UV-LED as a New Emerging Tool for Curable Polyurethane Acrylate Hydrophobic Coating
Abstract
:1. Introduction
2. Materials and Methods
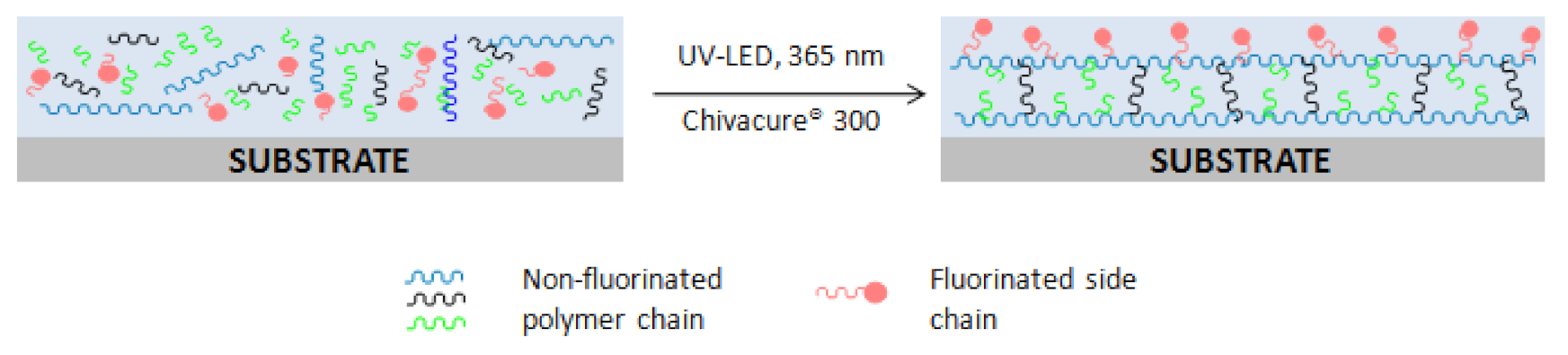
2.1. Preparation of UV-LED Polyurethane Acrylate
2.2. Characterisations of the UV-LED Polyurethane Acrylate
3. Results and Discussion
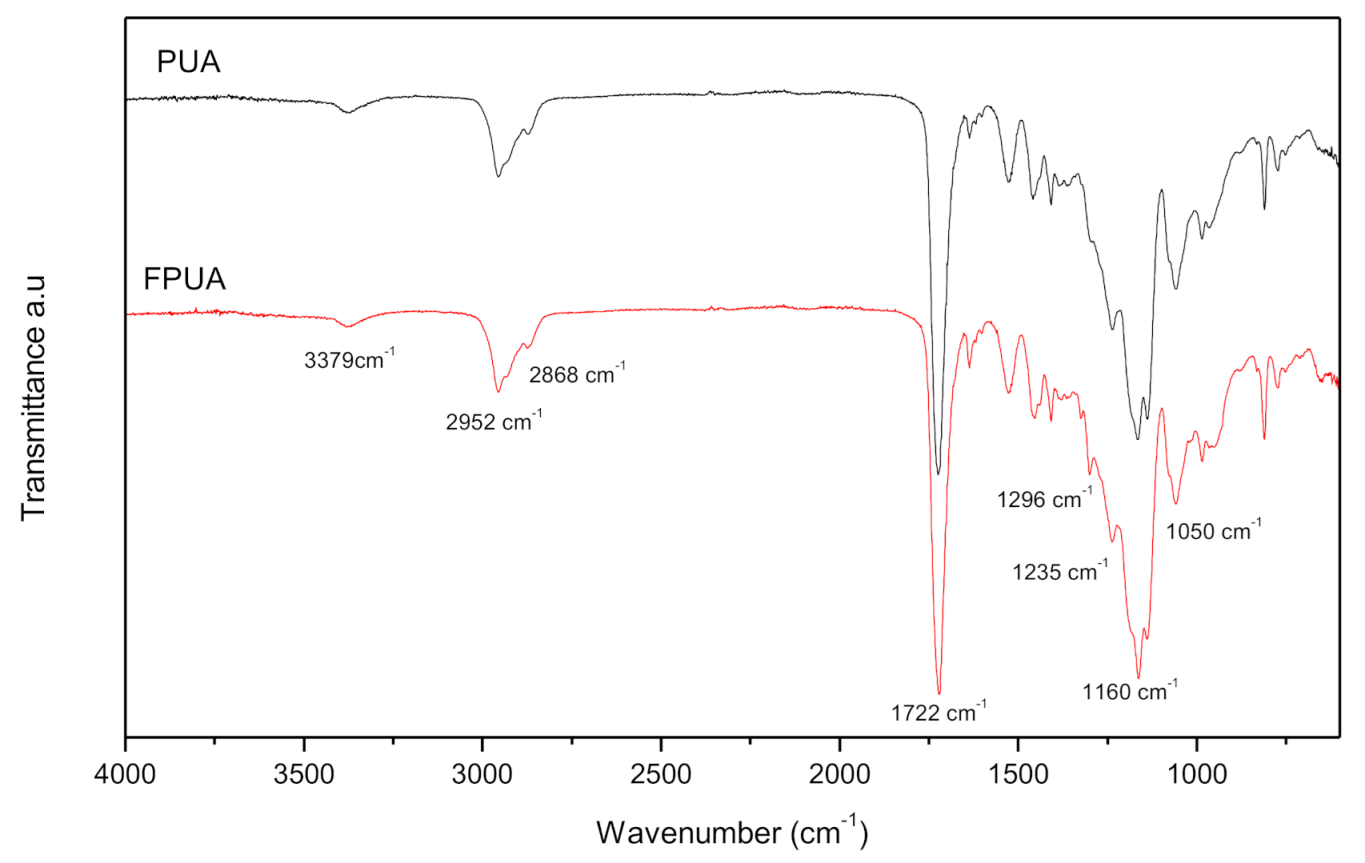
3.1. Chemical Analysis and Curing Behaviour
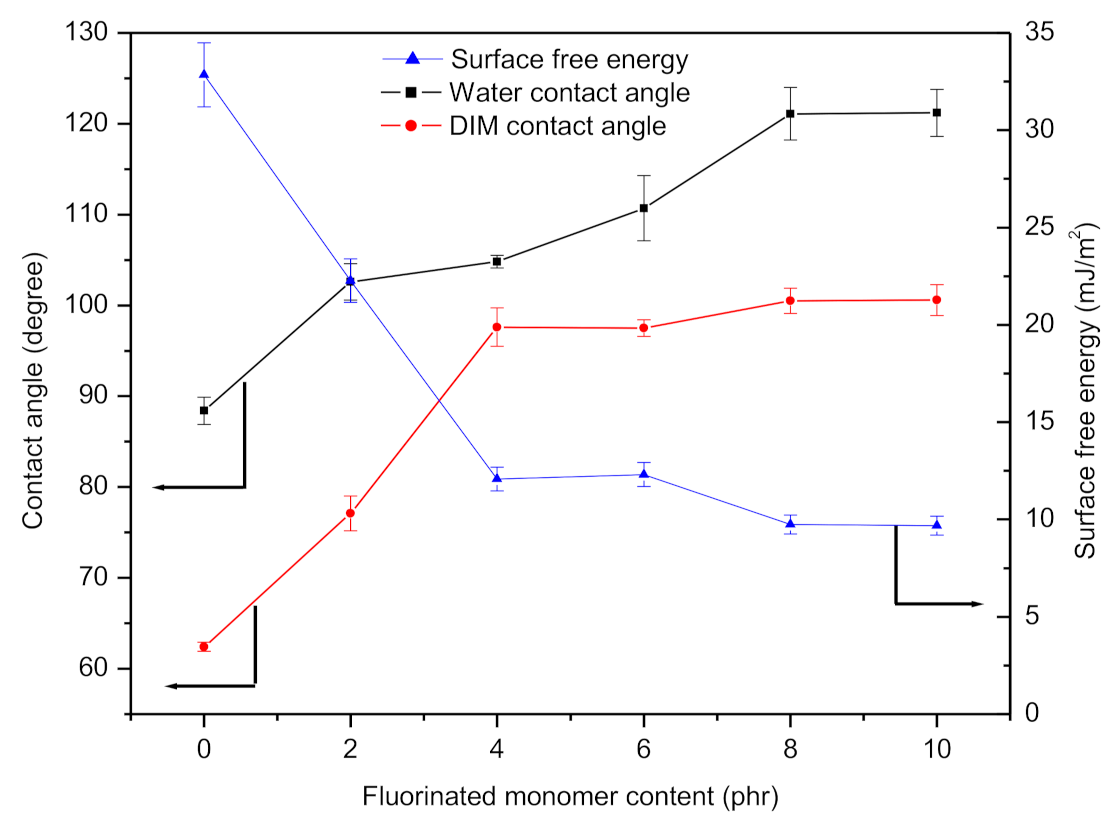
3.2. Contact Angle and Surface Free Energy
3.3. Surface Morphology
3.4. Surface Roughness
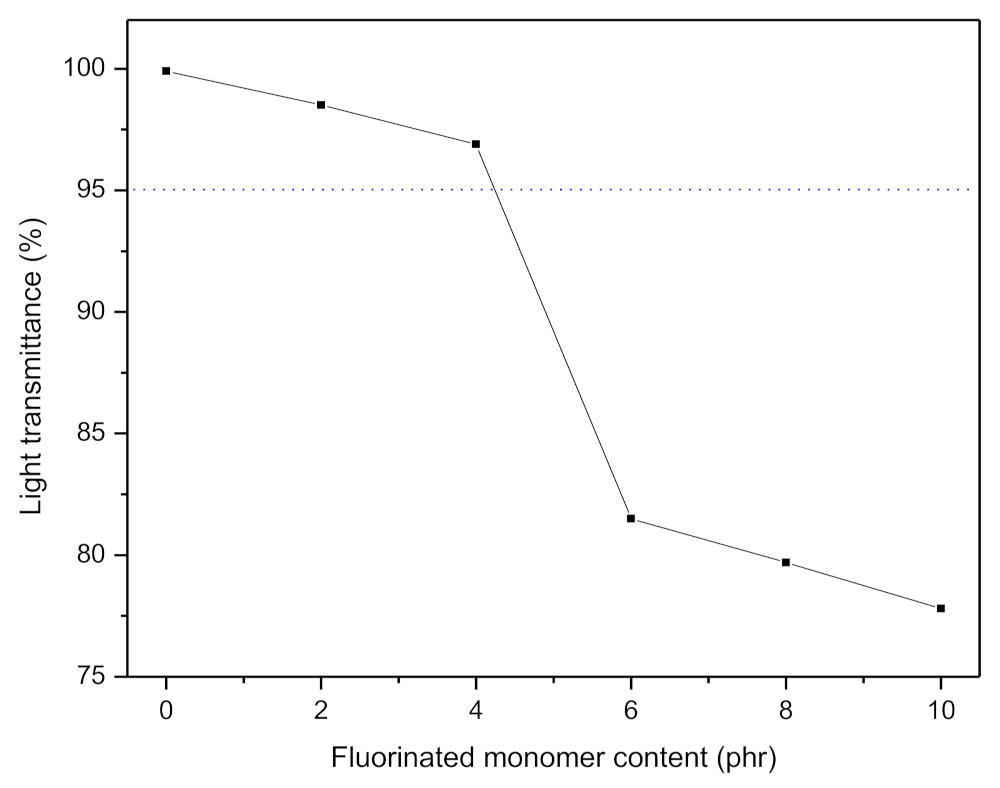
3.5. Optical Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yoshimitsu, Z.; Watanabe, T.; Hashimoto, K. Effects of Surface Structure on the Hydrophobicity and Sliding Behavior of Water Droplets. Langmuir 2002, 18, 5818–5822. [Google Scholar] [CrossRef]
- Ha, J.W.; Park, I.J.; Lee, S.B. Hydrophobicity and sliding behavior of liquid droplets on the fluorinated latex films. Macromolecules 2005, 38, 736–744. [Google Scholar] [CrossRef]
- Boinovich, L.B.; Emelyanenko, A.M. Hydrophobic materials and coatings: Principles of design, properties and applications. Russ. Chem. Rev. 2008, 77, 583–600. [Google Scholar] [CrossRef]
- Latthe, S.S.; Nadargi, D.Y.; Rao, A.V. TMOS based water repellent silica thin films by co-precursor method using TMES as a hydrophobic agent. Appl. Surf. Sci. 2009, 255, 3600–3604. [Google Scholar] [CrossRef]
