Enhancing Photoelectric Powder Deposition of Polymers by Charge Control Substances
Abstract
1. Introduction
2. Materials and Methods
2.1. Dry Coating with Charge Control Substances
2.2. Scanning Electron Microscopy
2.3. Powder Flow Characterization
2.4. Differential Scanning Calorimetry
2.5. Electrostatic Surface Potential
2.6. Powder Deposition
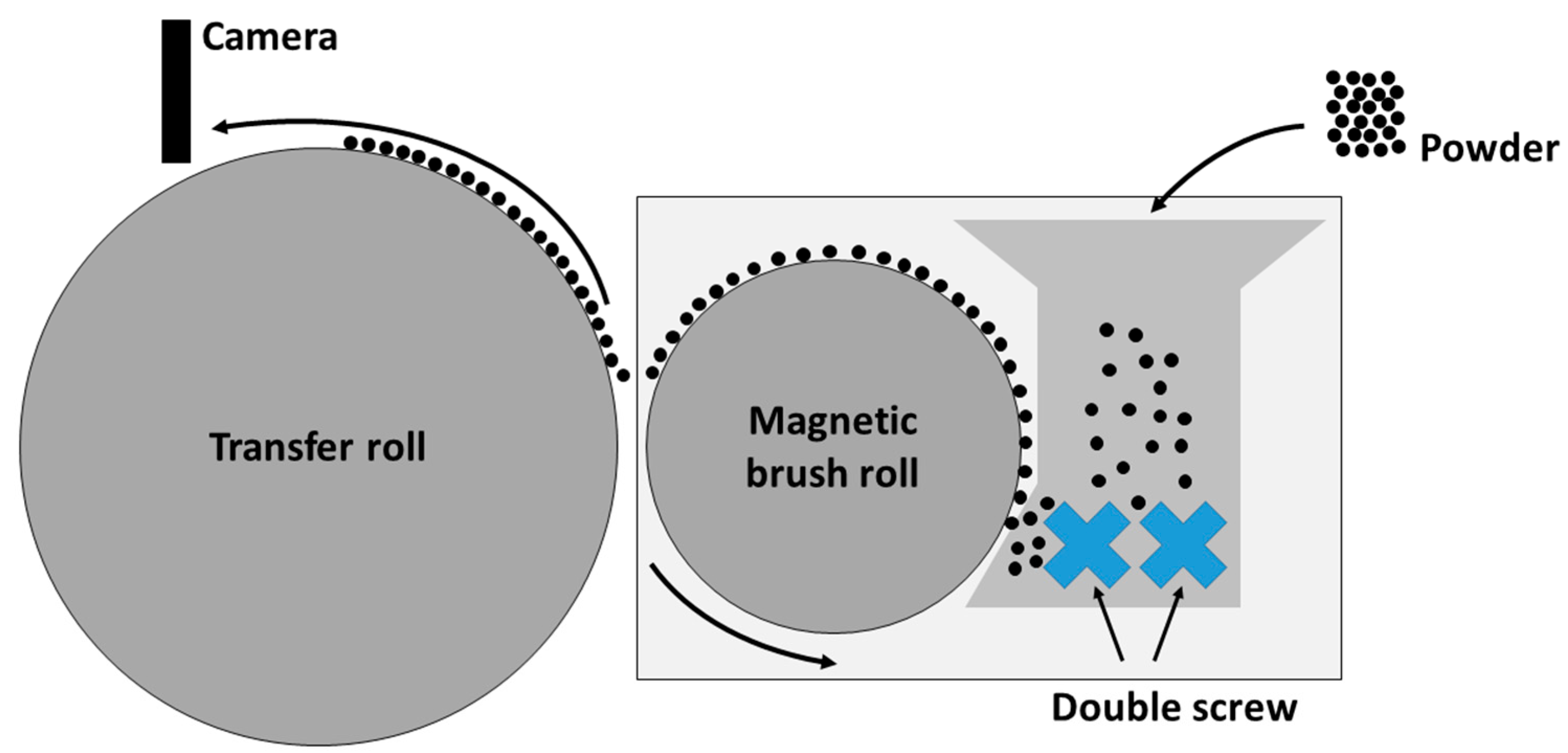
2.6.1. Photoelectric Powder Deposition
2.6.2. Mechanical Powder Deposition and Subsequent Powder Bed Fusion
3. Results and Discussion
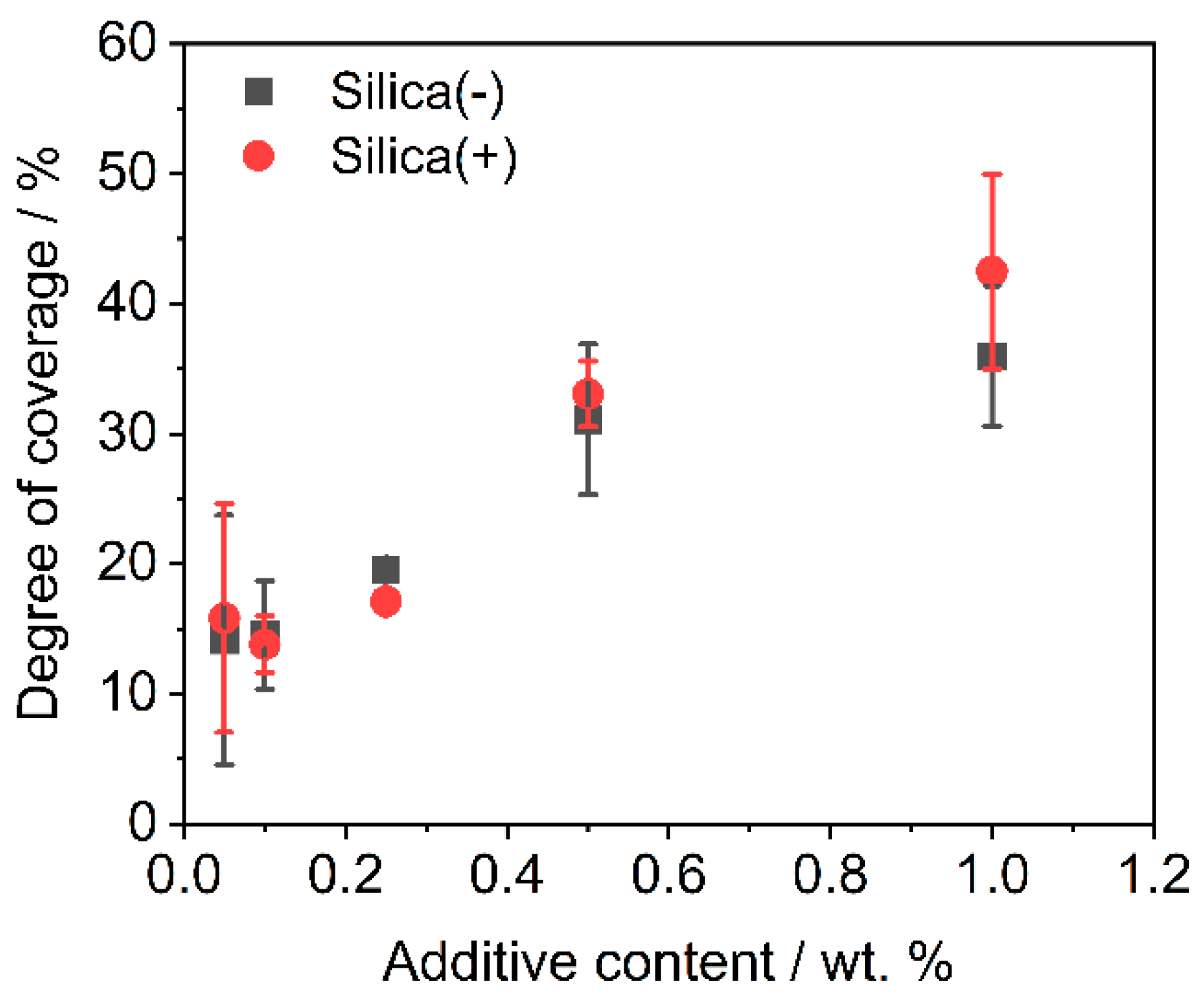
3.1. Degree of Coverage
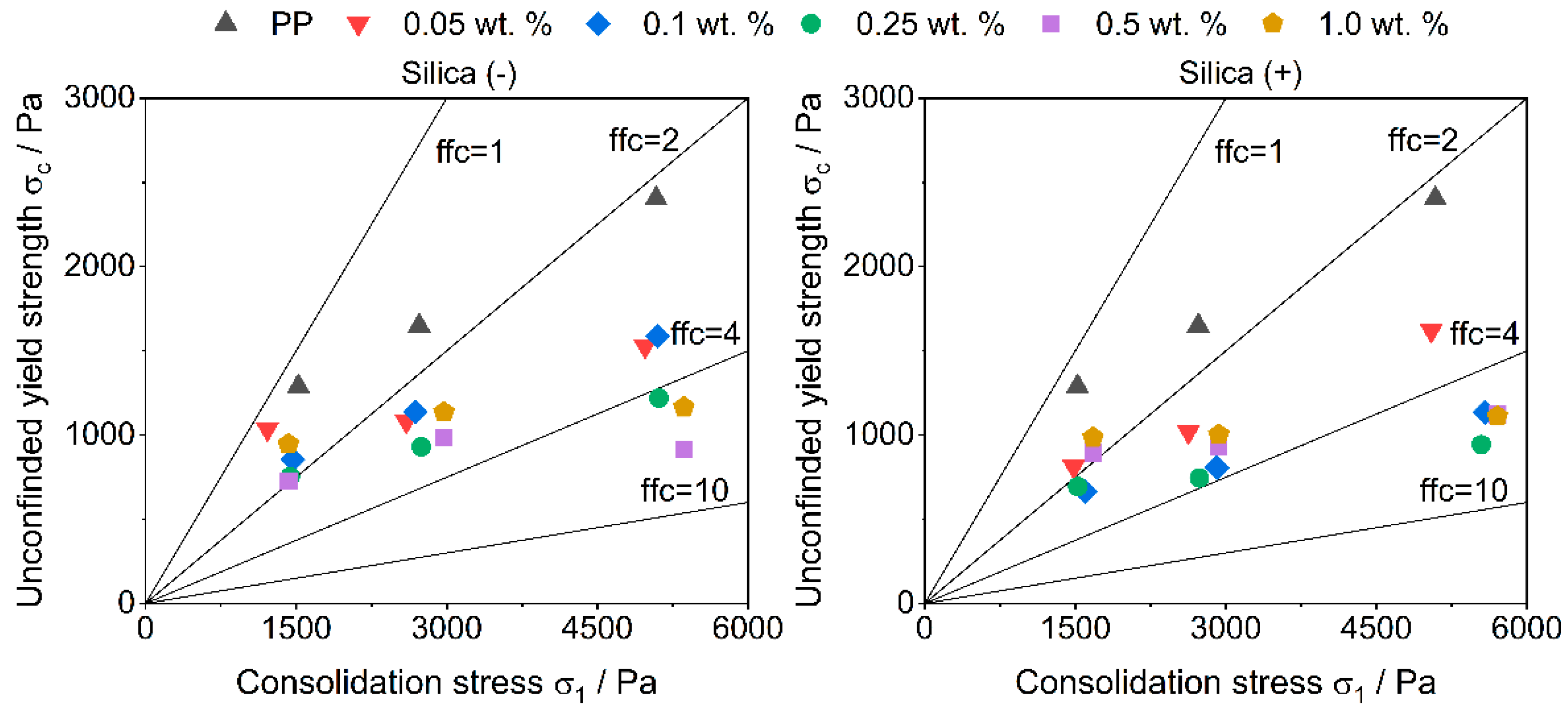
3.2. Flowability
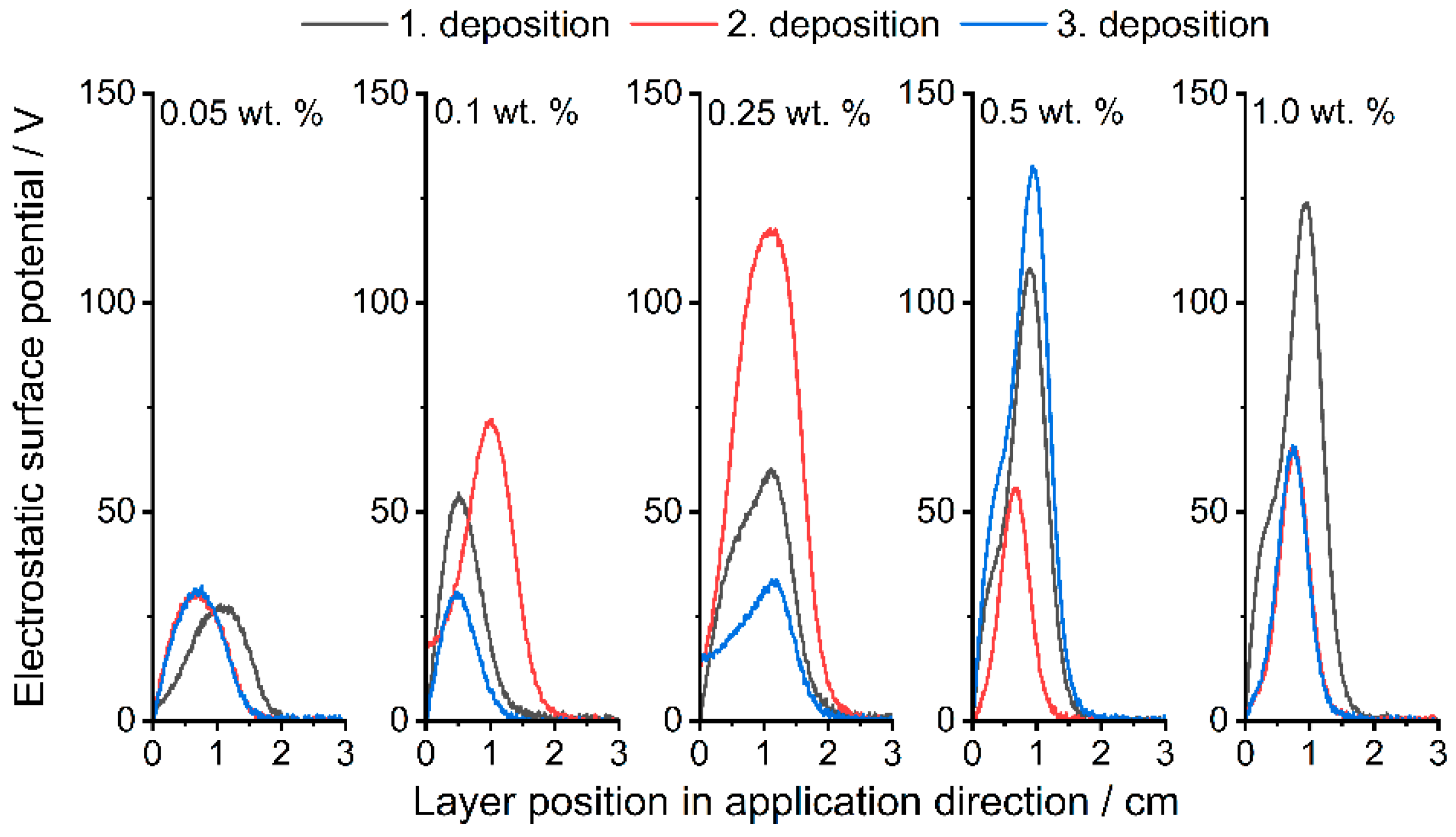
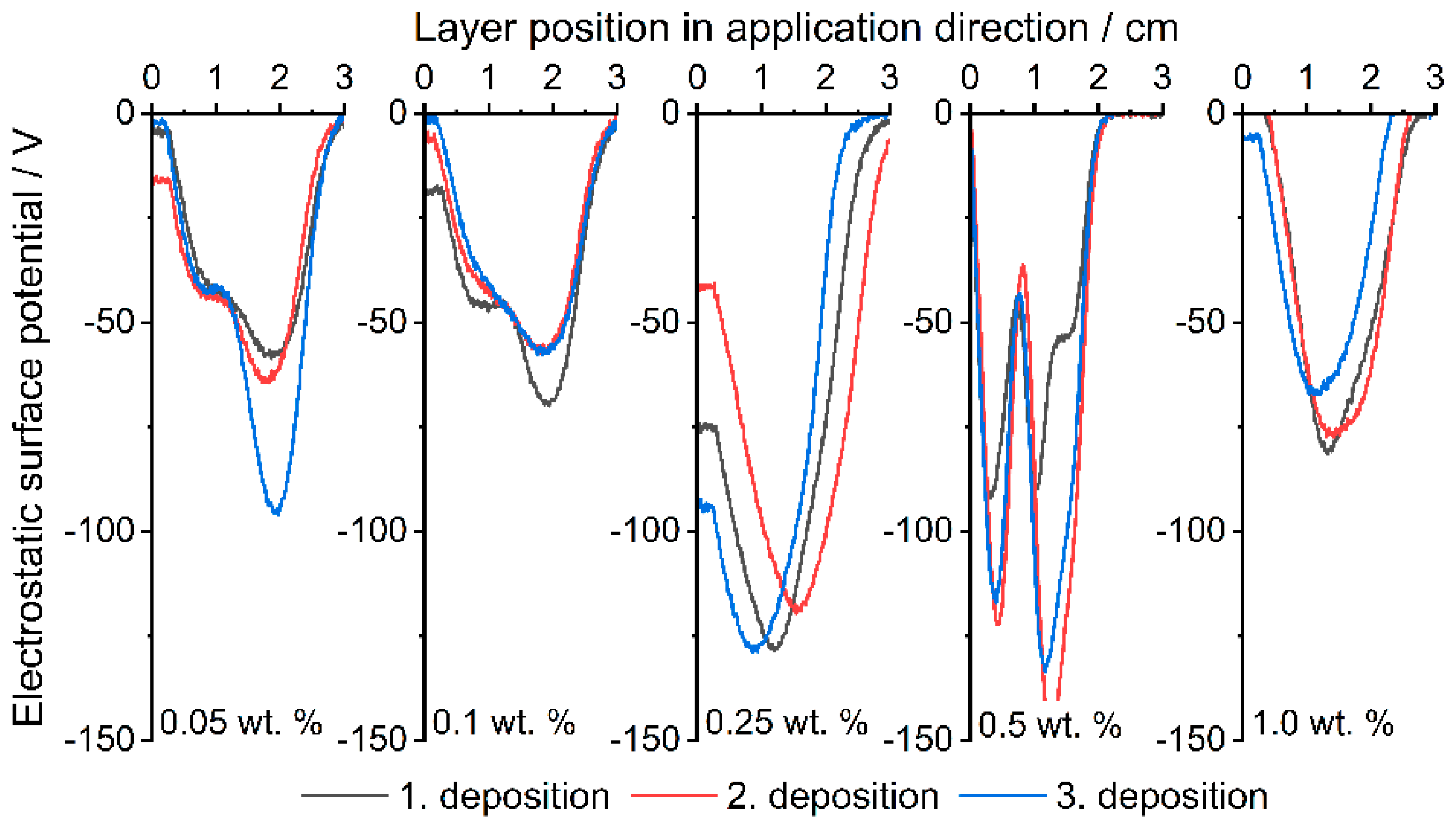
3.3. Electrostatic Surface Potential
3.4. Thermal Analysis
3.5. Powder Deposition
3.5.1. Photoelectric Powder Deposition
3.5.2. Mechanical Powder Deposition and Subsequent Powder Bed Fusion
3.5.3. Characterization of Tensile Strength Specimens
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gibson, I.; Rosen, D.; Stucker, B. Additive Manufacturing Technologies 3D Printing, Rapid Prototyping, and Direct Digital Manufacturing; Springer: Berlin, Germany, 2015. [Google Scholar]
- Schmid, M. Laser Sintering with Plastics: Technology, Processes, and Materials; Carl Hanser Verlag GmbH Co KG: Munich, Germany, 2018; ISBN 156990684X. [Google Scholar]
- Dechet, M.A.; Baumeister, I.; Schmidt, J. Development of polyoxymethylene particles via the solution-dissolution process and application to the powder bed fusion of polymers. Materials 2020, 13, 1535. [Google Scholar] [CrossRef]
- 4. Ziegelmeier, S.; Christou, P.; Wöllecke, F.; Tuck, C.; Goodridge, R.; Hague, R.; Krampe, E.; Wintermantel, E. An experimental study into the effects of bulk and flow behaviour of laser sintering polymer powders on resulting part properties. J. Mater. Processing Technol. 2015, 215, 239–250. [Google Scholar] [CrossRef]
