Electrospun Poly(methyl methacrylate)/TiO2 Composites for Photocatalytic Water Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Electrospinning
2.3. Characterization
2.4. Photocatalytic Activity
3. Results and Discussion
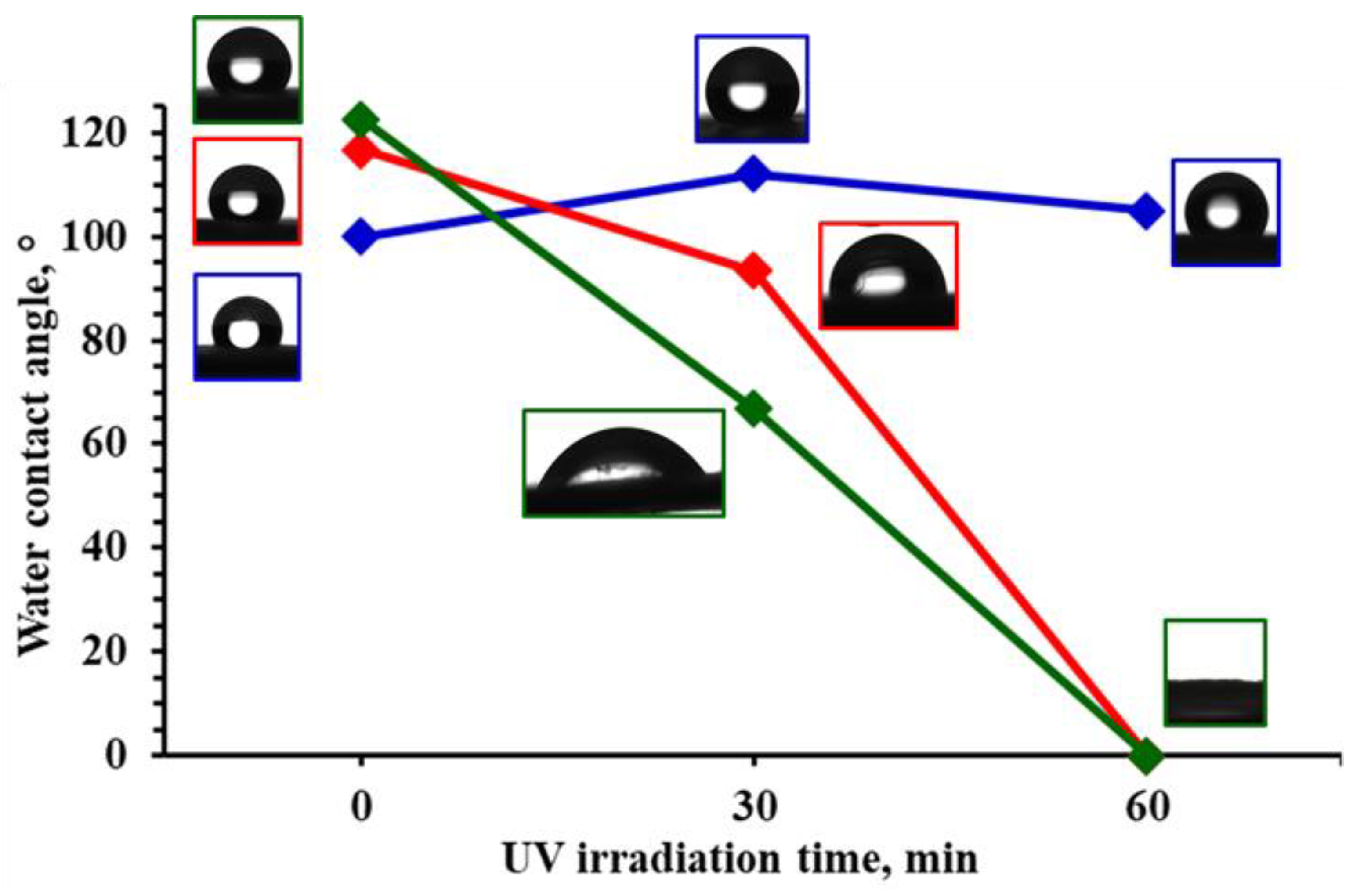
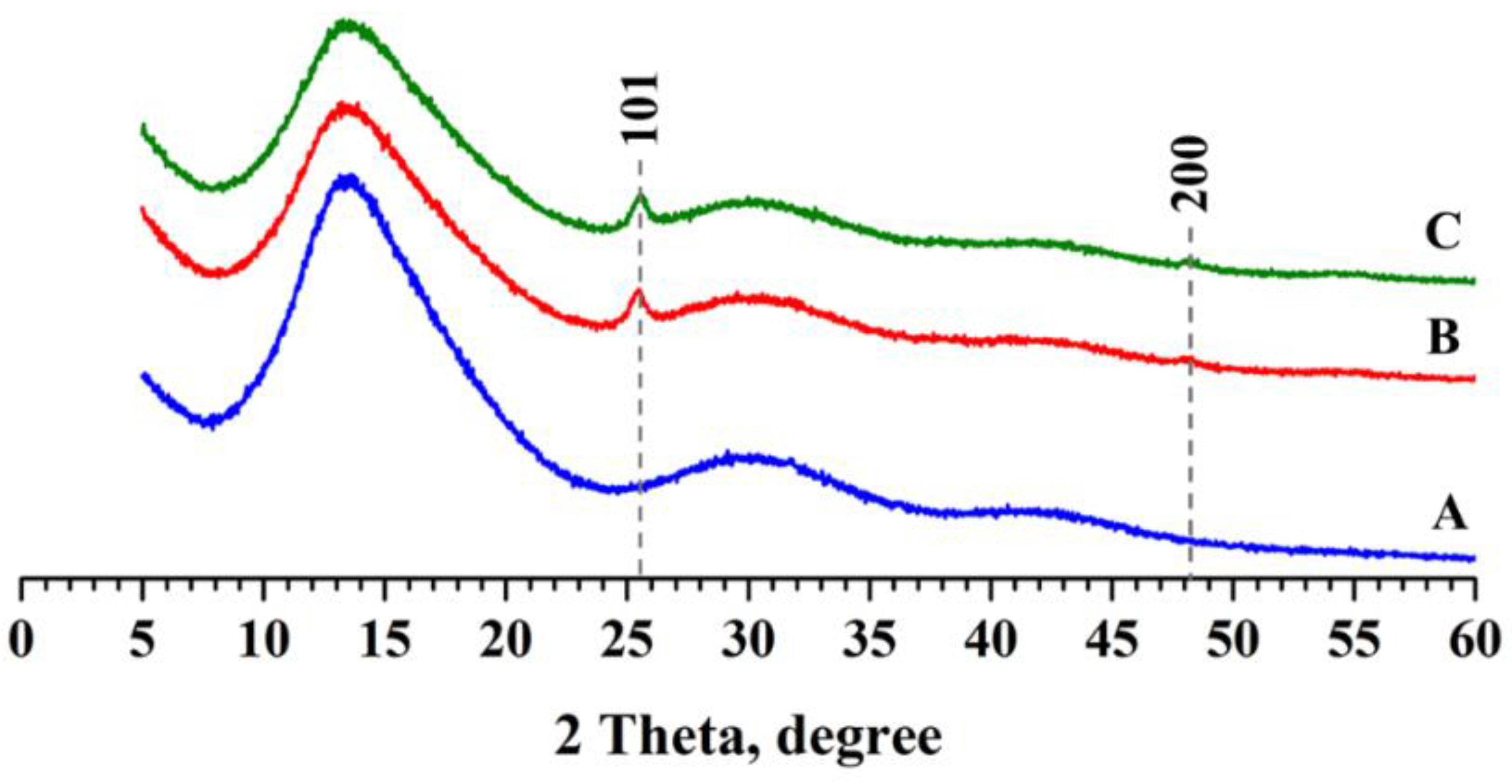
3.1. PMMA/TiO2 Composites Fabrication and Characterization
3.2. Tensile Properties of the PMMA/TiO2 Composites
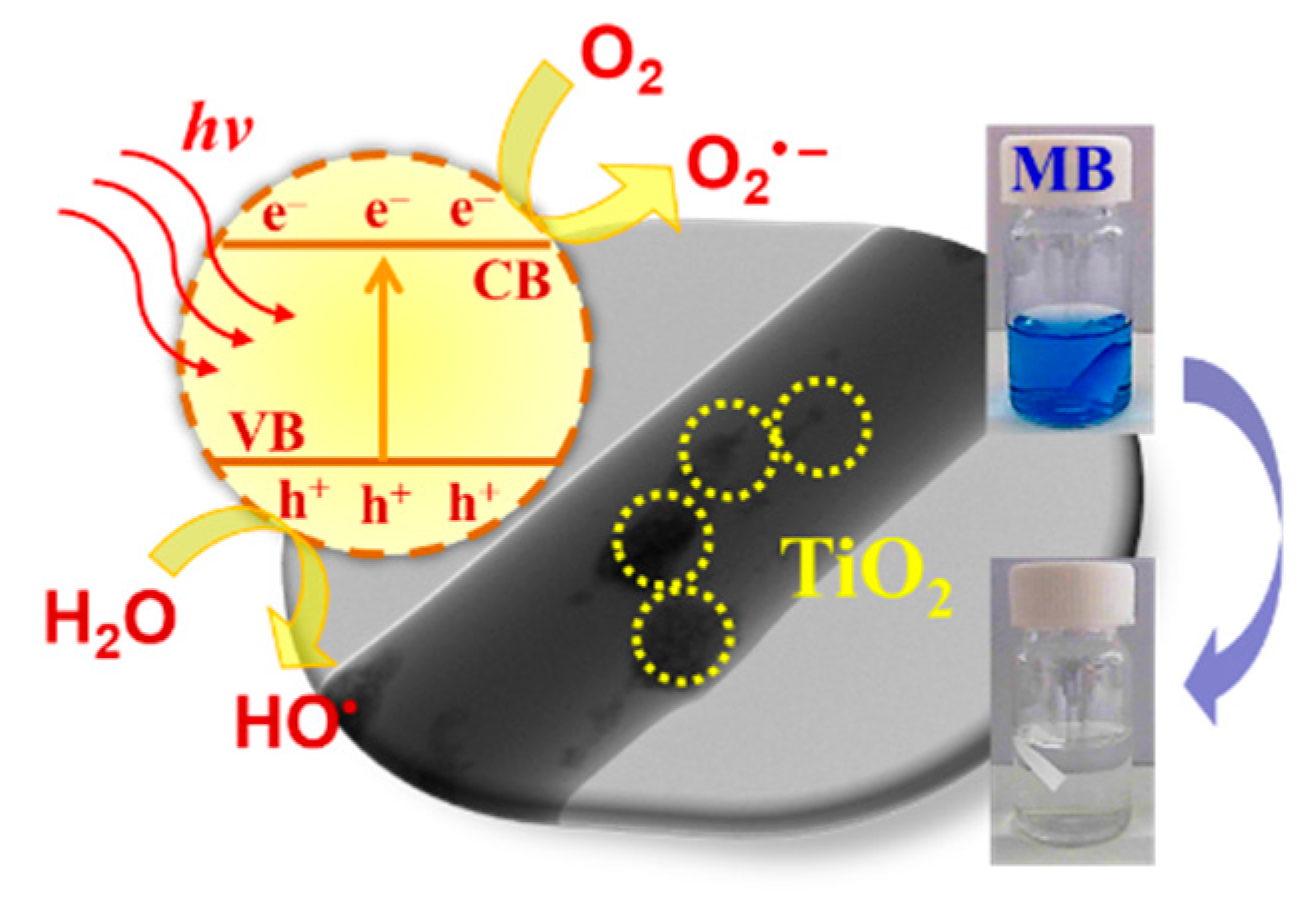
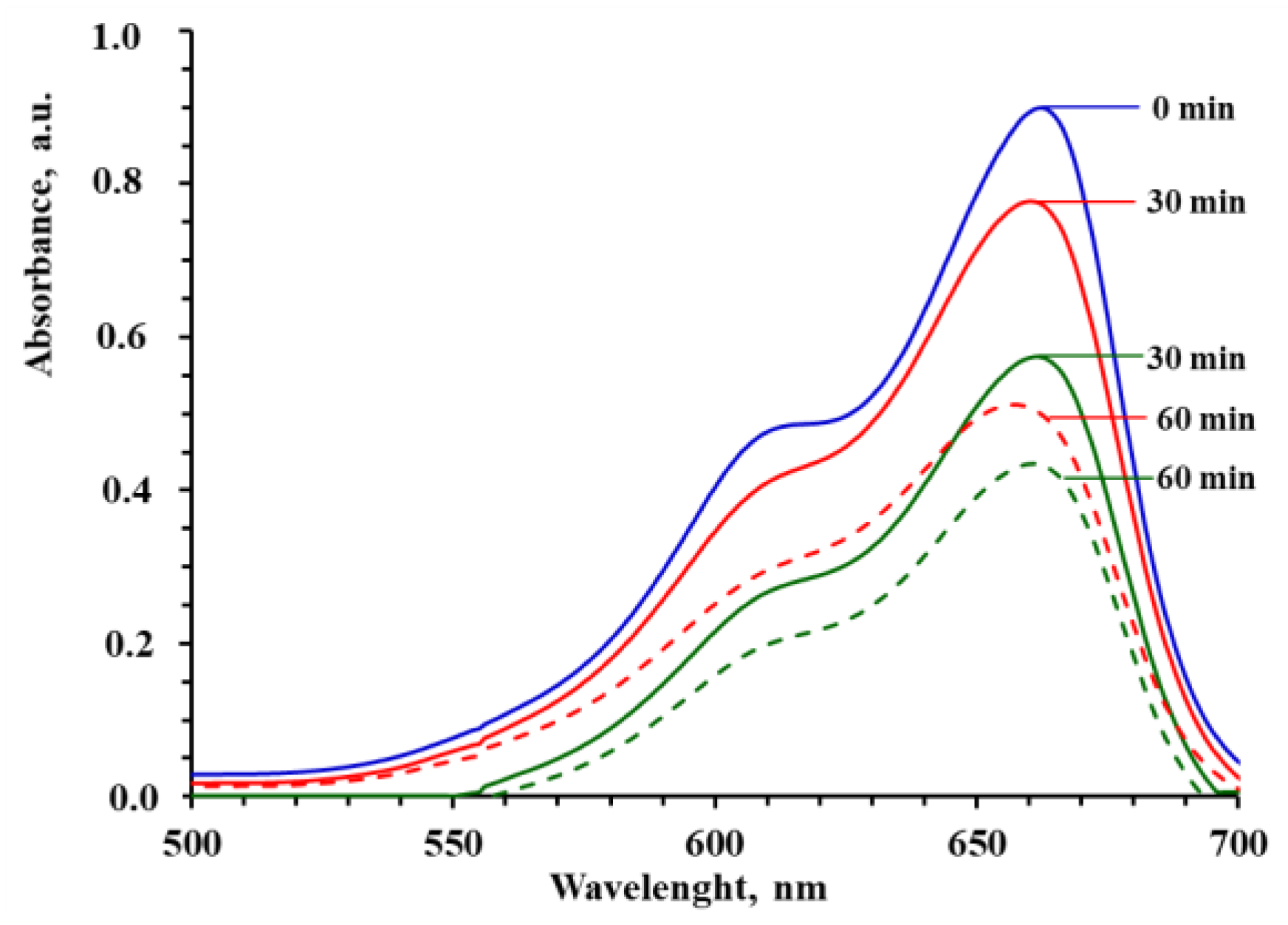
3.3. Photocatalytic Degradation of MB and Reusability of PMMA/TiO2 Composites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liao, Y.; Loh, C.-H.; Tian, M.; Wang, R.; Fane, A.G. Progress in electrospun polymeric nanofibrous membranes for water treatment: Fabrication, modification and applications. Prog. Polym. Sci. 2018, 77, 69–94. [Google Scholar] [CrossRef]
- Asmatulu, R.; Khan, W.S. Chapter 7—Electrospun nanofibers for filtration applications. In Synthesis and Applications of Electrospun Nanofibers (Micro and Nano Technologies), 1st ed.; Asmatulu, R., Khan, W.S., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 135–152. [Google Scholar]
