The Effect of Chitosan Type on Biological and Physicochemical Properties of Films with Propolis Extract
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chitosan Films Preparation
2.2. Chitosan-Propolis Films Preparation
2.3. Antimicrobial Properties of Films
2.4. Oxygen Transmission Rate
2.5. Water Vapor Transmission Rate
2.6. Sorption Experiments
2.7. Sorption Modelling
2.8. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy
3. Results and Discussion
3.1. Antimicrobial Activity
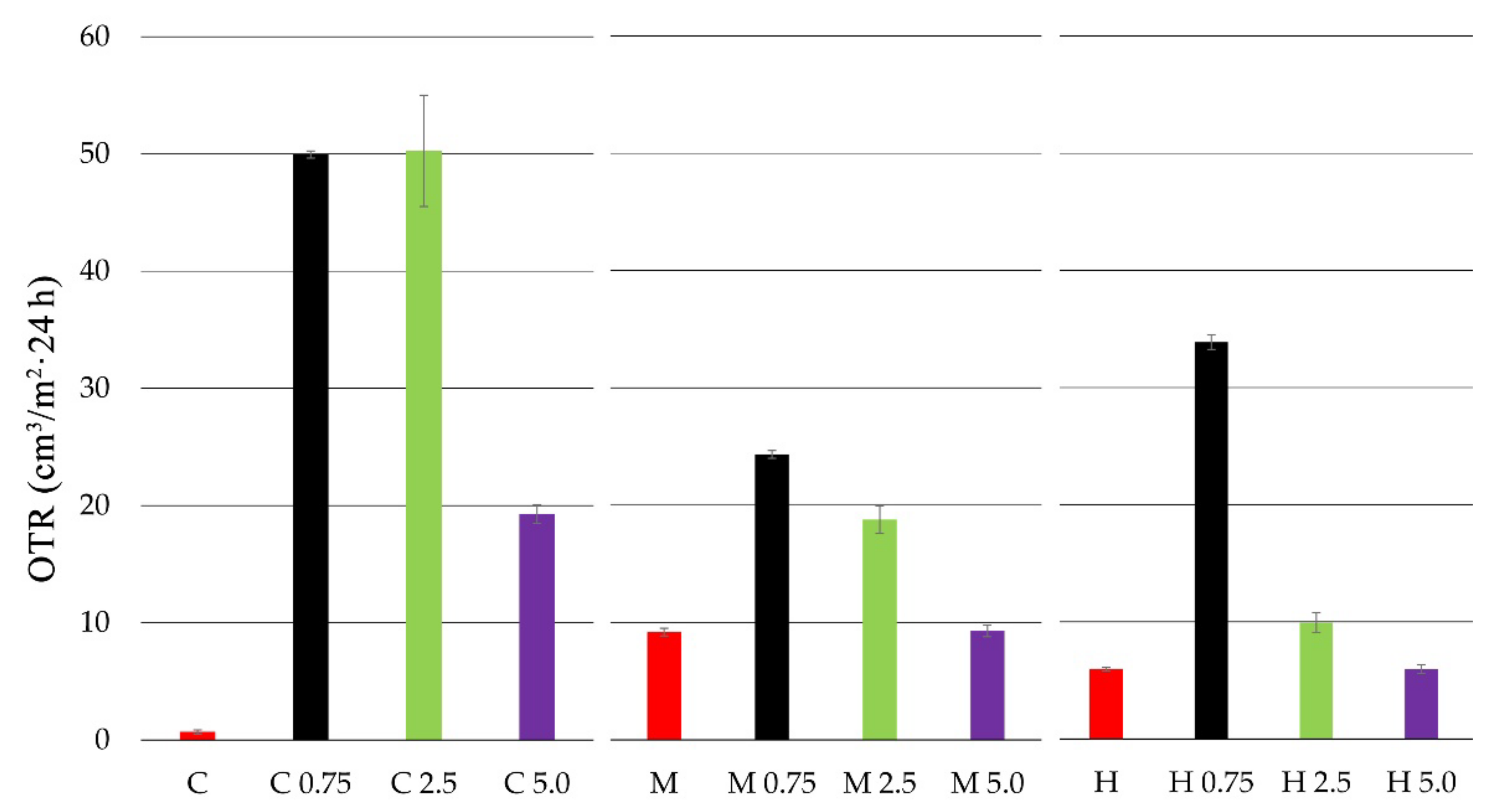
