Recovery Observations from Alkali, Nanoparticles and Polymer Flooding as Combined Processes
Abstract
:1. Introduction
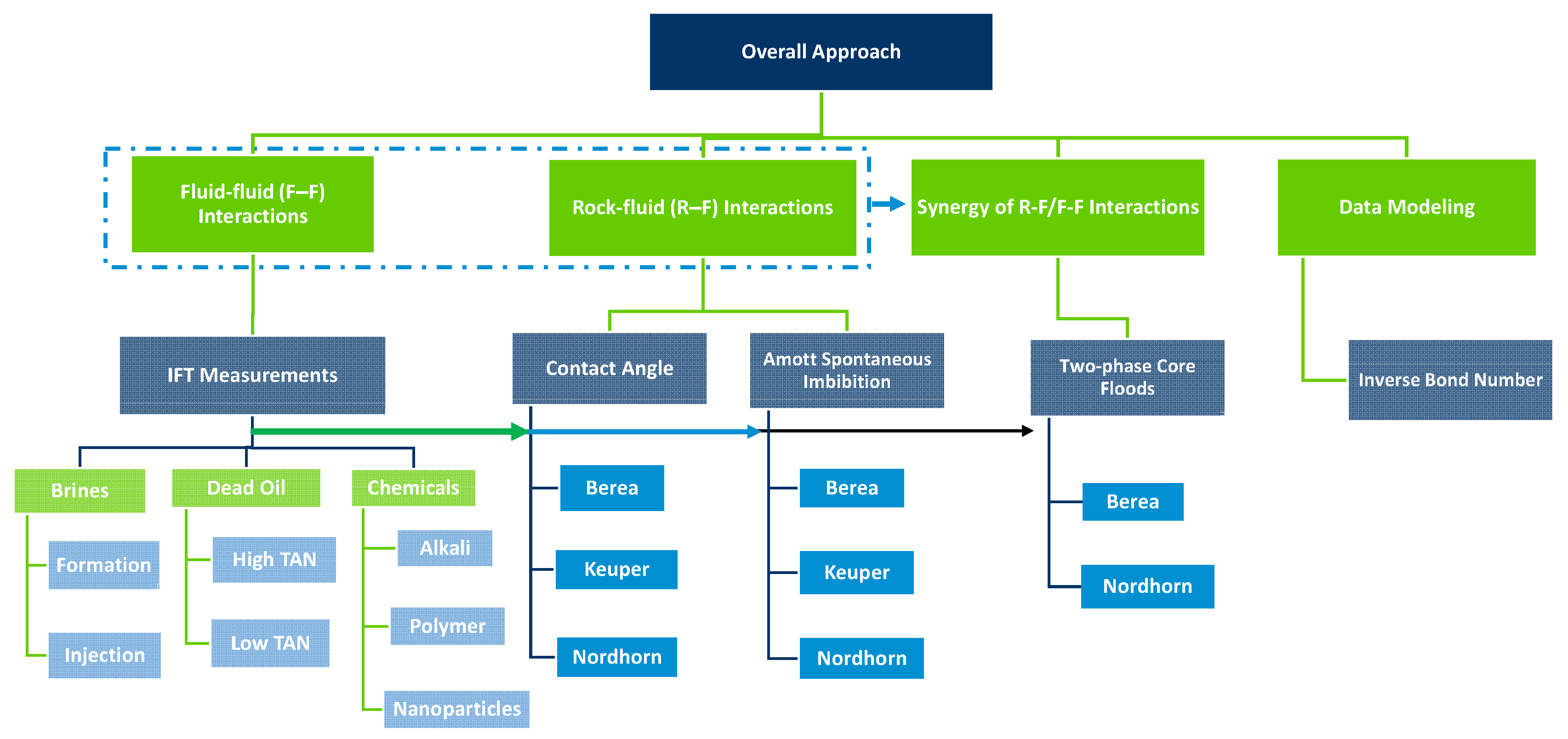
2. Overall Approach
3. Materials and Methods
3.1. Synthetic Brines
3.2. Crude Oils
3.3. Alkali, Polymer and Nanoparticles
3.4. Outcrop Samples
3.5. Interfacial Tension (IFT) Evaluations
3.6. Amott Spontaneous Imbibition Experiments
3.7. Contact Angle Experiments
3.8. Two-Phase Core Floods
3.9. Inverse Bond Number Modeling
3.10. Post Spontaneous Amott Imbibition Test—Rock Property Evaluation
4. Results and Discussion
4.1. Interfacial Tension (IFT) Evaluations
4.2. Contact Angle Observations
4.3. Amott Spontaneous Imbibition Experiments
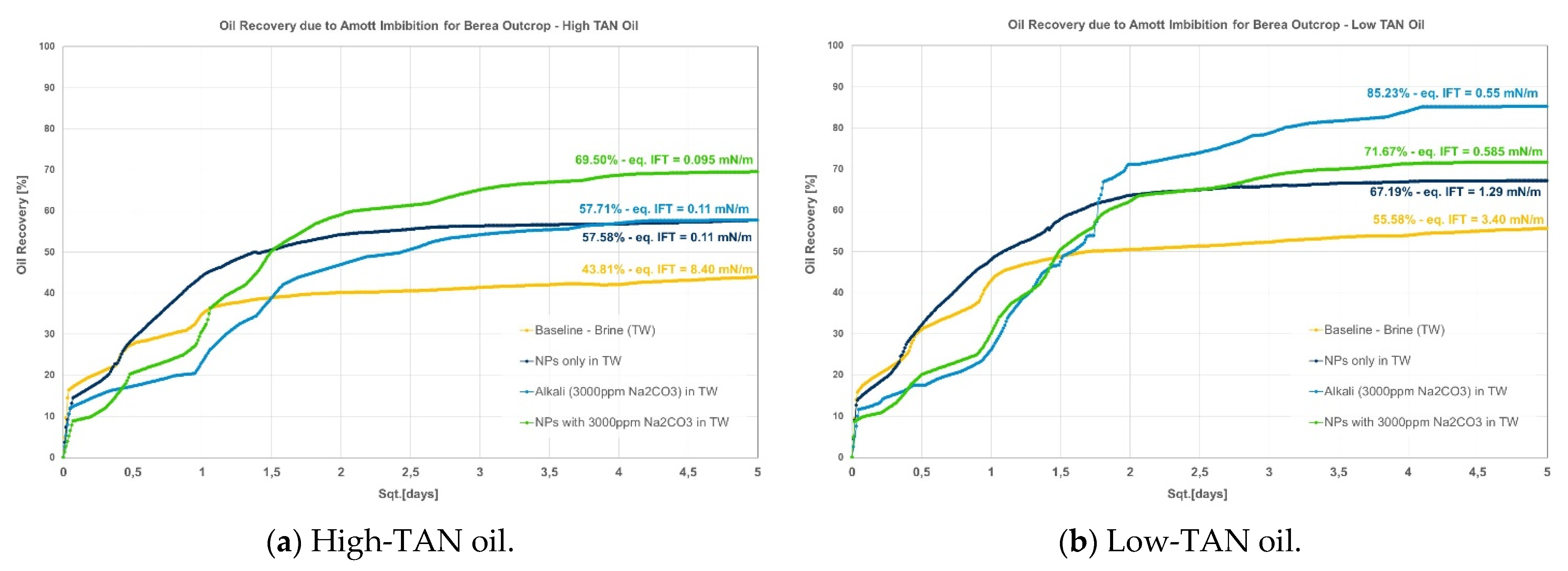
- Berea: it was observed that combining nanomaterials with alkali leads to the highest incremental oil, as shown in Figure 2. This is explained by acid–alkali reactions and the generation of in situ soaps, as reported by Saleh et al. [49]. The solution also depicted the lowest IFT of the compared group in Figure 2a for the high-TAN oil. Surprisingly, the contributions of each chemical fluid separately were much lower. In contrast, for the low-TAN oil, it was observed that alkali achieved the highest incremental oil recovery, followed by the nanomaterials and then the alkali;
