TPV Foaming by CO2 Extrusion: Processing and Modelling
Abstract
:1. Introduction
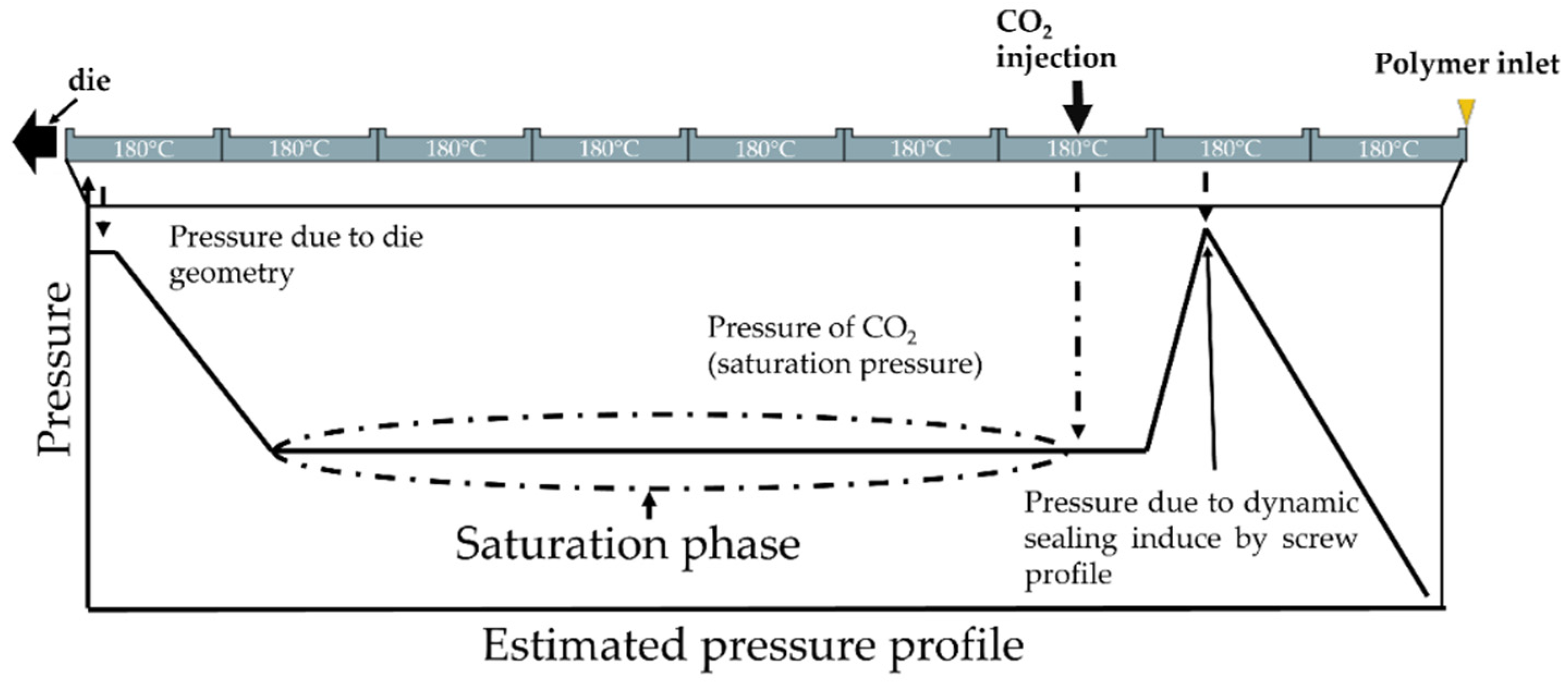
2. Materials and Methods
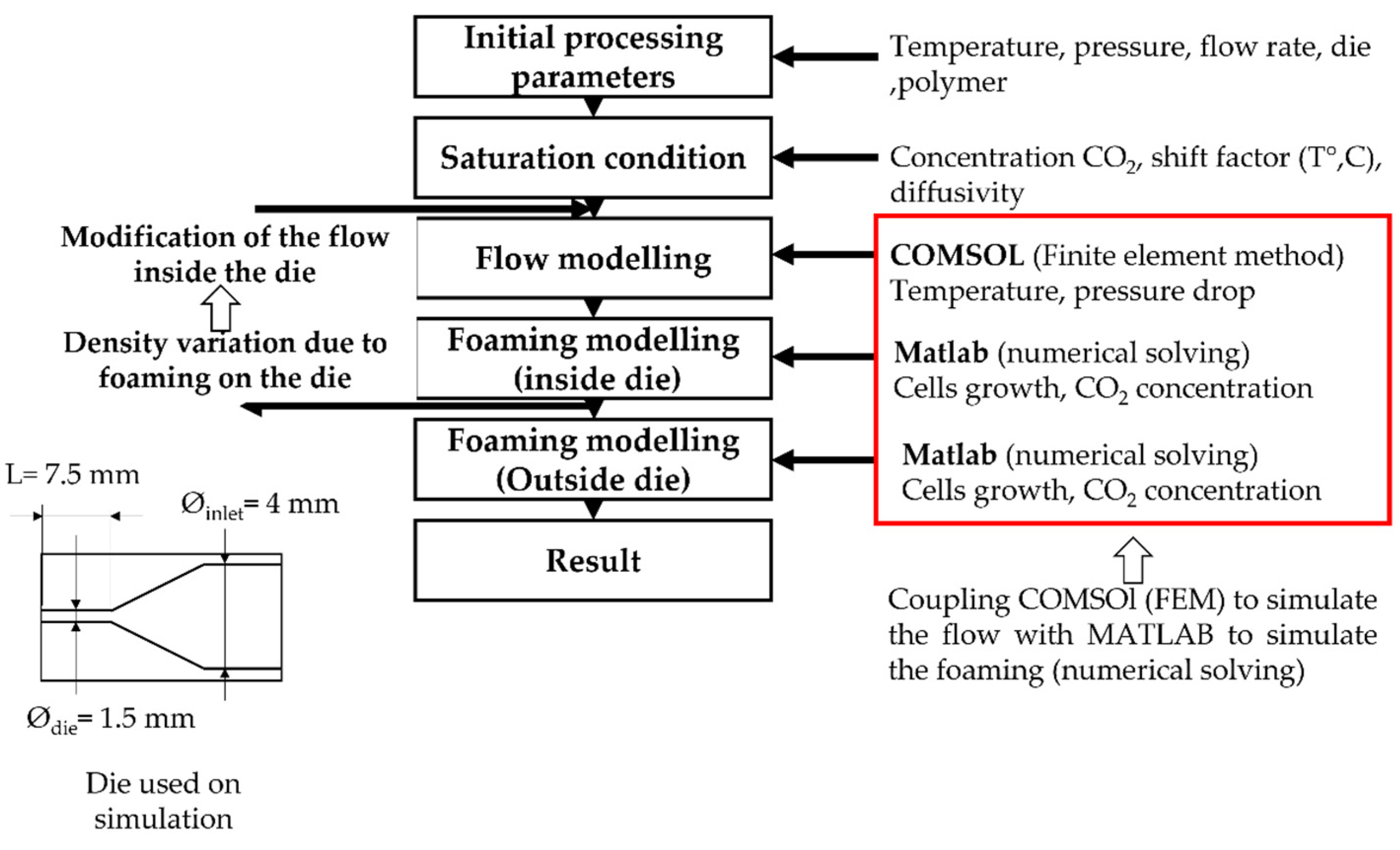
- Modelling
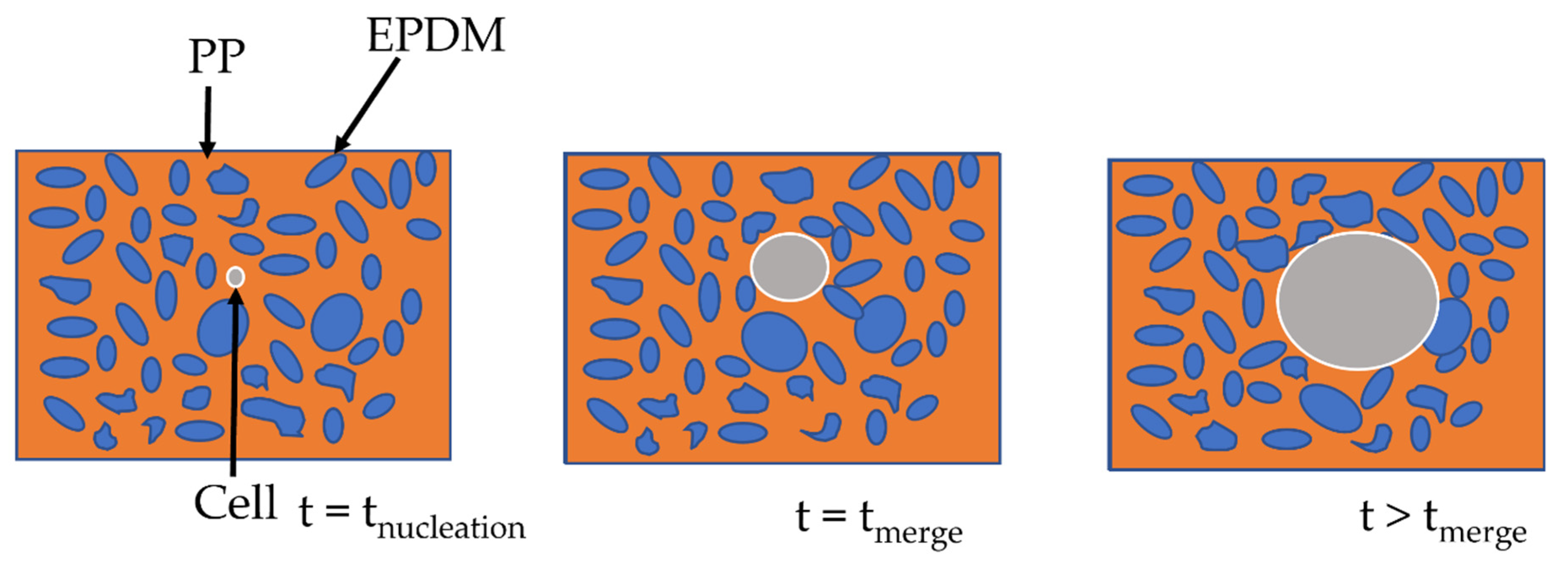
- Cell growth Simulation
- Nucleation simulation
- Plasticizing model:
- Foam growth resolution
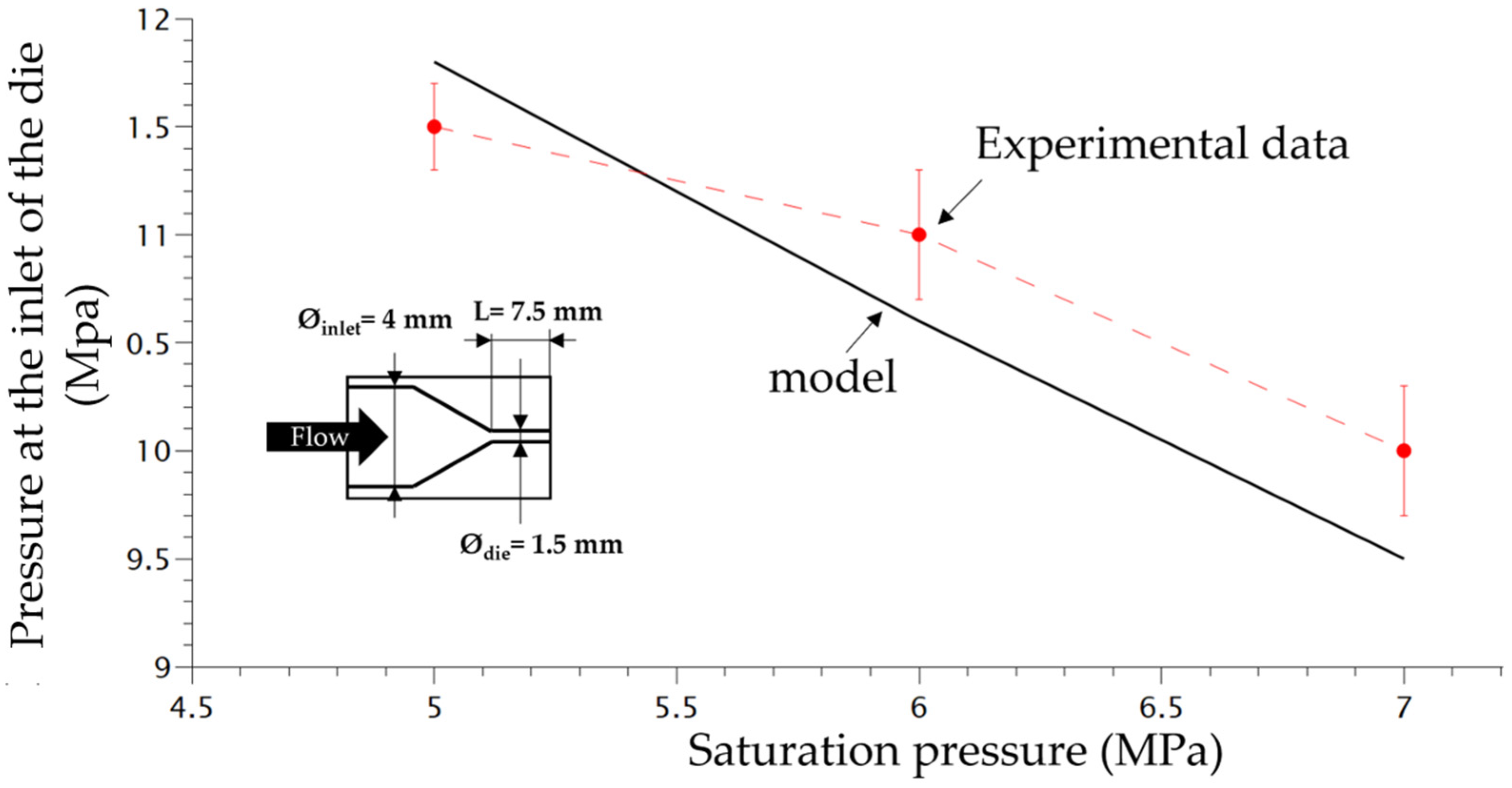
3. Results and Discussion
3.1. Influence of CO2 Content on Foaming Behaviour
3.2. Numerical Modelling of Cell Growth
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhai, W.; Jiang, J.; Park, C.B. A review on physical foaming of thermoplastic and vulcanized elastomers. Polym. Rev. 2022, 62, 95–141. [Google Scholar] [CrossRef]
- Banerjee, R.; Ray, S.S. Foamability and Special Applications of Microcellular Thermoplastic Polymers: A Review on Recent Advances and Future Direction. Macromol. Mater. Eng. 2020, 305, 2000366. [Google Scholar] [CrossRef]