3.2. Oxygen Transmission Rate
3.3. Water Vapor Transmission Rate
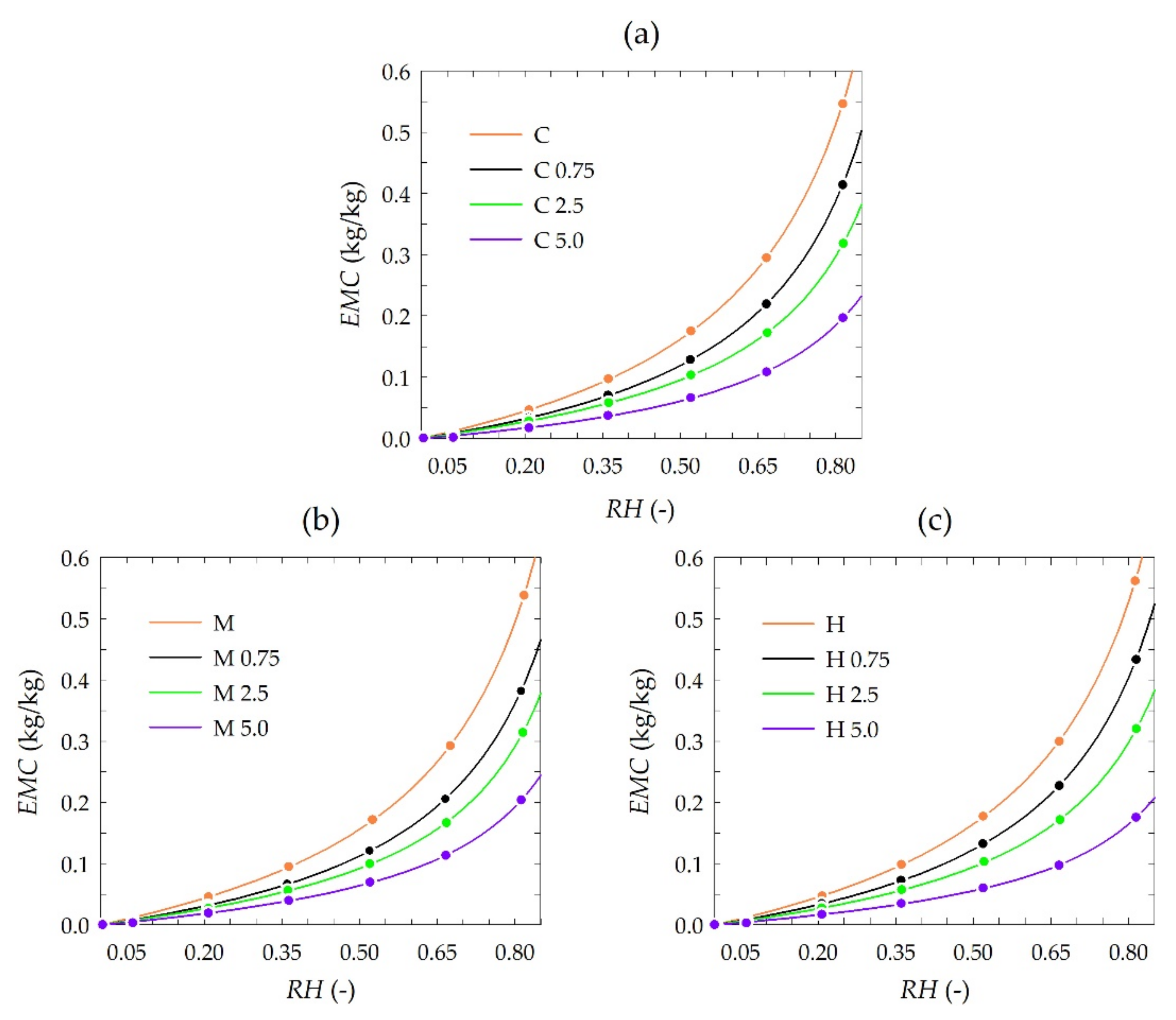
3.4. Sorption Behavior Derived from Experimental GAB Sorption Isotherms
3.5. Attenuated Total Reflectance Fourier Transform Infrared Spectroscopy
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Directive 2019/904 of the European Parliament and of the Council of 5 June 2019 on the Reduction of the Impact of Certain Plastic Products on the Environment; EU: Brussels, Belgium, 2019.
