Synthesis of Biodegradable Polyester–Polyether with Enhanced Hydrophilicity, Thermal Stability, Toughness, and Degradation Rate
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of Copolymers
2.3. Characterization of Copolymers
3. Results and Discussion
3.1. The Chemical Structures and Compositions
3.2. FTIR-ATR
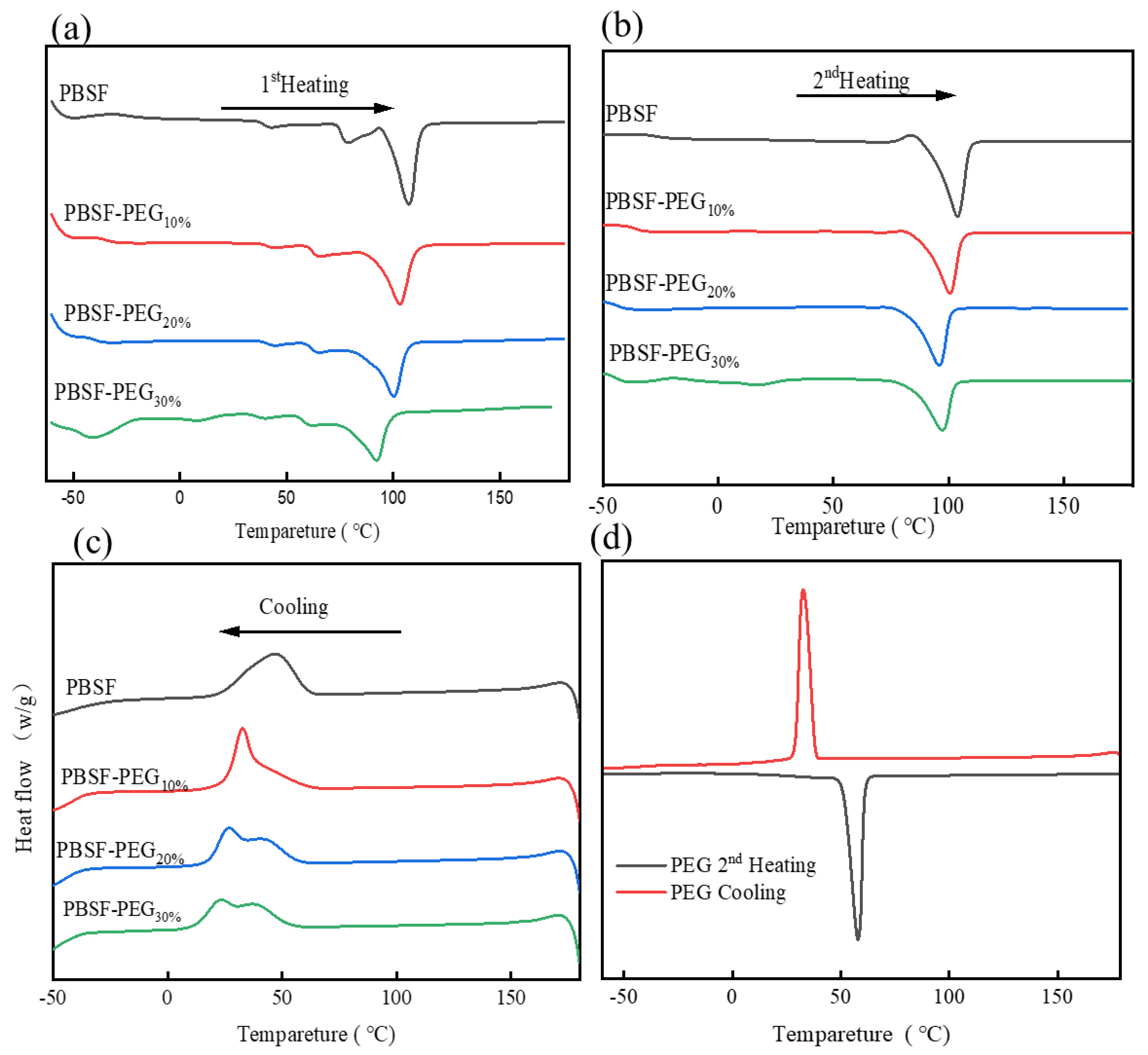
3.3. Thermal Properties of Copolymers
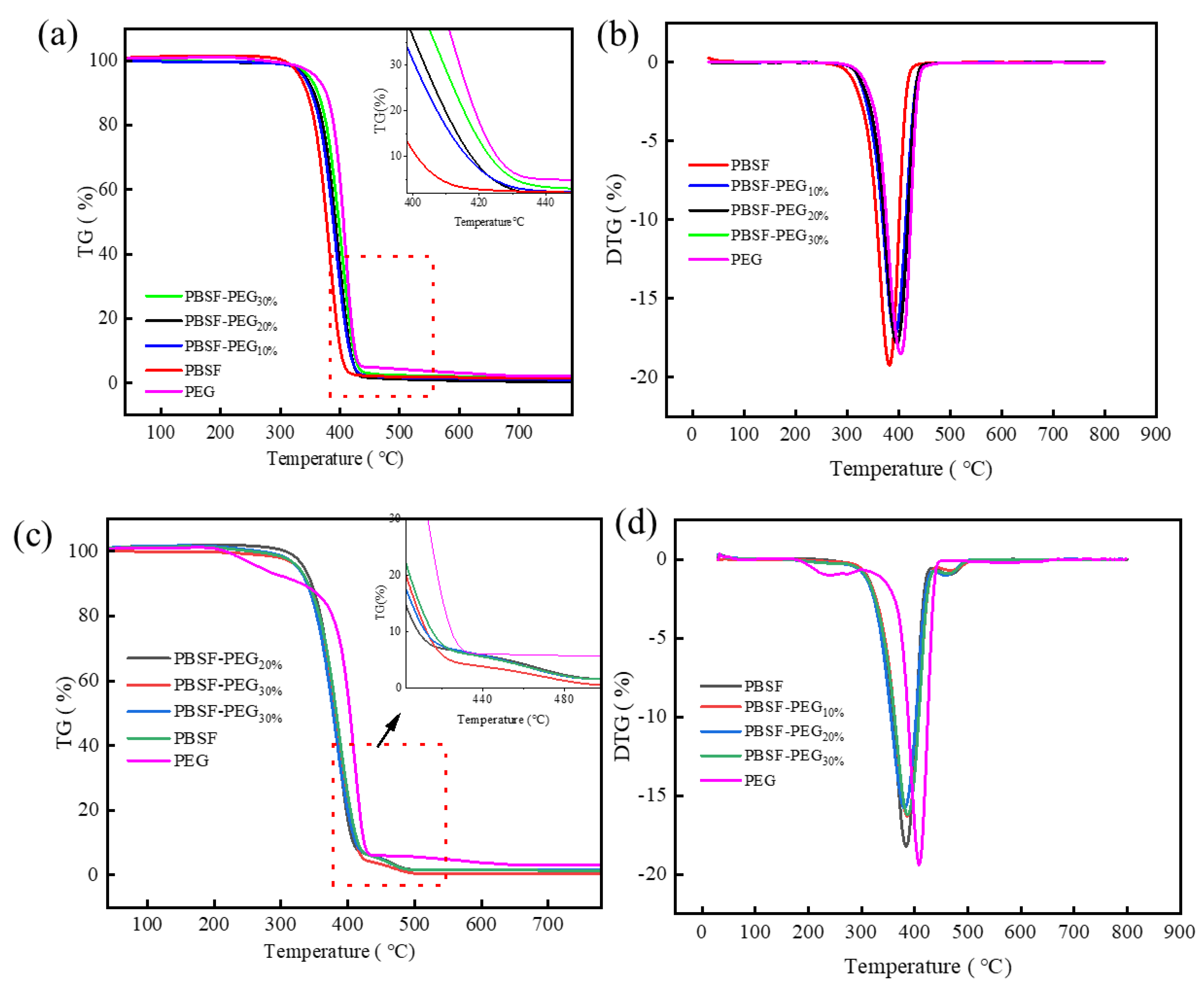
3.3.1. Thermal Stability of PBSF and PBSF-PEG under N2 and Air
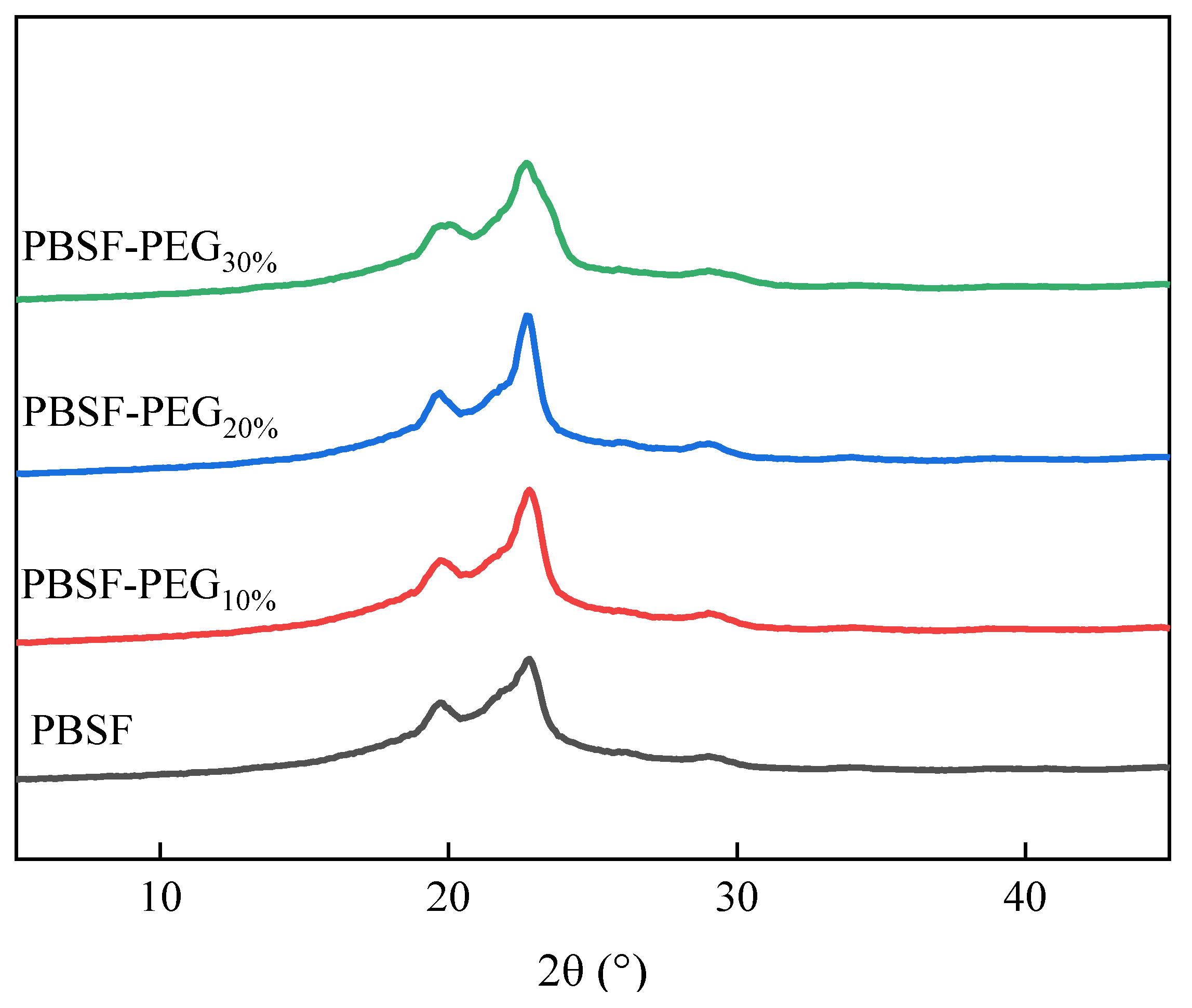
3.3.2. Wide-Angle X-ray Analysis (WAXD) Analysis of PBSF and PBSF-PEG
3.3.3. Water Contact Angle (WCA)
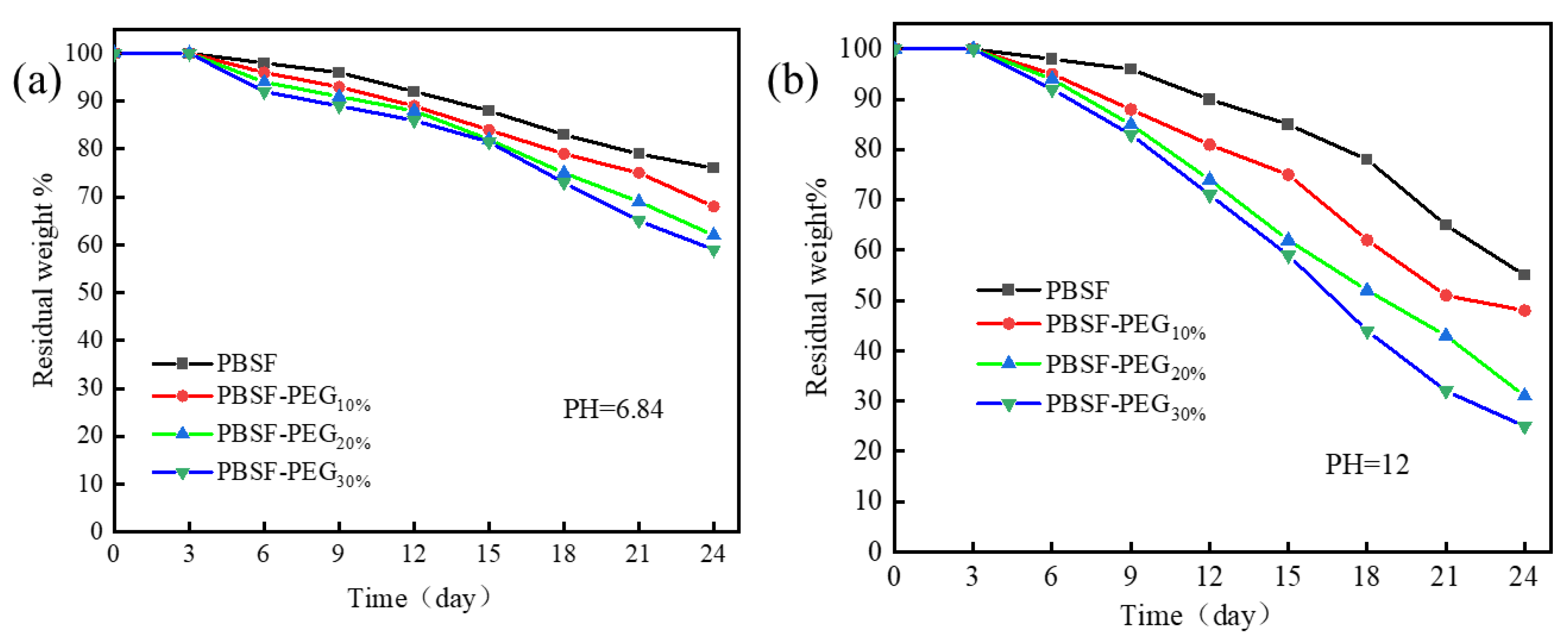
3.3.4. Degradation Properties of PBSF and PBSF-PEG
3.3.5. Mechanical Properties of PBSF and PBSF-PEG
3.3.6. Transmittance Properties of PBSF-PEG
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barletta, M.; Aversa, C.; Ayyoob, M.; Gisario, A.; Hamad, K.; Mehrpouya, M.; Vahabi, H. Poly(butylene succinate) (PBS): Materials, processing, and industrial applications. Prog. Polym. Sci. 2022, 132, 101579. [Google Scholar] [CrossRef]
- Aliotta, L.; Seggiani, M.; Lazzeri, A.; Gigante, V.; Cinelli, P. A Brief Review of Poly(Butylene Succinate) (PBS) and Its Main Copolymers: Synthesis, Blends, Composites, Biodegradability, and Applications. Polymers 2022, 14, 844. [Google Scholar] [CrossRef]
- Zhang, Q.; Song, M.; Xu, Y.; Wang, W.; Wang, Z.; Zhang, L. Bio-based polyesters: Recent progress and future prospects. Prog. Polym. Sci. 2021, 120, 101430. [Google Scholar] [CrossRef]
- Dirauf, M.; Muljajew, I.; Weber, C.; Schubert, U.S. Recent advances in degradable synthetic polymers for biomedical applications - Beyond polyesters. Prog. Polym. Sci. 2022, 129, 101547. [Google Scholar] [CrossRef]
- Badia, J.D.; Gil-Castell, O.; Ribes-Greus, A. Long-term properties and end-of-life of polymers from renewable resources. Polym. Degrad. Stab. 2017, 137, 35–57. [Google Scholar] [CrossRef]