- Chauvet, M. Extrusion Assistee Par CO2 Supercritique Appliquee au Moussage D’un Biopolymere, le Poly(Acide Lactique), Seul ou en Melange a de L’amidon: Etude Experimentale et Modelisation; Ecole des Mines d’Albi-Carmaux: Albi, France, 2017. [Google Scholar]
- Kim, S.G.; Leung, S.N.; Park, C.B.; Sain, M. The effect of dispersed elastomer particle size on heterogeneous nucleation of TPO with N2 foaming. Chem. Eng. Sci 2011, 66, 3675–3686. [Google Scholar] [CrossRef]
- Kim, S.G.; Park, C.B.; Kang, B.S.; Sain, M. Foamability of Thermoplastic Vulcanizates (TPVs) with Carbon Dioxide and Nitrogen. Cell. Polym. 2006, 25, 19–33. [Google Scholar] [CrossRef]
- Kim, S.G.; Park, C.B.; Sain, M. Foamability of thermoplastic vulcanizates blown with various physical blowing agents. J. Cell Plast. 2008, 44, 53–67. [Google Scholar] [CrossRef]
- Taki, K.; Nakayama, T.; Yatsuzuka, T.; Ohshima, M. Visual Observations of Batch and Continuous Foaming Processes. J. Cell Plast. 2003, 39, 155–169. [Google Scholar] [CrossRef]