- Ray, S.S.; Chen, S.-S.; Nguyen, N.C.; Nguyen, H.T. Chapter 9—Electrospinning: A Versatile Fabrication Technique for Nanofibrous Membranes for Use in Desalination. In Nanoscale Materials in Water Purification (Micro and Nano Technologies); Thomas, S., Pasquini, D., Leu, S.-Y., Gopakumar, D.A., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 247–273. [Google Scholar]
- El Nazer, H.E.A.F.A.H.; Gaballah, S.T. Nanocomposites and Its Importance in Photocatalysis. In Nanocomposites for Visible Light-induced Photocatalysis; Khan, M., Pradhan, D., Sohn, Y., Eds.; Springer Series on Polymer and Composite Materials; Springer: Berlin/Heidelberg, Germany, 2017; pp. 41–67. [Google Scholar]
- Pirilä, M.; Saouabe, M.; Ojala, S.; Rathnayake, B.; Drault, F.; Valtanen, A.; Huuhtanen, M.; Brahmi, R.; Keiski, R.L. Photocatalytic degradation of organic pollutants in wastewater. Top. Catal. 2015, 58, 1085–1099. [Google Scholar] [CrossRef]
- Rajeshwar, K.; Osugi, M.E.; Chanmanee, W.; Chenthamaraksha, C.R.; Zanoni, M.V.B.; Kajitvichyanukul, P.; Krishnan-Ayer, R. Heterogeneous photocatalytic treatment of organic dyes in air and aqueous media. J. Photochem. Photobiol. C Photochem. Rev. 2008, 9, 171–192. