Graphene Oxide–Protein-Based Scaffolds for Tissue Engineering: Recent Advances and Applications
Abstract
:1. Introduction
2. Proteins for Tissue Engineering
2.1. Collagen and Gelatin
2.2. Fibronectin
2.3. Elastin
2.4. Human Serum Albumin
2.5. Bovine Serum Albumin (BSA)
2.6. Laminins
2.7. Growth Factors
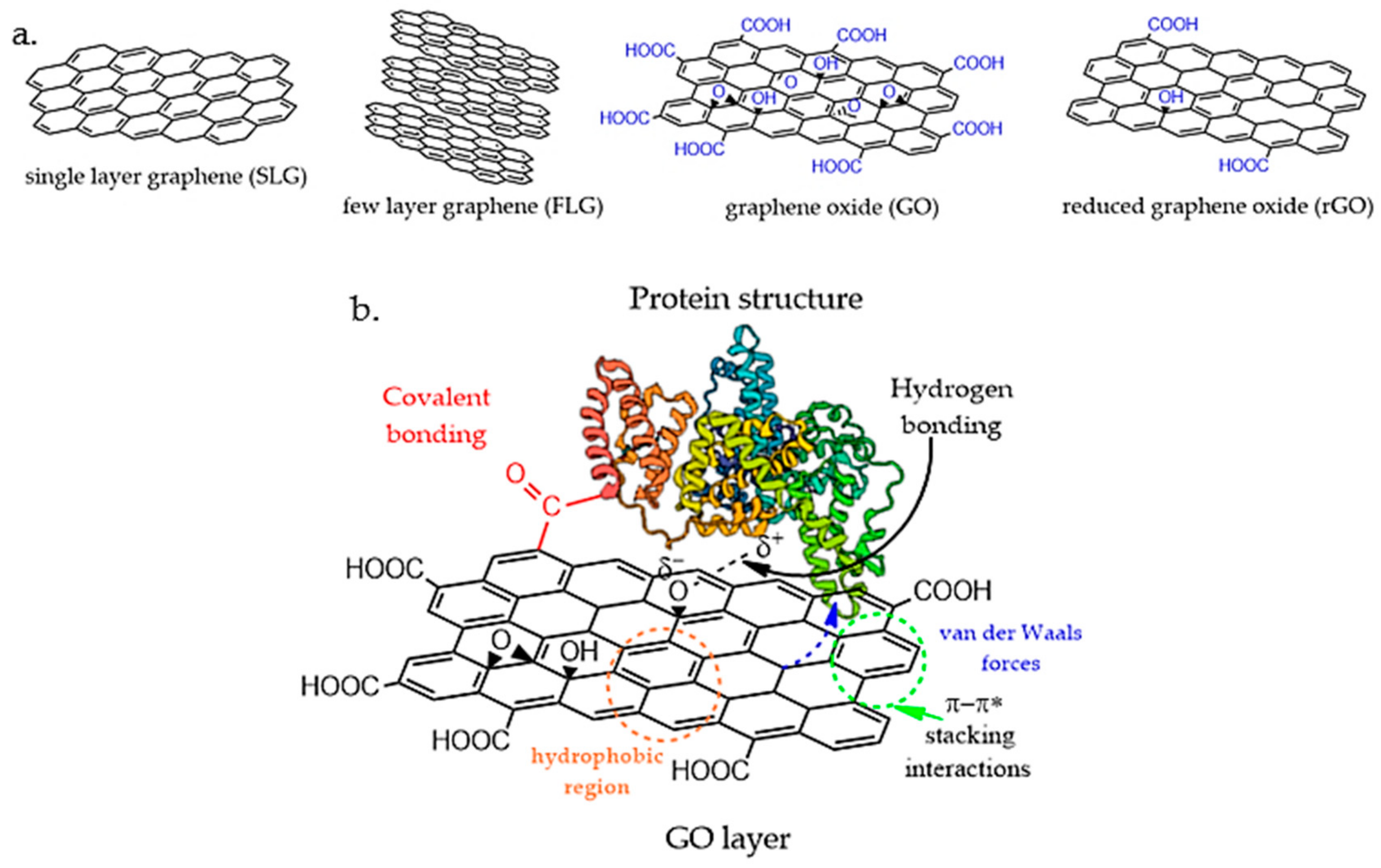
3. Graphene Oxide–Protein Interactions
3.1. Non-Covalent Interactions
3.2. Covalent Attachment
4. Applications in Tissue Engineering and Regenerative Medicine
4.1. Bone Tissue Engineering
4.2. Cardiac Tissue
4.3. Nerve Tissue
5. Advances and Challenges in Tissue Engineering Platforms
6. Conclusions and Future Considerations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tiwari, S.; Patil, R.; Dubey, S.K.; Bahadur, P. Graphene nanosheets as reinforcement and cell-instructive material in soft tissue scaffolds. Adv. Colloid Interface Sci. 2020, 281, 102167. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.C.; Southgate, J. 11—Bladder tissue regeneration. In Electrospinning for Tissue Regeneration; Bosworth, L.A., Downes, S., Eds.; Woodhead Publishing: Cambridge, UK, 2011; pp. 225–241. [Google Scholar] [CrossRef]
- Peranidze, K.; Safronova, T.V.; Kildeeva, N.R. Fibrous Polymer-Based Composites Obtained by Electrospinning for Bone Tissue Engineering. Polymers 2022, 14, 96. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.S.; Jardini, A.L.; Filho, R.M. Poly (Lactic Acid) Production for Tissue Engineering Applications. Procedia Eng. 2012, 42, 1402–1413. [Google Scholar] [CrossRef] [Green Version]
- Kumar, A.; Han, S.S. PVA-based hydrogels for tissue engineering: A review. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 159–182. [Google Scholar] [CrossRef]
- Gentile, P.; Chiono, V.; Carmagnola, I.; Hatton, P.V. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014, 15, 3640–3659. [Google Scholar] [CrossRef]
- Siddiqui, N.; Asawa, S.; Birru, B.; Baadhe, R.; Rao, S. PCL-Based Composite Scaffold Matrices for Tissue Engineering Applications. Mol. Biotechnol. 2018, 60, 506–532. [Google Scholar] [CrossRef] [PubMed]
- Echave, M.C.; Saenz del Burgo, L.; Pedraz, J.L.; Orive, G. Gelatin as Biomaterial for Tissue Engineering. Curr. Pharm. Des. 2017, 23, 3567–3584. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Li, R.; Bai, H.; Zhu, Z.; Zhu, L.; Zhu, C.; Che, Z.; Liu, H.; Wang, J.; et al. Collagen-based biomaterials for bone tissue engineering. Mater. Des. 2021, 210, 110049. [Google Scholar] [CrossRef]
- Sukpaita, T.; Chirachanchai, S.; Pimkhaokham, A.; Ampornaramveth, R.S. Chitosan-Based Scaffold for Mineralized Tissues Regeneration. Mar. Drugs 2021, 19, 551. [Google Scholar] [CrossRef]
- Sahoo, D.R.; Biswal, T. Alginate and its application to tissue engineering. SN Appl. Sci. 2021, 3, 30. [Google Scholar] [CrossRef]
- Pandini, F.E.; Kubo, F.M.M.; Plepis, A.M.d.G.; Martins, V.d.C.A.; da Cunha, M.R.; Silva, V.R.; Hirota, V.B.; Lopes, E.; Menezes, M.A.; Pelegrine, A.A.; et al. In Vivo Study of Nasal Bone Reconstruction with Collagen, Elastin and Chitosan Membranes in Abstainer and Alcoholic Rats. Polymers 2022, 14, 188. [Google Scholar] [CrossRef] [PubMed]
- Della Coletta, B.B.; Jacob, T.B.; Moreira, L.A.d.C.; Pomini, K.T.; Buchaim, D.V.; Eleutério, R.G.; Pereira, E.d.S.B.M.; Roque, D.D.; Rosso, M.P.d.O.; Shindo, J.V.T.C.; et al. Photobiomodulation Therapy on the Guided Bone Regeneration Process in Defects Filled by Biphasic Calcium Phosphate Associated with Fibrin Biopolymer. Molecules 2021, 26, 847. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, C.M.; McKinney, J.S.; Lanctot, D.; Athanasiou, K.A. Effects of fluid flow on the in vitro degradation kinetics of biodegradable scaffolds for tissue engineering. Biomaterials 2000, 21, 2443–2452. [Google Scholar] [CrossRef]
- Gomes, S.; Leonor, I.B.; Mano, J.F.; Reis, R.L.; Kaplan, D.L. Natural and Genetically Engineered Proteins for Tissue Engineering. Prog. Polym. Sci. 2012, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Uherek, C.; Wels, W. DNA-carrier proteins for targeted gene delivery. Adv. Drug Deliv. Rev. 2000, 44, 153–166. [Google Scholar] [CrossRef]
- Hong, S.; Choi, D.W.; Kim, H.N.; Park, C.G.; Lee, W.; Park, H.H. Protein-Based Nanoparticles as Drug Delivery Systems. Pharmaceutics 2020, 12, 604. [Google Scholar] [CrossRef]
- Green, E.M.; Lee, R.T. Proteins and small molecules for cellular regenerative medicine. Physiol. Rev. 2013, 93, 311–325. [Google Scholar] [CrossRef]
- Ren, X.; Zhao, M.; Lash, B.; Martino, M.M.; Julier, Z. Growth Factor Engineering Strategies for Regenerative Medicine Applications. Front. Bioeng. Biotechnol. 2020, 7, 469. [Google Scholar] [CrossRef]
- Misawa, N.; Osaki, T.; Takeuchi, S. Membrane protein-based biosensors. J. R. Soc. Interface 2018, 15, 20170952. [Google Scholar] [CrossRef]
- Shi, M.; Fu, Z.; Pan, W.; Chen, Y.; Wang, K.; Zhou, P.; Li, N.; Tang, B. A Protein-Binding Molecular Photothermal Agent for Tumor Ablation. Angew. Chem. Int. Ed. 2021, 60, 13564–13568. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Jin, N.; Mao, C.; Yang, M. Protein-Induced Gold Nanoparticle Assembly for Improving the Photothermal Effect in Cancer Therapy. ACS Appl. Mater. Interfaces 2019, 11, 11136–11143. [Google Scholar] [CrossRef] [PubMed]
- Gromiha, M.M. (Ed.) Chapter 6—Protein Stability. In Protein Bioinformatics; Academic Press: Singapore, 2010; pp. 209–245. [Google Scholar] [CrossRef]
