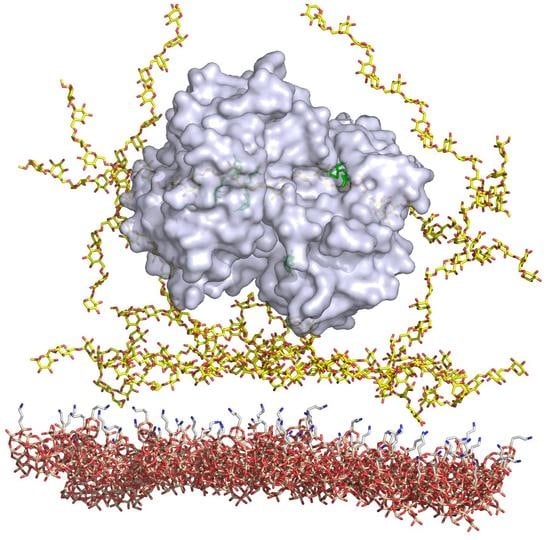
The Elaboration of Effective Coatings for Photonic Crystal Chips in Optical Biosensors
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
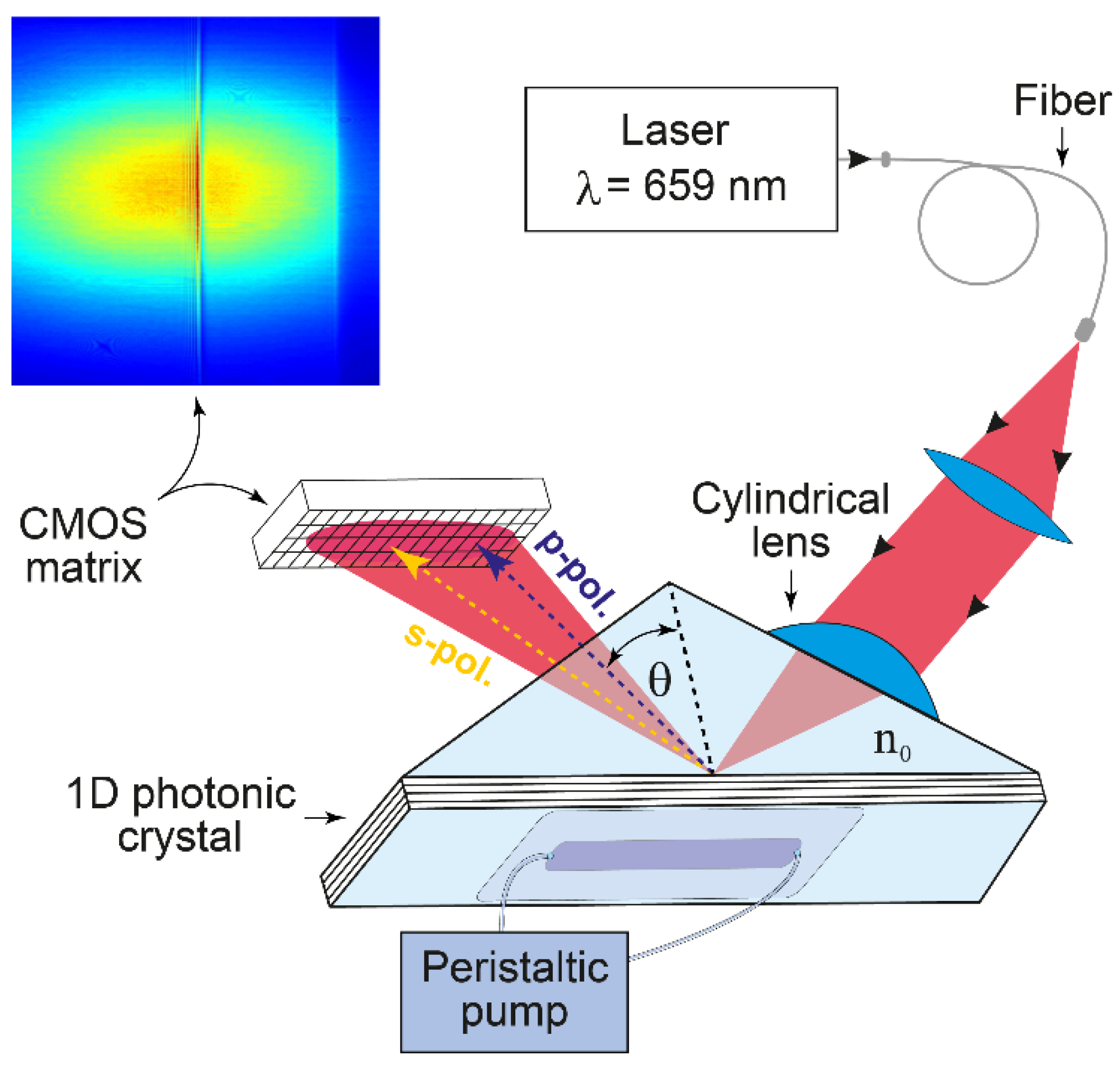
2.2. Photonic Crystal Surface Modes Detection System
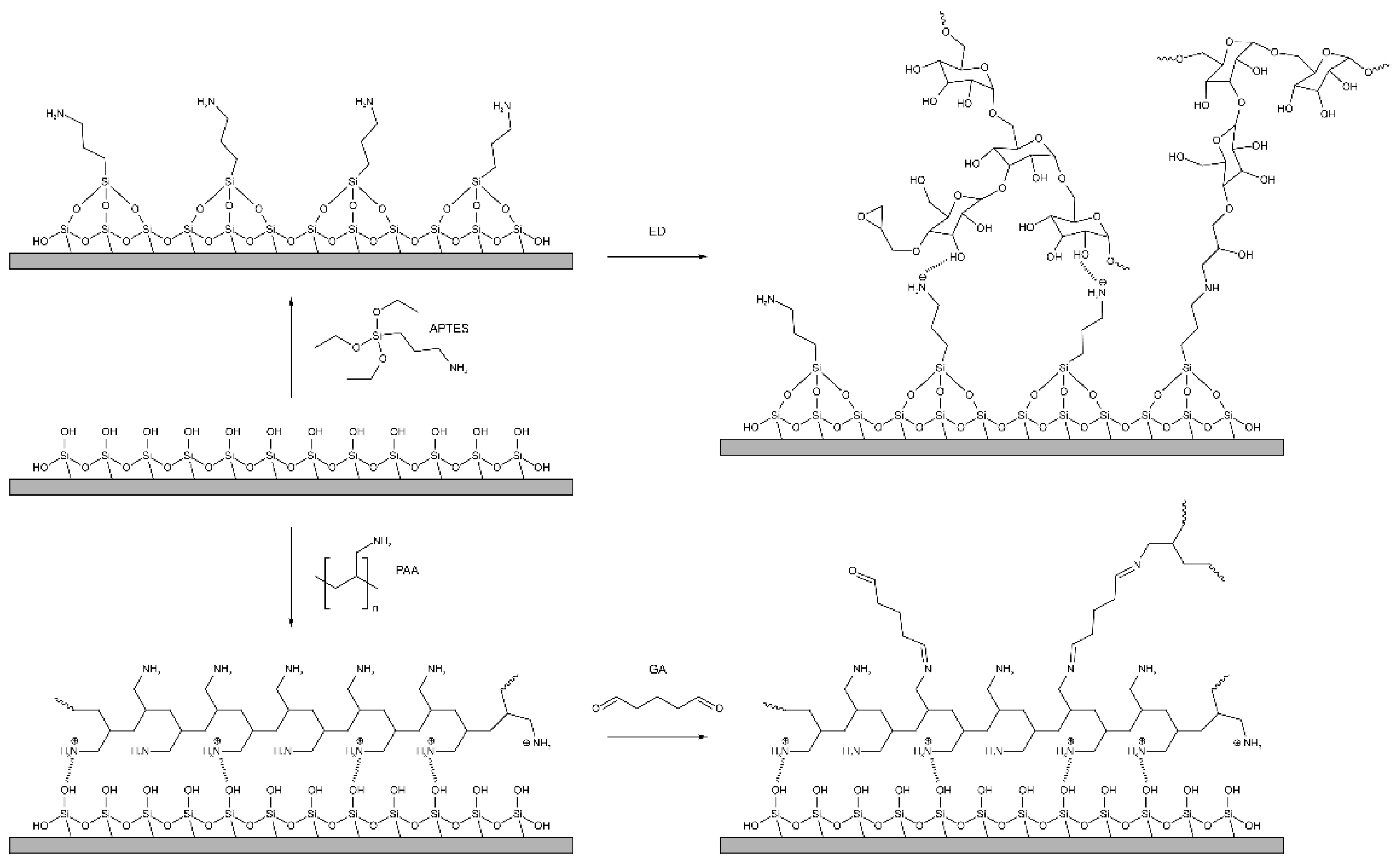
2.3. Synthesis of Aldehyde Dextran
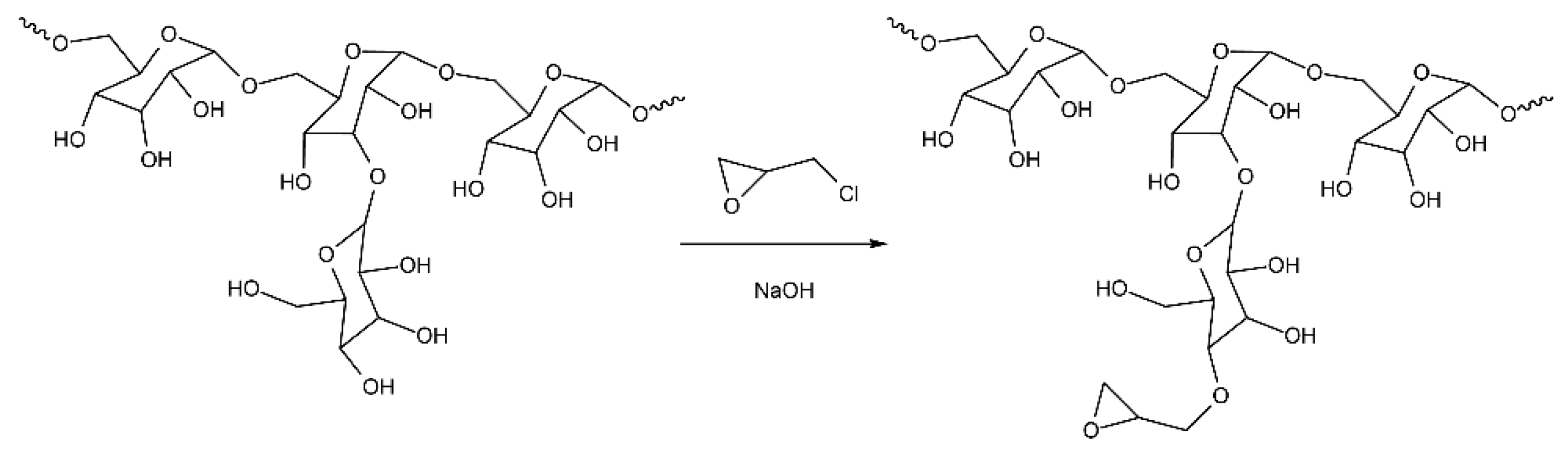
2.4. Synthesis of Epoxydextran
2.5. Fabrication of the PC Biochip: Activation and Functionalization
2.6. Proteins and Low Molecular Weight Biomolecules Binding Detection
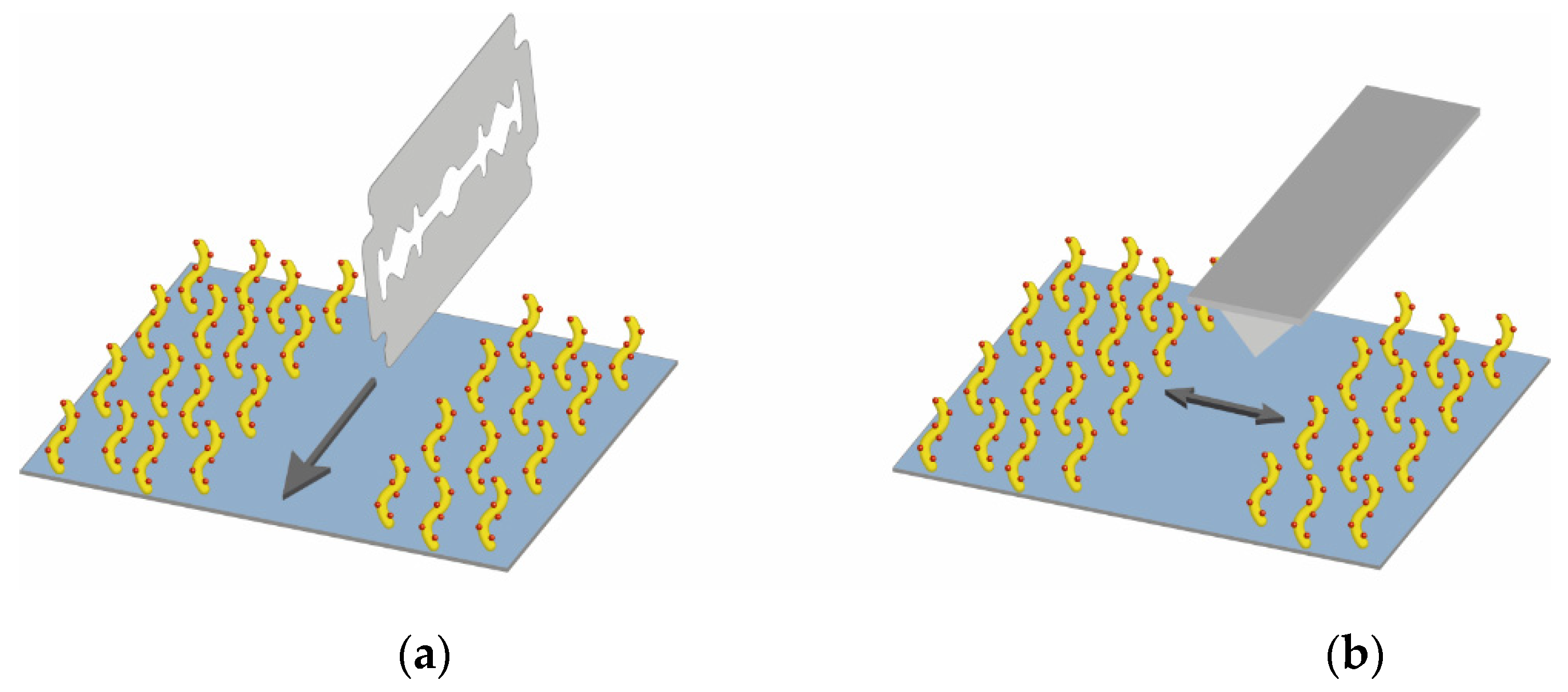
2.7. Atomic Force Microscopy
3. Results
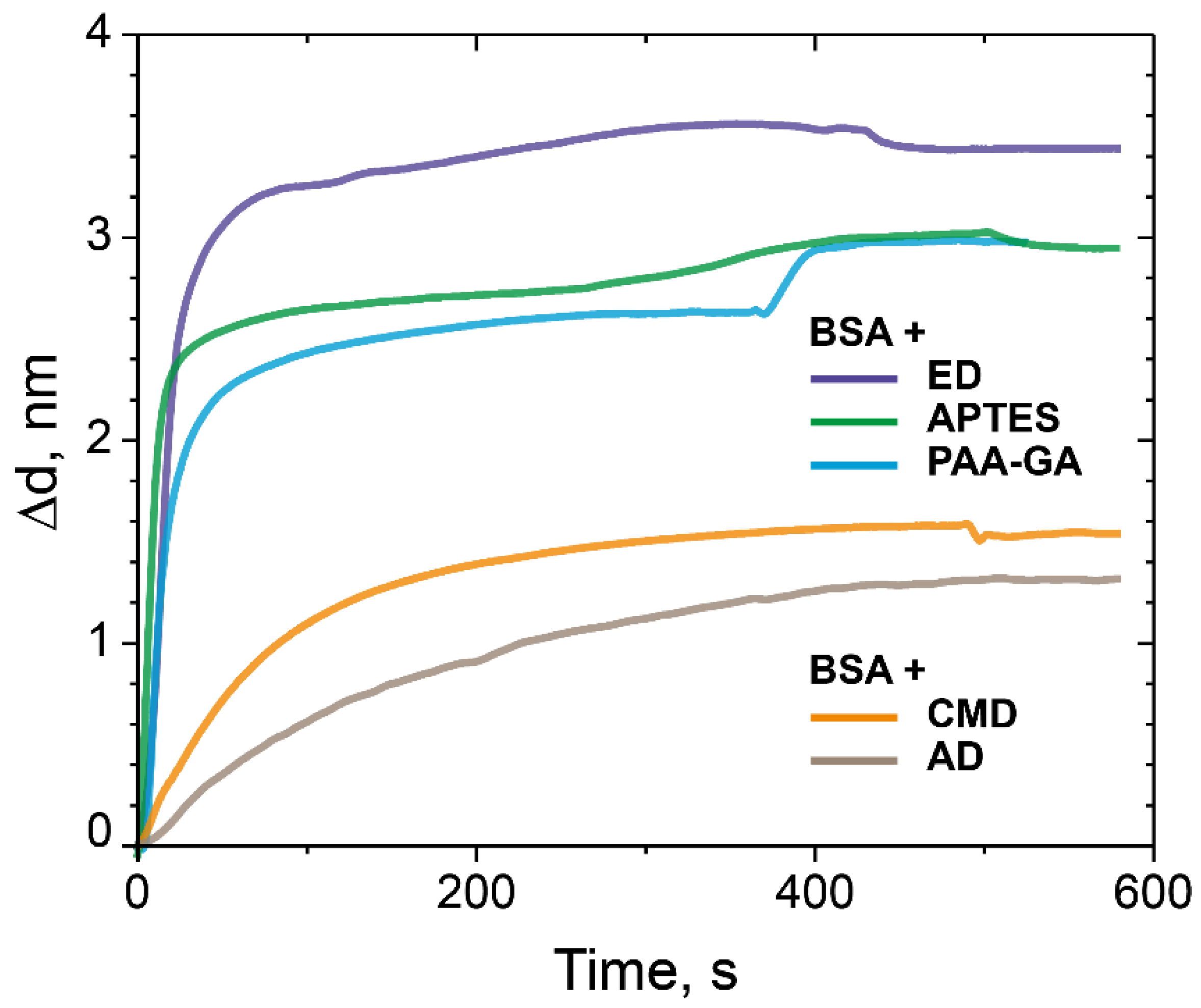
3.1. Comparison of Model Protein Binding Capacity on Dextrans with Anchor Carboxy, Aldehyde, and Epoxy Groups
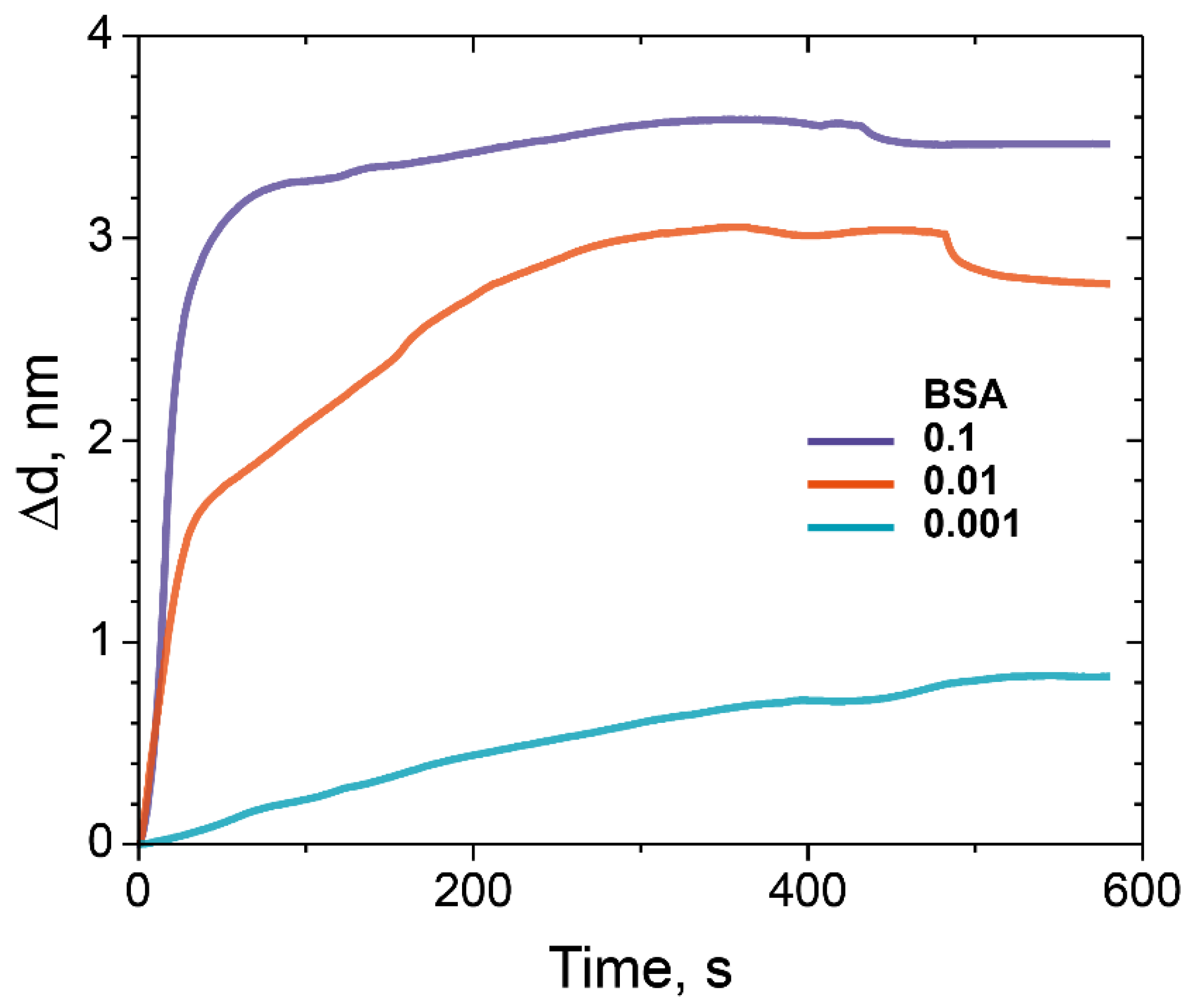
3.2. Investigation of Sorption Capacity of ED with Different Molecular Weight
3.3. Normalized Sensograms of BSA Binding to ED
3.4. Detection of Low Molecular Weight Biomolecules Interaction by PC SM Biosensor
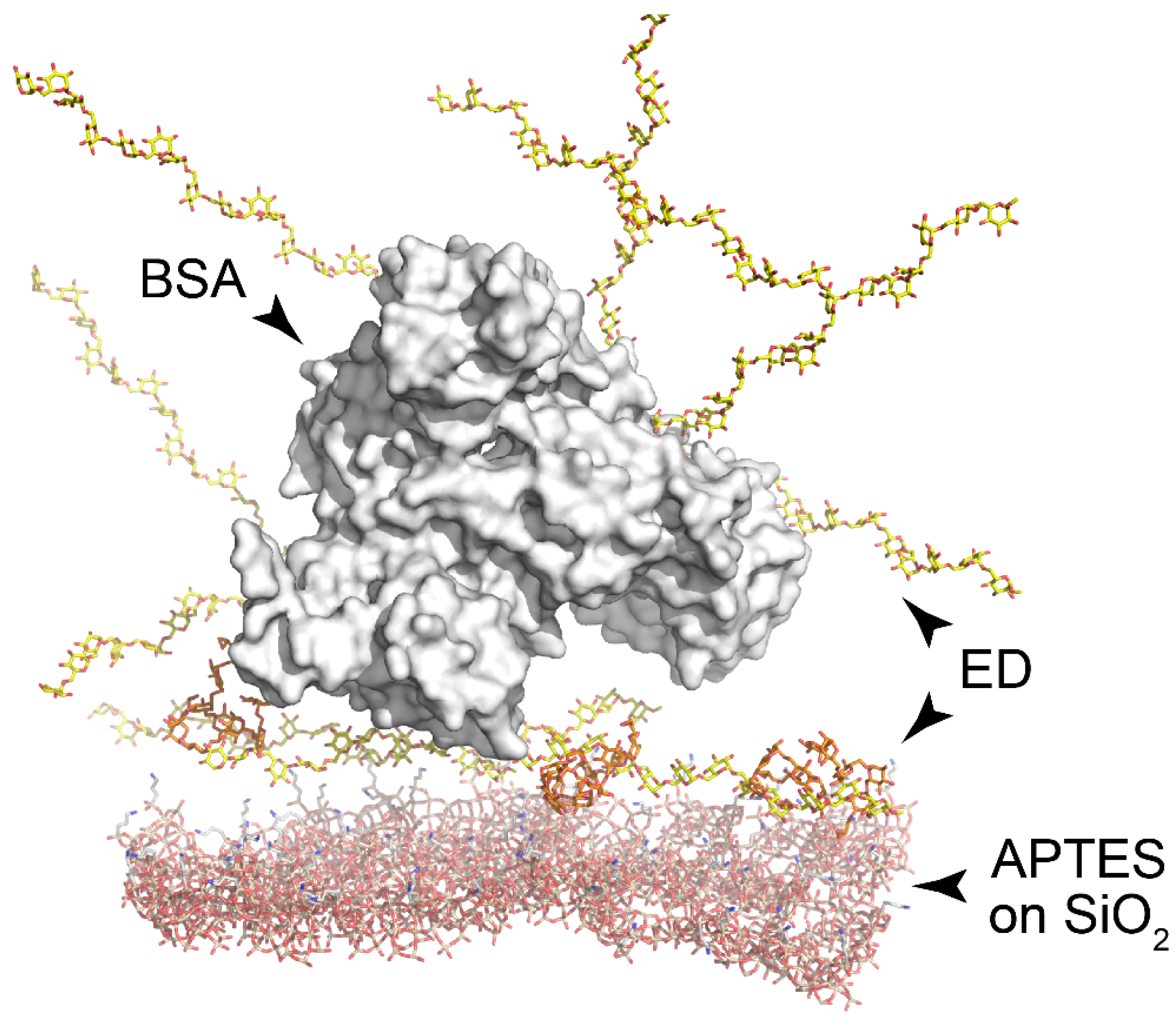
3.5. Measuring of the Thickness of Fabricated 3D Dextran Matrix on PC Chip
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heydarnezhad, H.R.; Pourabbas, B.; Tayefi, M. Conducting Electroactive Polymers via Photopolymerization: A Review on Synthesis and Applications. Polym.-Plast. Technol. Eng. 2018, 57, 1093–1109. [Google Scholar] [CrossRef]
- Shi, J.; Porterfield, D.M. Surface Modification Approaches for Electrochemical Biosensors; In Biosensors-Emerging Materials and Applications, 1st ed.; Serra, P.A., Ed.; In-Tech.: Rijeka, Croatia, 2011; pp. 209–226. [Google Scholar] [CrossRef]
- Schäferling, M.; Riepl, M.; Pavlickova, P.; Paul, H.; Kambhampati, D.; Liedberg, B. Functionalized Self-Assembled Monolayers on Gold as Binding Matrices for the Screening of Antibody-Antigen Interactions. Microchim. Acta 2003, 142, 193–203. [Google Scholar] [CrossRef]