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: Design and applications. J. Photochem. Photobiol. C Photochem. Rev. 2008, 13, 169–189. [Google Scholar] [CrossRef]
- Linsebigler, A.L.; Lu, G.; Yates, J.T. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Dahl, M.; Liu, Y.; Yin, Y. Composite titanium dioxide nanomaterials. Chem. Rev. 2014, 114, 9853–9889. [Google Scholar] [CrossRef]
- Ali, U.; Abd Karim, K.J.B.; Buang, N.A. A review of the properties and applications of poly (methyl methacrylate) (PMMA). Polym. Rev. 2015, 55, 678–705. [Google Scholar] [CrossRef]
- Cantarella, M.; Sanz, R.; Buccheri, M.A.; Ruffino, F.; Rappazzo, G.; Scalese, S.; Impellizzeri, G.; Romano, L.; Privitera, V. Immobilization of nanomaterials in PMMA composites for photocatalytic removal of dyes, phenols and bacteria from water. J. Photochem. Photobiol. A Chem. 2016, 321, 1–11. [Google Scholar] [CrossRef]
- Sengwa, R.J.; Dhatarwal, P. Polymer nanocomposites comprising PMMA matrix and ZnO, SnO2, and TiO2 nanofillers: A comparative study of structural, optical, and dielectric properties for multifunctional technological applications. Opt. Mater. 2021, 113, 110837–110848. [Google Scholar] [CrossRef]
