Gelatin/Chitosan Films Incorporated with Curcumin Based on Photodynamic Inactivation Technology for Antibacterial Food Packaging
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Bacterial Strains
2.2. Films Preparation
2.3. Characterizations of the Composite Films
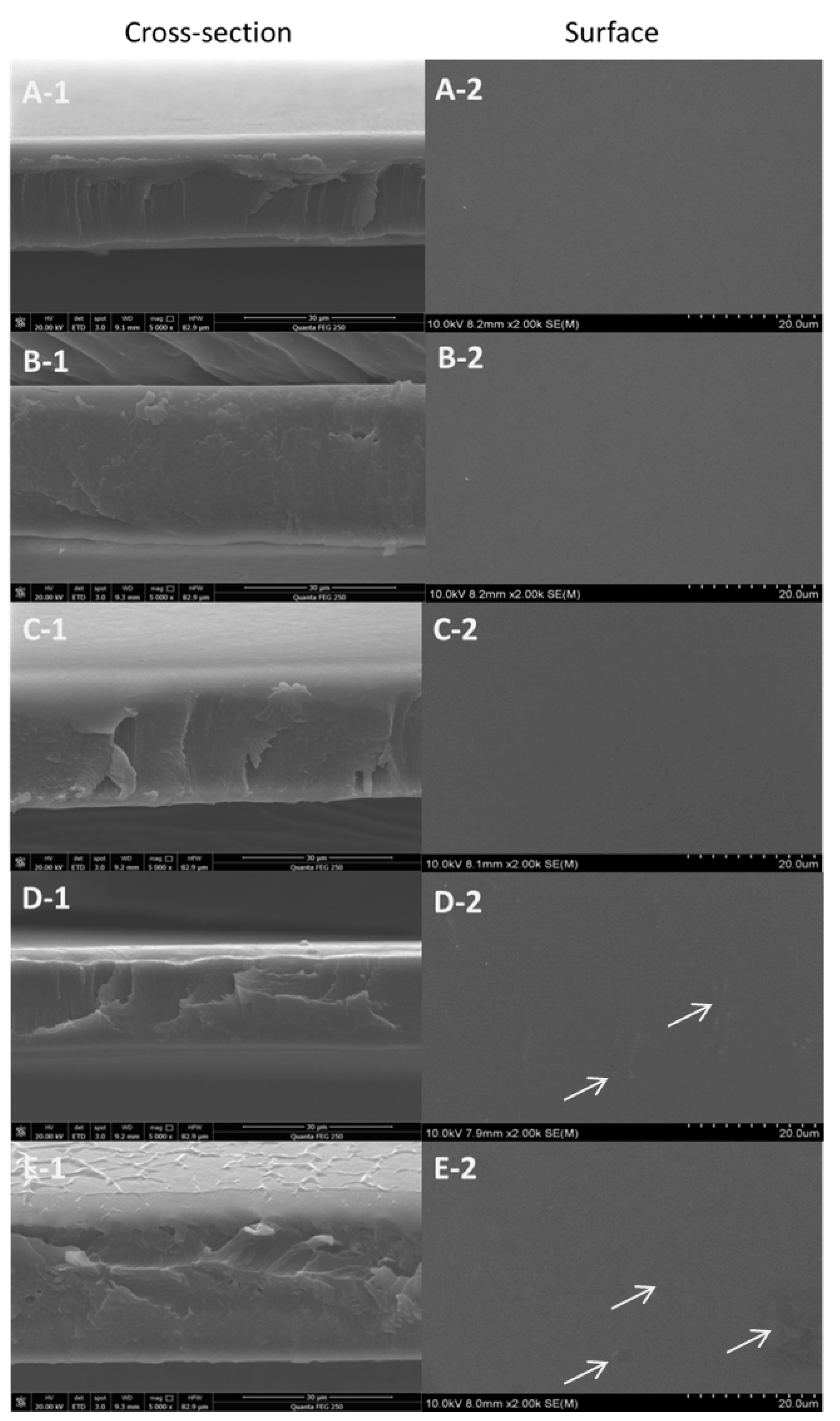
2.3.1. Scanning Electron Microscopy (SEM)
2.3.2. Color
2.3.3. UV–Visible Spectra
2.3.4. Thickness and Mechanical Properties
2.3.5. Moisture Content (MC), Water Solubility (WS), and Water Vapor Permeability (WVP)
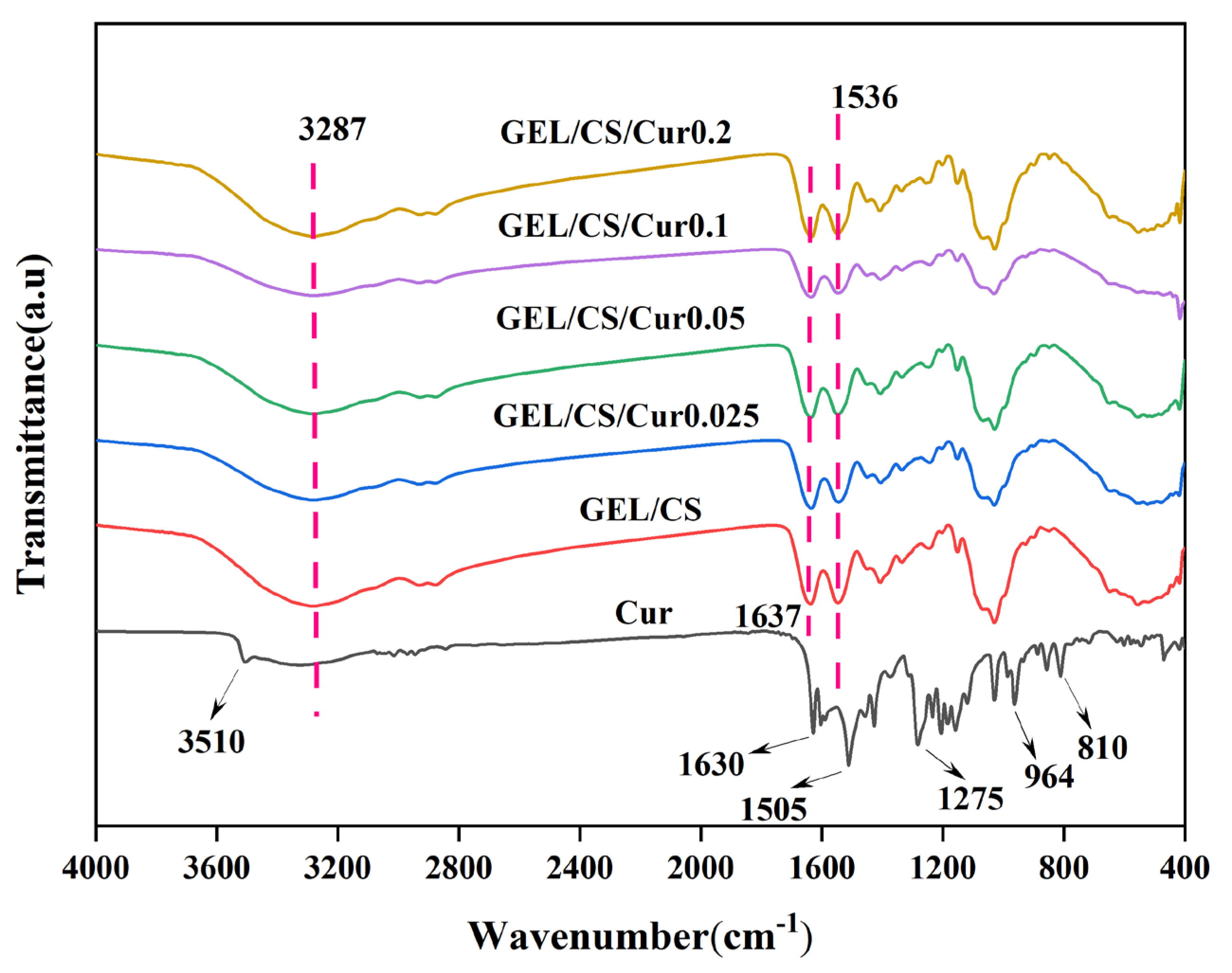
2.3.6. Fourier-Transform Infrared (FTIR) Spectroscopy
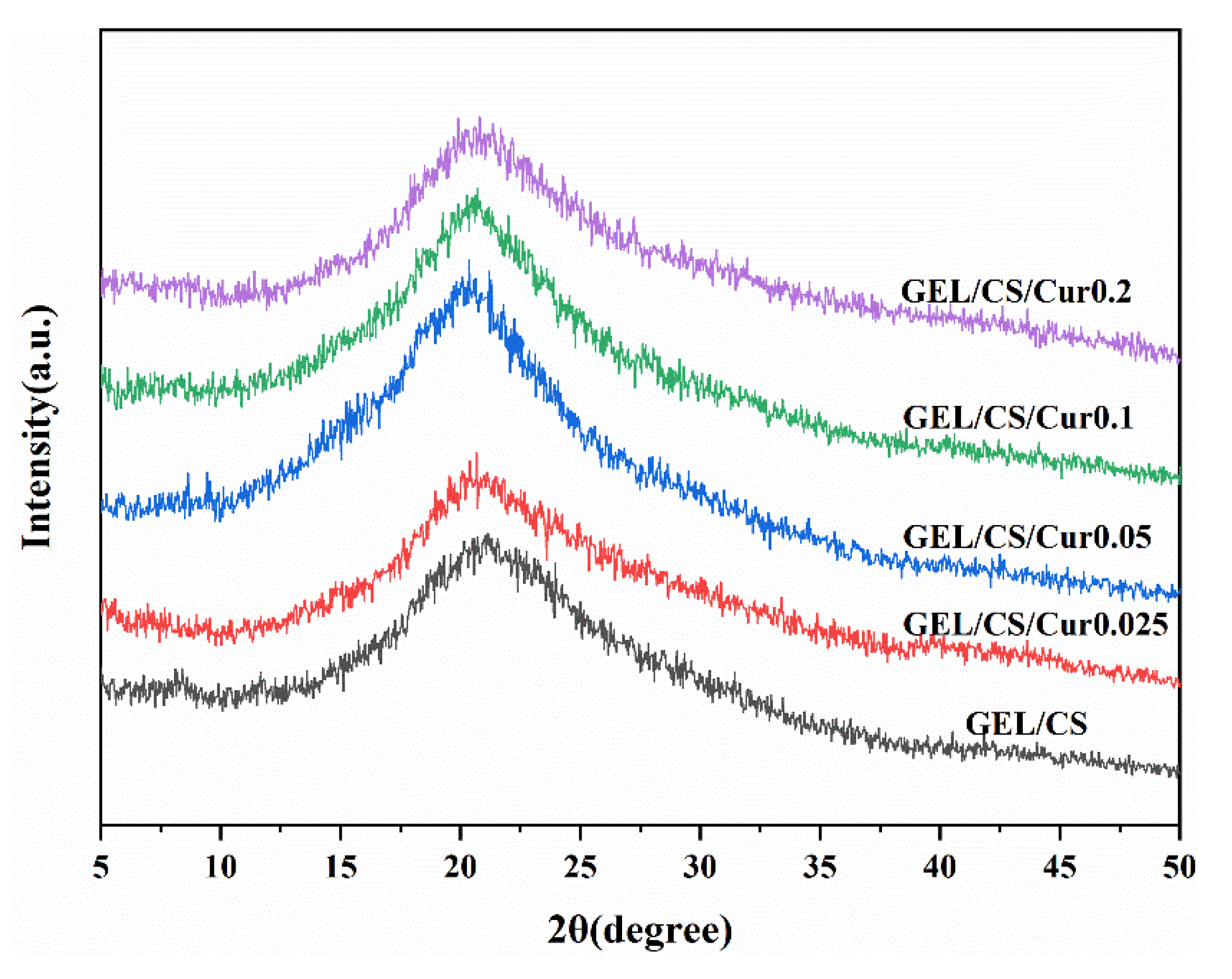
2.3.7. X-Ray Diffraction (XRD)
2.3.8. Thermogravimetric Analysis (TGA)
2.4. Photodynamic Inactivation of the Composite Films
2.4.1. Light-Emitting Diodes (LEDs) System
2.4.2. Antimicrobial Activity
2.5. Statistical Analysis
3. Results and Discussion
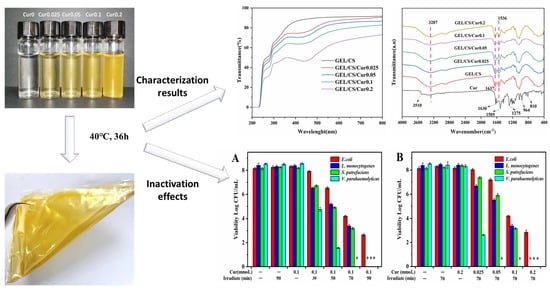
3.1. Optical Properties of Films
3.2. Microstructure of Films
3.3. Mechanical Properties
3.4. Moisture Content (MC), Water Solubility (WS), and Water Vapor Permeability (WVP)
3.5. Physicochemical and Structural Properties of Films
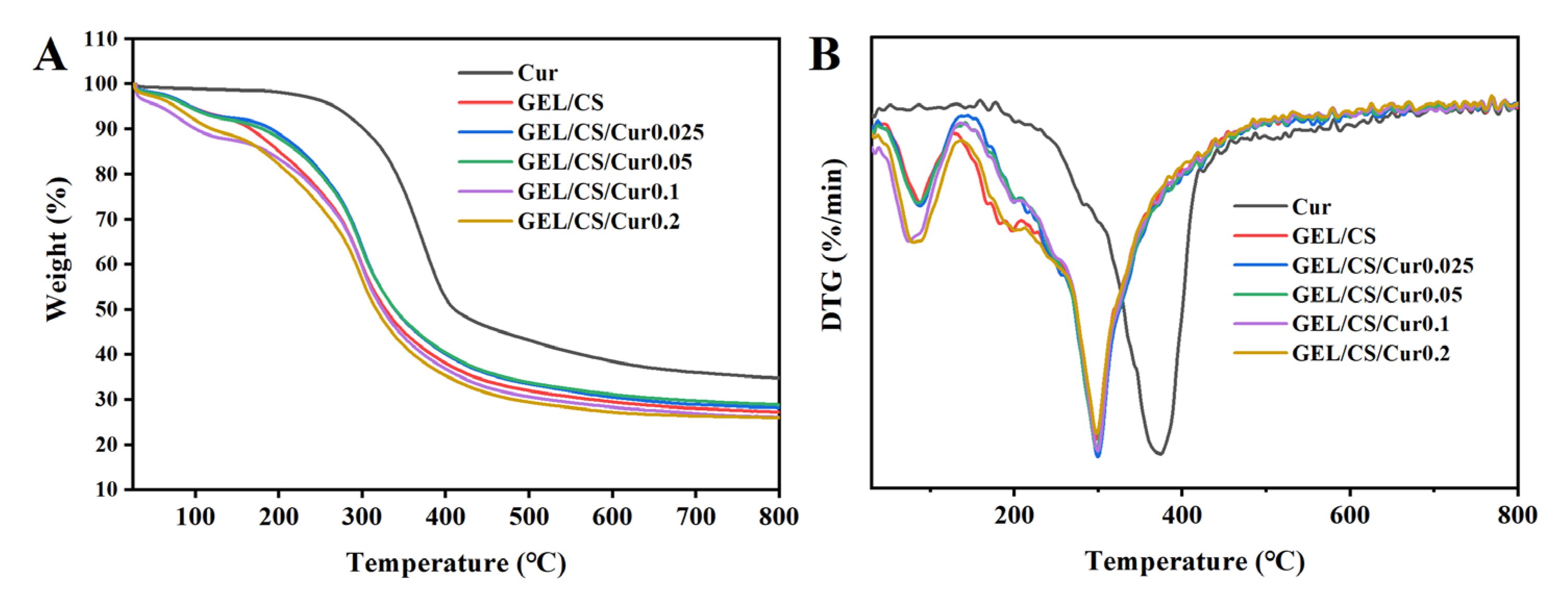
3.6. Thermal Properties
3.7. In Vitro Antimicrobial Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sutherland, W.J.; Aveling, R.; Bennun, L.; Chapman, E.; Clout, M.; Côté, I.M.; Depledge, M.H.; Dicks, L.V.; Dobson, A.P.; Fellman, L.; et al. A horizon scan of global conservation issues for 2012. Trends Ecol. Evol. 2012, 27, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Beikzadeh, S.; Khezerlou, A.; Jafari, S.M.; Pilevar, Z.; Mortazavian, A.M. Seed mucilages as the functional ingredients for biodegradable films and edible coatings in the food industry. Adv. Colloid Interface Sci. 2020, 280, 102164. [Google Scholar] [CrossRef] [PubMed]
- Espitia, P.J.P.; Du, W.-X.; Avena-Bustillos, R.D.J.; Soares, N.D.F.F.; McHugh, T.H. Edible films from pectin: Physical-mechanical and antimicrobial properties—A review. Food Hydrocoll. 2014, 35, 287–296. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.Z.; Ramakrishna, S.; Lim, C.T. Electrospinning and mechanical characterization of gelatin nanofibers. Polymer 2004, 45, 5361–5368. [Google Scholar] [CrossRef]
