Current Trends in Biomedical Hydrogels: From Traditional Crosslinking to Plasma-Assisted Synthesis
Abstract
1. Introduction
2. Hydrogels as Biomaterials
3. Evolution of Hydrogels
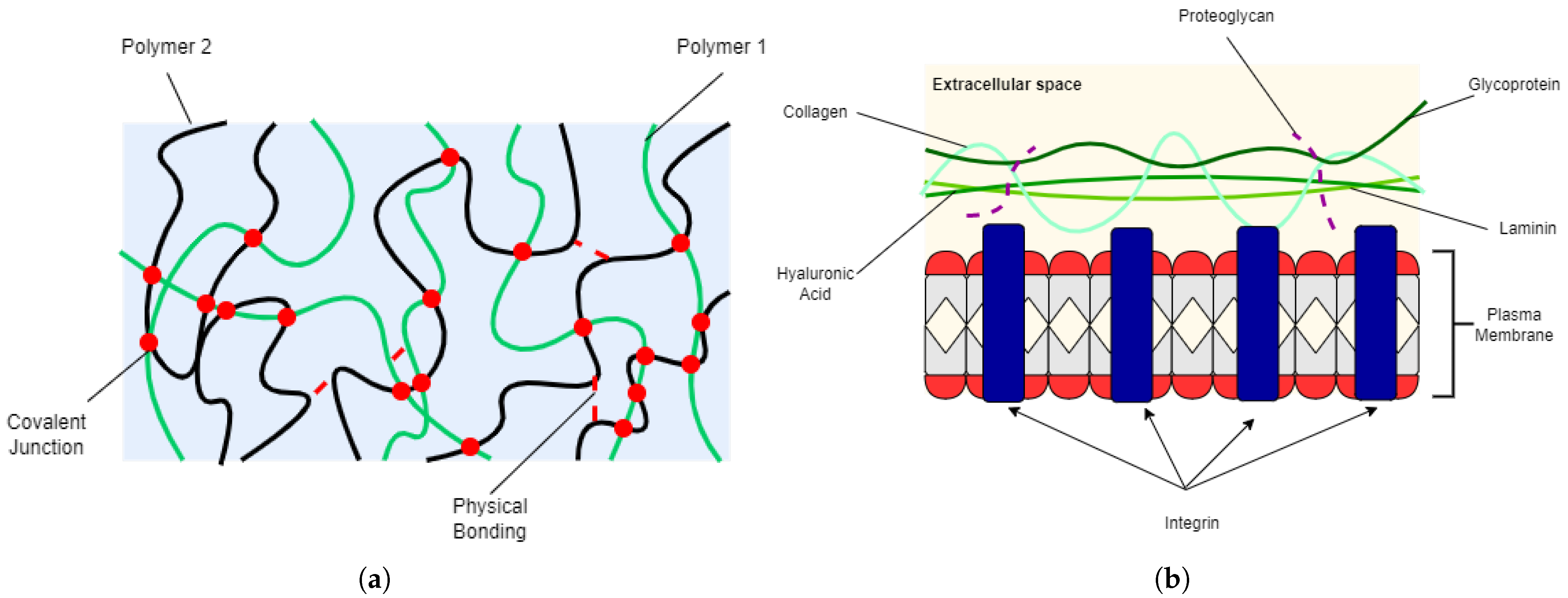
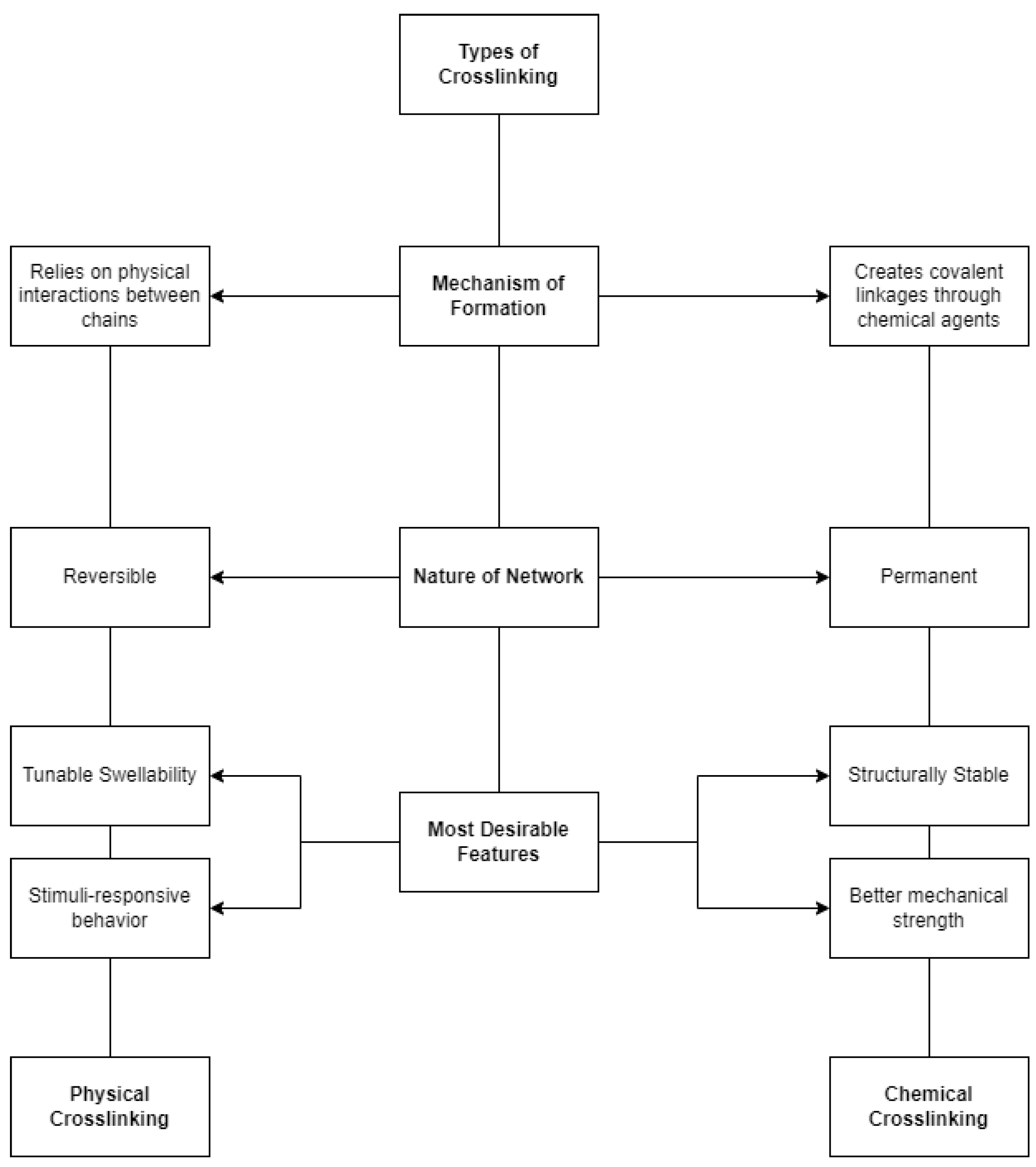
4. Hydrogel Crosslinking Strategies
4.1. Physical Crosslinking in Hydrogels
4.1.1. Hydrogen Bonding
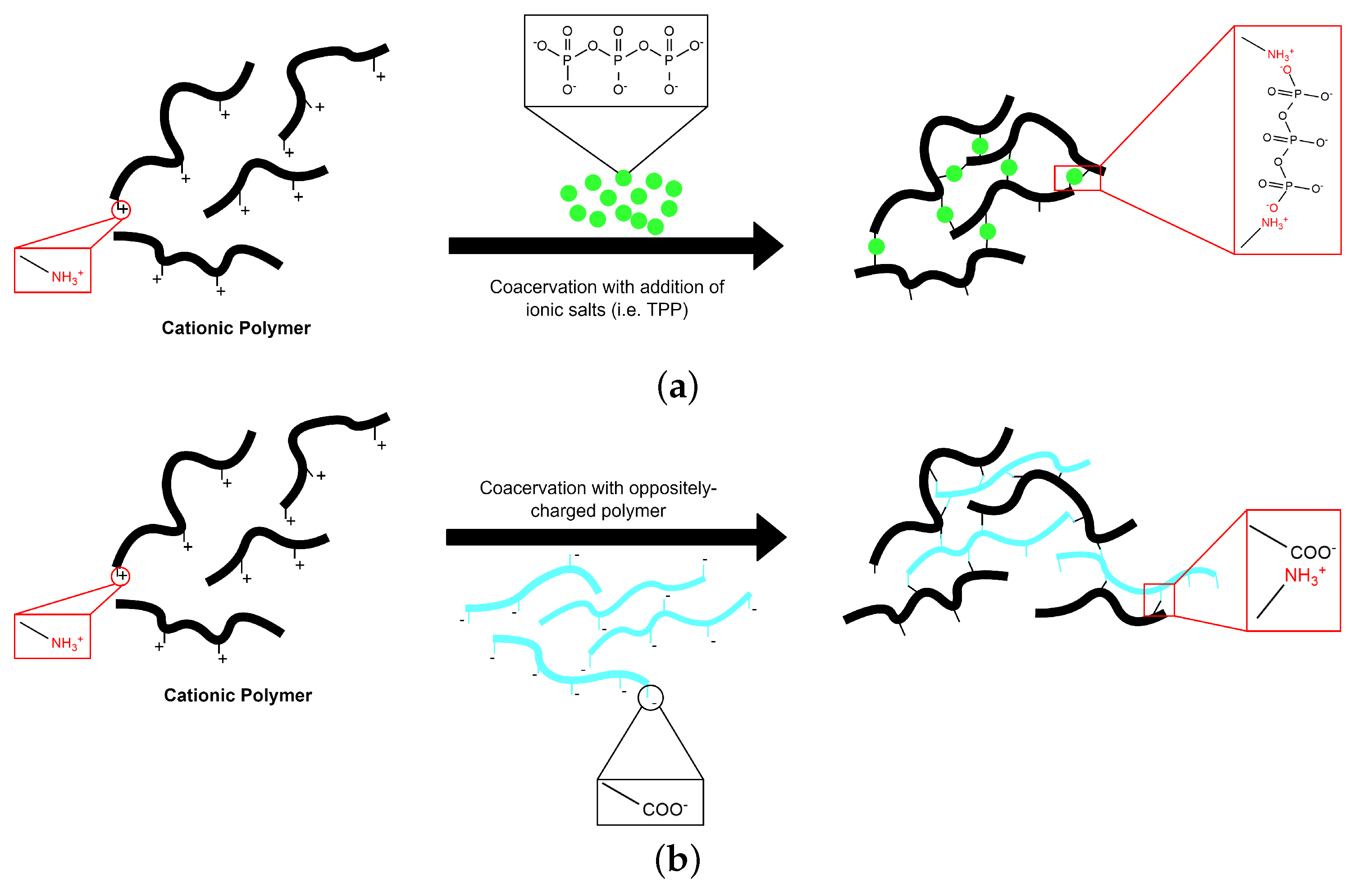
4.1.2. Coacervation Process
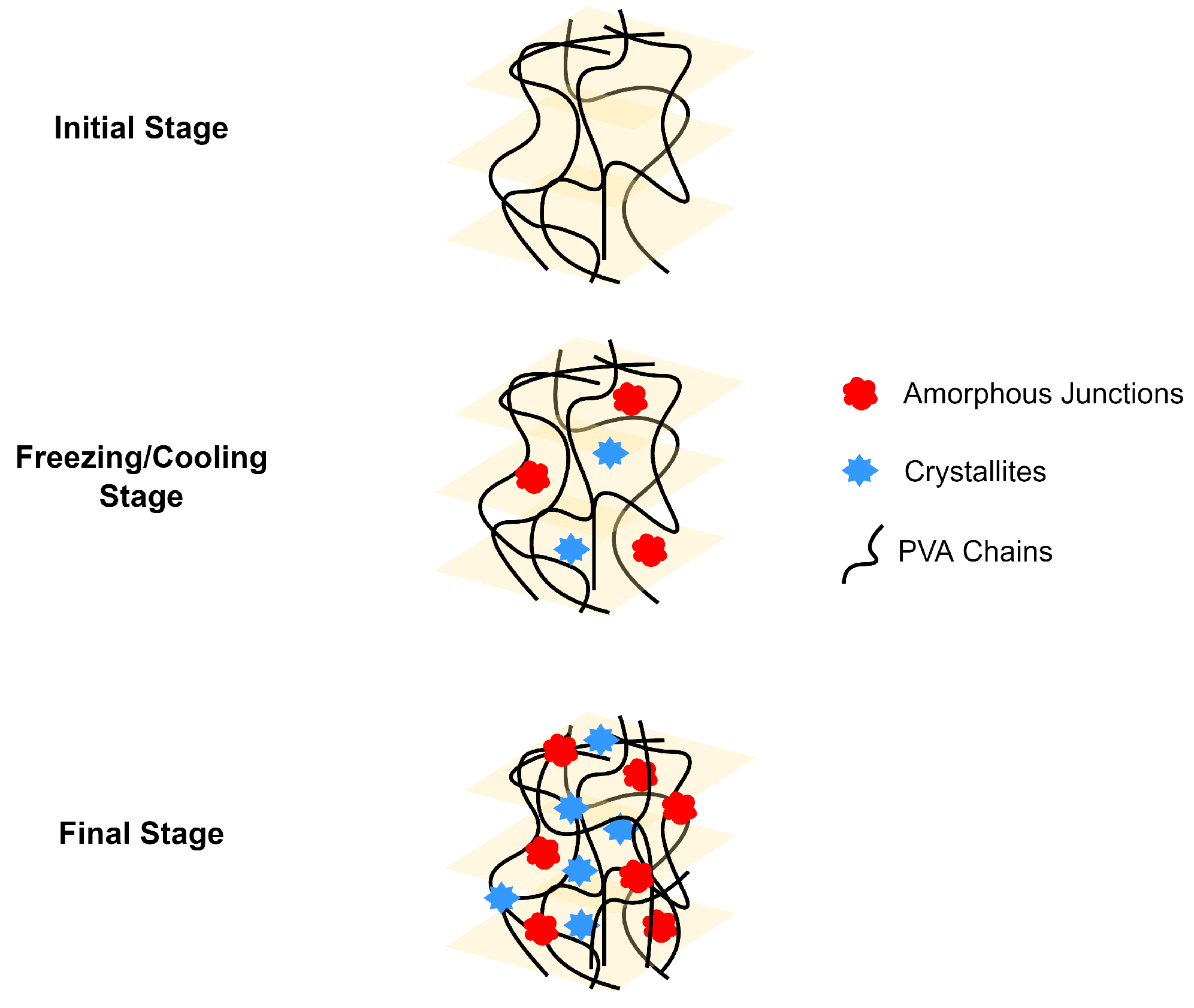
4.1.3. Heating or Cooling a Polymer Solution
4.1.4. Crosslinking by Crystallization
4.2. Chemical Crosslinking in Hydrogels
4.2.1. Free Radical Polymerization
4.2.2. Photopolymerization
4.2.3. Crosslinking Induced by Enzymatic Reactions
| Hydrogel | Application | Reference |
|---|---|---|
| Carboxymethylated pullulanchondroitin sulfate | Cartilage scaffold | [80] |
| Polyphenol-epigallocatechin gallates | Tissue adhesive | [81] |
| Tyramine-silk fibroin | Cell delivery | [82] |
| Agarose-chitosan | Biocatalysis | [83,85] |
4.2.4. Crosslinking by “Click Chemistry”
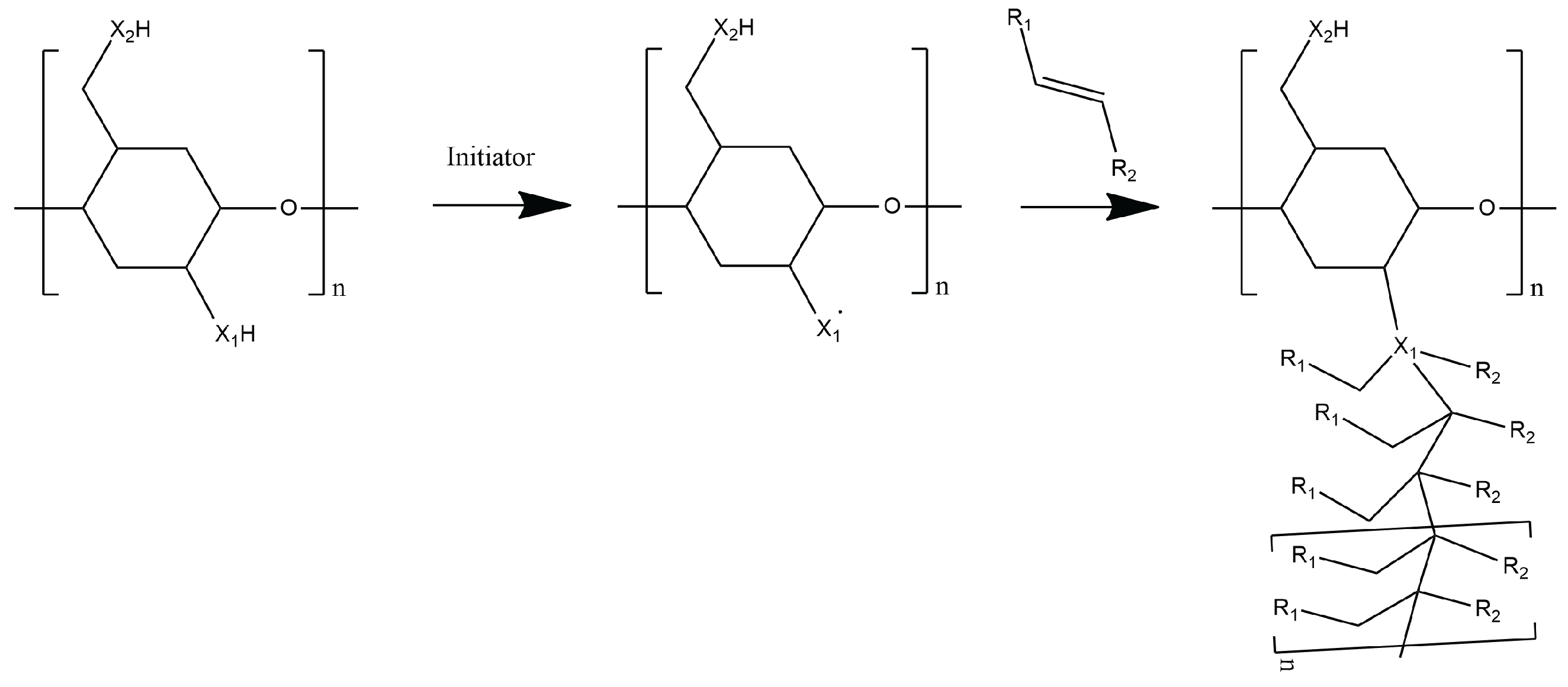
4.2.5. Grafting
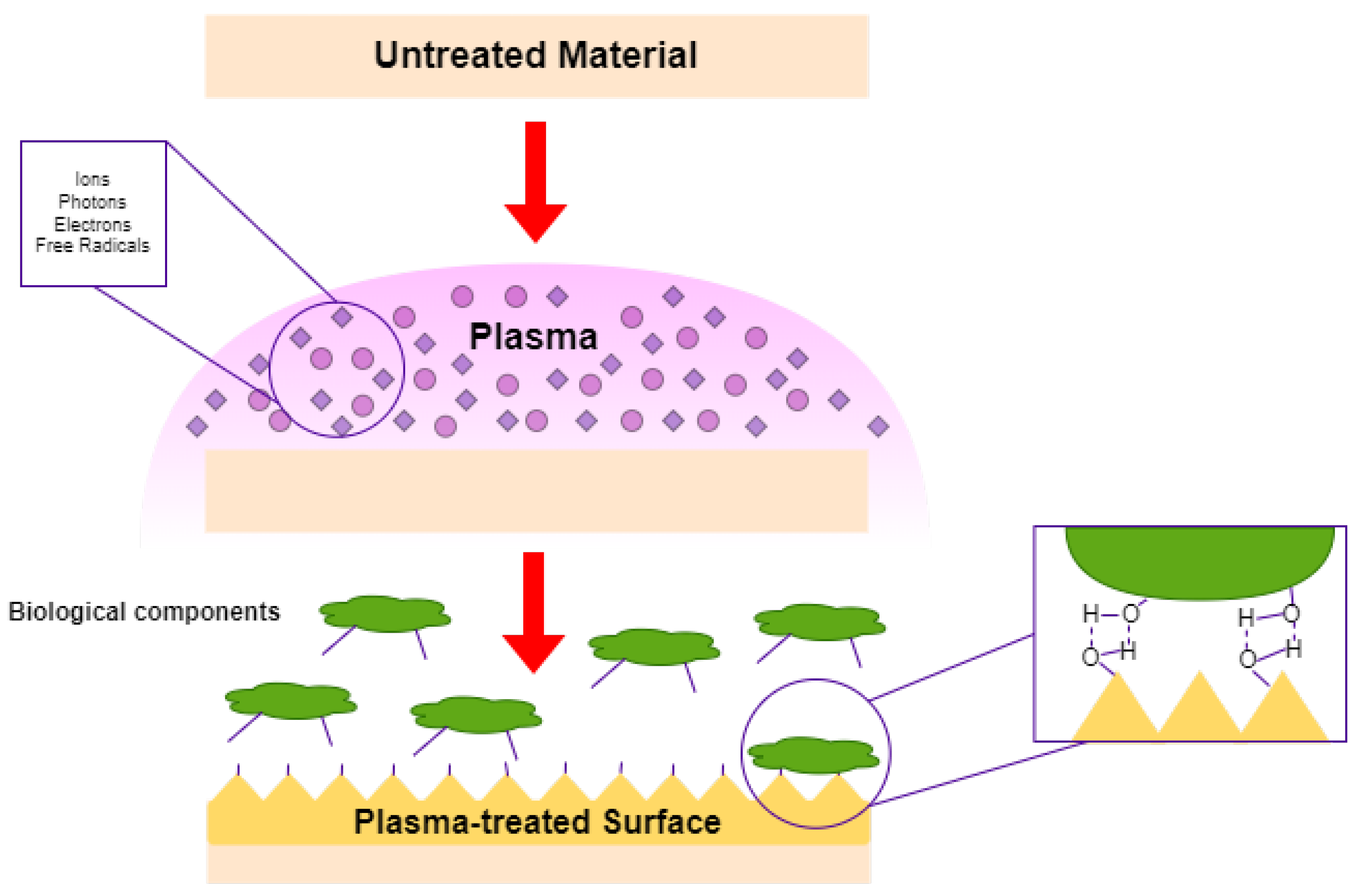
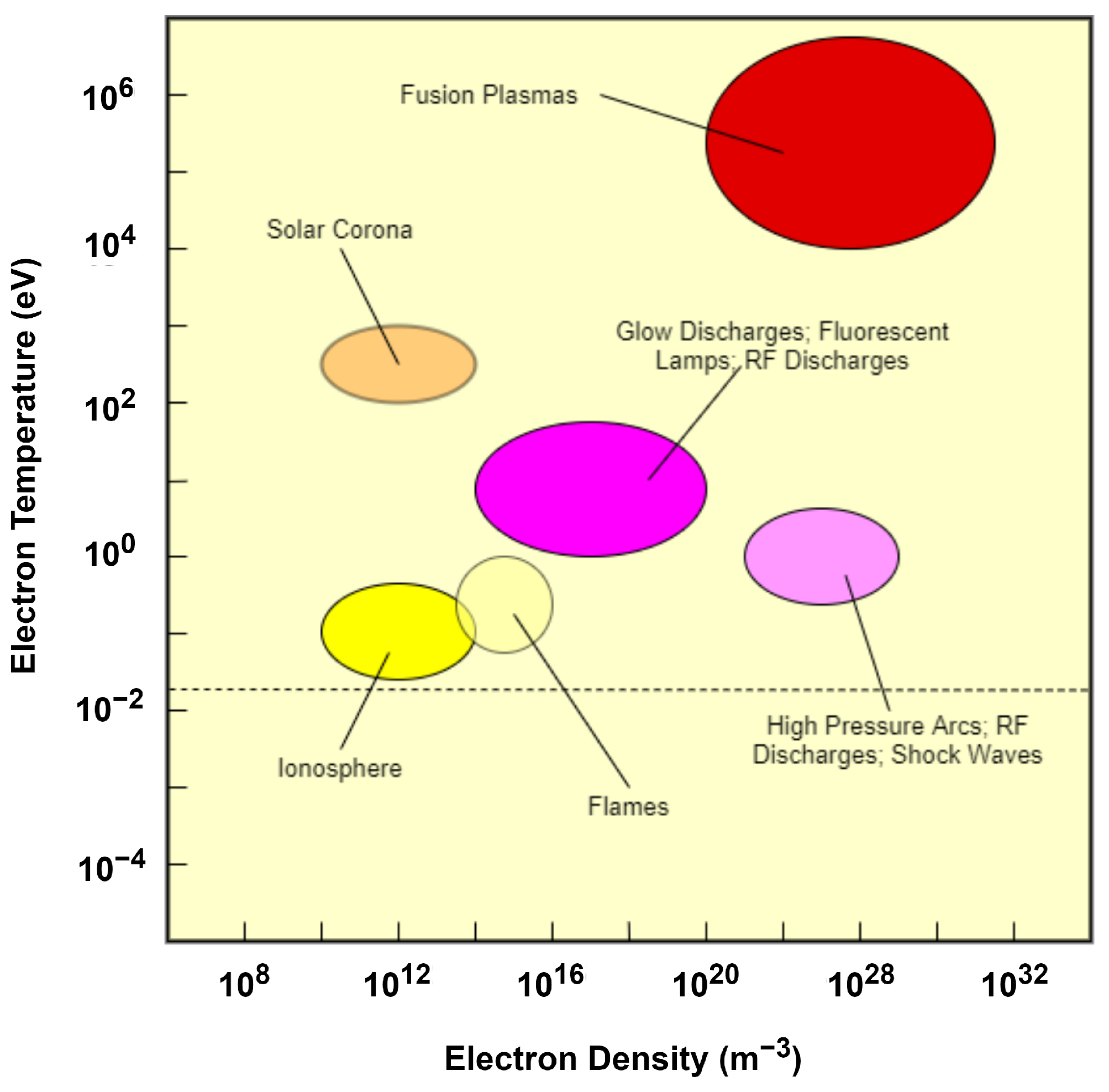
5. Plasma–Material Interactions
6. Surface Modification by Plasma Technology
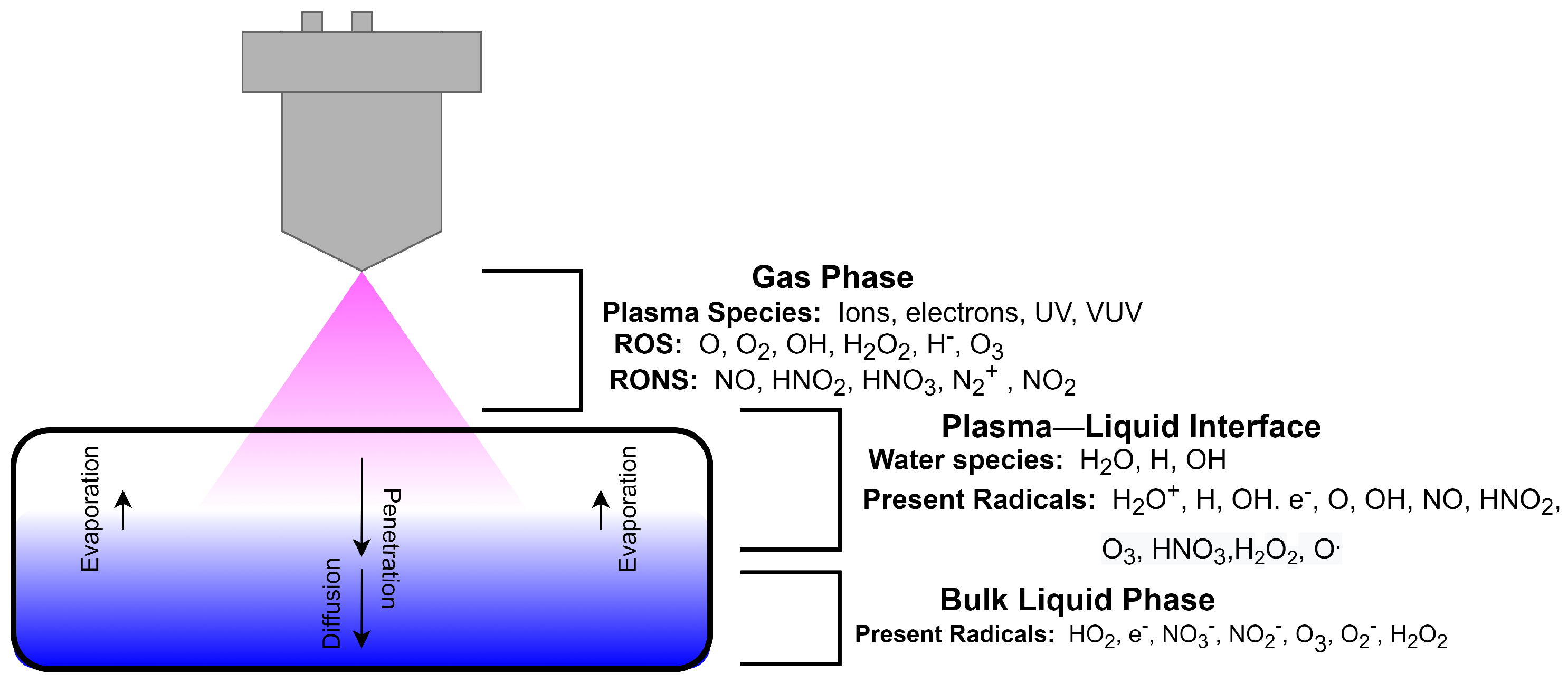
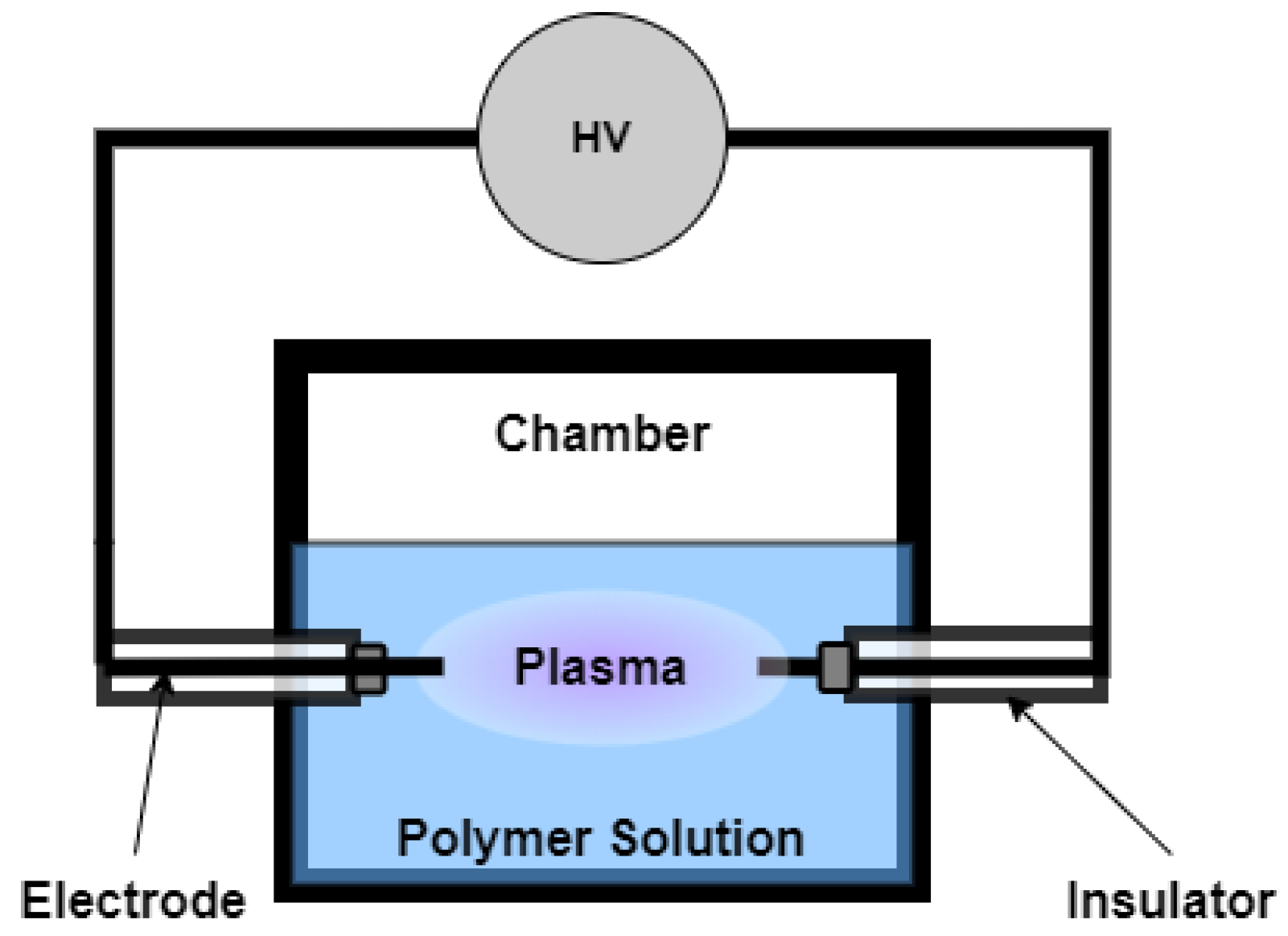
7. Early Use of Plasma Treatment in Liquid Solutions
8. Mechanisms in a Plasma-Assisted Hydrogel Synthesis
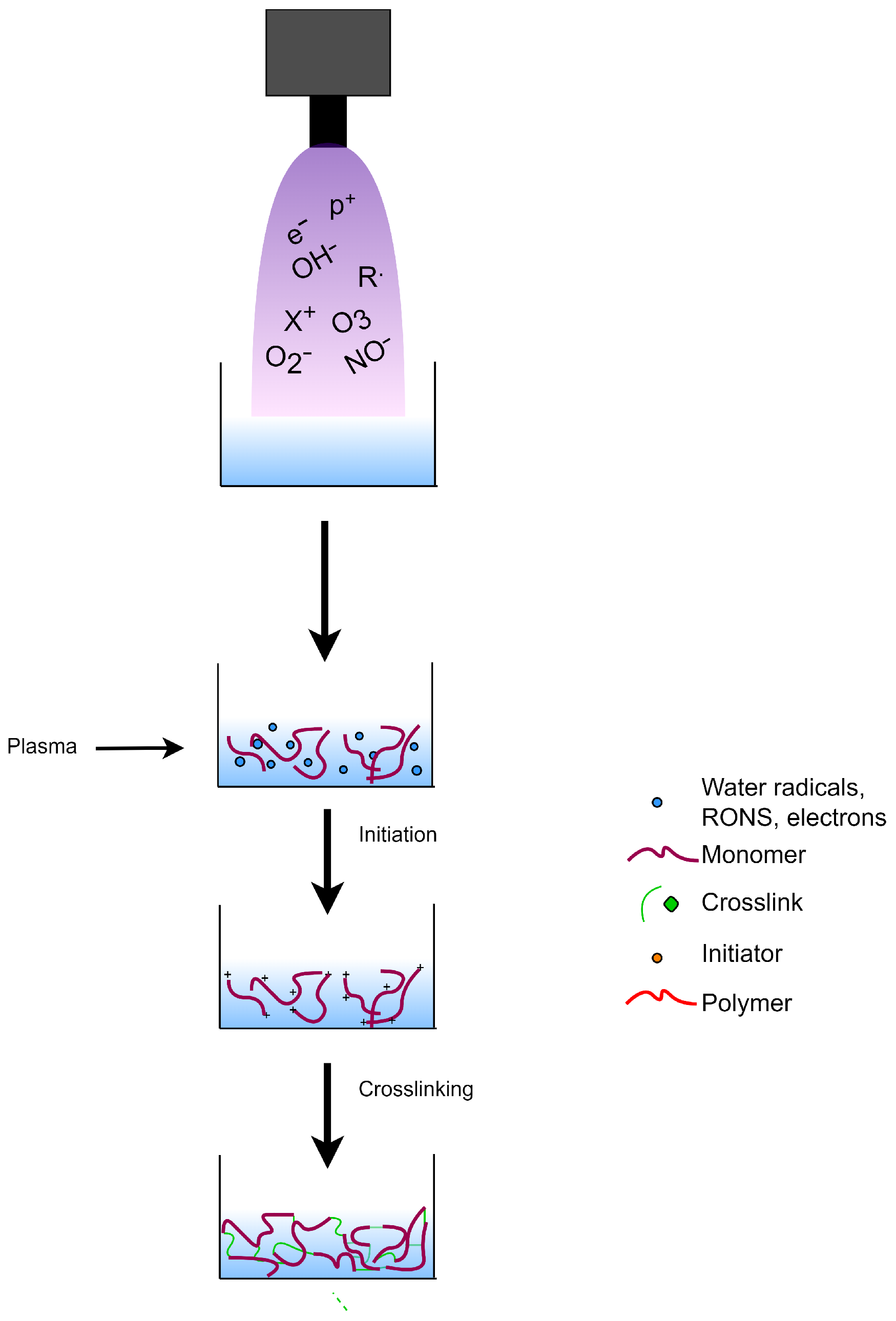
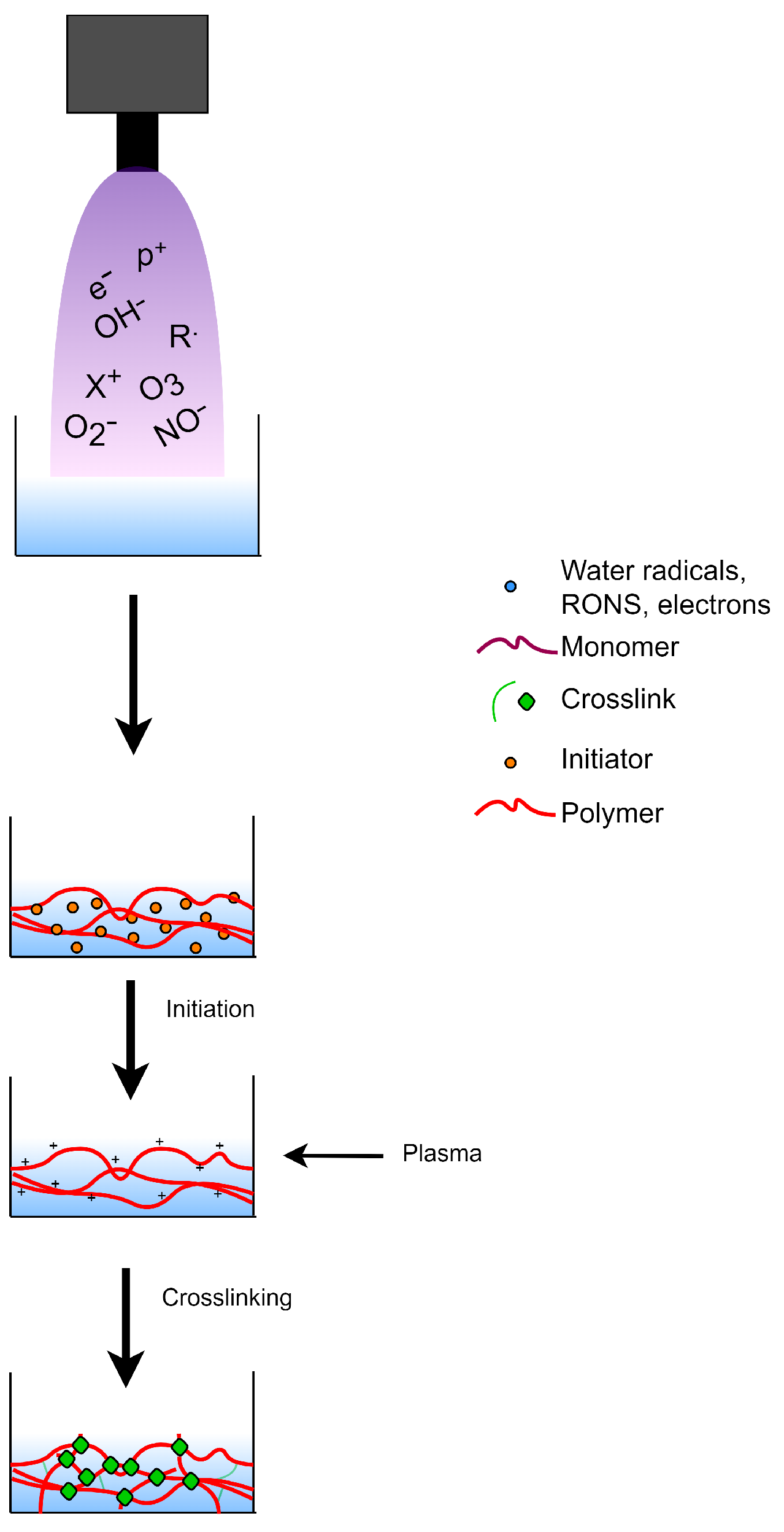
8.1. Plasma-Initiated Polymerization
8.2. Plasma-Induced Crosslinking
9. Roles of Plasma-Assisted Hydrogel Biomaterials
10. Perspectives
10.1. Future Direction of Hydrogels
10.2. Plasma-Synthesized Hydrogels
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ratner, B.D.; Hoffman, A.S.; Schoen, F.J.; Lemons, J.E. (Eds.) Biomaterials Science: An Introduction to Materials in Medicine, 3rd ed.; Academic Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Hasirci, V.; Hasirci, N. (Eds.) Fundamentals of Biomaterials; Springer Science+Business Media, LLC.: New York, NY, USA, 2009. [Google Scholar] [CrossRef]
- Patel, A.; Mequanint, K. Hydrogel Biomaterials. In Biomedical Engineering; Fazel-Rezai, R., Ed.; IntechOpen: Rijeka, Croatia, 2011; Chapter 14. [Google Scholar] [CrossRef]
- Rogero, S.O.; Malmonge, S.M.; Lugão, A.B.; Ikeda, T.I.; Miyamaru, L.; Cruz, Á.S. Biocompatibility study of polymeric biomaterials. Artif. Organs 2003, 27, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Rickert, D.; Lendlein, A.; Peters, I.; Moses, M.A.; Franke, R.P. Biocompatibility testing of novel multifunctional polymeric biomaterials for tissue engineering applications in head and neck surgery: An overview. Eur. Arch. Oto-Rhino-Laryngol. 2006, 263, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Murphy, M.; Li, C.; Ting, K.; Soo, C.; Zheng, Z. Current development of biodegradable polymeric materials for biomedical applications. Drug Des. Dev. Ther. 2018, 12, 3117–3145. [Google Scholar] [CrossRef] [PubMed]
- Francis PJ, J. Biomedical Applications of Polymers—An Overview. Curr. Trends Biomed. Eng. Biosci. 2018, 15, 44–45. [Google Scholar] [CrossRef]
- Bahram, M.; Mohseni, N.; Moghtader, M. An Introduction to Hydrogels and Some Recent Applications. Emerg. Concepts Anal. Appl. Hydrogels 2016. [Google Scholar] [CrossRef]
- Singh, G.; Lohani, A.; Bhattacharya, S. Niosome as a novel drug delivery system a review. J. Fundam. Pharm. Res. 2014, 2, 35–48. [Google Scholar]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Hydrogels as Drug Delivery Systems; Pros and Cons. Trends Pharm. Sci. 2019, 5, 7–24. [Google Scholar] [CrossRef]
- Utech, S.; Boccaccini, A.R. A review of hydrogel-based composites for biomedical applications: Enhancement of hydrogel properties by addition of rigid inorganic fillers. J. Mater. Sci. 2015, 51, 271–310. [Google Scholar] [CrossRef]
