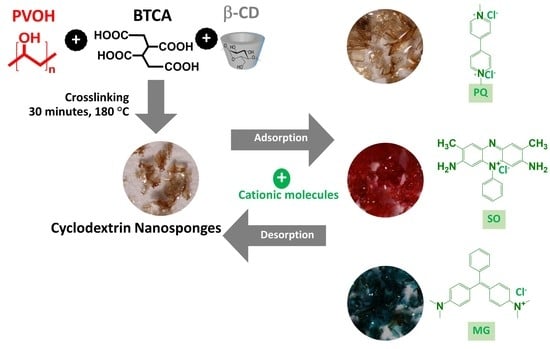
Adsorption of Cationic Contaminants by Cyclodextrin Nanosponges Cross-Linked with 1,2,3,4-Butanetetracarboxylic Acid and Poly(vinyl alcohol)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Nanosponges Preparation
2.3. Nanosponges Characterization
2.4. Adsorption Study
2.4.1. Preliminary Adsorption Study
2.4.2. Kinetics Study
2.4.3. Isotherm Study
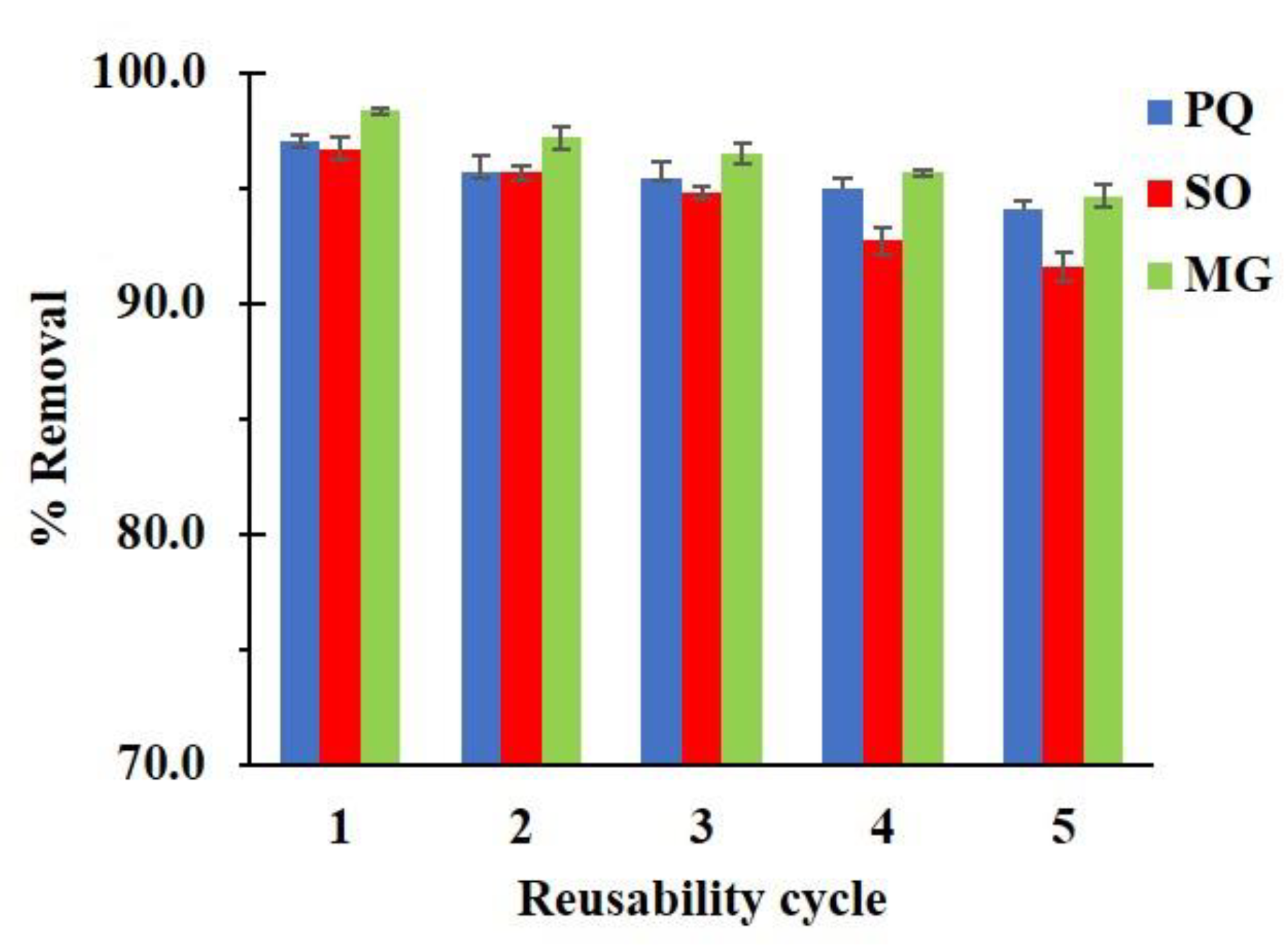
2.4.4. Reusability Study
3. Results and Discussion
3.1. Synthesis and Characterization of Nanosponges
3.1.1. Physicochemical Properties of Nanosponges
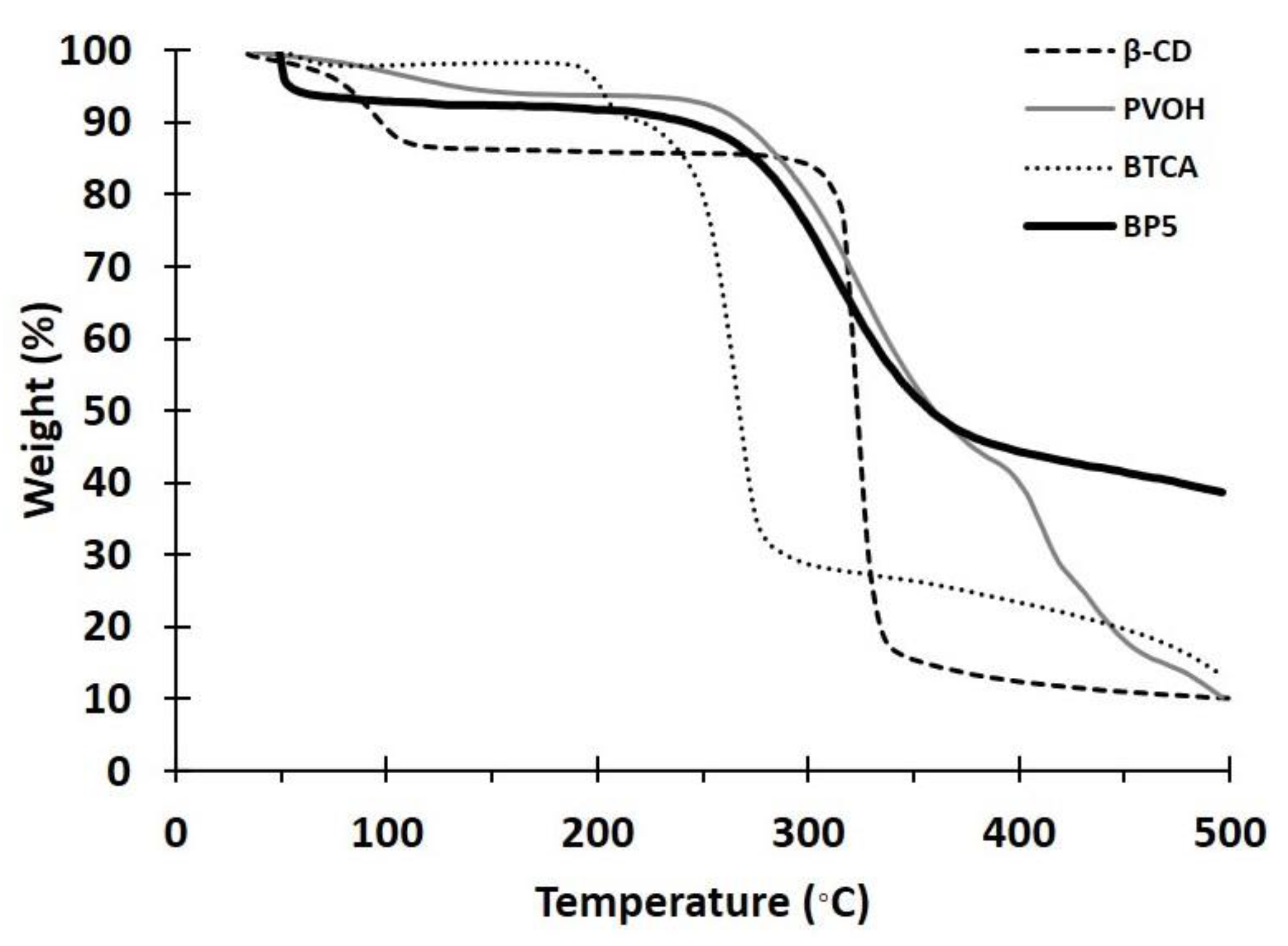
3.1.2. TGA Analysis
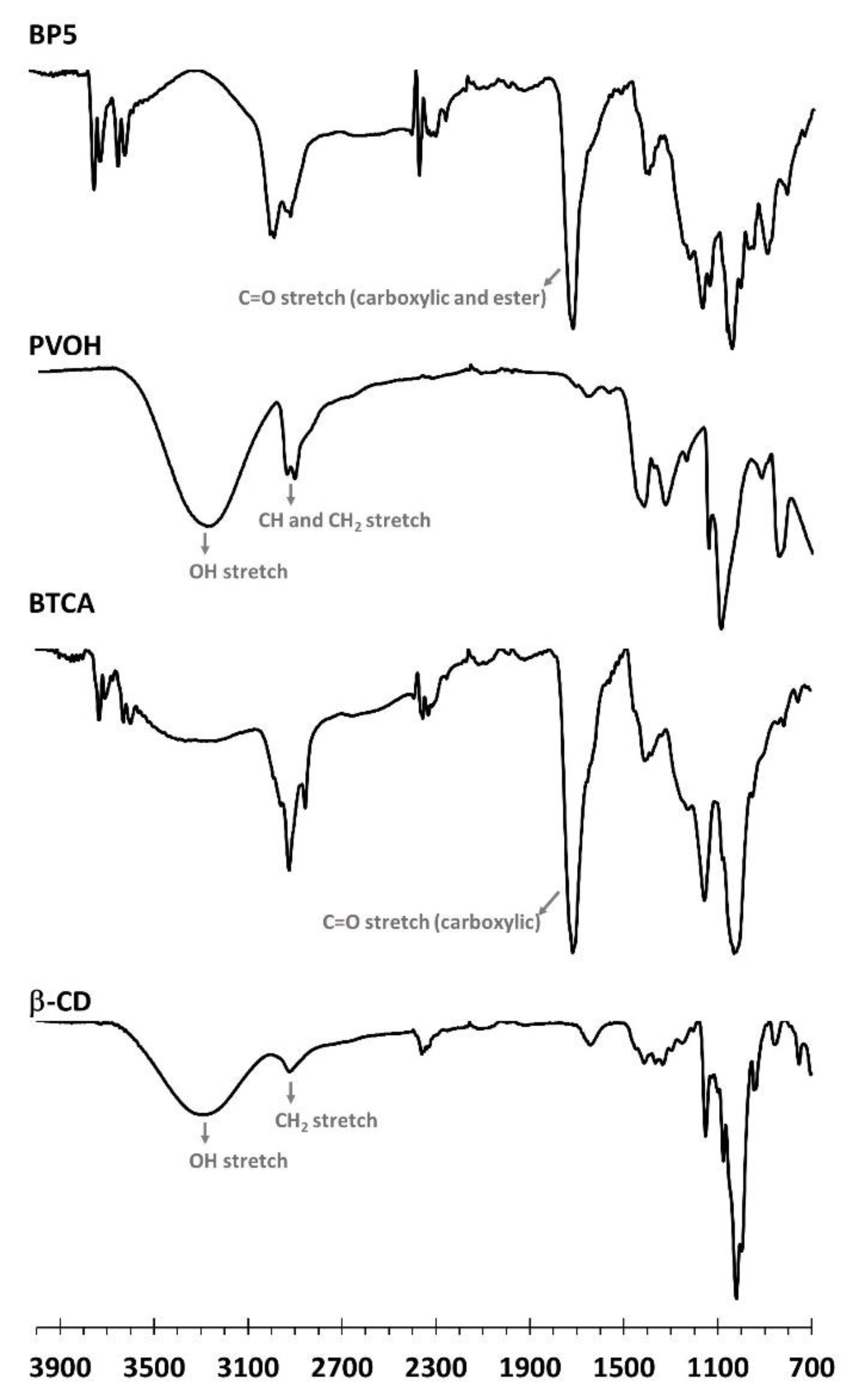
3.1.3. ATR-FTIR Exploration
3.1.4. Raman Investigation
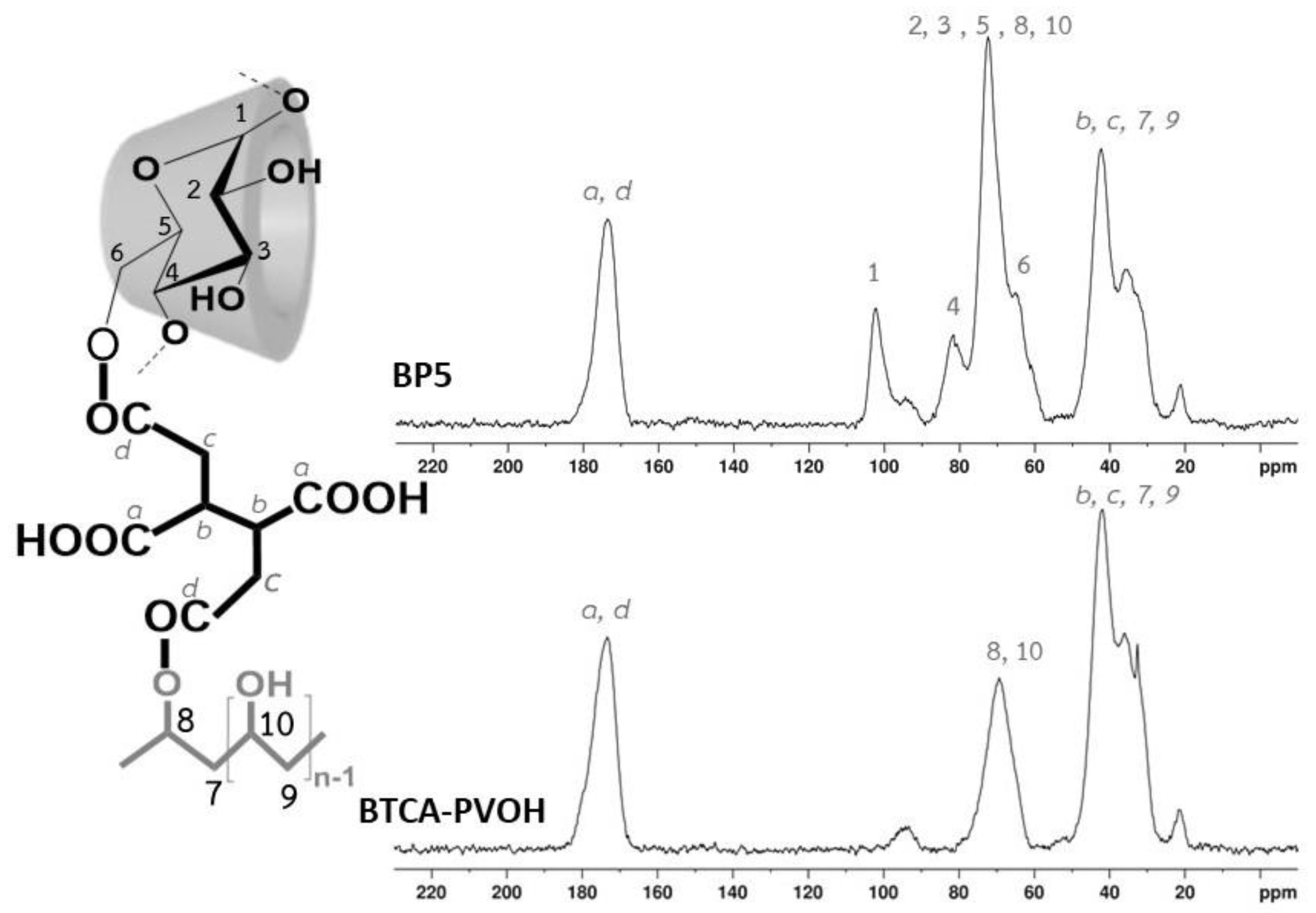
3.1.5. NMR Characterization
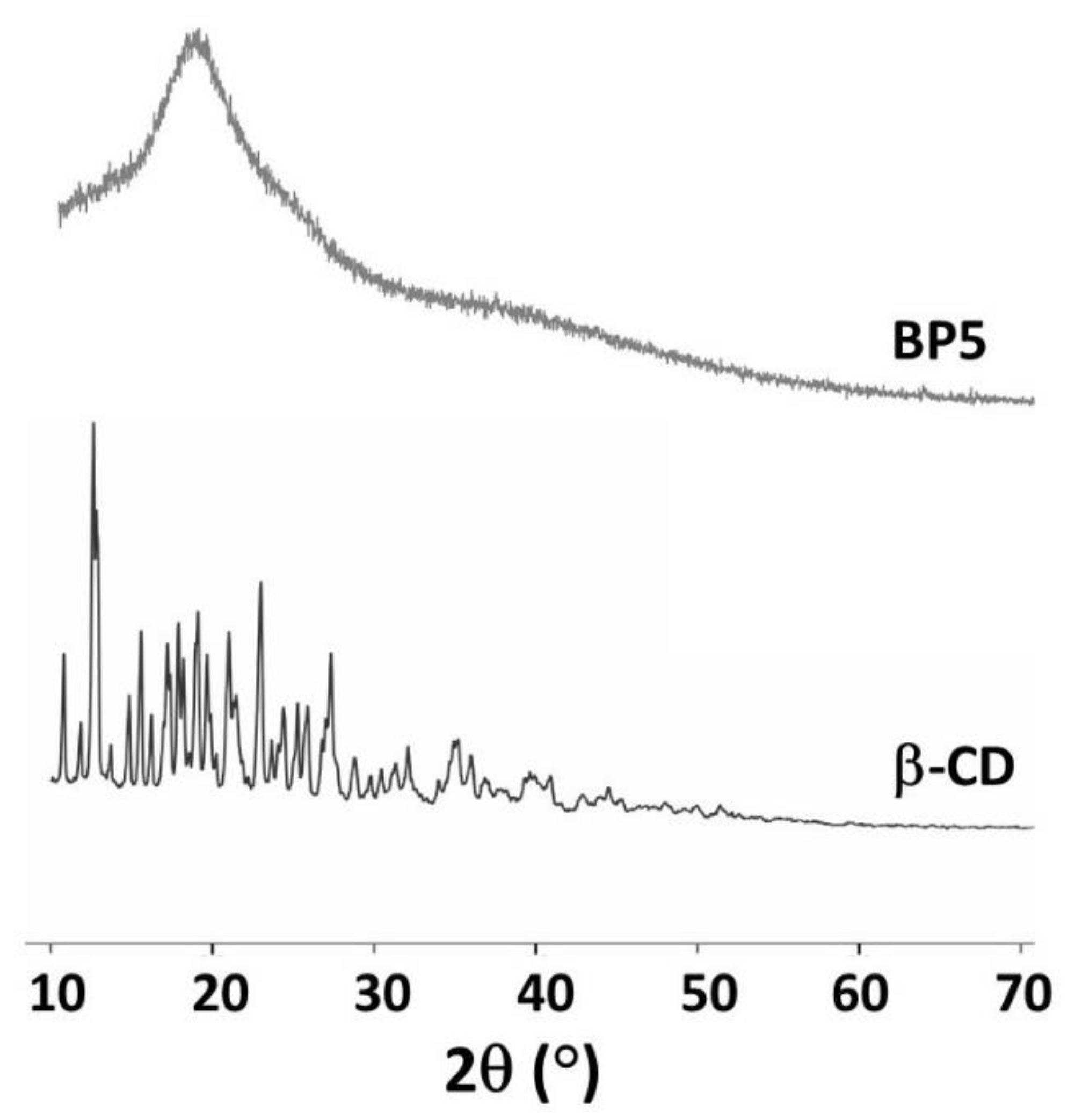
3.1.6. XRD Measurement
3.2. Adsorption Study
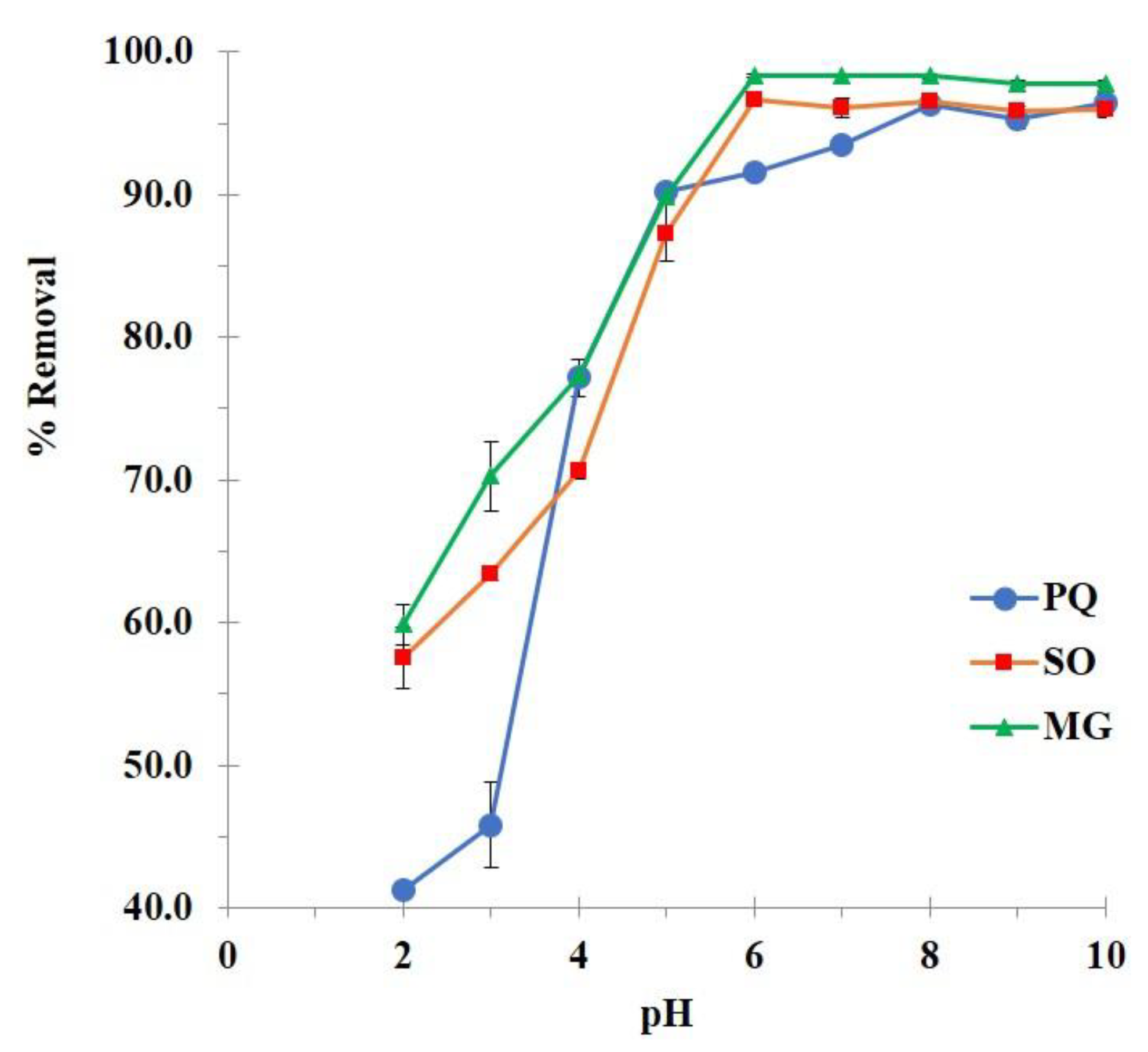
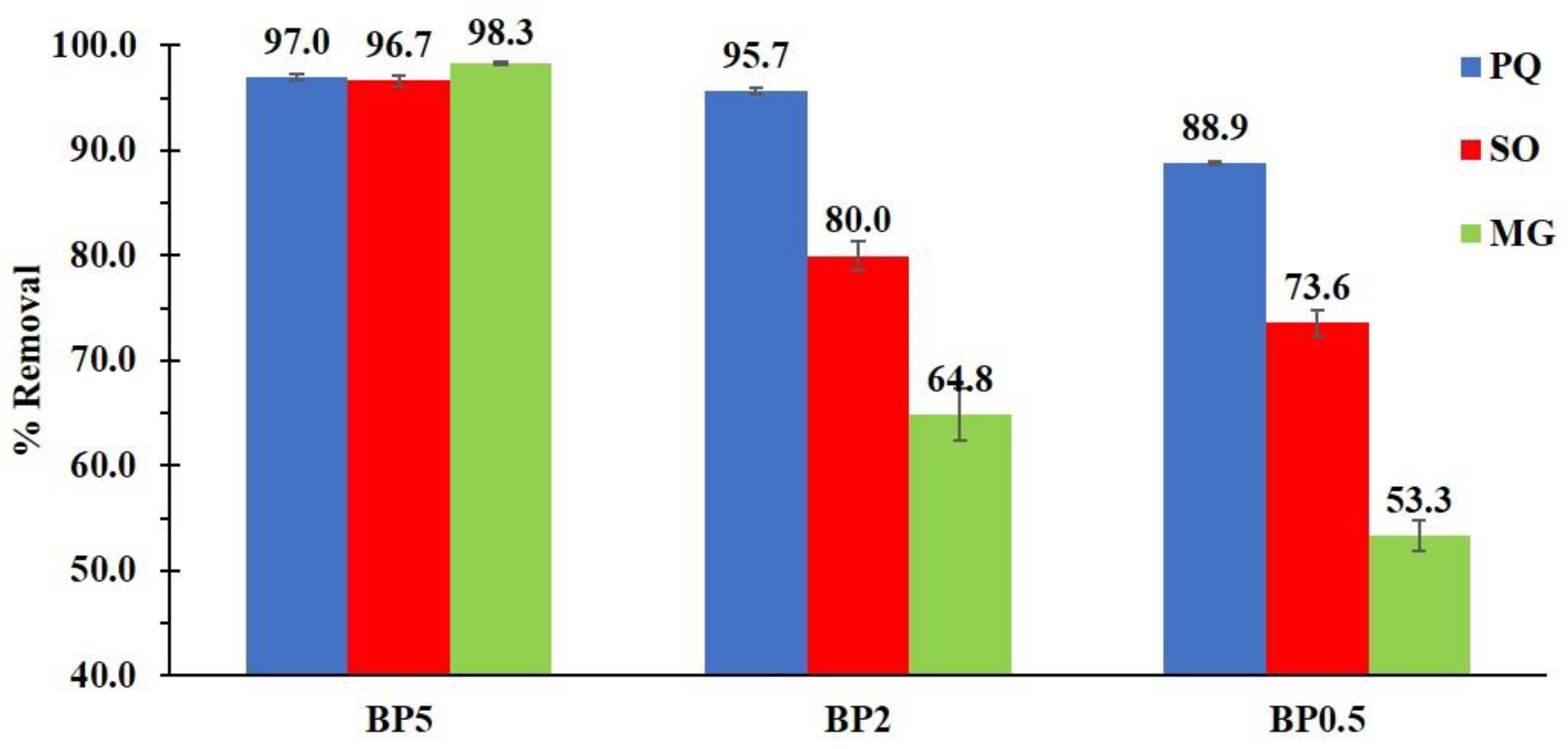
3.2.1. Preliminary Adsorption Study
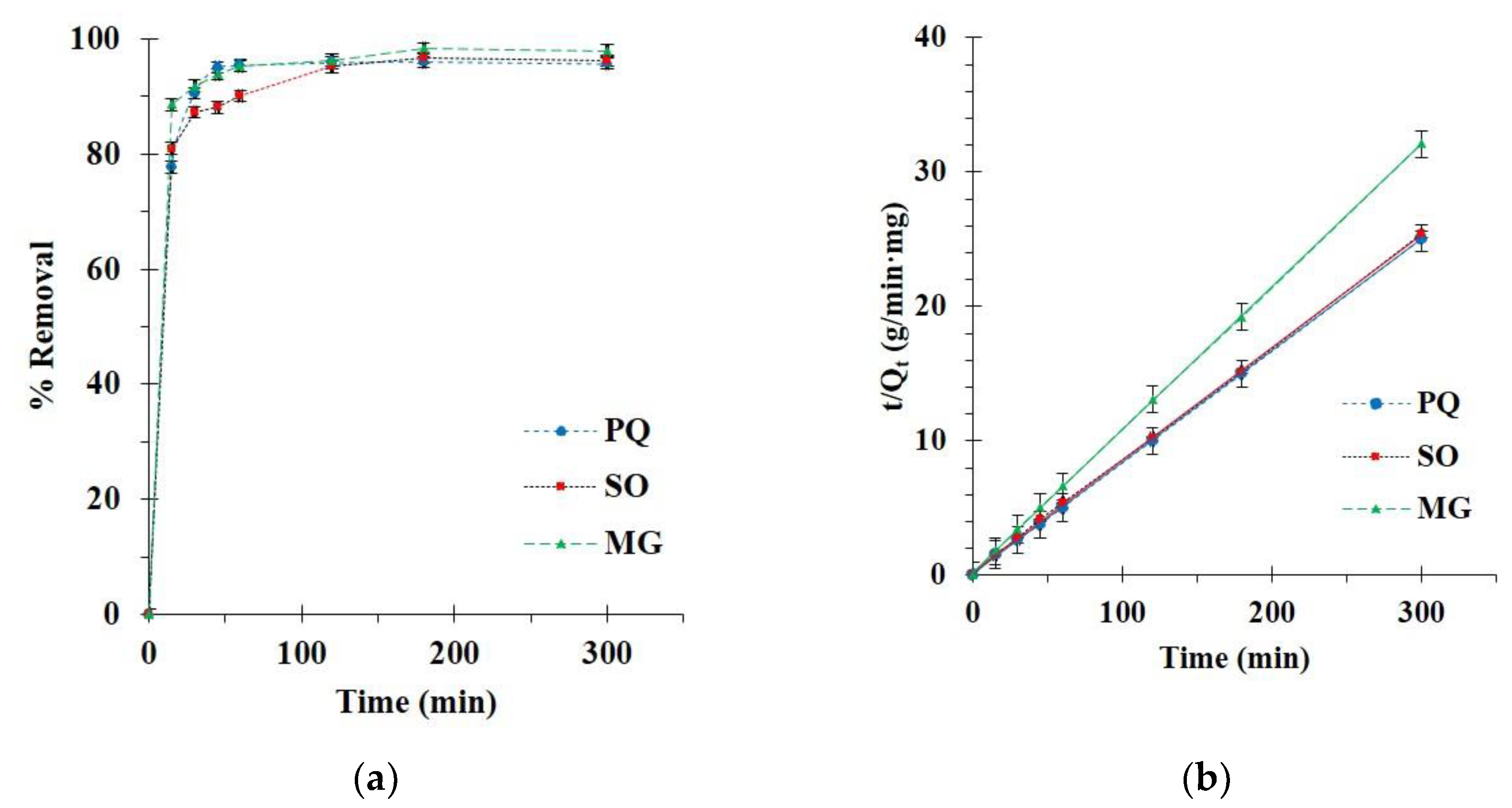
3.2.2. Kinetics Study
3.2.3. Isotherm Study
3.2.4. Reusability Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, Y.; Zhan, H.; Bhatt, P.; Chen, S. Paraquat Degradation from Contaminated Environments: Current Achievements and Perspectives. Front. Microbiol. 2019, 10, 1754. [Google Scholar] [CrossRef] [Green Version]
- Dennis, P.G.; Kukulies, T.; Forstner, C.; Orton, T.G.; Pattison, A.B. The Effects of Glyphosate, Glufosinate, Paraquat and Paraquat-Diquat on Soil Microbial Activity and Bacterial, Archaeal and Nematode Diversity. Sci. Rep. 2018, 8, 2119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shadnia, S.; Ebadollahi-Natanzi, A.; Ahmadzadeh, S.; Karami-Mohajeri, S.; Pourshojaei, Y.; Rahimi, H.R. Delayed Death Following Paraquat Poisoning: Three Case Reports and a Literature Review. Toxicol. Res. 2018, 7, 745–753. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Jiang, X.; Lu, G.; Zhou, J.; Kang, J.; Zhang, J. Identify the Early Predictor of Mortality in Patients with Acute Paraquat Poisoning. BioMed Res. Int. 2020, 2020, 8894180. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Kim, D.-S. Paraquat: Toxicology and Impacts of Its Ban on Human Health and Agriculture. Weed Sci. 2020, 68, 208–213. [Google Scholar] [CrossRef]
- Jindakaraked, M.; Khan, E.; Kajitvichyanukul, P. Biodegradation of Paraquat by Pseudomonas Putida and Bacillus Subtilis Immobilized on Ceramic with Supplemented Wastewater Sludge. Environ. Pollut. 2021, 286, 117307. [Google Scholar] [CrossRef]
- Thongpitak, J.; Pumas, P.; Pumas, C. Paraquat Degradation by Biological Manganese Oxide (BioMnOx) Catalyst Generated from Living Microalga Pediastrum Duplex AARL G060. Front. Microbiol. 2020, 11, 575361. [Google Scholar] [CrossRef]
- Burns, I.G.; Hayes, M.H.B.; Stacey, M. Studies of the Adsorption of Paraquat on Soluble Humic Fractions by Gel Filtration and Ultrafiltration Techniques. Pestic. Sci. 1973, 4, 629–641. [Google Scholar] [CrossRef]
- Humbert, H.; Gallard, H.; Suty, H.; Croué, J.-P. Natural Organic Matter (NOM) and Pesticides Removal Using a Combination of Ion Exchange Resin and Powdered Activated Carbon (PAC). Water Res. 2008, 42, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Ghavi, A.; Bagherian, G.; Rezaei-Vahidian, H. Degradation of Paraquat Herbicide Using Hybrid AOP Process: Statistical Optimization, Kinetic Study, and Estimation of Electrical Energy Consumption. Environ. Sci. Eur. 2021, 33, 117. [Google Scholar] [CrossRef]
- Tariq, S.R.; Chotana, G.A.; Rashid, A. Photocatalytic Degradation of Paraquat Dichloride in the Presence of ZnO.WO3 Composite. Int. J. Environ. Sci. Technol. 2021, 1–16. [Google Scholar] [CrossRef]
- Mueanpun, N.; Srisuk, N.; Chaiammart, N.; Panomsuwan, G. Nanoporous Activated Carbons Derived from Water Ferns as an Adsorbent for Removal of Paraquat from Contaminated Water. Materialia 2021, 15, 100986. [Google Scholar] [CrossRef]
- Rasaie, A.; Sabzehmeidani, M.M.; Ghaedi, M.; Ghane-Jahromi, M.; Sedaratian-Jahromi, A. Removal of Herbicide Paraquat from Aqueous Solutions by Bentonite Modified with Mesoporous Silica. Mater. Chem. Phys. 2021, 262, 124296. [Google Scholar] [CrossRef]
- Mehmandost, N.; García-Valverde, M.T.; Soriano, M.L.; Goudarzi, N.; Lucena, R.; Chamjangali, M.A.; Cardenas, S. Heracleum Persicum Based Biosorbent for the Removal of Paraquat and Diquat from Waters. J. Environ. Chem. Eng. 2020, 8, 104481. [Google Scholar] [CrossRef]
- Kamga, F.T. Modeling Adsorption Mechanism of Paraquat onto Ayous (Triplochiton Scleroxylon) Wood Sawdust. Appl. Water Sci. 2019, 9, 1. [Google Scholar] [CrossRef] [Green Version]
- Ha, N.T.H.; Toan, N.C.; Kajitvichyanukul, P. Enhanced Paraquat Removal from Contaminated Water Using Cell-Immobilized Biochar. Clean Technol. Environ. Policy 2021, 1–13. [Google Scholar] [CrossRef]
- Li, H.; Qi, H.; Yin, M.; Chen, Y.; Deng, Q.; Wang, S. Carbon Tubes from Biomass with Prominent Adsorption Performance for Paraquat. Chemosphere 2021, 262, 127797. [Google Scholar] [CrossRef] [PubMed]