- Ma, L.; Zhang, M.; Bhandari, B.; Gao, Z. Recent developments in novel shelf life extension technologies of fresh-cut fruits and vegetables. Trends Food Sci. Technol. 2017, 64, 23–38. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, M.Á.V.; Marangon, C.A.; Martins, V.d.C.A.; Plepis, A.M.D.G. Chitosan/gelatin films with jatobá resin: Control of properties by vegetal resin inclusion and degree of acetylation modification. Int. J. Biol. Macromol. 2021, 182, 1737–1745. [Google Scholar] [CrossRef]
- Nuvoli, L.; Conte, P.; Fadda, C.; Reglero Ruiz, J.A.; García, J.M.; Baldino, S.; Mannu, A. Structural, thermal, and mechanical properties of gelatin-based films integrated with tara gum. Polymer 2021, 214, 123244. [Google Scholar] [CrossRef]
- Łupina, K.; Kowalczyk, D.; Lis, M.; Raszkowska-Kaczor, A.; Drozłowska, E. Controlled release of water-soluble astaxanthin from carboxymethyl cellulose/gelatin and octenyl succinic anhydride starch/gelatin blend films. Food Hydrocoll. 2022, 123, 107179. [Google Scholar] [CrossRef]
- Cheng, Y.; Gao, S.; Wang, W.; Hou, H.; Lim, L.-T. Low temperature extrusion blown ε-polylysine hydrochloride-loaded starch/gelatin edible antimicrobial films. Carbohydr. Polym. 2022, 278, 118990. [Google Scholar] [CrossRef]
- Ahmadi, S.; Hivechi, A.; Bahrami, S.H.; Milan, P.B.; Ashraf, S.S. Cinnamon extract loaded electrospun chitosan/gelatin membrane with antibacterial activity. Int. J. Biol. Macromol. 2021, 173, 580–590. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Sarbon, N.M. A comparative study: Physical, mechanical and antibacterial properties of bio-composite gelatin films as influenced by chitosan and zinc oxide nanoparticles incorporation. Food Biosci. 2021, 43, 101250. [Google Scholar] [CrossRef]
- Zhao, J.; Wei, F.; Xu, W.; Han, X. Enhanced antibacterial performance of gelatin/chitosan film containing capsaicin loaded MOFs for food packaging. Appl. Surf. Sci. 2020, 510, 145418. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, Z.; Shi, J.; Zou, X.; Zhai, X.; Huang, X.; Li, Z.; Holmes, M.; Daglia, M.; Xiao, J. Physical properties and bioactivities of chitosan/gelatin-based films loaded with tannic acid and its application on the preservation of fresh-cut apples. LWT 2021, 144, 111223. [Google Scholar] [CrossRef]