- Rafiqah, S.A.; Khalina, A.; Harmaen, A.S.; Tawakkal, I.A.; Zaman, K.; Asim, M.; Nurrazi, M.N.; Lee, C.H. A Review on Properties and Application of Bio-Based Poly(Butylene Succinate). Polymers 2021, 13, 1436. [Google Scholar] [CrossRef]
- Mochane, M.J.; Magagula, S.I.; Sefadi, J.S.; Mokhena, T.C. A Review on Green Composites Based on Natural Fiber-Reinforced Polybutylene Succinate (PBS). Polymers 2021, 13, 1200. [Google Scholar] [CrossRef]
- Henke, L.; Zarrinbakhsh, N.; Endres, H.-J.; Misra, M.; Mohanty, A.K. Biodegradable and Bio-based Green Blends from Carbon Dioxide-Derived Bioplastic and Poly(Butylene Succinate). J. Polym. Environ. 2017, 25, 499–509. [Google Scholar] [CrossRef]
- Platnieks, O.; Gaidukovs, S.; Kumar Thakur, V.; Barkane, A.; Beluns, S. Bio-based poly(butylene succinate): Recent progress, challenges and future opportunities. Eur. Polym. J. 2021, 161, 110855. [Google Scholar] [CrossRef]
- Hsu, K.-H.; Chen, C.-W.; Wang, L.-Y.; Chan, H.-W.; He, C.-L.; Cho, C.-J.; Rwei, S.-P.; Kuo, C.-C. Bio-based thermoplastic poly(butylene succinate-co-propylene succinate) copolyesters: Effect of glycerol on thermal and mechanical properties. Soft Matter 2019, 15, 9710–9720. [Google Scholar] [CrossRef]
- Jin, C.; Wang, B.; Liu, L.; Tu, Z.; Wei, Z. Biodegradable Poly(butylene succinate) Copolyesters Modified by Bioresoured 2,5-Tetrahydrofurandimethanol. ACS Sustain. Chem. Eng. 2022, 10, 11203–11214. [Google Scholar] [CrossRef]
- Chen, B.-M.; Cheng, T.-L.; Roffler, S.R. Polyethylene Glycol Immunogenicity: Theoretical, Clinical, and Practical Aspects of Anti-Polyethylene Glycol Antibodies. ACS Nano 2021, 15, 14022–14048. [Google Scholar] [CrossRef]
- Kwizera, E.A.; Ou, W.; Lee, S.; Stewart, S.; Shamul, J.G.; Xu, J.; Tait, N.; Tkaczuk, K.H.R.; He, X. Greatly Enhanced CTC Culture Enabled by Capturing CTC Heterogeneity Using a PEGylated PDMS–Titanium–Gold Electromicrofluidic Device with Glutathione-Controlled Gentle Cell Release. ACS Nano 2022, 16, 11374–11391. [Google Scholar] [CrossRef]
