Analytical and Numerical Modeling of Degradation and Pyrolysis of Polyethylene: Measuring Aging with Thermogravimetry
Abstract
1. Introduction
2. Theoretical Model
2.1. Analytical Model for Cross-Linked Polymer
2.2. Analytical Degradation Model
2.3. Analytical Expression for Volatile Fraction
2.4. Coarse-Grained Kinetics of Pyrolysis
3. Materials and Methods
3.1. TGA Experiments with Aged XLPE
3.2. Numerical Aging and Decomposition Model
3.3. Evaporation Limit
4. Results
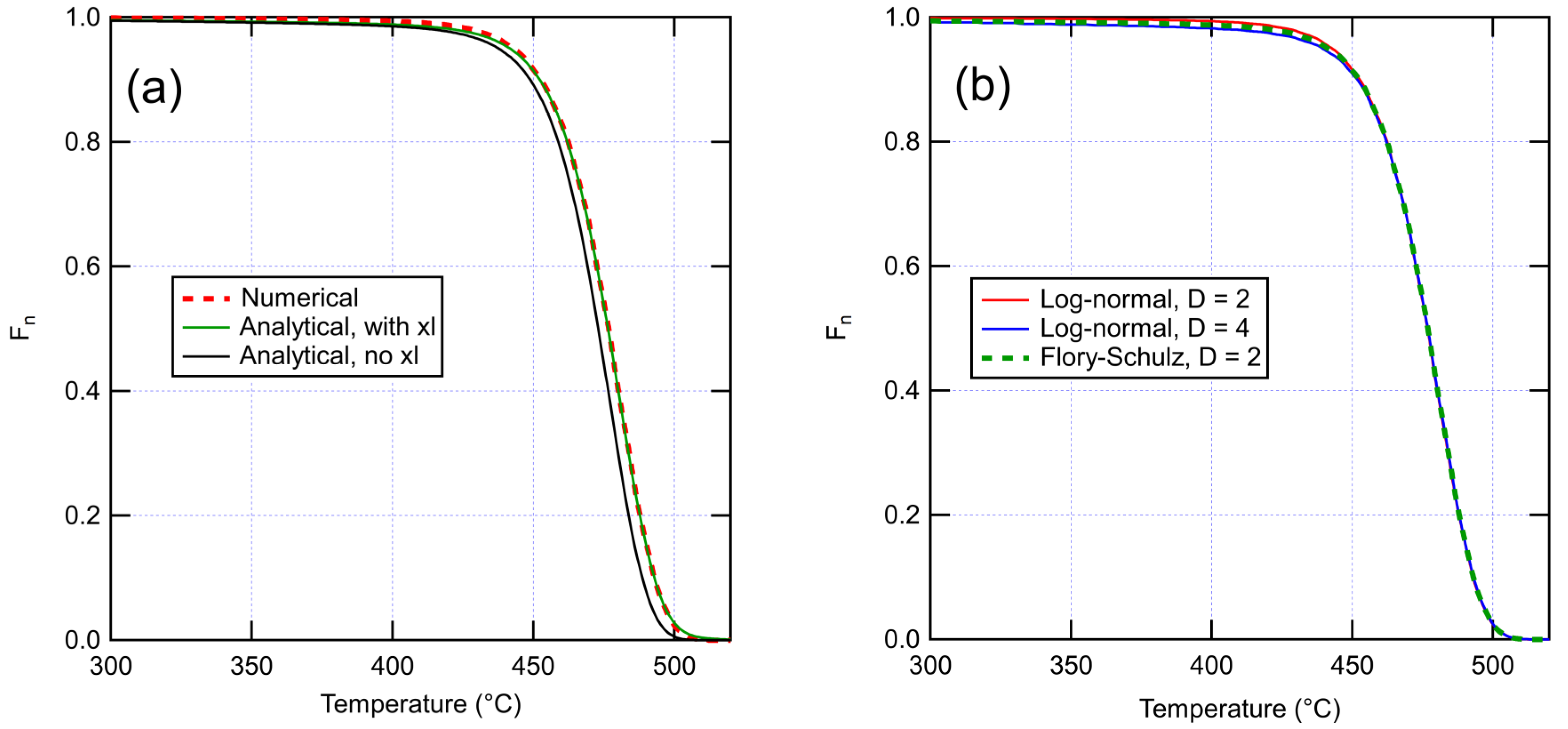
4.1. Model for Unaged XLPE
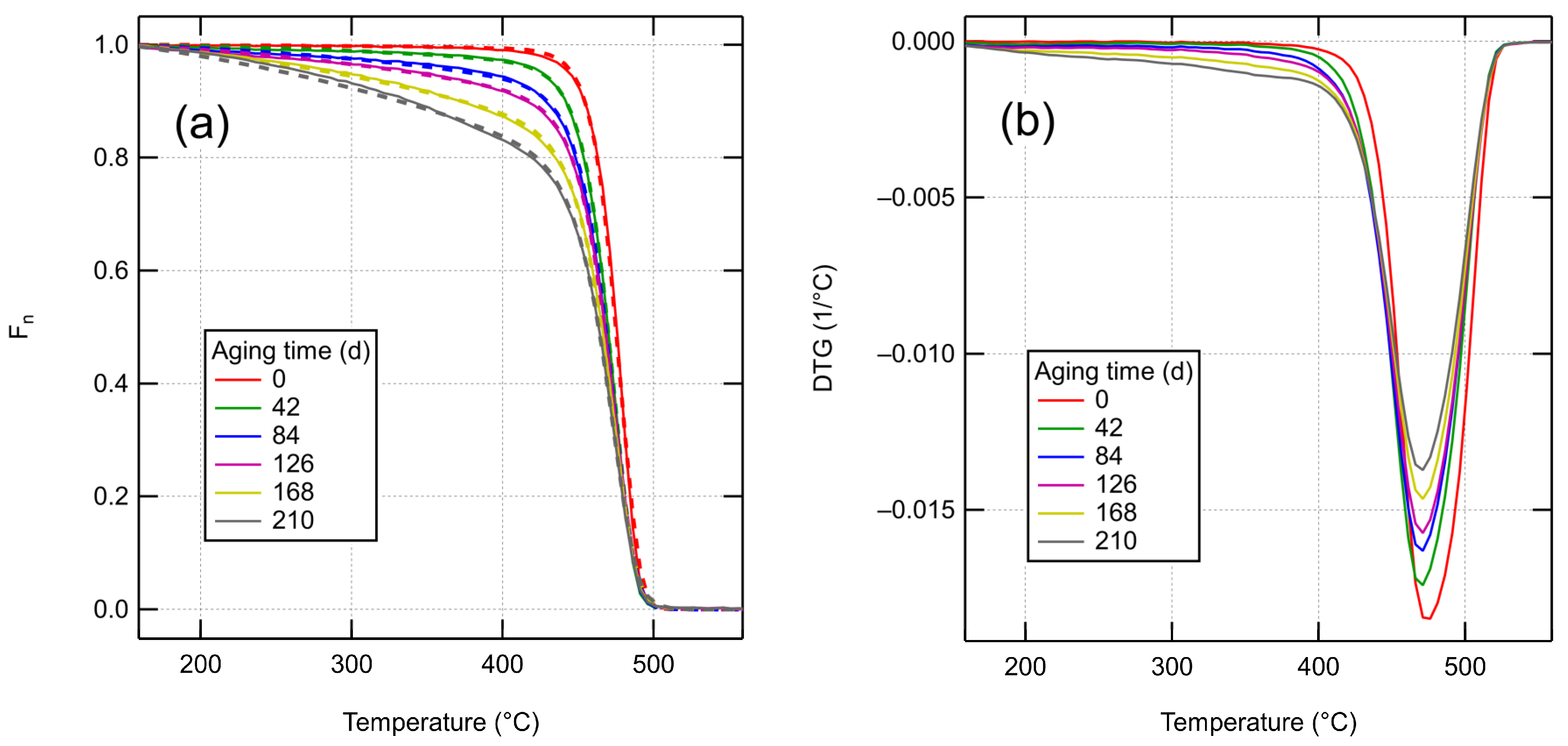
4.2. Aging of XLPE
4.3. Aging Characterization by TGA
5. Discussion
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Celina, M. Review of polymer oxidation and its relationship with materials performance and lifetime prediction. Polym. Degrad. Stab. 2013, 98, 2419–2429. [Google Scholar] [CrossRef]
- Celina, M.; Linde, E.; Brunson, D.; Quintana, A.; Giron, N. Overview of accelerated aging and polymer degradation kinetics for combined radiation-thermal environments. Polym. Degrad. Stab. 2019, 166, 353–378. [Google Scholar] [CrossRef]
- Burnay, S. Degradation of Polymeric Components in Nuclear Power Applications. Energiforsk AB Report 2018:480. Available online: https://energiforsk.se/program/polymera-material-i-karnkraft/rapporter/degradation-of-polymeric-components-in-nuclear-power-applications-2018-480/ (accessed on 1 June 2022).