- Miao, Q.; Mi, Y.; Cui, J.; Zhang, J.; Tan, W.; Li, Q.; Guo, Z. Determination of chitosan content with Schiff base method and HPLC. Int. J. Biol. Macromol. 2021, 182, 1537–1542. [Google Scholar] [CrossRef]
- Kumar, D.; Kumar, P.; Pandey, J. Binary grafted chitosan film: Synthesis, characterization, antibacterial activity and prospects for food packaging. Int. J. Biol. Macromol. 2018, 115, 341–348. [Google Scholar] [CrossRef]
- Yi, E.; Yang, C.K.; Leem, C.; Park, Y.; Chang, J.-E.; Cho, S.; Jheon, S. Clinical outcome of photodynamic therapy in esophageal squamous cell carcinoma. J. Photochem. Photobiol. B Biol. 2014, 141, 20–25. [Google Scholar] [CrossRef]
- Machado, F.C.; de Matos, R.P.A.; Primo, F.L.; Tedesco, A.; Rahal, P.; Calmon, M.F. Effect of curcumin-nanoemulsion associated with photodynamic therapy in breast adenocarcinoma cell line. Bioorg. Med. Chem. 2019, 27, 1882–1890. [Google Scholar] [CrossRef]
- Paiva, A.d.C.M.d.; Ferreira, M.D.C.; Fonseca, A.D.S.D. Photodynamic therapy for treatment of bacterial keratitis. Photodiagn. Photodyn. Ther. 2022, 37, 102717. [Google Scholar] [CrossRef]
- Li, X.; Farid, M. A review on recent development in non-conventional food sterilization technologies. J. Food Eng. 2016, 182, 33–45. [Google Scholar] [CrossRef]
- Su, J.; Cavaco-Paulo, A. Effect of ultrasound on protein functionality. Ultrason. Sonochem. 2021, 76, 105653. [Google Scholar] [CrossRef]
- Luksiene, Z.; Zukauskas, A. Prospects of photosensitization in control of pathogenic and harmful micro-organisms. J. Appl. Microbiol. 2009, 107, 1415–1424. [Google Scholar] [CrossRef] [PubMed]
- Broekgaarden, M.; Weijer, R.; Van Gulik, T.M.; Hamblin, M.R.; Heger, M. Tumor cell survival pathways activated by photodynamic therapy: A molecular basis for pharmacological inhibition strategies. Cancer Metastasis Rev. 2015, 34, 643–690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. Curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014, 32, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Srikrishna, S.; Gupta, S.C. Cancer drug development using drosophila as an in vivo tool: From bedside to bench and back. Trends Pharmacol. Sci. 2016, 37, 789–806. [Google Scholar] [CrossRef]