- Liu, K.; He, Y.; Yao, Y.; Zhang, Y.; Cai, Z.; Ru, J.; Zhang, X.; Jin, X.; Xu, M.; Li, Y.; et al. Methoxy polyethylene glycol modification promotes adipogenesis by inducing the production of regulatory T cells in xenogeneic acellular adipose matrix. Mater. Today Bio 2021, 12, 100161. [Google Scholar] [CrossRef]
- Takashima, R.; Ohira, M.; Yokochi, H.; Aoki, D.; Li, X.; Otsuka, H. Characterization of N-phenylmaleimide-terminated poly(ethylene glycol)s and their application to a tetra-arm poly(ethylene glycol) gel. Soft Matter 2020, 16, 10869–10875. [Google Scholar] [CrossRef]
- Feng, X.; Wang, G.; Neumann, K.; Yao, W.; Ding, L.; Li, S.; Sheng, Y.; Jiang, Y.; Bradley, M.; Zhang, R. Synthesis and characterization of biodegradable poly(ether-ester) urethane acrylates for controlled drug release. Mater. Sci. Eng. C 2017, 74, 270–278. [Google Scholar] [CrossRef]
- Sha, D.; Wu, Z.; Zhang, J.; Ma, Y.; Yang, Z.; Yuan, Y. Development of modified and multifunctional poly(glycerol sebacate) (PGS)-based biomaterials for biomedical applications. Eur. Polym. J. 2021, 161, 110830. [Google Scholar] [CrossRef]
- Dethe, M.R.; Prabakaran, A.; Ahmed, H.; Agrawal, M.; Roy, U.; Alexander, A. PCL-PEG copolymer based injectable thermosensitive hydrogels. J. Control. Release 2022, 343, 217–236. [Google Scholar] [CrossRef]
- Perinelli, D.R.; Cespi, M.; Bonacucina, G.; Palmieri, G.F. PEGylated polylactide (PLA) and poly(lactic-co-glycolic acid) (PLGA) copolymers for the design of drug delivery systems. J. Pharm. Investig. 2019, 49, 443–458. [Google Scholar] [CrossRef]