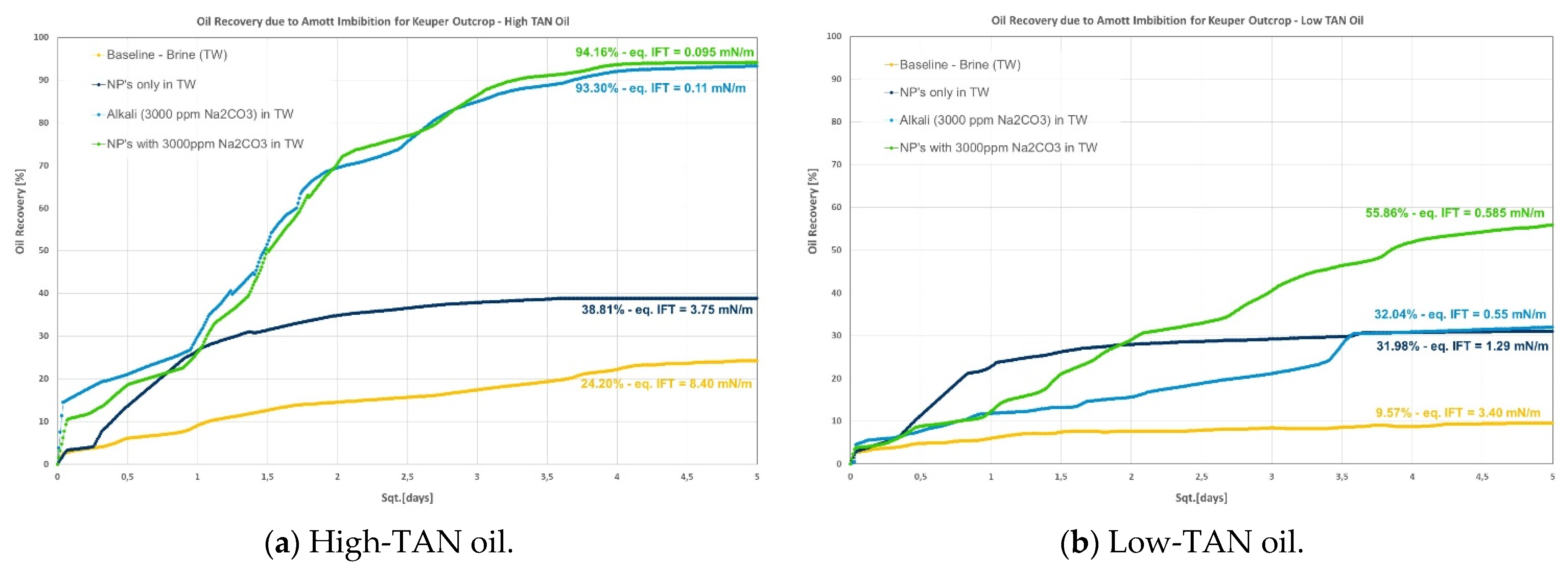
- Keuper: recoveries in this outcrop clearly presented a difference between high- and low-TAN oil, as shown in Figure 3. The highest recoveries were achieved with the combination of nanomaterials and alkali or by alkali alone. In addition to the IFT, the wettability nature of this outcrop (discussed in the contact angle section) helps explain the observations;
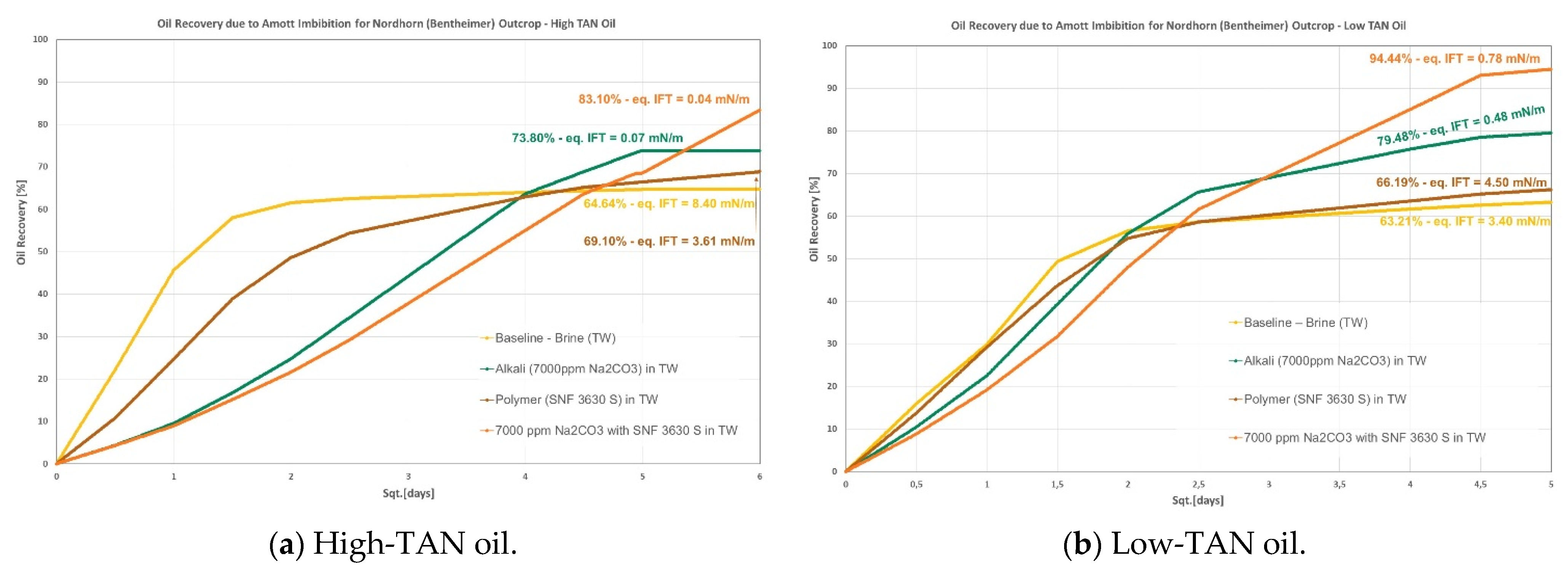
- Nordhorn (Bentheimer): it can be observed from Figure 4 that alkali in combination with a polymer on a similar slug leads to the highest incremental recovery in both oils. Observations were reported by Arekhov et al. [1] for the system with similar IFT and oil. The polymer alone did not lead to any further additional recovery greater than 5%, as shown in Table 6 and Table 7;
- As predicted in the contact angle section, Keuper plugs showed higher additional oil recoveries compared with Berea and Nordhorn core plugs for NPs in alkali chemical systems for both low-TAN and high-TAN oil. This significant additional oil recovery is due to the synergetic effect of NPs and alkali, and resulted in the IFT reduction and wettability alteration from oil-wet to water-wet systems.
