Mechanical, Structural, and Biological Characteristics of Polylactide/Wollastonite 3D Printed Scaffolds
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Wollastonite
2.2. Composites and Scaffolds Preparation
2.3. Particles and Scaffolds Characterization
2.4. Scaffolds Mechanical Testing
2.5. Study of Cell Colonization and Antibacterial Properties
2.5.1. MSCs Colonization
2.5.2. Antibacterial Activity
2.5.3. Statistics
3. Results and Discussion
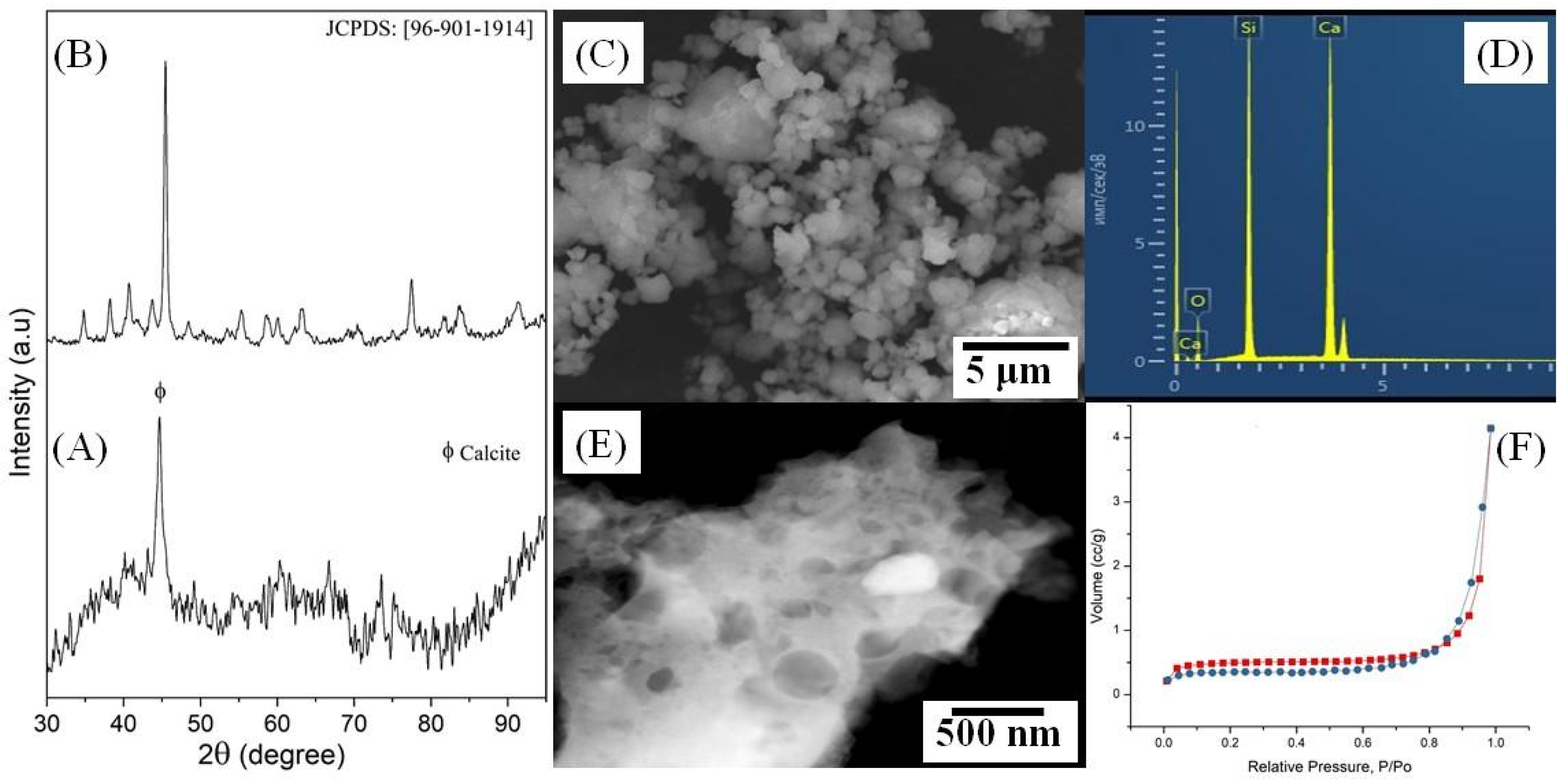
3.1. Characterization of Wollastonite
3.2. Characterization of HAp
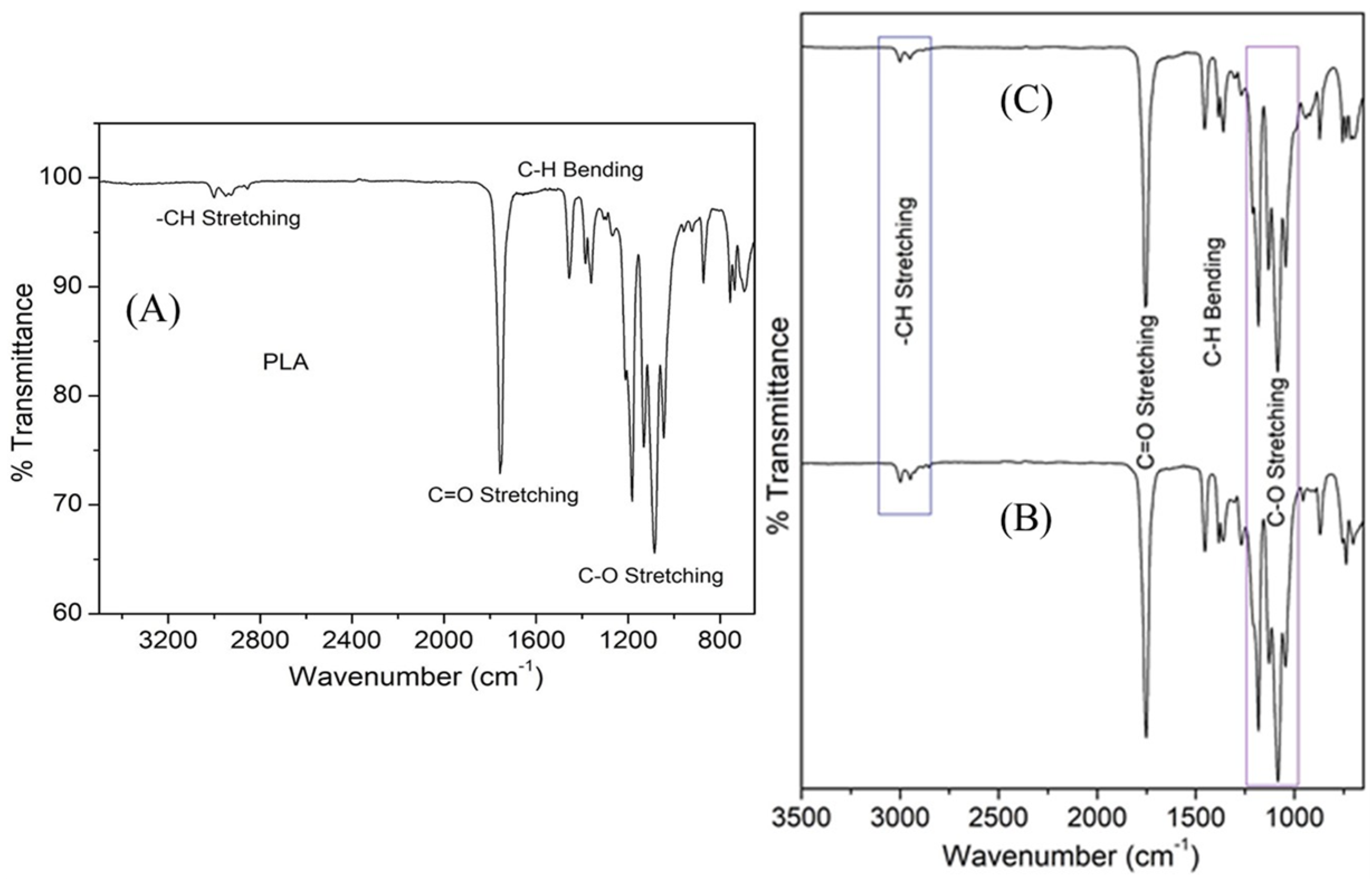
3.3. FT-IR Characterization of PLA and 3D Printed Scaffolds
3.4. Mechanical Testing of 3D Printed Scaffolds
3.5. In Vitro Studies
3.5.1. Antibacterial Activity
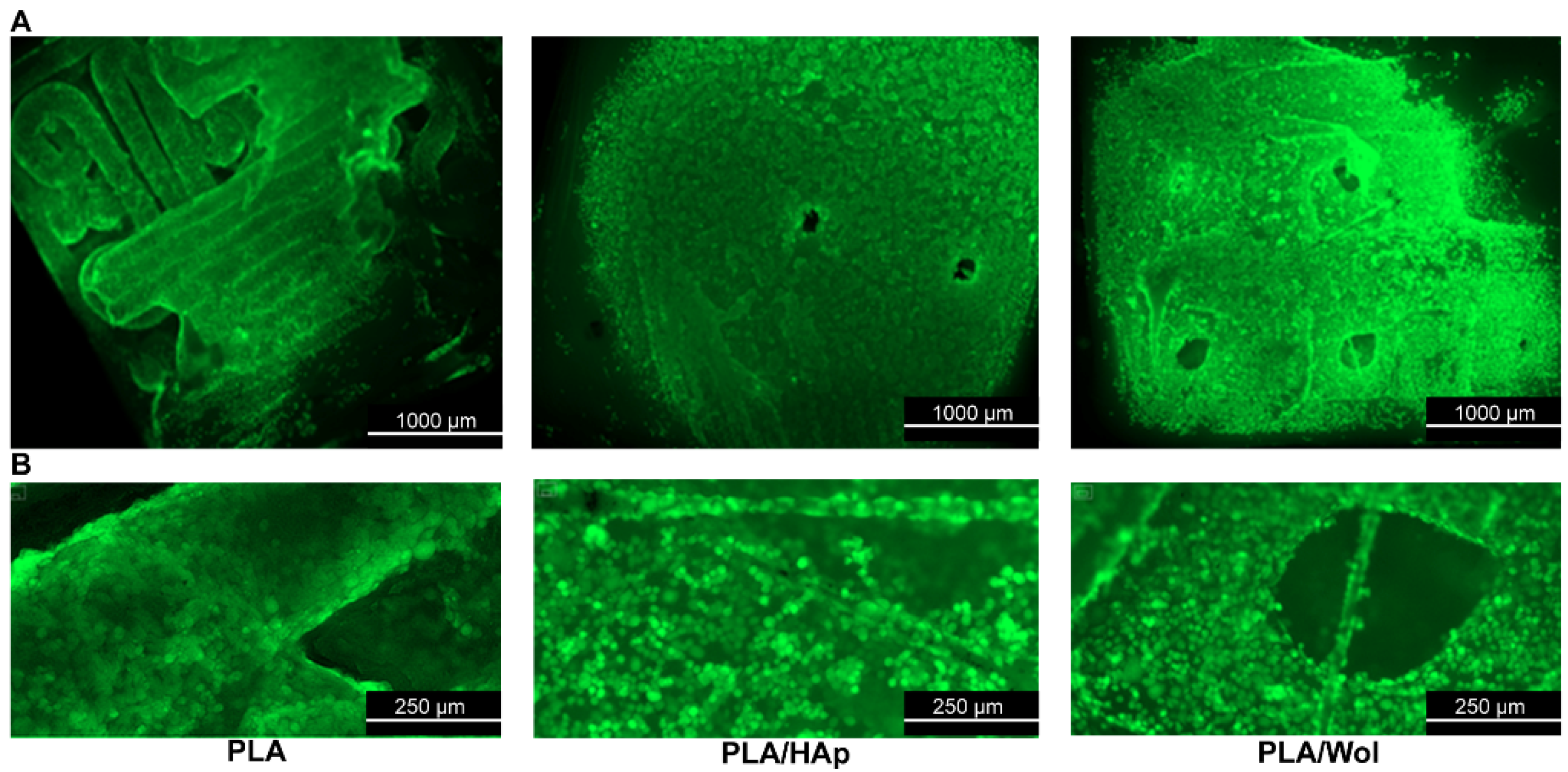
3.5.2. MSCs Colonization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Xu, Y.; Zhang, F.; Zhai, W.; Cheng, S.; Li, J.; Wang, Y. Unraveling of Advances in 3D-Printed Polymer-Based Bone Scaffolds. Polymers 2022, 14, 566. [Google Scholar] [CrossRef] [PubMed]
- Indurkar, A.; Bangde, P.; Gore, M.; Agrawal, A.K.; Jain, R.; Dandekar, P. Fabrication of Guar Gum-Gelatin Scaffold for Soft Tissue Engineering. Carbohydr. Polym. Technol. Appl. 2020, 1, 100006. [Google Scholar] [CrossRef]
- Roseti, L.; Parisi, V.; Petretta, M.; Cavallo, C.; Desando, G.; Bartolotti, I.; Grigolo, B. Scaffolds for Bone Tissue Engineering: State of the Art and New Perspectives. Mater. Sci. Eng. C 2017, 78, 1246–1262. [Google Scholar] [CrossRef] [PubMed]
- Pedde, R.D.; Mirani, B.; Navaei, A.; Styan, T.; Wong, S.; Mehrali, M.; Thakur, A.; Mohtaram, N.K.; Bayati, A.; Dolatshahi-Pirouz, A.; et al. Emerging Biofabrication Strategies for Engineering Complex Tissue Constructs. Adv. Mater. 2017, 29, 1606061. [Google Scholar] [CrossRef]
- Arif, Z.U.; Khalid, M.Y.; Noroozi, R.; Sadeghianmaryan, A.; Jalalvand, M.; Hossain, M. Recent Advances in 3D-Printed Polylactide and Polycaprolactone-Based Biomaterials for Tissue Engineering Applications. Int. J. Biol. Macromol. 2022, 218, 930–968. [Google Scholar] [CrossRef]