- Shetty, D.; Boutros, S.; Skorjanc, T.; Garai, B.; Asfari, Z.; Raya, J.; Trabolsi, A. Fast and Efficient Removal of Paraquat in Water by Porous Polycalix[n]Arenes (n = 4, 6, and 8). J. Mater. Chem. A 2020, 8, 13942–13945. [Google Scholar] [CrossRef]
- Huang, C.-F.; Tu, C.-W.; Lee, R.-H.; Yang, C.-H.; Hung, W.-C.; Andrew Lin, K.-Y. Study of Various Diameter and Functionality of TEMPO-Oxidized Cellulose Nanofibers on Paraquat Adsorptions. Polym. Degrad. Stab. 2019, 161, 206–212. [Google Scholar] [CrossRef]
- Junthip, J. Water-Insoluble Cyclodextrin Polymer Crosslinked with Citric Acid for Paraquat Removal from Water. J. Macromol. Sci. Part A 2019, 56, 555–563. [Google Scholar] [CrossRef]
- Junthip, J.; Promma, W.; Sonsupap, S.; Boonyanusith, C. Adsorption of Paraquat from Water by Insoluble Cyclodextrin Polymer Crosslinked with 1,2,3,4-Butanetetracarboxylic Acid. Iran. Polym. J. 2019, 28, 213–223. [Google Scholar] [CrossRef]
- Dehghani, Z.; Sedghi-Asl, M.; Ghaedi, M.; Sabzehmeidani, M.M.; Adhami, E. Ultrasound-Assisted Adsorption of Paraquat Herbicide from Aqueous Solution by Graphene Oxide/ Mesoporous Silica. J. Environ. Chem. Eng. 2021, 9, 105043. [Google Scholar] [CrossRef]
- Kakhki, R.M.; Karimian, A.; Rad, M.S. Highly Efficient Removal of Paraquat Pesticide from Aqueous Solutions Using a Novel Nano Kaolin Modified with Sulfuric Acid via Host–Guest Interactions. J. Incl. Phenom. Macrocycl. Chem. 2020, 96, 307–313. [Google Scholar] [CrossRef]
- Fernandes, T.; Soares, S.F.; Trindade, T.; Daniel-da-Silva, A.L. Magnetic Hybrid Nanosorbents for the Uptake of Paraquat from Water. Nanomaterials 2017, 7, 68. [Google Scholar] [CrossRef]
- Pan, S.; Zhang, J.; He, Q.; Chen, X.; Jin, M. Fabrication of Benzenesulfonic Acid Groups Modified Magnetic Microspheres as an MSPE Adsorbent for Fast Determination of Paraquat and Diquat in Human Urine Combined with UPLC-HRMS. J. Chromatogr. B 2020, 1136, 121880. [Google Scholar] [CrossRef]
- Brenes-Guillén, L.; Fuentes-Schweizer, P.; García-Piñeres, A.; Uribe-Lorío, L. Tolerance and Sorption of Bromacil and Paraquat by Thermophilic Cyanobacteria Leptolyngbya 7M from Costa Rican Thermal Springs. J. Contam. Hydrol. 2019, 226, 103539. [Google Scholar] [CrossRef]
- Wang, M.; Orr, A.A.; He, S.; Dalaijamts, C.; Chiu, W.A.; Tamamis, P.; Phillips, T.D. Montmorillonites Can Tightly Bind Glyphosate and Paraquat Reducing Toxin Exposures and Toxicity. ACS Omega 2019, 4, 17702–17713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.-H.; Zhang, Y.-F.; Sun, X.-W.; Li, Y.-J.; Zhang, Y.-M.; Wei, T.-B.; Yao, H.; Lin, Q. Linear Tri-Pillar[5]Arene-Based Acceptor for Efficiently Separate Paraquat from Water through Collaboration Effect. Mater. Sci. Eng. C 2021, 118, 111358. [Google Scholar] [CrossRef]
- Jodeh, S.; Hanbali, G.; Tighadouini, S.; Radi, S.; Hamed, O.; Jodeh, D. Removal and Extraction Efficiency of Quaternary Ammonium Herbicides Paraquat (PQ) from Aqueous Solution by Ketoenol–Pyrazole Receptor Functionalized Silica Hybrid Adsorbent (SiNPz). BMC Chem. 2019, 13, 86. [Google Scholar] [CrossRef]
- Brigante, M.; Schulz, P.C. Adsorption of Paraquat on Mesoporous Silica Modified with Titania: Effects of PH, Ionic Strength and Temperature. J. Colloid Interface Sci. 2011, 363, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Sim, S.J.; Kang, J.H.; Choi, S.S. Isotherm and Thermodynamic Modelling of Malachite Green on CO2-Activated Carbon Fibers. Chem. Phys. Lett. 2021, 780, 138962. [Google Scholar] [CrossRef]
- Matpang, P.; Sriuttha, M.; Piwpuan, N. Effects of Malachite Green on Growth and Tissue Accumulation in Pak Choy (Brassica Chinensis Tsen & Lee). Agric. Nat. Resour. 2017, 51, 96–102. [Google Scholar] [CrossRef]
- Sinha, R.; Jindal, R. Elucidation of Malachite Green Induced Behavioural, Biochemical, and Histo-Architectural Defects in Cyprinus Carpio, as Piscine Model. Environ. Sustain. Indic. 2020, 8, 100055. [Google Scholar] [CrossRef]
- Pipoyan, D.; Stepanyan, S.; Beglaryan, M.; Stepanyan, S.; Mantovani, A. Health Risk Assessment of Toxicologically Relevant Residues in Emerging Countries: A Pilot Study on Malachite Green Residues in Farmed Freshwater Fish of Armenia. Food Chem. Toxicol. 2020, 143, 111526. [Google Scholar] [CrossRef]
- Mawad, A.M.M.; Albasri, H.; Temerk, H.A. Biosorption of Malachite Green by Dry Cells of Isolated Free Living Nitrogen Fixing Bacteria. Nat. Environ. Pollut. Technol. 2021, 20. [Google Scholar] [CrossRef]
- Panahi, M.; Behnam, S. Biosorption of Malachite Green Dye by the Brown Alga Dictyota Cervicornis: Kinetics and Isotherm Study. Color. Technol. 2018, 134, 292–298. [Google Scholar] [CrossRef]
- Song, J.; Han, G.; Wang, Y.; Jiang, X.; Zhao, D.; Li, M.; Yang, Z.; Ma, Q.; Parales, R.E.; Ruan, Z.; et al. Pathway and Kinetics of Malachite Green Biodegradation by Pseudomonas Veronii. Sci. Rep. 2020, 10, 4502. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.; Shah, L.A.; Sayed, M.; Siddiq, M.; Amin, N.U. Micellar Supported Ultrafiltration of Malachite Green: Experimental Verification of Theoretical Approach. Z. Für Phys. Chem. 2019, 233, 289–301. [Google Scholar] [CrossRef]