- Lalevée, J.; El-Roz, M.; Allonas, X.; Fouassier, J.P. Surface modification of a UV curable acrylate coating: In situ introduction of hydrophobic properties. Prog. Org. Coat. 2009, 65, 457–461. [Google Scholar] [CrossRef]
- Faustini, M.; Nicole, L.; Boissière, C.; Innocenzi, P.; Sanchez, C.; Grosso, D. Hydrophobic, antireflective, self-cleaning, and antifogging sol-gel coatings: An example of multifunctional nanostructured materials for photovoltaic cells. Chem. Mater. 2010, 22, 4406–4413. [Google Scholar] [CrossRef]
- Nadargi, D.Y.; Gurav, J.L.; El-Hawi, N.; Rao, A.V.; Koebel, M. Synthesis and characterization of transparent hydrophobic silica thin films by single step sol-gel process and dip coating. J. Alloys Compd. 2010, 496, 436–441. [Google Scholar] [CrossRef]
- Subasri, R.; Madhav, C.S.; Somaraju, K.R.C.; Padmanabham, G. Decorative, hydrophobic sol-gel coatings densified using near-infrared radiation. Surf. Coatings Technol. 2012, 206, 2417–2421. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, L.; Han, D.; Li, W.; Chen, Y. Preparation and Hydrophobicity Failure Behavior of Two Kinds of Fluorine-containing Acrylic Polyurethane Coatings. RSC Adv. 2015, 5, 95230–95239. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, L.; Liu, J.; Wang, R.; Wang, H.; Tian, Q.; Li, X. Hydrophobic films of acrylic emulsion by incorporation of fluorine-based copolymer prepared through the RAFT emulsion copolymerization. J. Fluor. Chem. 2016, 183, 82–91. [Google Scholar] [CrossRef]
- Zhang, X.; Sato, O.; Taguchi, M. Self-Cleaning Particle Coating with AntiReflection Properties. Chem. Mater. 2005, 696–700. [Google Scholar] [CrossRef]