- Spitael, P.; Macosko, C.W. Strain Hardening in Polypropylenes and Its Role in Extrusion Foaming. Polym. Eng. Sci. 2004, 44, 2090–2100. [Google Scholar] [CrossRef]
- Xu, Z.-M.; Jiang, X.-L.; Liu, T.; Hu, G.-H.; Zhao, L.; Zhu, Z.-N.; Yuan, W.-K. Foaming of polypropylene with supercritical carbon dioxide. J. Supercrit. Fluids 2007, 41, 299–310. [Google Scholar] [CrossRef]
- Mohebbi, A.; Mighri, F.; Ajji, A.; Rodrigue, D. Current issues and challenges in polypropylene foaming: A review. Cell Polym. 2015, 34, 299–337. [Google Scholar] [CrossRef]
- Naguib, H.E.; Park, C.B.; Reichelt, N. Fundamental foaming mechanisms governing the volume expansion of extruded polypropylene foams. J. Appl. Polym. Sci. 2004, 91, 2661–2668. [Google Scholar] [CrossRef]
- Arefmanesh, A.; Advani, S.G. Diffusion-induced growth of a gas bubble in a viscoelastic fluid. Rheol Acta 1991, 30, 274–283. [Google Scholar] [CrossRef]
- Shimoda, M.; Tsujimura, I.; Tanigaki, M.; Ohshima, M. Polymeric foaming simulation for extrusion processes. J. Cell Plast. 2001, 37, 517–536. [Google Scholar] [CrossRef]