- Khamrai, M.; Banerjee, S.L.; Paul, S.; Samanta, S.; Kundu, P.P. Curcumin entrapped gelatin/ionically modified bacterial cellulose based self-healable hydrogel film: An eco-friendly sustainable synthesis method of wound healing patch. Int. J. Biol. Macromol. 2019, 122, 940–953. [Google Scholar] [CrossRef]
- Vaughn, A.R.; Haas, K.N.; Burney, W.; Andersen, E.; Clark, A.K.; Crawford, R.; Sivamani, R.K. Potential role of curcumin against biofilm-producing organisms on the skin: A review. Phytother. Res. 2017, 31, 1807–1816. [Google Scholar] [CrossRef]
- Li, T.; Zhao, Y.; Matthews, K.; Gao, J.; Hao, J.; Wang, S.; Han, J.; Jia, Y. Antibacterial activity against Staphylococcus aureus of curcumin-loaded chitosan spray coupled with photodynamic treatment. LWT 2020, 134, 110073. [Google Scholar] [CrossRef]
- Lin, Y.-L.; Liu, Y.-K.; Tsai, N.-M.; Hsieh, J.-H.; Chen, C.-H.; Lin, C.-M.; Liao, K.-W. A Lipo-PEG-PEI complex for encapsulating curcumin that enhances its antitumor effects on curcumin-sensitive and curcumin-resistance cells. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 318–327. [Google Scholar] [CrossRef]
- Yu, T.; Ji, J.; Guo, Y.-L. MST1 activation by curcumin mediates JNK activation, Foxo3a nuclear translocation and apoptosis in melanoma cells. Biochem. Biophys. Res. Commun. 2013, 441, 53–58. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Preparation of antimicrobial and antioxidant gelatin/curcumin composite films for active food packaging application. Colloids Surf. B Biointerfaces 2020, 188, 110761. [Google Scholar] [CrossRef]
- Ma, T.; Chen, Y.; Zhi, X.; Du, B. Cellulose laurate films containing curcumin as photoinduced antibacterial agent for meat preservation. Int. J. Biol. Macromol. 2021, 193, 1986–1995. [Google Scholar] [CrossRef]
- Filho, J.G.D.O.; Bertolo, M.R.V.; Rodrigues, M.Á.V.; Marangon, C.A.; Silva, G.D.C.; Odoni, F.C.A.; Egea, M.B. Curcumin: A multifunctional molecule for the development of smart and active biodegradable polymer-based films. Trends Food Sci. Technol. 2021, 118, 840–849. [Google Scholar] [CrossRef]
- Benbettaïeb, N.; Chambin, O.; Karbowiak, T.; Debeaufort, F. Release behavior of quercetin from chitosan-fish gelatin edible films influenced by electron beam irradiation. Food Control 2016, 66, 315–319. [Google Scholar] [CrossRef]
- Da Silva, A.O.; Cortez-Vega, W.R.; Prentice, C.; Fonseca, G.G. Development and characterization of biopolymer films based on bocaiuva (Acromonia aculeata) flour. Int. J. Biol. Macromol. 2020, 155, 1157–1168. [Google Scholar] [CrossRef]