- Gu, H.; AlFayez, F.; Ahmed, T.; Bashir, Z. Poly (ethylene terephthalate) powder—a versatile material for additive manufacturing. Polymers 2019, 11, 2041. [Google Scholar] [CrossRef]
- Dechet, M.A.; Goblirsch, A.; Romeis, S.; Zhao, M.; Lanyi, F.J.; Kaschta, J.; Schubert, D.W.; Drummer, D.; Peukert, W.; Schmidt, J. Production of polyamide 11 microparticles for Additive Manufacturing by liquid-liquid phase separation and precipitation. Chem. Eng. Sci. 2019, 197, 11–25. [Google Scholar] [CrossRef]
- Vidakis, N.; Petousis, M.; Tzounis, L.; Maniadi, A.; Velidakis, E.; Mountakis, N.; Kechagias, J.D. Sustainable additive manufacturing: Mechanical response of polyamide 12 over multiple recycling processes. Materials 2021, 14, 466. [Google Scholar] [CrossRef]
- Berretta, S.; Evans, K.E.; Ghita, O. Processability of PEEK, a new polymer for High Temperature Laser Sintering (HT-LS). Eur. Polym. J. 2015, 68, 243–266. [Google Scholar] [CrossRef]
- Berretta, S.; Ghita, O.; Evans, K.E. Morphology of polymeric powders in Laser Sintering (LS): From Polyamide to new PEEK powders. Eur. Polym. J. 2014, 59, 218–229. [Google Scholar] [CrossRef]
- Strobbe, D.; Dadbakhsh, S.; Verbelen, L.; van Puyvelde, P.; Kruth, J.-P. Selective laser sintering of polystyrene: A single-layer approach. Plast. Rubber Compos. 2018, 47, 2–8. [Google Scholar] [CrossRef]
- Gómez Bonilla, J.S.; Düsenberg, B.; Lanyi, F.; Schmuki, P.; Schubert, D.W.; Schmidt, J.; Peukert, W.; Bück, A. Improvement of polymer properties for powder bed fusion by combining in situ PECVD nanoparticle synthesis and dry coating. Plasma Processes Polym. 2021, 18, e2000247. [Google Scholar] [CrossRef]
- Vidakis, N.; Petousis, M.; Tzounis, L.; Maniadi, A.; Velidakis, E.; Mountakis, N.; Papageorgiou, D.; Liebscher, M.; Mechtcherine, V. Sustainable additive manufacturing: Mechanical response of polypropylene over multiple recycling processes. Sustainability 2021, 13, 159. [Google Scholar] [CrossRef]