- Azimi, H.; Jahani, D. The experimental and numerical relation between the solubility, diffusivity and bubble nucleation of supercritical CO2 in Polystyrene via visual observation apparatus. J. Supercrit. Fluids 2018, 139, 30–37. [Google Scholar] [CrossRef]
- Leung, S.N.; Park, C.B.; Xu, D.; Li, H.; Fenton, R.G. Computer simulation of bubble-growth phenomena in foaming. Ind. Eng. Chem. Res. 2006, 45, 7823–7831. [Google Scholar] [CrossRef]
- Rainglet, B.; Chalamet, Y.; Bounor-Legaré, V.; Delage, K.; Forest, C.; Cassagnau, P. Polypropylene foams under CO2 batch conditions: From formulation and rheological modeling to cell-growth simulation. Polymer 2021, 218, 123496. [Google Scholar] [CrossRef]
- Leung, S.N.; Park, C.B.; Li, H. Numerical simulation of polymeric foaming processes using modified nucleation theory. Plast. Rubber Compos. 2006, 35, 93–100. [Google Scholar] [CrossRef]
- Daoud, M.; Cotton, J.P.; Farnoux, B.; Jannink, G.; Sarma, G.; Benoit, H.; Duplessix, C.; Picot, C.; de Gennes, P.G. Solutions of Flexible Polymers. Neutron Experiments and Interpretation. Macromolecules 1975, 8, 804–818. [Google Scholar] [CrossRef]
- Lei, Z.; Ohyabu, H.; Sato, Y.; Inomata, H.; Smith, R.L. Solubility, swelling degree and crystallinity of carbon dioxide-polypropylene system. J. Supercrit. Fluids 2007, 40, 452–461. [Google Scholar] [CrossRef]
- Ramesh, N.S. Foam Growth in Polymers. In Foam Extrusion, 2nd ed.; Lee, S.-T., Park, C.B., Eds.; C.R.C. Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Sun, Y.; Ueda, Y.; Suganaga, H.; Haruki, M.; Kihara, S.I.; Takishima, S. Pressure drop threshold in the foaming of low-density polyethylene, polystyrene, and polypropylene using CO2 and N2 as foaming agents. J. Supercrit. Fluids 2015, 103, 38–47. [Google Scholar] [CrossRef]



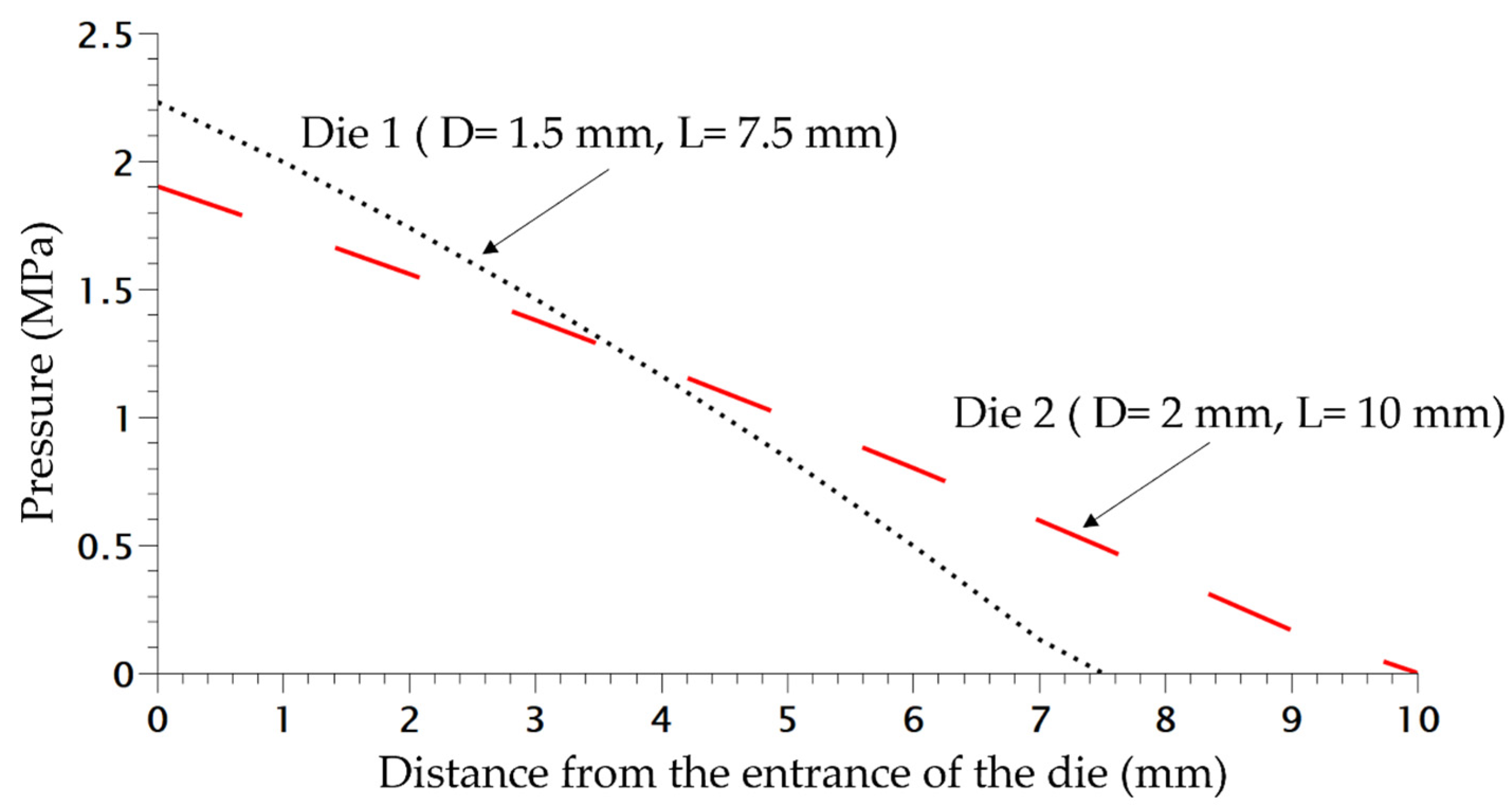


| Die | Radius (mm) | Length (mm) | Drop Pressure (MPa) 1 | Drop Rate Pressure (MPa/s) |
