Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review
Abstract
:1. Introduction
2. Application of Nanoparticles
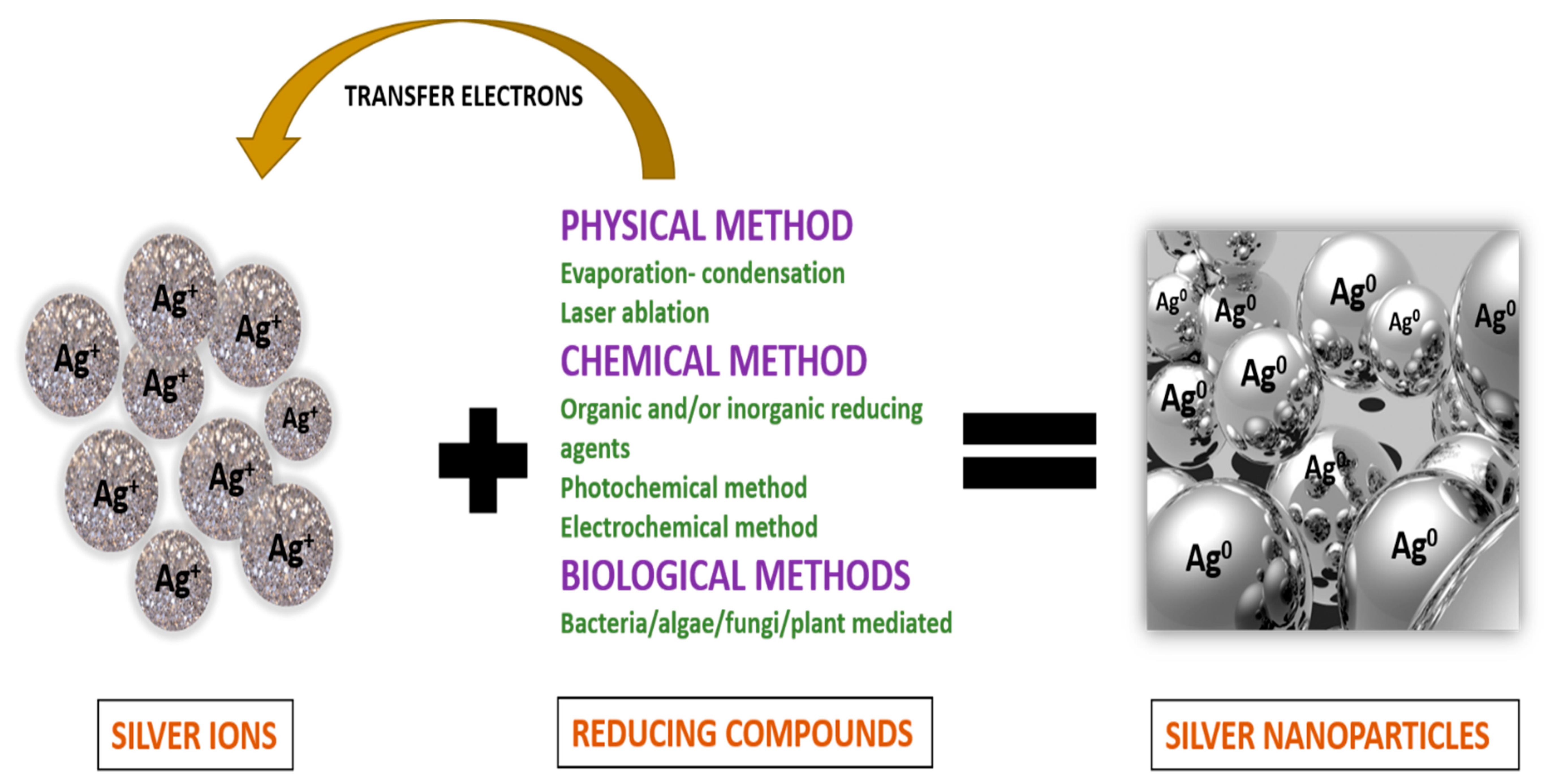
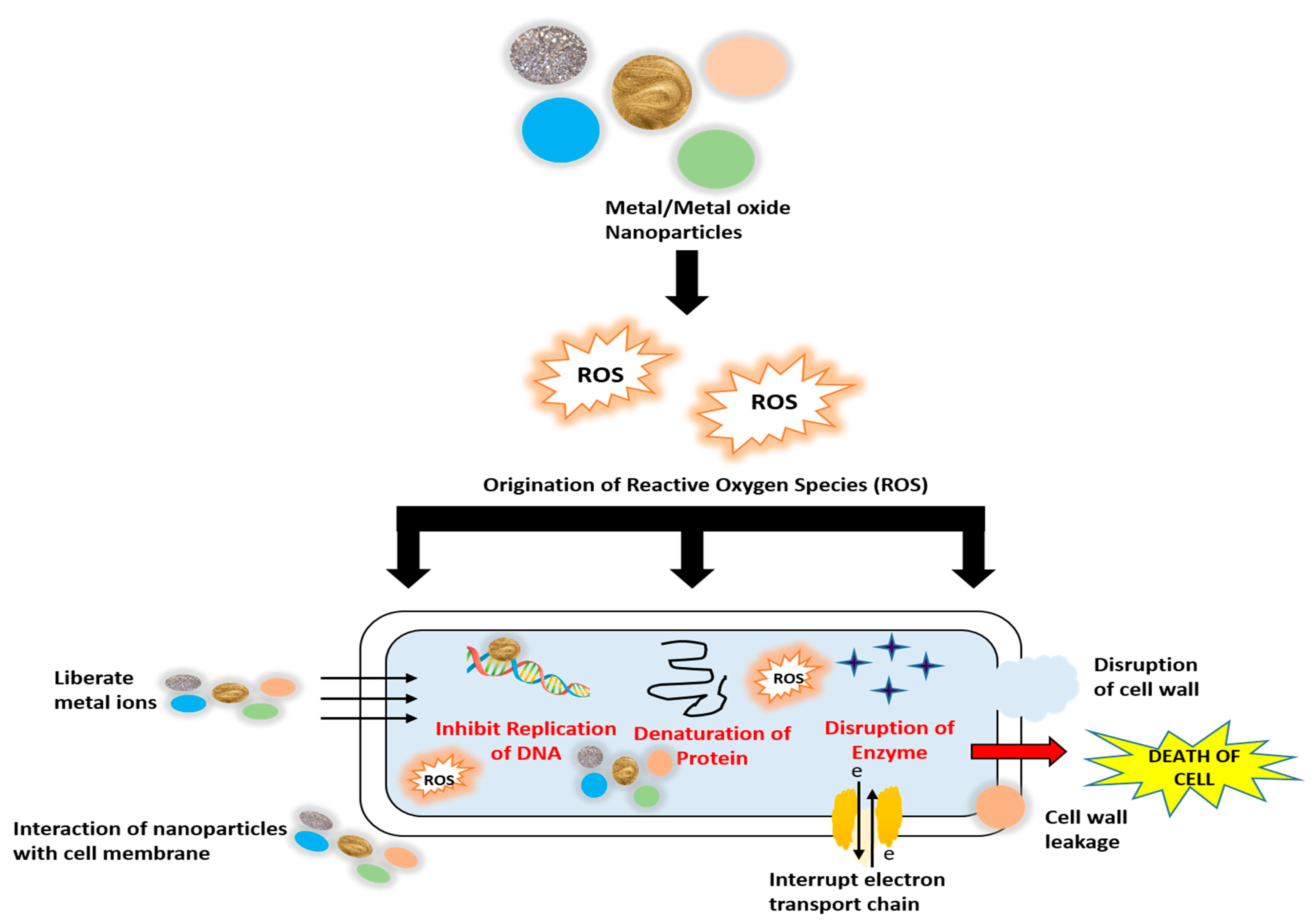
2.1. Silver Nanoparticles
2.2. Gold Nanoparticles
3. Metal Oxide Nanoparticles for Food Packaging Application
3.1. Zinc Oxide (ZnO) Nanoparticles for Food Packaging
3.2. Titanium Dioxide (TiO2) Nanoparticles for Food Packaging
3.3. Copper Oxide (CuO) Nanoparticles for Food Packaging
3.4. Silicon Dioxide (SiO2) Nanoparticles for Food Packaging
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kirwan, M.J.; Plant, S.; Strawbridge, J.W. Plastics in Food Packaging; Coles, R., Mc Dowel, D., Kirwan, M.J.., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2011; ISBN 9781405189101. [Google Scholar]
- Tharanathan, R.N. Biodegradable films and composite coatings: Past, present and future. Trends Food Sci. Technol. 2003, 14, 71–78. [Google Scholar] [CrossRef]
- Kimbuathong, N.; Leelaphiwat, P.; Harnkarnsujarit, N. Inhibition of melanosis and microbial growth in Pacific white shrimp (Litopenaeus vannamei) using high CO2 modified atmosphere packaging. Food Chem. 2020, 312, 126114. [Google Scholar] [CrossRef] [PubMed]
- Laorenza, Y.; Harnkarnsujarit, N. Carvacrol, citral and α-terpineol essential oil incorporated biodegradable films for functional active packaging of Pacific white shrimp. Food Chem. 2021, 363, 130252. [Google Scholar] [CrossRef] [PubMed]
- Katekhong, W.; Wongphan, P.; Klinmalai, P.; Harnkarnsujarit, N. Thermoplastic starch blown films functionalized by plasticized nitrite blended with PBAT for superior oxygen barrier and active biodegradable meat packaging. Food Chem. 2022, 374, 131709. [Google Scholar] [CrossRef]
- Kuswandi, B. Nanotechnology in Food Packaging. In Nanoscience in Food and Agriculture 1; Ranjan, S., Dasgupta, N., Lichtfouse, E., Eds.; Sustainable Agriculture Reviews; Springer: Cham, Switzerland, 2016; Volume 20. [Google Scholar] [CrossRef]
- Neethirajan, S.; Jayas, D.S. Nanotechnology for the Food and Bioprocessing Industries. Food Bioprocess Technol. 2011, 4, 39–47. [Google Scholar] [CrossRef]
- Wesley, S.J.; Raja, P.; Raj, A.A.; Tiroutchelvamae, D. Review on—Nanotechnology Applications in Food Packaging and Safety. Int. J. Eng. Res. 2014, 3, 645–651. [Google Scholar] [CrossRef]
- Singh Sekhon, B. Nanotechnology in agri-food production: An overview. Nanotechnol. Sci. Appl. 2014, 7, 31–53. [Google Scholar] [CrossRef] [Green Version]
- Lagarón, J.M.; Cabedo, L.; Cava, D.; Feijoo, J.L.; Gavara, R.; Gimenez, E. Improving packaged food quality and safety. Part 2: Nanocomposites. Food Addit. Contam. 2005, 22, 994–998. [Google Scholar] [CrossRef]
- Ray, S.S.; Bousmina, M. Biodegradable polymers and their layered silicate nanocomposites: In greening the 21st century materials world. Prog. Mater. Sci. 2005, 50, 962–1079. [Google Scholar] [CrossRef]
- Silvestre, C.; Duraccio, D.; Cimmino, S. Food packaging based on polymer nanomaterials. Prog. Polym. Sci. 2011, 36, 1766–1782. [Google Scholar] [CrossRef]
- Gordienko, M.G.; Palchikova, V.V.; Kalenov, S.V.; Belov, A.A.; Lyasnikova, V.N.; Poberezhniy, D.Y.; Chibisova, A.V.; Sorokin, V.V.; Skladnev, D.A. Antimicrobial activity of silver salt and silver nanoparticles in different forms against microorganisms of different taxonomic groups. J. Hazard. Mater. 2019, 378, 120754. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Abad, A.; Lagaron, J.M.; Ocio, M.J. Development and characterization of silver-based antimicrobial ethylene-vinyl alcohol copolymer (EVOH) films for food-packaging applications. J. Agric. Food Chem. 2012, 60, 5350–5359. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of silver nanoparticles: Chemical, physical and biological methods. Res. Pharm. Sci. 2014, 9, 385–406. [Google Scholar]
- Kanmani, P.; Lim, S.T. Synthesis and characterization of pullulan-mediated silver nanoparticles and its antimicrobial activities. Carbohydr. Polym. 2013, 97, 421–428. [Google Scholar] [CrossRef]
- Kraśniewska, K.; Pobiega, K.; Gniewosz, M. Pullulan-Biopolymer with Potential for Use as Food Packaging. Int. J. Food Eng. 2019, 15, 15. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, S.; Zhong, L.; Wu, G. Preparation of high-stable silver nanoparticle dispersion by using sodium alginate as a stabilizer under gamma radiation. Radiat. Phys. Chem. 2009, 78, 251–255. [Google Scholar] [CrossRef]