- Chabira, S.F.; Sebaa, M.; G’sell, C. Oxidation and Crosslinking Processes During Thermal Aging of Low-Density Polyethylene Films. J. Appl. Polym. Sci. 2012, 124, 5200–5208. [Google Scholar] [CrossRef]
- Suraci, S.V.; Fabiani, D.; Roland, S.; Colin, X. Multi scale aging assessment of low-voltage cables subjected to radio-chemical aging: Towards an electrical diagnostic technique. Polym. Test. 2021, 103, 107352. [Google Scholar] [CrossRef]
- Vahabi, H.; Sonnier, R.; Ferry, L. Effects of ageing on the fire behaviour of flame-retarded polymers: A review. Polym. Int. 2015, 64, 313–328. [Google Scholar] [CrossRef]
- Matala, A.; Hostikka, S. Pyrolysis Modelling of PVC Cable Materials. Fire Saf. Sci. 2011, 10, 917–930. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, R.; Ouyang, D.; Wang, J. Investigation on thermal stability and flame spread behavior of new and aged fine electrical wires. J. Therm. Anal. Calorim. 2020, 140, 157–165. [Google Scholar] [CrossRef]
- Weon, J.-I. Effects of thermal ageing on mechanical and thermal behaviors of linear low density polyethylene pipe. Polym. Degrad. Stab. 2010, 95, 14–20. [Google Scholar] [CrossRef]
- Boersma, A. Predicting the efficiency of antioxidants in polymers. Polym. Degrad. Stab. 2006, 91, 472–478. [Google Scholar] [CrossRef]
- Wei, X.-F.; Linde, E.; Hedenqvist, M.S. Plasticiser loss from plastic or rubber products through diffusion and evaporation. NPJ Mater. Degrad. 2019, 3, 18. [Google Scholar] [CrossRef]
- Xu, A.; Roland, S.; Colin, X. Thermal ageing of a silane-crosslinked polyethylene stabilised with a thiodipropionate antioxidant. Polym. Degrad. Stab. 2020, 181, 109276. [Google Scholar] [CrossRef]
- Barbosa, A.P.C.; Fulco, A.P.P.; Guerra, E.S.S.; Arakaki, F.K.; Tosatto, M.; Costa, M.C.B.; Melo, J.D.D. Accelerated aging effects on carbon fiber/epoxy composites. Compos. B Eng. 2017, 110, 298–306. [Google Scholar] [CrossRef]
- Pablos, J.L.; Abrusci, C.; Marín, I.; López-Marín, J.; Catalina, F.; Espí, E.; Corrales, T. Photodegradation of polyethylenes: Comparative effect of Fe and Ca-stearates as pro-oxidant additives. Polym. Degrad. Stab. 2010, 95, 2057–2064. [Google Scholar] [CrossRef]
- Fayolle, B.; Colin, X.; Audouin, L.; Verdu, J. Mechanism of degradation induced embrittlement in polyethylene. Polym. Degrad. Stab. 2007, 92, 231–238. [Google Scholar] [CrossRef]
- Nyden, M.R.; Forney, G.P.; Brown, J.E. Molecular Modeling of Polymer Flammability: Application to the Design of Flame-Resistant Polyethylene. Macromolecules 1992, 25, 1658–1666. [Google Scholar] [CrossRef]
- Vyazovkin, S. Computational aspects of kinetic analysis. Part C. The ICTAC Kinetics Project—The light at the end of the tunnel? Thermochim. Acta 2000, 355, 155–163. [Google Scholar] [CrossRef]
- Nemeth, A.; Blaszó, M.; Baranyai, P.; Vidóczy, T. Thermal degradation of polyethylene modeled on tetracontane. J. Anal. Appl. Pyrolysis 2008, 81, 237–242. [Google Scholar] [CrossRef]