- Dechet, M.A.; Gómez Bonilla, J.S.; Lanzl, L.; Drummer, D.; Bück, A.; Schmidt, J.; Peukert, W. Spherical Polybutylene Terephthalate (PBT)—Polycarbonate (PC) Blend Particles by Mechanical Alloying and Thermal Rounding. Polymers 2018, 10, 1373. [Google Scholar] [CrossRef] [PubMed]
- Riedel, F.; Düsenberg, B.; Schmidt, J.; Bück, A.; Peukert, W. Production of PBT/PC multi-material particles via a combination of co-grinding and spray-agglomeration for powder bed fusion. Procedia CIRP 2020, 94, 100–104. [Google Scholar] [CrossRef]
- Park, S.J.; Lee, J.E.; Lee, H.B.; Park, J.; Lee, N.-K.; Son, Y.; Park, S.-H. 3D printing of bio-based polycarbonate and its potential applications in ecofriendly indoor manufacturing. Addit. Manuf. 2020, 31, 100974. [Google Scholar] [CrossRef]
- Meier, C.; Weissbach, R.; Weinberg, J.; Wall, W.A.; Hart, A.J. Critical influences of particle size and adhesion on the powder layer uniformity in metal additive manufacturing. J. Mater. Processing Technol. 2019, 266, 484–501. [Google Scholar] [CrossRef]
- Kusoglu, I.M.; Doñate-Buendía, C.; Barcikowski, S.; Gökce, B. Laser powder bed fusion of polymers: Quantitative research direction indices. Materials 2021, 14, 1169. [Google Scholar] [CrossRef]
- Götzinger, M.; Peukert, W. Dispersive forces of particle–surface interactions: Direct AFM measurements and modelling. Powder Technol. 2003, 130, 102–109. [Google Scholar] [CrossRef]
- Nan, W.; Pasha, M.; Bonakdar, T.; Lopez, A.; Zafar, U.; Nadimi, S.; Ghadiri, M. Jamming during particle spreading in additive manufacturing. Powder Technol. 2018, 338, 253–262. [Google Scholar] [CrossRef]
- Rumpf, H. Die wissenschaft des agglomerierens. Chem. Ing. Tech. 1974, 46, 1–11. [Google Scholar] [CrossRef]
- Blümel, C.; Sachs, M.; Laumer, T.; Winzer, B.; Schmidt, J.; Schmidt, M.; Peukert, W.; Wirth, K.-E. Increasing flowability and bulk density of PE-HD powders by a dry particle coating process and impact on LBM processes. Rapid Prototyp. J. 2015, 21, 697–704. [Google Scholar] [CrossRef]
- Zhou, H.; Götzinger, M.; Peukert, W. The influence of particle charge and roughness on particle–substrate adhesion. Powder Technol. 2003, 135, 82–91. [Google Scholar] [CrossRef]