|---|---|---|---|---|
| 1 | 0.75 | 7.5 | 9.7 | 135 |
| 2 | 1 | 10 | 9.2 | 45 |
| Parameters | Value |
|---|---|
| Range of Rsup | Between 10 and 12.5 µm |
| KH (at 433 K) | 1.64 × 10−4 mol Pa/m3 [11] |
| D (at 433 K) | 6 × 10−9 m2/s [2] |
| R0 | 38 nm |
| Mass flow rate | 2 kg·h−1 |
| Saturation Pressure (MPa) | Die | T (°C) | Mean Radius Experimental (µm) | Mean Modelized Radius (µm) | Experimental Density (kg/m3) | Modelized Density (kg/m3) | Modelized Number of Cells (Cells/m3) | Experimental Number of Cells (Cells/m3) |
|---|---|---|---|---|---|---|---|---|
| 7 | 1 | 160 | Deformed cell | 12 | 750 | 520 | 8.5 × 1013 | None |
| 6 | 1 | 160 | Deformed cell | 26 | 610 | 410 | 1.6 × 1013 | None |
| 5 | 1 | 160 | 30 | 31 | 480 | 510 | 8 × 1012 | 8 × 1012 |
| 4 | 2 | 160 | 25 | 27 | 700 | 650 | 6 × 1012 | 5 × 1012 |