- Haghighi, H.; Leugoue, S.K.; Pfeifer, F.; Siesler, H.W.; Licciardello, F.; Fava, P.; Pulvirenti, A. Development of antimicrobial films based on chitosan-polyvinyl alcohol blend enriched with ethyl lauroyl arginate (LAE) for food packaging applications. Food Hydrocoll. 2020, 100, 105419. [Google Scholar] [CrossRef]
- Sun, L.; Sun, J.; Chen, L.; Niu, P.; Yang, X.; Guo, Y. Preparation and characterization of chitosan film incorporated with thinned young apple polyphenols as an active packaging material. Carbohydr. Polym. 2017, 163, 81–91. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Tang, Z.; Lu, J.; Cheng, Y.; Qian, F.; Zhai, S.; An, Q.; Wang, H. The fabrication of a degradable film with high antimicrobial and antioxidant activities. Ind. Crop. Prod. 2019, 140, 111692. [Google Scholar] [CrossRef]
- Liu, X.; You, L.; Tarafder, S.; Zou, L.; Fang, Z.; Chen, J.; Lee, C.H.; Zhang, Q. Curcumin-releasing chitosan/aloe membrane for skin regeneration. Chem. Eng. J. 2019, 359, 1111–1119. [Google Scholar] [CrossRef]
- Chen, B.; Huang, J.; Li, H.; Zeng, Q.-H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Eradication of planktonic Vibrio parahaemolyticus and its sessile biofilm by curcumin-mediated photodynamic inactivation. Food Control 2020, 113, 107181. [Google Scholar] [CrossRef]
- Huang, J.; Chen, B.; Zeng, Q.-H.; Liu, Y.; Liu, H.; Zhao, Y.; Wang, J.J. Application of the curcumin-mediated photodynamic inactivation for preserving the storage quality of salmon contaminated with L. monocytogenes. Food Chem. 2021, 359, 129974. [Google Scholar] [CrossRef]
- Li, H.; Tan, L.; Chen, B.; Huang, J.; Zeng, Q.; Liu, H.; Zhao, Y.; Wang, J.J. Antibacterial potency of riboflavin-mediated photodynamic inactivation against Salmonella and its influences on tuna quality. LWT—Food Sci. Technol. 2021, 146, 111462. [Google Scholar]
- Bing, S.; Zang, Y.; Li, Y.; Zhang, B.; Mo, Q.; Zhao, X.; Yang, C. A combined approach using slightly acidic electrolyzed water and tea polyphenols to inhibit lipid oxidation and ensure microbiological safety during beef preservation. Meat Sci. 2022, 183, 108643. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, J.; Sobral, P.J.A. Investigation of the physicochemical, antimicrobial and antioxidant properties of gelatin-chitosan edible film mixed with plant ethanolic extracts. Food Biosci. 2016, 16, 17–25. [Google Scholar] [CrossRef]
