrossRef]
- Bonaduce, I.; Ribechini, E.; Modugno, F.; Colombini, M.P. Analytical approaches based on gas chromatography mass spectrometry (GC/MS) to study organic materials in artworks and archaeological objects. Top Curr. Chem. 2016, 374, 291–327. [Google Scholar] [CrossRef]
- Ploeger, R.; Scalarone, D.; Chiantore, O. The characterization of commercial artists’ alkyd paints. J. Cult. Herit. 2008, 9, 412–419. [Google Scholar] [CrossRef]
- Colombini, M.P.; Andreotti, A.; Bonaduce, I.; Modugno, F.; Ribechini, E. Analytical strategies for characterizing organic paint media using gas chromatography/mass spectrometry. Acc. Chem. Res. 2010, 43, 715–727. [Google Scholar] [CrossRef]
- Silva, M.F.; Doménech-Carbó, M.T.; Fuster-López, L.; Mecklenburg, M.F.; Martin-Rey, S. Identification of additives in poly(vinylacetate) artist’s paints using PY-GC-MS. Anal. Bioanal. Chem. 2010, 397, 357–367. [Google Scholar] [CrossRef]
- Grzywacz, C.M. Monitoring for Gaseous Pollutants in Museum Environments; Getty Publications: Los Angeles, CA, USA, 2006. [Google Scholar]
- Berni, A.; Mennig, M.S.H. Doctor blades. In Sol-Gel Technologies for Glass Producers and Users; Springer: London, UK, 2004; pp. 89–92. [Google Scholar]
- European Environmental Agency. 2020. Available online: https://www.eea.europa.eu/ (accessed on 18 June 2020).
- Wiesinger, R.; Pagnin, L.; Anghelone, M.; Moretto, L.M.; Orsega, E.F.; Schreiner, M. Pigment and Binder Concentrations in Modern Paint Samples Determined by IR and Raman Spectroscopy. Angew. Chem. Int. Ed. 2018, 57, 7401–7407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagnin, L.; Calvini, R.; Wiesinger, R.; Weber, J.; Schreiner, M. Photodegradation Kinetics of Alkyd Paints: The Influence of Varying Amounts of Inorganic Pigments on the Stability of the Synthetic Binder. Front. Mater. 2020, 7, 423. [Google Scholar] [CrossRef]
- Schilling, M.R.; Keeney, J.; Learner, T. Characterization of Alkyd Paint Media By Gas Chromatography-Mass Spectrometry. Stud. Conserv. 2004, 49, 197–201. [Google Scholar] [CrossRef]
- La Nasa, J.; Degano, I.; Modugno, F.; Colombini, M.P. Industrial alkyd resins: Characterization of pentaerythritol and phthalic acid esters using integrated mass spectrometry. Rapid Commun. Mass Spectrom. 2015, 29, 225–237. [Google Scholar] [CrossRef]
