Removal of Dyes by Polymer-Enhanced Ultrafiltration: An Overview
Abstract
1. Introduction
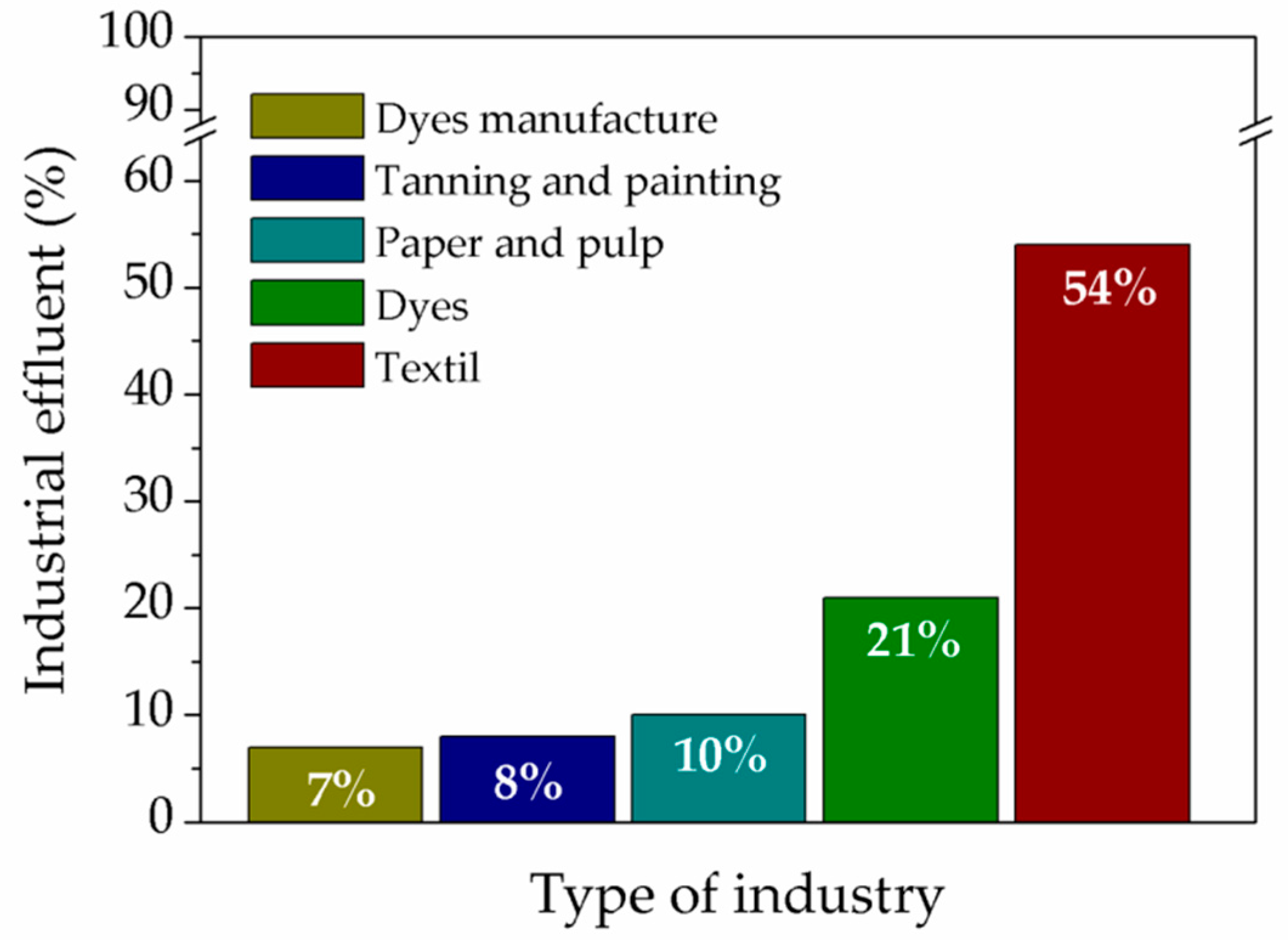
2. Dyes in Industrial Wastewater
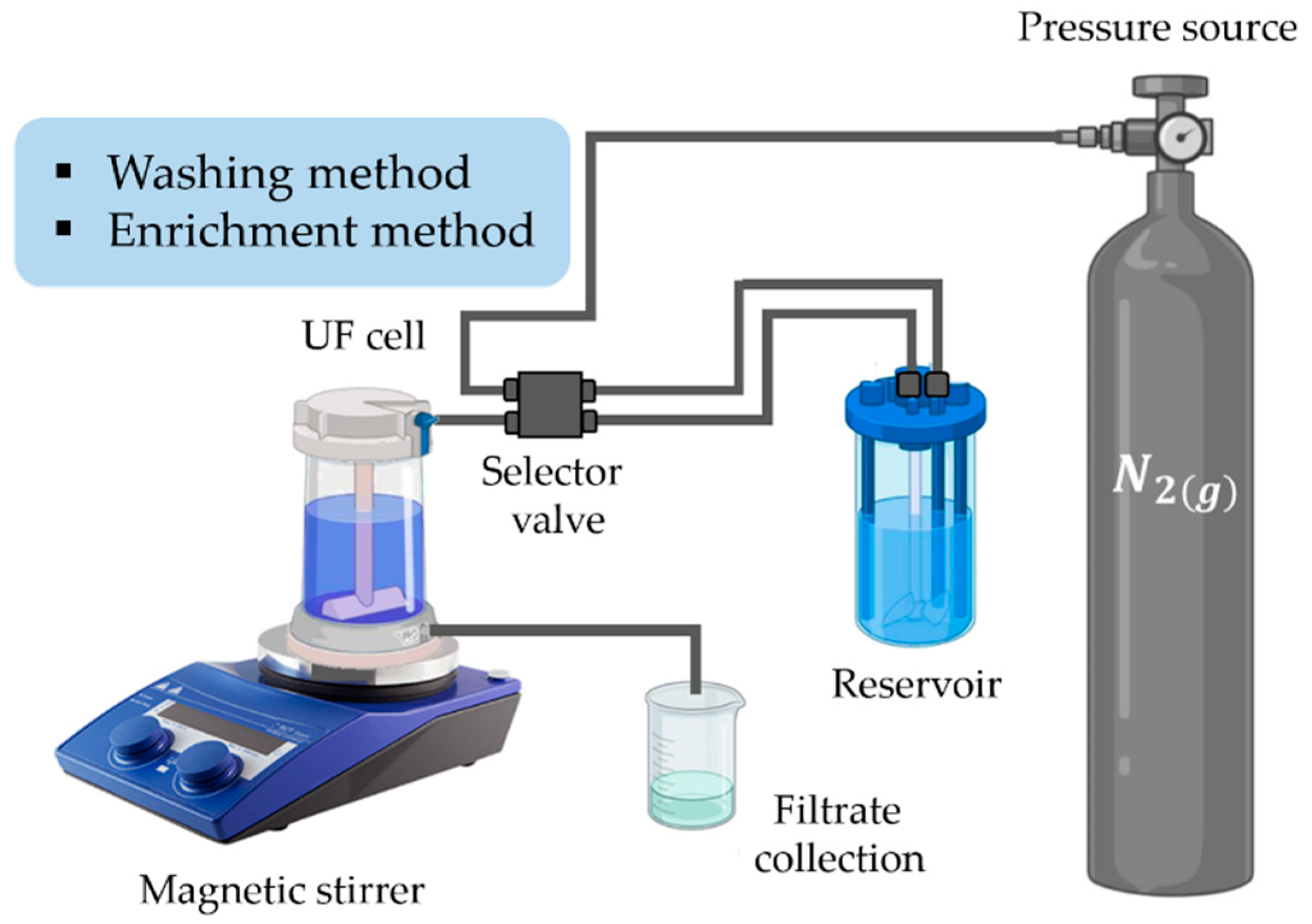
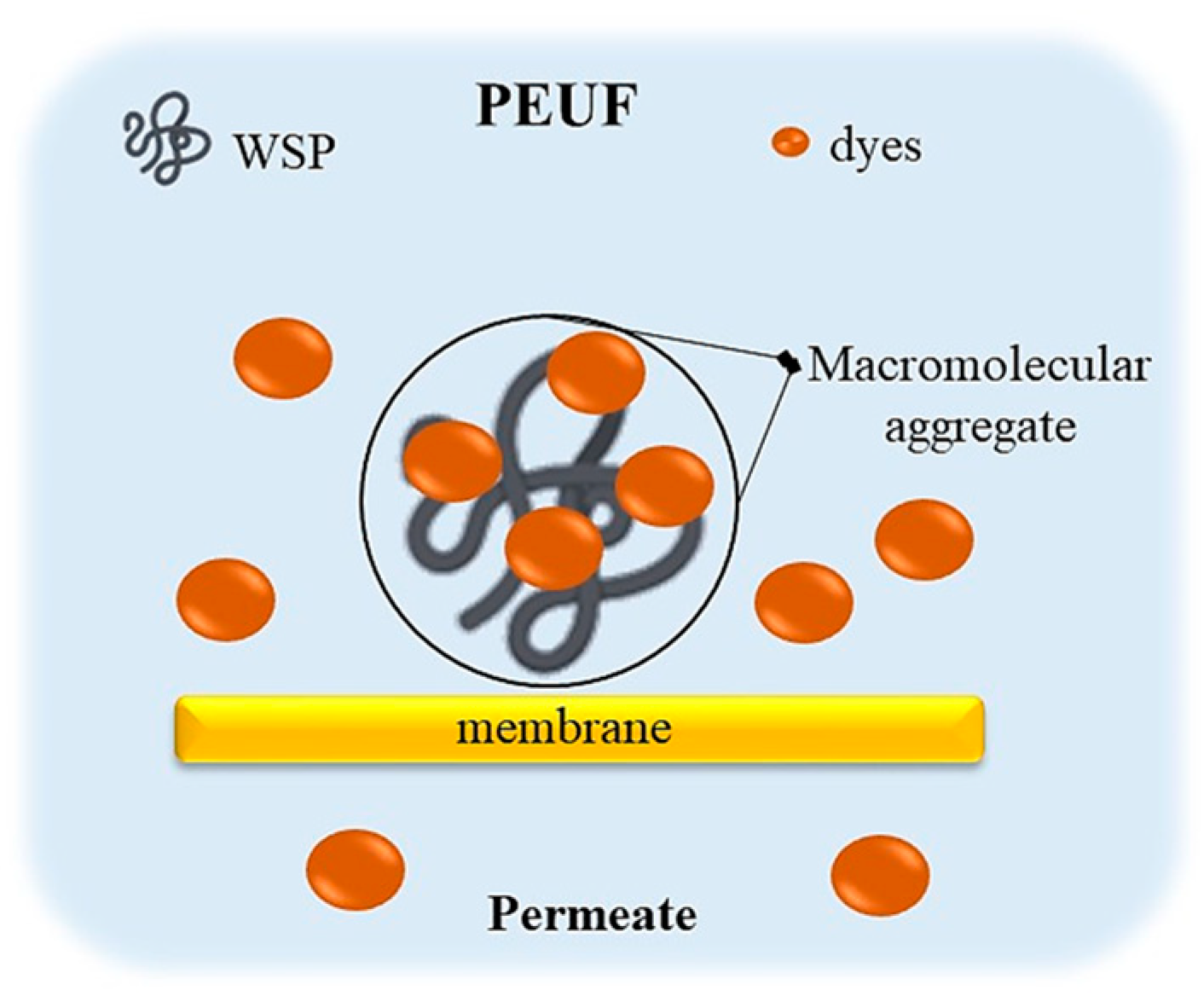
3. Polymer-Enhanced Ultrafiltration (PEUF) Technique
3.1. MB Removal by PEUF
3.2. MO Removal by PEUF
4. Synthetic and Natural Water-Soluble Polymers Used as Extractants of Dyes
5. Removal of Dyes on Function of Different Variables
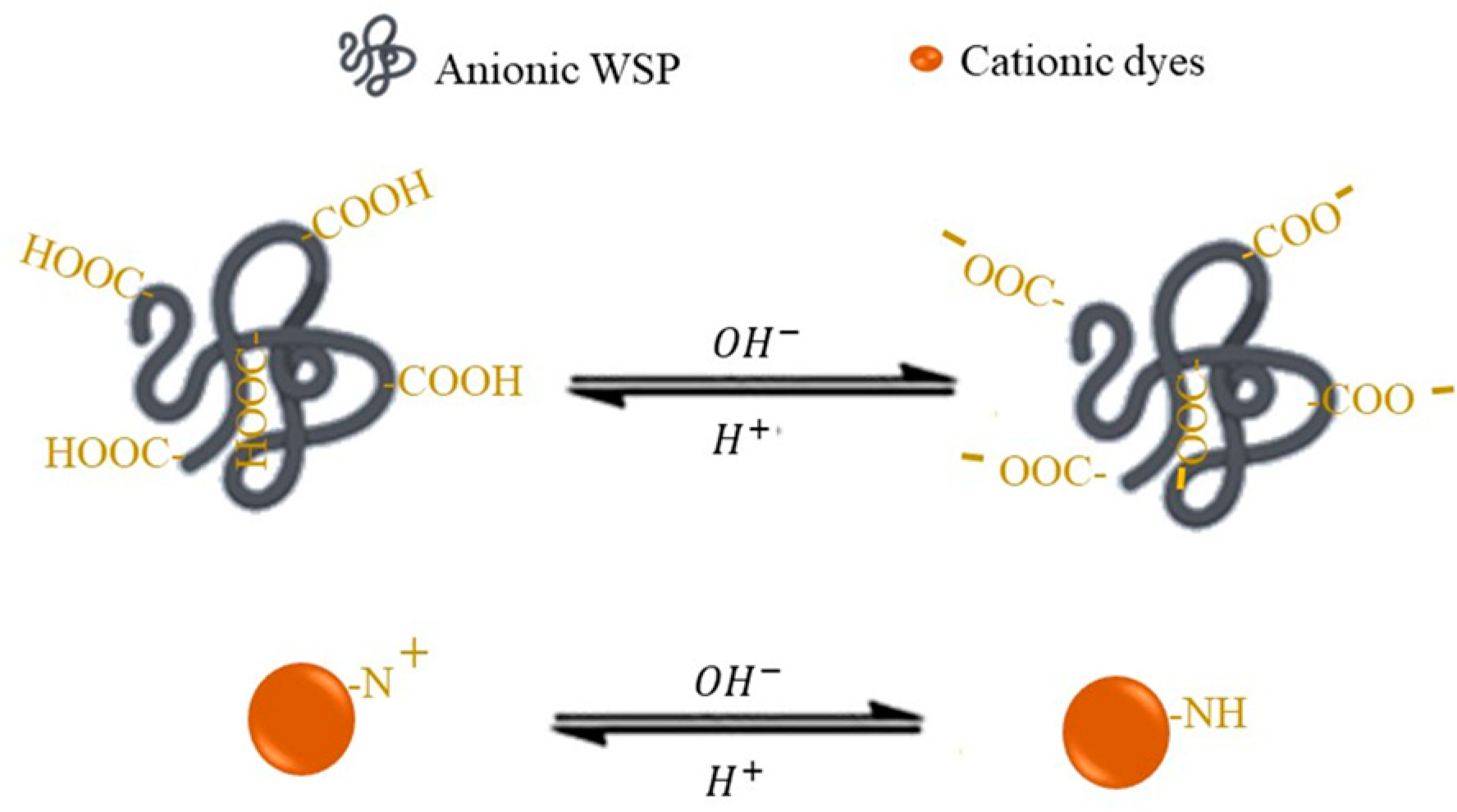
5.1. Effect of pH on Dye Removal
5.2. Effect of Ionic Strength on Dye Removal
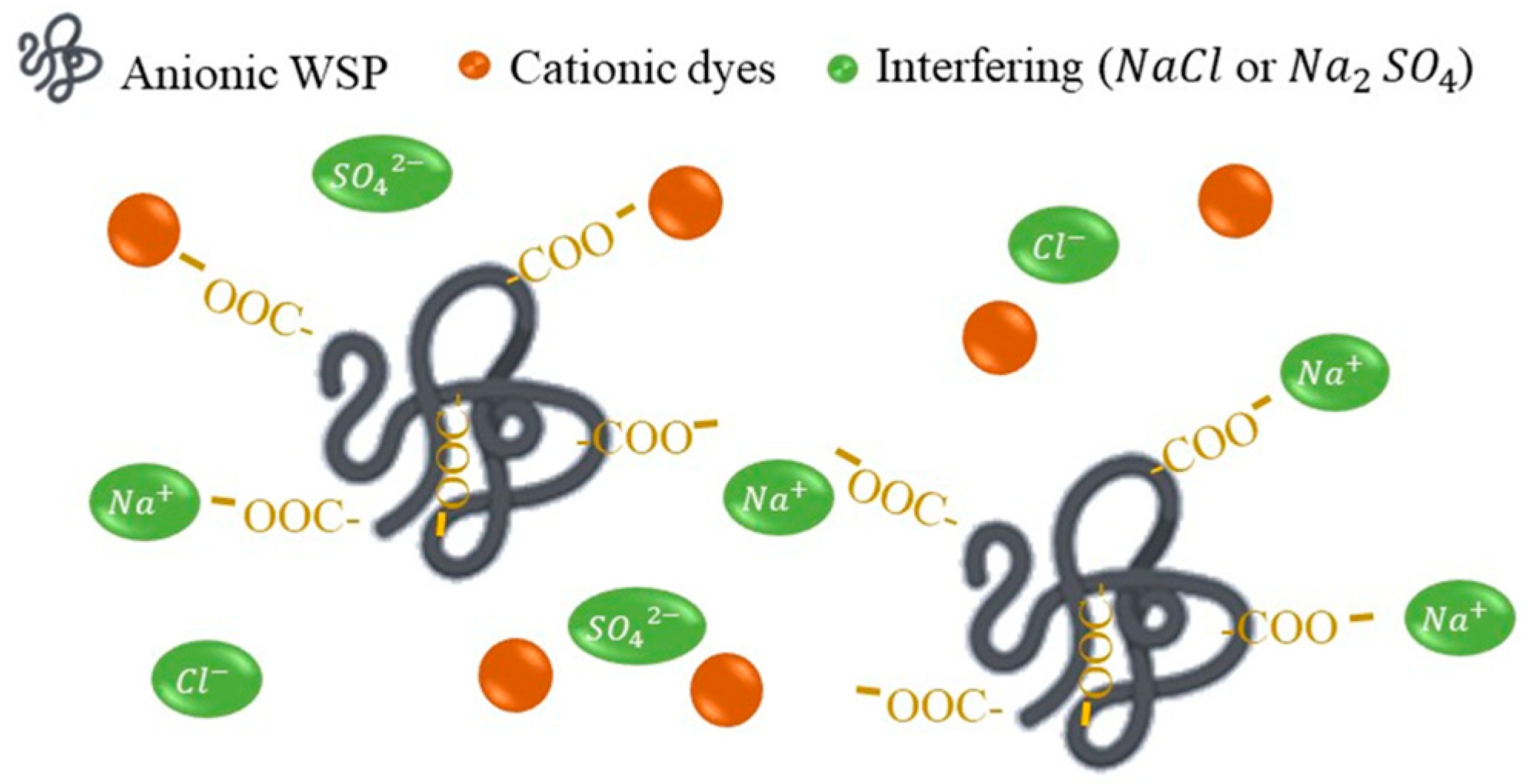
5.3. Effect of Interfering Ions on Dye Removal
5.4. Effect of Polymer and Dye Concentration on Dye Removal
5.5. Effect of the Membrane on Dye Removal
6. Computational Studies
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Islam, A.; Teo, S.; Taufiq-Yap, Y.; Ng, C.; Vo, D.-V.; Ibrahim, M.; Hasan, M.; Khan, M.; Nur, A.; Awual, M. Step towards the sustainable toxic dyes and heavy metals removal and recycling from aqueous solution-A comprehensive review. Resour. Conserv. Recycl. 2021, 175, 105849. [Google Scholar] [CrossRef]
- Fradj, A.B.; Boubakri, A.; Hafiane, A.; Hamouda, S.B. Removal of azoic dyes from aqueous solutions by chitosan enhanced ultrafiltration. Results Chem. 2019, 2, 100017. [Google Scholar] [CrossRef]
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile finishing dyes and their impact on aquatic environs. Heliyon 2019, 5, e02711. [Google Scholar] [CrossRef]
- Şen, F.; Demirbaş, Ö.; Çalımlı, M.; Aygün, A.; Alma, M.; Nas, M. The dye removal from aqueous solution using polymer composite films. Appl. Water Sci. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Tuncer, C.; Kilinc, M.; Butun, V. Removal of dye contaminants from waste water by using highly cross-linked polymer as novel adsorbent, referencial manual. J. Nanomed. Nanotechnol. 2016, 7, 7439. [Google Scholar]
- Dasgupta, J.; Sikder, J.; Mandal, T.; Adhikari, U. Reactive red 120 retentions through ultrafiltration enhanced by synthetic and natural polyelectrolytes. J. Hazard. Mater. 2015, 299, 192–205. [Google Scholar] [CrossRef]
- Garza-Montelongo, E.; Pérez-Rodríguez, N.; Sánchez-Anguiano, M.; Rivas-Quiroz, B.; Elizondo-Martínez, P. Molecularly imprinted polymer for the binding and recognition of basic blue 41 dye. J. Chil. Chem. Soc. 2021, 66, 5215–5219. [Google Scholar] [CrossRef]
- Beluci, N.D.; Mateus, G.A.; Miyashiro, C.S.; Homem, N.C.; Gomes, R.G.; Fagundes-klen, M.R.; Bergamasco, R.; Vieira, A.M. Hybrid treatment of coagulation/flocculation process followed by ultrafiltration in TIO2 -modified membranes to improve the removal of reactive black 5 dye. Sci. Total Environ. 2019, 664, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Paz, A.; Carballo, J.; Pérez, M.J.; Domínguez, J.M. Biological treatment of model dyes and textile wastewaters. Chemosphere 2017, 181, 168–177. [Google Scholar] [CrossRef]
- Nidheesh, P.V.; Zhou, M.; Oturan, M.A. An overview on the removal of synthetic dyes from water by electrochemical advanced oxidation processes. Chemosphere 2018, 197, 210–227. [Google Scholar] [CrossRef]
- Zhan, C.; Sharma, P.; He, H.; Sharma, S.; McCauley-Pearl, A.; Wang, R.; Hsiao, B. Rice husk based nanocellulose scaffolds for highly efficient removal of heavy metal ions from contaminated water. Environ. Sci. Water Res. Technol. 2020, 6, 3080–3090. [Google Scholar] [CrossRef]
- Ghaedi, A.M.; Vafaei, A. Applications of artificial neural networks for adsorption removal of dyes from aqueous solution: A review. Adv. Colloid Interface Sci. 2017, 245, 20–39. [Google Scholar] [CrossRef]
- Sharma, P.; Sharma, S.; Lindström, T.; Hsiao, B. Nanocellulose-enabled membranes for water purification: Perspectives. Adv. Sustain. Syst. 2020, 4, 1900114. [Google Scholar] [CrossRef]