- Ganguly, S.; Margel, S. 3D Printed Magnetic Polymer Composite Hydrogels for Hyperthermia and Magnetic Field Driven Structural Manipulation. Prog. Polym. Sci. 2022, 131, 101574. [Google Scholar] [CrossRef]
- Saranti, A.; Tiron-Stathopoulos, A.; Papaioannou, L.; Gioti, C.; Ioannou, A.; Karakassides, M.A.; Avgoustakis, K.; Koutselas, I.; Dimos, K. 3D-Printed Bioactive Scaffolds for Bone Regeneration Bearing Carbon Dots for Bioimaging Purposes. Smart Mater. Med. 2022, 3, 12–19. [Google Scholar] [CrossRef]
- Indurkar, A.; Bangde, P.; Gore, M.; Reddy, P.; Jain, R.; Dandekar, P. Optimization of Guar Gum-Gelatin Bioink for 3D Printing of Mammalian Cells. Bioprinting 2020, 20, e00101. [Google Scholar] [CrossRef]
- Mirkhalaf, M.; Men, Y.; Wang, R.; No, Y.; Zreiqat, H. Personalized 3D Printed Bone Scaffolds: A Review. Acta Biomater. 2022; in press. [Google Scholar] [CrossRef]
- Zieliński, P.S.; Gudeti, P.K.R.; Rikmanspoel, T.; Włodarczyk-Biegun, M.K. 3D Printing of Bio-Instructive Materials: Toward Directing the Cell. Bioact. Mater. 2023, 19, 292–327. [Google Scholar] [CrossRef] [PubMed]
- Noroozi, R.; Tatar, F.; Zolfagharian, A.; Brighenti, R.; Shamekhi, M.A.; Rastgoo, A.; Hadi, A.; Bodaghi, M. Additively Manufactured Multi-Morphology Bone-like Porous Scaffolds: Experiments and Micro-Computed Tomography-Based Finite Element Modeling Approaches. Int. J. Bioprint. 2022, 8, 40–53. [Google Scholar] [CrossRef]
- Khalid, M.Y.; Arif, Z.U.; Noroozi, R.; Zolfagharian, A.; Bodaghi, M. 4D Printing of Shape Memory Polymer Composites: A Review on Fabrication Techniques, Applications, and Future Perspectives. J. Manuf. Process. 2022, 81, 759–797. [Google Scholar] [CrossRef]
- Gkika, D.A.; Maliaris, G.; Vordos, N.; Mitropoulos, A.C.; Kyzas, G.Z. Cost Profile of 3D Printing Using Biomaterials on a Lab Scale. Biointerface Res. Appl. Chem. 2023, 13, 93. [Google Scholar] [CrossRef]