- Drechsel, D.; Dettmer, K.; Engewald, W. Studies of thermally assisted hydrolysis and methylation-GC-MS of fatty acids and triglycerides using different reagents and injection systems. Chromatographia 2003, 57, S283–S289. [Google Scholar] [CrossRef]
- Yu, H.H.; Lim, J.-A.; Ham, S.W.; Lee, K.-B.; Lee, Y. Quantitative Analysis of Blended Asian Lacquers Using ToF-SIMS, Py-GC/MS and HPLC. Polymers 2020, 13, 97. [Google Scholar] [CrossRef]
- Lee, Y.S.; Lee, W.K.; Cho, S.G.; Kim, I.; Ha, C.S. Quantitative analysis of unknown compositions in ternary polymer blends: A model study on NR/SBR/BR system. J. Anal. Appl. Pyrol. 2007, 8, 85–94. [Google Scholar] [CrossRef]
- Fuster-López, L.; Izzo, F.C.; Piovesan, M.; Yusá-Marco, D.J.; Sperni, L.; Zendri, E. Study of the chemical composition and the mechanical behaviour of 20th century commercial artists’ oil paints containing manganese-based pigments. Microchem. J. 2016, 124, 962–973. [Google Scholar] [CrossRef] [Green Version]
- Fuster-López, L.; Izzo, F.C.; Andersen, C.K.; Murray, A.; Vila, A.; Picollo, M.; Stefani, L.; Jiménez, R.; Aguado-Guardiola, E. Picasso’s 1917 paint materials and their influence on the condition of four paintings. SN Appl. Sci. 2020, 2, 2159. [Google Scholar] [CrossRef]
- Fuster-López, L.; Izzo, F.C.; Damato, V.; Yusá-Marco, D.J.; Zendri, E. An insight into the mechanical properties of selected commercial oil and alkyd paint films containing cobalt blue. J. Cult. Herit. 2019, 35, 225–234. [Google Scholar] [CrossRef]
- Mills, J.; White, R. Organic Mass-Spectrometry of Art Materials: Work in Progress. Natl. Gallery Tech. Bull. 1982, 6, 3–16. [Google Scholar]
- Duce, C.; Bernazzani, L.; Bramanti, E.; Spepi, A.; Colombini, M.P.; Tiné, M.R. Alkyd artists’ paints: Do pigments affect the stability of the resin? A TG and DSC study on fast-drying oil colours. Polym. Degrad. Stab. 2014, 105, 48–58. [Google Scholar] [CrossRef]