- Wang, J.-Z.; You, M.-L.; Ding, Z.-Q.; Ye, W.-B. A review of emerging bone tissue engineering via PEG conjugated biodegradable amphiphilic copolymers. Mater. Sci. Eng. C 2019, 97, 1021–1035. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, W.; Wang, Z.; Wang, Z.; Xie, Q.; Niu, H.; Guo, H.; Yuan, Y.; Liu, C. PEGylated poly(glycerol sebacate)-modified calcium phosphate scaffolds with desirable mechanical behavior and enhanced osteogenic capacity. Acta Biomater. 2016, 44, 110–124. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, H.; Wang, Z.; Zhang, J.; Zhu, J.; Ma, Y.; Yang, Z.; Yuan, Y. Optimized Synthesis of Biodegradable Elastomer PEGylated Poly(glycerol sebacate) and Their Biomedical Application. Polymers 2019, 11, 965. [Google Scholar] [CrossRef]
- Zhou, X.-M. Synthesis and characterization of polyester copolymers based on poly(butylene succinate) and poly(ethylene glycol). Mater. Sci. Eng. C 2012, 32, 2459–2463. [Google Scholar] [CrossRef]
- Huang, C.-L.; Jiao, L.; Zhang, J.-J.; Zeng, J.-B.; Yang, K.-K.; Wang, Y.-Z. Poly(butylene succinate)-poly(ethylene glycol) multiblock copolymer: Synthesis, structure, properties and shape memory performance. Polym. Chem. 2012, 3, 800–808. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, M.; Zhang, Q.; Wang, R.; Zhou, G. Biobased multiblock copolymers: Synthesis, properties and shape memory performance of poly(ethylene 2,5-furandicarboxylate)-b-poly(ethylene glycol). Polym. Degrad. Stab. 2017, 144, 121–127. [Google Scholar] [CrossRef]
- Hu, B.; Xia, H.; Liu, F.; Ni, Q.-Q. Heat-stimuli controllability of shape memory thermoplastic epoxy filaments by adding polyethylene glycol. Polymer 2022, 250, 124818. [Google Scholar] [CrossRef]
- Guo, Y.; Ma, J.; Lv, Z.; Zhao, N.; Wang, L.; Li, Q. The effect of plasticizer on the shape memory properties of poly(lactide acid)/poly(ethylene glycol) blends. J. Mater. Res. 2018, 33, 4101–4112. [Google Scholar] [CrossRef]
- Feng, L.-d.; Xiang, S.; Sun, B.; Liu, Y.-l.; Sun, Z.-q.; Bian, X.-c.; Li, G.; Chen, X.-s. Thermal, morphological, mechanical and aging properties of polylactide blends with poly(ether urethane) based on chain-extension reaction of poly(ethylene glycol) using diisocyanate. Chin. J. Polym. Sci. 2016, 34, 1070–1078. [Google Scholar] [CrossRef]