- Choudhury, A.J.; Gogoi, D.; Kandimalla, R.; Kalita, S.; Chaudhari, Y.B.; Khan, M.R.; Kotoky, J.; Chutia, J. Penicillin impregnation on oxygen plasma surface functionalized chitosan/Antheraea assama silk fibroin: Studies of antibacterial activity and antithrombogenic property. Mater. Sci. Eng. C 2016, 60, 475–484. [Google Scholar] [CrossRef]
- Ma, F.; Wang, Z.; Zhao, H.; Tian, S. Plasma depolymerization of chitosan in the presence of hydrogen peroxide. Int. J. Mol. Sci. 2012, 13, 7788–7797. [Google Scholar] [CrossRef]
- Geyter, N.D.; Morent, R. Non-Thermal Plasma Surface Modification of Biodegradable Polymers. In Biomedical Science, Engineering and Technology; Ghista, D., Ed.; InTech: London, UK, 2012. [Google Scholar]
- Slepička, P.; Kasálková, N.S.; Stránská, E.; Bačáková, L.; Švorčík, V. Surface characterization of plasma treated polymers for applications as biocompatible carriers. Express Polym. Lett. 2013, 7, 535–545. [Google Scholar] [CrossRef]
- Prasertsung, I.; Damrongsakkul, S.; Saito, N. Crosslinking of a gelatin solutions induced by pulsed electrical discharges in solutions. Plasma Process. Polym. 2013, 10, 792–797. [Google Scholar] [CrossRef]
- Valerio, J.K.C.; Nakajima, H.; Vasquez, M.R. Grafting of acrylic acid onto microwave plasma-treated polytetrafluoroethylene (PTFE) substrates. Jpn. J. Appl. Phys. 2018, 58, SAAC02. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; Nakajima, H.; Thumanu, K.; Janphuang, P.; Chanlek, N.; Vasquez, M.R. Spectroscopic studies of plasma-modified silver-exchanged zeolite and chitosan composites. Mater. Chem. Phys. 2020, 250, 122980. [Google Scholar] [CrossRef]
- Lao, T.L.; Cordura, S.; Diaz, L.J.; Vasquez, M. Influence of plasma treatment on the dissolution of cellulose in lithium chloride–dimethylacetamide. Cellulose 2020, 27, 9801–9811. [Google Scholar] [CrossRef]
- Liguori, A.; Bigi, A.; Colombo, V.; Focarete, M.L.; Gherardi, M.; Gualandi, C.; Oleari, M.C.; Panzavolta, S. Atmospheric Pressure Non-Equilibrium Plasma as a Green Tool to Crosslink Gelatin Nanofibers. Sci. Rep. 2016, 6, 38542. [Google Scholar] [CrossRef]
- Wichterle, O.; Lim, D. Hydrophilic Gels for Biological Use. Nature 1960, 185, 117–118. [Google Scholar] [CrossRef]
- Kopeček, J. Hydrogel biomaterials: A smart future? Biomaterials 2007, 28, 5185–5192. [Google Scholar] [CrossRef]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for Biomedical Applications: Their Characteristics and the Mechanisms behind Them. Gels 2017, 3, 6. [Google Scholar] [CrossRef]
- Li, X.; Sun, Q.; Li, Q.; Kawazoe, N.; Chen, G. Functional hydrogels with tunable structures and properties for tissue engineering applications. Front. Chem. 2018, 6, 1–20. [Google Scholar] [CrossRef]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Mohite, P.B.; Adhav, S.S. A hydrogels: Methods of preparation and applications. Int. J. Adv. Pharm. 2017, 06, 79–85. [Google Scholar]
- Nguyen, K.T.; West, J.L. Photopolymerizable hydrogels for tissue engineering applications. Biomaterials 2002, 23, 4307–4314. [Google Scholar] [CrossRef]
- Yang, J.; Sun, X.; Zhang, Y.; Chen, Y. The Application of Natural Polymer-Based Hydrogels in Tissue Engineering; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 273–307. [Google Scholar] [CrossRef]
- Saludas, L.; Pascual-Gil, S.; Prósper, F.; Garbayo, E.; Blanco-Prieto, M. Hydrogel based approaches for cardiac tissue engineering. Int. J. Pharm. 2017, 523, 454–475. [Google Scholar] [CrossRef]
- Lee, S.C.; Kwon, I.K.; Park, K. Hydrogels for Delivery of Bioactive Agents: A Historical Perspective. Adv. Drug Deliv. Rev. 2013, 65, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Van Bemmelen, J. Das hydrogel und das krystallinische hydrat des kupferoxyds. Z. Anorg. Chem. 1894, 5, 466–483. [Google Scholar] [CrossRef]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Azzahari, A.D.; Selvanathan, V.; Sonsudin, F.; Abouloula, C.N. pH sensitive hydrogels in drug delivery: Brief history, properties, swelling, and release mechanism, material selection and applications. Polymers 2017, 9, 137. [Google Scholar] [CrossRef]