- Schmidt, J.; Sachs, M.; Fanselow, S.; Zhao, M.; Romeis, S.; Drummer, D.; Wirth, K.-E.; Peukert, W. Optimized polybutylene terephthalate powders for selective laser beam melting. Chem. Eng. Sci. 2016, 156, 1–10. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Mujumdar, A.; Dave, R. Fluidized bed film coating of cohesive Geldart group C powders. Powder Technol. 2009, 189, 466–480. [Google Scholar] [CrossRef]
- Yang, J.; Sliva, A.; Banerjee, A.; Dave, R.N.; Pfeffer, R. Dry particle coating for improving the flowability of cohesive powders. Powder Technol. 2005, 158, 21–33. [Google Scholar] [CrossRef]
- Pfeffer, R.; Dave, R.N.; Wei, D.; Ramlakhan, M. Synthesis of engineered particulates with tailored properties using dry particle coating. Powder Technol. 2001, 117, 40–67. [Google Scholar] [CrossRef]
- Matsusaka, S.; Maruyama, H.; Matsuyama, T.; Ghadiri, M. Triboelectric charging of powders: A review. Chem. Eng. Sci. 2010, 65, 5781–5807. [Google Scholar] [CrossRef]
- Bailey, A.G. Charging of solids and powders. J. Electrost. 1993, 30, 167–180. [Google Scholar] [CrossRef]
- Hesse, N.; Dechet, M.A.; Bonilla, J.S.G.; Lübbert, C.; Roth, S.; Bück, A.; Schmidt, J.; Peukert, W. Analysis of tribo-charging during powder spreading in Selective Laser Sintering: Assessment of polyamide 12 powder ageing effects on charging behavior. Polymers 2019, 11, 609. [Google Scholar] [CrossRef]
- Bailey, A.G. Electrostatic phenomena during powder handling. Powder Technol. 1984, 37, 71–85. [Google Scholar] [CrossRef]
- Bauser, H. Static electrification of organic solids. Dechema Monogr. 1974, 71, 11–28. [Google Scholar]
- Taylor, D.M.; Secker, P.E. Industrial Electrostatics: Fundamentals and Measurements; Research Studies Press: Boston, MA, USA, 1994; ISBN 0863801587. [Google Scholar]
- Henniker, J. Triboelectricity in polymers. Nature 1962, 196, 474. [Google Scholar] [CrossRef]