- Palza, H. Antimicrobial polymers with metal nanoparticles. Int. J. Mol. Sci. 2015, 16, 2099–2116. [Google Scholar] [CrossRef] [Green Version]
- Cakić, M.; Glišić, S.; Nikolić, G.; Nikolić, G.M.; Cakić, K.; Cvetinov, M. Synthesis, characterization and antimicrobial activity of dextran sulphate stabilized silver nanoparticles. J. Mol. Struct. 2016, 1110, 156–161. [Google Scholar] [CrossRef]
- Hettiarachchi, M.A.; Wickramarachchi, P.A.S.R. Synthesis of chitosan stabilized silver nanoparticles using gamma ray irradiation and characterization. J. Sci. Univ. Kelaniya Sri Lanka 2012, 6, 65. [Google Scholar] [CrossRef] [Green Version]
- Ishihara, M.; Nguyen, V.Q.; Mori, Y.; Nakamura, S.; Hattori, H. Adsorption of silver nanoparticles onto different surface structures of chitin/chitosan and correlations with antimicrobial activities. Int. J. Mol. Sci. 2015, 16, 13973–13988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, B.; Luo, Y.; Teng, Z.; Zhang, B.; Zhou, B.; Wang, Q. Development of silver/titanium dioxide/chitosan adipate nanocomposite as an antibacterial coating for fruit storage. LWT 2015, 63, 1206–1213. [Google Scholar] [CrossRef]
- De Moura, M.R.; Mattoso, L.H.C.; Zucolotto, V. Development of cellulose-based bactericidal nanocomposites containing silver nanoparticles and their use as active food packaging. J. Food Eng. 2012, 109, 520–524. [Google Scholar] [CrossRef]
- Bahrami, A.; Rezaei Mokarram, R.; Sowti Khiabani, M.; Ghanbarzadeh, B.; Salehi, R. Physico-mechanical and antimicrobial properties of tragacanth/hydroxypropyl methylcellulose/beeswax edible films reinforced with silver nanoparticles. Int. J. Biol. Macromol. 2019, 129, 1103–1112. [Google Scholar] [CrossRef]
- Pinto, R.J.B.; Almeida, A.; Fernandes, S.C.M.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P.; Trindade, T. Antifungal activity of transparent nanocomposite thin films of pullulan and silver against Aspergillus niger. Colloids Surf. B Biointerfaces 2013, 103, 143–148. [Google Scholar] [CrossRef]
- Khalaf, H.; Sharoba, A.; El-Tanahi, H.; Morsy, M. Stability of Antimicrobial Activity of Pullulan Edible Films Incorporated with Nanoparticles and Essential Oils and Their Impact on Turkey Deli Meat Quality. J. Food Dairy Sci. 2013, 4, 557–573. [Google Scholar] [CrossRef]
- Sharma, C.; Dhiman, R.; Rokana, N.; Panwar, H. Nanotechnology: An untapped resource for food packaging. Front. Microbiol. 2017, 8, 1735. [Google Scholar] [CrossRef] [Green Version]
- Roy, S.; Shankar, S.; Rhim, J.W. Melanin-mediated synthesis of silver nanoparticle and its use for the preparation of carrageenan-based antibacterial films. Food Hydrocoll. 2019, 88, 237–246. [Google Scholar] [CrossRef]
- Shankar, S.; Teng, X.; Li, G.; Rhim, J.W. Preparation, characterization, and antimicrobial activity of gelatin/ZnO nanocomposite films. Food Hydrocoll. 2015, 45, 264–271. [Google Scholar] [CrossRef]
- Jiang, T.; Feng, L.; Wang, Y. Effect of alginate/nano-Ag coating on microbial and physicochemical characteristics of shiitake mushroom (Lentinus edodes) during cold storage. Food Chem. 2013, 141, 954–960. [Google Scholar] [CrossRef]
- Moussa, S.H.; Tayel, A.A.; Alsohim, A.S.; Abdallah, R.R. Botryticidal activity of nanosized silver-chitosan composite and its application for the control of gray mold in strawberry. J. Food Sci. 2013, 78, 1589–1594. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Conte, A.; Buonocore, G.G.; Lavorgna, M.; Del Nobile, M.A. Calcium-alginate coating loaded with silver-montmorillonite nanoparticles to prolong the shelf-life of fresh-cut carrots. Food Res. Int. 2012, 48, 164–169. [Google Scholar] [CrossRef]
- Hedayati, S.; Niakousari, M. Effect of Coatings of Silver Nanoparticles and Gum Arabic on Physicochemical and Microbial Properties of Green Bell Pepper (Capsicum annuum). J. Food Process. Preserv. 2015, 39, 2001–2007. [Google Scholar] [CrossRef]
- Shah, M.; Fawcett, D.; Sharma, S.; Tripathy, S.K.; Poinern, G.E.J. Green synthesis of metallic nanoparticles via biological entities. Materials 2015, 8, 7278–7308. [Google Scholar] [CrossRef] [Green Version]
- Gudadhe, J.A.; Yadav, A.; Gade, A.; Marcato, P.D.; Durán, N.; Rai, M. Preparation of an agar-silver nanoparticles (A-AgNp) film for increasing the shelf-life of fruits. IET Nanobiotechnology 2014, 8, 190–195. [Google Scholar] [CrossRef]
- Zimoch-Korzycka, A.; Jarmoluk, A. The use of chitosan, lysozyme, and the nanosilver as antimicrobial ingredients of edible protective hydrosols applied into the surface of meat. J. Food Sci. Technol. 2015, 52, 5996–6002. [Google Scholar] [CrossRef] [Green Version]