- Li, D.; Liu, T.; Yu, X.; Wu, D.; Su, Z. Fabrication of graphene–biomacromolecule hybrid materials for tissue engineering application. Polym. Chem. 2017, 8, 4309–4321. [Google Scholar] [CrossRef]
- Bosi, S.; Ballerini, L.; Prato, M. Carbon nanotubes in tissue engineering. Top. Curr. Chem. 2014, 348, 181–204. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Sundarrajan, S.; Hussain, T.; Nazir, A.; Ayyoob, M.; Berto, F.; Ramakrishna, S. Sustainable nanofibers in tissue engineering and biomedical applications. Mater. Des. Process. Commun. 2021, 3, e202. [Google Scholar] [CrossRef]
- Yadid, M.; Feiner, R.; Dvir, T. Gold Nanoparticle-Integrated Scaffolds for Tissue Engineering and Regenerative Medicine. Nano Lett. 2019, 19, 2198–2206. [Google Scholar] [CrossRef]
- Kim, S.W.; Im, G.B.; Kim, Y.J.; Kim, Y.H.; Lee, T.J.; Bhang, S.H. Bio-application of Inorganic Nanomaterials in Tissue Engineering. Adv. Exp. Med. Biol. 2020, 1249, 115–130. [Google Scholar] [CrossRef]
- Geetha Bai, R.; Muthoosamy, K.; Manickam, S.; Hilal-Alnaqbi, A. Graphene-based 3D scaffolds in tissue engineering: Fabrication, applications, and future scope in liver tissue engineering. Int. J. Nanomed. 2019, 14, 5753–5783. [Google Scholar] [CrossRef] [Green Version]
- Eivazzadeh-Keihan, R.; Maleki, A.; de la Guardia, M.; Bani, M.S.; Chenab, K.K.; Pashazadeh-Panahi, P.; Baradaran, B.; Mokhtarzadeh, A.; Hamblin, M.R. Carbon based nanomaterials for tissue engineering of bone: Building new bone on small black scaffolds: A review. J. Adv. Res. 2019, 18, 185–201. [Google Scholar] [CrossRef]
- Fox, K.; Ratwatte, R.; Booth, M.A.; Tran, H.M.; Tran, P.A. High Nanodiamond Content-PCL Composite for Tissue Engineering Scaffolds. Nanomaterials 2020, 10, 948. [Google Scholar] [CrossRef]
- Zhao, Y.; Shen, X.; Ma, R.; Hou, Y.; Qian, Y.; Fan, C. Biological and biocompatible characteristics of fullerenols nanomaterials for tissue engineering. Histol. Histopathol. 2021, 36, 725–731. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K. Advances in Drug Delivery Nanosystems Using Graphene-Based Materials and Carbon Nanotubes. Materials 2021, 14, 1059. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.C.; Lee, S.; Lalwani, G.; Suhrland, C.; Chowdhury, S.M.; Sitharaman, B. Graphene-based platforms for cancer therapeutics. Ther. Deliv. 2016, 7, 101–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, S.; Shen, H.; Wang, Y.; Chu, X.; Xie, J.; Zhou, N.; Shen, J. Biomedical application of graphene: From drug delivery, tumor therapy, to theranostics. Colloids Surf. B Biointerfaces 2020, 185, 110596. [Google Scholar] [CrossRef] [PubMed]
- Lage, T.; Rodrigues, R.O.; Catarino, S.; Gallo, J.; Bañobre-López, M.; Minas, G. Graphene-Based Magnetic Nanoparticles for Theranostics: An Overview for Their Potential in Clinical Application. Nanomaterials 2021, 11, 1073. [Google Scholar] [CrossRef]
- Volkov, Y.; McIntyre, J.; Prina-Mello, A. Graphene toxicity as a double-edged sword of risks and exploitable opportunities: A critical analysis of the most recent trends and developments. 2D Mater. 2017, 4, 022001. [Google Scholar] [CrossRef]
- Lalwani, G.; D’Agati, M.; Khan, A.M.; Sitharaman, B. Toxicology of graphene-based nanomaterials. Adv. Drug Deliv. Rev. 2016, 105, 109–144. [Google Scholar] [CrossRef] [Green Version]
- Guo, X.; Mei, N. Assessment of the toxic potential of graphene family nanomaterials. J. Food Drug Anal. 2014, 22, 105–115. [Google Scholar] [CrossRef] [Green Version]
- Achawi, S.; Feneon, B.; Pourchez, J.; Forest, V. Structure–Activity Relationship of Graphene-Based Materials: Impact of the Surface Chemistry, Surface Specific Area and Lateral Size on Their In Vitro Toxicity. Nanomaterials 2021, 11, 2963. [Google Scholar] [CrossRef]
- Ullah, S.; Chen, X. Fabrication, applications and challenges of natural biomaterials in tissue engineering. Appl. Mater. Today 2020, 20, 100656. [Google Scholar] [CrossRef]
- Klimek, K.; Ginalska, G. Proteins and Peptides as Important Modifiers of the Polymer Scaffolds for Tissue Engineering Applications—A Review. Polymers 2020, 12, 844. [Google Scholar] [CrossRef] [Green Version]
- Shoulders, M.D.; Raines, R.T. Collagen structure and stability. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koeck, K.; Salehi, S.; Humenik, M.; Scheibel, T. Processing of Continuous Non-Crosslinked Collagen Fibers for Microtissue Formation at the Muscle-Tendon Interface. Adv. Funct. Mater. 2021, 2112238. [Google Scholar] [CrossRef]
- Xie, Y.; Kawazoe, N.; Yang, Y.; Chen, G. Preparation of mesh-like collagen scaffolds for tissue engineering. Mater. Adv. 2022, 3, 1556–1564. [Google Scholar] [CrossRef]
- Hayat, U.; Raza, A.; Bilal, M.; Iqbal, H.M.N.; Wang, J.-Y. Biodegradable polymeric conduits: Platform materials for guided nerve regeneration and vascular tissue engineering. J. Drug Deliv. Sci. Technol. 2022, 67, 103014. [Google Scholar] [CrossRef]
- Filippi, M.; Born, G.; Chaaban, M.; Scherberich, A. Natural Polymeric Scaffolds in Bone Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 474. [Google Scholar] [CrossRef] [PubMed]
- Ebhodaghe, S.; Fattahi, F.; Ndibe, H.; Imanah, O. Emerging Fabrication Techniques for Engineering Extracellular Matrix Biomimetic Materials. 2021, 2021110389. Preprints. Available online: https://www.preprints.org/manuscript/202111.0389/v1 (accessed on 24 January 2022).