- Nandimath, M.; Bhajantri, R.F.; Naik, J.; Hebbar, V. Effect of Rhodamine 6G dye on chromaticity co-ordinates and photoluminescence properties of TiO2/PMMA polymer nanocomposites for LED applications. J. Lumin. 2019, 207, 571–584. [Google Scholar] [CrossRef]
- Li, C.; Yu, H.; Song, Y.; Liang, H.; Yan, H. Preparation and characterization of PMMA/TiO2 hybrid shell microencapsulated PCMs for thermal energy storage. Energy 2019, 167, 1031–1039. [Google Scholar] [CrossRef]
- Cantarella, M.; Sanz, R.; Buccheri, M.A.; Romano, L.; Privitera, V. PMMA/TiO2 nanotubes composites for photocatalytic removal of organic compounds and bacteria from water. Mater. Sci. Semicond. Process. 2016, 42, 58–61. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Y.; Li, Y.; Zhao, L.; Wang, H.; Song, G.; Tang, G. Microencapsulated phase change materials with TiO2-doped PMMA shell for thermal energy storage and UV-shielding. Sol. Energy Mater. Sol. Cells 2017, 168, 62–68. [Google Scholar] [CrossRef]
- Sugumaran, S.; Bellan, C. Transparent nano composite PVA–TiO2 and PMMA–TiO2 thin films: Optical and dielectric properties. Optik 2014, 125, 5128–5133. [Google Scholar] [CrossRef]