- Morsy, M.K.; Khalaf, H.H.; Sharoba, A.M.; El-Tanahi, H.H.; Cutter, C.N. Incorporation of Essential Oils and Nanoparticles in Pullulan Films to Control Foodborne Pathogens on Meat and Poultry Products. J. Food Sci. 2014, 79, 675–684. [Google Scholar] [CrossRef]
- Marchiore, N.G.; Manso, I.J.; Kaufmann, K.C.; Lemes, G.F.; Pizolli, A.P.d.O.; Droval, A.A.; Bracht, L.; Gonçalves, O.H.; Leimann, F.V. Migration evaluation of silver nanoparticles from antimicrobial edible coating to sausages. LWT—Food Sci. Technol. 2017, 76, 203–208. [Google Scholar] [CrossRef]
- Incoronato, A.L.; Conte, A.; Buonocore, G.G.; Del Nobile, M.A. Agar hydrogel with silver nanoparticles to prolong the shelf life of Fior di Latte cheese. J. Dairy Sci. 2011, 94, 1697–1704. [Google Scholar] [CrossRef]
- Silvan, J.M.; Zorraquin-Peña, I.; de Llano, D.G.; Moreno-Arribas, V.V.; Martinez-Rodriguez, A.J. Antibacterial activity of glutathione-stabilized silver nanoparticles against Campylobacter multidrug-resistant strains. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Kumar, S.; Shukla, A.; Baul, P.P.; Mitra, A.; Halder, D. Biodegradable hybrid nanocomposites of chitosan/gelatin and silver nanoparticles for active food packaging applications. Food Packag. Shelf Life 2018, 16, 178–184. [Google Scholar] [CrossRef]
- He, Y.; Li, H.; Fei, X.; Peng, L. Carboxymethyl cellulose/cellulose nanocrystals immobilized silver nanoparticles as an effective coating to improve barrier and antibacterial properties of paper for food packaging applications. Carbohydr. Polym. 2021, 252, 117156. [Google Scholar] [CrossRef] [PubMed]
- Kowsalya, E.; MosaChristas, K.; Balashanmugam, P.; Tamil Selvi, A.; Jaquline Chinna, R.I. Biocompatible silver nanoparticles/poly(vinyl alcohol) electrospun nanofibers for potential antimicrobial food packaging applications. Food Packag. Shelf Life 2019, 21, 100379. [Google Scholar] [CrossRef]
- Mathew, S.; Snigdha, S.; Mathew, J.; Radhakrishnan, E.K. Biodegradable and active nanocomposite pouches reinforced with silver nanoparticles for improved packaging of chicken sausages. Food Packag. Shelf Life 2019, 19, 155–166. [Google Scholar] [CrossRef]
- Orsuwan, A.; Shankar, S.; Wang, L.F.; Sothornvit, R.; Rhim, J.W. One-step preparation of banana powder/silver nanoparticles composite films. J. Food Sci. Technol. 2017, 54, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Silva, L.S.C.; Martim, S.R.; Gomes, D.M.D.; Prado, F.B.; Marinho, N.M.V.; de Amorim Silva, T.; Castillo, T.A.; de Arimatéia Rodrigues do Rego, J.; Seabra, A.B.; Durán, N.; et al. Amazonian tuber starch based films incorporated with silver nanoparticles for preservation of fruits. Res. Soc. Dev. 2021, 10, e23510615304. [Google Scholar] [CrossRef]
- Gu, H.; Chen, X.; Chen, F.; Zhou, X.; Parsaee, Z. Ultrasound-assisted biosynthesis of CuO-NPs using brown alga Cystoseira trinodis: Characterization, photocatalytic AOP, DPPH scavenging and antibacterial investigations. Ultrason. Sonochem. 2018, 41, 109–119. [Google Scholar] [CrossRef]
- Paidari, S.; Ibrahim, S.A. Potential application of gold nanoparticles in food packaging: A mini review. Gold Bull. 2021, 54, 31–36. [Google Scholar] [CrossRef]
- Lima, E.; Guerra, R.; Lara, V.; Guzmán, A. Gold nanoparticles as efficient antimicrobial agents for Escherichia coli and Salmonella typhi. Chem. Cent. J. 2013, 7, 11. [Google Scholar] [CrossRef] [Green Version]
- Pagno, C.H.; Costa, T.M.H.; De Menezes, E.W.; Benvenutti, E.V.; Hertz, P.F.; Matte, C.R.; Tosati, J.V.; Monteiro, A.R.; Rios, A.O.; Flôres, S.H. Development of active biofilms of quinoa (Chenopodium quinoa W.) starch containing gold nanoparticles and evaluation of antimicrobial activity. Food Chem. 2015, 173, 755–762. [Google Scholar] [CrossRef]
- Thirumurugan, A.; Ramachandran, S.; Shiamala Gowri, A. Combined effect of bacteriocin with gold nanoparticles against food spoiling bacteria—An approach for food packaging material preparation. Int. Food Res. J. 2013, 20, 1909–1912. [Google Scholar]
- Zawrah, M.F.; Abd El-Moez, S.I. Antimicrobial activities of gold nanoparticles against major foodborne pathogens. Life Sci. J. 2011, 8, 37–44. [Google Scholar]