| 3 | 2 | 165 | 19 | 22 | 770 | 740 | 4 × 1012 | 2.5 × 1012 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rainglet, B.; Besognet, P.; Benoit, C.; Delage, K.; Bounor-Legaré, V.; Forest, C.; Cassagnau, P.; Chalamet, Y. TPV Foaming by CO2 Extrusion: Processing and Modelling. Polymers 2022, 14, 4513. https://doi.org/10.3390/polym14214513
Rainglet B, Besognet P, Benoit C, Delage K, Bounor-Legaré V, Forest C, Cassagnau P, Chalamet Y. TPV Foaming by CO2 Extrusion: Processing and Modelling. Polymers. 2022; 14(21):4513. https://doi.org/10.3390/polym14214513
Chicago/Turabian StyleRainglet, Benoit, Paul Besognet, Cyril Benoit, Karim Delage, Véronique Bounor-Legaré, Charlène Forest, Philippe Cassagnau, and Yvan Chalamet. 2022. "TPV Foaming by CO2 Extrusion: Processing and Modelling" Polymers 14, no. 21: 4513. https://doi.org/10.3390/polym14214513
APA StyleRainglet, B., Besognet, P., Benoit, C., Delage, K., Bounor-Legaré, V., Forest, C., Cassagnau, P., & Chalamet, Y. (2022). TPV Foaming by CO2 Extrusion: Processing and Modelling. Polymers, 14(21), 4513. https://doi.org/10.3390/polym14214513