- Koysuren, O.; Koysuren, H.N. Photocatalytic activities of poly(methyl methacrylate)/titanium dioxide nanofiber mat. J. Macromol. Sci. Part A Pure Appl. Chem. 2017, 54, 80–84. [Google Scholar] [CrossRef]
- Korina, E.; Stoilova, O.; Manolova, N.; Rashkov, I. Multifunctional hybrid materials from poly(3-hydroxybutyrate), TiO2 nanoparticles, and chitosan oligomers by combining electrospinning/electrospraying and impregnation. Macromol. Biosci. 2013, 13, 707–716. [Google Scholar] [CrossRef]
- Korina, E.; Stoilova, O.; Manolova, N.; Rashkov, I. Poly(3-hydroxybutyrate)-based hybrid materials with photocatalytic and magnetic properties prepared by electrospinning and electrospraying. J. Mater. Sci. 2014, 49, 2144–2153. [Google Scholar] [CrossRef]
- Korina, E.; Stoilova, O.; Manolova, N.; Rashkov, I. Polymer fibers with magnetic core decorated with titanium dioxide prospective for photocatalytic water treatment. J. Environ. Chem. Eng. 2018, 6, 2075–2084. [Google Scholar] [CrossRef]
- Casaletto, M.P.; Ingo, G.M.; Kaciulis, S.; Mattogno, G.; Pandol, L.; Scavia, G. Surface studies of in vitro biocompatibility of titanium oxide coatings. Appl. Surf. Sci. 2001, 172, 167–177. [Google Scholar] [CrossRef]
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induced amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- Ma, M.; Gupta, M.; Li, Z.; Zhai, L.; Gleason, K.K.; Cohen, R.E.; Rubner, M.F.; Rutledge, G.C. Decorated electrospun fibers exhibiting superhydrophobicity. Adv. Mater. 2007, 19, 255–259. [Google Scholar] [CrossRef]
- Spasova, M.; Stoilova, O.; Manolova, N.; Rashkov, I.; Naydenov, M. Electrospun eco-friendly materials based on poly(3-hydroxybutyrate) and TiO2 with antifungal activity prospective for Esca treatment. Polymers 2020, 12, 1384–1395. [Google Scholar] [CrossRef] [PubMed]
- Nowak, E.; Combes, G.; Hugh Stitt, E.; Pacek, A.W. A comparison of contact angle measurement techniques applied to highly porous catalyst supports. Powder Technol. 2013, 233, 52–64. [Google Scholar] [CrossRef]
- Borisova, I.; Stoilova, O.; Manolova, N.; Rashkov, I. Effect of coating on the mechanical properties of electrospun poly(3-hydroxybutyrate) materials with targeted fibers alignment. J. Polym. Res. 2021, 28, 1–13. [Google Scholar] [CrossRef]