- Levine, S.E.; Broadbelt, L.J. Detailed mechanistic modeling of high-density polyethylene pyrolysis: Low molecular weight product evolution. Polym. Degrad. Stab. 2009, 94, 810–822. [Google Scholar] [CrossRef]
- Gascoin, N.; Navarro-Rodriguez, A.; Fau, G.; Gillard, P. Kinetic modelling of High Density PolyEthylene pyrolysis: Part 2. Reduction of existing detailed mechanism. Polym. Degrad. Stab. 2012, 97, 1142–1150. [Google Scholar] [CrossRef][Green Version]
- Voter, A. Introduction to the Kinetic Monte Carlo Method. In Radiation Effects in Solids; Springer: Dordrecht, The Netherlands, 2007; Volume 235, pp. 1–23. [Google Scholar]
- Gillespie, D.T. Exact Stochastic Simulation of Coupled Chemical Reactions. J. Phys. Chem. 1977, 81, 2340–2361. [Google Scholar] [CrossRef]
- Galina, H.; Lechowicz, J.B. Monte-Carlo modeling of degradation of polymer networks. Polym. Gels Netw. 1998, 6, 103–111. [Google Scholar] [CrossRef]
- Adema, K.N.S.; Makki, H.; Peters, E.A.J.F.; Laven, J.; Van der Ven, L.G.J.; Van Benthem, R.A.T.M.; De With, G. Kinetic Monte Carlo simulation of the photodegradation process of polyester-urethane coatings. Phys. Chem. Chem. Phys. 2015, 17, 19962–19976. [Google Scholar] [CrossRef]
- Bystritskaya, E.V.; Karpukhin, O.N.; Kutsenova, A. Monte Carlo Simulation of Linear Polymer Thermal Depolymerization under Isothermal and Dynamic Modes. Int. J. Polym. Sci. 2011, 2011, 849370. [Google Scholar] [CrossRef][Green Version]
- Vinu, R.; Levine, S.E.; Wang, L.; Broadbelt, L.J. Detailed mechanistic modeling of poly(styrene peroxide) pyrolysis using kinetic Monte Carlo simulation. Chem. Eng. Sci. 2012, 69, 456–471. [Google Scholar] [CrossRef]
- Peterson, B.K.; Formolo, M.J.; Lawson, M. Molecular and detailed isotopic structures of petroleum: Kinetic Monte Carlo analysis of alkane cracking. Geochim. Cosmochim. Acta 2018, 243, 169–185. [Google Scholar] [CrossRef]
- Younker, J.M.; Saito, T.; Hunt, M.A.; Naskar, A.K.; Beste, A. Pyrolysis Pathways of Sulfonated Polyethylene, an Alternative Carbon Fiber Precursor. J. Am. Chem. Soc. 2013, 135, 6130–6141. [Google Scholar] [CrossRef]
- Charlesby, A. Gel formation and molecular weight distribution in long-chain polymers. Proc. R. Soc. A 1954, 222, 542–557. [Google Scholar] [CrossRef]
- St. Cholakov, G.; Wakeham, W.A.; Stateva, R.P. Estimation of normal boiling points of hydrocarbons from descriptors of molecular structure. Fluid Phase Equilib. 1999, 163, 21–42. [Google Scholar] [CrossRef]
- Da Cruz, M.; Van Schoors, L.; Benzarti, K.; Colin, X. Thermo-oxidative degradation of additive free polyethylene. Part I. Analysis of chemical modifications at molecular and macromolecular scales. J. Appl. Polym. Sci. 2016, 133, 43287. [Google Scholar] [CrossRef]
- Hettal, S.; Roland, S.; Sipilä, K.; Joki, H.; Colin, X. A new analytical model for predicting the radio-thermal oxidation kinetics and the lifetime of electric cable insulation in nuclear power plants: Application to silane cross-linked polyethylene. Polym. Degrad. Stab. 2021, 185, 109492. [Google Scholar] [CrossRef]