- Hoseinnejad, M.; Jafari, S.M.; Katouzian, I. Inorganic and metal nanoparticles and their antimicrobial activity in food packaging applications. Crit. Rev. Microbiol. 2018, 44, 161–181. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Prabhune, A.; Perry, C.C. Antibiotic mediated synthesis of gold nanoparticles with potent antimicrobial activity and their application in antimicrobial coatings. J. Mater. Chem. 2010, 20, 6789–6798. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, S.; Teoh, Y.L.; Ong, K.M.; Rafflisman Zaidi, N.S.; Mah, S.K. Poly(vinyl) alcohol crosslinked composite packaging film containing gold nanoparticles on shelf life extension of banana. Food Packag. Shelf Life 2020, 24, 100463. [Google Scholar] [CrossRef]
- Virgili, A.H.; Laranja, D.C.; Malheiros, P.S.; Pereira, M.B.; Costa, T.M.H.; de Menezes, E.W. Nanocomposite film with antimicrobial activity based on gold nanoparticles, chitosan and aminopropylsilane. Surf. Coatings Technol. 2021, 415, 127086. [Google Scholar] [CrossRef]
- Gomashe, A.V.; Dharmik, P.G. Synergistic Effect of Gold Nanoparticles and Bacteriocin Against Food Blemishing Microbes: A Novel Approach for Food Packaging Material Preparation. Glob. J. Res. Anal. 2012, 3, 1–3. [Google Scholar] [CrossRef]
- Chow, C.F. Biogenic amines- and sulfides-responsive gold nanoparticles for real-time visual detection of raw meat, fish, crustaceans, and preserved meat. Food Chem. 2020, 311, 125908. [Google Scholar] [CrossRef]
- Ashe, B. A Detail Investigation to Observe the Effect of Zinc Oxide and Silver Nanoparticles in Biological System. Ph.D. Thesis, National Institute Of Technology, Odisha, India, 2011; pp. 1–228. [Google Scholar]
- Rasmussen, J.W.; Martinez, E.; Louka, P.; Wingett, D.G. Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications. Expert Opin. Drug Deliv. 2010, 7, 1063–1077. [Google Scholar] [CrossRef] [Green Version]
- Wahid, F.; Zhong, C.; Wang, H.S.; Hu, X.H.; Chu, L.Q. Recent advances in antimicrobial hydrogels containing metal ions and metals/metal oxide nanoparticles. Polymers 2017, 9, 636. [Google Scholar] [CrossRef] [Green Version]
- Fan, Z.; Lu, J.G. Zinc oxide nanostructures: Synthesis and properties. J. Nanosci. Nanotechnol. 2005, 5, 1561–1573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zanet, V.; Vidic, J.; Auger, S.; Vizzini, P.; Lippe, G.; Iacumin, L.; Comi, G.; Manzano, M. Activity evaluation of pure and doped zinc oxide nanoparticles against bacterial pathogens and Saccharomyces cerevisiae. J. Appl. Microbiol. 2019, 127, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Seil, J.T.; Webster, T.J. Antimicrobial applications of nanotechnology: Methods and literature. Int. J. Nanomed. 2012, 7, 2767–2781. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.; Kelf, T.A.; Sanchez, W.H.; Roberts, M.S.; Rička, J.; Frenz, M.; Zvyagin, A.V. Characterization of optical properties of ZnO nanoparticles for quantitative imaging of transdermal transport. Biomed. Opt. Express 2011, 2, 3321. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jiang, Y.; Ding, Y.; Povey, M.; York, D. Investigation into the antibacterial behaviour of suspensions of ZnO nanoparticles (ZnO nanofluids). J. Nanoparticle Res. 2007, 9, 479–489. [Google Scholar] [CrossRef]
- Yu, J.; Yang, J.; Liu, B.; Ma, X. Preparation and characterization of glycerol plasticized-pea starch/ZnO-carboxymethylcellulose sodium nanocomposites. Bioresour. Technol. 2009, 100, 2832–2841. [Google Scholar] [CrossRef] [PubMed]
- Sawai, J.; Kawada, E.; Kanou, F.; Igarashi, H.; Hashimoto, A.; Kokugan, T.; Shimizu, M. Detection of active oxygen generated from ceramic powders having antibacterial activity. J. Chem. Eng. Japan 1996, 29, 627–633. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, O. Influence of particle size on the antibacterial activity of zinc oxide. Int. J. Inorg. Mater. 2001, 3, 643–646. [Google Scholar] [CrossRef]
- Yang, H.; Liu, C.; Yang, D.; Zhang, H.; Xi, Z. Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: The role of particle size, shape and composition. J. Appl. Toxicol. 2009, 29, 69–78. [Google Scholar] [CrossRef]
- Jeng, H.A.; Swanson, J. Toxicity of metal oxide nanoparticles in mammalian cells. J. Environ. Sci. Heal.—Part A Toxic/Hazardous Subst. Environ. Eng. 2006, 41, 2699–2711. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.T.; Manna, A.C. Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef] [Green Version]