- Caravá, S.; Roldán García, C.; Vázquez de Agredos-Pascual, M.L.; Murcia Mascarós, S.; Izzo, F.C. Investigation of modern oil paints through a physico-chemical integrated approach. Emblematic cases from Valencia, Spain. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 240, 118633. [Google Scholar] [CrossRef] [PubMed]
- Van Keulen, H.; Schilling, M. AMDIS & EXCEL: A Powerful Combination for Evaluating THM-Py-GC/MS Results from European Lacquers. Stud. Conserv. 2019, 64, S74–S80. [Google Scholar] [CrossRef]
- Ellis, G.; Claybourn, M.; Richards, S.E. The application of fourier transform raman spectroscopy to the study of paint systems. Spectrochim. Acta Part A Mol. Spectrosc. 1990, 46, 227–241. [Google Scholar] [CrossRef]
- Derrick, M.R.; Stulik, D.; Landry, J.M. Infrared Spectroscopy in Conservation Science; Getty Publications: Los Angeles, CA, USA, 1999. [Google Scholar]
- Shearer, G.L. An Evaluation of Fourier Transform Infrared Spectroscopy for the Characterization of Organic Compounds in Art and Archaeology. Food Chem. 1989, 190, 1109–1115. [Google Scholar]
- Hase, Y.; Davanzo, C.U.; Kawai, K.; Sala, O. The vibrational spectra of phthalic anhydride. J. Mol. Struct. 1976, 30, 37–44. [Google Scholar] [CrossRef]
- Bechtold, K.; Ligner, G. Stabilization of Paints. Canadian Patent 2,251,318, 23 October 1997. [Google Scholar]
- Yang, Y.; Garcia de Visicaro, J.; Sheerin, R.; Minassian, S.; Cooper, G. Aqueous Cross-Linking Compositions and Methods. U.S. Patent 9,790,374, 26 May 2015. [Google Scholar]
- Duce, C.; Della Porta, V.; Tiné, M.R.; Spepi, A.; Ghezzi, L.; Colombini, M.P.; Bramanti, E. FTIR study of ageing of fast drying oil colour (FDOC) alkyd paint replicas. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 130, 214–221. [Google Scholar] [CrossRef]
- Karis, T.E.; Miller, J.L.; Hunziker, H.E.; de Vries, M.S.; Hopper, D.A.; Nagaraj, H.S. Oxidation Chemistry of a Pentaerythritol Tetraester Oil. Tribol. Trans. 1999, 42, 431–442. [Google Scholar] [CrossRef]
- Gorkum, R.; Bouwman, E. The oxidative drying of alkyd paint catalysed by metal complexes. Coord. Chem. Rev. 2005, 249, 1709–1728. [Google Scholar] [CrossRef]
- Šťáva, V.; Veselý, D.; Kalenda, P. Catalytic effects of transition metals in the form of the salts of organic acids in the cross linking of alkyds. Pigment Resin Technol. 2008, 37, 67–72. [Google Scholar] [CrossRef]