- Nicosia, C.; Huskens, J. Reactive Self-Assembled Monolayers: From Surface Functionalization to Gradient Formation. Mater. Horiz. 2014, 1, 32–45. [Google Scholar] [CrossRef]
- Ricciardi, S.; Castagna, R.; Severino, S.M.; Ferrante, I.; Frascella, F.; Celasco, E.; Mandracci, P.; Vallini, I.; Mantero, G.; Pirri, C.F.; et al. Surface Functionalization by Poly-Acrylic Acid Plasma-Polymerized Films for Microarray DNA Diagnostics. Surf. Coat. Technol. 2012, 207, 389–399. [Google Scholar] [CrossRef]
- Thierry, B.; Jasieniak, M.; De Smet, L.C.P.M.; Vasilev, K.; Griesser, H.J. Reactive Epoxy-Functionalized Thin Films by a Pulsed Plasma Polymerization Process. Langmuir 2008, 24, 10187–10195. [Google Scholar] [CrossRef] [PubMed]
- Gong, X. Self-Assembly Technique for Biomedical Applications. Nano LIFE 2015, 5, 1542002. [Google Scholar] [CrossRef]
- Mescola, A.; Canale, C.; Prato, M.; Diaspro, A.; Berdondini, L.; MacCione, A.; Dante, S. Specific Neuron Placement on Gold and Silicon Nitride-Patterned Substrates through a Two-Step Functionalization Method. Langmuir 2016, 32, 6319–6327. [Google Scholar] [CrossRef] [PubMed]
- Reimhult, E.; Höök, F. Design of Surface Modifications for Nanoscale Sensor Applications. Sensors 2015, 15, 1635–1675. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Du, L.; Krause, S.; Wu, C.; Wang, P. Surface Modification and Construction of LAPS towards Biosensing Applications. Sens. Actuators B Chem. 2018, 265, 161–173. [Google Scholar] [CrossRef]
- Akther, F.; Yakob, S.B.; Nguyen, N.T.; Ta, H.T. Surface Modification Techniques for Endothelial Cell Seeding in PDMS Microfluidic Devices. Biosensors 2020, 10, 182. [Google Scholar] [CrossRef]