- Cross, J. Electrostatics, Principles, Problems and Applications; CRC Press: Boca Raton, FL, USA, 1987; ISBN 0852745893. [Google Scholar]
- Biegaj, K.W.; Rowland, M.G.; Lukas, T.M.; Heng, J.Y.Y. Surface chemistry and humidity in powder electrostatics: A comparative study between tribocharging and corona discharge. Acs Omega 2017, 2, 1576–1582. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.; James, N.M.; Waitukaitis, S.R.; Jaeger, H.M. Collisional charging of individual submillimeter particles: Using ultrasonic levitation to initiate and track charge transfer. Phys. Rev. Mater. 2018, 2, 35602. [Google Scholar] [CrossRef]
- Zou, H.; Guo, L.; Xue, H.; Zhang, Y.; Shen, X.; Liu, X.; Wang, P.; He, X.; Dai, G.; Jiang, P. Quantifying and understanding the triboelectric series of inorganic non-metallic materials. Nat. Commun. 2020, 11, 2093. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.W. Triboelectric charging of insulating polymers–some new perspectives. Aip Adv. 2012, 2, 10701. [Google Scholar] [CrossRef]
- Zou, H.; Zhang, Y.; Guo, L.; Wang, P.; He, X.; Dai, G.; Zheng, H.; Chen, C.; Wang, A.C.; Xu, C. Quantifying the triboelectric series. Nat. Commun. 2019, 10, 1427. [Google Scholar] [CrossRef]
- Ndama, A.T.; Guigon, P.; Saleh, K. A reproducible test to characterise the triboelectric charging of powders during their pneumatic transport. J. Electrost. 2011, 69, 146–156. [Google Scholar] [CrossRef]
- Huber, G.; Wirth, K.-E. Electrostatically supported surface coating of solid particles in liquid nitrogen for use in dry-powder-inhalers. Powder Technol. 2003, 134, 181–192. [Google Scholar] [CrossRef]
- Stichel, T.; Brachmann, C.; Raths, M.; Dechet, M.A.; Schmidt, J.; Peukert, W.; Frick, T.; Roth, S. Electrophotographic multilayer powder pattern deposition for additive manufacturing. Jom 2020, 72, 1366–1375. [Google Scholar] [CrossRef]
- Kopp, S.-P.; Stichel, T.; Roth, S.; Schmidt, M. Investigation of the electrophotographic powder deposition through a transfer grid for efficient additive manufacturing. Procedia CIRP 2020, 94, 122–127. [Google Scholar] [CrossRef]