- Stanković, A.; Dimitrijević, S.; Uskoković, D. Influence of size scale and morphology on antibacterial properties of ZnO powders hydrothemally synthesized using different surface stabilizing agents. Colloids Surf. B Biointerfaces 2013, 102, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; He, Y.; Irwin, P.L.; Jin, T.; Shi, X. Antibacterial activity and mechanism of action of zinc oxide nanoparticles against Campylobacter jejuni. Appl. Environ. Microbiol. 2011, 77, 2325–2331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghupathi, K.R.; Koodali, R.T.; Manna, A.C. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2011, 27, 4020–4028. [Google Scholar] [CrossRef] [PubMed]
- Heinlaan, M.; Ivask, A.; Blinova, I.; Dubourguier, H.C.; Kahru, A. Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere 2008, 71, 1308–1316. [Google Scholar] [CrossRef]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 184–192. [Google Scholar] [CrossRef]
- Sevinç, B.A.; Hanley, L. Antibacterial activity of dental composites containing zinc oxide nanoparticles. J. Biomed. Mater. Res.—Part B Appl. Biomater. 2010, 94, 22–31. [Google Scholar] [CrossRef] [Green Version]
- Colon, G.; Ward, B.C.; Webster, T.J. Increased osteoblast and decreased Staphylococcus epidermidis functions on nanophase ZnO and TiO2. J. Biomed. Mater. Res.—Part A 2006, 78, 595–604. [Google Scholar] [CrossRef]
- Karami, H.; Fakoori, E. Synthesis and characterization of ZnO nanorods based on a new gel pyrolysis method. J. Nanomater. 2011, 2011, 628203. [Google Scholar] [CrossRef]
- Amna, T.; Yang, J.; Ryu, K.S.; Hwang, I.H. Electrospun antimicrobial hybrid mats: Innovative packaging material for meat and meat-products. J. Food Sci. Technol. 2015, 52, 4600–4606. [Google Scholar] [CrossRef]
- Wang, Z.L. Zinc oxide nanostructures: Growth, properties and applications. J. Phys. Condens. Matter 2004, 16, 829–858. [Google Scholar] [CrossRef]
- Mahmud, S. One-dimensional growth of zinc oxide nanostructures from large micro-particles in a highly rapid synthesis. J. Alloys Compd. 2011, 509, 4035–4040. [Google Scholar] [CrossRef]
- Talebian, N.; Amininezhad, S.M.; Doudi, M. Controllable synthesis of ZnO nanoparticles and their morphology-dependent antibacterial and optical properties. J. Photochem. Photobiol. B Biol. 2013, 120, 66–73. [Google Scholar] [CrossRef]
- Wahab, R.; Ansari, S.G.; Kim, Y.S.; Seo, H.K.; Kim, G.S.; Khang, G.; Shin, H.S. Low temperature solution synthesis and characterization of ZnO nano-flowers. Mater. Res. Bull. 2007, 42, 1640–1648. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, L.; Yin, J.; Su, H.; Liao, C.; Yan, C. Control of ZnO morphology via a simple solution route. Chem. Mater. 2002, 14, 4172–4177. [Google Scholar] [CrossRef]
- Wahab, R.; Mishra, A.; Yun, S., Il; Kim, Y.S.; Shin, H.S. Antibacterial activity of ZnO nanoparticles prepared via non-hydrolytic solution route. Appl. Microbiol. Biotechnol. 2010, 87, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Wahab, R.; Khan, F.; Lutfullah; Singh, R.B.; Khan, A. Enhance antimicrobial activity of ZnO nanomaterial’s (QDs and NPs) and their analytical applications. Phys. E Low-Dimens. Syst. Nanostructures 2014, 62, 111–117. [Google Scholar] [CrossRef]
- Wu, G.; Cheng, Y.; Xie, Q.; Jia, Z.; Xiang, F.; Wu, H. Facile synthesis of urchin-like ZnO hollow spheres with enhanced electromagnetic wave absorption properties. Mater. Lett. 2015, 144, 157–160. [Google Scholar] [CrossRef]
- Kumar, S.; Boro, J.C.; Ray, D.; Mukherjee, A.; Dutta, J. Bionanocomposite films of agar incorporated with ZnO nanoparticles as an active packaging material for shelf life extension of green grape. Heliyon 2019, 5, e01867. [Google Scholar] [CrossRef] [Green Version]
- Duncan, T.V. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef]
- Xiao-e, L.; Green, A.N.M.; Haque, S.A.; Mills, A.; Durrant, J.R. Light-driven oxygen scavenging by titania/polymer nanocomposite films. J. Photochem. Photobiol. A Chem. 2004, 162, 253–259. [Google Scholar] [CrossRef]
- Zhu, Z.; Cai, H.; Sun, D.W. Titanium dioxide (TiO2) photocatalysis technology for nonthermal inactivation of microorganisms in foods. Trends Food Sci. Technol. 2018, 75, 23–35. [Google Scholar] [CrossRef]
- He, Q.; Zhang, Y.; Cai, X.; Wang, S. Fabrication of gelatin-TiO2 nanocomposite film and its structural, antibacterial and physical properties. Int. J. Biol. Macromol. 2016, 84, 153–160. [Google Scholar] [CrossRef]
- Flores-López, L.Z.; Espinoza-Gómez, H.; Somanathan, R. Silver nanoparticles: Electron transfer, reactive oxygen species, oxidative stress, beneficial and toxicological effects. Mini review. J. Appl. Toxicol. 2019, 39, 16–26. [Google Scholar] [CrossRef] [Green Version]