- Ashraf, M.W.; Abulibdeh, N.; Salam, A. Selective Removal of Malachite Green Dye from Aqueous Solutions by Supported Liquid Membrane Technology. Int. J. Environ. Res. Public Health 2019, 16, 3484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elkady, M.F.; Hassan, H.S. Photocatalytic Degradation of Malachite Green Dye from Aqueous Solution Using Environmentally Compatible Ag/ZnO Polymeric Nanofibers. Polymers 2021, 13, 2033. [Google Scholar] [CrossRef] [PubMed]
- Lavand, A.B.; Bhatu, M.N.; Malghe, Y.S. Visible Light Photocatalytic Degradation of Malachite Green Using Modified Titania. J. Mater. Res. Technol. 2019, 8, 299–308. [Google Scholar] [CrossRef]
- Raval, N.P.; Shah, P.U.; Shah, N.K. Malachite Green “a Cationic Dye” and Its Removal from Aqueous Solution by Adsorption. Appl. Water Sci. 2017, 7, 3407–3445. [Google Scholar] [CrossRef] [Green Version]
- Tewari, K.; Singhal, G.; Arya, R.K. Adsorption Removal of Malachite Green Dye from Aqueous Solution. Rev. Chem. Eng. 2018, 34, 427–453. [Google Scholar] [CrossRef]
- Igwegbe, C.A.; Ighalo, J.O.; Onyechi, K.K.; Onukwuli, O.D. Adsorption of Congo Red and Malachite Green Using H3PO4 and NaCl-Modified Activated Carbon from Rubber (Hevea Brasiliensis) Seed Shells. Sustain. Water Resour. Manag. 2021, 7, 63. [Google Scholar] [CrossRef]
- Bulut, E.; Özacar, M.; Şengil, İ.A. Adsorption of Malachite Green onto Bentonite: Equilibrium and Kinetic Studies and Process Design. Microporous Mesoporous Mater. 2008, 115, 234–246. [Google Scholar] [CrossRef]
- Wan Ngah, W.S.; Ariff, N.F.M.; Hashim, A.; Hanafiah, M.A.K.M. Malachite Green Adsorption onto Chitosan Coated Bentonite Beads: Isotherms, Kinetics and Mechanism. Clean Soil Air Water 2010, 38, 394–400. [Google Scholar] [CrossRef]
- Deng, H.; Li, Y.F.; Tao, S.Q.; Li, A.Y.; Li, Q.Y.; Hu, L.N. Efficient Adsorption Capability of Banana and Cassava Biochar for Malachite Green: Removal Process and Mechanism Exploration. Environ. Eng. Res. 2021, 27, 200575. [Google Scholar] [CrossRef]
- Khan, T.A.; Rahman, R.; Ali, I.; Khan, E.A.; Mukhlif, A.A. Removal of Malachite Green from Aqueous Solution Using Waste Pea Shells as Low-Cost Adsorbent–Adsorption Isotherms and Dynamics. Toxicol. Environ. Chem. 2014, 96, 569–578. [Google Scholar] [CrossRef]
- Ahmad, R.; Kumar, R. Adsorption Studies of Hazardous Malachite Green onto Treated Ginger Waste. J. Environ. Manag. 2010, 91, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Rajabi, M.; Mirza, B.; Mahanpoor, K.; Mirjalili, M.; Najafi, F.; Moradi, O.; Sadegh, H.; Shahryari-Ghoshekandi, R.; Asif, M.; Tyagi, I.; et al. Adsorption of Malachite Green from Aqueous Solution by Carboxylate Group Functionalized Multi-Walled Carbon Nanotubes: Determination of Equilibrium and Kinetics Parameters. J. Ind. Eng. Chem. 2016, 34, 130–138. [Google Scholar] [CrossRef]
- Chinthalapudi, N.; Kommaraju, V.V.D.; Kannan, M.K.; Nalluri, C.B.; Varanasi, S. Composites of Cellulose Nanofibers and Silver Nanoparticles for Malachite Green Dye Removal from Water. Carbohydr. Polym. Technol. Appl. 2021, 2, 100098. [Google Scholar] [CrossRef]
- Sekine, Y.; Nankawa, T.; Yunoki, S.; Sugita, T.; Nakagawa, H.; Yamada, T. Eco-Friendly Carboxymethyl Cellulose Nanofiber Hydrogels Prepared via Freeze Cross-Linking and Their Applications. ACS Appl. Polym. Mater. 2020, 2, 5482–5491. [Google Scholar] [CrossRef]
- Sadiq, A.C.; Rahim, N.Y.; Suah, F.B.M. Adsorption and Desorption of Malachite Green by Using Chitosan-Deep Eutectic Solvents Beads. Int. J. Biol. Macromol. 2020, 164, 3965–3973. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Ur Rahman, A.; Ullah, F.; Rashid, A.; Arshad, T.; Viglašová, E.; Galamboš, M.; Mahmoodi, N.M.; Ullah, H. Adsorption of Malachite Green Dye onto Mesoporous Natural Inorganic Clays: Their Equilibrium Isotherm and Kinetics Studies. Water 2021, 13, 965. [Google Scholar] [CrossRef]
- Crini, G.; Peindy, H.; Gimbert, F.; Robert, C. Removal of C.I. Basic Green 4 (Malachite Green) from Aqueous Solutions by Adsorption Using Cyclodextrin-Based Adsorbent: Kinetic and Equilibrium Studies. Sep. Purif. Technol. 2007, 53, 97–110. [Google Scholar] [CrossRef]
- Wang, D.; Liu, L.; Jiang, X.; Yu, J.; Chen, X. Adsorption and Removal of Malachite Green from Aqueous Solution Using Magnetic β-Cyclodextrin-Graphene Oxide Nanocomposites as Adsorbents. Colloids Surf. A Physicochem. Eng. Asp. 2015, 466, 166–173. [Google Scholar] [CrossRef]
- Chilakapati, R.B.; Hemanth Kumar, S.; Satyanarayana, S.V.; Behara, D.K. Adsorptive Removal of Methylene Blue (MB) and Malachite Green (MG) Dyes from Aqueous Solutions Using Graphene Oxide (GO). Z. Für Phys. Chem. 2021, 235, 1645–1660. [Google Scholar] [CrossRef]
- Suwandi, A.C.; Indraswati, N.; Ismadji, S. Adsorption of N-Methylated Diaminotriphenilmethane Dye (Malachite Green) on Natural Rarasaponin Modified Kaolin. Desalination Water Treat. 2012, 41, 342–355. [Google Scholar] [CrossRef] [Green Version]
- Yılmaz, Ş.; Zengin, A.; Şahan, T. A Novel Material Poly(N-Acryloyl-L-Serine)-Brush Grafted Kaolin for Efficient Elimination of Malachite Green Dye from Aqueous Environments. Colloids Surf. A Physicochem. Eng. Asp. 2020, 601, 125041. [Google Scholar] [CrossRef]
- Eltaweil, A.S.; Mohamed, H.A.; El-Monaem, E.M.A.; El-Subruiti, G.M. Mesoporous Magnetic Biochar Composite for Enhanced Adsorption of Malachite Green Dye: Characterization, Adsorption Kinetics, Thermodynamics and Isotherms. Adv. Powder Technol. 2020, 31, 1253–1263. [Google Scholar] [CrossRef]
