Towards the Development of Novel Hybrid Composite Steel Pipes: Electrochemical Evaluation of Fiber-Reinforced Polymer Layered Steel against Corrosion
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
Potentiodynamic Polarization (PDP) and Electrochemical Impedance Spectroscopy (EIS)
3. Results and Discussion
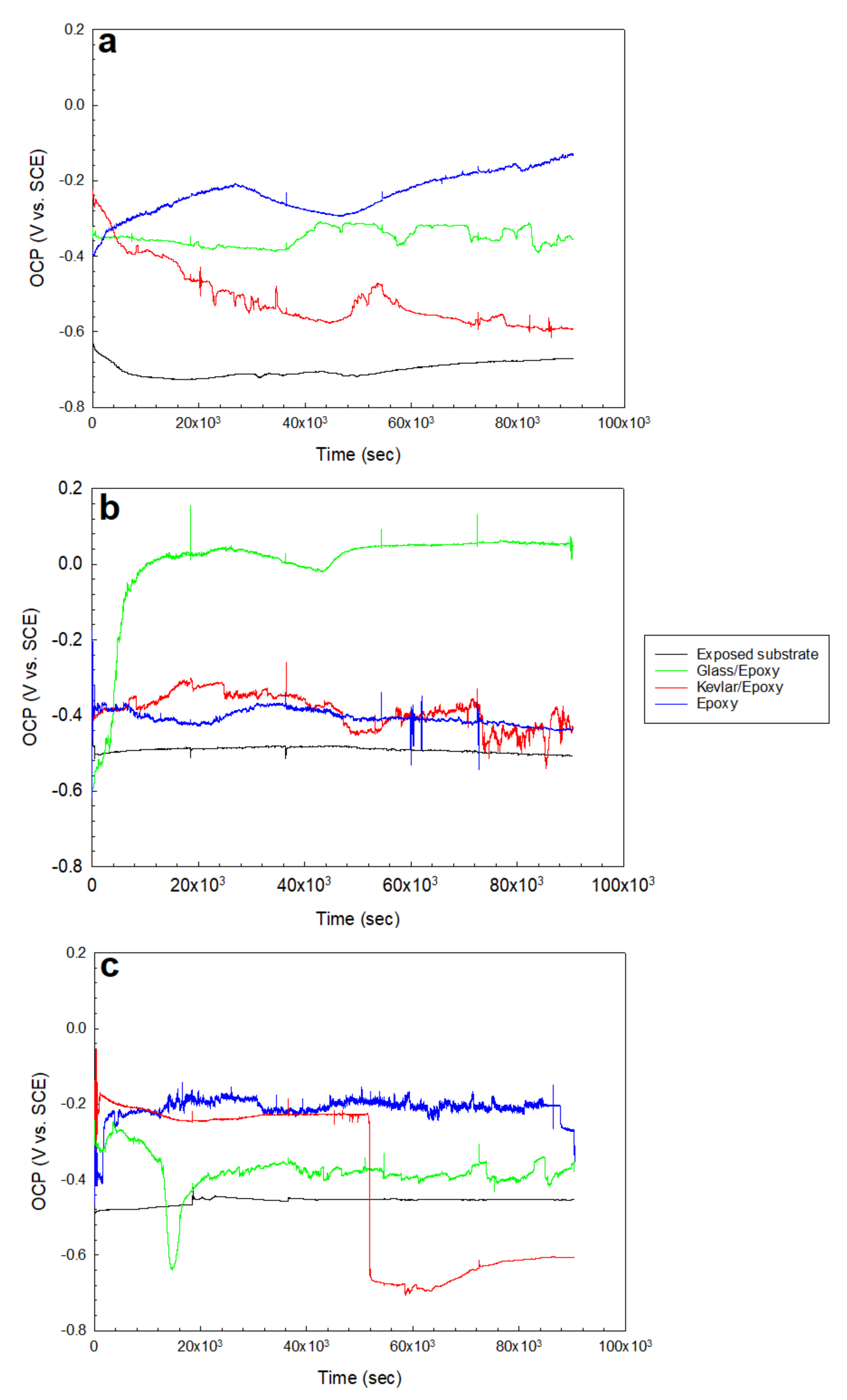