- Zhu, J.; Hsu, C.M.; Yu, Z.; Fan, S.; Cui, Y. Nanodome solar cells with efficient light management and self-cleaning. Nano Lett. 2010, 10, 1979–1984. [Google Scholar] [CrossRef]
- Park, Y.-B.; Im, H.; Im, M.; Choi, Y.-K. Self-Sleaning Effect of Highly Water-Repellent Microshell Structures for Solar Cell Applications. J. Mater. Chem. 2011, 21, 633. [Google Scholar] [CrossRef]
- Suradi, S.S.; Naemuddin, N.H.; Hashim, S.; Adrus, N. Impact of carboxylation and hydrolysis functionalisations on the anti-oil staining behaviour of textiles grafted with poly(N-isopropylacrylamide) hydrogel. RSC Adv. 2018, 8, 13423–13432. [Google Scholar] [CrossRef] [Green Version]
- Brassard, J.-D.; Sarkar, D.K.; Perron, J. Fluorine Based Superhydrophobic Coatings. Appl. Sci. 2012, 2, 453–464. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.G.; Ham, D.S.; Lee, D.Y.; Bong, H.; Cho, K. Transparent Superhydrophobic/Translucent Superamphiphobic Coatings based on Silica-Fluoropolymer Hybrid Nanoparticles. Langmuir 2013, 29, 15051–15057. [Google Scholar] [CrossRef] [PubMed]
- Çanak, T.Ç.; Serhatli, I.E. Synthesis of fluorinated urethane acrylate based UV-curable coatings. Prog. Org. Coat. 2013, 76, 388–399. [Google Scholar] [CrossRef]
- Ganesh, V.A.; Nair, A.S.; Raut, H.K.; Tan, T.T.Y.; He, C.; Ramakrishna, S.; Xu, J. Superhydrophobic fluorinated POSS–PVDF-HFP nanocomposite coating on glass by electrospinning. J. Mater. Chem. 2012, 22, 18479–18485. [Google Scholar] [CrossRef]