- Pavithra, K.G.; Kumar, P.S.; Jaikumar, V.; Rajan, P.S. Removal of colorants from wastewater: A review on sources and treatment strategies. J. Ind. Eng. Chem. 2019, 75, 1–19. [Google Scholar] [CrossRef]
- Huang, Y.; Feng, X. Polymer-enhanced ultrafiltration: Fundamentals, applications and recent developments. J. Membr. Sci. 2019, 586, 53–83. [Google Scholar] [CrossRef]
- Yang, L.; Wang, Z.; Zhang, J. Zeolite imidazolate framework hybrid nanofiltration (NF) membranes with enhanced permselectivity for dye removal. J. Membr. Sci. 2017, 532, 76–86. [Google Scholar] [CrossRef]
- Su, Y.; Yan, X.; Chen, Y.; Guo, X.; Chen, X.; Lang, W. Facile fabrication of COF-LZU1/PES composite membrane via interfacial polymerization on microfiltration substrate for dye/salt separation. J. Membr. Sci. 2021, 618, 118706. [Google Scholar] [CrossRef]
- Saja, S.; Bouazizi, A.; Achiou, B.; Ouaddari, H.; Karim, A.; Ouammou, M.; Addane, A.; Bennazha, J.; Younssi, S. Fabrication of low-cost ceramic ultrafiltration membrane made frombentonite clay and its application for soluble dyes removal. J. Eur. Ceram. Soc. 2020, 40, 2453–2462. [Google Scholar] [CrossRef]
- Bukman, L.; De Souza, V.; Camargo, N.; Caetano, W.; Batistela, V.; Hioka, N. Reverse micellar extraction of dyes based on fatty acids and recoverable organic solvents. Sep. Purif. Technol. 2020, 242, 116772. [Google Scholar] [CrossRef]
- Dang, T.; Li, C.-W.; Choo, K.-H. Comparison of low-pressure reverse osmosis filtration and polyelectrolyte-enhanced ultrafiltration for the removal of Co and Sr from nuclear plant wastewater. Sep. Purif. Technol. 2016, 157, 209–214. [Google Scholar] [CrossRef]
- Fradj, A.B.; Ouni, H.; Hamouda, S.B.; Hafiane, A. The effect of operating conditions on the retention of Toluidine blue by polyelectrolyte-enhanced ultrafiltration. Desalin. Water Treat. 2015, 56, 2766–2772. [Google Scholar] [CrossRef]
- Yuksel, S.; Rivas, R.; Sánchez, J.; Mansilla, H.; Yañez, J.; Kochifas, P.; Kabay, N.; Bryjak, M. Water-soluble polymer and photocatalysis for arsenic removal. J. Appl. Polym. Sci. 2014, 131, 40871–40876. [Google Scholar] [CrossRef]
- Arar, O.; Kabay, N.; Sánchez, J.; Rivas, B.; Bryjak, M.; Peña, C. Removal of arsenic from water by combination of electro-oxidation and polymer enhanced ultrafiltration. Environ. Prog. Sustain. Energy 2014, 33, 918–924. [Google Scholar] [CrossRef]
- Valle, H.; Sánchez, J.; Rivas, B. Poly(N-vinylpyrrolidone-co-2-acrylamido-2-methylpropanesulfonate sodium): Synthesis, characterization, and its potential application for the removal of metal ions from aqueous solution. J. Appl. Polym. Sci. 2015, 132, 41272–41278. [Google Scholar] [CrossRef]
- Pereira, E.; Homper, D.; Sánchez, J.; Rivas, B. Removal of metal ions from Bío Bío river water by liquid-phase polymer-based retention technique. J. Chil. Chem. Soc. 2015, 60, 2868–2872. [Google Scholar] [CrossRef]
- Sánchez, J.; Rivas, B. Liquid-phase polymer-based retention of chromate and arsenate oxy-anions. Macromol. Symp. 2012, 317–318, 123–136. [Google Scholar] [CrossRef]
- Toledo, L.; Rivas, B.; Urbano, B.; Sánchez, J. Novel N-methyl-D-glucamine-based water-soluble polymer and its potential application in the removal of arsenic. Sep. Purif. Technol. 2013, 103, 1–7. [Google Scholar] [CrossRef]
- Oyarce, E.; Santander, P.; Butter, B.; Pizarro, G.D.C.; Sánchez, J. Use of sodium alginate biopolymer as an extracting agent of methylene blue in the polymer-enhanced ultrafiltration technique. J. Appl. Polym. Sci. 2021, 138, 50844. [Google Scholar] [CrossRef]
- Sánchez, J.; Rivas, B. Arsenate retention from aqueous solution by hydrophilic polymers through ultrafiltration membranes. Desalination 2011, 270, 57–63. [Google Scholar] [CrossRef]