4.4. Two-Phase Corefloods
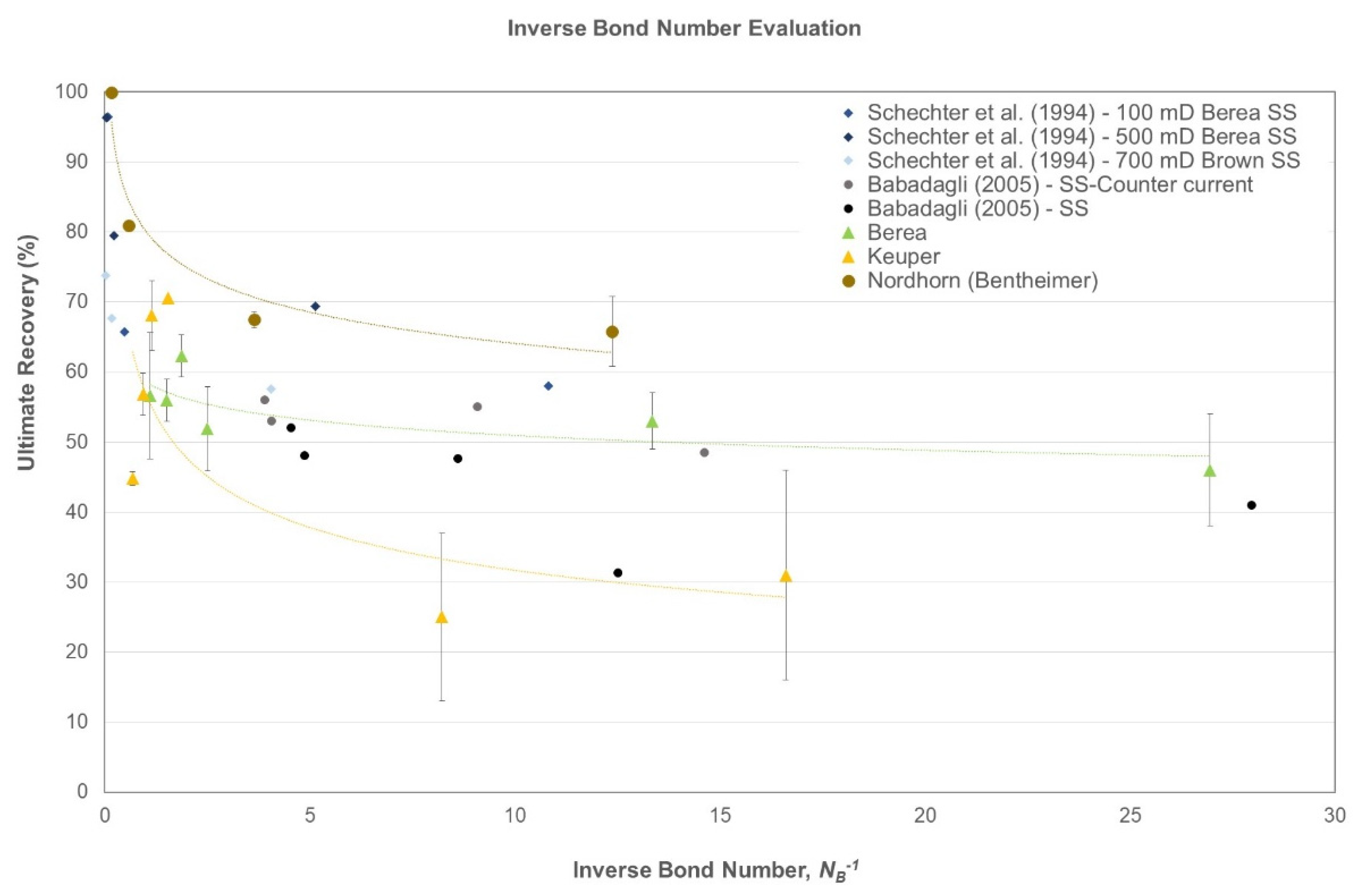
4.5. Data Modeling
- Berea: This outcrop exhibited general water-wet behavior and the data showed more consistency, even for the lower inverse bond numbers. As reported by Saleh et al. [49] and Neubauer et al. [46], nanomaterial usage indicates a change in wettability as one of the EOR mechanisms. However, the inverse bond number evaluation revealed that a significant amount of additional recovery results from improving the gravity drive by lowering the inverse bond number;
- Keuper: Data presented here fall within the general trend; the data tend to scatter at lower inverse bond numbers for oil-wet Keuper cores. We attribute this behavior to the change in wettability along with lowering of IFT. Moreover, we observed that once inverse bond number increased for baseline experiments, oil recovery dropped, which is in agreement with Schechter et al. [48]. The high standard deviation is attributed to Keuper core heterogeneity;
- Nordhorn (Bentheimer): The curve depicts the highest recovery values for this outcrop type. The general trend is followed, because once the inverse bond number decreases, the ultimate recovery drops.