- Mandala, R.; Bannoth, A.P.; Akella, S.; Rangari, V.K.; Kodali, D. A Short Review on Fused Deposition Modeling 3D Printing of Bio-Based Polymer Nanocomposites. J. Appl. Polym. Sci. 2022, 139, 51904. [Google Scholar] [CrossRef]
- Boccaccini, A. Bioresorbable and Bioactive Polymer/Bioglass® Composites with Tailored Pore Structure for Tissue Engineering Applications. Compos. Sci. Technol. 2003, 63, 2417–2429. [Google Scholar] [CrossRef]
- Choudhary, R.; Venkatraman, S.K.; Bulygina, I.; Senatov, F.; Kaloshkin, S.; Swamiappan, S. Designing of Porous PMMA/Diopside Bone Cement for Non-Load Bearing Applications. J. Asian Ceram. Soc. 2020, 8, 862–872. [Google Scholar] [CrossRef]
- Choudhary, R.; Koppala, S.; Srivastava, A.; Sasikumar, S. In-Vitro Bioactivity of Nanocrystalline and Bulk Larnite/Chitosan Composites: Comparative Study. J. Sol. Gel. Sci. Technol. 2015, 74, 631–640. [Google Scholar] [CrossRef]
- Alksne, M.; Kalvaityte, M.; Simoliunas, E.; Rinkunaite, I.; Gendviliene, I.; Locs, J.; Rutkunas, V.; Bukelskiene, V. In Vitro Comparison of 3D Printed Polylactic Acid/Hydroxyapatite and Polylactic Acid/Bioglass Composite Scaffolds: Insights into Materials for Bone Regeneration. J. Mech. Behav. Biomed. Mater. 2020, 104, 103641. [Google Scholar] [CrossRef]
- Diez-Escudero, A.; Andersson, B.; Persson, C.; Hailer, N.P. Hexagonal Pore Geometry and the Presence of Hydroxyapatite Enhance Deposition of Mineralized Bone Matrix on Additively Manufactured Polylactic Acid Scaffolds. Mater. Sci. Eng. C 2021, 125, 112091. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Scalera, F.; Gervaso, F.; Montagna, F.; Sannino, A.; Maffezzoli, A. One-Step Solvent-Free Process for the Fabrication of High Loaded PLA/HA Composite Filament for 3D Printing. J. Therm. Anal. Calorim. 2018, 134, 575–582. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, B.; Li, M.; Li, J.; Zhang, C.; Han, Y.; Wang, L.; Wang, K.; Zhou, C.; Liu, L.; et al. 3D Printing of PLA/n-HA Composite Scaffolds with Customized Mechanical Properties and Biological Functions for Bone Tissue Engineering. Compos. Part B Eng. 2021, 224, 109192. [Google Scholar] [CrossRef]