- Nešić, A.; Gordić, M.; Davidović, S.; Radovanović, Ž.; Nedeljković, J.; Smirnova, I.; Gurikov, P. Pectin-based nanocomposite aerogels for potential insulated food packaging application. Carbohydr. Polym. 2018, 195, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Noori Hashemabad, Z.; Shabanpour, B.; Azizi, H.; Ojagh, S.M.; Alishahi, A. Effect of Tio2 Nanoparticles on the Antibacterial and Physical Properties of Low-Density Polyethylene Film. Polym.—Plast. Technol. Eng. 2017, 56, 1516–1527. [Google Scholar] [CrossRef]
- Roilo, D.; Maestri, C.A.; Scarpa, M.; Bettotti, P.; Checchetto, R. Gas barrier and optical properties of cellulose nanofiber coatings with dispersed TiO2 nanoparticles. Surf. Coatings Technol. 2018, 343, 131–137. [Google Scholar] [CrossRef]
- Oleyaei, S.A.; Zahedi, Y.; Ghanbarzadeh, B.; Moayedi, A.A. Modification of physicochemical and thermal properties of starch films by incorporation of TiO2 nanoparticles. Int. J. Biol. Macromol. 2016, 89, 256–264. [Google Scholar] [CrossRef]
- Goudarzi, V.; Shahabi-Ghahfarrokhi, I.; Babaei-Ghazvini, A. Preparation of ecofriendly UV-protective food packaging material by starch/TiO2 bio-nanocomposite: Characterization. Int. J. Biol. Macromol. 2017, 95, 306–313. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Wongphan, P.; Promhuad, K.; Promsorn, J.; Harnkarnsujarit, N. Biodegradable Poly (Butylene Adipate-Co-Terephthalate) and Thermoplastic Starch-Blended TiO2 Nanocomposite Blown Films as Functional Active Packaging of Fresh Fruit. Polymers 2021, 13, 4192. [Google Scholar] [CrossRef]
- Grigore, M.E.; Biscu, E.R.; Holban, A.M.; Gestal, M.C.; Grumezescu, A.M. Methods of synthesis, properties and biomedical applications of CuO nanoparticles. Pharmaceuticals 2016, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, C.; Manikandan, G.; Gomathisankar, P. Microwave, sonochemical and combustion synthesized CuO nanostructures and their electrical and bactericidal properties. J. Alloys Compd. 2013, 580, 570–577. [Google Scholar] [CrossRef]
- Kamble, S.P.; Mote, V.D. Structural, optical and magnetic properties of Co doped CuO nano-particles by sol-gel auto combustion technique. Solid State Sci. 2019, 95, 105936. [Google Scholar] [CrossRef]
- Jadhav, S.; Gaikwad, S.; Nimse, M.; Rajbhoj, A. Copper Oxide Nanoparticles: Synthesis, Characterization and Their Antibacterial Activity. J. Clust. Sci. 2011, 22, 121–129. [Google Scholar] [CrossRef]
- Ibrahim, E.M.M.; Abdel-Rahman, L.H.; Abu-Dief, A.M.; Elshafaie, A.; Hamdan, S.K.; Ahmed, A.M. The synthesis of CuO and NiO nanoparticles by facile thermal decomposition of metal-Schiff base complexes and an examination of their electric, thermoelectric and magnetic Properties. Mater. Res. Bull. 2018, 107, 492–497. [Google Scholar] [CrossRef]
- Almasi, H.; Jafarzadeh, P.; Mehryar, L. Fabrication of novel nanohybrids by impregnation of CuO nanoparticles into bacterial cellulose and chitosan nanofibers: Characterization, antimicrobial and release properties. Carbohydr. Polym. 2018, 186, 273–281. [Google Scholar] [CrossRef]
- Eivazihollagh, A.; Bäckström, J.; Dahlström, C.; Carlsson, F.; Ibrahem, I.; Lindman, B.; Edlund, H.; Norgren, M. One-pot synthesis of cellulose-templated copper nanoparticles with antibacterial properties. Mater. Lett. 2017, 187, 170–172. [Google Scholar] [CrossRef]
- Beigmohammadi, F.; Peighambardoust, S.H.; Hesari, J.; Azadmard-Damirchi, S.; Peighambardoust, S.J.; Khosrowshahi, N.K. Antibacterial properties of LDPE nanocomposite films in packaging of UF cheese. LWT—Food Sci. Technol. 2016, 65, 106–111. [Google Scholar] [CrossRef]
- Shankar, S.; Wang, L.F.; Rhim, J.W. Preparation and properties of carbohydrate-based composite films incorporated with CuO nanoparticles. Carbohydr. Polym. 2017, 169, 264–271. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Sathiyaseelan, A.; Mariadoss, A.V.A.; Xiaowen, H.; Wang, M.H. Physical and bioactivities of biopolymeric films incorporated with cellulose, sodium alginate and copper oxide nanoparticles for food packaging application. Int. J. Biol. Macromol. 2020, 153, 207–214. [Google Scholar] [CrossRef]
- Goksungur, Y.; Uren, S.; Guvenc, U. Biosorption of Copper Ions by Caustic Treated Waste Baker’s Yeast Biomass. Turkish J. Biol. 2003, 27, 23–29. [Google Scholar]
- Tran, T.H.; Nguyen, V.T. Copper Oxide Nanomaterials Prepared by Solution Methods, Some Properties, and Potential Applications: A Brief Review. Int. Sch. Res. Not. 2014, 2014, 856592. [Google Scholar] [CrossRef] [PubMed]