4.6. Porosity and Permeability Changes
5. Summary and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arekhov, V.; Hincapie, R.E.; Clemens, T.; Tahir, M. Variations in Wettability and Interfacial Tension during Alkali–Polymer Application for High and Low TAN Oils. Polymers 2020, 12, 2241. [Google Scholar] [CrossRef]
- Sheng, J.J. Modern Chemical Enhanced Oil Recovery; Gulf Professional Publishing: Boston, MA, USA, 2011; p. 4. [Google Scholar] [CrossRef]
- Al-Saedi, H.N.; Flori, R.E.; Al-Jaberi, S.K. Eliminate the role of clay in sandstone: EOR low salinity water flooding. J. Pet. Explor. Prod. Technol. 2019, 9, 1475–1483. [Google Scholar] [CrossRef] [Green Version]
- Havre, T.E.; Sjöblom, J. Emulsion stabilization by means of combined surfactant multilayer (D-phase) and asphaltene particles. Colloids Surf. A Physicochem. Eng. Asp. 2003, 228, 131–142. [Google Scholar] [CrossRef]
- Horváth-Szabó, G.; Czarnecki, J.; Masliyah, J. Liquid crystals in aqueous solutions of sodium naphthenates. J. Colloid Interface Sci. 2001, 236, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.I.; Alvarado, V. Smart water flooding in berea sandstone at low temperature: Is wettability alteration the sole mechanism at play? In Proceedings of the SPE Annual Technical Conference and Exhibition, San Antonio, TX, USA, 9–11 October 2017. [CrossRef]
- Sullivan, A.P.; Zaki, N.N.; Sjöblom, J.; Kilpatrick, P.K. The stability of water-in-crude and model oil emulsions. Can. J. Chem. Eng. 2008, 85, 793–807. [Google Scholar] [CrossRef]
- Tahir, M.; Hincapie, R.E.; Langanke, N.; Ganzer, L.; Jaeger, P. Coupling microfluidics data with core flooding experiments to understand sulfonated/polymer water injection. Polymers 2020, 12, 1227. [Google Scholar] [CrossRef] [PubMed]
- Bagci, S.; Kok, M.V.; Turksoy, U. Effect of brine composition on oil recovery by waterflooding. Pet. Sci. Technol. 2001, 19, 359–372. [Google Scholar] [CrossRef]
- Bidhendi, M.M.; Garcia-Olvera, G.; Morin, B.; Oakey, J.S.; Alvarado, V. Interfacial viscoelasticity of crude oil/brine: An alternative enhanced-oil-recovery mechanism in smart waterflooding. SPE J. 2018, 23, 803–818. [Google Scholar] [CrossRef]
- Ligthelm, D.J.; Gronsveld, J.; Hofman, J.; Brussee, N.; Marcelis, F.; van der Linde, H. Novel waterflooding strategy by manipulation of injection brine composition. In Proceedings of the EUROPEC/EAGE Conference and Exhibition, Society of Petroleum Engineers, Amsterdam, The Netherlands, 8–11 June 2009. [Google Scholar] [CrossRef]
- Morrow, N.; Buckley, J. Improved oil recovery by low-salinity waterflooding. J. Pet. Technol. 2011, 63, 106–112. [Google Scholar] [CrossRef]
- Shaker Shiran, B.; Skauge, A. Wettability and oil recovery by low salinity injection. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Society of Petroleum Engineers, Muscat, Oman, 16–18 April 2012. [Google Scholar] [CrossRef] [Green Version]
- Webb, K.J.; Black, C.J.J.; Tjetland, G. A laboratory study investigating methods for improving oil recovery in carbonates. In Proceedings of the International Petroleum Technology Conference, Doha, Qatar, 21–23 November 2005. [Google Scholar] [CrossRef]
- Du, D.; Pu, W.; Jin, F.; Hou, D.-D.; Shi, L. Experimental investigation on plugging and transport characteristics of pore-scale microspheres in heterogeneous porous media for enhanced oil recovery. J. Dispers. Sci. Technol. 2021, 42, 1152–1162. [Google Scholar] [CrossRef]
- Sabooniha, E.; Rokhforouz, M.-R.; Ayatollahi, S. Pore-scale investigation of selective plugging mechanism in immiscible two-phase flow using phase-field method. Oil Gas Sci. Technol. Rev. IFP Energies Nouv. 2019, 74, 78. [Google Scholar] [CrossRef] [Green Version]
- Delamaide, E.; Zaitoun, A.; Renard, G.; Tabary, R. Pelican lake field: First successful application of polymer flooding in a heavy oil reservoir. In Proceedings of the SPE Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 2–4 July 2013. [Google Scholar] [CrossRef]
- Zhu, Y. Current developments and remaining challenges of chemical flooding EOR techniques in China. In Proceedings of the SPE Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 11–13 August 2015. [Google Scholar] [CrossRef]
- Kumar, P.; Raj, R.; Koduru, N.; Kumar, S.; Pandey, A. Field implementation of mangala polymer flood: Initial challenges, mitigation and management. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 21–23 March 2016. [Google Scholar] [CrossRef]
- Anand, A.; Al Sulaimani, H.; Riyami, O.; AlKindi, A. Success and challenges in ongoing field scale polymer flood in sultanate of Oman—A holistic reservoir simulation case study for polymer flood performance analysis & prediction. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 26–28 March 2018. [Google Scholar] [CrossRef]
- Sieberer, M.; Clemens, T.; Peisker, J.; Ofori, S. Polymer-flood field implementation: Pattern configuration and horizontal vs. vertical wells. SPE Reserv. Eval. Eng. 2019, 22, 577–596. [Google Scholar] [CrossRef]
- Watson, A.; Trahan, G.A.; Sorensen, W. An interim case study of an alkali-surfactant-polymer flood in the mooney field, Alberta, Canada. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 12–16 April 2014. [Google Scholar] [CrossRef]
- Yin, S.; Pu, H.; Zhou, S. An update on full field implementation of chemical flooding in Daqing Oilfield, China, and its future. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 14–18 April 2018. [Google Scholar] [CrossRef]
- Pitts, M.; Dean, E.; Wyatt, K.; Skeans, E.; Deo, D.; Galipeault, A.; Moohagen, D.; Humphry, C. Instow a full field, multi-patterned alkali-surfactant-polymer flood—Analysis and comparison of phases 1 and 2. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 18–22 April 2020. [Google Scholar] [CrossRef]