- Palencia, M.; Rivas, B.; Pereira, E. Metal ion recovery by polymer-enhanced ultrafiltration using poly (vinyl sulfonic acid): Fouling description and membrane–metal ion interaction. J. Membr. Sci. 2009, 345, 191–200. [Google Scholar] [CrossRef]
- Fradj, A.B.; Hamouda, S.B.; Ouni, H.; Lafi, R.; Gzara, L.; Hafiane, A. Removal of methylene blue from aqueous solutions by poly (acrylic acid) and poly (ammonium acrylate) assisted ultrafiltration. Sep. Purif. Technol. 2014, 133, 76–81. [Google Scholar] [CrossRef]
- Heo, J.; Flora, J.; Her, N.; Park, Y.; Cho, J.; Son, A.; Yoon, Y. Removal of bisphenol A and 17β-estradiol in single walled carbon nanotubes–ultrafiltration (SWNTs–UF) membrane systems. Sep. Purif. Technol. 2012, 90, 39–52. [Google Scholar] [CrossRef]
- Fradj, A.B.; Lafi, R.; Hamouda, S.B.; Gzara, L.; Hamzaoui, A.H.; Hafiane, A. Effect of chemical parameters on the interaction between cationic dyes and poly (acrylic acid). J. Photochem. Photobiol. A 2014, 284, 49–54. [Google Scholar] [CrossRef]
- Palacio, D.; Urbano, B.; Rivas, B. Water-soluble polymers with the ability to remove amoxicillin as emerging pollutant from water. Environ. Technol. Innov. 2021, 23, 101589. [Google Scholar] [CrossRef]
- Chakraborty, S.; Dasgupta, J.; Farooq, U.; Sikder, J.; Drioli, E.; Curcio, S. Experimental analysis, modeling and optimization of chromium (VI) removal from aqueous solutions by polymer-enhanced ultrafiltration. J. Membr. Sci. 2014, 456, 139–154. [Google Scholar] [CrossRef]
- Nicolai, S.; Tralau, T.; Luch, A.; Pirow, R. A scientific review of colorful textiles. J. Consum. Prot. Food Saf. 2021, 16, 5–17. [Google Scholar] [CrossRef]
- Benkhaya, S.; M’rabet, S.; El Harfi, A. A review on classifications, recent synthesis and applications of textile dyes. Inorg. Chem. Commun. 2020, 115, 107891. [Google Scholar] [CrossRef]
- Ruiz, C.; Vera, M.; Rivas, B.; Sánchez, S.; Urbano, B. Magnetic methacrylated gelatin-g-polyelectrolyte for methylene blue sorption. RSC Adv. 2020, 10, 43799–43810. [Google Scholar] [CrossRef]
- Dassanayake, R.; Acharya, S.; Abidi, N. Recent advances in biopolymer-based dye removal technologies. Molecules 2021, 26, 4697. [Google Scholar] [CrossRef] [PubMed]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.; Bilal, M.; Chandra, R.; Bharagavaunet, R.N. Ecotoxicological and health concerns of persistent coloring pollutants of textile industry wastewater and treatment approaches for environmental safety. J. Environ. Chem. Eng. 2021, 9, 105012. [Google Scholar] [CrossRef]
- Saini, R.D. Textile Organic Dyes: Polluting effects and elimination methods from textile waste water. Int. J. Chem. Eng. 2017, 9, 121–136. [Google Scholar]
- Mokif, L.A. Removal methods of synthetic dyes from industrial wastewater: A review. Mesop. Environ. J. 2019, 5, 23–40. [Google Scholar]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Ishak, S.; Murshed, M.; Akil, H.; Ismail, N.; Rasib, S.; Al-Gheethi, A. The application of modified natural polymers in toxicant dye compounds wastewater: A review. Water 2020, 12, 2032. [Google Scholar] [CrossRef]
- Roa, K.; Oyarce, E.; Boulett, A.; ALSamman, M.; Oyarzún, D.; Pizarro, G.D.C.; Sánchez, J. Lignocellulose-based materials and their application in the removal of dyes from water: A review. Sustain. Mater. Technol. 2021, 29, e00320. [Google Scholar]
- Rahimian, R.; Zarinabadi, S. A review of studies on the removal of methylene blue dye from industrial wastewater using activated carbon adsorbents made from almond bark. Prog. Chem. Biochem. Res. 2020, 3, 251–268. [Google Scholar]
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: A review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef] [PubMed]