- Pagnin, L.; Calvini, R.; Sterflinger, K.; Izzo, F.C. Data Fusion Approach to Simultaneously Evaluate the Degradation Process Caused by Ozone and Humidity on Modern Paint Materials. Polymers 2022, 14, 1787. [Google Scholar] [CrossRef]
- Korpany, K.V.; Majewski, D.D.; Chiu, C.T.; Cross, S.N.; Blum, A.S. Iron Oxide Surface Chemistry: Effect of Chemical Structure on Binding in Benzoic Acid and Catechol Derivatives. Langmuir 2017, 33, 3000–3013. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.; Hohner, C.; Mohr, S.; Libuda, J. Dissociative Adsorption of Benzoic Acid on Well-Ordered Cobalt Oxide Surfaces: Role of the Protons. J. Phys. Chem. C 2017, 121, 28317–28327. [Google Scholar] [CrossRef]
- Wiesinger, R.; Martina, I.; Kleber, C.; Schreiner, M. Influence of relative humidity and ozone on atmospheric silver corrosion. Corros. Sci. 2013, 77, 69–76. [Google Scholar] [CrossRef]
- Phipps, P.B.P.; Rice, D.W. The Role of Water in Atmospheric Corrosion. Corros. Chem. 1979, 89, 235–261. [Google Scholar]

| Binder | Pigments | Chemical Composition * | Colour Index (C.I.) Number |
|---|---|---|---|
| Alkyd resin (Alk) Commercial name: Alkyd Medium 4 | Polymer oil-modified polyester-resin | ||
| Titanium white | TiO2 | PW6 | |
| Cadmium yellow | CdS | PY37 | |
| Cobalt green | Co2TiO4 | PG50 | |
| Hydrated chromium oxide green | Cr2O3 · 2H2O | PG18 | |
| Cerulean blue | CoSnO3 | PB35 | |
| Cobalt blue | CoO · Al2O3 | PB28 | |
| Artificial ultramarine blue | Na8−10Al6Si6O29S2−4 | PB29 | |
| Iron oxide red | Fe2O3 | PR101 | |
| Manganese violet | NH4MnP2O7 | PV16 |
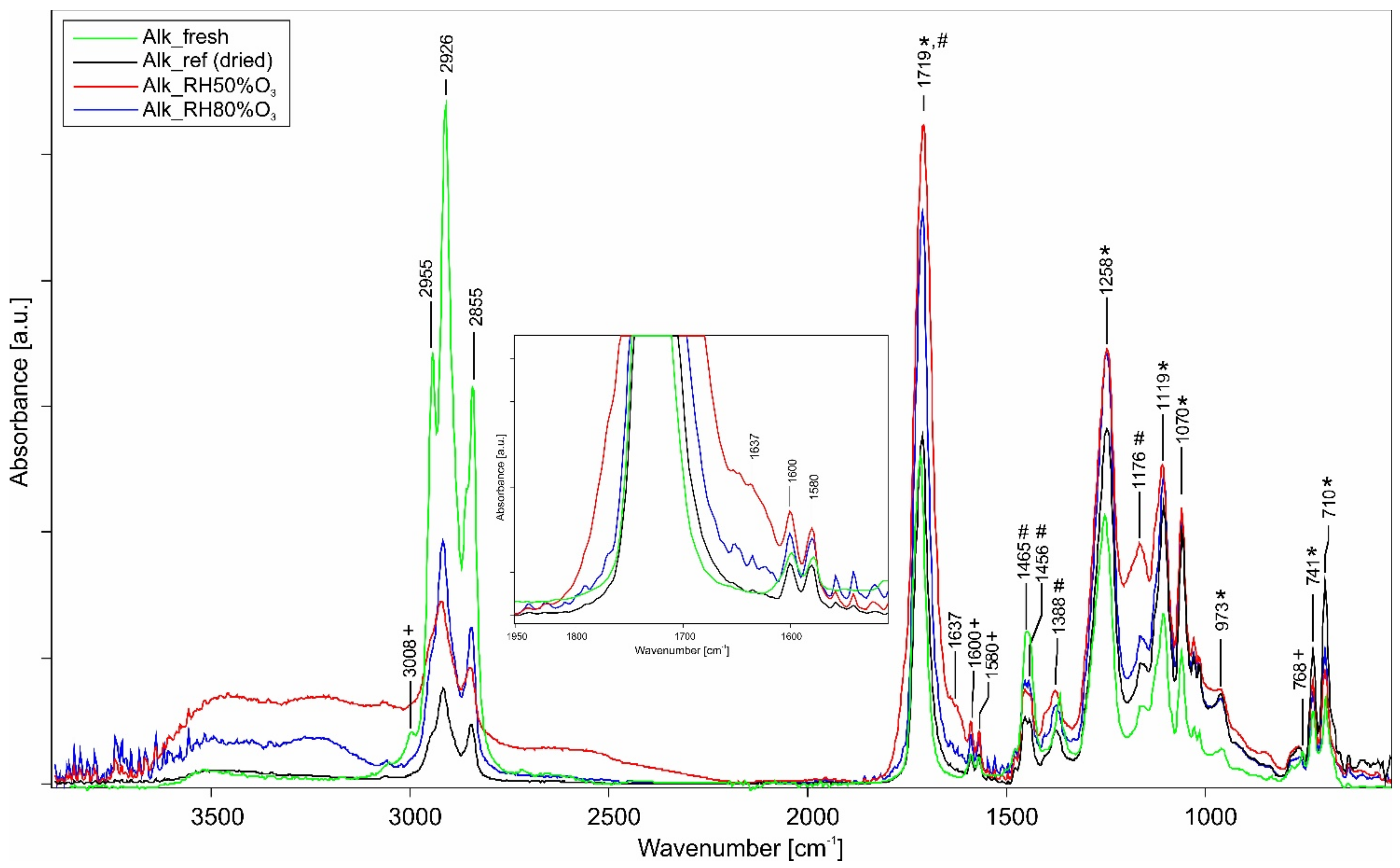
| Material | Wavenumber (cm−1) | Absorption Band | Assignment |
|---|---|---|---|
| Alkyd resin | 3008 | Vinyl proton of C–H stretching | Phthalate plasticizer |
| 2926–2855 | C–H stretching (sym–asym) | ||
| 1719 | C=O stretching | Oil and phthalic-based compound | |
| 1600–1580 | Aromatic ring C=C stretching | Phthalate plasticizer | |
| 1465–1453 | CH2 and CH3 bending (asym) | Oil | |
| 1388 | CH3 bending (sym) | Oil | |
| 1258 | C–O–C stretching (sym) | Phthalic-based compound | |