- Shin, M.; Lee, Y.; Rahman, M.; Kim, H. Synthesis and properties of waterborne fluorinated polyurethane-acrylate using a solvent-/emulsifier-free method. Polymer 2013, 54, 4873–4882. [Google Scholar] [CrossRef]
- Li, K.; Zeng, X.; Li, H.; Lai, X. Fabrication and Characterization of Stable Superhydrophobic Fluorinated-Polyacrylate/Silica Hybrid Coating. Appl. Surf. Sci. 2014, 298, 214–220. [Google Scholar] [CrossRef]
- Yao, W.; Li, Y.; Huang, X. Fluorinated poly(meth)acrylate: Synthesis and properties. Polymer 2014, 55, 6197–6211. [Google Scholar] [CrossRef] [Green Version]
- Decker, C. Kinetic Study and New Applications of UV radiation Curing. Macromol. Rapid Commun. 2002, 23, 1067–1093. [Google Scholar] [CrossRef]
- Schwalm, R. UV Coatings. Elsevier: Amsterdam, The Netherlands, 2007; ISBN 9780444529794. [Google Scholar]
- Decker, C.; Masson, F.; Schwalm, R. Weathering resistance of waterbased UV-cured polyurethane-acrylate coatings. Polym. Degrad. Stab. 2004, 83, 309–320. [Google Scholar] [CrossRef]
- Zhang, Y.; Asif, A.; Shi, W. Highly branched polyurethane acrylates and their waterborne UV curing coating. Prog. Org. Coat. 2011, 71, 295–301. [Google Scholar] [CrossRef]
- Hubert, M.; Dimas, C. High-power UV LED array for curing photoadhesives. In Applications of Photonic Technology; International Society for Optics and Photonics: Quebec, QC, Canada, 2003; Volume 5260, pp. 163–168. [Google Scholar]