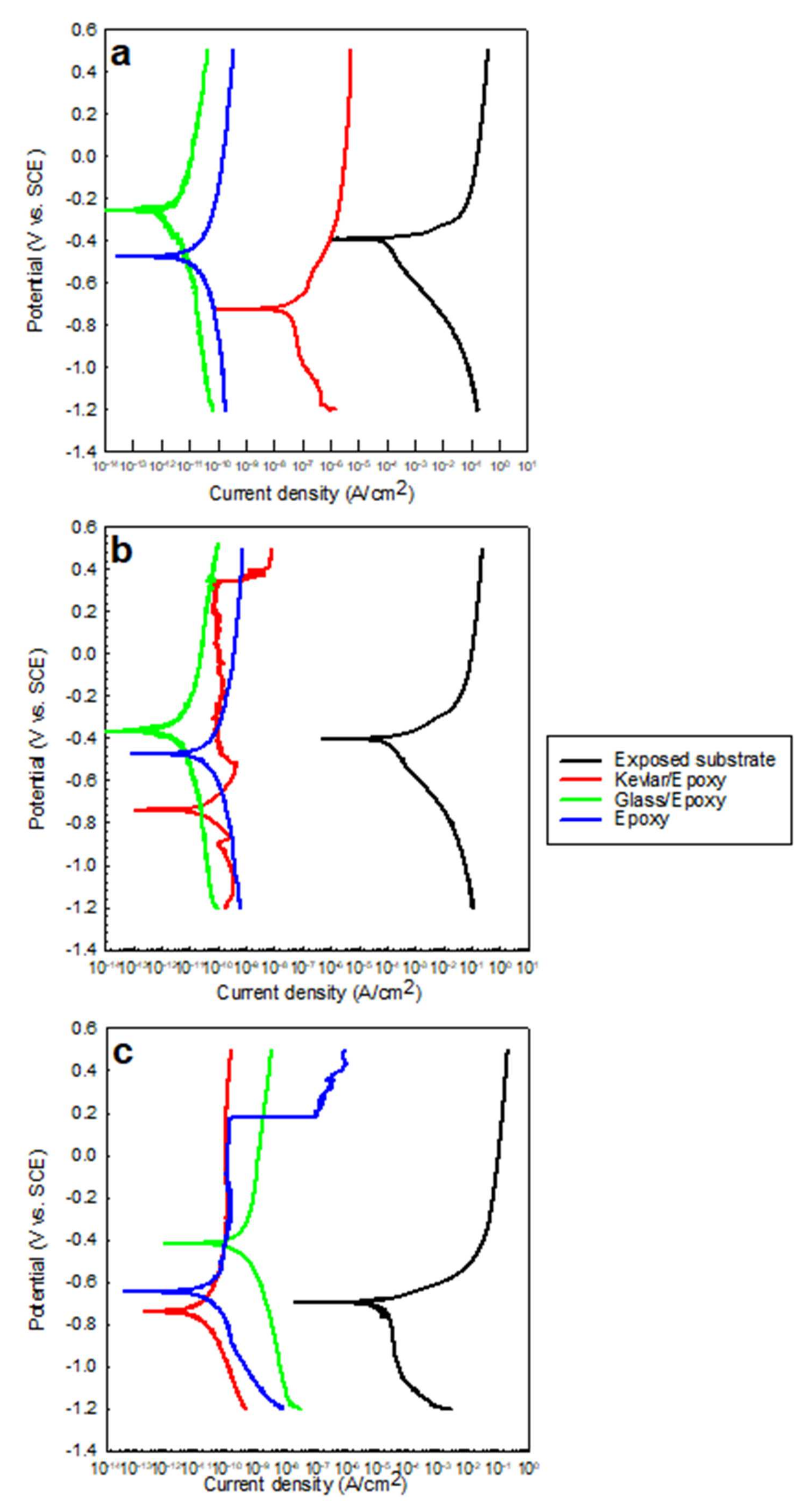
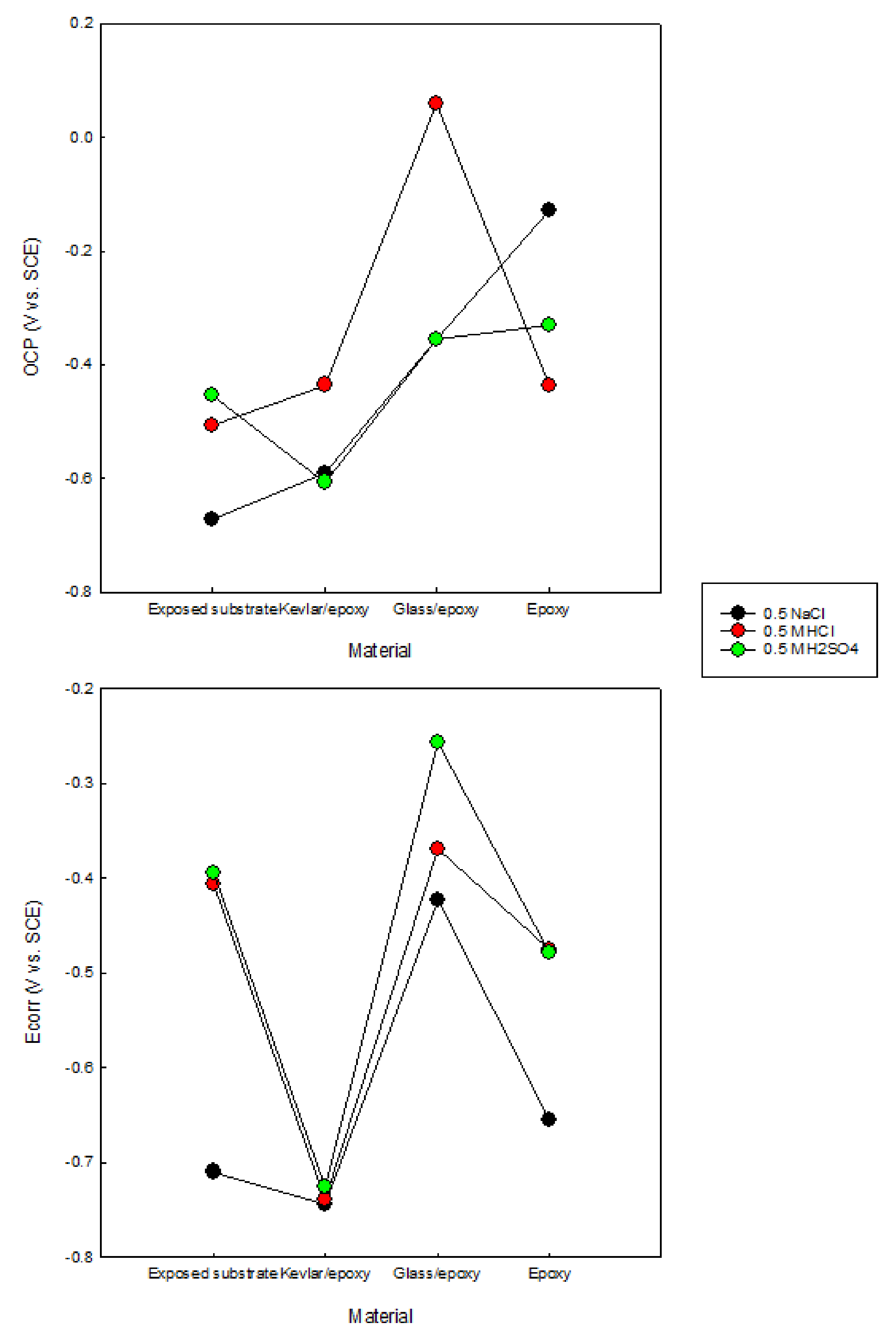
3.1. OCP/Potentiodynamic Polarization
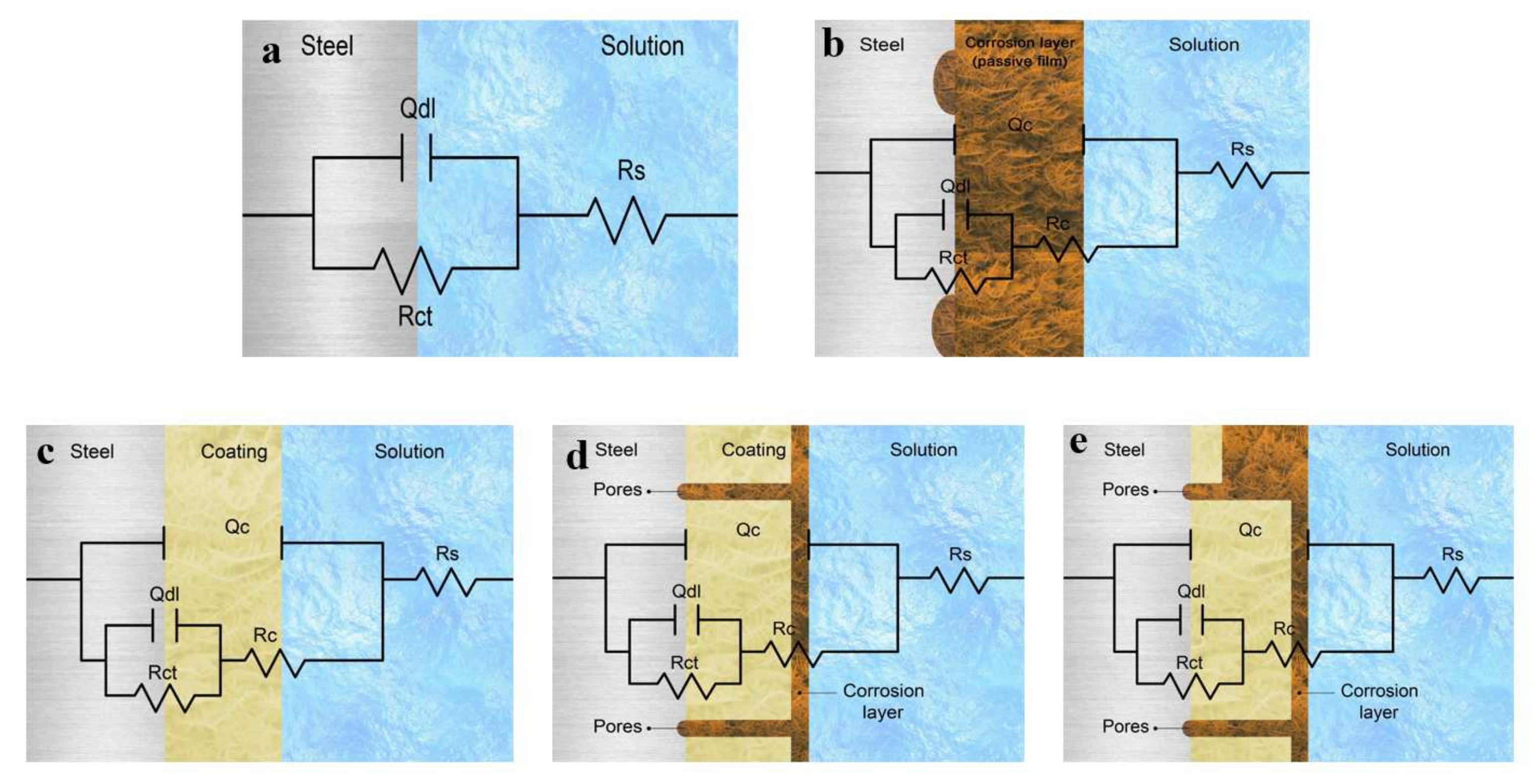
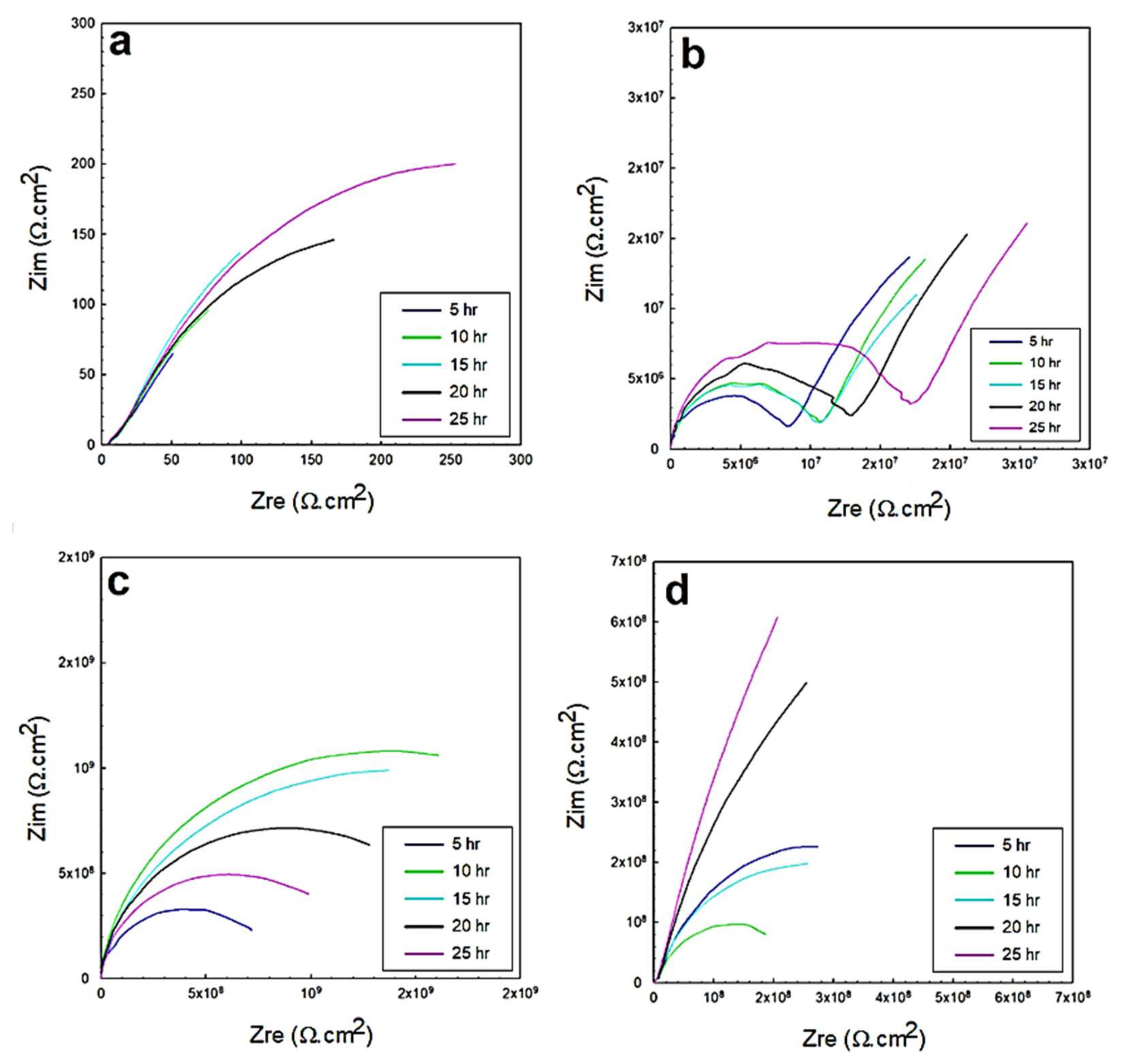
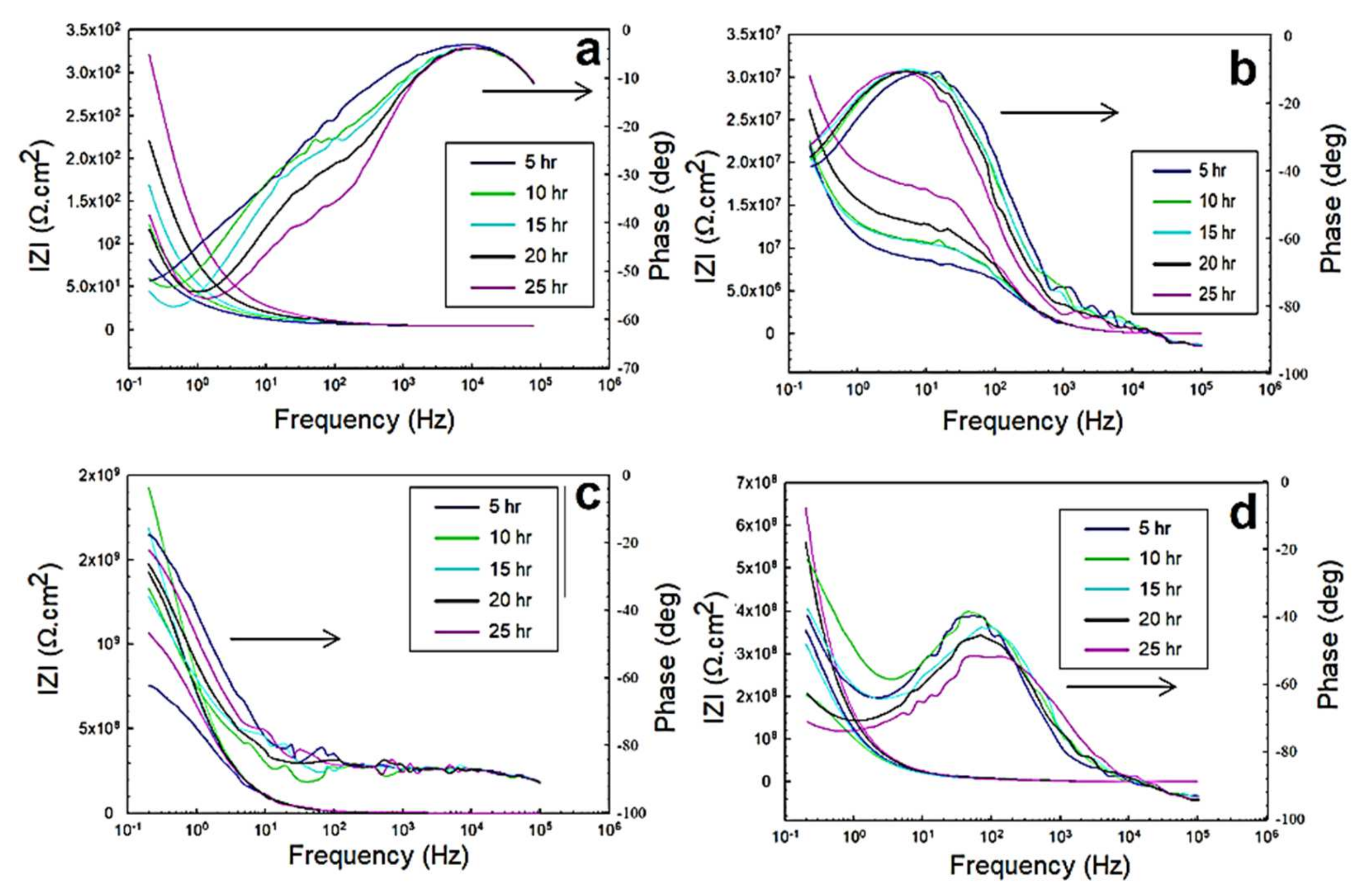
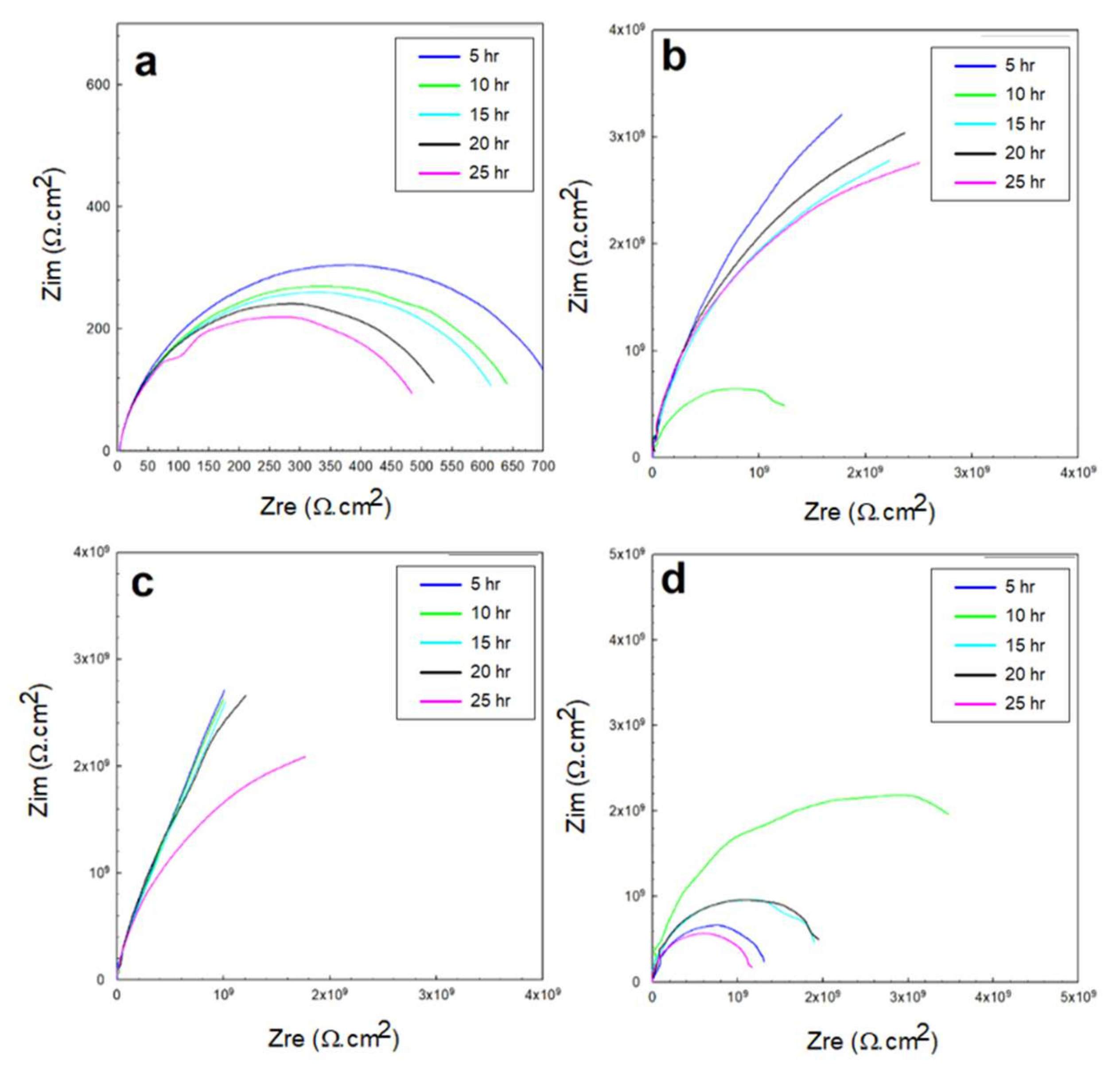
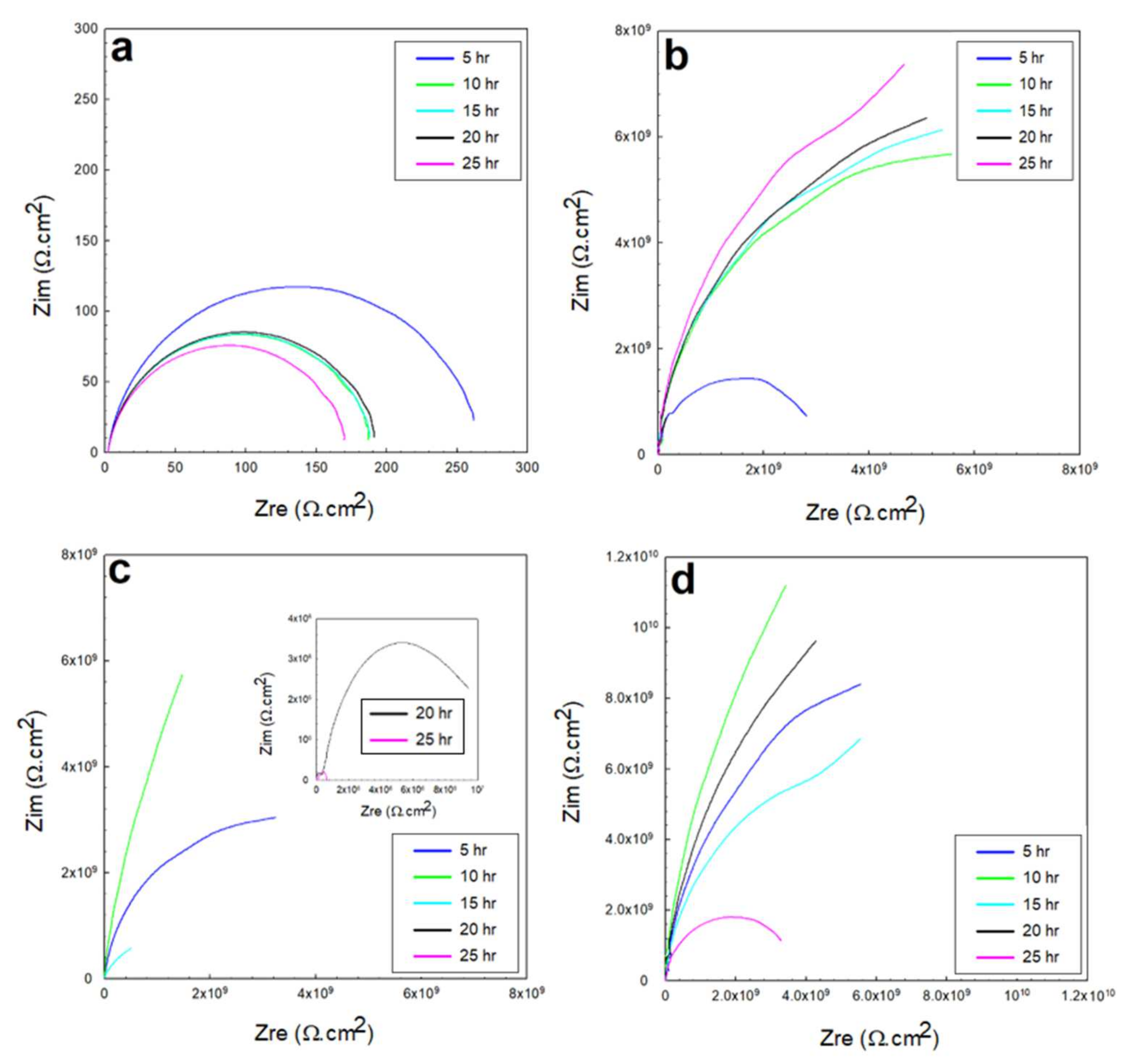
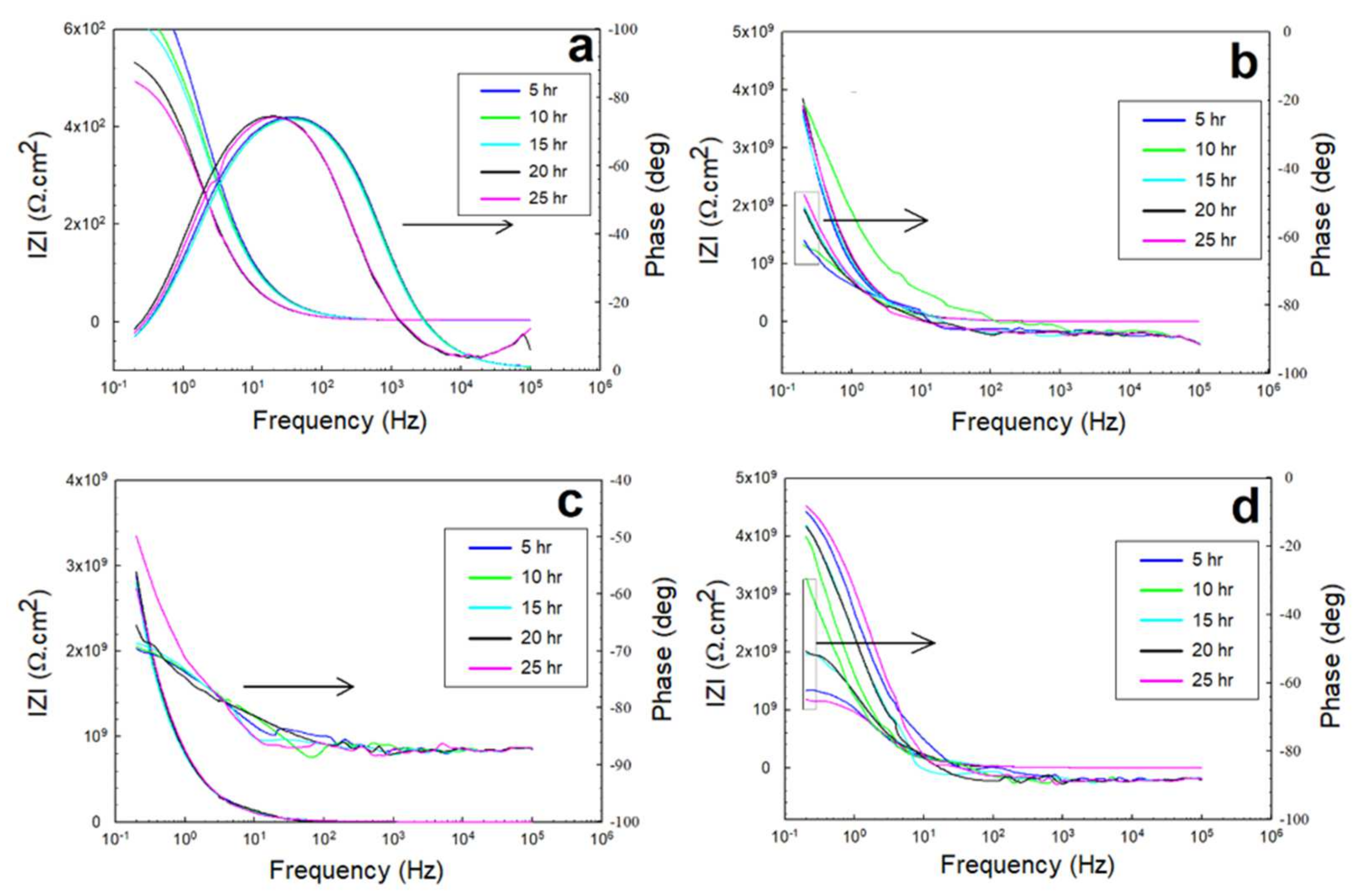
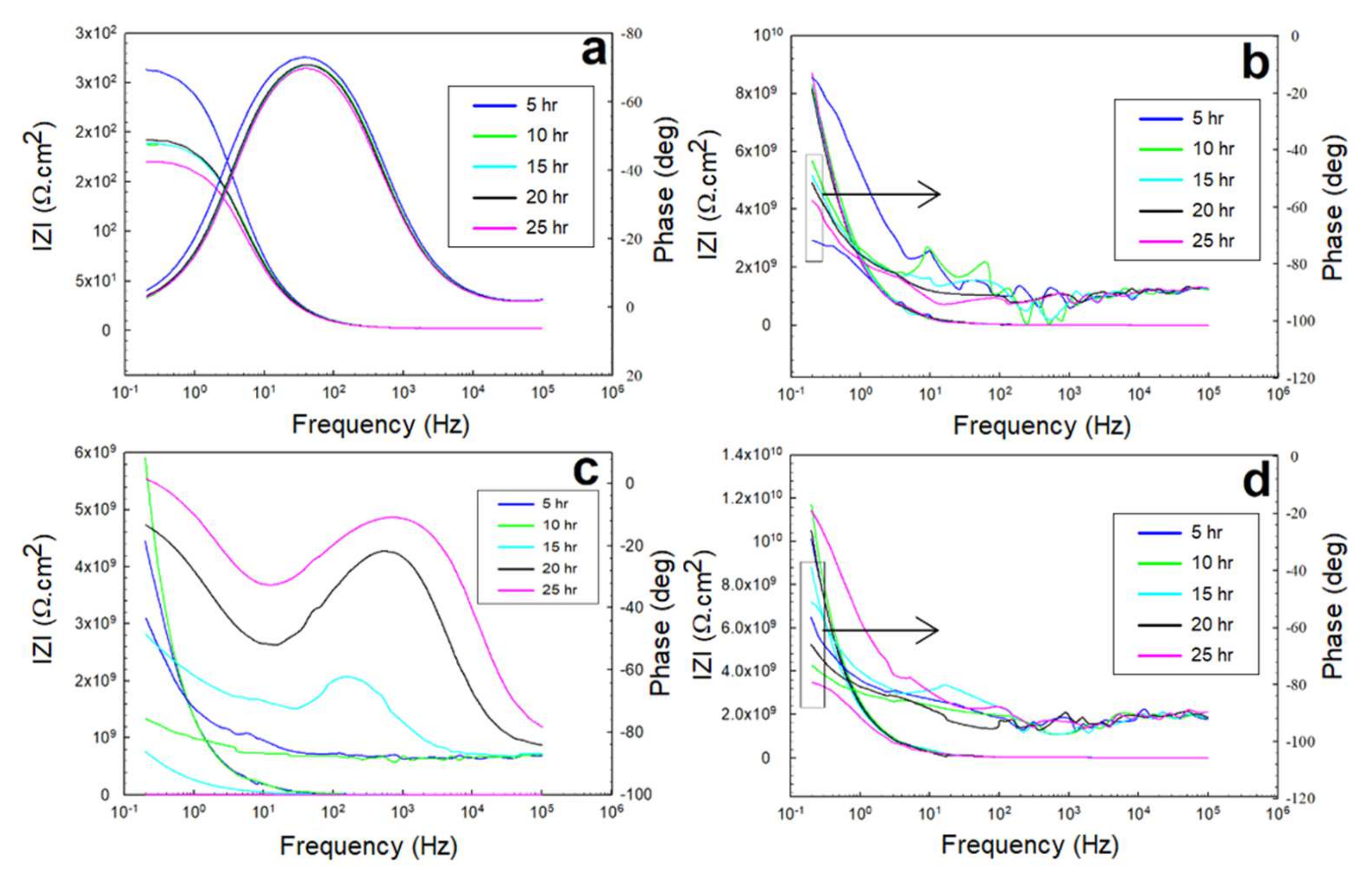