| 1176 | C–O stretching | Oil | |
| 1119 | C–O stretching (sym) | Phthalic-based compound | |
| 1071 | C–O stretching | Phthalic-based compound | |
| 973 | Out-of-plane CH deformation | Phthalic-based compound | |
| 768 | Aromatic C–H out-of-plane bending | Phthalate plasticizer | |
| 741–711 | Aromatic C–H out-of-plane bending | Phthalic-based compound | |
| Titanium white (PW6) | 606–546 | TiO2 vibrations | |
| Cadmium yellow (PY37) | / | Below detector cut-off | |
| Cobalt green (PG50) | 602 | Co–O vibrations | |
| Hydrated chromium oxide green (PG18) | 546–484 | Cr–O vibrations | |
| Cerulean blue (PB35) | 553 | Co–O vibrations | |
| Cobalt blue (PB28) | 641–553–486 | Al–O and Co–O vibrations | |
| Artificial ultramarine blue (PB29) | 1024–976 | Al,Si–O4 asymmetric stretching | |
| Iron oxide red (PR101) | 544–481 | Fe–O vibrations | |
| Manganese violet (PV16) | 3213–3068 | O–H in the mineral | |
| 1416 | [PO4]3– vibrations | ||
| 1032–995–905 | P–O stretching (asym) | ||
| 638–591–564–490 | O–P–O bending |
| Retention Time (min) | Compounds * | M+ (m/z) | Origin § |
|---|---|---|---|
| 4.999 | 6-Heptenoic acid ME | 142 (74, 41, 43) | Monobasic acids (oil) |
| 5.067 | Heptanoic acid ME | 144 (74, 87, 43) | Monobasic acids (oil) |
| 5.724 | Benzoic acid ME | 136 (105, 77, 51) | Stopping agent (BA) |
| 5.768 | 7-Octenoic acid ME | 156 (55, 74, 43) | Monobasic acids (oil) |
| 5.836 | Caprylic acid ME | 158 (74, 87, 43) | Monobasic acids (oil) |
| 5.870 | Pentaerythritol tetraME | 128 (75, 45, 71) | Polyol (PE) |
| 5.975 | Pentaerythritol diME | 131 (45, 71, 99) | Polyol (PE) |
| 6.418 | Pentaerythritol triME | 178 (45,75, 71) | Polyol (PE) |
| 6.642 | Nonanoic acid ME | 172 (74, 87, 55) | Monobasic acids (oil) |
| 6.751 | 4-Methyl-2-piperidone | 113 (42, 55, 69) | Additive (paint stabiliser) |
| 7.679 | 8-Methoxyoctanoic acid ME | 188 (45, 74, 124) | Monobasic acids (oil) |
| 7.856 | Phthalo lactone | 134 (105, 77, 134) | Additive |
| 8.026 | 1-Tetradecene | 196 (43, 55, 57) | Additive |
| 8.227 | 10-Undecenoic acid ME | 198 (74, 55, 87) | Monobasic acids (oil) |
| 8.394 | Nonanoic acid, 9-oxo ME | 186 (74, 87, 55) | Monobasic acids (oil) |