- Sani, M.A.; Azizi-Lalabadi, M.; Tavassoli, M.; Mohammadi, K.; McClements, D.J. Recent advances in the development of smart and active biodegradable packaging materials. Nanomaterials 2021, 11, 1331. [Google Scholar] [CrossRef] [PubMed]
- Junianto; Mametapo, M.M.N.; Aulia, A.F.; Fitriyanti; A’yun, N.Q. Chitosan application as edible packaging raw material. Asian J. Fish. Aquat. Res. 2021, 12, 44–54. [Google Scholar] [CrossRef]
- Fu, B.; Mei, S.; Su, X.; Chen, H.; Zhu, J.; Zheng, Z.; Lin, H.; Dai, C.; Luque, R.; Yang, D.-P. Integrating waste fish scale-derived gelatin and chitosan into edible nanocomposite film for perishable fruits. Int. J. Biol. Macromol. 2021, 191, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Abral, H.; Pratama, A.B.; Handayani, D.; Mahardika, M.; Aminah, I.; Sandrawati, N.; Sugiarti, E.; Muslimin, A.N.; Sapuan, S.M.; Ilyas, R.A. Antimicrobial edible film prepared from bacterial cellulose nanofibers/starch/chitosan for a food packaging alternative. Int. J. Polym. Sci. 2021, 2021, 6641284. [Google Scholar] [CrossRef]
- Zhao, R.; Guan, W.; Zheng, P.; Tian, F.; Zhang, Z.; Sun, Z.; Cai, L. Development of edible composite film based on chitosan nanoparticles and their application in packaging of fresh red sea bream fillets. Food Control 2022, 132, 108545. [Google Scholar] [CrossRef]
- Martínez-Camacho, A.P.; Cortez-Rocha, M.O.; Graciano-Verdugo, A.Z.; Rodríguez-Félix, F.; Castillo-Ortega, M.M.; Burgos-Hernández, A.; Ezquerra-Brauer, J.M.; Plascencia-Jatomea, M. Extruded films of blended chitosan, low density polyethylene and ethylene acrylic acid. Carbohydr. Polym. 2013, 91, 666–674. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [Green Version]
- Foster, L.J.R.; Butt, J. Chitosan films are NOT antimicrobial. Biotechnol. Lett. 2011, 33, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Dobrucka, R. Antimicrobial packaging with natural compunds—A review. LogForum 2016, 12, 193–202. [Google Scholar] [CrossRef]
- Mitelut, A.C.; Tanase, E.E.; Popa, V.I.; Popa, M.E. Sustainable alternative for food packaging: Chitosan biopolymer—A review. AgroLife Sci. J. 2015, 4, 52–61. [Google Scholar]
- Coma, V.; Deschamps, A.; Martial-Gros, A. Bioactive packaging materials from edible chitosan polymer-antimicrobial activity assessment on dairy-related contaminants. J. Food Sci. 2003, 68, 2788–2792. [Google Scholar] [CrossRef]
- Tripathi, S.; Mehrotra, G.K.; Dutta, P.K. Chitosan based antimicrobial films for food packaging applications. e-Polymers 2008, 8, 1082–1088. [Google Scholar] [CrossRef]
- Silva-Weiss, A.; Bifani, V.; Ihl, M.; Sobral, P.J.A.; Gómez-Guillén, M.C. Structural properties of films and rheology of film-forming solutions based on chitosan and chitosan-starch blend enriched with murta leaf extract. Food Hydrocoll. 2013, 31, 458–466. [Google Scholar] [CrossRef] [Green Version]
- Mayachiew, P.; Devahastin, S. Effects of drying methods and conditions on release characteristics of edible chitosan films enriched with Indian gooseberry extract. Food Chem. 2010, 118, 594–601. [Google Scholar] [CrossRef]