- Zimoch-Korzycka, A.; Bobak, Ł.; Jarmoluk, A. Antimicrobial and antioxidant activity of chitosan/hydroxypropyl methylcellulose film-forming hydrosols hydrolyzed by cellulase. Int. J. Mol. Sci. 2016, 17, 1436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asamoah, R.B.; Annan, E.; Mensah, B.; Nbelayim, P.; Apalangya, V.; Onwona-Agyeman, B.; Yaya, A. A Comparative Study of Antibacterial Activity of CuO/Ag and ZnO/Ag Nanocomposites. Adv. Mater. Sci. Eng. 2020, 2020, 7814324. [Google Scholar] [CrossRef]
- Khumkomgool, A.; Saneluksana, T.; Harnkarnsujarit, N. Active meat packaging from thermoplastic cassava starch containing sappan and cinnamon herbal extracts via LLDPE blown-film extrusion. Food Packag. Shelf Life 2020, 26, 100557. [Google Scholar] [CrossRef]
- Wangprasertkul, J.; Siriwattanapong, R.; Harnkarnsujarit, N. Antifungal packaging of sorbate and benzoate incorporated biodegradable films for fresh noodles. Food Control 2021, 123, 107763. [Google Scholar] [CrossRef]
- Toǧrul, H.; Arslan, N. Moisture sorption isotherms and thermodynamic properties of walnut kernels. J. Stored Prod. Res. 2007, 43, 252–264. [Google Scholar] [CrossRef]
- Bumbudsanpharoke, N.; Ko, S. Nano-Food Packaging: An Overview of Market, Migration Research, and Safety Regulations. J. Food Sci. 2015, 80, R910–R923. [Google Scholar] [CrossRef]
- Li, D.; Zhang, J.; Xu, W.; Fu, Y. Effect of SiO2/EVA on the mechanical properties, permeability, and residual solvent of polypropylene packaging films. Polym. Compos. 2016, 37, 101–107. [Google Scholar] [CrossRef]
- Bang, G.; Kim, S.W. Biodegradable poly(lactic acid)-based hybrid coating materials for food packaging films with gas barrier properties. J. Ind. Eng. Chem. 2012, 18, 1063–1068. [Google Scholar] [CrossRef]
- Luo, Z.; Xu, Y.; Ye, Q. Effect of nano-SiO2-LDPE packaging on biochemical, sensory, and microbiological quality of Pacific white shrimp Penaeus vannamei during chilled storage. Fish. Sci. 2015, 81, 983–993. [Google Scholar] [CrossRef]
- Bangar, S.P.; Purewal, S.S.; Trif, M.; Maqsood, S.; Kumar, M.; Manjunatha, V.; Rusu, A.V. Functionality and applicability of starch-based films: An eco-friendly approach. Foods 2021, 10, 2181. [Google Scholar] [CrossRef] [PubMed]
| Coating | Samples | Storage Conditions | Tested Strains | Result | References |
|---|---|---|---|---|---|
| Alginate coating with AgNPs | Shiitake mushrooms | 4 ± 1 °C | Mesophilic, psychrophilic, Pseudomonas, yeast, and molds | Extended shelf life to 16 days | [32] |
| Chitosan impregnated with AgNPs | Strawberries | 7 °C for 25 days | Botrytis cinerea | 10% fungal decay (coated samples) 90% (uncoated samples) | [33] |
| Silver montmorillonite nanoparticles and sodium alginate coating | Fresh cut carrots | - | Prolonged shelf life of 70 days (coated samples) as compared to 4 days (uncoated samples) | [34] | |
| Gum Arabic based coating dispersed with AgNPs | Green bell peppers | 21 days at 7 °C and 20 °C | Aerobic bacteria | Coated samples inhibited the growth of aerobic bacteria with improved appearance and delayed microbial decay compared to uncoated ones; also remains marketable even after 21 days of storage | [35] |
| Carboxymethylcellulose and guar gum-based coating with AgNPs | Kinnow mandarin | 10 °C and 4 °C | Psychrotrophic aerobic yeast and mold | Both coatings limit the growth of yeast and mold at 10 °C but completely prevent at 4 °C; also shelf life increased to 60 days (10 °C) and 120 days (4 °C) | [36] |
| Agar coating enriched with AgNPs | Citrus aurantifolia | Extended shelf life up to 9 days | [37] | ||
| Hydrosol based on chitosan, hydroxypropyl methylcellulose, and nanosilver | Meat surface | 4 °C for 4 weeks | Undesirable bacteria | The reduction of bacteria was 2.5 log CFU/g compared to the uncoated samples | [38] |
| Active pullulan edible packaging with silver nanoparticles | Vacuum-packaged ready-to-eat turkey deli meat | Stored at refrigerated temperature | Food-borne pathogens Listeria monocytogenes and Staphylococcus aureus | Reduction of count from 7 log CFU/g to below the detection limit during 14 days of storage at 4 °C | [39] |
| Edible coating containing silver nanoparticles | Vacuum-packaged sausages | 10 °C | lactic acid bacteria | Can inhibit bacterial activity till 30 days, thus increasing shelf life | [40] |
| Agar hydrosol with nanosilver | Cheese | 10 °C. | Pseudomonas spp. And coliforms bacteria | Inhibited growth of spoilage bacteria and also increased the shelf life to 6 days compared to the untreated one (1.5 days) | [41] |