- Martin, T.I. Method for Printing 3D Parts and Support Structures with Electrophotography-Based Additive Manufacturing. U.S. Patent US10065371B2, 29 September 2015. [Google Scholar]
- Benning, M.J.; Dalgarno, K. Proof of concept of a novel combined consolidation and transfer mechanism for electrophotographic 3D printing. Rapid Prototyp. J. 2018, 24, 1040–1048. [Google Scholar] [CrossRef]
- Wei, C.; Li, L. Recent progress and scientific challenges in multi-material additive manufacturing via laser-based powder bed fusion. Virtual Phys. Prototyp. 2021, 16, 347–371. [Google Scholar] [CrossRef]
- Kumar, A.V.; Dutta, A.; Fay, J.E. Electrophotographic printing of part and binder powders. Rapid Prototyp. J. 2004, 10, 7–13. [Google Scholar] [CrossRef]
- Kumar, A.V.; Dutta, A. Investigation of an electrophotography based rapid prototyping technology. Rapid Prototyp. J. 2003, 9, 95–103. [Google Scholar] [CrossRef]
- Schein, L.B. Electrophotography and Development Physics; Springer Science & Business Media: Berlin, Germany, 2013; ISBN 3642777449. [Google Scholar]
- Hays, D.A. Toner adhesion. J. Adhes. 1995, 51, 41–48. [Google Scholar] [CrossRef]
- Weigl, J.W. Electrophotography. Angew. Chem. Int. Ed. Engl. 1977, 16, 374–392. [Google Scholar] [CrossRef]
- Bejat, L.A.; Johnson, J.E.; Jones, R.O.; Livengood, B.P.; Srinivasan, K.R.; Strain, D.J.V. Toner Formulations with Tribocharge Control and Stability. U.S. Patent US20100040969A1, 18 February 2010. [Google Scholar]
- Anderson, J.H. The effect of additives on the tribocharging of electrophotographic toners. J. Electrost. 1996, 37, 197–209. [Google Scholar] [CrossRef]
- Sweeney, M.A.; Mang, M.E.; LaFica, S. ABS/Polycarbonate/Poly (Styrene-Co-Maleimide) Part Material for Electrophotography-Based Additive Manufacturing. U.S. Patent US20170190114A1, 11 February 2020. [Google Scholar]
- Blair, B.M.; Moore, M.T.; Morris, M.K.; Olson, J.M.; Walsh, J.L. Toner Formulations. Japan Patent JP200614 6268A, 2003.