- Ayub, N.F.; Hashim, S.; Jamaluddin, J.; Adrus, N. New UV LED curing approach for polyacrylamide and poly(N-isopropylacrylamide) hydrogels. New J. Chem. 2017, 41, 5613–5619. [Google Scholar] [CrossRef]
- Dietlin, C.; Schweizer, S.; Xiao, P.; Zhang, J.; Morlet-savary, F.; Graff, B.; Fouassier, J.-P.; Lalevée, J. Polymer Chemistry Photopolymerization upon LEDs: New photoinitiating systems and strategies. Polym. Chem. 2015, 6, 3895–3912. [Google Scholar] [CrossRef]
- Landry, V.; Blanchet, P.; Boivin, G.; Bouffard, J.-F.; Vlad, M. UV-LED Curing Efficiency of Wood Coatings. Coatings 2015, 5, 1019–1033. [Google Scholar] [CrossRef] [Green Version]
- Xie, R.; Li, D. An inexpensive stereolithography technology with high power UV-LED light. Rapid Prototyp. J. 2011, 17, 441–450. [Google Scholar] [CrossRef]
- Tasic, S.; Bozic, B.; Dunjic, B. Synthesis of new hyperbranched urethane-acrylates and their evaluation in UV-curable coatings. Prog. Org. Coat. 2004, 51, 321–328. [Google Scholar] [CrossRef]
- Kunwong, D.; Sumanochitraporn, N.; Kaewpirom, S. Curing behavior of a UV-curable coating based on urethane acrylate oligomer: The influence of reactive monomers. Songklanakarin J. Sci. Technol. 2011, 33, 201–207. [Google Scholar]
- Lin, Y.H.; Liao, K.H.; Chou, N.K.; Wang, S.S.; Chu, S.H.; Hsieh, K.H. UV-Curable Low-Surface-Energy Fluorinated Poly(urethane-acrylate)s for Biomedical Applications. Eur. Polym. J. 2008, 44, 2927–2937. [Google Scholar] [CrossRef]