- Petrova, I.; Konopsky, V.; Nabiev, I.; Sukhanova, A. Label-Free Flow Multiplex Biosensing via Photonic Crystal Surface Mode Detection. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Wang, Z.H.; Jin, G. Covalent Immobilization of Proteins for the Biosensor Based on Imaging Ellipsometry. J. Immunol. Methods 2004, 285, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Harpaz, D.; Koh, B.; Marks, R.S.; Seet, R.C.S.; Abdulhalim, I.; Tok, A.I.Y. Point-of-Care Surface Plasmon Resonance Biosensor for Stroke Biomarkers NT-ProBNP and S100β Using a Functionalized Gold Chip with Specific Antibody. Sensors 2019, 19, 2533. [Google Scholar] [CrossRef] [PubMed]
- Rehmat, Z.; Mohammed, W.S.; Anal, A.K. Chitosan-Based Nanomatrix for the Immobilization of Ochratoxin-A Conjugate on Surface Plasmon Resonance Chips. Colloid Polym. Sci. 2018, 296, 617–625. [Google Scholar] [CrossRef]
- Neugebauer, S.; Isik, S.; Schulte, A.; Schuhmann, W. Acrylic Acid-Based Copolymers as Immobilization Matrix for Amperometric Biosensors. Anal. Lett. 2003, 36, 2005–2020. [Google Scholar] [CrossRef]
- Löfås, S.; Johnsson, B. A Novel Hydrogel Matrix on Gold Surfaces in Surface Plasmon Resonance Sensors for Fast and Efficient Covalent Immobilization of Ligands. J. Chem. Soc. Chem. Commun. 1990, 21, 1526–1528. [Google Scholar] [CrossRef]
- Arnaud, J.A.; Saleh, A.A.M. Guidance of Surface Waves by Multilayer Coatings. Appl. Opt. 1974, 13, 2343–2345. [Google Scholar] [CrossRef]
- Yeh, P.; Yariv, A.; Hong, C.-S. Electromagnetic Propagation in Periodic Stratified Media. I. General Theory. J. Opt. Soc. Am. 1977, 67, 423–438. [Google Scholar] [CrossRef]
- Yeh, P.; Yariv, A.; Cho, A.Y. Optical Surface Waves in Periodic Layered Media. Appl. Phys. Lett. 1978, 32, 104–105. [Google Scholar] [CrossRef]
- Konopsky, V.N.; Alieva, E.V. Photonic Crystal Surface Waves for Optical Biosensors. Anal. Chem. 2007, 79, 4729–4735. [Google Scholar] [CrossRef]