- Mu, C.; Guo, J.; Li, X.; Lin, W.; Li, D. Preparation and properties of dialdehyde carboxymethyl cellulose crosslinked gelatin edible films. Food Hydrocoll. 2012, 27, 22–29. [Google Scholar] [CrossRef]
- Musso, Y.S.; Salgado, P.R.; Mauri, A.N. Smart edible films based on gelatin and curcumin. Food Hydrocoll. 2017, 66, 8–15. [Google Scholar] [CrossRef] [Green Version]
- Wu, C.; Sun, J.; Chen, M.; Ge, Y.; Ma, J.; Hu, Y.; Pang, J.; Yan, Z. Effect of oxidized chitin nanocrystals and curcumin into chitosan films for seafood freshness monitoring. Food Hydrocoll. 2019, 95, 308–317. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Wang, P.; Guo, M.; Jiang, S.; Li, X.; Jiang, S. Films based on κ-carrageenan incorporated with curcumin for freshness monitoring. Food Hydrocoll. 2018, 83, 134–142. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Balaguer, M.; López-Carballo, G.; Gavara, R.; Hernández-Muñoz, P. Improving antioxidant and antimicrobial properties of curcumin by means of encapsulation in gelatin through electrohydrodynamic atomization. Food Hydrocoll. 2017, 70, 313–320. [Google Scholar] [CrossRef]
- Łupina, K.; Kowalczyk, D.; Zięba, E.; Kazimierczak, W.; Mężyńska, M.; Basiura-Cembala, M.; Wiącek, A.E. Edible films made from blends of gelatin and polysaccharide-based emulsifiers—A comparative study. Food Hydrocoll. 2019, 96, 555–567. [Google Scholar] [CrossRef]
- Xu, T.; Gao, C.; Feng, X.; Huang, M.; Yang, Y.; Shen, X.; Tang, X. Cinnamon and clove essential oils to improve physical, thermal and antimicrobial properties of chitosan-gum Arabic polyelectrolyte complexed films. Carbohydr. Polym. 2019, 217, 116–125. [Google Scholar] [CrossRef]
- Wang, K.; Lim, P.N.; Tong, S.Y.; Thian, E.S. Development of grapefruit seed extract-loaded poly(ε-caprolactone)/chitosan films for antimicrobial food packaging. Food Packag. Shelf Life 2019, 22, 100396. [Google Scholar] [CrossRef]
- Abral, H.; Ariksa, J.; Mahardika, M.; Handayani, D.; Aminah, I.; Sandrawati, N.; Sugiarti, E.; Muslimin, A.N.; Rosanti, S.D. Effect of heat treatment on thermal resistance, transparency and antimicrobial activity of sonicated ginger cellulose film. Carbohydr. Polym. 2020, 240, 116287. [Google Scholar] [CrossRef]