- Roldan, C.J.; Nouri, K.; Chai, T.; Huh, B. Methylene blue for the treatment of intractable pain associated with oral mucositis. Pain Pract. 2017, 17, 1115–1121. [Google Scholar] [CrossRef]
- Alda, M. Methylene blue in the treatment of neuropsychiatric disorders. CNS Drugs 2019, 33, 719–725. [Google Scholar] [CrossRef]
- Rozenholc, A.; Samouelian, V.; Warkus, T.; Gauthier, P.; Provencher, D.; Sauthier, P.; Gauthier, F.; Drakopoulos, P.; Cormier, B. Green versus blue: Randomized controlled trial comparing indocyanine green with methylene blue for sentinel lymph node detection in endometrial cancer. Gynecol. Oncol. 2019, 153, 500–504. [Google Scholar] [CrossRef]
- Ighalo, J.O.; Iwuozor, K.O.; Igwegbe, C.A.; Adeniyi, A.G. Verification of pore size effect on aqueous-phase adsorption kinetics: A case study of methylene blue. Colloids Surf. A Physicochem. Eng. Asp. 2021, 626, 127119. [Google Scholar] [CrossRef]
- Minamoto, C.; Fujiwara, N.; Shigekawa, Y.; Tada, K.; Yano, J.; Yokoyama, T.; Minamoto, Y.; Nakayama, S. Effect of acidic conditions on decomposition of methylene blue in aqueous solution by air microbubbles. Chemosphere 2021, 263, 128141. [Google Scholar] [CrossRef]
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of methylene blue on low-cost adsorbents: A review. J. Hazard. Mater. 2010, 177, 70–80. [Google Scholar] [CrossRef]
- Ramirez, L.A.; Jacobo, A.; Martínez, J.M. Adsorción del naranja de metilo en solución acuosa sobre hidróxidos dobles laminares. Acta Univ. 2015, 25, 25–34. [Google Scholar]
- Wu, L.; Liu, X.; Lv, G.; Zhu, R.; Tian, L.; Liu, M.; Li, Y.; Rao, W.; Liu, T.; Liao, L. Study on the adsorption properties of methyl orange by natural one-dimensional nano-mineral materials with different structures. Sci. Rep. 2021, 11, 1–11. [Google Scholar]
- Palencia, M. Liquid-phase polymer-based retention: Theory, modeling, and application for the removal of pollutant inorganic ions. J. Chem. 2015, 2015, 965624. [Google Scholar] [CrossRef]
- Sánchez, J.; Rivas, B. Arsenic extraction from aqueous solution: Electrochemical oxidation combined to ultrafiltration membranes and water-soluble polymers. Chem. Eng. J. 2010, 165, 625–632. [Google Scholar] [CrossRef]
- Palacio, D.; Becerra, Y.; Urbano, B.F.; Rivas, B. Antibiotics removal using a chitosan-based polyelectrolyte in conjunction with ultrafiltration membranes. Chemosphere 2020, 258, 127416. [Google Scholar] [CrossRef]
- Palacio, D.; Leiton, L.; Urbano, B.; Rivas, B. Tetracycline removal by polyelectrolyte copolymers in conjunction with ultrafiltration membranes through liquid-phase polymer-based retention. Environ. Res. 2020, 182, 109014. [Google Scholar] [CrossRef]
- Crini, G.; Morin-Crini, N.; Fatin-Rouge, N.; Deon, S.; Fievet, P. Metal removal from aqueous media by polymer-assisted ultrafiltration with chitosan. Arab. J. Chem. 2017, 10, S3826–S3839. [Google Scholar] [CrossRef]
- Izumi, T.; Takahashi, K.; Hirata, M. Interactions of water-soluble polymers with small molecules I: Poly (2-diethylaminoethyl methacrylate)-orange II and ethyl orange systems. Colloid Polym. Sci. 1986, 264, 748–755. [Google Scholar] [CrossRef]
- Sánchez, J.; Toledo, L.; Rivas, B.; Rivera, N.; Muñoz, E. Water-soluble cationic cellulose coupled to a ultrafiltration membrane for the removal of arsenic and chromium. J. Chil. Chem. Soc. 2013, 58, 1842–1846. [Google Scholar] [CrossRef]
- Fane, A.G.; Tang, C.Y.; Wang, R. Membrane technology for water: Microfiltration, ulrafiltration, nanofiltration, and reverse osmosis. Treatise Water Sci. 2011, 4, 301–335. [Google Scholar]
- Yaghoubi, Z.; Basiri-Parsa, J. Modification of ultrafiltration membrane by thermo-responsive Bentonite-poly (N-isopropylacrylamide) nanocomposite to improve its antifouling properties. J. Water Process. Eng. 2020, 34, 101067. [Google Scholar] [CrossRef]