- Gul, K.; Khan, H.; Muhammad, N.; Ara, B.; Zia, T.U.H. Removal of Toxic Malachite Green Dye from Aqueous Environment Using Reduced Magnetic Graphene Oxide as an Efficient and Reusable Adsorbent. Sep. Sci. Technol. 2021, 56, 2507–2520. [Google Scholar] [CrossRef]
- Delpiano, G.R.; Tocco, D.; Medda, L.; Magner, E.; Salis, A. Adsorption of Malachite Green and Alizarin Red S Dyes Using Fe-BTC Metal Organic Framework as Adsorbent. Int. J. Mol. Sci. 2021, 22, 788. [Google Scholar] [CrossRef]
- Li, J.; Tao, J.; Ma, C.; Yang, J.; Gu, T.; Liu, J. Carboxylated Cellulose Nanofiber/Montmorillonite Nanocomposite for the Removal of Levofloxacin Hydrochloride Antibiotic from Aqueous Solutions. RSC Adv. 2020, 10, 42038–42053. [Google Scholar] [CrossRef]
- Fil, B.A. Isotherm, Kinetic, and Thermodynamic Studies on the Adsorption Behavior of Malachite Green Dye onto Montmorillonite Clay. Part. Sci. Technol. 2016, 34, 118–126. [Google Scholar] [CrossRef]
- Samiey, B.; Toosi, A.R. Adsorption of Malachite Green on Silica Gel: Effects of NaCl, PH and 2-Propanol. J. Hazard. Mater. 2010, 184, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.-Q.; Chen, Z.-Y.; Chen, J.-W.; Chen, Q.-H. Preparation of Mesoporous Silica Nanoparticles Modified by Urushiol and Their Adsorption on Malachite Green. J. Nanosci. Nanotechnol. 2021, 21, 4792–4796. [Google Scholar] [CrossRef] [PubMed]
- Mansa, R.F.; Sipaut, C.S.; Rahman, I.A.; Yusof, N.S.M.; Jafarzadeh, M. Preparation of Glycine–Modified Silica Nanoparticles for the Adsorption of Malachite Green Dye. J. Porous Mater. 2016, 23, 35–46. [Google Scholar] [CrossRef]
- Gao, L.; Yuan, H.; Xu, E.; Liu, J. Toxicology of Paraquat and Pharmacology of the Protective Effect of 5-Hydroxy-1-Methylhydantoin on Lung Injury Caused by Paraquat Based on Metabolomics. Sci. Rep. 2020, 10, 1790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, X.; Zhou, Z.; Wu, S.; Wang, D.; Zheng, S.; Wang, G. Adsorption Removal of Multiple Dyes Using Biogenic Selenium Nanoparticles from an Escherichia Coli Strain Overexpressed Selenite Reductase CsrF. Nanomaterials 2018, 8, 234. [Google Scholar] [CrossRef] [Green Version]
- Ogugbue, C.J.; Sawidis, T.; Oranusi, N.A. Bioremoval of Chemically Different Synthetic Dyes by Aeromonas Hydrophila in Simulated Wastewater Containing Dyeing Auxiliaries. Ann. Microbiol. 2012, 62, 1141–1153. [Google Scholar] [CrossRef]
- Acemioğlu, B.; Bilir, M.H.; Alma, M.H. Adsorption of Safranin-O Dye by Peanut Shell-Based Polyurethane Type Foam. Int. J. Chem. Technol. 2018, 2, 95–104. [Google Scholar] [CrossRef]
- Alabduljabbar, F.A.; Haider, S.; Alghyamah, A.A.; Haider, A.; Khan, R.; Almasry, W.A.; Patel, R.; Mujtaba, I.M.; Ali, F.A.A. Ethanol Amine Functionalized Electrospun Nanofibers Membrane for the Treatment of Dyes Polluted Wastewater. Appl. Nanosci. 2021, 1–14. [Google Scholar] [CrossRef]
- Mohamed, F.; Abukhadra, M.R.; Shaban, M. Removal of Safranin Dye from Water Using Polypyrrole Nanofiber/Zn-Fe Layered Double Hydroxide Nanocomposite (Ppy NF/Zn-Fe LDH) of Enhanced Adsorption and Photocatalytic Properties. Sci. Total Environ. 2018, 640–641, 352–363. [Google Scholar] [CrossRef]
- El-Kemary, M.; Abdel-Moneam, Y.; Madkour, M.; El-Mehasseb, I. Enhanced Photocatalytic Degradation of Safranin-O by Heterogeneous Nanoparticles for Environmental Applications. J. Lumin. 2011, 131, 570–576. [Google Scholar] [CrossRef]
- Sharma, K.; Vyas, R.K.; Singh, K.; Dalai, A.K. Reactive Adsorption of Safranin O: Surface–Pore Diffusion Modeling and Degradation Study. Water Sci. Technol. 2019, 80, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Al-Shahrani, S. Removal of Safranin Dye from Wastewater Using Khulays Natural Bentonite. J. King Abdulaziz Univ.-Eng. Sci. 2018, 29. [Google Scholar] [CrossRef]
- Suleman, M.; Zafar, M.; Ahmed, A.; Rashid, M.U.; Hussain, S.; Razzaq, A.; Mohidem, N.A.; Fazal, T.; Haider, B.; Park, Y.-K. Castor Leaves-Based Biochar for Adsorption of Safranin from Textile Wastewater. Sustainability 2021, 13, 6926. [Google Scholar] [CrossRef]
- Moawed, E.A.; Abulkibash, A.B. Selective Separation of Light Green and Safranin O from Aqueous Solution Using Salvadora Persica (Miswak) Powder as a New Biosorbent. J. Saudi Chem. Soc. 2016, 20, S178–S185. [Google Scholar] [CrossRef] [Green Version]
- Bayazit, Ş.S. Investigation of Safranin O Adsorption on Superparamagnetic Iron Oxide Nanoparticles (SPION) and Multi-Wall Carbon Nanotube/SPION Composites. Desalination Water Treat. 2014, 52, 6966–6975. [Google Scholar] [CrossRef]
- Abukhadra, M.R.; Adlii, A.; El-Sherbeeny, A.M.; Soliman, A.T.A.; Elgawad, A.E.E.A. Promoting the Decontamination of Different Types of Water Pollutants (Cd2+, Safranin Dye, and Phosphate) Using a Novel Structure of Exfoliated Bentonite Admixed with Cellulose Nanofiber. J. Environ. Manag. 2020, 273, 111130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Repo, E.; Yin, D.; Meng, Y.; Jafari, S.; Sillanpää, M. EDTA-Cross-Linked β-Cyclodextrin: An Environmentally Friendly Bifunctional Adsorbent for Simultaneous Adsorption of Metals and Cationic Dyes. Environ. Sci. Technol. 2015, 49, 10570–10580. [Google Scholar] [CrossRef] [PubMed]