- Buwalda, S.J.; Boere, K.W.; Dijkstra, P.J.; Feijen, J.; Vermonden, T.; Hennink, W.E. Hydrogels in a historical perspective: From simple networks to smart materials. J. Control. Release 2014, 190, 254–273. [Google Scholar] [CrossRef]
- Thakur, S.; Thakur, V.K.; Arotiba, O.A. Hydrogels: Recent Advances; Springer: Singapore, 2018; pp. 29–50. [Google Scholar]
- Yahia, L. History and Applications of Hydrogels. J. Biomed. Sci. 2015, 4, 1–23. [Google Scholar] [CrossRef]
- Mathur, A.M.; Moorjani, S.K.; Scranton, A.B. Methods for synthesis of hydrogel networks: A review. J. Macromol. Sci. Rev. Macromol. Chem. Phys. 1996, 36, 405–430. [Google Scholar] [CrossRef]
- Kuhn, W.; Hargitay, B.; Katchalsky, A.; Eisenberg, H. Reversible Dilation and Contraction by changing the state of ionization of high-polymer acid networks. Nature 1950, 165, 514–516. [Google Scholar] [CrossRef]
- Pauling, L.; Corey, R.B. Two Rippled-Sheet Configurations of Polypeptide Chains, and a Note about the Pleated Sheets. Proc. Natl. Acad. Sci. USA 1953, 39, 253–256. [Google Scholar] [CrossRef] [PubMed]
- De Jong, S.J.; Van Dijk-Wolthuis, W.N.; Kettenes-Van Den Bosch, J.J.; Schuyl, P.J.; Hennink, W.E. Monodisperse enantiomeric lactic acid oligomers: Preparation, characterization, and stereocomplex formation. Macromolecules 1998, 31, 6397–6402. [Google Scholar] [CrossRef]
- Peppas, N.A.; Hoffman, A.S. 1.3.2E—Hydrogels. In Biomaterials Science, 4th ed.; Wagner, W.R., Sakiyama-Elbert, S.E., Zhang, G., Yaszemski, M.J., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 153–166. [Google Scholar] [CrossRef]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef] [PubMed]
- Chuang, C.H.; Lin, R.Z.; Melero-Martin, J.M.; Chen, Y.C. Comparison of covalently and physically cross-linked collagen hydrogels on mediating vascular network formation for engineering adipose tissue. Artif. Cells Nanomed. Biotechnol. 2018, 46, S434–S447. [Google Scholar] [CrossRef]
- Echalier, C.; Valot, L.; Martinez, J.; Mehdi, A.; Subra, G. Chemical cross-linking methods for cell encapsulation in hydrogels. Mater. Today Commun. 2019, 20, 100536. [Google Scholar] [CrossRef]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart hydrogels in tissue engineering and regenerative medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef]
- Zhou, Q.; Dong, L.; Wu, J.; Shi, Y.; Feng, X.; Lu, X.; Zhu, J.; Mu, L. Versatile Ionic Gel Driven by Dual Hydrogen Bond Networks: Toward Advanced Lubrication and Self-Healing. ACS Appl. Polym. Mater. 2021, 3, 5932–5941. [Google Scholar] [CrossRef]
- Singh, S.K.; Dhyani, A.; Juyal, D. Hydrogel: Preparation, characterization, and applications. Pharma Innov. J. 2017, 6, 25–32. [Google Scholar] [CrossRef]
- Luo Zheng, L.; Vanchinathan, V.; Dalal, R.; Noolandi, J.; Waters, D.J.; Hartmann, L.; Cochran, J.R.; Frank, C.W.; Yu, C.Q.; Ta, C.N. Biocompatibility of poly(ethylene glycol) and poly(acrylic acid) interpenetrating network hydrogel by intrastromal implantation in rabbit cornea. J. Biomed. Mater. Res.-Part A 2015, 103, 3157–3165. [Google Scholar] [CrossRef]
- Elsayed, M.M. Hydrogel Preparation Technologies: Relevance Kinetics, Thermodynamics and Scaling up Aspects. J. Polym. Environ. 2019, 27, 871–891. [Google Scholar] [CrossRef]
- You, Y.; Yang, J.; Zheng, Q.; Wu, N.; Lv, Z.; Jiang, Z. Ultra-stretchable hydrogels with hierarchical hydrogen bonds. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Sun, Y.; Zhu, J.; Xu, J.; Zhang, C.; Liu, T. Hydrogen-bonded network enables polyelectrolyte complex hydrogels with high stretchability, excellent fatigue resistance and self-healability for human motion detection. Compos. Part B Eng. 2021, 217. [Google Scholar] [CrossRef]
- Timilsena, Y.P.; Akanbi, T.O.; Khalid, N.; Adhikari, B.; Barrow, C.J. Complex coacervation: Principles, mechanisms and applications in microencapsulation. Int. J. Biol. Macromol. 2019, 121, 1276–1286. [Google Scholar] [CrossRef] [PubMed]
- Nairm, J. 3 Coacervation-phase separation technology. In Advances in Pharmaceutical Sciences; Ganderton, D., Jones, T., McGinity, J., Eds.; Academic Press: Cambridge, MA, USA, 1995; Volume 7, pp. 93–219. [Google Scholar] [CrossRef]