- Tejo Otero, A.; Ritchie, A. Biological and mechanical evaluation of mineralized-hydrogel scaffolds for tissue engineering applications. J. Biomater. Appl. 2021, 36, 460–473. [Google Scholar] [CrossRef]
- Lenselink, E. Role of fibronectin in normal wound healing. Int. Wound J. 2013, 12, 313–316. [Google Scholar] [CrossRef]
- Feng, L.; Li, Y.; Zeng, W.; Xia, B.; Zhou, D.; Zhou, J. Enhancing effects of basic fibroblast growth factor and fibronectin on osteoblast adhesion to bone scaffolds for bone tissue engineering through extracellular matrix-integrin pathway. Exp. Ther. Med. 2017, 14, 6087–6092. [Google Scholar] [CrossRef]
- Nivison-Smith, L.; Rnjak, J.; Weiss, A.S. Synthetic human elastin microfibers: Stable cross-linked tropoelastin and cell interactive constructs for tissue engineering applications. Acta Biomater. 2010, 6, 354–359. [Google Scholar] [CrossRef]
- Shirzaei Sani, E.; Portillo-Lara, R.; Spencer, A.; Yu, W.; Geilich, B.M.; Noshadi, I.; Webster, T.J.; Annabi, N. Engineering Adhesive and Antimicrobial Hyaluronic Acid/Elastin-like Polypeptide Hybrid Hydrogels for Tissue Engineering Applications. ACS Biomater. Sci. Eng. 2018, 4, 2528–2540. [Google Scholar] [CrossRef] [Green Version]
- Biswal, T. Biopolymers for tissue engineering applications: A review. Mater. Today Proc. 2021, 41, 397–402. [Google Scholar] [CrossRef]
- Steglich, M.; Lombide, R.; López, I.; Portela, M.; Fló, M.; Marín, M.; Alvarez, B.; Turell, L. Expression, purification and initial characterization of human serum albumin domain I and its cysteine 34. PLoS ONE 2020, 15, e0240580. [Google Scholar] [CrossRef]
- Mishra, V.; Heath, R.J. Structural and Biochemical Features of Human Serum Albumin Essential for Eukaryotic Cell Culture. Int. J. Mol. Sci. 2021, 22, 8411. [Google Scholar] [CrossRef]
- Fu, L.; Li, N.; Ye, Y.; Ye, X.; Xiao, T.; Wu, X.; Ma, Y.; Yu, J. MicroRNA Hsa-Let-7b Regulates the Osteogenic Differentiation of Human Periodontal Ligament Stem Cells by Targeting CTHRC1. Stem Cells Int. 2021, 2021, 5791181. [Google Scholar] [CrossRef] [PubMed]
- Jahanban-Esfahlan, A.; Ostadrahimi, A.; Jahanban-Esfahlan, R.; Roufegarinejad, L.; Tabibiazar, M.; Amarowicz, R. Recent developments in the detection of bovine serum albumin. Int. J. Biol. Macromol. 2019, 138, 602–617. [Google Scholar] [CrossRef]
- Moghassemi, S.; Hadjizadeh, A.; Omidfar, K. Formulation and Characterization of Bovine Serum Albumin-Loaded Niosome. AAPS PharmSciTech 2017, 18, 27–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, T.; Wang, L.; Huang, Y.; Xin, B.; Liu, S. BSA loaded bead-on-string nanofiber scaffold with core-shell structure applied in tissue engineering. J. Biomater. Sci. Polym. Ed. 2020, 31, 1223–1236. [Google Scholar] [CrossRef]
- Yi, S.; Ding, F.; Gong, L.; Gu, X. Extracellular Matrix Scaffolds for Tissue Engineering and Regenerative Medicine. Curr. Stem Cell Res. Ther. 2017, 12, 233–246. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, T.L.; Toh, W.S.; Pei, M. The role of laminins in cartilaginous tissues: From development to regeneration. Eur. Cell Mater. 2017, 34, 40–54. [Google Scholar] [CrossRef]
- Barros, D.; Amaral, I.F.; Pêgo, A.P. Laminin-Inspired Cell-Instructive Microenvironments for Neural Stem Cells. Biomacromolecules 2020, 21, 276–293. [Google Scholar] [CrossRef]
- Setiawati, A.; Nguyen, H.; Shin, K. Future Research Directions in the Design of Versatile Extracellular Matrix in Tissue Engineering. Int. Neurourol. J. 2020, 22, S66–S75. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.P.; Pundir, S.; Sharma, A. Bone morphogenetic proteins: The anomalous molecules. J. Indian Soc. Periodontol. 2013, 17, 583–586. [Google Scholar] [CrossRef] [PubMed]
- El Bialy, I.; Jiskoot, W.; Reza Nejadnik, M. Formulation, Delivery and Stability of Bone Morphogenetic Proteins for Effective Bone Regeneration. Pharm. Res. 2017, 34, 1152–1170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A Review of the Clinical Side Effects of Bone Morphogenetic Protein-2. Tissue Eng. Part B Rev. 2016, 22, 284–297. [Google Scholar] [CrossRef]
- Benington, L.; Rajan, G.; Locher, C.; Lim, L.Y. Fibroblast Growth Factor 2—A Review of Stabilisation Approaches for Clinical Applications. Pharmaceutics 2020, 12, 508. [Google Scholar] [CrossRef]
- Rubert Pérez, C.M.; Álvarez, Z.; Chen, F.; Aytun, T.; Stupp, S.I. Mimicking the Bioactivity of Fibroblast Growth Factor-2 Using Supramolecular Nanoribbons. ACS Biomater. Sci. Eng. 2017, 3, 2166–2175. [Google Scholar] [CrossRef] [Green Version]
- Hoshiba, T. Decellularized Extracellular Matrix for Cancer Research. Materials 2019, 12, 1311. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Chen, X.; Hong, H.; Hu, R.; Liu, J.; Liu, C. Decellularized extracellular matrix scaffolds: Recent trends and emerging strategies in tissue engineering. Bioact. Mater. 2022, 10, 15–31. [Google Scholar] [CrossRef]
- Oliver-Cervelló, L.; Martin-Gómez, H.; Mas-Moruno, C. New trends in the development of multifunctional peptides to functionalize biomaterials. J. Pept. Sci. 2021, 28, e3335. [Google Scholar] [CrossRef]
- Simsikova, M.; Sikola, T. Interaction of Graphene Oxide with Proteins and Applications of their Conjugates. J. Nanomed. Res. 2017, 5, 1–4. [Google Scholar] [CrossRef]