- Orrock, J.E.; Chillscyzn, S.A.; Martin, T.I. High-Performance Consumable Materials for Electrophotography-Based Additive Manufacturing. U.S. Patent US20180046107A1, 22 January 2015. [Google Scholar]
- Baur, R.; Macholdt, H.-T. Charge control agents for triboelectric (friction) charging. J. Electrost. 1993, 30, 213–222. [Google Scholar] [CrossRef]
- Ni, W.; Wu, S.; Ren, Q. Silanized TiO2 nanoparticles and their application in toner as charge control agents: Preparation and characterization. Chem. Eng. J. 2013, 214, 272–277. [Google Scholar] [CrossRef]
- Alonso, M.; Alguacil, F.J. Dry mixing and coating of powders. Rev. De Metal. 1999, 35, 315–328. [Google Scholar] [CrossRef]
- Alonso, M.; Satoh, M.; Miyanami, K. Powder coating in a rotary mixer with rocking motion. Powder Technol. 1988, 56, 135–141. [Google Scholar] [CrossRef]
- Streich, C.; Koenen, S.; Lelle, M.; Peneva, K.; Barcikowski, S. Influence of ligands in metal nanoparticle electrophoresis for the fabrication of biofunctional coatings. Appl. Surf. Sci. 2015, 348, 92–99. [Google Scholar] [CrossRef]
- Wagener, P.; Brandes, G.; Schwenke, A.; Barcikowski, S. Impact of in situ polymer coating on particle dispersion into solid laser-generated nanocomposites. Phys. Chem. Chem. Phys. 2011, 13, 5120–5126. [Google Scholar] [CrossRef] [PubMed]
- Israelachvili, J.N. Intermolecular and Surface Forces; Academic Press: Cambridge, MA, USA, 2015; ISBN 0080923631. [Google Scholar]
- Krupp, H. Particles adhesion theory and experiment. Adv. Colloid Interface Sci. 1967, 1, 111–239. [Google Scholar]
- Dechet, M.A.; Bonilla, J.S.G.; Grünewald, M.; Popp, K.; Rudloff, J.; Lang, M.; Schmidt, J. A novel, precipitated polybutylene terephthalate feedstock material for powder bed fusion of polymers (PBF): Material development and initial PBF processability. Mater. Des. 2021, 197, 109265. [Google Scholar] [CrossRef]
- Laumer, T.; Stichel, T.; Amend, P.; Schmidt, M. Simultaneous laser beam melting of multimaterial polymer parts. J. Laser Appl. 2015, 27, S29204. [Google Scholar] [CrossRef]
- Laumer, T.; Stichel, T.; Riedlbauer, D.; Amend, P.; Mergheim, J.; Schmidt, M. Realization of Multi-material Polymer Parts by Simultaneous Laser Beam Melting. J. Laser Micro/Nanoeng. 2015, 10, 2. [Google Scholar] [CrossRef][Green Version]
- Evonik, A.G. Safety Data Sheet—Aerosil 200 Pharma. 2021. Available online: https://sds.evonik.com/msds-list/searchresult/aerosil/EN/99034598/SDS_GB/sds.pdf (accessed on 23 September 2021).
- Axalta Polymer Powders. Coathylene PD0580 Manufacturer Information. Available online: https://secure.axalta.com/content/dam/EMEA/Polymer%20Powders/EN/Public/Documents/polymer-powder-additives/Axalta-Coathylene-Paint-and-Varnishes-Flyer.pdf (accessed on 24 February 2022).
- McDonald, J.C.; Duffy, D.C.; Anderson, J.R.; Chiu, D.T.; Wu, H.; Schueller, O.J.A.; Whitesides, G.M. Fabrication of microfluidic systems in poly (dimethylsiloxane). Electrophor. Int. J. 2000, 21, 27–40. [Google Scholar] [CrossRef]
- Jankauskaitė, V.; Narmontas, P.; Lazauskas, A. Control of polydimethylsiloxane surface hydrophobicity by plasma polymerized hexamethyldisilazane deposition. Coatings 2019, 9, 36. [Google Scholar] [CrossRef]
- Jenike, A.W. Flow and storage of solids. In Utah Experimental Station; University of Utah: Salt Lake City, UT, USA, 1967. [Google Scholar]
- Schulze, D. Powders and bulk solids. In Behaviour, Characterization, Storage and Flow; Springer: Berlin, Germany, 2008; Volume 22. [Google Scholar]
- Meyer, K.; Zimmermann, I. Effect of glidants in binary powder mixtures. Powder Technol. 2004, 139, 40–54. [Google Scholar] [CrossRef]
- Schweiger, A.; Zimmermann, I. A new approach for the measurement of the tensile strength of powders. Powder Technol. 1999, 101, 7–15. [Google Scholar] [CrossRef]
- Otsu, N. A threshold selection method from gray-level histograms. IEEE Trans. Syst. Man Cybern. 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Lowell, J.; Rose-Innes, A.C. Contact electrification. Adv. Phys. 1980, 29, 947–1023. [Google Scholar] [CrossRef]
- Shaw, P.E. Experiments on tribo-electricity. I.—The tribo-electric series. Proc. R. Soc. London. Ser. A Contain. Pap. A Math. Phys. Character 1917, 94, 16–33. [Google Scholar]