- Parvez, M.K.; In, I.; Park, J.M.; Lee, S.H.; Kim, S.R. Long-term stable dye-sensitized solar cells based on UV photo-crosslinkable poly(ethylene glycol) and poly(ethylene glycol) diacrylate based electrolytes. Sol. Energy Mater. Sol. Cells 2011, 95, 318–322. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, Y.S.; Topolkaraev, V.; Hiltner, A.; Baer, E. Aging of poly(lactide)/poly(ethylene glycol) blends. Part 2. Poly(lactide) with high stereoregularity. Polymer 2003, 44, 5711–5720. [Google Scholar] [CrossRef]
- Lv, X.D.; Luo, F.L.; Zheng, L.C.; Niu, R.X.; Liu, Y.; Xie, Q.Q.; Song, D.Q.; Zhang, Y.C.A.; Zhou, T.B.; Zhu, S.F. Biodegradable poly(butylene succinate-co-butylene furandicarboxylate): Effect of butylene furandicarboxylate unit on thermal, mechanical, and ultraviolet shielding properties, and biodegradability. J. Appl. Polym. Sci. 2022, 139, e53122. [Google Scholar] [CrossRef]
- Correlo, V.M.; Boesel, L.F.; Bhattacharya, M.; Mano, J.F.; Neves, N.M.; Reis, R.L. Properties of melt processed chitosan and aliphatic polyester blends. Mater. Sci. Eng. A 2005, 403, 57–68. [Google Scholar] [CrossRef]
- Trapasso, G.; Annatelli, M.; Dalla Torre, D.; Aricò, F. Synthesis of 2,5-furandicarboxylic acid dimethyl ester from galactaric acid via dimethyl carbonate chemistry. Green Chem. 2022, 24, 2766–2771. [Google Scholar] [CrossRef]
- Hu, H.; Zhang, R.; Sousa, A.; Long, Y.; Ying, W.B.; Wang, J.; Zhu, J. Bio-based poly(butylene 2,5-furandicarboxylate)-b-poly(ethylene glycol) copolymers with adjustable degradation rate and mechanical properties: Synthesis and characterization. Eur. Polym. J. 2018, 106, 42–52. [Google Scholar] [CrossRef]
- León, A.; Reuquen, P.; Garín, C.; Segura, R.; Vargas, P.; Zapata, P.; Orihuela, P.A. FTIR and Raman Characterization of TiO2 Nanoparticles Coated with Polyethylene Glycol as Carrier for 2-Methoxyestradiol. Appl. Sci. 2017, 7, 49. [Google Scholar] [CrossRef]