- Lee, B.-H.; Choi, J.-H.; Kim, H.-J. Coating performance and characteristics for UV-curable aliphatic urethane acrylate coatings containing norrish type I photoinitiators. J. Coat. Technol. Res. 2006, 3, 221–229. [Google Scholar] [CrossRef]
- Džunuzović, E.S.; Tasić, S.V.; Božić, B.R.; Džunuzović, J.V.; Dunjić, B.M.; Jeremić, K.B. Mechanical and thermal properties of UV cured mixtures of linear and hyperbranched urethane acrylates. Prog. Org. Coat. 2012, 74, 158–164. [Google Scholar] [CrossRef]
- Park, J.; Lee, Y.; Park, H.; Kim, H. Preparation and Properties of UV-Curable Fluorinated Polyurethane Acrylates. J. Appl. Polym. 2014, 40603, 1–8. [Google Scholar] [CrossRef]
- Llorente, O.; Fernández-Berridi, M.J.; González, A.; Irusta, L. Study of the crosslinking process of waterborne UV curable polyurethane acrylates. Prog. Org. Coat. 2016, 99, 437–442. [Google Scholar] [CrossRef]
- Strongone, V.; Bartoli, M.; Jagdale, P.; Arrigo, R.; Tagliaferro, A.; Malucelli, G. Preparation and characterization of UV-LED curable acrylic films containing biochar and/or multiwalled carbon nanotubes: Effect of the filler loading on the rheological, thermal and optical properties. Polymers 2020, 12, 796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malucelli, G. Synthesis and Characterization of UV-LED Curable Nanocomposite Coatings. Curr. Org. Chem. 2017, 21, 2314–2321. [Google Scholar] [CrossRef]
- Ghazali, S.K.; Azim, F.S.S.; Adrus, N.; Jamaluddin, J. The effectiveness of uv-led photopolymerisation over conventional uv-mercury for polyurethane acrylate coating. J. Photopolym. Sci. Technol. 2019, 32, 705–710. [Google Scholar] [CrossRef]