- Yuan, G.; Lv, H.; Yang, B.; Chen, X.; Sun, H. Physical properties, antioxidant and antimicrobial activity of chitosan films containing carvacrol and pomegranate peel extract. Molecules 2015, 20, 11034–11045. [Google Scholar] [CrossRef] [Green Version]
- Zivanovic, S.; Chi, S.; Draughon, A.F. Antimicrobial activity of chitosan films enriched with essential oils. J. Food Sci. 2005, 70, M45–M51. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, W.; Tian, B.; Li, D.; Liu, C.; Jiang, B.; Feng, Z. Preparation and characterization of coating based on protein nanofibers and polyphenol and application for salted duck egg Yolks. Foods 2020, 9, 449. [Google Scholar] [CrossRef] [Green Version]
- Torlak, E.; Sert, D. Antibacterial effectiveness of chitosan-propolis coated polypropylene films against foodborne pathogens. Int. J. Biol. Macromol. 2013, 60, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Rollini, M.; Mascheroni, E.; Capretti, G.; Coma, V.; Musatti, A.; Piergiovanni, L. Propolis and chitosan as antimicrobial and polyphenols retainer for the development of paper based active packaging materials. Food Packag. Shelf Life 2017, 14, 75–82. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Vitchayakitti, W. Improving functional properties of chitosan films as active food packaging by incorporating with propolis. Food Hydrocoll. 2016, 61, 695–702. [Google Scholar] [CrossRef]
- Correa-Pacheco, Z.N.; Bautista-Baños, S.; Ramos-García, M.D.L.; Martínez-González, M.D.C.; Hernández-Romano, J. Physicochemical characterization and antimicrobial activity of edible propolis-chitosan nanoparticle films. Prog. Org. Coat. 2019, 137, 105326. [Google Scholar] [CrossRef]
- Wagh, V.D. Propolis: A wonder bees product and its pharmacological potentials. Adv. Pharmacol. Sci. 2013, 2013, 308249. [Google Scholar] [CrossRef] [Green Version]
- Toreti, V.C.; Sato, H.H.; Pastore, G.M.; Park, Y.K. Recent progress of propolis for its biological and chemical compositions and its botanical origin. Evid. Based Complement. Altern. Med. 2013, 2013, 697390. [Google Scholar] [CrossRef]
- Dos Santos, H.F.; Campos, J.F.; dos Santos, C.M.; Balestieri, J.B.P.; Silva, D.B.; Carollo, C.A.; Souza, K.D.P.; Estevinho, L.M.; dos Santos, E.L. Chemical profile and antioxidant, anti-inflammatory, antimutagenic and antimicrobial activities of geopropolis from the stingless bee Melipona orbignyi. Int. J. Mol. Sci. 2017, 18, 953. [Google Scholar] [CrossRef]
- Meneses, E.A.; Durango, D.L.; Garcia, C.M. Antifungal activity against postharvest fungi by extracts from Colombian propolis. Quim. Nova 2017, 32, 2011–2017. [Google Scholar] [CrossRef] [Green Version]
- Quiroga, E.N.; Sampietro, D.A.; Soberón, J.R.; Sgariglia, M.A.; Vattuone, M.A. Propolis from the northwest of Argentina as a source of antifungal principles. J. Appl. Microbiol. 2006, 101, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.W.; Ye, S.R.; Ting, C.; Yu, Y.H. Antibacterial activity of propolins from Taiwanese green propolis. J. Food Drug Anal. 2018, 26, 761–768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, F.A.; Bastos, E.M.A.; Uzeda, M.; Carvalho, M.A.R.; Farias, L.M.; Moreira, E.S.A.; Braga, F.C. Antibacterial activity of Brazilian propolis and fractions against oral anaerobic bacteria. J. Ethnopharmacol. 2002, 80, 1–7. [Google Scholar] [CrossRef]