- Huang, C.-L.; Jiao, L.; Zeng, J.-B.; Zhang, J.-J.; Yang, K.-K.; Wang, Y.-Z. Fractional Crystallization and Homogeneous Nucleation of Confined PEG Microdomains in PBS-PEG Multiblock Copolymers. J. Phys. Chem. B 2013, 117, 10665–10676. [Google Scholar] [CrossRef]
- Piccini, M.; Lightfoot, J.; Dominguez, B.C.; Buchard, A. Xylose-Based Polyethers and Polyesters Via ADMET Polymerization toward Polyethylene-Like Materials. ACS Appl. Polym. Mater. 2021, 3, 5870–5881. [Google Scholar] [CrossRef]
- Bai, Z.; Shi, K.; Su, T.; Wang, Z. Correlation between the chemical structure and enzymatic hydrolysis of Poly(butylene succinate), Poly(butylene adipate), and Poly(butylene suberate). Polym. Degrad. Stab. 2018, 158, 111–118. [Google Scholar] [CrossRef]
- De Falco, F.; Avolio, R.; Errico, M.E.; Di Pace, E.; Avella, M.; Cocca, M.; Gentile, G. Comparison of biodegradable polyesters degradation behavior in sand. J. Hazard. Mater. 2021, 416, 126231. [Google Scholar] [CrossRef]
- Müller, S.S.; Fritz, T.; Gimnich, M.; Worm, M.; Helm, M.; Frey, H. Biodegradable hyperbranched polyether-lipids with in-chain pH-sensitive linkages. Polym. Chem. 2016, 7, 6257–6268. [Google Scholar] [CrossRef][Green Version]
- Guo, J.; Liu, X.; Liu, M.; Han, M.; Liu, Y.; Ji, S. Effect of molecular weight of Poly(ethylene glycol) on plasticization of Poly(ʟ-lactic acid). Polymer 2021, 223, 123720. [Google Scholar] [CrossRef]
- Chen, H.; Liu, H.; Chen, M.; Ge, P.; Chen, S.; Yuan, H. Preparation of thermostable and compatible citrate-based polyesters for enhancing the ultraviolet shielding performance of thermoplastic resin. Polym. Chem. 2021, 12, 1939–1949. [Google Scholar] [CrossRef]
- Hung, A.; Fini, E.H. Surface Morphology and Chemical Mapping of UV-Aged Thin Films of Bitumen. ACS Sustain. Chem. Eng. 2020, 8, 11764–11771. [Google Scholar] [CrossRef]