- Su, L.; Huang, J.; Li, H.; Pan, Y.; Zhu, B.; Zhao, Y.; Liu, H. Chitosan-riboflavin composite film based on photodynamic inactivation technology for antibacterial food packaging. Int. J. Biol. Macromol. 2021, 172, 231–240. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, L.; Liu, C.; Zheng, X.; Tang, K. Tuning structure and properties of gelatin edible films through pullulan dialdehyde crosslinking. LWT 2021, 138, 110607. [Google Scholar] [CrossRef]
- Cai, L.; Shi, H.; Cao, A.; Jia, J. Characterization of gelatin/chitosan ploymer films integrated with docosahexaenoic acids fabricated by different methods. Sci. Rep. 2019, 9, 8375. [Google Scholar] [CrossRef]
- Wang, H.; Hao, L.; Wang, P.; Chen, M.; Jiang, S.; Jiang, S. Release kinetics and antibacterial activity of curcumin loaded zein fibers. Food Hydrocoll. 2017, 63, 437–446. [Google Scholar] [CrossRef]
- Herniou-Julien, C.; Mendieta, J.R.; Gutiérrez, T.J. Characterization of biodegradable/non-compostable films made from cellulose acetate/corn starch blends processed under reactive extrusion conditions. Food Hydrocoll. 2019, 89, 67–79. [Google Scholar] [CrossRef]
- Wang, T.; Ke, H.; Chen, S.; Wang, J.; Yang, W.; Cao, X.; Liu, J.; Wei, Q.; Ghiladi, R.A.; Wang, Q. Porous protoporphyrin IX-embedded cellulose diacetate electrospun microfibers in antimicrobial photodynamic inactivation. Mater. Sci. Eng. C 2021, 118, 111502. [Google Scholar] [CrossRef]
- Chen, L.; Dong, Q.; Shi, Q.; Du, Y.; Zeng, Q.; Zhao, Y.; Wang, J.J. Novel 2,3-dialdehyde cellulose-based films with photodynamic inactivation potency by incorporating the β-cyclodextrin/curcumin inclusion complex. Biomacromolecules 2021, 22, 2790–2801. [Google Scholar] [CrossRef]




| Film Nomenclature | Gelatin (%, w/v) | Chitosan (%, w/v) | Ratio (%, w/v) | Glycerol (%, w/v) | Curcumin (mmol/L) |
|---|---|---|---|---|---|
| GEL/CS | 1.5 | 1 | 4:6 | 0.3 | - |
| GEL/CS /Cur0.025 | 1.5 | 1 | 4:6 | 0.3 | 0.025 |
| GEL/CS /Cur0.05 | 1.5 | 1 | 4:6 | 0.3 | 0.05 |
| GEL/CS /Cur0.1 | 1.5 | 1 | 4:6 | 0.3 | 0.1 |
| GEL/CS /Cur 0.2 | 1.5 | 1 | 4:6 | 0.3 | 0.2 |
| Film Samples | L * | a * | b * | ∆E | Opacity | Image |
|---|---|---|---|---|---|---|