- Debnath, S.; Ballav, N.; Maity, A.; Pillay, K. Competitive Adsorption of Ternary Dye Mixture Using Pine Cone Powder Modified with β-Cyclodextrin. J. Mol. Liq. 2017, 225, 679–688. [Google Scholar] [CrossRef]
- Vidovix, T.B.; Quesada, H.B.; Bergamasco, R.; Vieira, M.F.; Vieira, A.M.S. Adsorption of Safranin-O Dye by Copper Oxide Nanoparticles Synthesized from Punica granatum Leaf Extract. Environ. Technol. 2021, 1–17. [Google Scholar] [CrossRef]
- Debnath, S.; Parashar, K.; Pillay, K. Ultrasound Assisted Adsorptive Removal of Hazardous Dye Safranin O from Aqueous Solution Using Crosslinked Graphene Oxide-Chitosan (GO-CH) Composite and Optimization by Response Surface Methodology (RSM) Approach. Carbohydr. Polym. 2017, 175, 509–517. [Google Scholar] [CrossRef]
- Adebowale, K.O.; Olu-Owolabi, B.I.; Chigbundu, E.C. Removal of Safranin-O from Aqueous Solution by Adsorption onto Kaolinite Clay. J. Encapsul. Adsorpt. Sci. 2014, 04, 89–104. [Google Scholar] [CrossRef] [Green Version]
- Abukhadra, M.R.; Mostafa, M.; El-Sherbeeny, A.M.; El-Meligy, M.A.; Nadeem, A. Instantaneous Adsorption of Synthetic Dyes from an Aqueous Environment Using Kaolinite Nanotubes: Equilibrium and Thermodynamic Studies. ACS Omega 2021, 6, 845–856. [Google Scholar] [CrossRef] [PubMed]
- Shariati, S.; Faraji, M.; Yamini, Y.; Rajabi, A.A. Fe3O4 Magnetic Nanoparticles Modified with Sodium Dodecyl Sulfate for Removal of Safranin O Dye from Aqueous Solutions. Desalination 2011, 270, 160–165. [Google Scholar] [CrossRef]
- Qin, L.; Chen, H.-Z.; Lei, J.; Wang, Y.-Q.; Ye, T.-Q.; Zheng, H.-G. Photodegradation of Some Organic Dyes over Two Metal–Organic Frameworks with Especially High Efficiency for Safranine T. Cryst. Growth Des. 2017, 17, 1293–1298. [Google Scholar] [CrossRef]
- Van Damme, H.; Crespan, M.; Cruz, M.I.; Fripiat, J.J. Adsorption of safranine by Na+, Ni2+ and Fe3+ montmorillonites. Clays Clay Miner. 1977, 25, 19–25. [Google Scholar] [CrossRef]
- Kamel, M.M.; Alsohaimi, I.H.; Alhumaimess, M.S.; Hassan, H.M.A.; Alshammari, M.S.; El-Sayed, M.Y. A Glassy Polyvinyl Alcohol/Silica Gel Hybrid Composite for Safranin Removal: Adsorption, Kinetic and Thermodynamic Studies. Res. Chem. Intermed. 2021, 47, 925–944. [Google Scholar] [CrossRef]
- Abukhadra, M.R.; Shaban, M. Recycling of Different Solid Wastes in Synthesis of High-Order Mesoporous Silica as Adsorbent for Safranin Dye. Int. J. Environ. Sci. Technol. 2019, 16, 7573–7582. [Google Scholar] [CrossRef]
- Köse, K.; Tüysüz, M.; Aksüt, D.; Uzun, L. Modification of Cyclodextrin and Use in Environmental Applications. Environ. Sci. Pollut. Res. 2021, 29, 182–209. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Crini, G. Environmental Applications of Water-Insoluble β-Cyclodextrin–Epichlorohydrin Polymers. Prog. Polym. Sci. 2013, 38, 344–368. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Winterton, P.; Fourmentin, S.; Wilson, L.D.; Fenyvesi, É.; Crini, G. Water-Insoluble β-Cyclodextrin–Epichlorohydrin Polymers for Removal of Pollutants from Aqueous Solutions by Sorption Processes Using Batch Studies: A Review of Inclusion Mechanisms. Prog. Polym. Sci. 2018, 78, 1–23. [Google Scholar] [CrossRef]
- Krabicová, I.; Appleton, S.L.; Tannous, M.; Hoti, G.; Caldera, F.; Rubin Pedrazzo, A.; Cecone, C.; Cavalli, R.; Trotta, F. History of Cyclodextrin Nanosponges. Polymers 2020, 12, 1122. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Hua, S.; Tian, Y.; Liu, J. Cyclodextrin-Based Adsorbents for the Removal of Pollutants from Wastewater: A Review. Environ. Sci. Pollut. Res. 2021, 28, 1317–1340. [Google Scholar] [CrossRef] [PubMed]
- Cova, T.F.; Murtinho, D.; Aguado, R.; Pais, A.A.C.C.; Valente, A.J.M. Cyclodextrin Polymers and Cyclodextrin-Containing Polysaccharides for Water Remediation. Polysaccharides 2021, 2, 2. [Google Scholar] [CrossRef]
- Zhao, B.; Jiang, L.; Jia, Q. Advances in Cyclodextrin Polymers Adsorbents for Separation and Enrichment: Classification, Mechanism and Applications. Chin. Chem. Lett. 2021, S1001841721004319. [Google Scholar] [CrossRef]
- Rizzi, V.; Gubitosa, J.; Signorile, R.; Fini, P.; Cecone, C.; Matencio, A.; Trotta, F.; Cosma, P. Cyclodextrin Nanosponges as Adsorbent Material to Remove Hazardous Pollutants from Water: The Case of Ciprofloxacin. Chem. Eng. J. 2021, 411, 128514. [Google Scholar] [CrossRef]
- Pedrazzo, A.R.; Smarra, A.; Caldera, F.; Musso, G.; Dhakar, N.K.; Cecone, C.; Hamedi, A.; Corsi, I.; Trotta, F.; Pedrazzo, R. Eco-Friendly β-Cyclodextrin and Linecaps Polymers for the Removal of Heavy Metals. Polymers 2019, 11, 1658. [Google Scholar] [CrossRef] [Green Version]
- Massaro, M.; Colletti, C.G.; Lazzara, G.; Guernelli, S.; Noto, R.; Riela, S. Synthesis and Characterization of Halloysite–Cyclodextrin Nanosponges for Enhanced Dyes Adsorption. ACS Sustain. Chem. Eng. 2017, 5, 3346–3352. [Google Scholar] [CrossRef] [Green Version]
- Varan, C.; Anceschi, A.; Sevli, S.; Bruni, N.; Giraudo, L.; Bilgiç, E.; Korkusuz, P.; İskit, A.B.; Trotta, F.; Bilensoy, E. Preparation and Characterization of Cyclodextrin Nanosponges for Organic Toxic Molecule Removal. Int. J. Pharm. 2020, 585, 119485. [Google Scholar] [CrossRef]