- Sacco, P.; Furlani, F.; de Marzo, G.; Marsich, E.; Paoletti, S.; Donati, I. Concepts for Developing Physical Gels of Chitosan and of Chitosan Derivatives. Gels 2018, 4, 67. [Google Scholar] [CrossRef]
- Madhumitha, G.; Fowsiya, J.; Roopan, S.M. Emerging Technology in Medical Applications for Hydrogel. In Hydrogels, Gels Horizons: From Science to Smart Materials; Thakur, V., Thakur, M., Eds.; Springer: Singapore, 2018; Chapter 8; pp. 29–50. [Google Scholar] [CrossRef]
- Leonard, M.; De Boisseson, M.R.; Hubert, P.; Dalençon, F.; Dellacherie, E. Hydrophobically modified alginate hydrogels as protein carriers with specific controlled release properties. J. Control. Release 2004, 98, 395–405. [Google Scholar] [CrossRef]
- Sun, P.; Li, P.; Li, Y.M.; Wei, Q.; Tian, L.H. A pH-sensitive chitosan-tripolyphosphate hydrogel beads for controlled glipizide delivery. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 97B, 175–183. [Google Scholar] [CrossRef]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Peppas, N.A.; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur. J. Pharm. Biopharm. 2004, 57, 19–34. [Google Scholar] [CrossRef]
- Argin, S.; Kofinas, P.; Lo, Y.M. The cell release kinetics and the swelling behavior of physically crosslinked xanthan-chitosan hydrogels in simulated gastrointestinal conditions. Food Hydrocoll. 2014, 40, 138–144. [Google Scholar] [CrossRef]
- Chu, C.H.; Sakiyama, T.; Yano, T. pH-Sensitive Swelling of a Polyelectrolyte Complex Gel Prepared from Xanthan and Chitosan. Biosci. Biotechnol. Biochem. 1995, 59, 717–719. [Google Scholar] [CrossRef]
- Malik, N.S.; Ahmad, M.; Minhas, M.U.; Tulain, R.; Barkat, K.; Khalid, I.; Khalid, Q. ChitosanXanthan Gum Based Hydrogels as Potential Carrier for an Antiviral Drug: Fabrication, Characterization, and Safety Evaluation. Front. Chem. 2020, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Varghese, J.S.; Chellappa, N.; Fathima, N.N. Gelatin–carrageenan hydrogels: Role of pore size distribution on drug delivery process. Colloids Surfaces B Biointerfaces 2014, 113, 346–351. [Google Scholar] [CrossRef]
- Voron, N.G.; Derkach, S.R.; Vovk, M.A.; Tolstoy, P.M. Formation of κ-carrageenan—Gelatin polyelectrolyte complexes studied by 1 H NMR, UV spectroscopy and kinematic viscosity measurements. Carbohydr. Polym. 2016, 151, 1152–1161. [Google Scholar] [CrossRef]
- Mao, H.; Wang, C.; Chang, X.; Cao, H.; Shan, G.; Bao, Y.; Pan, P. Poly(lactic acid)/poly(ethylene glycol) stereocomplexed physical hydrogels showing thermally-induced gel-sol-gel multiple phase transitions. Mater. Chem. Front. 2018, 2, 313–322. [Google Scholar] [CrossRef]
- Nakano, T.; Nakaoki, T. Coagulation size of freezable water in poly (vinyl alcohol) hydrogels formed by different freeze/thaw cycle periods. Polym. J. 2011, 43, 875–880. [Google Scholar] [CrossRef]
- Watase, M.; Nishinari, K. Effects of the Degree of Saponification and Concentration on the Thermal and Rheological Properties of Poly (vinyl alcohol)-Dimethyl Sulfoxide-Water Gels. Polymer 1989, 21, 567–575. [Google Scholar] [CrossRef][Green Version]
- Shapiro, Y.E. 1H NMR Self-Diffusion Study of Morphology and Structure of Polyvinyl Alcohol Cryogels. J. Colloid Interface Sci. 1999, 212, 453–465. [Google Scholar] [CrossRef]
- Ranganathan, N.; Joseph Bensingh, R.; Abdul Kader, M.; Nayak, S.K. Synthesis and Properties of Hydrogels Prepared by Various Polymerization Reaction Systems. In Cellulose-Based Superabsorbent Hydrogels; Mondal, M.I.H., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–25. [Google Scholar] [CrossRef]
- Pérez-Salinas, P.; Jaramillo-Soto, G.; Rosas-Aburto, A.; Vázquez-Torres, H.; Bernad-Bernad, M.J.; Licea-Claverie, Á.; Vivaldo-Lima, E. Comparison of polymer networks synthesized by conventional free radical and RAFT Copolymerization Processes In Supercritical Carbon Dioxide. Processes 2017, 5, 26. [Google Scholar] [CrossRef]