- Lieberman, R.L. How does a protein’s structure spell the difference between health and disease? Our journey to understand glaucoma-associated myocilin. PLoS Biol. 2019, 17, e3000237. [Google Scholar] [CrossRef] [PubMed]
- Savojardo, C.; Babbi, G.; Martelli, P.L.; Casadio, R. Functional and Structural Features of Disease-Related Protein Variants. Int. J. Mol. Sci. 2019, 20, 1530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhen, Z.; Zhu, H. 1—Structure and Properties of Graphene. In Graphene; Zhu, H., Xu, Z., Xie, D., Fang, Y., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 1–12. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhou, S.; Hu, P.; Zhao, G.; Li, Y.; Zhang, X.; Han, W. Enhanced mechanical, thermal, and electric properties of graphene aerogels via supercritical ethanol drying and high-temperature thermal reduction. Sci. Rep. 2017, 7, 1439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papageorgiou, D.G.; Kinloch, I.A.; Young, R.J. Mechanical properties of graphene and graphene-based nanocomposites. Prog. Mater. Sci. 2017, 90, 75–127. [Google Scholar] [CrossRef]
- Wassei, J.K.; Kaner, R.B. Graphene, a promising transparent conductor. Mater. Today 2010, 13, 52–59. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, H.; Liu, J.; Bao, C. Measuring the specific surface area of monolayer graphene oxide in water. Mater. Lett. 2020, 261, 127098. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Zhou, C. Review of Chemical Vapor Deposition of Graphene and Related Applications. Acc. Chem. Res. 2013, 46, 2329–2339. [Google Scholar] [CrossRef]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Achee, T.C.; Sun, W.; Hope, J.T.; Quitzau, S.G.; Sweeney, C.B.; Shah, S.A.; Habib, T.; Green, M.J. High-yield scalable graphene nanosheet production from compressed graphite using electrochemical exfoliation. Sci. Rep. 2018, 8, 14525. [Google Scholar] [CrossRef]
- Zaaba, N.I.; Foo, K.L.; Hashim, U.; Tan, S.J.; Liu, W.-W.; Voon, C.H. Synthesis of Graphene Oxide using Modified Hummers Method: Solvent Influence. Procedia Eng. 2017, 184, 469–477. [Google Scholar] [CrossRef]
- Mouhat, F.; Coudert, F.-X.; Bocquet, M.-L. Structure and chemistry of graphene oxide in liquid water from first principles. Nat. Commun. 2020, 11, 1566. [Google Scholar] [CrossRef] [PubMed]
- Goenka, S.; Sant, V.; Sant, S. Graphene-based nanomaterials for drug delivery and tissue engineering. J. Control. Release 2014, 173, 75–88. [Google Scholar] [CrossRef]
- Riley, P.R.; Joshi, P.; Penchev, H.; Narayan, J.; Narayan, R.J. One-Step Formation of Reduced Graphene Oxide from Insulating Polymers Induced by Laser Writing Method. Crystals 2021, 11, 1308. [Google Scholar] [CrossRef]
- Park, S.; An, J.; Potts, J.R.; Velamakanni, A.; Murali, S.; Ruoff, R.S. Hydrazine-reduction of graphite- and graphene oxide. Carbon 2011, 49, 3019–3023. [Google Scholar] [CrossRef]
- De Silva, K.K.H.; Huang, H.-H.; Yoshimura, M. Progress of reduction of graphene oxide by ascorbic acid. Appl. Surf. Sci. 2018, 447, 338–346. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, H.; Shen, G.; Cheng, P.; Zhang, J.; Guo, S. Reduction of graphene oxide vial-ascorbic acid. Chem. Commun. 2010, 46, 1112–1114. [Google Scholar] [CrossRef]
- Lingaraju, K.; Raja Naika, H.; Nagaraju, G.; Nagabhushana, H. Biocompatible synthesis of reduced graphene oxide from Euphorbia heterophylla (L.) and their in-vitro cytotoxicity against human cancer cell lines. Biotechnol. Rep. 2019, 24, e00376. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, L.; Li, X.; Zeng, J.; Jia, X.; Liu, P. Biocompatible Graphene Oxide Nanoparticle-Based Drug Delivery Platform for Tumor Microenvironment-Responsive Triggered Release of Doxorubicin. Langmuir 2014, 30, 10419–10429. [Google Scholar] [CrossRef]
- Karimi Hajishoreh, N.; Baheiraei, N.; Naderi, N.; Salehnia, M. Reduced graphene oxide facilitates biocompatibility of alginate for cardiac repair. J. Bioact. Compat. Polym. 2020, 35, 363–377. [Google Scholar] [CrossRef]
- Chaudhary, K.; Kumar, K.; Venkatesu, P.; Masram, D.T. Protein immobilization on graphene oxide or reduced graphene oxide surface and their applications: Influence over activity, structural and thermal stability of protein. Adv. Colloid Interface Sci. 2021, 289, 102367. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhu, Z.; Wang, Y.; Fei, Z.; Cao, J. Changing the activities and structures of bovine serum albumin bound to graphene oxide. Appl. Surf. Sci. 2018, 427, 1019–1029. [Google Scholar] [CrossRef]
- Wu, C.; He, Q.; Zhu, A.; Yang, H.; Liu, Y. Probing the protein conformation and adsorption behaviors in nanographene oxide-protein complexes. J. Nanosci. Nanotechnol. 2014, 14, 2591–2598. [Google Scholar] [CrossRef]
- Hampitak, P.; Melendrez, D.; Iliut, M.; Fresquet, M.; Parsons, N.; Spencer, B.; Jowitt, T.A.; Vijayaraghavan, A. Protein interactions and conformations on graphene-based materials mapped using a quartz-crystal microbalance with dissipation monitoring (QCM-D). Carbon 2020, 165, 317–327. [Google Scholar] [CrossRef]
- Bai, Y.; Ming, Z.; Cao, Y.; Feng, S.; Yang, H.; Chen, L.; Yang, S.T. Influence of graphene oxide and reduced graphene oxide on the activity and conformation of lysozyme. Colloids Surf. B Biointerfaces 2017, 154, 96–103. [Google Scholar] [CrossRef]