- Liao, X.; Wang, B.; Zhang, Q. Synthesis of Glycopolymer Nanosponges with Enhanced Adsorption Performances for Boron Removal and Water Treatment. J. Mater. Chem. A 2018, 6, 21193–21206. [Google Scholar] [CrossRef]
- Salazar, S.; Yutronic, N.; Jara, P. Magnetic β-Cyclodextrin Nanosponges for Potential Application in the Removal of the Neonicotinoid Dinotefuran from Wastewater. Int. J. Mol. Sci. 2020, 21, 4079. [Google Scholar] [CrossRef] [PubMed]
- Kumari, P.; Singh, P.; Singhal, A. Cyclodextrin-Based Nanostructured Materials for Sustainable Water Remediation Applications. Environ. Sci. Pollut. Res. 2020, 27, 32432–32448. [Google Scholar] [CrossRef]
- Huang, W.; Hu, Y.; Li, Y.; Zhou, Y.; Niu, D.; Lei, Z.; Zhang, Z. Citric Acid-Crosslinked β-Cyclodextrin for Simultaneous Removal of Bisphenol A, Methylene Blue and Copper: The Roles of Cavity and Surface Functional Groups. J. Taiwan Inst. Chem. Eng. 2018, 82, 189–197. [Google Scholar] [CrossRef]
- Moulahcene, L.; Skiba, M.; Senhadji, O.; Milon, N.; Benamor, M.; Lahiani-Skiba, M. Inclusion and Removal of Pharmaceutical Residues from Aqueous Solution Using Water-Insoluble Cyclodextrin Polymers. Chem. Eng. Res. Des. 2015, 97, 145–158. [Google Scholar] [CrossRef]
- Zhao, D.; Zhao, L.; Zhu, C.-S.; Shen, X.; Zhang, X.; Sha, B. Comparative Study of Polymer Containing β-Cyclodextrin and –COOH for Adsorption toward Aniline, 1-Naphthylamine and Methylene Blue. J. Hazard. Mater. 2009, 171, 241–246. [Google Scholar] [CrossRef]
- Li, W.; Liu, H.; Li, L.; Liu, K.; Liu, J.; Tang, T.; Jiang, W. Green Synthesis of Citric Acid-Crosslinked β-Cyclodextrin for Highly Efficient Removal of Uranium(VI) from Aqueous Solution. J. Radioanal. Nucl. Chem. 2019, 322, 2033–2042. [Google Scholar] [CrossRef]
- Pirouzmand, M.; Sani, P.S.; Ghasemi, Z.; Azizi, S. Citric Acid-Crosslinked β-Cyclodextrin Supported Zinc Peroxide as a Biocompatible H2O2 Scavenger. J. Biol. Inorg. Chem. 2020, 25, 411–417. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Y.X.; Wang, P.L.; Zhang, Y.; Cheng, B.W.; Sun, Q.M.; Li, F. Synthesis of β-Cyclodextrin Immobilized Starch and Its Application for the Removal of Dyestuff from Waste-Water. J. Polym. Environ. 2019, 27, 929–941. [Google Scholar] [CrossRef]
- Zhao, J.; Zou, Z.; Ren, R.; Sui, X.; Mao, Z.; Xu, H.; Zhong, Y.; Zhang, L.; Wang, B. Chitosan Adsorbent Reinforced with Citric Acid Modified β-Cyclodextrin for Highly Efficient Removal of Dyes from Reactive Dyeing Effluents. Eur. Polym. J. 2018, 108, 212–218. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, Y.; Wang, J.; Lu, J.; Zhou, Y. Polydopamine Modified Cyclodextrin Polymer as Efficient Adsorbent for Removing Cationic Dyes and Cu2+. J. Hazard. Mater. 2020, 389, 121897. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhang, Y.; Zhao, B.; Jia, Q. Supramolecular Adsorbents in Extraction and Separation Techniques-A Review. Anal. Chim. Acta 2020, 1122, 97–113. [Google Scholar] [CrossRef]
- Trotta, F. Cyclodextrin Nanosponges and Their Applications. In Cyclodextrins in Pharmaceutics, Cosmetics, and Biomedicine; Bilensoy, E., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 323–342. ISBN 978-0-470-92681-9. [Google Scholar]
- Trotta, F.; Zanetti, M.; Cavalli, R. Cyclodextrin-Based Nanosponges as Drug Carriers. Beilstein J. Org. Chem. 2012, 8, 2091–2099. [Google Scholar] [CrossRef] [PubMed]
- Kardooni, R.; Kiasat, A.R.; Eskandari Sabzi, N. Hyper-Cross-Linked β-Cyclodextrin Nanosponge: A Three-Dimensional, Porous and Biodegradable Catalyst in the One-Pot Synthesis of Kojic Acid-Based Heterocyclic Compounds. Res. Chem. Intermed. 2020, 46, 1857–1868. [Google Scholar] [CrossRef]
- Haimhoffer, Á.; Rusznyák, Á.; Réti-Nagy, K.; Vasvári, G.; Váradi, J.; Vecsernyés, M.; Bácskay, I.; Fehér, P.; Ujhelyi, Z.; Fenyvesi, F. Cyclodextrins in Drug Delivery Systems and Their Effects on Biological Barriers. Sci. Pharm. 2019, 87, 33. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Porcino, M.; Qiu, J.; Constantin, D.; Martineau-Corcos, C.; Gref, R. Doxorubicin-Loaded Metal-Organic Frameworks Nanoparticles with Engineered Cyclodextrin Coatings: Insights on Drug Location by Solid State NMR Spectroscopy. Nanomaterials 2021, 11, 945. [Google Scholar] [CrossRef] [PubMed]
- Martwong, E.; Chuetor, S.; Junthip, J. Adsorption of Paraquat by Poly(Vinyl Alcohol)-Cyclodextrin Nanosponges. Polymers 2021, 13, 4110. [Google Scholar] [CrossRef]
- Kono, H.; Nakamura, T. Polymerization of β-Cyclodextrin with 1,2,3,4-Butanetetracarboxylic Dianhydride: Synthesis, Structural Characterization, and Bisphenol A Adsorption Capacity. React. Funct. Polym. 2013, 73, 1096–1102. [Google Scholar] [CrossRef]
- Tang, T.; Liu, H.; Liu, J.; Jiang, W.; Liu, Z.; Yan, J.; Xie, L.; Li, L. Facile Synthesis of an Environment-Friendly Cyclodextrin-Based Polycarboxylic Acid Polymer for Efficient Removal of U(VI) and Eu(III). J. Radioanal. Nucl. Chem. 2021, 329, 1247–1260. [Google Scholar] [CrossRef]
- Mok, C.F.; Ching, Y.C.; Muhamad, F.; Osman, N.A.A.; Hai, N.D.; Hassan, C.R.C. Adsorption of Dyes Using Poly(Vinyl Alcohol) (PVA) and PVA-Based Polymer Composite Adsorbents: A Review. J. Polym. Environ. 2020, 28, 775–793. [Google Scholar] [CrossRef]