- Przybyłek, I.; Karpiński, T.M. Antibacterial properties of propolis. Molecules 2019, 24, 2047. [Google Scholar] [CrossRef] [Green Version]
- Kujumgiev, A.; Tsvetkova, I.; Serkedjieva, Y.; Bankova, V.; Christov, R.; Popov, S. Antibacterial, antifungal and antiviral activity of propolis of different geographic origin. J. Ethnopharmacol. 1999, 64, 235–240. [Google Scholar] [CrossRef]
- Uzel, A.; Sorkun, K.; Önçaǧ, Ö.; Çoǧulu, D.; Gençay, Ö.; Salih, B. Chemical compositions and antimicrobial activities of four different Anatolian propolis samples. Microbiol. Res. 2005, 160, 189–195. [Google Scholar] [CrossRef]
- Kartal, M.; Yildiz, S.; Kaya, S.; Kurucu, S.; Topçu, G. Antimicrobial activity of propolis samples from two different regions of Anatolia. J. Ethnopharmacol. 2003, 86, 69–73. [Google Scholar] [CrossRef]
- Mohammadzadeh, S.; Shariatpanahi, M.; Hamedi, M.; Ahmadkhaniha, R.; Samadi, N.; Ostad, S.N. Chemical composition, oral toxicity and antimicrobial activity of Iranian propolis. Food Chem. 2007, 103, 1097–1103. [Google Scholar] [CrossRef]
- Popova, M.; Giannopoulou, E.; Skalicka-Woźniak, K.; Graikou, K.; Widelski, J.; Bankova, V.; Kalofonos, H.; Sivolapenko, G.; Gaweł-Bȩben, K.; Antosiewicz, B.; et al. Characterization and biological evaluation of propolis from Poland. Molecules 2017, 22, 1159. [Google Scholar] [CrossRef]
- Socha, R.; Gałkowska, D.; Bugaj, M.; Juszczak, L. Phenolic composition and antioxidant activity of propolis from various regions of Poland. Nat. Prod. Res. 2015, 29, 416–422. [Google Scholar] [CrossRef]
- Szliszka, E.; Kucharska, A.Z.; Sokół-Łętowska, A.; Mertas, A.; Czuba, Z.P.; Król, W. Chemical composition and anti-inflammatory effect of ethanolic extract of Brazilian green propolis on activated J774A.1 macrophages. Evid. Based Complement. Altern. Med. 2013, 2013, 976415. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, M.; Mrówczyńska, L.; Kwaśniewska-Sip, P.; Waśkiewicz, A.; Nowak, P.; Ratajczak, I. Effect of the solvent on propolis phenolic profile and its antifungal, antioxidant, and in vitro cytoprotective activity in human erythrocytes under oxidative stress. Molecules 2020, 25, 4266. [Google Scholar] [CrossRef] [PubMed]
- Petruzzi, L.; Corbo, M.R.; Campaniello, D.; Speranza, B.; Sinigaglia, M.; Bevilacqua, A. Antifungal and antibacterial effect of propolis: A comparative hit for food-borne Pseudomonas, Enterobacteriaceae and fungi. Foods 2020, 9, 559. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Ponce, A.G.; Moreira, M.D.R. Antimicrobial efficiency of chitosan coating enriched with bioactive compounds to improve the safety of fresh cut broccoli. LWT Food Sci. Technol. 2013, 50, 78–87. [Google Scholar] [CrossRef]
- ASTM F3136-15—Standard Test Method for Oxygen Gas Transmission Rate through Plastic Film and Sheeting Using a Dynamic Accumulation Method; ASTM International: West Conshohocken, PA, USA, 2015.
- ASTM F2714-08—Standard Test Method for Oxygen Headspace Analysis of Packages Using Fluorescent Decay; ASTM International: West Conshohocken, PA, USA, 2013.