- Noroozi, R.; Shamekhi, M.A.; Mahmoudi, R.; Zolfagharian, A.; Asgari, F.; Mousavizadeh, A.; Bodaghi, M.; Hadi, A.; Haghighipour, N. In vitro static and dynamic cell culture study of novel bone scaffolds based on 3D-printed PLA and cell-laden alginate hydrogel. Biomed. Mater. 2022, 17, 045024. [Google Scholar] [CrossRef]
- Bernardo, M.P.; da Silva, B.C.R.; Hamouda, A.E.I.; de Toledo, M.A.S.; Schalla, C.; Rütten, S.; Goetzke, R.; Mattoso, L.H.C.; Zenke, M.; Sechi, A. PLA/Hydroxyapatite Scaffolds Exhibit In Vitro Immunological Inertness and Promote Robust Osteogenic Differentiation of Human Mesenchymal Stem Cells without Osteogenic Stimuli. Sci. Rep. 2022, 12, 2333. [Google Scholar] [CrossRef]
- Vallet-Regí, M.; Arcos, D. Silicon Substituted Hydroxyapatites. A Method to Upgrade Calcium Phosphate Based Implants. J. Mater. Chem. 2005, 15, 1509–1516. [Google Scholar] [CrossRef]
- Patel, N.; Best, S.M.; Bonfield, W.; Gibson, I.R.; Hing, K.A.; Damien, E.; Revell, P.A. A comparative study on the in vivo behavior of hydroxyapatite and silicon substituted hydroxyapatite granules. J. Mater. Sci. Mater. Med. 2002, 13, 1199–1206. [Google Scholar] [CrossRef]
- Liu, X.; Ding, C.; Chu, P.K. Mechanism of Apatite Formation on Wollastonite Coatings in Simulated Body Fluids. Biomaterials 2004, 25, 1755–1761. [Google Scholar] [CrossRef]
- Choudhary, R.; Venkatraman, S.K.; Bulygina, I.; Senatov, F.; Kaloshkin, S.; Anisimova, N.; Kiselevskiy, M.; Knyazeva, M.; Kukui, D.; Walther, F.; et al. Biomineralization, Dissolution and Cellular Studies of Silicate Bioceramics Prepared from Eggshell and Rice Husk. Mater. Sci. Eng. C 2021, 118, 111456. [Google Scholar] [CrossRef]
- Hoppe, A.; Mourino, V.; Boccaccini, A.R. Therapeutic inorganic ions in bioactive glasses to enhance bone formation and beyond. Biomater. Sci. 2013, 1, 254–256. [Google Scholar] [CrossRef]