- Zhao, J.; Liu, T.; Xia, K.; Liu, X.; Zhang, X. Preparation and application of edible agar-based composite films modified by cellulose nanocrystals. Food Packag. Shelf Life 2022, 34, 100936. [Google Scholar] [CrossRef]

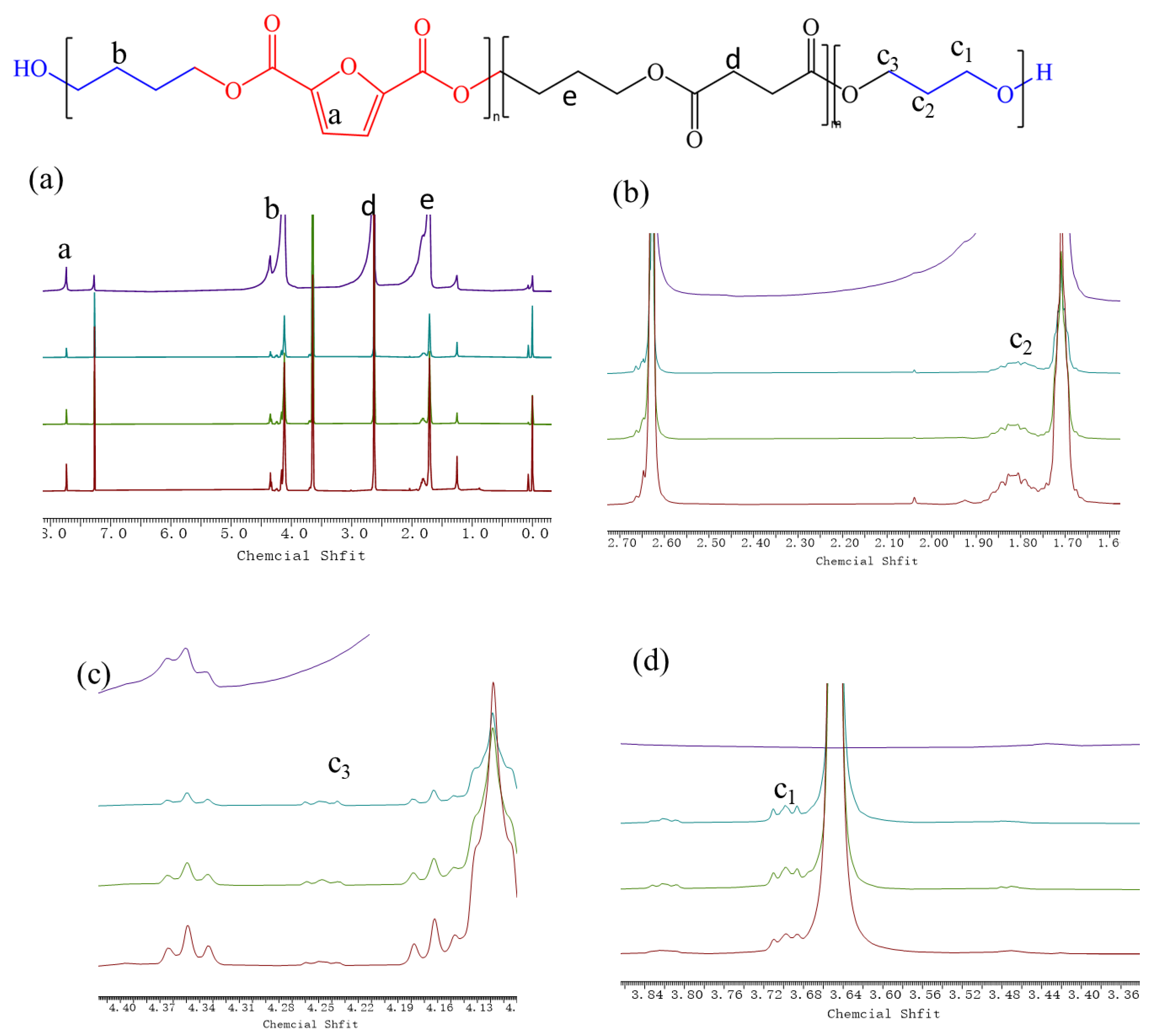





| Sample | PEG (mol%) | Polycondensation Temperature (°C) | [η] | Mn × 104 (g/mol) | Mw × 104 (g/mol) | PDI | |
|---|---|---|---|---|---|---|---|
| Feed | Found | ||||||
| PBSF10% | nd | nd | 230 | 1.65 | 5.48 | 10.81 | 1.96 |
| PBSF-PEG10% | 10.00 | 9.52 | 230 | 2.03 | 6.88 | 12.19 | 1.77 |
| PBSF-PEG20% | 20.00 | 19.04 | 230 | 2.25 | 7.90 | 13.68 | 1.73 |
| PBSF-PEG30% | 30.00 | 28.32 | 230 | 1.82 | 6.05 | 12.16 | 2.00 |
| * PBS-PEG10% | -- | -- | -- | -- | 6.32 | 8.59 | 1.36 |
| * PBS-PEG15% | -- | -- | -- | -- | 3.41 | 6.75 | 1.98 |
| Sample | DSC | WAXD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st Heating Scan | Cooling Scan | 2nd Heating Scan | |||||||||
| Tm (°C) | ΔHm (J/g) | Tc (°C) | ΔHc (J/g) | Tg (°C) | Tcc (°C) | ΔHcc (J/g) | Tm (°C) | ΔHm (J/g) | Xc,h (%) | Xc (%) | |
| PBSF | 107.4 | 40.52 | 44.6 | 55.27 | −26.6 | 84.5 | 5.78 | 105.2 | 45.89 | 37.5 | 29.5 |
| PBSF-PEG10% | 103.2 | 35.31 | 32.7 | 50.24 | −35.3 | 80.2 | 1.37 | 100.7 | 41.41 | 33.4 | 27.8 |
| PBSF-PEG20% | 100.2 | 34.07 | 27.2 | 46.76 | −41.1 | 64.6 | 3.08 | 98.5 | 43.15. | 31.5 | 25.1 |
| PBSF-PEG30% | 98.3 | 25.92 | 23.5 | 43.81 | −47.9 | 44.1 | 0.25 | 96.8 | 40.49 | 25.4 | 22.6 |
| PEG | -- | -- | 32.6 | 194.8 | -- | -- | -- | 57.9 | 195.4 | -- | -- |
| Sample | TGA | |||||
|---|---|---|---|---|---|---|
| N2 | Air | |||||
| T5% (°C) | Td,max (°C) | R600 (%) | T5% (°C) | Td,max (°C) | R600 (%) | |
| PBSF | 358.1 | 389.1 | 1.51 | 352.1 | 382.0 | 4.51 |
| PBSF-PEG10% | 360.4 | 393.2 | 1.10 | 355.8 | 391.7 | 3.56 |
| PBSF-PEG20% | 365.2 | 396.5 | 0.49 | 354.2 | 387.7 | 4.91 |
| PBSF-PEG30% | 370.0 | 404.1 | 1.31 | 359.8 | 393.9 | 4.57 |
| PEG | 265.5 | 409.3 | 2.24 | 243.5 | 408.4 | 3.23 |