- Zhao, D.; Zhao, L.; Zhu, C.; Tian, Z.; Shen, X. Synthesis and Properties of Water-Insoluble β-Cyclodextrin Polymer Crosslinked by Citric Acid with PEG-400 as Modifier. Carbohydr. Polym. 2009, 78, 125–130. [Google Scholar] [CrossRef]
- Patil, N.V.; Netravali, A.N. Cyclodextrin-Based “Green” Wrinkle-Free Finishing of Cotton Fabrics. Ind. Eng. Chem. Res. 2019, 58, 20496–20504. [Google Scholar] [CrossRef]
- Yang, C.Q.; Lu, Y.; Lickfield, G.C. Chemical Analysis of 1,2,3,4-Butanetetracarboxylic Acid. Text. Res. J. 2002, 72, 817–824. [Google Scholar] [CrossRef]
- Çay, A.; Kumbasar, E.P.A.; Keskin, Z.; Akduman, Ç.; Ürkmez, A.S. Crosslinking of Poly(Vinyl Alcohol) Nanofibres with Polycarboxylic Acids: Biocompatibility with Human Skin Keratinocyte Cells. J. Mater. Sci 2017, 52, 12098–12108. [Google Scholar] [CrossRef]
- Liao, G.-M.; Yang, C.-C.; Hu, C.-C.; Pai, Y.-L.; Lue, S.J. Novel Quaternized Polyvinyl Alcohol/Quaternized Chitosan Nano-Composite as an Effective Hydroxide-Conducting Electrolyte. J. Membr. Sci. 2015, 485, 17–29. [Google Scholar] [CrossRef]
- Salazar, J.D.R. Study of Structural, Thermic, μ-Raman and Optic Transformation of PVA/TiO2 Polymeric Membranes. Sci. Tech. 2018, 23, 543–552. [Google Scholar] [CrossRef]
- Casanova, E.; Pelé-Meziani, C.; Guilminot, É.; Mevellec, J.-Y.; Riquier-Bouclet, C.; Vinçotte, A.; Lemoine, G. The Use of Vibrational Spectroscopy Techniques as a Tool for the Discrimination and Identification of the Natural and Synthetic Organic Compounds Used in Conservation. Anal. Methods 2016, 8, 8514–8527. [Google Scholar] [CrossRef]
- Munnier, E.; Al Assaad, A.; David, S.; Mahut, F.; Vayer, M.; Van Gheluwe, L.; Yvergnaux, F.; Sinturel, C.; Soucé, M.; Chourpa, I.; et al. Homogeneous Distribution of Fatty Ester-based Active Cosmetic Ingredients in Hydrophilic Thin Films by Means of Nanodispersion. Int. J. Cosmet. Sci. 2020, 42, 512–519. [Google Scholar] [CrossRef]
- Gu, X.; Yang, C.Q. FT-IR and FT-Raman Spectroscopy Study of the Cyclic Anhydride Intermediates for Esterification of Cellulose: I. Formation of Anhydrides without a Catalyst. Res. Chem. Intermed. 1998, 24, 979–996. [Google Scholar] [CrossRef]
- Gieroba, B.; Kalisz, G.; Sroka-Bartnicka, A.; Płazińska, A.; Płaziński, W.; Starek, M.; Dąbrowska, M. Molecular Structure of Cefuroxime Axetil Complexes with α-, β-, γ-, and 2-Hydroxypropyl-β-Cyclodextrins: Molecular Simulations and Raman Spectroscopic and Imaging Studies. Int. J. Mol. Sci. 2021, 22, 5238. [Google Scholar] [CrossRef] [PubMed]
- Sierpe, R.; Lang, E.; Jara, P.; Guerrero, A.R.; Chornik, B.; Kogan, M.J.; Yutronic, N. Gold Nanoparticles Interacting with β-Cyclodextrin–Phenylethylamine Inclusion Complex: A Ternary System for Photothermal Drug Release. ACS Appl. Mater. Interfaces 2015, 7, 15177–15188. [Google Scholar] [CrossRef] [PubMed]
- Rachmawati, H.; Edityaningrum, C.A.; Mauludin, R. Molecular Inclusion Complex of Curcumin–β-Cyclodextrin Nanoparticle to Enhance Curcumin Skin Permeability from Hydrophilic Matrix Gel. AAPS PharmSciTech 2013, 14, 1303–1312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ceborska, M.; Zimnicka, M.; Kowalska, A.A.; Dąbrowa, K.; Repeć, B. Structural Diversity in the Host–Guest Complexes of the Antifolate Pemetrexed with Native Cyclodextrins: Gas Phase, Solution and Solid State Studies. Beilstein J. Org. Chem. 2017, 13, 2252–2263. [Google Scholar] [CrossRef] [PubMed]
- Euvrard, É.; Morin-Crini, N.; Druart, C.; Bugnet, J.; Martel, B.; Cosentino, C.; Moutarlier, V.; Crini, G. Cross-Linked Cyclodextrin-Based Material for Treatment of Metals and Organic Substances Present in Industrial Discharge Waters. Beilstein J. Org. Chem. 2016, 12, 1826–1838. [Google Scholar] [CrossRef]
- Kono, H.; Fujita, S.; Oeda, I. Comparative Study of Homogeneous Solvents for the Esterification Crosslinking of Cellulose with 1,2,3,4-Butanetetracarboxylic Dianhydride and Water Absorbency of the Reaction Products. J. Appl. Polym. Sci. 2013, 127, 478–486. [Google Scholar] [CrossRef]
- Musuc, A.M.; Anuta, V.; Atkinson, I.; Popa, V.T.; Sarbu, I.; Mircioiu, C.; Abdalrb, G.A.; Mitu, M.A.; Ozon, E.A. Development and Characterization of Orally Disintegrating Tablets Containing a Captopril-Cyclodextrin Complex. Pharmaceutics 2020, 12, 744. [Google Scholar] [CrossRef]
- Bekkouche, S.; Merouani, S.; Hamdaoui, O.; Bouhelassa, M. Efficient Photocatalytic Degradation of Safranin O by Integrating Solar-UV/TiO2/Persulfate Treatment: Implication of Sulfate Radical in the Oxidation Process and Effect of Various Water Matrix Components. J. Photochem. Photobiol. A Chem. 2017, 345, 80–91. [Google Scholar] [CrossRef]
- Ojediran, J.O.; Dada, A.O.; Aniyi, S.O.; David, R.O.; Adewumi, A.D. Mechanism and Isotherm Modeling of Effective Adsorption of Malachite Green as Endocrine Disruptive Dye Using Acid Functionalized Maize Cob (AFMC). Sci. Rep. 2021, 11, 21498. [Google Scholar] [CrossRef]
- Zbair, M.; Anfar, Z.; Ahsaine, H.A.; Khallok, H. Kinetics, Equilibrium, Statistical Surface Modeling and Cost Analysis of Paraquat Removal from Aqueous Solution Using Carbonated Jujube Seed. RSC Adv. 2019, 9, 1084–1094. [Google Scholar] [CrossRef] [Green Version]