- Pooley, S.A.; Rivas, B.L.; Riquelme, F.J. Stimuli-responsive Hydrogels from Acrylamide with N-[3-Dimethylamine)Propyl] Methacrylamde: Synthesis and Properties. J. Chil. Chem. Soc. 2007, 52, 1160–1163. [Google Scholar] [CrossRef]
- Das, D.; Pham, T.T.H.; Noh, I. Characterizations of hyaluronate-based terpolymeric hydrogel synthesized via free radical polymerization mechanism for biomedical applications. Colloids Surf. B Biointerfaces 2018, 170, 64–75. [Google Scholar] [CrossRef]
- Campan, R.; Cazaux, F.; Coqueret, X. Controlled Swelling of Poly(hydroxyethyl methacrylate) Hydrogels by Photochemical Grafting of Hydrophobic Acrylates. Macromol. Mater. Eng. 2003, 287, 924–930. [Google Scholar] [CrossRef]
- Qavi, S.; Pourmahdian, S.; Eslami, H. Acrylamide Hydrogels Preparation via Free Radical Crosslinking Copolymerization: Kinetic Study and Morphological Investigation. J. Macromol. Sci. Part A 2014, 51, 842–848. [Google Scholar] [CrossRef]
- Hasan, M.M.; Uddin, M.F.; Zabin, N.; Shakil, M.S.; Alam, M.; Achal, F.J.; Begum, M.H.A.; Hossen, M.S.; Hasan, M.A.; Morshed, M.M. Fabrication and Characterization of Chitosan-Polyethylene Glycol (Ch-Peg) Based Hydrogels and Evaluation of Their Potency in Rat Skin Wound Model. Int. J. Biomater. 2021, 2021. [Google Scholar] [CrossRef] [PubMed]
- Elisseeff, J.; McIntosh, W.; Anseth, K.; Riley, S.; Ragan, P.; Langer, R. Photoencapsulation of Chondrocytes in Poly(Ethylene Oxide)-Based Semi-Interpenetrating Networks; John Wiley & Sons: Hoboken, NJ, USA, 2000; pp. 164–171. [Google Scholar] [CrossRef]
- Ifkovits, J.L.; Burdick, J.A. Review: Photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 2007, 13, 2369–2385. [Google Scholar] [CrossRef] [PubMed]
- Nge, T.T.; Yamaguchi, M.; Hori, N.; Takemura, A.; Ono, H. Synthesis and characterization of chitosan/poly(acrylic acid) polyelectrolyte complex. J. Appl. Polym. Sci. 2002, 83, 1025–1035. [Google Scholar] [CrossRef]
- Mironi-Harpaz, I.; Wang, D.Y.; Venkatraman, S.; Seliktar, D. Photopolymerization of cell-encapsulating hydrogels: Crosslinking efficiency versus cytotoxicity. Acta Biomater. 2012, 8, 1838–1848. [Google Scholar] [CrossRef] [PubMed]
- Ritz, U.; Kögler, P.; Höfer, I.; Frank, P.; Klees, S.; Gebhard, S.; Brendel, C.; Kaufmann, K.; Hofmann, A.; Rommens, P.M.; et al. Photocrosslinkable polysaccharide hydrogel composites based on dextran or pullulan-amylose blends with cytokines for a human co-culture model of human osteoblasts and endothelial cells. J. Mater. Chem. B 2016, 4, 6552–6564. [Google Scholar] [CrossRef]
- Bae, H.; Ahari, A.F.; Shin, H.; Nichol, J.W.; Hutson, C.B.; Masaeli, M.; Kim, S.H.; Aubin, H.; Yamanlar, S.; Khademhosseini, A. Cell-laden microengineered pullulan methacrylate hydrogels promote cell proliferation and 3D cluster formation. Soft Matter 2011, 7, 1903–1911. [Google Scholar] [CrossRef]
- Chen, F.; Yu, S.; Liu, B.; Ni, Y.; Yu, C.; Su, Y.; Zhu, X.; Yu, X.; Zhou, Y.; Yan, D. An Injectable Enzymatically Crosslinked Carboxymethylated Pullulan/Chondroitin Sulfate Hydrogel for Cartilage Tissue Engineering. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, K.; Kim, B.S.; An, Y.H.; Lee, U.J.; Lee, S.H.; Kim, S.L.; Kim, B.G.; Hwang, N.S. Fabrication of polyphenol-incorporated anti-inflammatory hydrogel via high-affinity enzymatic crosslinking for wet tissue adhesion. Biomaterials 2020, 242, 119905. [Google Scholar] [CrossRef]
- Hasturk, O.; Jordan, K.E.; Choi, J.; Kaplan, D.L. Enzymatically crosslinked silk and silk-gelatin hydrogels with tunable gelation kinetics, mechanical properties and bioactivity for cell culture and encapsulation. Biomaterials 2020, 232, 119720. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, G.; Xu, X.; Abdou, P.; Jiang, Q.; Shi, D.; Gu, Z. Advances of injectable hydrogel-based scaffolds for cartilage regeneration. Regen. Biomater. 2019, 6, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Khanmohammadi, M.; Dastjerdi, M.B.; Ai, A.; Ahmadi, A.; Godarzi, A.; Rahimi, A.; Ai, J. Horseradish peroxidase-catalyzed hydrogelation for biomedical applications. Biomater. Sci. 2018, 6, 1286–1298. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Zhao, Y.; Iqbal, H.M. Agarose-chitosan hydrogel-immobilized horseradish peroxidase with sustainable bio-catalytic and dye degradation properties. Int. J. Biol. Macromol. 2019, 124, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Hein, C.D.; Liu, X.M.; Wang, D. Click chemistry, a powerful tool for pharmaceutical sciences. Pharm. Res. 2008, 25, 2216–2230. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Philip Ball. The click concept. In Chemistry World; Philip Ball: London, UK, 2007; pp. 46–51. [Google Scholar]