- Bera, S.; Dhar, J.; Dasgupta, R.; Basu, G.; Chakraborti, S.; Chakrabarti, P. Molecular features of interaction involving hen egg white lysozyme immobilized on graphene oxide and the effect on activity. Int. J. Biol. Macromol. 2018, 120, 2390–2398. [Google Scholar] [CrossRef]
- Kumar, S.; Chatterjee, K. Comprehensive Review on the Use of Graphene-Based Substrates for Regenerative Medicine and Biomedical Devices. ACS Appl. Mater. Interfaces 2016, 8, 26431–26457. [Google Scholar] [CrossRef]
- Eckhart, K.E.; Schmidt, S.J.; Starvaggi, F.A.; Wolf, M.E.; Vickery, W.M.; Sydlik, S.A. Peptide- and Protein-Graphene Oxide Conjugate Materials for Controlling Mesenchymal Stem Cell Fate. Regen. Eng. Transl. Med. 2021, 7, 460–484. [Google Scholar] [CrossRef]
- Su, R.; Shi, P.; Zhu, M.; Hong, F.; Li, D. Studies on the properties of graphene oxide–alkaline protease bio-composites. Bioresour. Technol. 2012, 115, 136–140. [Google Scholar] [CrossRef]
- Onaș, A.M.; Bîru, I.E.; Gârea, S.A.; Iovu, H. Novel Bovine Serum Albumin Protein Backbone Reassembly Study: Strongly Twisted β-Sheet Structure Promotion upon Interaction with GO-PAMAM. Polymers 2020, 12, 2603. [Google Scholar] [CrossRef]
- Hermanová, S.; Zarevúcká, M.; Bouša, D.; Pumera, M.; Sofer, Z. Graphene oxide immobilized enzymes show high thermal and solvent stability. Nanoscale 2015, 7, 5852–5858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ligorio, C.; O’Brien, M.; Hodson, N.W.; Mironov, A.; Iliut, M.; Miller, A.F.; Vijayaraghavan, A.; Hoyland, J.A.; Saiani, A. TGF-β3-loaded graphene oxide—Self-assembling peptide hybrid hydrogels as functional 3D scaffolds for the regeneration of the nucleus pulposus. Acta Biomater 2021, 127, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Sun, J.; Liu, G.; Li, L.; Wu, S.; Xiang, Z. Graphene oxide-modified 3D acellular cartilage extracellular matrix scaffold for cartilage regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 119, 111603. [Google Scholar] [CrossRef] [PubMed]
- Borges-Vilches, J.; Figueroa, T.; Guajardo, S.; Aguayo, C.; Fernández, K. Improved hemocompatibility for gelatin-graphene oxide composite aerogels reinforced with proanthocyanidins for wound dressing applications. Colloids Surf. B Biointerfaces 2021, 206, 111941. [Google Scholar] [CrossRef]
- Ryu, S.B.; Park, K.M.; Park, K.D. In situ graphene oxide-gelatin hydrogels with enhanced mechanical property for tissue adhesive and regeneration. Biochem. Biophys. Res. Commun. 2022, 592, 24–30. [Google Scholar] [CrossRef]
- Patel, M.; Moon, H.J.; Ko, D.Y.; Jeong, B. Composite System of Graphene Oxide and Polypeptide Thermogel as an Injectable 3D Scaffold for Adipogenic Differentiation of Tonsil-Derived Mesenchymal Stem Cells. ACS Appl. Mater. Interfaces 2016, 8, 5160–5169. [Google Scholar] [CrossRef]
- Kang, M.S.; Kang, J.I.; Le Thi, P.; Park, K.M.; Hong, S.W.; Choi, Y.S.; Han, D.-W.; Park, K.D. Three-Dimensional Printable Gelatin Hydrogels Incorporating Graphene Oxide to Enable Spontaneous Myogenic Differentiation. ACS Macro Lett. 2021, 10, 426–432. [Google Scholar] [CrossRef]
- Fang, X.; Guo, H.; Zhang, W.; Fang, H.; Li, Q.; Bai, S.; Zhang, P. Reduced graphene oxide-GelMA-PCL hybrid nanofibers for peripheral nerve regeneration. J. Mater. Chem. B 2020, 8, 10593–10601. [Google Scholar] [CrossRef]
- Ghitman, J.; Biru, E.I.; Cojocaru, E.; Pircalabioru, G.G.; Vasile, E.; Iovu, H. Design of new bioinspired GO-COOH decorated alginate/gelatin hybrid scaffolds with nanofibrous architecture: Structural, mechanical and biological investigations. RSC Adv. 2021, 11, 13653–13665. [Google Scholar] [CrossRef]
- Aidun, A.; Safaei Firoozabady, A.; Moharrami, M.; Ahmadi, A.; Haghighipour, N.; Bonakdar, S.; Faghihi, S. Graphene oxide incorporated polycaprolactone/chitosan/collagen electrospun scaffold: Enhanced osteogenic properties for bone tissue engineering. Artif. Organs 2019, 43, E264–E281. [Google Scholar] [CrossRef]
- Shin, Y.C.; Kim, J.; Kim, S.E.; Song, S.-J.; Hong, S.W.; Oh, J.-W.; Lee, J.; Park, J.-C.; Hyon, S.-H.; Han, D.-W. RGD peptide and graphene oxide co-functionalized PLGA nanofiber scaffolds for vascular tissue engineering. Regen. Biomater. 2017, 4, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Park, J.B.; Lee, T.-J.; Ryu, S.; Bhang, S.H.; La, W.-G.; Noh, M.-K.; Hong, B.H.; Kim, B.-S. Covalent conjugation of mechanically stiff graphene oxide flakes to three-dimensional collagen scaffolds for osteogenic differentiation of human mesenchymal stem cells. Carbon 2015, 83, 162–172. [Google Scholar] [CrossRef]
- Shin, Y.C.; Song, S.-J.; Lee, J.; Park, R.; Kang, M.; Lee, Y.; Hong, S.W.; Han, D.-W. Different Alignment Between Skeletal and Smooth Muscle Cells on Reduced Graphene Oxide-Patterned Arrays. Sci. Adv. Mater. 2020, 12, 474–480. [Google Scholar] [CrossRef]
- Zhou, C.; Liu, S.; Li, J.; Guo, K.; Yuan, Q.; Zhong, A.; Yang, J.; Wang, J.; Sun, J.; Wang, Z. Collagen Functionalized With Graphene Oxide Enhanced Biomimetic Mineralization and in Situ Bone Defect Repair. ACS Appl. Mater. Interfaces 2018, 10, 44080–44091. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Kim, H.-M.; Kawashita, M. Novel bioactive materials with different mechanical properties. Biomaterials 2003, 24, 2161–2175. [Google Scholar] [CrossRef]