| 8.475 | Suberic acid diME | 202 (129, 138, 74) | Monobasic acids (oil) |
| 8.710 | Phthalic acid diME | 194 (163, 77, 76) | Polybasic acid (PA) |
| 9.291 | Azelaic acid diME | 216 (152, 55, 74) | Monobasic acids (oil) |
| 10.064 | Sebacic acid diME | 230 (55, 74, 125) | Monobasic acids (oil) |
| 10.427 | Allyl methyl phthalate | 220 (163, 164, 104) | Additive |
| 10.774 | Butanal, dimethylhydrazone | 114 (44, 85, 42) | Additive |
| 11.866 | 2-Methoxyethyl methyl phthalate | 238 (163, 58, 77) | Additive (plasticiser) |
| 12.046 | Palmitic acid ME | 270 (74, 87, 43) | Monobasic acids (oil) |
| 13.264 | Oleic acid ME | 296 (55, 69, 74) | Monobasic acids (oil) |
| 13.427 | Stearic Acid ME | 298 (74, 87, 43) | Monobasic acids (oil) |
| 13.594 | Phthalic acid, furfuryl hexyl ester | 334 (71, 149, 84) | Additive (plasticiser) |
| 13.747 | Methyl nonyl phthalate | 306 (163, 149, 181) | Additive (plasticiser) |
| 13.883 | Methyl octyl phthalate | 292 (163, 149, 181) | Additive (plasticiser) |
| 14.203 | Nonadecanoic acid ME I.S. | 312 (74, 87, 43) | Internal standard |
| 14.403 | Linoleic acid ME | 294 (67, 81, 95) | Monobasic acids (oil) |
| 14.516 | Methyl 4-methylpentan-2-yl phthalate | 264 (163, 149, 181) | Additive (plasticiser) |
| 14.665 | Oxiraneoctanoic acid, 3-octyl ME | 312 (155, 55, 41) | Monobasic acids (oil) |
| 14.784 | Oxiraneoctanoic acid, 3-octyl-, ME, cis- | 312 (55, 74, 155) | Monobasic acids (oil) |
| 14.869 | Octadecanoic acid, 10-oxo- ME | 312 (55, 43, 57) | Monobasic acids (oil) |
| 15.029 | Arachidic Acid ME | 326 (74, 87, 43) | Monobasic acids (oil) |
| 15.264 | Hexadecanoic acid, 9,10,16-trimethoxy ME | 360 (71, 95, 201) | Monobasic acids (oil) |
| 15.907 | Octadecanoic acid, 9,10-dihydroxy-, ME | 330 (155, 55, 41) | Monobasic acids (oil) |
| 16.907 | Behenic acid ME | 354 (74, 87, 43) | Monobasic acids (oil) |
| Alk_Ref | Alk_50%RHO3 | Alk_80%RHO3 | |
|---|---|---|---|
| Palmitic acid | 9.01 | 11.66 | 9.12 |
| Stearic acid | 5.41 | 6.92 | 5.52 |
| Azelaic acid | 5.71 | 6.78 | 5.46 |
| Suberic acid | 1.19 | 2.73 | 1.15 |
| Sebacic acid | 0.67 | 0.72 | 0.67 |
| Linoleic acid | 0.36 | 0.94 | 0.98 |
| Oleic acid | 7.49 | 7.84 | 8.73 |