- Venkatraman, S.K.; Choudhary, R.; Genasan, K.; Murali, M.R.; Raghavendran, H.R.B.; Kamarul, T.; Suresh, A.; Abraham, J.; Venkateswaran, S.; Livingston, A.; et al. Antibacterial Wollastonite Supported Excellent Proliferation and Osteogenic Differentiation of Human Bone Marrow Derived Mesenchymal Stromal Cells. J. Sol. Gel. Sci. Technol. 2021, 100, 506–516. [Google Scholar] [CrossRef]
- Tcacencu, I.; Rodrigues, N.; Alharbi, N.; Benning, M.; Toumpaniari, S.; Mancuso, E.; Marshall, M.; Bretcanu, O.; Birch, M.; McCaskie, A.; et al. Osseointegration of Porous Apatite-Wollastonite and Poly(Lactic Acid) Composite Structures Created Using 3D Printing Techniques. Mater. Sci. Eng. C 2018, 90, 1–7. [Google Scholar] [CrossRef]
- Goswami, J.; Bhatnagar, N.; Mohanty, S.; Ghosh, A.K. Processing and Characterization of Poly(Lactic Acid) Based Bioactive Composites for Biomedical Scaffold Application. Express Polym. Lett. 2013, 7, 767–777. [Google Scholar] [CrossRef]
- Saravana, S.; Kandaswamy, R. Investigation on the Mechanical and Thermal Properties of PLA/Calcium Silicate Biocomposites for Injection Molding Applications. Silicon 2019, 11, 1143–1150. [Google Scholar] [CrossRef]
- Chaiwutthinan, P.; Chuayjuljit, S.; Srasomsub, S.; Boonmahitthisud, A. Composites of Poly(Lactic Acid)/Poly(Butylene Adipate-Co-Terephthalate) Blend with Wood Fiber and Wollastonite: Physical Properties, Morphology, and Biodegradability. J. Appl. Polym. Sci. 2019, 136, 47543. [Google Scholar] [CrossRef]
- Chaiwutthinan, P.; Chauyjuljit, S.; Thipkham, N.; Kowalski, C.P.; Boonmahitthisud, A. Poly(Lactic Acid)/Ethylene Vinyl Acetate Copolymer Blend Composites with Wood Flour and Wollastonite: Physical Properties, Morphology, and Biodegradability. J. Vinyl Addit. Technol. 2019, 25, 313–327. [Google Scholar] [CrossRef]
- Rajendran, A.; Barik, R.C.; Natarajan, D.; Kiran, M.S.; Pattanayak, D.K. Synthesis, Phase Stability of Hydroxyapatite–Silver Composite with Antimicrobial Activity and Cytocompatability. Ceram. Int. 2014, 40, 10831–10838. [Google Scholar] [CrossRef]
- El-Kady, A.M.; Ali, A.F.; Rizk, R.A.; Ahmed, M.M. Synthesis, Characterization and Microbiological Response of Silver Doped Bioactive Glass Nanoparticles. Ceram. Int. 2012, 38, 177–188. [Google Scholar] [CrossRef]