| Silver nanoparticles stabilized with glutathione | -- | Vacuum sealing and Modified atmospheric packaging | Multidrug-resistant strains of Campylobacter | Susceptibility of Campylobacter strains to silver nanoparticles at a concentration of less than or equal to 9.85 micrograms/mL | [42] |
| Composite films of gelatin, chitosan, polyethylene, and silver nanoparticles | Red Grapes | -- | Molds | Extended the storage life by 14 days | [43] |
| Coating of silver nanoparticles stabilized with cellulose nanocrystals on paper | Strawberries | -- | E. coli and S. aureus | Augmented the shelf life up to 7 days | [44] |
| Green organic-inorganic hybrid nanofibers by the conglomeration of poly(vinyl alcohol) and silver nanoparticles | Lemon and strawberries | Room temperature | B. subtilis, S. aureus, E. coli and P. aeruginosa | Repressed the proliferation of pathogens up to 10 days | [45] |
| Biobased and compostable composite film of PVA-montmorillonite K10 clay ginger extract mediated AgNPs | Chicken sausages | 4 °C | S. Typhimurium and S. aureus | Depreciation in the growth of pathogens when packed in the pouches made from composite films | [46] |
| AgNPs incorporated in agar and banana-based films | E. coli and L. monocytogenes | Superior bacteriocidal property | [47] | ||
| Amazonian tuber starch-based films with AgNPs | Camu Camu fruit | S. aureus and E. coli | Good inhibition towards pathogens; in addition, ripening was delayed | [48] |
| Coating/Packaging | Food Samples | Experimental Condition | Antimicrobial Activity | Result | References |
|---|---|---|---|---|---|
| Incorporation of AuNPs and grapheme oxide separately on PVA (Polyvinyl alcohol) composite films | Banana | Room temperature for 5 days | E. coli | Zone of inhibition for E. coli increased to 10 mm for PVA-Glyoxal-AuNPs as compared to 0 mm for PVA alone | [57] |
| Blend of 3-aminopropyltrimethoxysilane, chitosan, and gold nanoparticles | -- | -- | S. Typhimurium | Exhibited excellent activity against Salmonella sp. owing to the interaction of the constituents with cell membrane leading to their death | [58] |
| AuNPs dispersed on zeolites | -- | -- | E. coli and S. typhi | Materials contained particles sized 5 nm on the surface eliminated 95% of both microorganisms | [51] |
| Combination of bacteriocin and gold nanoparticles | -- | -- | Micrococcus luteus, Bacillus cereus, S. aureus, E. coli | Antibacterial activity increased against food-spoiling bacteria | [53] |
| Gold nanoparticles with nisin | -- | -- | E. coli, S. aureus, B. cereus, K. pneumonia, Proteus mirabilis | Antibacterial activity against food-spoilage microorganisms | [59] |
| AuNPs based colorimetric sensor | Meat and fish spoilage | Dimethyl sulfide and histamine are two significant volatile biogenic markers | -- | Able to detect histamine at 0.035 ppm and dimethyl sulfide at 0.5 ppm | [60] |
| Shapes | Methods | References |
|---|---|---|
| Flower | Solution process at low temperature (90 °C) using zinc acetate dehydrate and NaOH | [87] |
| Flower, prism, snowflakes | Solution process at high temperature (180 °C for 13 h) | [88] |
| Prism like and prickly sphere like | Decomposition method at 100 °C for 13 h | [83] |
| Spherical | Non hydrolytic solution process using zinc acetate | [89] |
| Spherical | Soft chemical solution process | [90] |
| Nanorods of hexagonal prismatic and hexagonal pyramid like | Hydrothermal treatment with stabilizing agents | [75] |
| Nanowires | UV light decomposition process | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dash, K.K.; Deka, P.; Bangar, S.P.; Chaudhary, V.; Trif, M.; Rusu, A. Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review. Polymers 2022, 14, 521. https://doi.org/10.3390/polym14030521
Dash KK, Deka P, Bangar SP, Chaudhary V, Trif M, Rusu A. Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review. Polymers. 2022; 14(3):521. https://doi.org/10.3390/polym14030521
Chicago/Turabian StyleDash, Kshirod Kumar, Pinky Deka, Sneh Punia Bangar, Vandana Chaudhary, Monica Trif, and Alexandru Rusu. 2022. "Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review" Polymers 14, no. 3: 521. https://doi.org/10.3390/polym14030521
APA StyleDash, K. K., Deka, P., Bangar, S. P., Chaudhary, V., Trif, M., & Rusu, A. (2022). Applications of Inorganic Nanoparticles in Food Packaging: A Comprehensive Review. Polymers, 14(3), 521. https://doi.org/10.3390/polym14030521