3.2. Electrochemical Impedance Spectroscopy (EIS)
3.2.1. EIS in 0.5 M NaCl Solutions
3.2.2. EIS in 0.5 M HCl and 0.5 M H2SO4 Solutions
3.3. Failure Modes
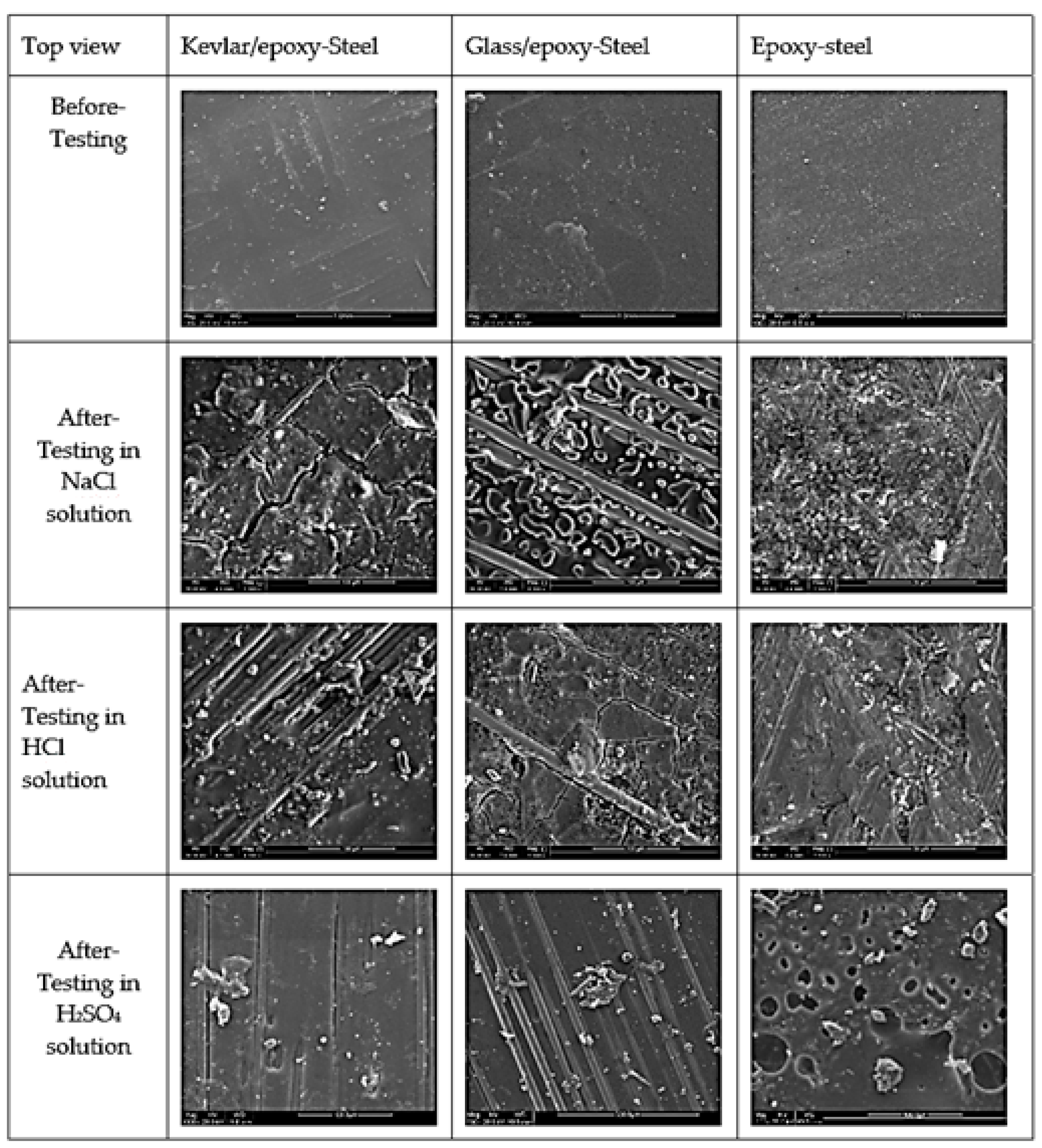
3.3.1. Optical Microscope Images
3.3.2. SEM Analysis
4. Conclusions
- As demonstrated by the open circuit potential results, the Kevlar/epoxy system interestingly showed deterioration in the H2SO4 solution comparable to an exposed substrate after nearly 50 ks.
- The corrosion rates for the coated substrates were 1 million to 10 million times less than that of the exposed substrates, as reported by the PDP tests.
- The equivalent circuit of the configuration, {R(Q(R(QR)))}, was correctly fitted to the experimental data across the entire frequency range.
- The peaks of the glass/epoxy and epoxy coating systems in the Bode diagrams indicated reliability against activity at both the double and coating layers, appearing at frequencies two orders of magnitude higher than those of the Kevlar/epoxy system.
- The resistance of the glass/epoxy system in the HCl solution was the highest, although it showed a steady decrease with time. The Kevlar/epoxy system, on the contrary, had less resistance, but it noticeably increased with time. This result underlines the significance of Kevlar in promoting the formation of passive films that, with time, decelerate dissolution and confirm the significance of glass in stabilizing the epoxy system that initially protects the system.