- Delamaide, E. Is chemical EOR finally coming of age? In Proceedings of the SPE Asia Pacific Oil & Gas Conference and Exhibition, Virtual, 17–19 November 2020. [CrossRef]
- Rock, A.; Hincapie, R.E.; Tahir, M.; Langanke, N.; Ganzer, L. On the role of polymer viscoelasticity in enhanced oil recovery: Extensive laboratory data and review. Polymers 2020, 12, 2276. [Google Scholar] [CrossRef]
- Sheng, J.; Leonhardt, B.; Al Azri, N.S. Status of polymer-flooding technology. J. Can. Pet. Technol. 2015, 54, 116–126. [Google Scholar] [CrossRef]
- Hincapie, R.E. Pore-Scale Investigation of the Viscoelastic Phenomenon during Enhanced Oil Recovery (EOR) Polymer Flooding through Porous Media; Papierflieger: Clausthal-Zellerfeld, Germany, 2016; ISBN 978-3-86948-531-7. [Google Scholar]
- Hashmet, M.R.; Qaiser, Y.; Mathew, E.S.; AlAmeri, W.; AlSumaiti, A. Injection of polymer for improved sweep efficiency in high temperature high salinity carbonate reservoirs: Linear X-ray aided flood front monitoring. In Proceedings of the SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition, Dammam, Saudi Arabia, 24–27 April 2017. [Google Scholar] [CrossRef]
- Castro-García, R.H.; Maya-Toro, G.A.; Jimenez-Diaz, R.; Quintero-Perez, H.I.; Díaz-Guardia, V.M.; Colmenares-Vargas, K.M.; Palma-Bustamante, J.M.; Delgadillo-Aya, C.L.; Pérez-Romero, R.A. Polymer flooding to improve volumetric sweep efficiency in waterflooding processes. CT&F Cienc. Tecnol. Futuro 2019, 6, 71–90. [Google Scholar]
- Al-Bahar, M.A.; Merrill, R.; Peake, W.; Jumaa, M.; Oskui, R. Evaluation of IOR potential within Kuwait. In Proceedings of the Abu Dhabi International Conference and Exhibition, Abu Dhabi, United Arab Emirates, 10–13 October 2004. [Google Scholar] [CrossRef]
- Berret, J.-F.; Yokota, K.; Morvan, M. Interactions between polymers and nanoparticles: Formation of “Supermicellar” hybrid aggregates. Soft Mater. 2004, 2, 71–84. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.K.; Son, H.A.; Kim, H.T.; Kim, J.W. Nanofluid enhanced oil recovery using hydrophobically associative zwitterionic polymer-coated silica nanoparticles. Energy Fuels 2017, 31, 7777–7782. [Google Scholar] [CrossRef]
- Galas, C.; Clements, A.; Jaafar, E.; Jeje, O.; Holst, D.; Holst, R.; Limited, S.A. Identification of Enhanced Oil Recovery Potential in Alberta: Phase 2 Final Report for Energy Resources Conservation Board; Sproule Associated Limited: Ottawa, ON, Canada, 2012. [Google Scholar]
- Maurya, N.; Mandal, A. Studies on behavior of suspension of silica nanoparticle in aqueous polyacrylamide solution for application in enhanced oil recovery. Pet. Sci. Technol. 2016, 34, 429–436. [Google Scholar] [CrossRef]
- Pitts, M.J.; Wyatt, K.; Surkalo, H. Alkaline-polymer flooding of the David Pool, Lloydminster Alberta. In Proceedings of the SPE/DOE Symposium on Improved Oil Recovery, Tulsa, OK, USA, 17–21 April 2004. [Google Scholar] [CrossRef]
- Wu, Y.; Dong, M.; Shirif, E. Study of Alkaline/Polymer Flooding for Heavy-Oil Recovery Using Channeled Sandpacks. SPE Reserv. Eval. Eng. 2011, 14, 310–319. [Google Scholar] [CrossRef]
- Clarke, A.; Howe, A.M.; Mitchell, J.; Staniland, J.; Hawkes, L.A. How viscoelastic polymer flooding enhances displacement efficiency. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Society of Petroleum Engineers, Kuala Lumpur, Malaysia, 11–13 August 2015. [Google Scholar] [CrossRef]
- Clarke, A.; Howe, A.M.; Mitchell, J.; Staniland, J.; Hawkes, L.; Leeper, K. Mechanism of anomalously increased oil displacement with aqueous viscoelastic polymer solutions. Soft Matter 2015, 11, 3536–3541. [Google Scholar] [CrossRef]
- Hincapie, R.E.; Rock, A.; Wegner, J.; Ganzer, L. Oil mobilization by viscoelastic flow instabilities effects during polymer EOR: A pore-scale visualization approach. In Proceedings of the SPE Latin America and Caribbean Petroleum Engineering Conference, Society of Petroleum Engineers, Buenos Aires, Argentina, 17–19 May 2017. [Google Scholar] [CrossRef]
- Sheng, J. Critical review of alkaline-polymer flooding. J. Pet. Explor. Prod. Technol. 2016, 7, 147–153. [Google Scholar] [CrossRef]
- Wang, D.; Wang, G.; Xia, H. Large scale high visco-elastic fluid flooding in the field achieves high recoveries. In Proceedings of the SPE Enhanced Oil Recovery Conference, Society of Petroleum Engineers, Kuala Lumpur, Malaysia, 19–20 July 2011. [Google Scholar] [CrossRef]
- Yin, D.; Zhao, N.; Gao, J.; Gai, J. Experimental study of enhancing oil recovery with weak base alkaline/surfactant/polymer. Int. J. Polym. Sci. 2017, 2017, 4652181. [Google Scholar] [CrossRef] [Green Version]
- Schumi, B.; Clemens, T.; Wegner, J.; Ganzer, L.; Kaiser, A.; Hincapie, R.E.; Leitenmueller, V. Alkali/cosolvent/polymer flooding of high-TAN oil: Using phase experiments, micromodels, and corefloods for injection-agent selection. SPE Reserv. Eval. Eng. 2020, 23, 463–478. [Google Scholar] [CrossRef]
- ShamsiJazeyi, H.; Miller, C.A.; Wong, M.S.; Tour, J.M.; Verduzco, R. Polymer-coated nanoparticles for enhanced oil recovery. J. Appl. Polym. Sci. 2014, 131, 15. [Google Scholar] [CrossRef]
- Neubauer, E.; Hincapie, R.E.; Borovina, A.; Biernat, M.; Clemens, T.; Ahmad, Y.K. Influence of nanofluids on wettability changes and interfacial tension reduction. In Proceedings of the SPE Europec, Virtual, 1–3 December 2020. [Google Scholar]
- Neubauer, E.; Hincapie, R.E.; Clemens, T.; Maximilian, C. Selection of nanomaterials as emulsion stabilizers in alkali-polymer EOR of high-TAN number oil. In Proceedings of the SPE Improved Oil Recovery Conference, Virtual, 31 August–4 September 2020. [Google Scholar]