- Hu, S.; Chang, J.; Liu, M.; Ning, C. Study on Antibacterial Effect of 45S5 Bioglass®. J. Mater. Sci. Mater. Med. 2009, 20, 281–286. [Google Scholar] [CrossRef]
- Hu, S.; Ning, C.; Zhou, Y.; Chen, L.; Lin, K.; Chang, J. Antibacterial Activity of Silicate Bioceramics. J. Wuhan Univ. Technol. Mat. Sci. Ed. 2011, 26, 226–230. [Google Scholar] [CrossRef]
- Choudhary, R.; Venkatraman, S.K.; Chatterjee, A.; Vecstaudza, J.; Yáñez-Gascón, M.J.; Pérez-Sánchez, H.; Locs, J.; Abraham, J.; Swamiappan, S. Biomineralization, Antibacterial Activity and Mechanical Properties of Biowaste Derived Diopside Nanopowders. Adv. Powder Technol. 2019, 30, 1950–1964. [Google Scholar] [CrossRef]
- Choudhary, R.; Manohar, P.; Vecstaudza, J.; Yáñez-Gascón, M.J.; Sánchez, H.P.; Nachimuthu, R.; Locs, J.; Swamiappan, S. Preparation of Nanocrystalline Forsterite by Combustion of Different Fuels and Their Comparative In-Vitro Bioactivity, Dissolution Behaviour and Antibacterial Studies. Mater. Sci. Eng. C 2017, 77, 811–822. [Google Scholar] [CrossRef]
- Choudhary, R.; Chatterjee, A.; Venkatraman, S.K.; Koppala, S.; Abraham, J.; Swamiappan, S. Antibacterial Forsterite (Mg2SiO4) Scaffold: A Promising Bioceramic for Load Bearing Applications. Bioact. Mater. 2018, 3, 218–224. [Google Scholar] [CrossRef]
- Choudhary, R.; Venkatraman, S.K.; Bulygina, I.; Chatterjee, A.; Abraham, J.; Senatov, F.; Kaloshkin, S.; Ilyasov, A.; Abakumov, M.; Knyazeva, M.; et al. Impact of Forsterite Addition on Mechanical and Biological Properties of Composites. J. Asian Ceram. Soc. 2020, 8, 1051–1065. [Google Scholar] [CrossRef]
- Diba, M.; Goudouri, O.-M.; Tapia, F.; Boccaccini, A.R. Magnesium-Containing Bioactive Polycrystalline Silicate-Based Ceramics and Glass-Ceramics for Biomedical Applications. Curr. Opin. Solid State Mater. Sci. 2014, 18, 147–167. [Google Scholar] [CrossRef]