- The SEM images revealed cracks and the exposure of the embedded fibers from under the epoxy subjected to deterioration from the acid attack, sustained/assisted by the diffusion of the corrosion species.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Finšgar, M.; Jackson, J. Application of corrosion inhibitors for steels in acidic media for the oil and gas industry: A review. Corros. Sci. 2014, 86, 17–41. [Google Scholar] [CrossRef]
- Asmara, Y.P.; Kurniawan, T. Corrosion prediction for corrosion rate of carbon steel in oil and gas environment: A review. Indones. J. Sci. Technol. 2018, 3, 64–74. [Google Scholar] [CrossRef]
- Al-Abtah, F.G.; Al-Huniti, N.; Mahdi, E. Simulation-based parametric study for the hybrid superplastic forming of AZ31. Proc. Comput. Sci. 2019, 158, 177–197. [Google Scholar] [CrossRef]
- Ameh, E.; Ikpeseni, S.; Lawal, L. A Review of field corrosion control and monitoring techniques of the upstream oil and gas pipelines. Niger. J. Technol. Dev. 2018, 14, 67. [Google Scholar] [CrossRef]
- Mahdi, E.; Eltai, E. Development of cost-effective composite repair system for oil/gas pipelines. Compos. Struct. 2018, 202, 802–806. [Google Scholar] [CrossRef]
- Vairo, T.; Magrì, S.; Quagliati, M.; Reverberi, A.P.; Fabiano, B. An oil pipeline catastrophic failure: Accident scenario modelling and emergency response development. Chem. Eng. Trans. 2017, 57, 373–378. [Google Scholar] [CrossRef]
- Prabhakar, M.M.; Rajini, N.; Ayrilmis, N.; Mayandi, K.; Siengchin, S.; Senthilkumar, K.; Karthikeyan, S.; Ismail, S.O. An overview of burst, buckling, durability and corrosion analysis of lightweight FRP composite pipes and their applicability. Compos. Struct. 2019, 230, 111419. [Google Scholar] [CrossRef]
- Mazumder, M.A.J. Global impact of corrosion: Occurrence, cost and mitigation. Glob. J. Eng. Sci. 2020, 5, 1–5. [Google Scholar]
- Tan, M.Y.J.; Mahdavi, F.; Ranade, S.D. Recent progresses in understanding pipeline coating degraduation. In Proceedings of the Corrosion and Prevention Conference 2018, Adelaide, Australia, 11–14 November 2018. [Google Scholar]
- Bäßler, R. Corrosion control through organic coatings (Book review). Mater. Corros. 2017, 68, 1014. [Google Scholar] [CrossRef][Green Version]
- Fotovvati, B.; Namdari, N.; Dehghanghadikolaei, A. On coating techniques for surface protection: A review. J. Manuf. Mater. Process. 2019, 3, 28. [Google Scholar] [CrossRef]
- Khan, A.; Sliem, M.H.; Arif, A.; Salih, M.A.; Shakoor, R.; Montemor, F.; Kahraman, R.; Mansour, S.; Abdullah, A.M.; Hasan, A. Designing and performance evaluation of polyelectrolyte multilayered composite smart coatings. Prog. Org. Coat. 2019, 137, 105319. [Google Scholar] [CrossRef]
- Lee, H.; Neville, K. Handbook of Epoxy Resins; McGraw-Hill: New York, NY, USA, 1982. [Google Scholar]
- Sung, P.-H.; Lin, C.-Y. Polysiloxane modified epoxy polymer networks—I. Graft interpenetrating polymeric networks. Eur. Polym. J. 1997, 33, 903–906. [Google Scholar] [CrossRef]
- Alabtah, F.G.; Mahdi, E.; Khraisheh, M. External Corrosion Behavior of Steel/GFRP Composite Pipes in Harsh Conditions. Materials 2021, 14, 6501. [Google Scholar] [CrossRef]
- Alabtah, F.G.; Mahdi, E.; Eliyan, F.F. The Use of Fiber Reinforced Polymeric Composites in Pipelines: A Review. Compos. Struct. 2021, 276, 114595. [Google Scholar] [CrossRef]
- Gowid, S.; Mahdi, E.; Alabtah, F. Modeling and optimization of the crushing behavior and energy absorption of plain weave composite hexagonal quadruple ring systems using artificial neural network. Compos. Struct. 2019, 229, 111473. [Google Scholar] [CrossRef]
- Alabtah, F.G.; Mahdi, E. The effect of sizing optimization on the interface between high strength steel and fiber reinforced composite. Compos. Struct. 2021, 266, 113740. [Google Scholar] [CrossRef]
- Chen, L.; Wu, S.; Lu, H.; Huang, K.; Shi, J. Comparative analysis of FRP and seamless steel pipe on crude oil transportation performance. J. Chem. Pharm. Res. 2014, 6, 2364–2369. [Google Scholar]
- Esmaeel, R.A.; Khan, M.A.; Taheri, F. Assessment of the environmental effects on the performance of FRP repaired steel pipes subjected to internal pressure. J. Press. Vessel. Technol. 2012, 134, 041702. [Google Scholar] [CrossRef]
- Al-Abtah, F.G.; Mahdi, E.; Gowid, S. The use of composite to eliminate the effect of welding on the bending behavior of metallic pipes. Compos. Struct. 2020, 235, 111793. [Google Scholar] [CrossRef]
- Eslami, S.; Raouf, A.H.; Eslami, S. Effects of moisture absorption on degradation of E-glass fiber reinforced Vinyl Ester composite pipes and modelling of transient moisture diffusion using finite element analysis. Corros. Sci. 2015, 90, 168–175. [Google Scholar] [CrossRef]
- Dalai, R.P.; Ray, B.C. Failure and fractography studies of FRP composites: Effects of loading speed and environments. In Proceedings of the Processing and Fabrication of Advanced Materials XIX, Auckland, New Zeland, 14–17 January 2011. [Google Scholar]
- Mahato, K.K.; Dutta, K.; Ray, B.C. Static and dynamic behavior of fibrous polymeric composite materials at different environmental conditions. J. Polym. Environ. 2018, 26, 1024–1050. [Google Scholar] [CrossRef]
- Ray, B.C.; Rathore, D. Durability and integrity studies of environmentally conditioned interfaces in fibrous polymeric composites: Critical concepts and comments. Adv. Colloid Interface Sci. 2014, 209, 68–83. [Google Scholar] [CrossRef]
- Guo, F.; Al-Saadi, S.; Raman, R.S.; Zhao, X. Durability of fiber reinforced polymer (FRP) in simulated seawater sea sand concrete (SWSSC) environment. Corros. Sci. 2018, 141, 1–13. [Google Scholar] [CrossRef]
- Wang, Z.; Zhao, X.-L.; Xian, G.; Wu, G.; Raman, R.S.; Al-Saadi, S. Effect of sustained load and seawater and sea sand concrete environment on durability of basalt- and glass-fibre reinforced polymer (B/GFRP) bars. Corros. Sci. 2018, 138, 200–218. [Google Scholar] [CrossRef]
- Dong, Z.-Q.; Wu, G.; Zhao, X.-L.; Lian, J.-L. Long-term bond durability of fiber-reinforced polymer bars embedded in seawater sea-sand concrete under ocean environments. J. Compos. Constr. 2018, 22, 04018042. [Google Scholar] [CrossRef]
- Li, S.; Guo, S.; Yao, Y.; Jin, Z.; Shi, C.; Zhu, D. The effects of aging in seawater and SWSSC and strain rate on the tensile performance of GFRP/BFRP composites: A critical review. Constr. Build. Mater. 2021, 282, 122534. [Google Scholar] [CrossRef]
- Wang, G.; Xiao, L.; Liu, Z.; Han, Z.; Ouyang, J.-H.; Zhang, D. Effect of supercritical CO2 on corrosion behavior and mechanism of fiber reinforced pipe. Corros. Sci. 2016, 113, 180–182. [Google Scholar] [CrossRef]
- Ellyin, F.; Maser, R. Environmental effects on the mechanical properties of glass-fiber epoxy composite tubular specimens. Compos. Sci. Technol. 2004, 64, 1863–1874. [Google Scholar] [CrossRef]
- Nakai, A.; Ikegaki, S.; Hamada, H.; Takeda, N. Degradation of braided composites in hot water. Compos. Sci. Technol. 2000, 60, 325–331. [Google Scholar] [CrossRef]
- Imielińska, K.; Guillaumat, L. The effect of water immersion ageing on low-velocity impact behaviour of woven aramid–glass fibre/epoxy composites. Compos. Sci. Technol. 2004, 64, 2271–2278. [Google Scholar] [CrossRef]