- Oughlis-Hammache, F.; Skiba, M.; Hallouard, F.; Moulahcene, L.; Kebiche-Senhadji, O.; Benamor, M.; Lahiani-Skiba, M. Synthesis and Characterization of Poly(Vinyl-Alcohol)-Poly(β-Cyclodextrin) Copolymer Membranes for Aniline Extraction. Membr. Water Treat. 2016, 7, 223–240. [Google Scholar] [CrossRef]
- Jiang, L.-W.; Zeng, F.-T.; Zhang, Y.; Xu, M.-Y.; Xie, Z.-W.; Wang, H.-Y.; Wu, Y.-X.; He, F.-A.; Jiang, H.-L. Preparation of a Novel Fe3O4/Graphite Oxide Nanosheet/Citric Acid-Crosslinked β-Cyclodextrin Polymer Composite to Remove Methylene Blue from Water. Adv. Powder Technol. 2021, 32, 492–503. [Google Scholar] [CrossRef]
- Zhou, K.; Li, Y.; Li, Q.; Du, Q.; Wang, D.; Sui, K.; Wang, C.; Li, H.; Xia, Y. Kinetic, Isotherm and Thermodynamic Studies for Removal of Methylene Blue Using β-Cyclodextrin/Activated Carbon Aerogels. J. Polym. Environ. 2018, 26, 3362–3370. [Google Scholar] [CrossRef]




| Name | Composition (% w/v) | % Yield | IEC (mmol/g) | % Swelling | β-CD Content (mmol/g) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| β-CD | BTCA | PVOH | Mean | S.D. | Mean | S.D. | Mean | S.D. | Mean | S.D. | |
| BP0.5 | 10 | 11.14 | 0.5 | 73.3 | 1.7 | 3.69 | 0.05 | 65.3 | 1.0 | 0.085 | 0.001 |
| BP2 | 10 | 11.14 | 2 | 71.5 | 1.8 | 3.55 | 0.03 | 59.1 | 1.2 | 0.097 | 0.006 |
| BP5 | 10 | 11.14 | 5 | 68.4 | 1.1 | 3.31 | 0.09 | 54.2 | 0.5 | 0.160 | 0.003 |
| Qe (exp) | Pseudo-First-Order | Pseudo-Second-Order | Adsorption Mechanism | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | Qe (cal) | k1 | R2 | Qe (cal) | k2 | h | t1/2 | k31 | k32 | ||
| PQ | 12.0 | 0.8097 | 1.8 | 0.0173 | 0.9999 | 12.0 | 0.1427 | 20.7 | 0.6 | 0.5795 | 0.0045 |
| SO | 11.9 | 0.8727 | 7.5 | 0.0041 | 0.9997 | 11.9 | 0.0994 | 14.2 | 0.8 | 0.2757 | 0.0016 |
| MG | 9.4 | 0.9233 | 8.4 | 0.0150 | 0.9999 | 9.4 | 0.1152 | 10.2 | 0.9 | 0.1643 | 0.0221 |
| Qm (exp) | Langmuir Isotherm | Freundlich Isotherm | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | Qm (cal) | KL | χ2 | RL for C0 (mg/L) | R2 | Qm (cal) | Kf | 1/n | χ2 | ||||||
| 25 | 50 | 150 | 250 | 300 | |||||||||||
| PQ | 112.9 | 0.9867 | 120.5 | 0.09 | 0.5 | 0.300 | 0.176 | 0.067 | 0.041 | 0.034 | 0.9486 | 143.9 | 14.1 | 0.49 | 6.7 |
| SO | 79.3 | 0.9964 | 92.6 | 0.16 | 1.9 | 0.201 | 0.112 | 0.040 | 0.025 | 0.021 | 0.9854 | 139.5 | 14.4 | 0.52 | 25.9 |
| MG | 61.1 | 0.9927 | 64.9 | 0.34 | 0.2 | 0.107 | 0.056 | 0.019 | 0.012 | 0.010 | 0.9602 | 83.4 | 16.3 | 0.40 | 6.0 |
| Contaminant | Adsorbent | Adsorption Dosage | Paraquat Concentration (mg/L) | Maximum Adsorption Capacity |
|---|---|---|---|---|
| PQ | BTCA cross-linked CD and PVOH nanosponges [This work] | 0.02 g in 0.01 L | 25–300 mg/L | 120.5 mg/g |
| Citric acid cross-linked CD and PVOH nanosponges [120] | 0.02 g in 0.01 L | 25–300 mg/L | 112.2 mg/g | |
| BTCA cross-linked CD nanosponges [21] | 0.02 g in 0.01 L | 10–200 mg/L | 26.7 mg/g | |
| Citric acid cross-linked CD nanosponges [20] | 0.02 g in 0.01 L | 10–200 mg/L | 21.9 mg/g | |
| Magnetic adsorbent [24] | 0.0025 g in 0.005 L | 30–900 mg/L | 242.2 mg/g | |
| Carbon nanotubes [17] | 0.002 g in 0.005 L | 70–250 mg/L | 218.6 mg/g | |
| Graphene oxide [22] | 0.02 g in 0.025 L | 4–24 mg/L | 29.15 mg/g | |
| Bentonite [13] | 0.04 g in 0.025 L | 4–24 mg/L | 11.75 mg/g | |
| SO | BTCA cross-linked CD and PVOH nanosponges [This work] | 0.02 g in 0.01 L | 25–250 mg/L | 92.6 mg/g |
| Magnetic adsorbent [87] | 0.01 g in 0.04 L | 50–500 mg/L | 769.23 mg/g | |
| Carbon nanotubes [79] | 0.02 g in 0.01 L | 1–15 mg/L | 8.42 mg/g | |
| Graphene oxide [84] | 0.01 g in 0.02 L | 50–1800 mg/L | 279.6 mg/g | |
| Bentonite [76] | 0.025 g in 0.05 L | 100–600 mg/L | 294.1 mg/g | |
| MG | BTCA cross-linked CD and PVOH nanosponges [This work] | 0.02 g in 0.01 L | 25–250 mg/L | 64.9 mg/g |
| Magnetic adsorbent [60] | 0.02 g in 0.04 L | 50–150 mg/L | 512.8 mg/g | |
| Carbon nanotubes [50] | 0.02 g in 0.02 L | 10–50 mg/L | 11.76 mg/g | |
| Graphene oxide [57] | - | - | 102.4 mg/g | |
| Bentonite [45] | 1 g in 0.1 L | 50–300 mg/L | 178.6 mg/g |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martwong, E.; Chuetor, S.; Junthip, J. Adsorption of Cationic Contaminants by Cyclodextrin Nanosponges Cross-Linked with 1,2,3,4-Butanetetracarboxylic Acid and Poly(vinyl alcohol). Polymers 2022, 14, 342. https://doi.org/10.3390/polym14020342
Martwong E, Chuetor S, Junthip J. Adsorption of Cationic Contaminants by Cyclodextrin Nanosponges Cross-Linked with 1,2,3,4-Butanetetracarboxylic Acid and Poly(vinyl alcohol). Polymers. 2022; 14(2):342. https://doi.org/10.3390/polym14020342
Chicago/Turabian StyleMartwong, Ekkachai, Santi Chuetor, and Jatupol Junthip. 2022. "Adsorption of Cationic Contaminants by Cyclodextrin Nanosponges Cross-Linked with 1,2,3,4-Butanetetracarboxylic Acid and Poly(vinyl alcohol)" Polymers 14, no. 2: 342. https://doi.org/10.3390/polym14020342
APA StyleMartwong, E., Chuetor, S., & Junthip, J. (2022). Adsorption of Cationic Contaminants by Cyclodextrin Nanosponges Cross-Linked with 1,2,3,4-Butanetetracarboxylic Acid and Poly(vinyl alcohol). Polymers, 14(2), 342. https://doi.org/10.3390/polym14020342