- Ribeiro, M.; Monteiro, F.J.; Ferraz, M.P. Infection of Orthopedic Implants with Emphasis on Bacterial Adhesion Process and Techniques Used in Studying Bacterial-Material Interactions. Biomatter 2012, 2, 176–194. [Google Scholar] [CrossRef]
- Voznesensky, E.F.; Timoshina, Y.A.; Karimullin, I.I.; Anisimova, N.Y.; Larin, I.I.; Statnik, E.S. Plasma Methods for Preparation of the Substrate and Fixing the Nanoparticles in the Obtaining of Disposable Antibacterial Synthetic Materials. Mater. Lett. 2022, 308, 131193. [Google Scholar] [CrossRef]
- Lakshmi, R.; Velmurugan, V.; Sasikumar, S. Preparation and Phase Evolution of Wollastonite by Sol-Gel Combustion Method Using Sucrose as the Fuel. Combust. Sci. Technol. 2013, 185, 1777–1785. [Google Scholar] [CrossRef]
- Charmforoushan, A.; Roknabadi, M.R.; Shahtahmassebi, N.; Malaekeh-Nikouei, B.; Bagherabadi, M. Synthesis and Controlled Drug Release Behavior of Micro-Mesoporous Wollastonite Nanoparticles. Effect of Calcination Temperature on the Structural and Biodegradability Properties. Mater. Chem. Phys. 2022, 280, 125825. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Ma, K.; Cui, H.; Zhou, A.; Wu, H.; Dong, X.; Zu, F.; Yi, J.; Wang, R.; Xu, Q. Mesoporous Hydroxyapatite: Synthesis in Molecular Self-Assembly and Adsorption Properties. Microporous Mesoporous Mater. 2021, 323, 111164. [Google Scholar] [CrossRef]
- Zimina, A.; Senatov, F.; Choudhary, R.; Kolesnikov, E.; Anisimova, N.; Kiselevskiy, M.; Orlova, P.; Strukova, N.; Generalova, M.; Manskikh, V.; et al. Biocompatibility and Physico-Chemical Properties of Highly Porous PLA/HA Scaffolds for Bone Reconstruction. Polymers 2020, 12, 2938. [Google Scholar] [CrossRef]
- Divya Rani, V.V.; Vinoth-Kumar, L.; Anitha, V.C.; Manzoor, K.; Deepthy, M.; Shantikumar, V.N. Osteointegration of Titanium Implant Is Sensitive to Specific Nanostructure Morphology. Acta Biomater. 2012, 8, 1976–1989. [Google Scholar] [CrossRef]
- Peraire, C.; Arias, J.L.; Bernal, D.; Pou, J.; León, B.; Arañó, A.; Roth, W. Biological Stability and Osteoconductivity in Rabbit Tibia of Pulsed Laser Deposited Hydroxylapatite Coatings. J. Biomed. Mater. Res. 2006, 77A, 370–379. [Google Scholar] [CrossRef]