- Fang, H.; Luo, C.; Liu, S.; Zhou, M.; Zeng, Y.; Hou, J.; Chen, L.; Mou, S.; Sun, J.; Zhenxing, W. A biocompatible vascularized graphene oxide (GO)-collagen chamber with osteoinductive and anti-fibrosis effects promotes bone regeneration in vivo. Theranostics 2020, 10, 2759–2772. [Google Scholar] [CrossRef]
- Yap, K.K.; Yeoh, G.C.; Morrison, W.A.; Mitchell, G.M. The Vascularised Chamber as an In Vivo Bioreactor. Trends Biotechnol. 2018, 36, 1011–1024. [Google Scholar] [CrossRef]
- Askari, E.; Naghib, S.M.; Zahedi, A.; Seyfoori, A.; Zare, Y.; Rhee, K.Y. Local delivery of chemotherapeutic agent in tissue engineering based on gelatin/graphene hydrogel. J. Mater. Res. Technol. 2021, 12, 412–422. [Google Scholar] [CrossRef]
- Mahdavi, R.; Belgheisi, G.; Haghbin-Nazarpak, M.; Omidi, M.; Khojasteh, A.; Solati-Hashjin, M. Bone tissue engineering gelatin-hydroxyapatite/graphene oxide scaffolds with the ability to release vitamin D: Fabrication, characterization, and in vitro study. J. Mater. Sci. Mater. Med. 2020, 31, 97. [Google Scholar] [CrossRef]
- Zhu, J.; Qi, Z.; Zheng, C.; Xue, P.; Fu, C.; Pan, S.; Yang, X. Enhanced Cell Proliferation and Osteogenesis Differentiation through a Combined Treatment of Poly-L-Lysine-Coated PLGA/Graphene Oxide Hybrid Fiber Matrices and Electrical Stimulation. J. Nanomater. 2020, 2020, 5892506. [Google Scholar] [CrossRef]
- Liu, S.; Mou, S.; Zhou, C.; Guo, L.; Zhong, A.; Yang, J.; Yuan, Q.; Wang, J.; Sun, J.; Wang, Z. Off-the-Shelf Biomimetic Graphene Oxide–Collagen Hybrid Scaffolds Wrapped with Osteoinductive Extracellular Matrix for the Repair of Cranial Defects in Rats. ACS Appl. Mater. Interfaces 2018, 10, 42948–42958. [Google Scholar] [CrossRef] [PubMed]
- Zuluaga-Vélez, A.; Quintero-Martinez, A.; Orozco, L.M.; Sepúlveda-Arias, J.C. Silk fibroin nanocomposites as tissue engineering scaffolds—A systematic review. Biomed. Pharmacother. 2021, 141, 111924. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Li, P.; Fan, Y. The assembly of silk fibroin and graphene-based nanomaterials with enhanced mechanical/conductive properties and their biomedical applications. J. Mater. Chem. B 2019, 7, 6890–6913. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, X.; Liu, A.; Pan, C.; Ding, H.; Ye, W. Reduced graphene oxide/titanium dioxide hybrid nanofiller-reinforced electrospun silk fibroin scaffolds for tissue engineering. Mater. Lett. 2021, 291, 129563. [Google Scholar] [CrossRef]
- Mohammadzadehmoghadam, S.; Dong, Y. 4—Electrospinning of silk fibroin-based nanofibers and their applications in tissue engineering. In Electrospun Polymers and Composites; Dong, Y., Baji, A., Ramakrishna, S., Eds.; Woodhead Publishing: Cambridge, UK, 2021; pp. 111–146. [Google Scholar] [CrossRef]
- Wu, J.; Zheng, A.; Liu, Y.; Jiao, D.; Zeng, D.; Wang, X.; Cao, L.; Jiang, X. Enhanced bone regeneration of the silk fibroin electrospun scaffolds through the modification of the graphene oxide functionalized by BMP-2 peptide. Int. J. Nanomed. 2019, 14, 733–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Wang, X.; Fan, S.; Lan, P.; Cao, C.; Zhang, Y. Silk fibroin/reduced graphene oxide composite mats with enhanced mechanical properties and conductivity for tissue engineering. Colloids Surf. B Biointerfaces 2021, 197, 111444. [Google Scholar] [CrossRef]
- Wang, L.; Lu, R.; Hou, J.; Nan, X.; Xia, Y.; Guo, Y.; Meng, K.; Xu, C.; Wang, X.; Zhao, B. Application of injectable silk fibroin/graphene oxide hydrogel combined with bone marrow mesenchymal stem cells in bone tissue engineering. Colloids Surf. A Physicochem. Eng. Asp. 2020, 604, 125318. [Google Scholar] [CrossRef]
- Cheng, J.; Liu, J.; Wu, B.; Liu, Z.; Li, M.; Wang, X.; Tang, P.; Wang, Z. Graphene and its Derivatives for Bone Tissue Engineering: In Vitro and In Vivo Evaluation of Graphene-Based Scaffolds, Membranes and Coatings. Front. Bioeng. Biotechnol. 2021, 9, 734688. [Google Scholar] [CrossRef]
- Pathmanapan, S.; Periyathambi, P.; Anandasadagopan, S.K. Fibrin hydrogel incorporated with graphene oxide functionalized nanocomposite scaffolds for bone repair—In vitro and in vivo study. Nanomedicine 2020, 29, 102251. [Google Scholar] [CrossRef]
- Nguyen, A.H.; Marsh, P.; Schmiess-Heine, L.; Burke, P.J.; Lee, A.; Lee, J.; Cao, H. Cardiac tissue engineering: State-of-the-art methods and outlook. J. Biol. Eng. 2019, 13, 57. [Google Scholar] [CrossRef] [Green Version]
- Karperien, L.; Navaei, A.; Godau, B.; Dolatshahi-Pirouz, A.; Akbari, M.; Nikkhah, M. Chapter 5—Nanoengineered biomaterials for cardiac regeneration. In Nanoengineered Biomaterials for Regenerative Medicine; Mozafari, M., Rajadas, J., Kaplan, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 95–124. [Google Scholar] [CrossRef]
- Alagarsamy, K.N.; Mathan, S.; Yan, W.; Rafieerad, A.; Sekaran, S.; Manego, H.; Dhingra, S. Carbon nanomaterials for cardiovascular theranostics: Promises and challenges. Bioact. Mater. 2021, 6, 2261–2280. [Google Scholar] [CrossRef] [PubMed]
- Ul Haq, A.; Carotenuto, F.; Nardo, P.; Francini, R.; Prosposito, P.; Pescosolido, F.; De Matteis, F. Extrinsically Conductive Nanomaterials for Cardiac Tissue Engineering Applications. Micromachines 2021, 12, 914. [Google Scholar] [CrossRef] [PubMed]