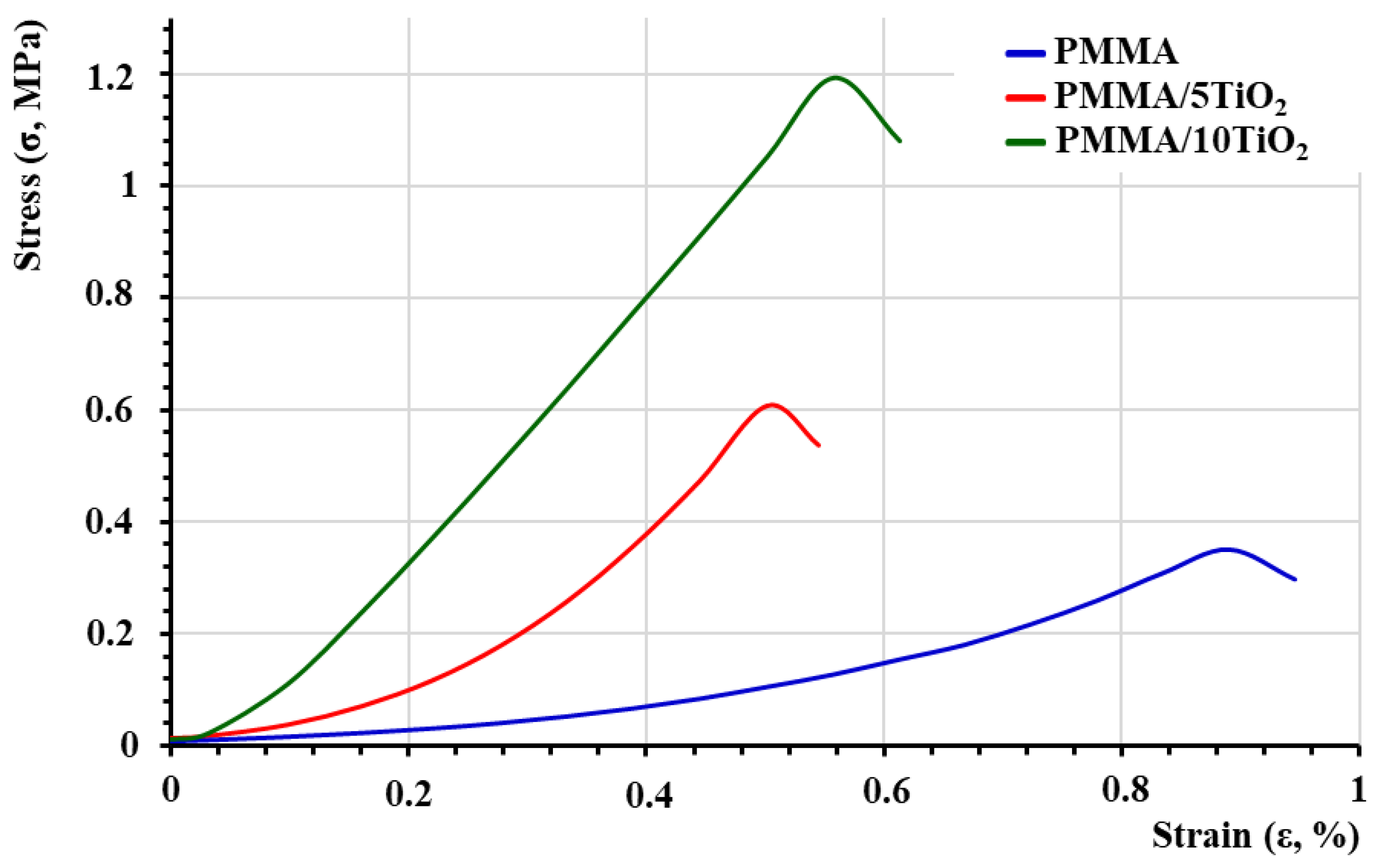

| Samples | Young’s Modulus (E, MPa) | Tensile Strength (σ, MPa) | Elongation at Break (εB, %) |
|---|---|---|---|
| PMMA | 82.4 | 0.50 | 1.95 |
| PMMA/5TiO2 | 237.4 | 0.61 | 0.50 |
| PMMA/10TiO2 | 251.9 | 1.19 | 0.56 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoilova, O.; Manolova, N.; Rashkov, I. Electrospun Poly(methyl methacrylate)/TiO2 Composites for Photocatalytic Water Treatment. Polymers 2021, 13, 3923. https://doi.org/10.3390/polym13223923
Stoilova O, Manolova N, Rashkov I. Electrospun Poly(methyl methacrylate)/TiO2 Composites for Photocatalytic Water Treatment. Polymers. 2021; 13(22):3923. https://doi.org/10.3390/polym13223923
Chicago/Turabian StyleStoilova, Olya, Nevena Manolova, and Iliya Rashkov. 2021. "Electrospun Poly(methyl methacrylate)/TiO2 Composites for Photocatalytic Water Treatment" Polymers 13, no. 22: 3923. https://doi.org/10.3390/polym13223923
APA StyleStoilova, O., Manolova, N., & Rashkov, I. (2021). Electrospun Poly(methyl methacrylate)/TiO2 Composites for Photocatalytic Water Treatment. Polymers, 13(22), 3923. https://doi.org/10.3390/polym13223923