- Xie, W.; Tiraferri, A.; Ji, X.; Chen, C.; Bai, Y.; Crittenden, J.; Liu, B. Green and sustainable method of manufacturing anti-fouling zwitterionic polymers-modified poly (vinyl chloride) ultrafiltration membranes. J. Colloid Interface Sci. 2021, 591, 343–351. [Google Scholar] [CrossRef]
- Al Aani, S.; Mustafa, T.; Hilal, N. Ultrafiltration membranes for wastewater and water process engineering: A comprehensive statistical review over the past decade. J. Water Process. Eng. 2020, 35, 101241. [Google Scholar] [CrossRef]
- Ilyas, A.; Mertens, M.; Oyaert, S.; Vankelecom, I. Synthesis of patterned PVDF ultrafiltration membranes: Spray-modified non-solvent induced phase separation. J. Membr. Sci. 2020, 612, 118383. [Google Scholar] [CrossRef]
- Rivas, B.; Pereira, E.; Moreno-Villoslada, I. Water-soluble polymer–metal ion interactions. Prog. Polym. Sci. 2003, 28, 173–208. [Google Scholar] [CrossRef]
- Palacio, D.; Rivas, B.; Urbano, B. Ultrafiltration membranes with three water-soluble polyelectrolyte copolymers to remove ciprofloxacin from aqueous systems. Chem. Eng. J. 2018, 351, 85–93. [Google Scholar] [CrossRef]
- Palencia, M.; Martínez, J.M.; Arrieta, Á. Polymer-enhanced ultrafiltration: Fundamentals, blue 129 dye by polymer-enhanced ultrafiltration (PEUF). J. Sci. Educ. Technol. 2017, 2, 65–74. [Google Scholar]
- Sánchez, J.; Rivas, B.; Pooley, S.; Basaez, L.; Pereira, E.; Pignot-Paintrand, I.; Bucher, C.; Royal, G.; Saint-Aman, E.; Moutet, J.-C. Electrocatalytic oxidation of As(III) to As(V) using noble metal-polymer nanocomposites. Electrochim. Acta 2010, 55, 4876–4882. [Google Scholar] [CrossRef]
- Mondal, S.; Ouni, H.; Dhahbi, M. Kinetic modeling for dye removal using polyelectrolyte enhanced ultrafiltration. J. Hazard. Mater. 2012, 229–230, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, J.; Sikder, J.; Mandal, D. Modeling and optimization of polymer enhanced ultrafiltration using hybrid neural-genetic algorithm based evolutionary approach. Appl. Soft Comput. 2017, 55, 108–126. [Google Scholar] [CrossRef]
- Chmilenko, F.A.; Korobova, I.V.; Nazarenko, S.V. Spectrophotometric determination of polyacrylamide in aqueous solutions using cationic dyes. J. Anal. Chem. 2004, 59, 143–148. [Google Scholar] [CrossRef]
- Dasgupta, J.; Singh, A.; Kumar, S.; Sikder, J.; Chakraborty, S.; Curcio, S.; Arafat, H. Poly (sodium-4-styrenesulfonate) assisted ultrafiltration for methylene blue dye removal from simulated wastewater: Optimization using response surface methodology. J. Environ. Chem. Eng. 2016, 4, 2008–2022. [Google Scholar] [CrossRef]
- Moreno-Villoslada, I.; Torres, C.; González, F.; Shibue, T.; Nishide, H. Binding of methylene blue to polyelectrolytes containing sulfonate groups. Macromol. Chem. Phys. 2009, 210, 1167–1175. [Google Scholar] [CrossRef]
- Majewska-Nowak, K. The effect of a polyelectrolyte on the efficiency of dye-surfactant solution treatment by ultrafiltration. Desalination 2008, 221, 395–404. [Google Scholar] [CrossRef]
- Ouni, H.; Hafiane, A.; Dhahbi, M. The effect of surfactant on dye removal by polyelectrolyte enhanced ultrafiltration. Desalin. Water Treat. 2015, 56, 1526–1535. [Google Scholar] [CrossRef]
- Schwarze, M.; Seo, D.; Bibouche, B.; Schomäcker, R. Comparison of positively charged polymer species and cationic surfactants for methyl orange removal via polyelectrolyte and micellar enhanced ultrafiltration. J. Water Process. Eng. 2020, 36, 101287. [Google Scholar] [CrossRef]
- Ouni, H.; Dhahbi, M. Removal of dyes from wastewater using polyelectrolyte enhanced ultrafiltration (PEUF). Desalin. Water Treat. 2010, 22, 355–362. [Google Scholar] [CrossRef]