- Kanoh, N.; Kyo, M.; Inamori, K.; Ando, A.; Asami, A.; Nakao, A.; Osada, H. SPR Imaging of Photo-Cross-Linked Small-Molecule Arrays on Gold. Anal. Chem. 2006, 78, 2226–2230. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Saint-Guirons, J.; Käck, C.; Ingemarsson, B.; Aastrup, T. Real-Time Analysis of the Carbohydrates on Cell Surfaces Using a QCM Biosensor: A Lectin-Based Approach. Biosens. Bioelectron. 2012, 35, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Mohan, T.; Čas, A.; Bračič, M.; Plohl, O.; Vesel, A.; Rupnik, M.; Zemljič, L.F.; Rebol, J. Highly Protein Repellent and Antiadhesive Polysaccharide Biomaterial Coating for Urinary Catheter Applications. ACS Biomater. Sci. Eng. 2019, 5, 5825–5832. [Google Scholar] [CrossRef]
- Hu, X.; Ricci, S.; Naranjo, S.; Hill, Z.; Gawason, P. Protein and Polysaccharide-Based Electroactive and Conductive Materials for Biomedical Applications. Molecules 2021, 26, 4499. [Google Scholar] [CrossRef]
- Fu, Z.; Han, L.; Zhang, P.; Mao, H.; Zhang, H.; Wang, Y.; Gao, X.; Liu, E. Cistanche Polysaccharides Enhance Echinacoside Absorption in Vivo and Affect the Gut Microbiota. Int. J. Biol. Macromol. 2020, 149, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Chatelier, R.C.; Zientek, P.; Gengenbach, T.R.; Griesser, H.J. Covalent Surface Attachment of Polysaccharides via Bifunctional Epoxides. In Surface Modification of Polymeric Biomaterials; Springer: Boston, MA, USA, 1996. [Google Scholar] [CrossRef]
- Song, S.; Lu, Y.; Li, X.; Cao, S.; Pei, Y.; Aastrup, T.; Pei, Z. Optimization of 3D Surfaces of Dextran with Different Moleculeweights for Real-Time Detection of Biomolecular Interactions by a QCM Biosensor. Polymers 2017, 9, 409. [Google Scholar] [CrossRef] [PubMed]
- Frascella, F.; Petri, C.; Ricciardi, S.; Napione, L.; Munzert, P.; Jonas, U.; Dostalek, J.; Bussolino, F.; Pirri, C.F.; Descrovi, E. Hydrogel-Terminated Photonic Crystal for Label-Free Detection of Angiopoietin-1. J. Lightwave Technol. 2016, 34, 3641–3645. [Google Scholar] [CrossRef]