- ISO 2528:2017—Sheet Materials—Determination of Water Vapour Transmission Rate (WVTR)—Gravimetric (Dish) Method; International Organization for Standarization: Geneva, Switzerland, 2017.
- Van den Berg, C. Vapour Sorption Equilibria and Other Water-Starch Interactions: A Physico-Chemical Approach. Ph.D. Thesis, Wageningen University & Research, Wageningen, The Netherlands, 1981. [Google Scholar]
- Basu, S.; Shivhare, U.S.; Mujumdar, A.S. Models for sorption isotherms for foods: A Review. Dry. Technol. 2007, 24, 917–930. [Google Scholar] [CrossRef]
- Gil, G.; Del Mónaco, S.; Cerrutti, P.; Galvagno, M. Selective antimicrobial activity of chitosan on beer spoilage bacteria and brewing yeasts. Biotechnol. Lett. 2004, 26, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Franca, J.R.; De Luca, M.P.; Ribeiro, T.G.; Castilho, R.O.; Moreira, A.N.; Santos, V.R.; Faraco, A.A. Propolis—Based chitosan varnish: Drug delivery, controlled release and antimicrobial activity against oral pathogen bacteria. BMC Complement. Altern. Med. 2014, 14, 478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kacániová, M.; Vuković, N.; Chlebo, R.; Haščík, P.; Rovná, K.; Cubon, J.; Dzugan, M.; Pasternakiewicz, A. The antimicrobial activity of honey, bee pollen loads and beeswax from Slovakia. Arch. Biol. Sci. 2012, 64, 927–934. [Google Scholar] [CrossRef]
- Garedew, A.; Schmolz, E.; Lamprecht, I. Microbiological and calorimetric investigations on the antimicrobial actions of different propolis extracts: An in vitro approach. Thermochim. Acta 2004, 422, 115–124. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Konteles, S.J.; Troullidou, E.; Mourtzinos, I.; Karathanos, V.T. Chemical composition, antioxidant activity and antimicrobial properties of propolis extracts from Greece and Cyprus. Food Chem. 2009, 116, 452–461. [Google Scholar] [CrossRef]
- Mavri, A.; Abramovič, H.; Polak, T.; Bertoncelj, J.; Jamnik, P.; Moažina, S.S.; Jeršek, B. Chemical properties and antioxidant and antimicrobial activities of Slovenian propolis. Chem. Biodivers. 2012, 9, 1545–1558. [Google Scholar] [CrossRef] [PubMed]
- Silici, S.; Ünlü, M.; Vardar-Ünlü, G. Antibacterial activity and phytochemical evidence for the plant origin of Turkish propolis from different regions. World J. Microbiol. Biotechnol. 2007, 23, 1797–1803. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G.; Nunes, S.; Dandlen, S.A.; Cavaco, A.M.; Antunes, M.D. Phenols, flavonoids and antioxidant activity of aqueous and methanolic extracts of propolis (Apis mellifera L.) from Algarve, South Portugal. Food Sci. Technol. 2014, 34, 16–23. [Google Scholar] [CrossRef] [Green Version]
- Papotti, G.; Bertelli, D.; Bortolotti, L.; Plessi, M. Chemical and functional characterization of Italian propolis obtained by different harvesting methods. J. Agric. Food Chem. 2012, 60, 2852–2862. [Google Scholar] [CrossRef] [PubMed]
- Haddadin, M.S.Y.; Nazer, I.; Abu Raddad, S.J.; Robinson, R.K. Effect of propolis on two bacterial species with probiotic potential. Pak. J. Nutr. 2008, 7, 391–394. [Google Scholar] [CrossRef] [Green Version]