- Dasgupta, J.; Singh, M.; Sikder, J.; Padarthi, V.; Chakraborty, S.; Curcio, S. Response surface-optimized removal of Reactive Red 120 dye from its aqueous solutions using polyethyleneimine enhanced ultrafiltration. Ecotoxicol. Environ. Saf. 2015, 121, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Cojocaru, C.; Clima, L. Polymer assisted ultrafiltration of AO7 anionic dye from aqueous solutions: Experimental design, multivariate optimization, and molecular docking insights. J. Membr. Sci. 2020, 604, 118054. [Google Scholar] [CrossRef]
- Guibal, E.; Touraud, E.; Roussy, J. Chitosan interactions with metal ions and dyes: Dissolved-state vs. solid-state application. World J. Microbiol. Biotechnol. 2005, 21, 913–920. [Google Scholar] [CrossRef]
- Izumi, T.; Hirata, M. Interactions of water-soluble polymers with small molecules II: Poly (2-diethylaminoethyl methacrylate)-methyl orange and its homolog systems at different temperatures. Colloid Polym. Sci. 1986, 264, 756–763. [Google Scholar] [CrossRef]
- Tan, X.; Kyaw, N.N.; Teo, W.K.; Li, K. Decolorization of dye-containing aqueous solutions by the polyelectrolyte-enhanced ultrafiltration (PEUF) process using a hollow fiber membrane module. Sep. Purif. Technol. 2006, 52, 110–116. [Google Scholar] [CrossRef]
- Ouni, H.; Dhahbi, M. Spectrometric study of crystal violet in presence of polyacrylic acid and polyethylenimine and its removal by polyelectrolyte enhanced ultrafiltration. Sep. Purif. Technol. 2010, 72, 340–346. [Google Scholar] [CrossRef]
- Rivas, B.; Sánchez, J.; Pooley, S.; Basaez, L.; Pereira, E.; Bucher, C.; Royal, G.; Saint-Aman, E.; Moutet, J.-C. Water-soluble polyelectrolyte with ability to remove arsenic. Macromol. Symp. 2010, 296, 416–428. [Google Scholar] [CrossRef]
- Sánchez, J.; Butter, B.; Rivas, B.L. Biopolymers applied to remove metal ions through ultrafiltration. A review. J. Chil. Chem. Soc. 2020, 3, 5004–5010. [Google Scholar] [CrossRef]
- Oyarce, E.; Butter, B.; Santander, P.; Sánchez, J. Polyelectrolytes applied to remove methylene blue and methyl orange dyes from water via polymer-enhanced ultrafiltration. J. Environ. Chem. Eng. 2021, 106297, in press. [Google Scholar] [CrossRef]
- Sánchez, J.; Rodriguez, C.; Oyarce, E.; Rivas, B.L. Removal of chromium ions by functional polymers in conjunction with ultrafiltration membranes. Pure Appl. Chem. 2020, 92, 883–896. [Google Scholar] [CrossRef]
- Sánchez, J.; Riffo, L.; Salazar, P.; Rivas, B. Removal of molybdate and vanadate ions by a copolymer adsorbent in a ultrafiltration system. J. Appl. Polym. Sci. 2019, 136, 48184. [Google Scholar] [CrossRef]
- Rivas, B.; Espinosa, C.; Sánchez, J. Application of the liquid-phase polymer-based retention technique to the sorption of molybdenum (VI) and vanadium(V). Polym. Bull. 2019, 76, 539–552. [Google Scholar] [CrossRef]
- Sánchez, J.; Espinosa, C.; Pooch, F.; Tenhu, H.; Pizarro, G.; Oyarzún, D. Poly(N,N-dimethylaminoethyl methacrylate) for removing chromium (VI) through polymer-enhanced ultrafiltration technique. React. Funct. Polym. 2018, 127, 67–73. [Google Scholar] [CrossRef]
- Rivas, B.; Pereira, E.; Palencia, M.; Sánchez, J. Water-soluble functional polymers in conjunction with membranes to remove pollutant ions from aqueous solutions. Prog. Polym. Sci. 2011, 36, 294–322. [Google Scholar] [CrossRef]
- Hegazy, S.; Abdel-AAl, S.; Abdel-Rehim, H.; Khalifa, N.; El-Hosseiny, E. Removal of some basic dyes by poly (Vinyl Alcohol/acrylic acid) Hydrogel. In Proceedings of the Seventh Conference of Nuclear Sciences and Applications, Cairo, Egypt, 6–10 January 2000. [Google Scholar]
- Rivas, B.; Urbano, B.; Sánchez, J. Water-soluble and insoluble polymers, nanoparticles, nanocomposites and hybrids with ability to remove hazardous inorganic pollutants in water. Front. Chem. 2018, 6, 320. [Google Scholar] [CrossRef]
- Hooda, R.; Singh, N.; Sharma, M. Chitosan nanoparticles: A marine polysaccharide for biomedical research. In Marine Polysaccharides, 1st ed.; Jenny Stanford Publishing: Singapore, 2019; pp. 141–168. [Google Scholar]