- Valiente-Alandi, I.; Schafer, A.E.; Blaxall, B.C. Extracellular matrix-mediated cellular communication in the heart. J. Mol. Cell. Cardiol. 2016, 91, 228–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.P.; Qu, K.Y.; Zhou, B.; Zhang, F.; Wang, Y.Y.; Abodunrin, O.D.; Zhu, Z.; Huang, N.P. Electrical stimulation of neonatal rat cardiomyocytes using conductive polydopamine-reduced graphene oxide-hybrid hydrogels for constructing cardiac microtissues. Colloids Surf. B Biointerfaces 2021, 205, 111844. [Google Scholar] [CrossRef] [PubMed]
- Norahan, M.H.; Amroon, M.; Ghahremanzadeh, R.; Mahmoodi, M.; Baheiraei, N. Electroactive graphene oxide-incorporated collagen assisting vascularization for cardiac tissue engineering. J. Biomed. Mater. Res. Part A 2018, 107, 204–219. [Google Scholar] [CrossRef] [Green Version]
- Norahan, M.H.; Pourmokhtari, M.; Saeb, M.R.; Bakhshi, B.; Soufi Zomorrod, M.; Baheiraei, N. Electroactive cardiac patch containing reduced graphene oxide with potential antibacterial properties. Mater. Sci. Eng. C 2019, 104, 109921. [Google Scholar] [CrossRef]
- Zhao, G.; Qing, H.; Huang, G.; Genin, G.M.; Lu, T.J.; Luo, Z.; Xu, F.; Zhang, X. Reduced graphene oxide functionalized nanofibrous silk fibroin matrices for engineering excitable tissues. NPG Asia Mater. 2018, 10, 982–994. [Google Scholar] [CrossRef]
- Grijalvo, S.; Díaz, D.D. Graphene-based hybrid materials as promising scaffolds for peripheral nerve regeneration. Neurochem. Int. 2021, 147, 105005. [Google Scholar] [CrossRef]
- Magaz, A.; Li, X.; Gough, J.E.; Blaker, J.J. Graphene oxide and electroactive reduced graphene oxide-based composite fibrous scaffolds for engineering excitable nerve tissue. Mater. Sci. Eng. C 2021, 119, 111632. [Google Scholar] [CrossRef]
- Ajiteru, O.; Sultan, M.; Lee, Y.J.; Seo, Y.; Hong, H.; Lee, J.; Lee, H.; Suh, Y.; Ju, H.W.; Lee, O.J.; et al. A 3D Printable Electroconductive Biocomposite Bioink Based on Silk Fibroin-Conjugated Graphene Oxide. Nano Lett. 2020, 20, 6873–6883. [Google Scholar] [CrossRef]
- Meng, C.; Jiang, W.; Huang, Z.; Liu, T.; Feng, J. Fabrication of a Highly Conductive Silk Knitted Composite Scaffold by Two-Step Electrostatic Self-Assembly for Potential Peripheral Nerve Regeneration. ACS Appl. Mater. Interfaces 2020, 12, 12317–12327. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Mikos, A.G. Emerging strategies in reprogramming and enhancing the fate of mesenchymal stem cells for bone and cartilage tissue engineering. J. Control. Release 2021, 330, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Swanson, W.B.; Omi, M.; Zhang, Z.; Nam, H.K.; Jung, Y.; Wang, G.; Ma, P.X.; Hatch, N.E.; Mishina, Y. Macropore design of tissue engineering scaffolds regulates mesenchymal stem cell differentiation fate. Biomaterials 2021, 272, 120769. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Xu, J.; Pan, X.; Ding, Z.; Xie, H.; Wang, X.; Xie, H. Advances of adipose-derived mesenchymal stem cells-based biomaterial scaffolds for oral and maxillofacial tissue engineering. Bioact. Mater. 2021, 6, 2467–2478. [Google Scholar] [CrossRef]
- Augustine, R.; Dan, P.; Hasan, A.; Khalaf, I.M.; Prasad, P.; Ghosal, K.; Gentile, C.; McClements, L.; Maureira, P. Stem cell-based approaches in cardiac tissue engineering: Controlling the microenvironment for autologous cells. Biomed. Pharmacother. 2021, 138, 111425. [Google Scholar] [CrossRef]
- Cho, S.-W.; Kim, J.; Kim, Y.H. Nanotechnology for stem cell and tissue engineering. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar] [CrossRef]
- Levingstone, T.J.; Moran, C.; Almeida, H.V.; Kelly, D.J.; O’Brien, F.J. Layer-specific stem cell differentiation in tri-layered tissue engineering biomaterials: Towards development of a single-stage cell-based approach for osteochondral defect repair. Mater. Today Bio 2021, 12, 100173. [Google Scholar] [CrossRef]
- Godbey, W.T. (Ed.) Chapter 17—Stem cells, tissue engineering, and regenerative medicine. In Biotechnology and its Applications, 2nd ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 389–409. [Google Scholar] [CrossRef]
- Rezaei, A.; Aligholi, H.; Zeraatpisheh, Z.; Gholami, A.; Mirzaei, E. Collagen/chitosan-functionalized graphene oxide hydrogel provide a 3D matrix for neural stem/precursor cells survival, adhesion, infiltration and migration. J. Bioact. Compat. Polym. 2021, 36, 296–313. [Google Scholar] [CrossRef]
- Ligorio, C.; Zhou, M.; Wychowaniec, J.K.; Zhu, X.; Bartlam, C.; Miller, A.F.; Vijayaraghavan, A.; Hoyland, J.A.; Saiani, A. Graphene oxide containing self-assembling peptide hybrid hydrogels as a potential 3D injectable cell delivery platform for intervertebral disc repair applications. Acta Biomater. 2019, 92, 92–103. [Google Scholar] [CrossRef]
- Rueda-Gensini, L.; Serna, J.A.; Cifuentes, J.; Cruz, J.C.; Muñoz-Camargo, C. Graphene Oxide-Embedded Extracellular Matrix-Derived Hydrogel as a Multiresponsive Platform for 3D Bioprinting Applications. Int. J. Bioprint. 2021, 7, 353. [Google Scholar] [CrossRef]
- Duran, M.; Luzo, A.C.M.; de Souza, J.G.; Favaro, W.J.; Garcia, P.; Duran, N. Graphene Oxide as Scaffolds for Stem Cells: An Overview. Curr. Mol. Med. 2017, 17, 619–626. [Google Scholar] [CrossRef]