| Sample | E (MPa) | (MPa) | (MPa) | εb (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Dry | Swollen | Dry | Swollen | Dry | Swollen | Dry | Swollen | |
| PBSF | 290 ± 20 | 290 ± 25 | 24 ± 2 | 24 ± 2 | 39 ± 3 | 39 ± 2 | 490 ± 30 | 510 ± 30 |
| PBSF-PEG10% | 249 ± 40 | 249 ± 40 | 18 ± 2 | 18 ± 1 | 31 ± 4 | 31 ± 3 | 520 ± 40 | 525 ± 40 |
| PBSF-PEG20% | 200 ± 20 | 198 ± 20 | 12 ± 2 | 12 ± 4 | 25 ± 3 | 25 ± 3 | 900 ± 30 | 915 ± 30 |
| PBSF-PEG30% | 178 ± 50 | 175 ± 50 | 8 ± 1 | 8 ± 2 | 23 ± 1 | 23 ± 1 | 1580 ± 40 | 1240 ± 40 |
| Sample | T320 (%) | T380 (%) | T400 (%) | T450 (%) |
|---|---|---|---|---|
| PBSF | 3.2 | 47.5 | 83.3 | 89.1 |
| PBSF-PEG10% | 32.4 | 75.9 | 88.7 | 90.6 |
| PBCBS-PEG20% | 58.9 | 76.6 | 89.6 | 91.5 |
| PBCBS-PEG30% | 58.9 | 86.5 | 93.2 | 95.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, X.; Lin, H.; Wang, Z.; Niu, R.; Liu, Y.; Wei, Y.; Zheng, L. Synthesis of Biodegradable Polyester–Polyether with Enhanced Hydrophilicity, Thermal Stability, Toughness, and Degradation Rate. Polymers 2022, 14, 4895. https://doi.org/10.3390/polym14224895
Lv X, Lin H, Wang Z, Niu R, Liu Y, Wei Y, Zheng L. Synthesis of Biodegradable Polyester–Polyether with Enhanced Hydrophilicity, Thermal Stability, Toughness, and Degradation Rate. Polymers. 2022; 14(22):4895. https://doi.org/10.3390/polym14224895
Chicago/Turabian StyleLv, Xuedong, Haitao Lin, Zhengxiang Wang, Ruixue Niu, Yi Liu, Yen Wei, and Liuchun Zheng. 2022. "Synthesis of Biodegradable Polyester–Polyether with Enhanced Hydrophilicity, Thermal Stability, Toughness, and Degradation Rate" Polymers 14, no. 22: 4895. https://doi.org/10.3390/polym14224895
APA StyleLv, X., Lin, H., Wang, Z., Niu, R., Liu, Y., Wei, Y., & Zheng, L. (2022). Synthesis of Biodegradable Polyester–Polyether with Enhanced Hydrophilicity, Thermal Stability, Toughness, and Degradation Rate. Polymers, 14(22), 4895. https://doi.org/10.3390/polym14224895