- Rosales-Leal, J.I.; Rodríguez-Valverde, M.A.; Mazzaglia, G.; Ramón-Torregrosa, P.J.; Díaz-Rodríguez, L.; García-Martínez, O.; Vallecillo-Capilla, M.; Ruiz, C.; Cabrerizo-Vílchez, M.A. Effect of Roughness, Wettability and Morphology of Engineered Titanium Surfaces on Osteoblast-like Cell Adhesion. Colloids Surf. A Physicochem. Eng. Asp. 2010, 365, 222–229. [Google Scholar] [CrossRef]
- Nakao, R.; Myint, S.L.; Wai, S.N.; Uhlin, B.E. Enhanced Biofilm Formation and Membrane Vesicle Release by Escherichia coli Expressing a Commonly Occurring Plasmid Gene, kil. Front. Microbiol. 2018, 9, 2605. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choudhary, R.; Bulygina, I.; Lvov, V.; Zimina, A.; Zhirnov, S.; Kolesnikov, E.; Leybo, D.; Anisimova, N.; Kiselevskiy, M.; Kirsanova, M.; et al. Mechanical, Structural, and Biological Characteristics of Polylactide/Wollastonite 3D Printed Scaffolds. Polymers 2022, 14, 3932. https://doi.org/10.3390/polym14193932
Choudhary R, Bulygina I, Lvov V, Zimina A, Zhirnov S, Kolesnikov E, Leybo D, Anisimova N, Kiselevskiy M, Kirsanova M, et al. Mechanical, Structural, and Biological Characteristics of Polylactide/Wollastonite 3D Printed Scaffolds. Polymers. 2022; 14(19):3932. https://doi.org/10.3390/polym14193932
Chicago/Turabian StyleChoudhary, Rajan, Inna Bulygina, Vladislav Lvov, Anna Zimina, Sergey Zhirnov, Evgeny Kolesnikov, Denis Leybo, Natalya Anisimova, Mikhail Kiselevskiy, Maria Kirsanova, and et al. 2022. "Mechanical, Structural, and Biological Characteristics of Polylactide/Wollastonite 3D Printed Scaffolds" Polymers 14, no. 19: 3932. https://doi.org/10.3390/polym14193932
APA StyleChoudhary, R., Bulygina, I., Lvov, V., Zimina, A., Zhirnov, S., Kolesnikov, E., Leybo, D., Anisimova, N., Kiselevskiy, M., Kirsanova, M., & Senatov, F. (2022). Mechanical, Structural, and Biological Characteristics of Polylactide/Wollastonite 3D Printed Scaffolds. Polymers, 14(19), 3932. https://doi.org/10.3390/polym14193932