- Song, S.; Hao, C.; Zhang, X.; Zhang, Q.; Sun, R. Sonocatalytic degradation of methyl orange in aqueous solution using Fe-doped TiO2 nanoparticles under mechanical agitation. Open Chem. J. 2018, 16, 122–135. [Google Scholar] [CrossRef]
- Li, S. Removal of crystal violet from aqueous solution by sorption into semi-interpenetrated networks hydrogels constituted of poly (acrylic acid-acrylamide-methacrylate) and amylose. Bioresour. Technol. 2010, 101, 2197–2202. [Google Scholar] [CrossRef]
- Sánchez, J.; Rivas, B. Cationic hydrophilic polymers coupled to ultrafiltration membranes to remove chromium (VI) from aqueous solution. Desalination 2011, 279, 338–343. [Google Scholar] [CrossRef]
- Ouyang, C.; Chen, S.; Che, B.; Xue, G. Aggregation of azo dye orange I induced by polyethylene glycol in aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2007, 301, 346–351. [Google Scholar] [CrossRef]
- Lau, W.-J.; Ismail, A. Polymeric nanofiltration membranes for textile dye wastewater treatment: Preparation, performance evaluation, transport modelling, and fouling control—A review. Desalination 2009, 245, 321–348. [Google Scholar] [CrossRef]
- Takagishi, T.; Kozuka, H.; Kuroki, N. Binding of methyl orange and its homologs by polyion complexes in aqueous solution. J. Polym. Sci. 1981, 19, 3237–3246. [Google Scholar] [CrossRef]
- Rivas, B.; Pereira, E.; Paredes, J.; Sánchez, J. Removal of arsenate from ionic mixture by anion exchanger water-soluble polymers and ultrafiltration membranes. Polym. Bull. 2012, 69, 1007–1022. [Google Scholar] [CrossRef]
- Allègre, C.; Moulin, P.; Maisseu, M.; Charbit, F. Treatment and reuse of reactive dyeing effluents. J. Membr. Sci. 2006, 269, 15–34. [Google Scholar] [CrossRef]
- Sánchez, J.; Bastrzyk, A.; Rivas, B.; Bryjak, M.; Kabay, N. Removal of As(V) using liquid-phase polymer-based retention (LPR) technique with regenerated cellulose membrane as a filter. Polym. Bull. 2013, 70, 2633–2644. [Google Scholar] [CrossRef]
- Rivas, B.; Hube, S.; Sánchez, J.; Pereira, E. Chelating water-soluble polymers associated with ultrafiltration membranes for metal ion removal. Polym. Bull. 2012, 69, 881–898. [Google Scholar] [CrossRef]
- Sánchez, J.; Rivas, B.; Nazar, E.; Bryjak, M.; Kabay, N. Boron removal by liquid-phase polymer-based retention (LPR) technique using poly(glycidyl methacrylate N-methyl D-glucamine). J. Appl. Polym. Sci. 2013, 129, 1541–1545. [Google Scholar] [CrossRef]
- Musale, D.; Kulkarni, S. Effect of membrane-solute interactions on ultrafiltration performance. J. Macromol. Sci. Polymer Rev. 1998, 38, 615–636. [Google Scholar] [CrossRef]
- Singh, A. Industrial membrane separation processes. Chemtech 1998, 28, 33–44. [Google Scholar]
- Prathipati, S.; Aangothu, S.; Munnangi, S.; Krishna, B.; Bollikolla, H. Volumetric, ultrasonic, spectroscopic, and molecular dynamics simulations studies of molecular interactions in binary mixtures of [EMIm][NTf2] with 2-propoxyethanol at temperatures from 298.15 to 318.15 K. J. Chem. Eng. Data. 2020, 65, 3566–3578. [Google Scholar] [CrossRef]
- Jabbari, F.; Saedodin, S.; Rajabpour, A. Experimental investigation and molecular dynamics simulations of viscosity of CNT-water nanofluid at different temperatures and volume fractions of nanoparticles. J. Chem. Eng. Data. 2019, 64, 262–272. [Google Scholar] [CrossRef]
- Cojocaru, C.; Clima, L. Binding assessment of methylene blue to human serum albumin and poly (acrylic acid): Experimental and computer-aided modeling studies. J. Mol. Liq. 2019, 285, 811–821. [Google Scholar] [CrossRef]
- Krieger, E.; Vriend, G. YASARA View-molecular graphics for all devices-from smartphones to workstations. Bioinformatics 2014, 30, 2981–2982. [Google Scholar] [CrossRef] [PubMed]