- Żenkiewicz, M. Methods for the calculation of surface free energy of solids. J. Achiev. Mater. Manuf. Eng. 2007, 24, 137–145. [Google Scholar]
- Forch, R.; Schönherr, H.; Jenkins, A.T.A. Appendix C: Contact Angle Goniometry. In Surface Design: Applications in Bioscience and Nanotechnology; John Wiley & Sons: Hoboken, NJ, USA, 2009; pp. 471–473. ISBN 9783527407897. [Google Scholar]
- Park, I.J.; Lee, S.-B.; Choi, C.K. Surface properties of the fluorine-containing graft copolymer of poly((perfluoroalkyl)ethylmethacrylate)-g-poly(methyl methacrylate). Macromolecules 1998, 31, 7555–7558. [Google Scholar] [CrossRef]
- Li, K.; Wu, P.; Han, Z. Preparation and surface properties of fluorine-containing diblock copolymers. Polymer 2002, 43, 4079–4086. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, X.; Duan, H.; Ma, J.; Ma, Y. Synthesis and Characterization of nano-SiO2 modified fluorine-containing polyacrylate emulsifier-free emulsion. Appl. Surf. Sci. 2015, 331, 504–511. [Google Scholar] [CrossRef]
- Gan, D.; Mueller, A.; Wooley, K.L. Amphiphilic and Hydrophobic Surface Patterns Generated from Hyperbranched Fluoropolymer/Linear Polymer Networks: Minimally Adhesive Coatings via the Crosslinking of Hyperbranched Fluoropolymers. J. Polym. Sci. Part A Polym. Chem. 2003, 41, 3531–3540. [Google Scholar] [CrossRef]
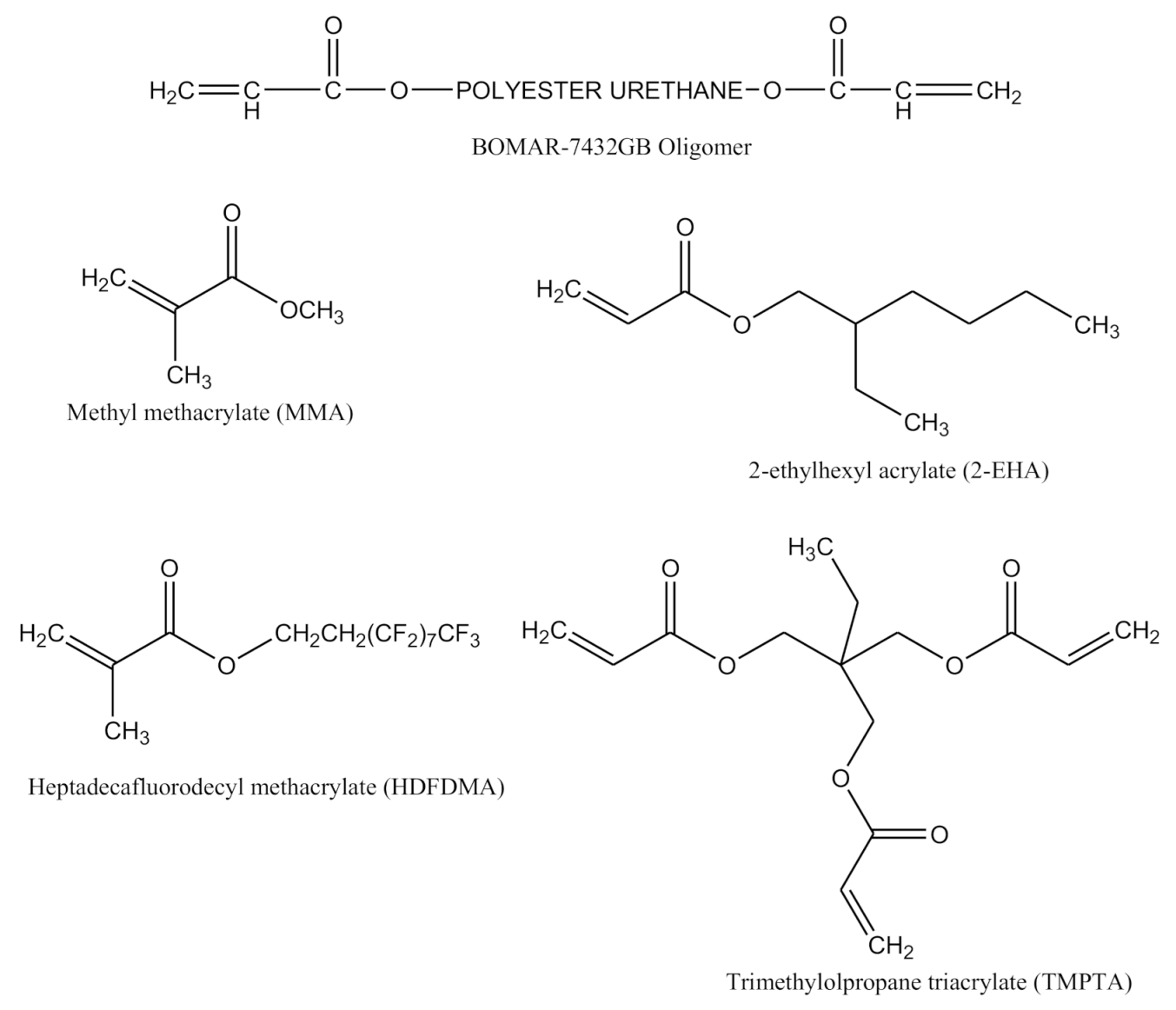


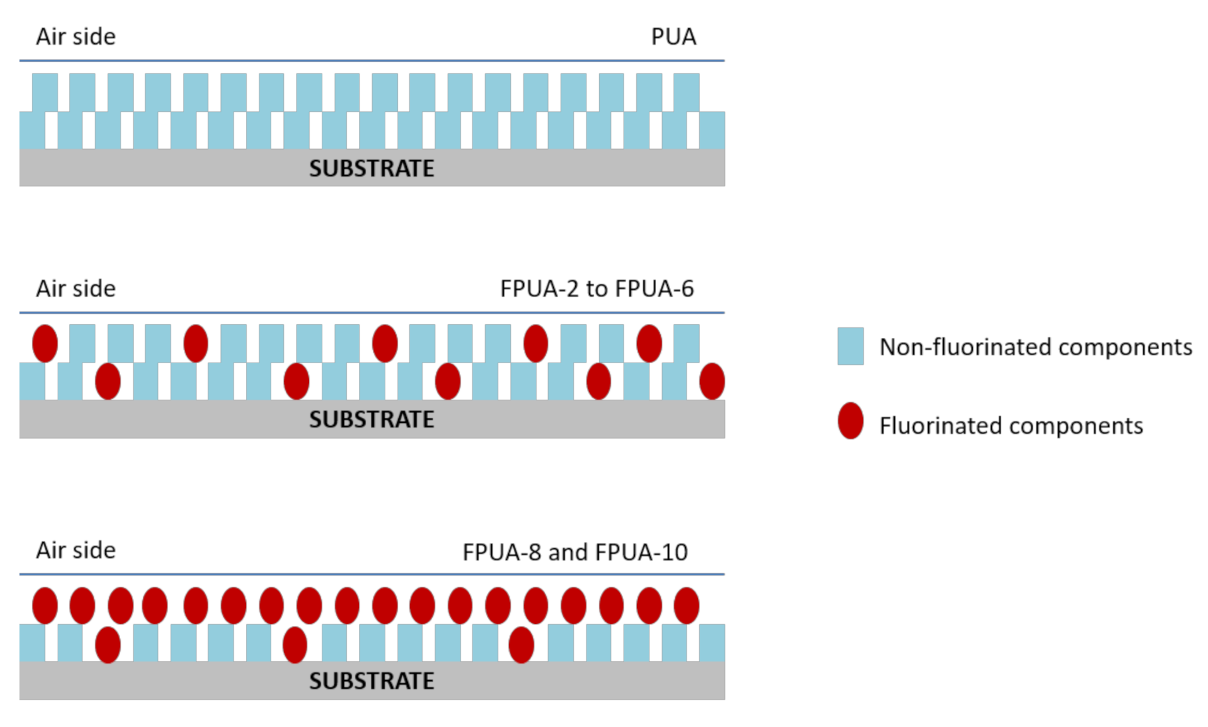

| Sample Designation | Formulations | |||||