- Konopsky, V.N.; Karakouz, T.; Alieva, E.V.; Vicario, C.; Sekatskii, S.K.; Dietler, G. Photonic Crystal Biosensor Based on Optical Surfacewaves. Sensors 2013, 13, 2566–2578. [Google Scholar] [CrossRef] [PubMed]
- Konopsky, V.N.; Alieva, E.V. A Biosensor Based on Photonic Crystal Surface Waves with an Independent Registration of the Liquid Refractive Index. Biosens. Bioelectron. 2010, 25, 1212–1216. [Google Scholar] [CrossRef]
- Tacias-Pascacio, V.G.; Ortiz, C.; Rueda, N.; Berenguer-Murcia, Á.; Acosta, N.; Aranaz, I.; Civera, C.; Fernandez-Lafuente, R.; Alcántara, A.R. Dextran Aldehyde in Biocatalysis: More than a Mere Immobilization System. Catalysts 2019, 9, 622. [Google Scholar] [CrossRef]
- Fuentes, M.; Segura, R.L.; Abian, O.; Betancor, L.; Hidalgo, A.; Mateo, C.; Fernandez-Lafuente, R.; Guisan, J.M. Determination of Protein-Protein Interactions through Aldehyde-Dextran Intermolecular Cross-Linking. Proteomics 2004, 4, 2602–2607. [Google Scholar] [CrossRef]
- Generalova, A.N.; Sizova, S.V.; Zdobnova, T.A.; Zarifullina, M.M.; Artemyev, M.V.; Baranov, A.V.; Oleinikov, V.A.; Zubov, V.P.; Deyev, S.M. Submicron Polymer Particles Containing Fluorescent Semiconductor Nanocrystals CdSe/ZnS for Bioassays. Nanomedicine 2011, 6, 195–209. [Google Scholar] [CrossRef]
- Dyatlov, V.; Seregina, T.; Luss, A.; Zaitsev, V.; Artyukhov, A.; Shtilman, M.; Chumakova, A.; Kushnerev, K.; Tsatsakis, A.; Mezhuev, Y. Immobilization of Amikacin on Dextran: Biocomposite Materials That Release an Antibiotic in the Presence of Bacterial Dextranase. Polym. Int. 2020, 70, 837–844. [Google Scholar] [CrossRef]
- Gunda, N.S.K.; Singh, M.; Norman, L.; Kaur, K.; Mitra, S.K. Optimization and Characterization of Biomolecule Immobilization on Silicon Substrates Using (3-Aminopropyl)Triethoxysilane (APTES) and Glutaraldehyde Linker. Appl. Surf. Sci. 2014, 305, 522–530. [Google Scholar] [CrossRef]