- Schechter, D.; Zhou, D.; Orr, F. Low IFT drainage and imbibition. J. Pet. Sci. Eng. 1994, 11, 283–300. [Google Scholar] [CrossRef]
- Saleh, S.; Neubauer, E.; Borovina, A.; Hincapie, R.E.; Clemens, T.; Ness, D. Wettability changes due to nanomaterials and alkali—A proposed formulation for EOR. Nanomaterials 2021, 11, 2351. [Google Scholar] [CrossRef]
- Sharma, M.; Jang, L.; Yen, T. Transient interfacial tension behavior of crude-oil/caustic interfaces. SPE Reserv. Eng. 1989, 4, 228–236. [Google Scholar] [CrossRef]
- Al-Quraishi, A.A. Oil Recovery by Dynamic Imbibition in Low Tension Aqueous Systems. Oil Gas Sci. Technol. Rev. IFP Energies Nouv. 2004, 59, 267–273. [Google Scholar] [CrossRef]
- Babadagli, T. Analysis of oil recovery by spontaneous imbibition of surfactant solution. Oil Gas Sci. Technol. Rev. IFP Energies Nouv. 2005, 60, 697–710. [Google Scholar] [CrossRef] [Green Version]
- Kamal, M.S.; Adewunmi, A.A.; Sultan, A.S.; Al-Hamad, M.F.; Mehmood, U. Recent advances in nanoparticles enhanced oil recovery: Rheology, interfacial tension, oil recovery, and wettability alteration. J. Nanomater. 2017, 2017, e2473175. [Google Scholar] [CrossRef]
- Ershadi, M.; Alaei, M.; Rashidi, A.; Ramazani, A.; Khosravani, S. Carbonate and sandstone reservoirs wettability improvement without using surfactants for chemical enhanced oil recovery (C-EOR). Fuel 2015, 153, 408–415. [Google Scholar] [CrossRef]
- Bayat, A.E.; Junin, R.; Samsuri, A.; Piroozian, A.; Hokmabadi, M. Impact of Metal Oxide Nanoparticles on Enhanced Oil Recovery from Limestone Media at Several Temperatures. Energy Fuels 2014, 28, 6255–6266. [Google Scholar] [CrossRef]
- Joonaki, E.; Ghanaatian, S. The application of nanofluids for enhanced oil recovery: Effects on interfacial tension and coreflooding process. Pet. Sci. Technol. 2014, 32, 2599–2607. [Google Scholar] [CrossRef]
- Ma, H.; Luo, M.; Dai, L.L. Influences of surfactant and nanoparticle assembly on effective interfacial tensions. Phys. Chem. Chem. Phys. 2008, 10, 2207–2213. [Google Scholar] [CrossRef] [PubMed]
- Moradi, B.; Pourafshary, P.; Jalali, F.; Mohammadi, M.; Emadi, M. Experimental study of water-based nanofluid alternating gas injection as a novel enhanced oil-recovery method in oil-wet carbonate reservoirs. J. Nat. Gas Sci. Eng. 2015, 27, 64–73. [Google Scholar] [CrossRef]
- Roustaei, A.; Bagherzadeh, H. Experimental investigation of SiO2 nanoparticles on enhanced oil recovery of carbonate reservoirs. J. Pet. Explor. Prod. Technol. 2015, 5, 27–33. [Google Scholar] [CrossRef] [Green Version]
- Roustaei, A.; Saffarzadeh, S.; Mohammadi, M. An evaluation of modified silica nanoparticles’ efficiency in enhancing oil recovery of light and intermediate oil reservoirs. Egypt. J. Pet. 2013, 22, 427–433. [Google Scholar] [CrossRef] [Green Version]
- Esfandiarian, A.; Azdarpour, A.; Santos, R.M.; Mohammadian, E.; Hamidi, H.; Sedaghat, M.; Dehkordi, P.B. Mechanistic investigation of LSW/surfactant/alkali synergism for enhanced oil recovery: Fluid–fluid interactions. ACS Omega 2020, 5, 30059–30072. [Google Scholar] [CrossRef]
- Auflem, I.H.; Westvik, A.; Sjöblom, J. Destabilization of water-in-crude oil emulsions based on recombined oil samples at various pressures. J. Dispers. Sci. Technol. 2003, 24, 103–112. [Google Scholar] [CrossRef]
- Acevedo, S.; Borges, B.; Quintero, F.; Piscitelly, V.; Gutierrez, L.B. Asphaltenes and other natural surfactants from cerro negro crude oil. Stepwise adsorption at the water/toluene interface: Film formation and hydrophobic effects. Energy Fuels 2005, 19, 1948–1953. [Google Scholar] [CrossRef]
- Czarnecki, J.; Moran, K. On the stabilization mechanism of water-in-oil emulsions in petroleum systems. Energy Fuels 2005, 19, 2074–2079. [Google Scholar] [CrossRef]
- Verruto, V.J.; Kilpatrick, P.K. Water-in-model oil emulsions studied by small-angle neutron scattering: Interfacial film thickness and composition. Langmuir 2008, 24, 12807–12822. [Google Scholar] [CrossRef]
- Elyaderani, S.M.G.; Jafari, A. Investigation of interactions between silica nanoparticle, alkaline, and polymer in micromodel flooding for enhanced oil recovery. Energy Sour. Part A Recover. Util. Environ. Eff. 2020, 1–18. [Google Scholar] [CrossRef]
- Rueda, E.; Akarri, S.; Torsæter, O.; Moreno, R.B. Experimental investigation of the effect of adding nanoparticles to polymer flooding in water-wet micromodels. Nanomaterials 2020, 10, 1489. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, Z.; Dong, H.; Mikhailova, M.V.; Davarpanah, A. Hybrid application of nanoparticles and polymer in enhanced oil recovery processes. Polymers 2021, 13, 1414. [Google Scholar] [CrossRef] [PubMed]
- Gbadamosi, A.O.; Junin, R.; Manan, M.A.; Agi, A.; Yusuff, A.S. An overview of chemical enhanced oil recovery: Recent advances and prospects. Int. Nano Lett. 2019, 9, 171–202. [Google Scholar] [CrossRef] [Green Version]
- Ali, J.A.; Kolo, K.; Manshad, A.K.; Mohammadi, A.H. Recent advances in application of nanotechnology in chemical enhanced oil recovery: Effects of nanoparticles on wettability alteration, interfacial tension reduction, and flooding. Egypt. J. Pet. 2018, 27, 1371–1383. [Google Scholar] [CrossRef]
- Cheraghian, G.; Rostami, S.; Afrand, M. Nanotechnology in enhanced oil recovery. Process. 2020, 8, 1073. [Google Scholar] [CrossRef]
- Perez, H.I.Q.; Cañas, M.C.R.; Garcia, R.H.C.; Bohorquez, A.R.R. Use of nanoparticles to improve thermochemical resistance of synthetic polymer to enhanced oil recovery applications: A review. CT&F Cienc. Tecnol. Futuro 2020, 10, 85–97. [Google Scholar] [CrossRef]