- Scida, D.; Aboura, Z.; Benzeggagh, M. The effect of ageing on the damage events in woven-fibre composite materials under different loading conditions. Compos. Sci. Technol. 2002, 62, 551–557. [Google Scholar] [CrossRef]
- Her, R.; Renard, J.; Gaffard, V.; Favry, Y.; Wiet, P. Design of pipeline composite repairs: From lab scale tests to fea and full scale testing. In Proceedings of the 10th International Pipeline Conference, Calgary, AB, Canada, 29 September–3 October 2014. [Google Scholar]
- Morampudi, P.; Namala, K.K.; Gajjela, Y.K.; Barath, M.; Prudhvi, G. Review on glass fiber reinforced polymer composites. Mater. Today Proc. 2021, 43, 314–319. [Google Scholar] [CrossRef]
- Akbarinezhad, E.; Ebrahimi, M.; Faridi, H. Corrosion inhibition of steel in sodium chloride solution by undoped polyaniline epoxy blend coating. Prog. Org. Coat. 2009, 64, 361–364. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, X.; Xing, F.; Sui, L.; Zheng, Y.; Huang, X. Investigation on the electrochemical and mechanical performance of CFRP and steel-fiber composite bar used for impressed current cathodic protection anode. Constr. Build. Mater. 2020, 255, 119377. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, Y.; Pan, J.; Sui, L.; Xing, F.; Sun, H.; Li, P. Experimental investigations on corrosion resistance of innovative steel-FRP composite bars using X-ray microcomputed tomography. Compos. Part B Eng. 2019, 161, 272–284. [Google Scholar] [CrossRef]
- Benea, L.; Simionescu, N.; Mardare, L. The effect of polymeric protective layers and the immersion time on the corrosion behavior of naval steel in natural seawater. J. Mater. Res. Technol. 2020, 9, 13174–13184. [Google Scholar] [CrossRef]
- Hsissou, R.; Benhiba, F.; Echihi, S.; Benkhaya, S.; Hilali, M.; Berisha, A.; Briche, S.; Zarrouk, A.; Nouneh, K.; Elharfi, A. New epoxy composite polymers as a potential anticorrosive coatings for carbon steel in 3.5% NaCl solution: Experimental and computational approaches. Chem. Data Collect. 2021, 31, 100619. [Google Scholar] [CrossRef]
- Zhang, C.; Zheng, D.; Song, G.-L.; Guo, Y.; Liu, M.; Kia, H. Influence of microstructure of carbon fibre reinforced polymer on the metal in contact. J. Mater. Res. Technol. 2020, 9, 560–573. [Google Scholar] [CrossRef]
- Holland, R.I. Use of potentiodynamic polarization technique for corrosion testing of dental alloys. Eur. J. Oral Sci. 1991, 99, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Bierwagen, G.; Tallman, D.; Li, J.; He, L.; Jeffcoate, C. EIS studies of coated metals in accelerated exposure. Prog. Org. Coat. 2003, 46, 149–158. [Google Scholar] [CrossRef]
- Eliyan, F.F.; Mahdi, E.-S.; Alfantazi, A. Electrochemical evaluation of the corrosion behaviour of API-X100 pipeline steel in aerated bicarbonate solutions. Corros. Sci. 2012, 58, 181–191. [Google Scholar] [CrossRef]
- Boukamp, B. Equivalent Circuit User’s Manual, 2nd ed.; University of Twente: Enschede, The Netherlands, 1989. [Google Scholar]
- Kumar, S.A.; Alagar, M.; Mohan, V. Studies on corrosion-resistant behavior of siliconized epoxy interpenetrating coatings over mild steel surface by electrochemical methods. J. Mater. Eng. Perform. 2002, 11, 123–129. [Google Scholar] [CrossRef]
- Medrano-Vaca, M.; Gonzalez-Rodriguez, J.; Nicho, M.; Casales, M.; Salinas-Bravo, V. Corrosion protection of carbon steel by thin films of poly(3-alkyl thiophenes) in 0.5M H2SO4. Electrochim. Acta 2008, 53, 3500–3507. [Google Scholar] [CrossRef]
- Wang, L.L.; Chen, H.J.; Hao, L.; Lin, A.; Gan, F.X. Electrochemical corrosion behavior of electroless Ni-P coating in NaCl and H2SO4 solutions. Mater. Corros. 2010, 62, 1003–1007. [Google Scholar] [CrossRef]
- Liao, K.; Schultheisz, C.; Hunston, D. Effects of environmental aging on the properties of pultruded GFRP. Compos. Part B Eng. 1999, 30, 485–493. [Google Scholar] [CrossRef]
- Sousa, J.; Correia, J.; Cabral-Fonseca, S. Durability of glass fibre reinforced polymer pultruded profiles: Comparison between QUV accelerated exposure and natural weathering in a mediterranean climate. Exp. Tech. 2016, 40, 207–219. [Google Scholar] [CrossRef]
- Bazli, M.; Ashrafi, H.; Oskouei, A.V. Effect of harsh environments on mechanical properties of GFRP pultruded profiles. Compos. Part B Eng. 2016, 99, 203–215. [Google Scholar] [CrossRef]
- Reddy, E.V.S.; Reddy, G.R.; Varadarajulu, A.; Reddy, K.H. Chemical resistance, compressive properties and impact strength of glass and bamboo fibres reinforced polyester hybrid composites. J. Inst. Eng. Chem. Eng. Div. 2010, 91, 5–8. [Google Scholar]





| Refs. | Year | Coating System | Immersion Period | Soln. | Test | Behavior |
|---|---|---|---|---|---|---|
| [37] | 2009 | Undoped polyaniline epoxy blend coating | 300 days | NaCl | OCP and EIS | Coating resistance decreased at day 14 and then stabilized during the remaining immersion time. |
| [38] | 2020 | CFRP | 42 days | Seawater | Anodic polarization and EIS | Carbon fiber degraded under the action of corrosive medium and polarization due to the corrosion damage of resin matrix, carbon fiber, and interface. |
| [39] | 2019 | CFRP and GFRP | 48 h | NaCl | Electrochemical method (climate accelerated process) | The corrosion rates of carbon-type and glass-type were less than 1/10 and 1/100, respectively, than that of an ordinary steel bar. |
| [40] | 2020 | Epoxy primer and Polyurethane acrylic paint | 98 days | Seawater | OCP, linear polarization, and EIS | A loss of thickness over time was recorded. |
| [41] | 2021 | sulfur epoxy resin SER/HMDA and SER/MDA composite polymeric coatings | 24 h | NaCl | PDP and EIS | Composite polymers could retard and chloride ions attack. |
| [42] | 2020 | CFRP | 8 h | NaCl+ CaCl2+ NaHCO3 | OCP, PDP, and EIS | CFRP can cause galvanic corrosion to engineering metal in an aggressive environment. |
| Current Study | -- | GFRP, KFRP, and pure epoxy | 25 h | NaCl, HCl, and H2SO4 | OCP, PDP, and EIS | Results and conclusions are discussed in the following sections. |
| Fe | C | Mn | P | S | Si |
|---|---|---|---|---|---|
| 99.09 | 0.12 | 0.6 | 0.045 | 0.045 | 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alabtah, F.G.; Mahdi, E.; Eliyan, F.F.; Eltai, E.; Khraisheh, M. Towards the Development of Novel Hybrid Composite Steel Pipes: Electrochemical Evaluation of Fiber-Reinforced Polymer Layered Steel against Corrosion. Polymers 2021, 13, 3805. https://doi.org/10.3390/polym13213805
Alabtah FG, Mahdi E, Eliyan FF, Eltai E, Khraisheh M. Towards the Development of Novel Hybrid Composite Steel Pipes: Electrochemical Evaluation of Fiber-Reinforced Polymer Layered Steel against Corrosion. Polymers. 2021; 13(21):3805. https://doi.org/10.3390/polym13213805
Chicago/Turabian StyleAlabtah, Fatima Ghassan, Elsadig Mahdi, Faysal Fayez Eliyan, Elsadig Eltai, and Marwan Khraisheh. 2021. "Towards the Development of Novel Hybrid Composite Steel Pipes: Electrochemical Evaluation of Fiber-Reinforced Polymer Layered Steel against Corrosion" Polymers 13, no. 21: 3805. https://doi.org/10.3390/polym13213805
APA StyleAlabtah, F. G., Mahdi, E., Eliyan, F. F., Eltai, E., & Khraisheh, M. (2021). Towards the Development of Novel Hybrid Composite Steel Pipes: Electrochemical Evaluation of Fiber-Reinforced Polymer Layered Steel against Corrosion. Polymers, 13(21), 3805. https://doi.org/10.3390/polym13213805