- Konopsky, V.; Mitko, T.; Aldarov, K.; Alieva, E.; Basmanov, D.; Moskalets, A.; Matveeva, A.; Morozova, O.; Klinov, D. Photonic Crystal Surface Mode Imaging for Multiplexed and High-Throughput Label-Free Biosensing. Biosens. Bioelectron. 2020, 168, 112575. [Google Scholar] [CrossRef] [PubMed]
- Hermanson, G.T. Bioconjugate Techniques; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar] [CrossRef]
- Kamerbeek, J.; Schouls, L.; Kolk, A.; Van Agterveld, M.; Van Soolingen, D.; Kuijper, S.; Bunschoten, A.; Molhuizen, H.; Shaw, R.; Goyal, M.; et al. Simultaneous Detection and Strain Differentiation of Mycobacterium Tuberculosis for Diagnosis and Epidemiology. J. Clin. Microbiol. 1997, 35, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Herr, A.E. Protein Immobilization Techniques for Microfluidic Assays. Biomicrofluidics 2013, 7, 4151. [Google Scholar] [CrossRef]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeerschd, T.; Zurek, E.; Hutchison, G.R. Avogadro: An Advanced Semantic Chemical Editor, Visualization, and Analysis Platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef]
- Elender, G.; Kfihner, M.; Sackmann, E. Functionalisation of Si/SiO2 and Glass Surfaces with Ultrathin Dextran Films and Deposition of Lipid Bilayers. Biosens. Bioelectron. 1996, 11, 565–577. [Google Scholar] [CrossRef]
- Hu, Q.; Lu, Y.; Luo, Y. Recent Advances in Dextran-Based Drug Delivery Systems: From Fabrication Strategies to Applications. Carbohydr. Polym. 2021, 264, 117999. [Google Scholar] [CrossRef]
- Malaprade, L. The Oxydation of Certain Polyhydric Alcohol by the Application of Periodic Acid. R Hebd. Seances Acad. Sci. 1928, 186, 382–385. [Google Scholar]
- Chu, M.; Dong, C.; Zhu, H.; Cai, X.; Dong, H.; Ren, T.; Su, J.; Li, Y. Biocompatible Polyethylenimine-Graft-Dextran Catiomer for Highly Efficient Gene Delivery Assisted by a Nuclear Targeting Ligand. Polym. Chem. 2013, 4, 2528–2539. [Google Scholar] [CrossRef]