- Puah, P.Y.; Moh, P.; Sipaut, C.; Lee, P.; How, S.E. Peptide Conjugate on Multilayer Graphene Oxide Film for the Osteogenic Differentiation of Human Wharton’s Jelly-Derived Mesenchymal Stem Cells. Polymers 2021, 13, 3290. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Shi, M.; Yan, B.; Ma, H.; Li, N.; Hu, Y.; Ye, M. Covalent attaching protein to graphene oxide via diimide-activated amidation. Colloids Surf. B Biointerfaces 2010, 81, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, R.; Digiacomo, L.; Quagliarini, E.; Capriotti, A.L.; Laganà, A.; Zenezini Chiozzi, R.; Caputo, D.; Cascone, C.; Coppola, R.; Pozzi, D.; et al. Personalized Graphene Oxide-Protein Corona in the Human Plasma of Pancreatic Cancer Patients. Front. Bioeng. Biotechnol. 2020, 8, 491. [Google Scholar] [CrossRef] [PubMed]
- Zare, P.; Aleemardani, M.; Seifalian, A.; Bagher, Z.; Seifalian, A.M. Graphene Oxide: Opportunities and Challenges in Biomedicine. Nanomaterials 2021, 11, 1083. [Google Scholar] [CrossRef]
- Orsu, P.; Koyyada, A. Recent progresses and challenges in graphene based nano materials for advanced therapeutical applications: A comprehensive review. Mater. Today Commun. 2020, 22, 100823. [Google Scholar] [CrossRef]
- Ikram, R.; Shamsuddin, S.A.A.; Mohamed Jan, B.; Abdul Qadir, M.; Kenanakis, G.; Stylianakis, M.M.; Anastasiadis, S.H. Impact of Graphene Derivatives as Artificial Extracellular Matrices on Mesenchymal Stem Cells. Molecules 2022, 27, 379. [Google Scholar] [CrossRef]
- Pandit, S.; De, M. Roles of Edges and Surfaces of Graphene Oxide in Molecular Recognition of Proteins: Implications for Enzymatic Inhibition of α-Chymotrypsin. ACS Appl. Nano Mater. 2020, 3, 3829–3838. [Google Scholar] [CrossRef]
- Bullock, C.J.; Bussy, C. Biocompatibility Considerations in the Design of Graphene Biomedical Materials. Adv. Mater. Interfaces 2019, 6, 1900229. [Google Scholar] [CrossRef]
- Li, D.; Hu, X.; Zhang, S. Biodegradation of graphene-based nanomaterials in blood plasma affects their biocompatibility, drug delivery, targeted organs and antitumor ability. Biomaterials 2019, 202, 12–25. [Google Scholar] [CrossRef]



| Biocomplex | Formulation | Tissue Engineering Application | Conclusions | Ref. |
|---|---|---|---|---|
| GO/peptide FEFKFEFK (F: phenylalanine; K: lysine; E: glutamic acid) | Hydrogel | Nucleus pulposus (NP) regeneration | GO provides mechanical reinforcement to the hydrogel, facilitates cell adhesion, and can also load and deliver growth factors. | Ligorio et al. [106] |
| GO/acellular cartilage extracellular matrix | Scaffold | Cartilage tissue regeneration | Composite scaffolds showed increased biocompatibility and reduced inflammatory response after implantation and favored cartilage tissue regeneration. | Gong et al. [107] |
| GO/gelatin | Aerogel | Skin tissue regeneration/wound healing | The nanocomposite aerogel exhibits hemostatic activity and clogging properties suitable for wound dressing applications. | Borges-Vilches et al. [108] |
| GO/gelatin | Hydrogel | Tissue adhesive and regeneration | The synthesized formulation showed increased biocompatibility, high mechanical properties, and the ability to promote fibroblast proliferation. | Ryu et al. [109] |
| GO/poly L-alanine | Thermogel | Adipose tissue engineering | GO–peptide thermogel favored cell differentiation of seeded tonsil-derived mesenchymal stem cells. GO improved cell adhesion and acted as a carrier for growth factors. | Patel et al. [110] |
| GO/gelatin | Hydrogel | Skeletal muscle regeneration | Nanocomposite hydrogel favored the instinctive myogenic differentiation of C2C12 myoblasts without the aid of external growth factors. | Kang et al. [111] |
| GO/GelMA/PCL | Nanofibers | Peripheral nerve regeneration | rGO improved the mechanical and electrical properties of the formulation and, at lower concentration of about 0.25–0.5 wt%, enhanced Schwann cell (RSC96) proliferation. | Fang et al. [112] |
| GO/gelatin/alginate | Nanofibrous scaffolds | Skin tissue engineering | The hybrid electrospun scaffold comprising carboxylated GO exhibited increased biocompatibility and proved to be an appropriate environment for cell adhesion and proliferation. | Ghitman et al. [113] |
| GO/collagen/PCL/chitosan | Electrospun scaffold | Bone tissue engineering | The concentration of GO within the polymeric scaffold strongly influenced cell adhesion and proliferation, and the nanocomposite with a high ratio of GO showed the most increased osteogenic activity. | Aidun et al. [114] |
| GO/RGD peptide/PLGA | Nanofibrous mat | Vascular tissue engineering | The 3D structure of the electrospun network was similar to the ECM. The presence of both GO and RGD sequence favored cellular adhesion and proliferation. | Shin et al. [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biru, E.I.; Necolau, M.I.; Zainea, A.; Iovu, H. Graphene Oxide–Protein-Based Scaffolds for Tissue Engineering: Recent Advances and Applications. Polymers 2022, 14, 1032. https://doi.org/10.3390/polym14051032
Biru EI, Necolau MI, Zainea A, Iovu H. Graphene Oxide–Protein-Based Scaffolds for Tissue Engineering: Recent Advances and Applications. Polymers. 2022; 14(5):1032. https://doi.org/10.3390/polym14051032
Chicago/Turabian StyleBiru, Elena Iuliana, Madalina Ioana Necolau, Adriana Zainea, and Horia Iovu. 2022. "Graphene Oxide–Protein-Based Scaffolds for Tissue Engineering: Recent Advances and Applications" Polymers 14, no. 5: 1032. https://doi.org/10.3390/polym14051032
APA StyleBiru, E. I., Necolau, M. I., Zainea, A., & Iovu, H. (2022). Graphene Oxide–Protein-Based Scaffolds for Tissue Engineering: Recent Advances and Applications. Polymers, 14(5), 1032. https://doi.org/10.3390/polym14051032