- Magli, S.; Rossi, G.B.; Risi, G.; Bertini, S.; Cosentino, C.; Crippa, L.; Ballarini, E.; Cavaletti, G.; Piazza, L.; Masseroni, E.; et al. Design and Synthesis of Chitosan—Gelatin Hybrid Hydrogels for 3D Printable in vitro Models. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Li, D.Q.; Wang, S.Y.; Meng, Y.J.; Guo, Z.W.; Cheng, M.M.; Li, J. Fabrication of self-healing pectin/chitosan hybrid hydrogel via Diels-Alder reactions for drug delivery with high swelling property, pH-responsiveness, and cytocompatibility. Carbohydr. Polym. 2021, 268. [Google Scholar] [CrossRef]
- Bashir, S.; Teo, Y.Y.; Naeem, S.; Ramesh, S.; Ramesh, K. pH responsive N-succinyl chitosan/Poly(acrylamide-co-acrylic acid) hydrogels and in vitro release of 5- fluorouracil. PLoS ONE 2017, 12, e0185505. [Google Scholar]
- Xu, C.; Zhan, W.; Tang, X.; Mo, F.; Fu, L.; Lin, B. Self-healing chitosan/vanillin hydrogels based on Schiff-base bond/hydrogen bond hybrid linkages. Polym. Test. 2018, 66, 155–163. [Google Scholar] [CrossRef]
- Grover, G.N.; Braden, R.L.; Christman, K.L. Oxime cross-linked injectable hydrogels for catheter delivery. Adv. Mater. 2013, 25, 2937–2942. [Google Scholar] [CrossRef]
- Hardy, J.G.; Lin, P.; Schmidt, C.E. Biodegradable hydrogels composed of oxime crosslinked poly(ethylene glycol), hyaluronic acid and collagen: A tunable platform for soft tissue engineering. J. Biomater. Sci. Polym. Ed. 2015, 26, 143–161. [Google Scholar] [CrossRef] [PubMed]
- Guaresti, O.; Basasoro, S.; González, K.; Eceiza, A.; Gabilondo, N. In situ cross–linked chitosan hydrogels via Michael addition reaction based on water–soluble thiol–maleimide precursors. Eur. Polym. J. 2019, 119, 376–384. [Google Scholar] [CrossRef]
- Quadrado, R.F.; Macagnan, K.L.; Moreira, A.S.; Fajardo, A.R. Chitosan-based hydrogel crosslinked through an aza-Michael addition catalyzed by boric acid. Int. J. Biol. Macromol. 2021, 193, 1032–1042. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Wang, Q.; Han, Y.; Tan, Y. Boronic ester-based self-healing hydrogels formed by using intermolecular B-N coordination. Polymer 2020, 202. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Wang, Q.; Han, Y.; Chen, H.; Tan, Y. Doubly dynamic hydrogel formed by combining boronate ester and acylhydrazone bonds. Polymers 2020, 12, 487. [Google Scholar] [CrossRef]
- Savina, I.N.; Mattiasson, B.O.; Galaev, I.Y.U. Graft Polymerization of Vinyl Monomers Inside Macroporous Polyacrylamide Gel, Cryogel, in Aqueous and Aqueous-Organic Media Initiated by Diperiodatocuprate (III) Complexes. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 1952–1963. [Google Scholar] [CrossRef]
- Sand, A.; Vyas, A. Introductory Chapter: Organic Polymer - Graft Copolymers. In Organic Polymers; Sand, A., Zaki, E., Eds.; IntechOpen: Rijeka, Croatia, 2020; Chapter 1. [Google Scholar] [CrossRef]
- Djordjevic, S.; Nikolic, L.; Kovacevic, S.; Miljkovic, M.; Djordjevic, D. Graft copolymerization of acrylic acid onto hydrolyzed potato starch using various initiators. Period. Polytech. Chem. Eng. 2013, 57, 55–61. [Google Scholar] [CrossRef]
- Xiang, J.; Shen, L.; Hong, Y. Status and future scope of hydrogels in wound healing: Synthesis, materials and evaluation. Eur. Polym. J. 2020, 130, 109609. [Google Scholar] [CrossRef]
- Gowda, D.V.; Koshy, T.; Godugu, K. Polymer Grafting-An Overview. Am. J. Pharmtech Res. 2016, 6. Available online: https://www.researchgate.net/publication/301521716_Polymer_Grafting-An_Overview (accessed on 26 May 2022).