| Pentaerythritol (di-, tri-, tetra-) | 29.52 | 17.42 | 27.28 |
| Phthalic acid diME | 27.45 | 25.17 | 27.35 |
| Benzoic acid ME | 13.17 | 19.83 | 13.74 |
| Fatty Acids from Oil | Polyol/Oil | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P/S | A/P | D/P | O/S | %D | PhA/P | PhA/A | BA/P | PhA/PE | BA/PE | PE/Oil | |||
| Pure alkyd resin | Alk_ref | 1.64 | 0.46 | 0.61 | 1.96 | 17.64 | 3.05 | 4.80 | 1.46 | 0.93 | 0.45 | 0.99 | |
| Alk_50%RHO3 | 1.66 | 0.42 | 0.64 | 1.60 | 19.25 | 2.20 | 3.72 | 1.70 | 1.45 | 1.14 | 0.46 | ||
| Alk_80%RHO3 | 1.63 | 0.44 | 0.58 | 2.24 | 15.66 | 3.00 | 5.01 | 1.51 | 1.00 | 0.50 | 0.86 | ||
| Alkyd paints | Ref. | PV16 | 1.69 ± 0.28 | 0.49 | 0.65 | 1.68 | 18.38 | 3.31 | 4.11 | 1.55 | 1.08 | 0.50 | 1.15 |
| PR101 | 0.40 | 0.64 | 1.63 | 22.93 | 2.98 | 5.14 | 1.72 | 1.11 | 0.64 | 1.06 | |||
| PB29 | 0.42 | 0.71 | 1.90 | 16.80 | 1.64 | 2.83 | 1.34 | 0.36 | 0.30 | 1.55 | |||
| PB35 | 0.49 | 0.65 | 1.57 | 19.18 | 2.41 | 5.09 | 1.44 | 0.85 | 0.51 | 1.12 | |||
| PB28 | 0.50 | 0.62 | 1.82 | 18.12 | 2.31 | 4.27 | 1.63 | 0.86 | 0.61 | 1.07 | |||
| PG18 | 0.49 | 0.61 | 0.97 | 19.99 | 2.25 | 4.29 | 1.76 | 0.77 | 0.60 | 1.33 | |||
| PG50 | 0.67 | 0.95 | 1.89 | 27.45 | 2.99 | 3.27 | 2.00 | 0.68 | 0.45 | 1.26 | |||
| PY37 | 0.77 | 0.74 | 1.76 | 20.57 | 1.35 | 2.09 | 1.57 | 0.35 | 0.41 | 1.26 | |||
| PW6 | 0.52 | 0.67 | 1.36 | 20.18 | 1.94 | 4.45 | 2.09 | 0.93 | 1.01 | 0.74 | |||
| 50%RHO3 | PV16 | 1.66 ± 0.32 | 0.63 | 0.73 | 0.86 | 25.89 | 2.47 | 2.84 | 2.07 | 1.76 | 1.12 | 0.52 | |
| PR101 | 0.85 | 0.85 | 1.30 | 28.22 | 3.52 | 3.00 | 3.32 | 1.43 | 1.43 | 0.47 | |||
| PB29 | 0.63 | 0.70 | 1.40 | 23.27 | 3.64 | 4.21 | 4.08 | 1.34 | 1.62 | 0.39 | |||
| PB35 | 0.52 | 0.60 | 1.44 | 20.27 | 1.51 | 2.60 | 1.25 | 1.26 | 1.15 | 0.43 | |||
| PB28 | 0.48 | 0.58 | 0.54 | 18.23 | 2.91 | 4.44 | 2.38 | 1.41 | 1.62 | 0.51 | |||
| PG18 | 0.55 | 0.72 | 1.01 | 26.68 | 1.65 | 4.23 | 1.45 | 1.24 | 1.17 | 0.53 | |||
| PG50 | 0.70 | 0.82 | 1.59 | 29.45 | 2.23 | 3.52 | 1.90 | 1.32 | 1.32 | 0.45 | |||
| PY37 | 0.50 | 0.71 | 1.35 | 23.12 | 3.15 | 4.56 | 2.09 | 1.47 | 1.19 | 0.49 | |||
| PW6 | 0.60 | 0.70 | 0.51 | 24.64 | 2.38 | 2.89 | 0.52 | 1.39 | 1.23 | 0.50 | |||