- Woźniak, M.; Mrówczyńska, L.; Sip, A.; Babicka, M.; Rogoziński, T.; Ratajczak, I. Aktywność przeciwutleniająca i przeciwbakteryjna miodu, propolisu oraz pyłku kwiatowego. Postępy Fitoter. 2020, 21, 123–128. [Google Scholar] [CrossRef]
- Xing, R.; Liu, S.; Guo, Z.; Yu, H.; Wang, P.; Li, C.; Li, Z.; Li, P. Relevance of molecular weight of chitosan and its derivatives and their antioxidant activities in vitro. Bioorg. Med. Chem. 2005, 13, 1573–1577. [Google Scholar] [CrossRef]
- Díaz-Montes, E.; Castro-Muñoz, R. Trends in chitosan as a primary biopolymer for functional films and coatings manufacture for food and natural products. Polymers 2021, 13, 767. [Google Scholar] [CrossRef] [PubMed]
- Vargas, M.; Albors, A.; Chiralt, A.; González-Martínez, C. Water interactions and microstructure of chitosan-methylcellulose composite films as affected by ionic concentration. LWT Food Sci. Technol. 2011, 44, 2290–2295. [Google Scholar] [CrossRef]
- Quirijns, E.J.; Van Boxtel, A.J.; Van Loon, W.K.; Van Straten, G. Sorption isotherms, GAB parameters and isosteric heat of sorption. J. Sci. Food Agric. 2005, 85, 1805–1814. [Google Scholar] [CrossRef]
- Hess, K.M.; Killgore, J.P.; Srubar, W.V. Nanoscale hygromechanical behavior of lignin. Cellulose 2018, 25, 6345–6360. [Google Scholar] [CrossRef]
- De Oliveira, G.H.H.; Corrêa, P.C.; De Oliveira, A.P.L.R.; Dos Reis, R.C.; Devilla, I.A. Application of GAB model for water desorption isotherms and thermodynamic analysis of sugar beet seeds. J. Food Process Eng. 2017, 40, e12278. [Google Scholar] [CrossRef]
- Timmermann, E.O. Multilayer sorption parameters: BET or GAB values? Colloids Surf. A Physicochem. Eng. Asp. 2003, 220, 235–260. [Google Scholar] [CrossRef] [Green Version]
- Timmermann, E.O.; Chirife, J.; Iglesias, H.A. Water sorption isotherms of foods and foodstuffs: BET or GAB parameters? J. Food Eng. 2001, 48, 19–31. [Google Scholar] [CrossRef]
- Grząbka-Zasadzińska, A.; Ratajczak, I.; Król, K.; Woźniak, M.; Borysiak, S. The influence of crystalline structure of cellulose in chitosan-based biocomposites on removal of Ca(II), Mg(II), Fe(III) ion in aqueous solutions. Cellulose 2021, 28, 5745–5759. [Google Scholar] [CrossRef]
- Shen, Z.; Kamdem, D.P. Development and characterization of biodegradable chitosan films containing two essential oils. Int. J. Biol. Macromol. 2015, 74, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Honary, S.; Maleki, M.; Karami, M. The effect of chitosan molecular weight on the properties of alginate/chitosan microparticles containing prednisolone. Trop. J. Pharm. Res. 2009, 8, 53–61. [Google Scholar] [CrossRef]
- Wu, Y.-W.; Sun, S.-Q.; Zhao, J.; Li, Y.; Zhou, Q. Rapid discrimination of extracts of Chinese propolis and poplar buds by FT-IR and 2D IR correlation spectroscopy. J. Mol. Struct. 2008, 883–884, 48–54. [Google Scholar] [CrossRef]
- Moţ, A.C.; Silaghi-Dumitrescu, R.; Sârbu, C. Rapid and effective evaluation of the antioxidant capacity of propolis extracts using DPPH bleaching kinetic profiles, FT-IR and UV-vis spectroscopic data. J. Food Compos. Anal. 2011, 24, 516–522. [Google Scholar] [CrossRef]