- O’Connor, N.A.; Abugharbieh, A.; Yasmeen, F.; Buabeng, E.; Mathew, S.; Samaroo, D.; Cheng, H.P. The Crosslinking of Polysaccharides with Polyamines and Dextran-Polyallylamine Antibacterial Hydrogels. Int. J. Biol. Macromol. 2015, 72, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.R.; Sriram, R.; Carter, J.A.; Miller, B.L. Comparative Study of Solution-Phase and Vapor-Phase Deposition of Aminosilanes on Silicon Dioxide Surfaces. Mater. Sci. Eng. C 2014, 35, 283–290. [Google Scholar] [CrossRef]
- Howarter, J.A.; Youngblood, J.P. Optimization of Silica Silanization by 3-Aminopropyltriethoxysilane. Langmuir 2006, 22, 11142–11147. [Google Scholar] [CrossRef]
- Seitz, O.; Fernandes, P.G.; Tian, R.; Karnik, N.; Wen, H.C.; Stiegler, H.; Chapman, R.A.; Vogel, E.M.; Chabal, Y.J. Control and Stability of Self-Assembled Monolayers under Biosensing Conditions. J. Mater. Chem. 2011, 21, 4384–4392. [Google Scholar] [CrossRef]
- Morozova, O.V.; Levchenko, O.A.; Cherpakova, Z.A.; Prokhorov, V.V.; Barinov, N.A.; Obraztsova, E.A.; Belova, A.M.; Prusakov, K.A.; Aldarov, K.G.; Basmanov, D.V.; et al. Surface Modification with Polyallylamines for Adhesion of Biopolymers and Cells. Int. J. Adhes. Adhes. 2019, 92, 125–132. [Google Scholar] [CrossRef]
- Fan, X.; White, I.M.; Shopova, S.I.; Zhu, H.; Suter, J.D.; Sun, Y. Sensitive Optical Biosensors for Unlabeled Targets: A Review. Anal. Chim. Acta 2008, 620, 8–26. [Google Scholar] [CrossRef]
- Tatarinova, O.; Tsvetkov, V.; Basmanov, D.; Barinov, N.; Smirnov, I.; Timofeev, E.; Kaluzhny, D.; Chuvilin, A.; Klinov, D.; Varizhuk, A.; et al. Comparison of the “chemical” and “Structural” Approaches to the Optimization of the Thrombin-Binding Aptamer. PLoS ONE 2014, 9, e89383. [Google Scholar] [CrossRef]