| 80%RHO3 | PV16 | 1.63 ± 0.21 | 0.61 | 0.72 | 2.05 | 19.71 | 3.06 | 4.39 | 1.58 | 1.14 | 0,59 | 0,76 | |
| PR101 | 0.43 | 0.68 | 1.70 | 16.52 | 2.38 | 2.89 | 0.52 | 4.35 | 2.2 | 0.29 | |||
| PB29 | 0.52 | 0.67 | 2.04 | 14.95 | 2.82 | 6.35 | 1.63 | 1.55 | 0.90 | 0.64 | |||
| PB35 | 0.54 | 0.76 | 1.94 | 21.25 | 2.20 | 3.66 | 0.86 | 1.24 | 0.48 | 0.56 | |||
| PB28 | 0.60 | 0.76 | 2.01 | 21.49 | 2.82 | 4.11 | 1.48 | 0.99 | 0.52 | 0.79 | |||
| PG18 | 0.49 | 0.64 | 0.93 | 22.98 | 2.08 | 3.92 | 1.61 | 1.47 | 1.14 | 0.54 | |||
| PG50 | 0.54 | 0.71 | 2.13 | 19.26 | 2.85 | 4.67 | 1.45 | 1.41 | 0.71 | 0.61 | |||
| PY37 | 05 | 0.54 | 1.74 | 15.06 | 1.99 | 4.90 | 1.99 | 0.95 | 0.96 | 0.84 | |||
| PW6 | 0.56 | 0.66 | 1.85 | 20.97 | 2.53 | 3.98 | 2.17 | 0.87 | 0.74 | 1.06 | |||
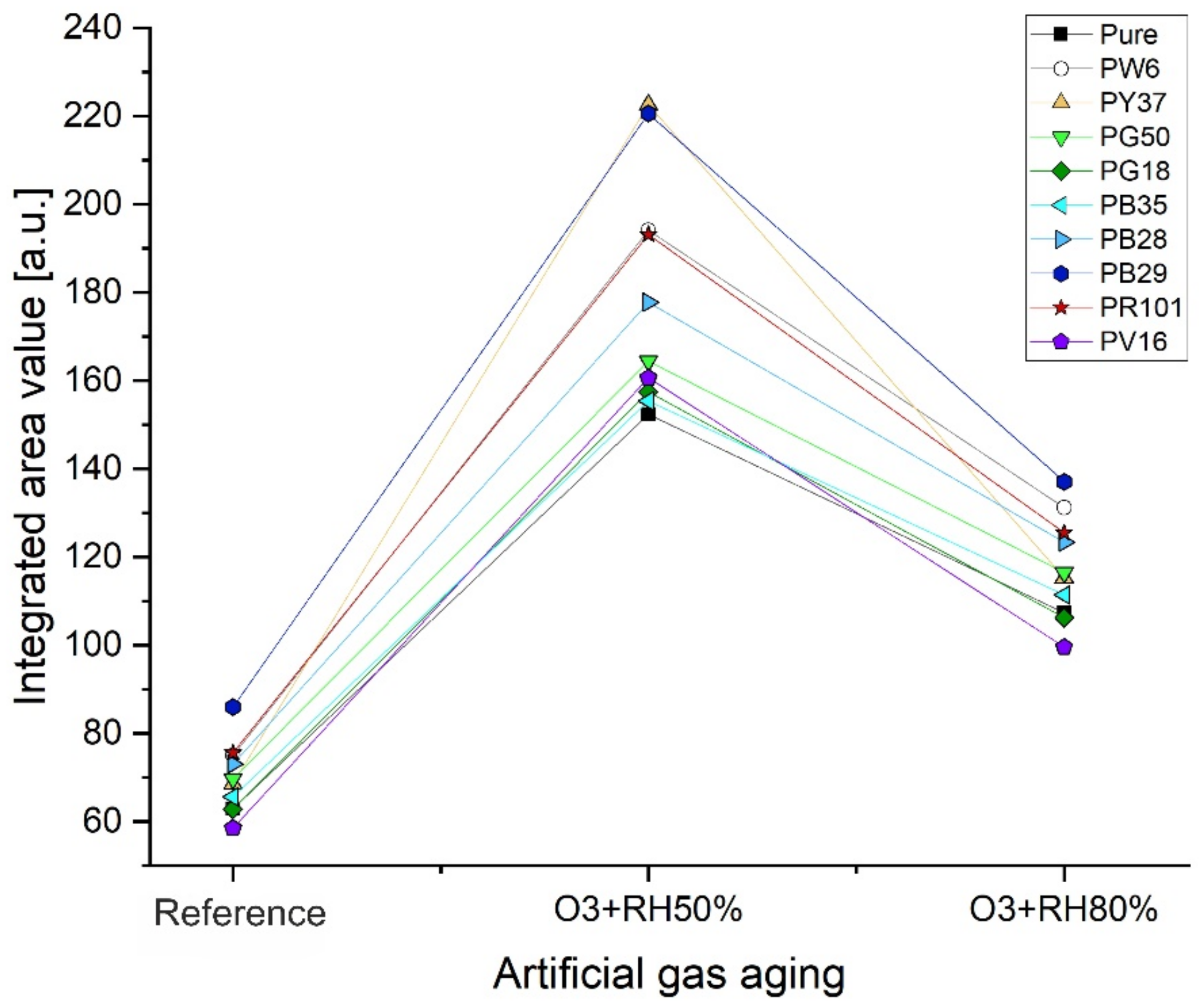
| Reference | O3 + 50%RH | O3 + 80%RH | |
|---|---|---|---|
| Pure alkyd | 62.9 | 152.3 | 107.2 |
| Alk_PW6 | 74.9 | 194.1 | 131.1 |
| Alk_PY37 | 68.6 | 222.6 | 115.3 |
| Alk_PG50 | 69.6 | 164.4 | 116.4 |
| Alk_PG18 | 62.8 | 157.4 | 106.2 |
| Alk_PB35 | 65.6 | 155.4 | 111.4 |
| Alk_PB28 | 73.1 | 177.7 | 123.3 |
| Alk_PB29 | 85.9 | 220.5 | 137.1 |
| Alk_PR101 | 75.6 | 193.1 | 125.4 |
| Alk_PV16 | 58.5 | 160.6 | 99.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pagnin, L.; Zendri, E.; Izzo, F.C. How Can Ozone and Relative Humidity Affect Artists’ Alkyd Paints? A FT-IR and Py-GC/MS Systematic Study. Polymers 2022, 14, 1831. https://doi.org/10.3390/polym14091831
Pagnin L, Zendri E, Izzo FC. How Can Ozone and Relative Humidity Affect Artists’ Alkyd Paints? A FT-IR and Py-GC/MS Systematic Study. Polymers. 2022; 14(9):1831. https://doi.org/10.3390/polym14091831
Chicago/Turabian StylePagnin, Laura, Elisabetta Zendri, and Francesca Caterina Izzo. 2022. "How Can Ozone and Relative Humidity Affect Artists’ Alkyd Paints? A FT-IR and Py-GC/MS Systematic Study" Polymers 14, no. 9: 1831. https://doi.org/10.3390/polym14091831
APA StylePagnin, L., Zendri, E., & Izzo, F. C. (2022). How Can Ozone and Relative Humidity Affect Artists’ Alkyd Paints? A FT-IR and Py-GC/MS Systematic Study. Polymers, 14(9), 1831. https://doi.org/10.3390/polym14091831