- Sakhare, M.S.; Rajput, H.H. Polymer Grafting and Applications in Pharmaceutical Drug Delivery Systems—A Brief Review. Asian J. Pharm. Clin. Res. 2017, 10, 59–63. [Google Scholar] [CrossRef]
- Witono, J.R.; Noordergraaf, I.W.; Heeres, H.J.; Janssen, L.P. Graft copolymerization of acrylic acid to cassava starch—Evaluation of the influences of process parameters by an experimental design method. Carbohydr. Polym. 2012, 90, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Doba, T.; Rodehed, C.; Rånby, B. Mechanism of Graft Copolymerization onto Polysaccharides Initiated by Metal Ion Oxidation Reactions of Model Compounds for Starch and Cellulose. Macromolecules 1984, 17, 2512–2519. [Google Scholar] [CrossRef]
- Danial, E.N.; Hamza, A.H.; Mahmoud, R.H. Characteristics of immobilized urease on grafted alginate bead systems. Braz. Arch. Biol. Technol. 2015, 58, 147–153. [Google Scholar] [CrossRef]
- Soledad Lencina, M.; Iatridi, Z.; Villar, M.A.; Tsitsilianis, C. Thermoresponsive hydrogels from alginate-based graft copolymers. Eur. Polym. J. 2014, 61, 33–44. [Google Scholar] [CrossRef]
- Essawy, H.A.; Ghazy, M.B.; El-Hai, F.A.; Mohamed, M.F. Superabsorbent hydrogels via graft polymerization of acrylic acid from chitosan-cellulose hybrid and their potential in controlled release of soil nutrients. Int. J. Biol. Macromol. 2016, 89, 144–151. [Google Scholar] [CrossRef]
- Rop, K.; Mbui, D.; Njomo, N.; Karuku, G.N.; Michira, I.; Ajayi, R.F. Biodegradable water hyacinth cellulose-graft-poly(ammonium acrylate-co-acrylic acid) polymer hydrogel for potential agricultural application. Heliyon 2019, 5, e01416. [Google Scholar] [CrossRef]
- Harish Prashanth, K.V.; Tharanathan, R.N. Studies on graft copolymerization of chitosan with synthetic monomers. Carbohydr. Polym. 2003, 54, 343–351. [Google Scholar] [CrossRef]
- Marcasuzaa, P.; Reynaud, S.; Ehrenfeld, F.; Khoukh, A.; Desbrieres, J. Chitosan-graft-Polyaniline-Based Hydrogels: Elaboration and Properties. Biomacromolecules 2010, 11, 1684–1691. [Google Scholar] [CrossRef]
- Carlos, J.; St, Q.D.; Abundis-correa, V.; Herrera-flores, S.D.; Alvarez, A.J. pH-Sensitive Starch-Based Hydrogels: Synthesis and Effect of Molecular Components on Drug Release Behavior. Polymers 2020, 12, 1–14. [Google Scholar]
- Chapiro, A. Radiation Grafting of Hydrogels to Improve the Thrombo-resistance of Polymers. Eur. Polym. J. 1983, 19, 859–861. [Google Scholar] [CrossRef]
- Ratner, B.D.; Weathersby, P.K.; Hoffman, A.S.; Kelly, M.A.; Scharpen, L.H. Radiation-grafted hydrogels for biomaterial applications as studied by the ESCA technique. J. Appl. Polym. Sci. 1978, 22, 643–664. [Google Scholar] [CrossRef]
- Mozetič, M.; Vesel, A.; Primc, G.; Zaplotnik, R. Introduction to Plasma and Plasma Diagnostics. In Non-Thermal Plasma Technology for Polymeric Materials; Thomas, S., Mozetič, M., Cvelbar, U., Špatenka, P., Praveen, K.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Chapter 2; pp. 23–65. [Google Scholar] [CrossRef]
- Kulkarni, S. Plasma Assisted Polymer Synthesis and Processing; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 67–94. [Google Scholar] [CrossRef]
- Gilliam, M.A. A Plasma Polymerization Investigation and Low Temperature Cascade Arc Plasma for Polymeric Surface Modification. Ph.D. Thesis, University of Missouri-Columbia, Columbia, MO, USA, 2006. [Google Scholar]
- Latag, G.V.; Vasquez, M.R. Effects of RF plasma modification on the thermal and mechanical properties of electrospun chitosan/poly(vinyl alcohol) nanofiber mats. J. Vac. Sci. Technol. B 2018, 36, 04I101. [Google Scholar] [CrossRef]
- Bruggeman, P.J.; Kushner, M.J.; Locke, B.R.; Gardeniers, J.G.; Graham, W.G.; Graves, D.B.; Hofman-Caris, R.C.; Maric, D.; Reid, J.P.; Ceriani, E.; et al. Plasma-liquid interactions: A review and roadmap. Plasma Sources Sci. Technol. 2016, 25, 1–59. [Google Scholar] [CrossRef]
- Mariotti, D.; Patel, J.; Švrček, V.; Maguire, P. Plasma–Liquid Interactions at Atmospheric Pressure for Nanomaterials Synthesis and Surface Engineering. Plasma Process. Polym. 2012, 9, 1074–1085. [Google Scholar] [CrossRef]
- Ogino, A.; Yamashita, M.; Nagatsu, M. Surface Characterization of Plasma Modified Chitosan Film Using Surface-wave Plasma. Energy 2007, 779–782. [Google Scholar]
- Silva, S.S.; Luna, S.M.; Gomes, M.E.; Benesch, J.; Pashkuleva, I.; Mano, J.F.; Reis, R.L. Plasma surface modification of chitosan membranes: Characterization and preliminary cell response studies. Macromol. Biosci. 2008, 8, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Ojah, N.; Kandimalla, R.; Mohan, K.; Gogoi, D.; Dolui, S.K.; Choudhury, A.J. Surface modification of electrospun PVA/chitosan nanofibers by dielectric barrier discharge plasma at atmospheric pressure and studies of their mechanical properties and biocompatibility. Int. J. Biol. Macromol. 2018, 114, 1026–1032. [Google Scholar] [CrossRef]
- López-Pérez, P.M.; Marques, A.P.; Silva, R.M.; Pashkuleva, I.; Reis, R.L. Effect of chitosan membrane surface modification via plasma induced polymerization on the adhesion of osteoblast-like cells. J. Mater. Chem. 2007, 17, 4064–4071. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Cleymand, F.; Noël, C.; Kahn, C.J.; Linder, M.; Dahoun, A.; Henrion, G.; Arab-Tehrany, E. Effects of Ar-H2-N2 microwave plasma on chitosan and its nanoliposomes blend thin films designed for tissue engineering applications. Carbohydr. Polym. 2013, 93, 401–411. [Google Scholar] [CrossRef]
- Chang, S.H.; Chian, C.H. Plasma surface modification effects on biodegradability and protein adsorption properties of chitosan films. Appl. Surf. Sci. 2013, 282, 735–740. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; Vasquez, M.R. Fabrication of Ag-exchanged zeolite/chitosan composites and effects of plasma treatment. Microporous Mesoporous Mater. 2017, 241, 383–391. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; Vasquez, M.R. Hemocompatibility and cytocompatibility of pristine and plasma-treated silver-zeolite-chitosan composites. Appl. Surf. Sci. 2018, 432, 324–331. [Google Scholar] [CrossRef]
- Pappa, A.M.; Karagkiozaki, V.; Krol, S.; Kassavetis, S.; Konstantinou, D.; Pitsalidis, C.; Tzounis, L.; Pliatsikas, N.; Logothetidis, S. Oxygen-plasma-modified biomimetic nanofibrous scaffolds for enhanced compatibility of cardiovascular implants. Beilstein J. Nanotechnol. 2015, 6, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Puppolo, M.M.; Hughey, J.R.; Weber, B.; Dillon, T.; Storey, D.; Cerkez, E.; Jansen-Varnum, S. Plasma modification of microporous polymer membranes for application in biomimetic dissolution studies. AAPS Open 2017, 3. [Google Scholar] [CrossRef]
- Dang, M.; Saunders, L.; Niu, X.; Fan, Y.; Ma, P.X. Biomimetic delivery of signals for bone tissue engineering. Bone Res. 2018, 6. [Google Scholar] [CrossRef]
- Rana, D.; Ramasamy, K.; Leena, M.; Pasricha, R.; Manivasagam, G.; Ramalingam, M. Chapter 21—Surface Functionalization of Biomaterials; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 331–343. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; Olegario, E.M.; Vasquez, M.R. Impregnation of silver in zeolite–chitosan composite: Thermal stability and sterility study. Clay Miner. 2019, 54, 145–151. [Google Scholar] [CrossRef]
- Vasquez, M.; Prieto, E.; Wada, M. RF plasma induced biocompatibility of polyimide substrates. Plasma Med. 2018, 8, 35–44. [Google Scholar] [CrossRef]
- Salapare, H.S.; Suarez, B.A.T.; Cosiñero, H.S.O.; Bacaoco, M.Y.; Ramos, H.J. Irradiation of poly(tetrafluoroethylene) surfaces by CF4 plasma to achieve robust superhydrophobic and enhanced oleophilic properties for biological applications. Mater. Sci. Eng. C 2015, 46, 270–275. [Google Scholar] [CrossRef]
- Ren, Y.; Ding, Z.; Wang, C.; Zang, C.; Zhang, Y.; Xu, L. Influence of DBD plasma pretreatment on the deposition of chitosan onto UHMWPE fiber surfaces for improvement of adhesion and dyeing properties. Appl. Surf. Sci. 2017, 396, 1571–1579. [Google Scholar] [CrossRef]
- Ratner, B.D. Plasma deposition for biomedical applications: A brief review. J. Biomater. Sci. Polym. Ed. 1993, 4, 3–11. [Google Scholar] [CrossRef]
- Gomathi, N.; Sureshkumar, A.; Neogi, S. RF plasma-treated polymers for biomedical applications. Curr. Sci. 2008, 94, 1478–1486. [Google Scholar]
- Baroch, P.; Anita, V.; Saito, N.; Takai, O. Bipolar pulsed electrical discharge for decomposition of organic compounds in water. J. Electrost. 2008, 66, 294–299. [Google Scholar] [CrossRef]
- Manolache, S.; Shamamian, V.; Denes, F. Dense Medium Plasma-Plasma-Enhanced Decontamination of Water of Aromatic Compounds. J. Environ. Eng. 2004, 130, 17–25. [Google Scholar] [CrossRef]
- Zou, J.J.; Liu, C.J.; Eliasson, B. Modification of starch by glow discharge plasma. Carbohydr. Polym. 2004, 55, 23–26. [Google Scholar] [CrossRef]
- Kogelschatz, U. Atmospheric-pressure plasma technology. Plasma Phys. Control. Fusion 2004, 46. [Google Scholar] [CrossRef]
- Tendero, C.; Tixier, C.; Tristant, P.; Desmaison, J.; Leprince, P. Atmospheric pressure plasmas: A review. Spectrochim. Acta Part B At. Spectrosc. 2006, 61, 2–30. [Google Scholar] [CrossRef]
- Schütze, A.; Jeong, J.Y.; Babayan, S.E.; Park, J.; Selwyn, G.S.; Hicks, R.F. The atmospheric-pressure plasma jet: A review and comparison to other plasma sources. IEEE Trans. Plasma Sci. 1998, 26, 1685–1694. [Google Scholar] [CrossRef]
- Winter, J.; Brandenburg, R.; Weltmann, K.D. Atmospheric pressure plasma jets: An overview of devices and new directions. Plasma Sources Sci. Technol. 2015, 24, 64001. [Google Scholar] [CrossRef]
- Lu, Q.; Yu, J.; Gao, J.; Yang, W.; Li, Y. Glow-discharge Electrolysis Plasma Induced Synthesis of Polyvinylpyrrolidone/Acrylic Acid Hydrogel and its Adsorption Properties for Heavy-metal Ions. Plasma Process. Polym. 2011, 8, 803–814. [Google Scholar] [CrossRef]
- Yu, J.; Yang, G.; Li, Y.; Yang, W.; Gao, J.; Lu, Q. Synthesis, Characterization, and swelling behaviors of acrylic acid/carboxymethyl cellulose superabsorbent hydrogel by glow-discharge electrolysis plasma. Polym. Eng. Sci. 2014, 54, 2310–2320. [Google Scholar] [CrossRef]
- Zhang, W.; Sha, Z.; Huang, Y.; Bai, Y.; Xi, N.; Zhang, Y. Glow discharge electrolysis plasma induced synthesis of cellulose-based ionic hydrogels and their multiple response behaviors. RSC Adv. 2015, 5, 6505–6511. [Google Scholar] [CrossRef]