| Name | Sequence, 5′→3′ | Length, nt | Mw, Da |
|---|---|---|---|
| S43, oligotarget | GTA TAC GTT GCT GCA CCT CCC GCA CCC GGT GCG ATT CTG CGT CCA GTT TCC GTC CCC TCT CG | 62 | 18827.1 |
| Z43, oligosensor | GGA GGT GCA GCA ACG TAT AC-(Biotin-TEG) | 20 | 6761.1 |
| SR, random oligotarget | TTC GAC ACT GCT AAA ATC ATT AAT CAA CCT GGA TAT TCT CTC GTG TTC TAT GCG TCT CTC AA | 62 | 18902.4 |
| ZR, random oligosensor | ACC GGT TAC CGC CTC CAC TG-(Biotin-TEG) | 20 | 6583.9 |
| ΔdPAA−GA, nm | ΔdAPTES, nm | ΔdED, nm | (ΔdED−ΔdPAA−GA)/ ΔdPAA-GA, % | (ΔdED−ΔdAPTES)/ ΔdAPTES, % |
|---|---|---|---|---|
| 0.14 | 0.19 | 0.31 | 121 | 63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sizova, S.; Shakurov, R.; Mitko, T.; Shirshikov, F.; Solovyeva, D.; Konopsky, V.; Alieva, E.; Klinov, D.; Bespyatykh, J.; Basmanov, D. The Elaboration of Effective Coatings for Photonic Crystal Chips in Optical Biosensors. Polymers 2022, 14, 152. https://doi.org/10.3390/polym14010152
Sizova S, Shakurov R, Mitko T, Shirshikov F, Solovyeva D, Konopsky V, Alieva E, Klinov D, Bespyatykh J, Basmanov D. The Elaboration of Effective Coatings for Photonic Crystal Chips in Optical Biosensors. Polymers. 2022; 14(1):152. https://doi.org/10.3390/polym14010152
Chicago/Turabian StyleSizova, Svetlana, Ruslan Shakurov, Tatiana Mitko, Fedor Shirshikov, Daria Solovyeva, Valery Konopsky, Elena Alieva, Dmitry Klinov, Julia Bespyatykh, and Dmitry Basmanov. 2022. "The Elaboration of Effective Coatings for Photonic Crystal Chips in Optical Biosensors" Polymers 14, no. 1: 152. https://doi.org/10.3390/polym14010152
APA StyleSizova, S., Shakurov, R., Mitko, T., Shirshikov, F., Solovyeva, D., Konopsky, V., Alieva, E., Klinov, D., Bespyatykh, J., & Basmanov, D. (2022). The Elaboration of Effective Coatings for Photonic Crystal Chips in Optical Biosensors. Polymers, 14(1), 152. https://doi.org/10.3390/polym14010152