- Colin, X.; Monchy-Leroy, C.; Verdu, J. Effect of gamma irradiation on tensile properties of low molecular weight polyethylene samples. Radiat. Phys. Chem. 2011, 80, 895–901. [Google Scholar] [CrossRef][Green Version]
- Peltzer, M.; Wagner, J.R.; Jiménez, A. Thermal characterization of UHWMPE stabilized with natural antioxidants. J. Therm. Anal. Calorim. 2007, 87, 493–497. [Google Scholar] [CrossRef]
- Busolo, M.A.; Lagaron, J.M. Antioxidant polyethylene films based on a resveratrol containing Clay of Interest in Food Packaging Applications. Food Packag. Shelf Life 2015, 6, 30–41. [Google Scholar] [CrossRef]
- Kabir, A.S.; Li, H.; Yuan, H.; Kuboki, T.; Xu, C. Effects of de-polymerized lignin content on thermo-oxidative and thermal stability of polyethylene. J. Anal. Appl. Pyrolysis 2019, 140, 413–422. [Google Scholar] [CrossRef]
- Westerhout, R.W.J.; Waanders, J.; Kuipers, J.A.M.; van Swaaij, W.P.M. Kinetics of the Low-Temperature Pyrolysis of Polyethylene, Polypropene, and Polystyrene Modeling, Experimental Determination, and Comparison with Literature Models and Data. Ind. Eng. Chem. Res. 1997, 36, 1955–1964. [Google Scholar] [CrossRef]
- Snegirev, A.Y.; Talalov, V.A.; Stepanov, V.V.; Korobeinichev, O.P.; Gerasimov, I.E. Autocatalysis in thermal decomposition of polymers. Polym. Degrad. Stab. 2017, 137, 151–161. [Google Scholar] [CrossRef]




| Aging Time d | Mw,chains g/mol | A 1/s | E kJ/mol | rsc @ 450 °C mol/L/s | ρxl mol/L | S |
|---|---|---|---|---|---|---|
| 0 | 5630 | 1.00 × 1016 | 276 | 1.09 × 10−4 | 0.195 | 0 |
| 42 | 2390 | 1.16 × 1015 | 262 | 1.45 × 10−4 | 0.283 | 0.0067 |
| 84 | 1500 | 1.16 × 1015 | 262 | 1.45 × 10−4 | 0.372 | 0.0137 |
| 126 | 1220 | 1.16 × 1015 | 262 | 1.45 × 10−4 | 0.460 | 0.0180 |
| 168 | 903 | 1.16 × 1015 | 262 | 1.45 × 10−4 | 0.548 | 0.0260 |
| 210 | 724 | 1.16 × 1015 | 262 | 1.45 × 10−4 | 0.637 | 0.0337 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verho, T.; Vaari, J. Analytical and Numerical Modeling of Degradation and Pyrolysis of Polyethylene: Measuring Aging with Thermogravimetry. Polymers 2022, 14, 2709. https://doi.org/10.3390/polym14132709
Verho T, Vaari J. Analytical and Numerical Modeling of Degradation and Pyrolysis of Polyethylene: Measuring Aging with Thermogravimetry. Polymers. 2022; 14(13):2709. https://doi.org/10.3390/polym14132709
Chicago/Turabian StyleVerho, Tuukka, and Jukka Vaari. 2022. "Analytical and Numerical Modeling of Degradation and Pyrolysis of Polyethylene: Measuring Aging with Thermogravimetry" Polymers 14, no. 13: 2709. https://doi.org/10.3390/polym14132709
APA StyleVerho, T., & Vaari, J. (2022). Analytical and Numerical Modeling of Degradation and Pyrolysis of Polyethylene: Measuring Aging with Thermogravimetry. Polymers, 14(13), 2709. https://doi.org/10.3390/polym14132709