- Bila, A.; Torsæter, O. Experimental investigation of polymer-coated silica nanoparticles for EOR under harsh reservoir conditions of high temperature and salinity. Nanomaterials 2021, 11, 765. [Google Scholar] [CrossRef]
- Mohnot, S.; Bae, J.; Foley, W. A Study of mineral/alkali reactions. SPE Reserv. Eng. 1987, 2, 653–663. [Google Scholar] [CrossRef]
- Jiecheng, C.; Wanfu, Z.; Qingfuo, W.; Gang, C.; Wenguang, B.; Changming, Z.; Meie, L. Technical breakthrough in production engineering ensures economic development of ASP flooding in daqing oilfield. In Proceedings of the SPE Asia Pacific Oil & Gas Conference and Exhibition, Adelaide, Australia, 14–16 October 2014. [Google Scholar] [CrossRef]
- AlGeer, M.A.; Gmira, A.; Al-Enezi, S.M.; Yousef, A.A. A new insight on the impact of individual ions on fluid/fluid interactions and smart water recovery. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 21–23 March 2016. [Google Scholar]
- Tahir, M.; Hincapie, R.E.; Ganzer, L. Unlocking the effects of fluid optimization on remaining oil saturation for the combined sulfate-modified water and polymer flooding. Energies 2020, 13, 3049. [Google Scholar] [CrossRef]
- Scheurer, C.; Hincapie, R.E.; Neubauer, E.; Metz, A.; Ness, D. Sorption of nanomaterials to sandstone rock. Nanomaterials 2022, 12, 200. [Google Scholar] [CrossRef] [PubMed]
| Synergy | Reported Recovery Mechanisms | Interaction | Media | Main Focus | Ref. |
|---|---|---|---|---|---|
| NPs, P, A | Increase in viscosity + reduction in IFT | R–F and F–F | Micromodel | Contact angle (CA, polymer adsorption | [66] |
| NPs, P | Increase in polymer viscosity, low polymer adsorption, homogenous NPs dispersion | R–F and F–F | Water-wet micromodels | IFT, CA and nano size distribution | [67] |
| NPs, P | Higher polymer viscoelasticity, low polymer retention and capillary forces reduction | R–F and F–F | Sandpacks | IFT, contact angle, RRF, viscosity, and relative permeability curves | [68] |
| A, P, S, NPs, F | Review summary of IFT, polymer adsorption, viscoelasticity, mobility control, wettability alteration, emulsion stabilization, | R–F and F–F | Laboratory to field applications | Review paper covering technical challenges and possible remediations, field projects. | [69] |
| A, P | IFT reduction and wettability alteration | R–F & F–F | Sandstone cores (oil-wet, water-wet) | Spontaneous imbibition, IFT, contact angle, oil TAN value | [1] |
| NPs-P, NPs-S, NPs-S-P | Review on IFT reduction & wettability alteration | R–F and F–F | Laboratory to field applications | Review paper covering nanotechnology applications in chemical EOR | [70] |
| NPs, P, S | IFT reduction, improved rheological properties and wettability alteration | R–F and F–F | Laboratory to field applications | Review on nanotechnology in EOR, challenges, and future research | [71] |
| NPs, P | Improving viscosity and thermal stability of HPAM polymer | NA | Laboratory to field applications | Review on nanotechnology for improving viscosity and stability | [72] |
| NPs, A | IFT reduction and wettability alteration | R–F and F–F | Sandstone core plugs | Spontaneous imbibition, IFT tests and phase behavior | [49] |
| NPs-P | IFT reduction, in situ emulsion generation, microscopic flow and wettability alteration | R–F and F–F | Neutral-wet core plugs | Flooding experiments, IFT, imbibition tests focusing on pressure and recovery | [73] |
| A, NPs, P | NPs as emulsion stabilizers in AP. IFT and phase behavior. Wettability alteration. | R–F and F–F | Sandstone outcrops | Flooding experiments, IFT, imbibition tests focusing on pressure and recovery | [46,47] |
| Property | High TAN | Low TAN |
|---|---|---|
| Reservoir/Well | 16 TH/Bockfliess 112 | St. Ulrich/St.U. 65 |
| TVD top [m] | 1622 | 1060 |
| TAN [mg KOH/g] | 1.61 | 0.39 |
| Saturates [%] | 39 | 55 |
| Aromatics [%] | 20 | 25.6 |
| Resins [%] | 39 | 18.6 |
| Asphaltene [%] | 2 | 0.8 |
| Saponifiable Acids [µmol/g] | 26 | n.m. |
| µ @ 60 °C [mPa.s] | 11.9 | 6 |
| ρ @ 20 °C/60 °C [g/cm3] | 0.917/0.884 | 0.866/0.842 |
| Parameter | Units | Berea 1 | Keuper 2 | Nordhorn (Bentheimer) 3 | |||
|---|---|---|---|---|---|---|---|
| Mean | SD * | Mean | SD * | Mean | SD * | ||
| Length | cm | 6.97 | 0.02 | 8.12 | 0.09 | 8.01 | 0.11 |
| Diameter | 2.96 | 0.01 | 2.98 | 0.01 | 2.96 | 0.01 | |
| Bulk Volume | cm3 | 47.76 | 0.26 | 55.76 | 0.73 | 54.42 | 0.87 |
| Pore Volume | 10.77 | 0.19 | 12.75 | 0.22 | 13.07 | 0.26 | |
| Grain Volume | kg/cm3 | 37.00 | 0.31 | 42.98 | 0.67 | 41.36 | 0.70 |
| Porosity | % | 22.60 | 0.40 | 23.30 | 0.80 | 23.96 | 0.35 |
| N2 permeability (kg) | mD | 447.60 | 37.40 | 1425.20 | 349.60 | 2313.02 | 162.10 |
| Water (Test Water) permeability (kw) | 223.90 | 17.90 | 890.00 | 193.90 | 1501.00 | 190.12 | |
| Irreducible water saturation | % | 24.00 | 8.00 | 21.40 | 7.90 | 25.60 | 4.00 |
| Fluid | Viscosity [mPa.s] 60 °C, 7.984 s−1 | High-TAN Oil, [mN/m] | Low-TAN Oil, [mN/m] | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Initial IFT | Equilibrium IFT | Initial IFT | Equilibrium IFT | ||||||
| Mean | SD * | Mean | SD * | Mean | SD * | Mean | SD * | ||
| Baseline—Brine (TW) | 0.571 | 7.84 | 0.43 | 8.40 | 0.50 | 4.31 | 0.62 | 3.40 | 0.56 |
| NPs only in TW 1 | 5.325 | 3.67 | 0.20 | 3.75 | 0.20 | 2.02 | 0.18 | 1.29 | 0.01 |
| Alkali (3000 ppm Na2CO3) in TW | 0.559 | 0.41 | 0.62 | 0.11 | 0.07 | 0.70 | 0.28 | 0.55 | 0.15 |
| Alkali (7000 ppm Na2CO3) in TW | 0.601 | 0.87 | 0.19 | 0.07 | 0.01 | 0.34 | 0.11 | 0.48 | 0.18 |
| NPs with 3000 ppm Na2CO3 in TW | 7.254 | 0.27 | 0.02 | 0.095 | 0.01 | 0.775 | 0.04 | 0.585 | 0.01 |