|---|---|---|---|---|---|---|
| Oligomer (wt %) | Chivacure® 300 (wt %) | MMA (wt %) | 2-EHA (wt %) | TMPTA (wt %) | HDFDMA (phr 1) | |
| PUA | 70 | 4 | 13 | 8 | 5 | 0 |
| FPUA-2 | 70 | 4 | 13 | 8 | 5 | 2 |
| FPUA-4 | 70 | 4 | 13 | 8 | 5 | 4 |
| FPUA-6 | 70 | 4 | 13 | 8 | 5 | 6 |
| FPUA-8 | 70 | 4 | 13 | 8 | 5 | 8 |
| FPUA-10 | 70 | 4 | 13 | 8 | 5 | 10 |
| FPUA-12 | 70 | 4 | 13 | 8 | 5 | 12 |
| Sample Designation | Observation of the Uncured Solution | Conversion (%) | Gel Fraction (%) |
|---|---|---|---|
| PUA | Homogeneous | 98 | 95 |
| FPUA-2 | Homogeneous | 98 | 94 |
| FPUA-4 | Homogeneous | 98 | 94 |
| FPUA-6 | Homogeneous | 98 | 94 |
| FPUA-8 | Homogeneous | 97 | 94 |
| FPUA-10 | Homogeneous | 96 | 93 |
| FPUA-12 1 | Heterogeneous | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghazali, S.K.; Adrus, N.; Majid, R.A.; Ali, F.; Jamaluddin, J. UV-LED as a New Emerging Tool for Curable Polyurethane Acrylate Hydrophobic Coating. Polymers 2021, 13, 487. https://doi.org/10.3390/polym13040487
Ghazali SK, Adrus N, Majid RA, Ali F, Jamaluddin J. UV-LED as a New Emerging Tool for Curable Polyurethane Acrylate Hydrophobic Coating. Polymers. 2021; 13(4):487. https://doi.org/10.3390/polym13040487
Chicago/Turabian StyleGhazali, Siti Khairunisah, Nadia Adrus, Rohah A. Majid, Fathilah Ali, and Jamarosliza Jamaluddin. 2021. "UV-LED as a New Emerging Tool for Curable Polyurethane Acrylate Hydrophobic Coating" Polymers 13, no. 4: 487. https://doi.org/10.3390/polym13040487