- Schubert, G.; Németh, E.; Simon, F.; Albrecht, V. Beeinflussung der Kontaktaufladung von Kunststoffen durch Modifizierung ihrer Oberfläche. Forsch. Sonderforschungsbereich 2004, 285, 183–211. [Google Scholar]
- Greason, W.D. Investigation of a test methodology for triboelectrification. J. Electrost. 2000, 49, 245–256. [Google Scholar] [CrossRef]
- Fillon, B.; Thierry, A.; Lotz, B.; Wittmann, J.C. Efficiency scale for polymer nucleating agents. J. Therm. Anal. 1994, 42, 721–731. [Google Scholar] [CrossRef]
- Beck, H.N.; Ledbetter, H.D. DTA study of heterogeneous nucleation of crystallization in polypropylene. J. Appl. Polym. Sci. 1965, 9, 2131–2142. [Google Scholar] [CrossRef]
- Velasco, J.I.; de Saja, J.A.; Martinez, A.B. Crystallization behavior of polypropylene filled with surface-modified talc. J. Appl. Polym. Sci. 1996, 61, 125–132. [Google Scholar] [CrossRef]
- Huang, L.; Zhan, R.; Lu, Y. Mechanical Properties and Crystallization Behavior of Polypropylene/Nano-SiO2 Composites. J. Reinf. Plast. Compos. 2006, 25, 1001–1012. [Google Scholar] [CrossRef]
- Qian, J.; He, P.; Nie, K. Nonisothermal crystallization of PP/nano-SiO2 composites. J. Appl. Polym. Sci. 2004, 91, 1013–1019. [Google Scholar] [CrossRef]
- Nomura, T.; Satoh, T.; Masuda, H. The environment humidity effect on the tribo-charge of powder. Powder Technol. 2003, 135, 43–49. [Google Scholar] [CrossRef]






| Property | Silica(−) HDK H05TX | Silica(+) HDK H05TA 3 |
|---|---|---|
| Particles tend to charge | negative | positive |
| Specific surface area (BET)/m² g−1 | 50 ± 20 | 50 ± 20 |
| Mean particle size/nm | 50 | 50 |
| Agglomerate particle size/µm | <20 | <20 |
| Specific charge/µC g−1 | −450 | +50 |
| Surface modification | HMDS/PDMS | PDMS/−NR2/−NR3 |
| No | Polypropylene | Silica(−) | Silica(+) | Mixing Aids | Coating Time |
|---|---|---|---|---|---|
| - | g | wt.% | wt.% | g | Min |
| 1.1 | 3 × 60 | 0.05 | - | 60 | 60 |
| 1.2 | 1 × 500 | 0.05 | - | 260 | |
| 2 | 3 × 60 | 0.1 | - | 60 | |
| 3 | 3 × 60 | 0.25 | - | 60 | |
| 4 | 3 × 60 | 0.5 | - | 60 | |
| 5.1 | 3 × 60 | 1.0 | - | 60 | |
| 5.2 | 1 × 500 | 1.0 | - | 260 | |
| 6.1 | 3 × 60 | - | 0.05 | 60 | |
| 6.2 | 1 × 500 | - | 0.05 | 260 | |
| 7 | 3 × 60 | - | 0.1 | 60 | |
| 8 | 3 × 60 | - | 0.25 | 60 | |
| 9 | 3 × 60 | - | 0.5 | 60 | |
| 10.1 | 3 × 60 | - | 1.0 | 60 | |
| 10.2 | 1 × 500 | - | 1.0 | 260 |
| Formulation (cf. Table 2) | Enthalpy J gpolymer−1 | Crystallization Temperature °C |
|---|---|---|
| Raw PP | −106 | 120 |
| 1.1 | −96.9 ± 0.6 | 122.7 ± 0.08 |
| 2 | −97.4 ± 1.2 | 122.2 ± 0.001 |
| 3 | −95.6 ± 1.1 | 122.4 ± 0.001 |
| 4 | −96,6 ± 2.0 | 122.8 ± 0.12 |
| 5.1 | −99,4 ± 1.8 | 123.4 ± 0.12 |
| 6.1 | −94.9 ± 0.9 | 122.1 ± 0.05 |
| 7 | −97.6 ± 0.6 | 122.3 ± 0.05 |
| 8 | −96.5 ± 1.7 | 122.3 ± 0.12 |
| 9 | −96.4 ± 1.3 | 122.4 ± 0.08 |
| 10.1 | −100.0 ± 0.6 | 144.8 ± 0.05 |
| Corresponding Image from Figure 8 | Formulation (cf. Table 2) | Degree of Coverage/% | Polarity of the Electrical Field of the Transfer Roll |
|---|---|---|---|
| a | Raw PP | 73.1 ± 4.8 | Positive |
| b | 1.2 | 99.74 ± 0.5 | |
| c | 5.2 | 99.6 ± 0.3 | |
| d | Raw PP | 66.14 ± 7.3 | Negative |
| e | 6.2 | 75.26 ± 11.0 | |
| f | 10.2 | 100 ± 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Düsenberg, B.; Kopp, S.-P.; Tischer, F.; Schrüfer, S.; Roth, S.; Schmidt, J.; Schmidt, M.; Schubert, D.W.; Peukert, W.; Bück, A. Enhancing Photoelectric Powder Deposition of Polymers by Charge Control Substances. Polymers 2022, 14, 1332. https://doi.org/10.3390/polym14071332
Düsenberg B, Kopp S-P, Tischer F, Schrüfer S, Roth S, Schmidt J, Schmidt M, Schubert DW, Peukert W, Bück A. Enhancing Photoelectric Powder Deposition of Polymers by Charge Control Substances. Polymers. 2022; 14(7):1332. https://doi.org/10.3390/polym14071332
Chicago/Turabian StyleDüsenberg, Björn, Sebastian-Paul Kopp, Florentin Tischer, Stefan Schrüfer, Stephan Roth, Jochen Schmidt, Michael Schmidt, Dirk W. Schubert, Wolfgang Peukert, and Andreas Bück. 2022. "Enhancing Photoelectric Powder Deposition of Polymers by Charge Control Substances" Polymers 14, no. 7: 1332. https://doi.org/10.3390/polym14071332
APA StyleDüsenberg, B., Kopp, S.-P., Tischer, F., Schrüfer, S., Roth, S., Schmidt, J., Schmidt, M., Schubert, D. W., Peukert, W., & Bück, A. (2022). Enhancing Photoelectric Powder Deposition of Polymers by Charge Control Substances. Polymers, 14(7), 1332. https://doi.org/10.3390/polym14071332