- Woźniak, M.; Kwaśniewska-Sip, P.; Krueger, M.; Roszyk, E.; Ratajczak, I. Chemical, biological and mechanical characterization of wood treated with propolis extract and silicon compounds. Forests 2020, 11, 907. [Google Scholar] [CrossRef]


| Type of Chitosan | Concentration of Propolis Extract (%) | Symbols |
|---|---|---|
| Crab shells | - | C |
| 0.75 | C 0.75 | |
| 2.5 | C 2.5 | |
| 5.0 | C 5.0 | |
| Medium molecular weight | - | M |
| 0.75 | M 0.75 | |
| 2.5 | M 2.5 | |
| 5.0 | M 5.0 | |
| High molecular weight | - | H |
| 0.75 | H 0.75 | |
| 2.5 | H 2.5 | |
| 5.0 | H 5.0 |
| Type of Microbe | Inhibition Zone (mm) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crab Shells Chitosan (C) | Medium Molecular Weight Chitosan (M) | High Molecular Weight Chitosan (H) | ||||||||||
| Ethanolic Extract of Propolis (%) | ||||||||||||
| - | 0.75 | 2.5 | 5.0 | - | 0.75 | 2.5 | 5.0 | - | 0.75 | 2.5 | 5.0 | |
| Pathogenic bacteria | ||||||||||||
| Lysteria monocytogenes | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 17 | 26 | 26 |
| Staphylococcus aureus | 0 | 0 | 0 | 12 | 0 | 0 | 0 | 12 | 0 | 14 | 21 | 22 |
| Bacillus cereus | 0 | 12 | 13 | 13 | 0 | 12.5 | 13 | 13 | 0 | 12 | 13 | 14 |
| Type of Chitosan | Concentration of Propolis Extract (%) | Symbol | Mm (kg/kg) | K | C | R2 |
|---|---|---|---|---|---|---|
| Crab shells | - | C | 0.176 | 0.922 | 1.150 | 0.99996 |
| 0.75 | C 0.75 | 0.129 | 0.931 | 1.112 | 0.99997 | |
| 2.5 | C 2.5 | 0.095 | 0.931 | 1.324 | 0.99993 | |
| 5.0 | C 5.0 | 0.069 | 0.900 | 1.143 | 0.99963 | |
| Medium molecular weight | - | M | 0.159 | 0.933 | 1.256 | 0.99995 |
| 0.75 | M 0.75 | 0.122 | 0.927 | 1.140 | 0.99995 | |
| 2.5 | M 2.5 | 0.089 | 0.942 | 1.367 | 0.99993 | |
| 5.0 | M 5.0 | 0.065 | 0.918 | 1.368 | 0.99986 | |
| High molecular weight | - | H | 0.171 | 0.934 | 1.208 | 0.99996 |
| 0.75 | H 0.75 | 0.134 | 0.932 | 1.096 | 0.99997 | |
| 2.5 | H 2.5 | 0.098 | 0.928 | 1.242 | 0.99992 | |
| 5.0 | H 5.0 | 0.056 | 0.913 | 1.412 | 0.99984 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanicka, K.; Dobrucka, R.; Woźniak, M.; Sip, A.; Majka, J.; Kozak, W.; Ratajczak, I. The Effect of Chitosan Type on Biological and Physicochemical Properties of Films with Propolis Extract. Polymers 2021, 13, 3888. https://doi.org/10.3390/polym13223888
Stanicka K, Dobrucka R, Woźniak M, Sip A, Majka J, Kozak W, Ratajczak I. The Effect of Chitosan Type on Biological and Physicochemical Properties of Films with Propolis Extract. Polymers. 2021; 13(22):3888. https://doi.org/10.3390/polym13223888
Chicago/Turabian StyleStanicka, Karolina, Renata Dobrucka, Magdalena Woźniak, Anna Sip, Jerzy Majka, Wojciech Kozak, and Izabela Ratajczak. 2021. "The Effect of Chitosan Type on Biological and Physicochemical Properties of Films with Propolis Extract" Polymers 13, no. 22: 3888. https://doi.org/10.3390/polym13223888
APA StyleStanicka, K., Dobrucka, R., Woźniak, M., Sip, A., Majka, J., Kozak, W., & Ratajczak, I. (2021). The Effect of Chitosan Type on Biological and Physicochemical Properties of Films with Propolis Extract. Polymers, 13(22), 3888. https://doi.org/10.3390/polym13223888