| Polymer (SNF 3630 S) in TW | 19.536 | 3.31 | 0.25 | 3.61 | 0.36 | 4.03 | 0.37 | 4.50 | 0.22 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | 18.054 | 2.41 | 0.55 | 0.04 | 0.01 | 0.48 | 0.09 | 0.78 | 0.18 |
| Fluid | Berea | Keuper | Nordhorn (Bentheimer) | |||
|---|---|---|---|---|---|---|
| High TAN | Low TAN | High TAN | Low TAN | High TAN | Low TAN | |
| Baseline—Brine (TW) | 30.00 | n.m. | 149.00 | 60.70 | 58.70 | 60.70 |
| NPs only in TW1 | 33.50 | n.m. | 31.50 | n.m. | n.m. | n.m. |
| Alkali (3000 ppm Na2CO3) in TW | 35.01 | n.m. | 55.76 | n.m. | n.m. | n.m. |
| Alkali (7000 ppm Na2CO3) in TW | n.m. | n.m. | 54.20 | 47.80 | 57.20 | 42.70 |
| NPs with 3000 ppm Na2CO3 in TW | 33.20 | n.m. | 46.02 | n.m. | n.m. | n.m. |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | n.m. | n.m. | 56.10 | 55.90 | 57.80 | 59.80 |
| Imbibing Fluid | Berea, [%] | Keuper, [%] | Nordhorn (Bentheimer), [%] | |||
|---|---|---|---|---|---|---|
| R.O | I.O. | R.O | I.O. | R.O | I.O. | |
| Baseline—Brine (TW) | 43.81 | - | 24.20 | - | 64.64 | - |
| NPs only in TW 1 | 57.58 | 13.77 | 38.31 | 14.11 | n.m. | n.m. |
| Alkali (3000 ppm Na2CO3) in TW | 57.71 | 13.90 | 93.30 | 69.10 | n.m. | n.m. |
| Alkali (7000 ppm Na2CO3) in TW | n.m. | n.m. | n.m. | n.m. | 73.80 | 9.16 |
| NPs with 3000 ppm Na2CO3 in TW | 69.50 | 25.69 | 94.16 | 69.96 | n.m. | n.m. |
| Polymer (SNF 3630 S) in TW | n.m. | n.m. | n.m. | n.m. | 69.10 | 4.46 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | n.m. | n.m. | n.m. | n.m. | 83.10 | 18.46 |
| Imbibing Fluid | Berea, [%] | Keuper, [%] | Nordhorn (Bentheimer), [%] | |||
|---|---|---|---|---|---|---|
| R.O | I.O. | R.O | I.O. | R.O | I.O. | |
| Baseline—Brine (TW) | 55.58 | - | 9.57 | - | 63.21 | - |
| NPs only in TW 1 | 67.19 | 11.61 | 31.98 | 22.41 | n.m. | n.m. |
| Alkali (3000 ppm Na2CO3) in TW | 85.23 | 29.65 | 32.04 | 22.47 | n.m. | n.m. |
| Alkali (7000 ppm Na2CO3) in TW | n.m. | n.m. | n.m. | n.m. | 74.48 | 11.27 |
| NPs with 3000 ppm Na2CO3 in TW | 71.67 | 16.09 | 55.86 | 46.29 | n.m. | n.m. |
| Polymer (SNF 3630 S) in TW | n.m. | n.m. | n.m. | n.m. | 66.19 | 2.98 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | n.m. | n.m. | n.m. | n.m. | 94.44 | 31.23 |
| Injected Fluid | Equilibrium IFT | Berea | Nordhorn (Bentheimer) | ||
|---|---|---|---|---|---|
| High TAN, [mN/m] | Incremental Recovery% | Injected PV | Incremental Recovery% | Injected PV | |
| NPs only in TW 1 | 3.75 | 3.90 | 2.00 | n.p. | n.p. |
| Alkali (3000 ppm Na2CO3) in TW | 0.11 | 14.00 | 2.00 | 12.00 | 1.5 |
| Alkali (7000 ppm Na2CO3) in TW | 0.07 | n.p. | n.p. | 19.00 | 1.5 |
| NPs with 3000 ppm Na2CO3 in TW | 0.095 | 18.00 | 2.00 | n.p. | n.p. |
| Polymer (SNF 3630 S) in TW | 3.61 | 9.00 | 2.00 | 3.00 | 3.0 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | 0.04 | n.p. | n.p. | 29.00 | 2.00 |
| Injected Fluid | Equilibrium IFT | Berea, Incremental Oil [%] | Nordhorn (Bentheimer), Berea, Incremental Oil [%] | ||
|---|---|---|---|---|---|
| High TAN, [mN/m] | Imbibition | Flooding | Imbibition | Flooding | |
| NPs only in TW 1 | 3.75 | 13.77 | 3.90 | n.m. | n.p. |
| Alkali (3000 ppm Na2CO3) in TW | 0.11 | 13.90 | 14.00 | n.m. | 12.00 |
| Alkali (7000 ppm Na2CO3) in TW | 0.07 | n.m. | n.p. | 11.27 | 19.00 |
| NPs with 3000 ppm Na2CO3 in TW | 0.095 | 25.69 | 18.00 | n.m. | n.p. |
| Polymer (SNF 3630 S) in TW | 3.61 | n.m. | 9.00 | 2.98 | 3.00 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | 0.04 | n.m. | n.p. | 31.23 | 29.00 |
| Imbibing Fluid | Outcrop | Porosity Φ, [%] | Permeability, [mD] | ||||
|---|---|---|---|---|---|---|---|
| Before | After | Diff. (%) | Before | After | Diff. (%) | ||
| NPs only in TW 1 | Berea | 22.60 | 21.34 | −1.83 | 477.62 | 419.82 | −12.14 |
| Keuper | 23.49 | 23.17 | 3.57 | 1381.62 | 1311.50 | −5.17 | |
| Alkali (3000 ppm Na2CO3) in TW | Berea | 22.58 | 22.12 | −2.04 | 399.55 | 364.44 | −8.82 |
| Keuper | 24.09 | 23.99 | −1.10 | 1238.83 | 1225.16 | −1.10 | |
| Nord. (Bent) | 23.30 | 23.05 | −1.07 | 2343.05 | 2340.02 | −0.15 | |
| Alkali (7000 ppm Na2CO3) in TW | Nord. (Bent) | 24.30 | 24.10 | −0.90 | 2448.62 | 2389.78 | −2.40 |
| NPs with 3000 ppm Na2CO3 in TW | Berea | 22.42 | 22.23 | −0.77 | 442.05 | 391.859 | −11.32 |
| Keuper | 24.28 | 23.69 | −2.63 | 1446.37 | 1320.94 | −8.03 | |
| Polymer (SNF 3630 S) in TW | Nord. (Bent) | 24.19 | 24.08 | 0.61 | 2346.49 | 2273.49 | −3.11 |
| 7000 ppm Na2CO3 with SNF 3630 S in TW | Nord. (Bent) | 23.98 | 24.45 | 1.95 | 2310.33 | 2113.95 | −8.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hincapie, R.E.; Borovina, A.; Neubauer, E.; Tahir, M.; Saleh, S.; Arekhov, V.; Biernat, M.; Clemens, T. Recovery Observations from Alkali, Nanoparticles and Polymer Flooding as Combined Processes. Polymers 2022, 14, 603. https://doi.org/10.3390/polym14030603
Hincapie RE, Borovina A, Neubauer E, Tahir M, Saleh S, Arekhov V, Biernat M, Clemens T. Recovery Observations from Alkali, Nanoparticles and Polymer Flooding as Combined Processes. Polymers. 2022; 14(3):603. https://doi.org/10.3390/polym14030603
Chicago/Turabian StyleHincapie, Rafael E., Ante Borovina, Elisabeth Neubauer, Muhammad Tahir, Samhar Saleh, Vladislav Arekhov, Magdalena Biernat, and Torsten Clemens. 2022. "Recovery Observations from Alkali, Nanoparticles and Polymer Flooding as Combined Processes" Polymers 14, no. 3: 603. https://doi.org/10.3390/polym14030603
APA StyleHincapie, R. E., Borovina, A., Neubauer, E., Tahir, M., Saleh, S., Arekhov, V., Biernat, M., & Clemens, T. (2022). Recovery Observations from Alkali, Nanoparticles and Polymer Flooding as Combined Processes. Polymers, 14(3), 603. https://doi.org/10.3390/polym14030603