- Zhang, W.; Zhu, S.; Bai, Y.; Xi, N.; Wang, S.; Bian, Y.; Li, X.; Zhang, Y. Glow discharge electrolysis plasma initiated preparation of temperature/pH dual sensitivity reed hemicellulose-based hydrogels. Carbohydr. Polym. 2015, 122, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Miao, W.; Yue, Z.; Liu, S.; Li, L.; Zhang, Z.; Wang, H. Glow discharge electrolysis plasma initiated synthesis of upconversion luminescent and temperature sensitive multifunctional hydrogel. Soft Mater. 2018, 16, 192–200. [Google Scholar] [CrossRef]
- Molina, R.; Jovancic, P.; Vilchez, S.; Tzanov, T.; Solans, C. In situ chitosan gelation initiated by atmospheric plasma treatment. Carbohydr. Polym. 2014, 103, 472–479. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; De Leon, M.J.D.; Thumanu, K.; Nakajima, H.; Chanlek, N.; Prieto, E.I.; Vasquez, M.R. Probing the structural features of a plasma-treated chitosan-acrylic acid hydrogel. Colloids Surf. Physicochem. Eng. Asp. 2022, 637, 128233. [Google Scholar] [CrossRef]
- Hamouda, I.; Labay, C.; Ginebra, M.P.; Nicol, E.; Canal, C. Investigating the atmospheric pressure plasma jet modification of a photo-crosslinkable hydrogel. Polymer 2020, 192, 122308. [Google Scholar] [CrossRef]
- Das, S.P.; Dalei, G.; Sahoo, S.; Das, S. Cold atmospheric plasma surface nanoengineered carboxymethyl cellulose hydrogels as oral ibuprofen carriers. SN Appl. Sci. 2019, 1. [Google Scholar] [CrossRef]
- Dalei, G.; Das, S.; JENA, S.; Nayak, J.; Samanta, L.; Das, S. Improved Chemosensitization Activity of Carboxymethyl Chitosan/PVA Hydrogels by Plasma Surface Modification. J. Polym. Environ. 2021, 29, 1–17. [Google Scholar] [CrossRef]
- Martinez-Gomez, A.; Cruz-Barba, L.; Sánchez-Díaz, J.; Becerra-Bracamontes, F.; Martínez-Ruvalcaba, A. Plasma enhanced modification of xanthan and its use in chitosan/xanthan hydrogels. Polym. Eng. Sci. 2014, 54, 2264–2271. [Google Scholar] [CrossRef]
- Levien, M.; Farka, Z.; Pastucha, M.; Skládal, P.; Nasri, Z.; Weltmann, K.D.; Fricke, K. Functional plasma-polymerized hydrogel coatings for electrochemical biosensing. Appl. Surf. Sci. 2022, 584, 152511. [Google Scholar] [CrossRef]
- Dalei, G.; Das, S.; Das, S.P. Evaluation of TEOS Plasma Polymerized Carboxymethyl Starch/Alginate Hydrogels as Controlled Drug Delivery Systems. Starch-StäRke 2022, 74, 2100226. [Google Scholar] [CrossRef]
- Suresh, M.; Kondeti, V.S.S.K.; Bruggeman, P.J. Production and diffusion of H2O2 during the interaction of a direct current pulsed atmospheric pressure plasma jet on a hydrogel. J. Phys. Appl. Phys. 2022, 55, 185201. [Google Scholar] [CrossRef]
- Lee, H.R.; Lee, H.Y.; Heo, J.; Jang, J.Y.; Shin, Y.S.; Kim, C.H. Liquid-type nonthermal atmospheric plasma enhanced regenerative potential of silk–fibrin composite gel in radiation-induced wound failure. Mater. Sci. Eng. C 2021, 128, 112304. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Bolouki, N.; Manga, Y.B.; Hsieh, J.H.; Chuang, E.Y.; Chen, C.H. Novel gelatin–graphene oxide crosslinking induced by nonthermal atmospheric pressure plasma for alendronate delivery system. Plasma Process. Polym. 2020, 17, 2000110. [Google Scholar] [CrossRef]
- Dalei, G.; Swain, S.; Das, S.; Das, S.P. Controlled Release of 5-Fluorouracil from Alginate Hydrogels by Cold HMDSO− Plasma Surface Engineering. ChemistrySelect 2020, 5, 2168–2178. [Google Scholar] [CrossRef]
- Rogojanu, A.; Rusu, E.; Dorohoi, D.O. Characterization of Structural Modifications Induced on Poly(Vinyl Alcohol) Surface by Atmospheric Pressure Plasma. Int. J. Polym. Anal. Charact. 2010, 15, 210–221. [Google Scholar] [CrossRef]
- Xu, C.; Ruiyu, H.; Xie, B.; Ismail, M.; Yao, C.; Luan, J.; Xinsong, L. Improved Protein Resistance of Silicone Hydrogels by Grafting Short Peptides for Ophthalmological Application. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 547–554. [Google Scholar] [CrossRef]
- Labay, C.; Roldán, M.; Tampieri, F.; Stancampiano, A.; Escot Bocanegra, P.; Ginebra, M.P.; Canal, C. Enhanced Generation of Reactive Species by Cold Plasma in Gelatin Solutions for Selective Cancer Cell Death. ACS Appl. Mater. Interfaces 2020, 12, 47256. [Google Scholar] [CrossRef]
- Jovancic, P.; Vílchez, A.; Molina, R. Synthesis of Thermo-Sensitive Hydrogels from Free Radical Copolymerization of NIPAAm with MBA Initiated by Atmospheric Plasma Treatment. Plasma Process. Polym. 2016, 13, 752–760. [Google Scholar] [CrossRef]
- Nolan, H.; Sun, D.; Falzon, B.G.; Chakrabarti, S.; Padmanaba, D.B.; Maguire, P.; Mariotti, D.; Yu, T.; Jones, D.; Andrews, G.; et al. Metal nanoparticle-hydrogel nanocomposites for biomedical applications—An atmospheric pressure plasma synthesis approach. Plasma Process. Polym. 2018, 15. [Google Scholar] [CrossRef]
- Dalei, G.; Das, S.; Das, S.P. Non-thermal plasma assisted surface nano-textured carboxymethyl guar gum/chitosan hydrogels for biomedical applications. RSC Adv. 2019, 9, 1705–1716. [Google Scholar] [CrossRef] [PubMed]
- Paradiso, P.; Chu, V.; Santos, L.; Serro, A.P.; Colaço, R.; Saramago, B. Effect of plasma treatment on the performance of two drug-loaded hydrogel formulations for therapeutic contact lenses. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Jumalon, J.; Basubas, C.; Taaca, K.; Capuli, C.J.D.; Castillo, A.P.; Pasela, B.R.; Tumacder, D.v.; Simon, R. Surface modification of polyvinyl alcohol-chitosan blend hydrogels using RF plasma treatment. In Proceedings of the 37th Samahang Pisika ng Pilipinas Physics Conference, Tagbilaran City, Philippines, 29 May–1 June 2019; Volume 37. SPP–2019–2E–05. [Google Scholar]
- Dalei, G.; Das, S.; Das, S.P. Low-pressure nitrogen and ammonia plasma treatment on carboxymethyl guar gum/PVA hydrogels: Impact on drug delivery, biocompatibility and biodegradability. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 75–89. [Google Scholar] [CrossRef]
- Rout, B.; Girard-Lauriault, P.L. Permeation-resistant and flexible plasma-polymerised films on 2-hydroxyethyl methacrylate hydrogels. Plasma Process. Polym. 2021, 18, 2000191. [Google Scholar] [CrossRef]
- Satapathy, M.K.; Chiang, W.H.; Chuang, E.Y.; Chen, C.H.; Liao, J.L.; 4 Huang, H.N. Microplasma-assisted hydrogel fabrication: A novel method for gelatin-graphene oxide nano composite hydrogel synthesis for biomedical application. PeerJ 2017, 5, e3498. [Google Scholar] [CrossRef]
- Satapathy, M.K.; Manga, Y.B.; Ostrikov, K.K.; Chiang, W.H.; Pandey, A.; Lekha, R.; Nyambat, B.; Chuang, E.Y.; Chen, C.H. Microplasma Cross-Linked Graphene Oxide-Gelatin Hydrogel for Cartilage Reconstructive Surgery. ACS Appl. Mater. Interfaces 2020, 12, 86–95. [Google Scholar] [CrossRef]
- Nolan, H.; Sun, D.; Falzon, B.G.; Maguire, P.; Mariotti, D.; Zhang, L.; Sun, D. Thermoresponsive nanocomposites incorporating microplasma synthesized magnetic nanoparticles—Synthesis and potential applications. Plasma Process. Polym. 2019, 16, 1–7. [Google Scholar] [CrossRef]
- Gorbanev, Y.; O’Connell, D.; Chechik, V. Non-Thermal Plasma in Contact with Water: The Origin of Species. Chem. A Eur. J. 2016, 22, 3496–3505. [Google Scholar] [CrossRef]




| Property | Sample Characteristics |
|---|---|
| Biocompatibility | Non-toxic; non-carcinogenic; non-allergenic |
| Physical properties | Density; porosity; form; surface roughness |
| Chemical properties | Inert, stable, reactive, selective |
| Mechanical properties | Compressive; tensile; shear; impact |
| Scalability | Processable; sustainable; sterilizable |
| Service life | Stable; tunable degradation rate |
| Economical | Affordable; readily available |
| Hydrogel | Characteristics | References |
|---|---|---|
| AAm/NDAPM | Stimuli responsive where volume and elasticity change | [69] |
| HA/2-HEA/PEGDA | Porous and biocompatible; capable of sustained drug release | [70] |
| Acrylate-g-PHEMA | Hydrophobic with tunable hardness and swelling | [71] |
| AAm/MBA | Temperature-sensitive swelling with smooth surface | [72] |
| Xantham/Chitosan/Gelatin/PEG | High water content, porous and biodegradable wound dressing | [73] |
| Hydrogel | Plasma Source | Operating Gas | Target Application(s) | Reference |
|---|---|---|---|---|
| Chitosan/acrylic acid | Plasma jet | Air | Wound healing | [153] |
| tPEO | Plasma jet | He | Various | [154] |
| Carboxymethyl cellulose/PVA | CAP | Ar, N | Drug carrier | [155] |
| Chitosan/PVA | Glow discharge | Air, He, N | Drug delivery | [156] |
| Chitosan/xanthan | PECVD | He, epichlorohydrin | Drug release | [157] |
| HEMA:DEAEMA | Plasma jet | Ar | Biosensing | [158] |
| Starch/Alginate | PECVD | O, tetraethyl orthosilicate | Drug delivery | [159] |
| Gelatin | Plasma jet | - | - | [160] |
| Silk/fibrin | Plasma jet | Ar, N | Wound healing | [161] |
| Gelatin/GO | DBD | Ar | Drug delivery | [162] |
| PVA/alginate | CAP | Ar, hexamethyldisiloxane | Drug delivery | [163] |
| PVA | DBD | He | - | [164] |
| Silicone | Glow discharge | He | Contact lens | [165] |
| Gelatin | Plasma jet | He | Drug delivery | [166] |
| NIPAAm | DBD | He | - | [167] |
| PVA/nanoparticle | CAP | He | Various | [168] |
| Chitosan/guar gum | CAP | Ar, O | Drug delivery, antibiofilm | [169] |
| HEMA/PVP, TRIS/NVP/HEMA | PECVD | N | Contact lens | [170] |
| PVA/Chitosan | RF discharge | Ar | - | [171] |
| Carboxymethyl/guar gum/PVA | Glow discharge | N and NH | Drug delivery | [172] |
| 2-hydroxyethyl methacrylate | RF discharge | CH, CH, CO, NH | - | [173] |
| Gelatin/GO | Microplasma | Ar | Cartilage reconstructive surgey | [174,175] |
| FeO-PNIPAm | Microplasma | He | - | [176] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taaca, K.L.M.; Prieto, E.I.; Vasquez, M.R., Jr. Current Trends in Biomedical Hydrogels: From Traditional Crosslinking to Plasma-Assisted Synthesis. Polymers 2022, 14, 2560. https://doi.org/10.3390/polym14132560
Taaca KLM, Prieto EI, Vasquez MR Jr. Current Trends in Biomedical Hydrogels: From Traditional Crosslinking to Plasma-Assisted Synthesis. Polymers. 2022; 14(13):2560. https://doi.org/10.3390/polym14132560
Chicago/Turabian StyleTaaca, Kathrina Lois M., Eloise I. Prieto, and Magdaleno R. Vasquez, Jr. 2022. "Current Trends in Biomedical Hydrogels: From Traditional Crosslinking to Plasma-Assisted Synthesis" Polymers 14, no. 13: 2560. https://doi.org/10.3390/polym14132560
APA StyleTaaca, K. L. M., Prieto, E. I., & Vasquez, M. R., Jr. (2022). Current Trends in Biomedical Hydrogels: From Traditional Crosslinking to Plasma-Assisted Synthesis. Polymers, 14(13), 2560. https://doi.org/10.3390/polym14132560