| GEL/CS | 93.38 ± 0.07 a | 0.04 ± 0.03 d | 3.23 ± 0.82 c | 4.13 ± 0.46 c | 1.22 ± 0.04 e |  |
| GEL/CS /Cur0.025 | 90.73 ± 0.47 b | 1.23 ± 0.26 c | 15.00 ± 2.12 b | 15.64 ± 1.95 b | 1.73 ± 0.06 d |  |
| GEL/CS /Cur0.05 | 89.91 ± 0.20 bc | 1.69 ± 0.10 bc | 16.63 ± 0.39 b | 17.47 ± 0.43 b | 2.09 ± 0.07 c |  |
| GEL/CS /Cur0.1 | 89.45 ± 0.41 c | 2.22 ± 0.43 b | 17.80 ± 2.03 b | 18.79 ± 2.07 b | 2.83 ± 0.07 b |  |
| GEL/CS /Cur 0.2 | 86.09 ± 0.85 d | 6.47 ± 0.63 a | 30.76 ± 3.03 a | 32.65 ± 3.17 a | 4.69 ± 0.06 a |  |
| Thickness (μm) | TS (MPa) | EB (%) | MC (%) | WS (%) | WVP (g·mm/m2·h·kPa) | |
|---|---|---|---|---|---|---|
| GEL/CS | 0.33 ± 0.06 b | 14.12 ± 0.57 e | 53.19 ± 1.27 e | 21.96 ± 0.42 a | 20.46 ± 1.53 d | 0.304 ± 0.029 ab |
| GEL/CS/Cur0.025 | 0.34 ± 0.05 b | 14.74 ± 0.44 d | 56.01 ± 3.15 d | 21.29 ± 0.56 b | 22.06 ± 0.57 c | 0.296 ± 0.013 ab |
| GEL/CS/Cur0.05 | 0.36 ± 0.01 a | 15.43 ± 0.66 c | 59.08 ± 1.40 c | 20.04 ± 0.11 c | 22.33 ± 1.16 c | 0.289 ± 0.008 b |
| GEL/CS/Cur0.1 | 0.37 ± 0.02 a | 16.85 ± 0.45 b | 60.72 ± 1.59 b | 19.38 ± 0.73 d | 23.42 ± 0.63 b | 0.319 ± 0.026 ab |
| GEL/CS/Cur0.2 | 0.38 ± 0.02 a | 18.12 ± 0.31 a | 65.26 ± 0.62 a | 18.54 ± 1.12 e | 24.75 ± 0.73 a | 0.325 ± 0.014 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, F.; Wang, R.; Pan, Y.; Du, M.; Zhao, Y.; Liu, H. Gelatin/Chitosan Films Incorporated with Curcumin Based on Photodynamic Inactivation Technology for Antibacterial Food Packaging. Polymers 2022, 14, 1600. https://doi.org/10.3390/polym14081600
Wang F, Wang R, Pan Y, Du M, Zhao Y, Liu H. Gelatin/Chitosan Films Incorporated with Curcumin Based on Photodynamic Inactivation Technology for Antibacterial Food Packaging. Polymers. 2022; 14(8):1600. https://doi.org/10.3390/polym14081600
Chicago/Turabian StyleWang, Fan, Ronghan Wang, Yingjie Pan, Ming Du, Yong Zhao, and Haiquan Liu. 2022. "Gelatin/Chitosan Films Incorporated with Curcumin Based on Photodynamic Inactivation Technology for Antibacterial Food Packaging" Polymers 14, no. 8: 1600. https://doi.org/10.3390/polym14081600
APA StyleWang, F., Wang, R., Pan, Y., Du, M., Zhao, Y., & Liu, H. (2022). Gelatin/Chitosan Films Incorporated with Curcumin Based on Photodynamic Inactivation Technology for Antibacterial Food Packaging. Polymers, 14(8), 1600. https://doi.org/10.3390/polym14081600