| Dyes | Molecular Structure | References |
|---|---|---|
| Anionic | ||
| MO | 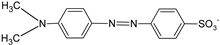 | [2,21,77,78,79] |
| [2,21,77,79] DB71 | 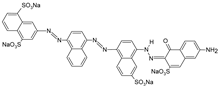 | [2] |
| EBBR | 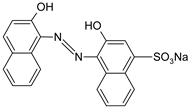 | [72,80] |
| RR 120 | 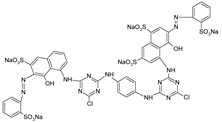 | [72,80,81] |
| AO7 | 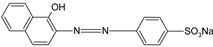 | [82] |
| IC | 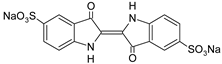 | [77] |
| AB | 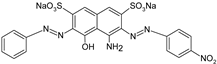 | [77] |
| DB |  | [77] |
| RB5 | 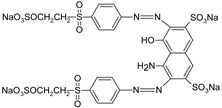 | [83] |
| AB129 | 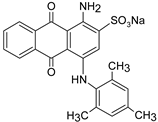 | [70] |
| OII | 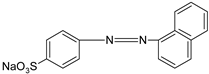 | [61,84] |
| EO |  | [61,84] |
| Cationic | ||
| ST | 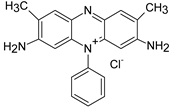 | [72,80] |
| MG | 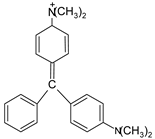 | [85] |
| BG | 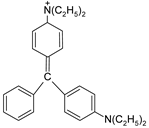 | [85] |
| NF |  | [85] |
| CV |  | [70,86] |
| MB |  | [28,74,76] |
| TB |  | [21,33,74] |
| AY |  | [74] |
| Anionic WSP | Basic Dyes | References |
PAA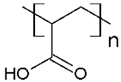 | CV, ST, TB and MB | [21,31,72,80] |
SA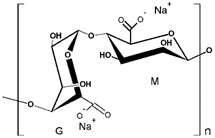 | MB | [28,31] |
PAMPS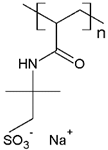 | MB | [76] |
PVS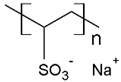 | MB | [76] |
PSS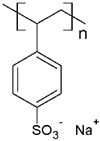 | MB, NF, BG and MG | [75,85] |
PANH4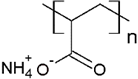 | MB, ST, CV and TB | [21,31,72] |
| Cationic WSP | Acid Dyes | References |
PEI | AO7 and RR 120 | [6,73,82] |
Poly(2-diethylaminoethyl methacrylate) (PDEAEMA) | OII and EO | [61,84] |
CHI | RB5, DB71 and MO | [2,83] |
| WSP | Dye | Membrane | pH | Initial Dye (mg L–1) | Polymer Dosage (mg L−1) | T (°C) | Time (min) | Retention Rate (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| CHI | MO | Cellulose | 4.5 | 32.7 | 116 | 25 | 5–80 | 86 | [2] |
| DB71 | 10 kDa | 102.9 | 89 | ||||||
| PEI | RR 120 | PES-30 | 8.4 | 50 | 50 | 35 | 10 | 99 | [6] |
| CHI | 30 kDa | 6.3 | 88 | ||||||
| SA | MB | Cellulose | 8 | 50 | 1250 | 20 | --- | 98 | [28] |
| 10 kDa | |||||||||
| PAA | MB | Cellulose | 4.06 | 31.9 | 28.8 | 25 | 10–100 | 99 | [31] |
| PANH4 | 10 kDa | 8.24 | 35.6 | 98 | |||||
| PAA | TB | --- | 2–6 | 27 | 8.6 | 25 | 180 | 89 | [33] |
| 4.5 | |||||||||
| MO | 2–6 | 32.7 | 14.4 | 86 | |||||
| 4.5 | |||||||||
| PEG | MO | Cellulose | 4 | 163.6 | 3360 | --- | 30 | 65 | [78] |
| 10kDa | |||||||||
| PANH4 | ST | Cellulose | 4–10 | 20 | 10 | --- | 10–90 | 99 | [80] |
| EBBR | 10kDa | 6–10 | 10 | 30 | --- | Oct-90 | 90 | ||
| PEI | RR120 | PES-30 | 10 | 50 | 150 | 35 | 10 | 99.94 | [81] |
| 30 kDa | |||||||||
| PAA | CV | Oss-flow | 10–12 | 2 | 20 | --- | 30 | 95 | [86] |
| 10kDa |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oyarce, E.; Roa, K.; Boulett, A.; Sotelo, S.; Cantero-López, P.; Sánchez, J.; Rivas, B.L. Removal of Dyes by Polymer-Enhanced Ultrafiltration: An Overview. Polymers 2021, 13, 3450. https://doi.org/10.3390/polym13193450
Oyarce E, Roa K, Boulett A, Sotelo S, Cantero-López P, Sánchez J, Rivas BL. Removal of Dyes by Polymer-Enhanced Ultrafiltration: An Overview. Polymers. 2021; 13(19):3450. https://doi.org/10.3390/polym13193450
Chicago/Turabian StyleOyarce, Estefanía, Karina Roa, Andrés Boulett, Sebastián Sotelo, Plinio Cantero-López, Julio Sánchez, and Bernabé L. Rivas. 2021. "Removal of Dyes by Polymer-Enhanced Ultrafiltration: An Overview" Polymers 13, no. 19: 3450. https://doi.org/10.3390/polym13193450
APA StyleOyarce, E., Roa, K., Boulett, A., Sotelo, S., Cantero-López, P., Sánchez, J., & Rivas, B. L. (2021). Removal of Dyes by Polymer-Enhanced Ultrafiltration: An Overview. Polymers, 13(19), 3450. https://doi.org/10.3390/polym13193450









