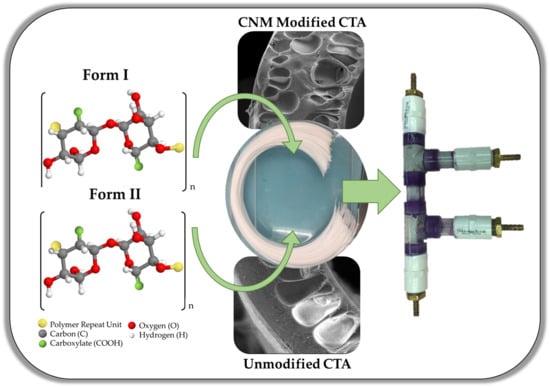
Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterial Ultrafiltration and Dialysis Mixed-Matrix Hollow Fiber Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Cellulose Solutions
2.3. Membrane Casting
2.4. Module Assembly
2.5. Dialysis and Ultrafiltration Testing
2.6. Sample Concentration Analysis
2.7. Scanning Election Microscopy
3. Results and Discussion
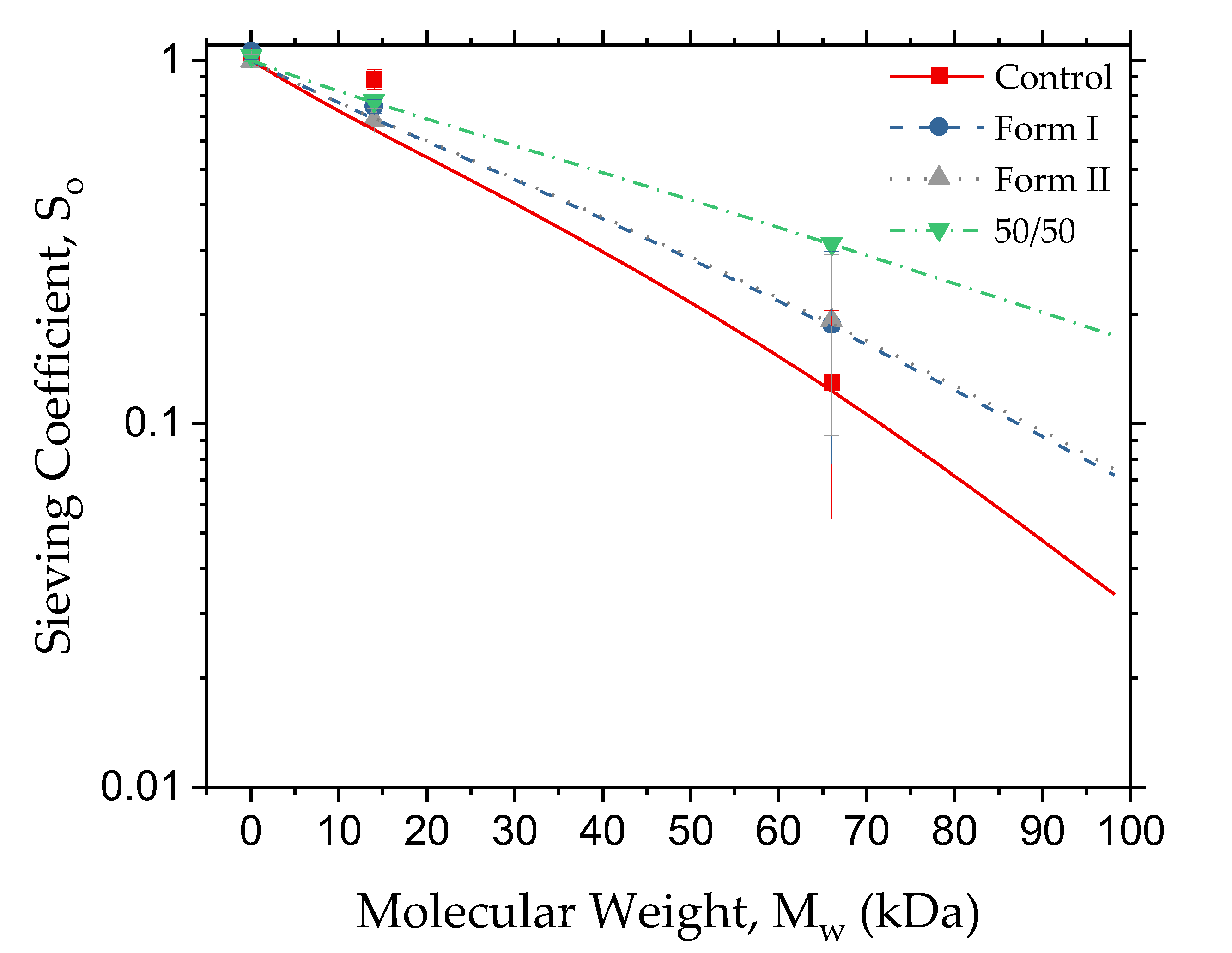
3.1. Modified Membrane Ultrafiltration Performance
3.2. Dialysis
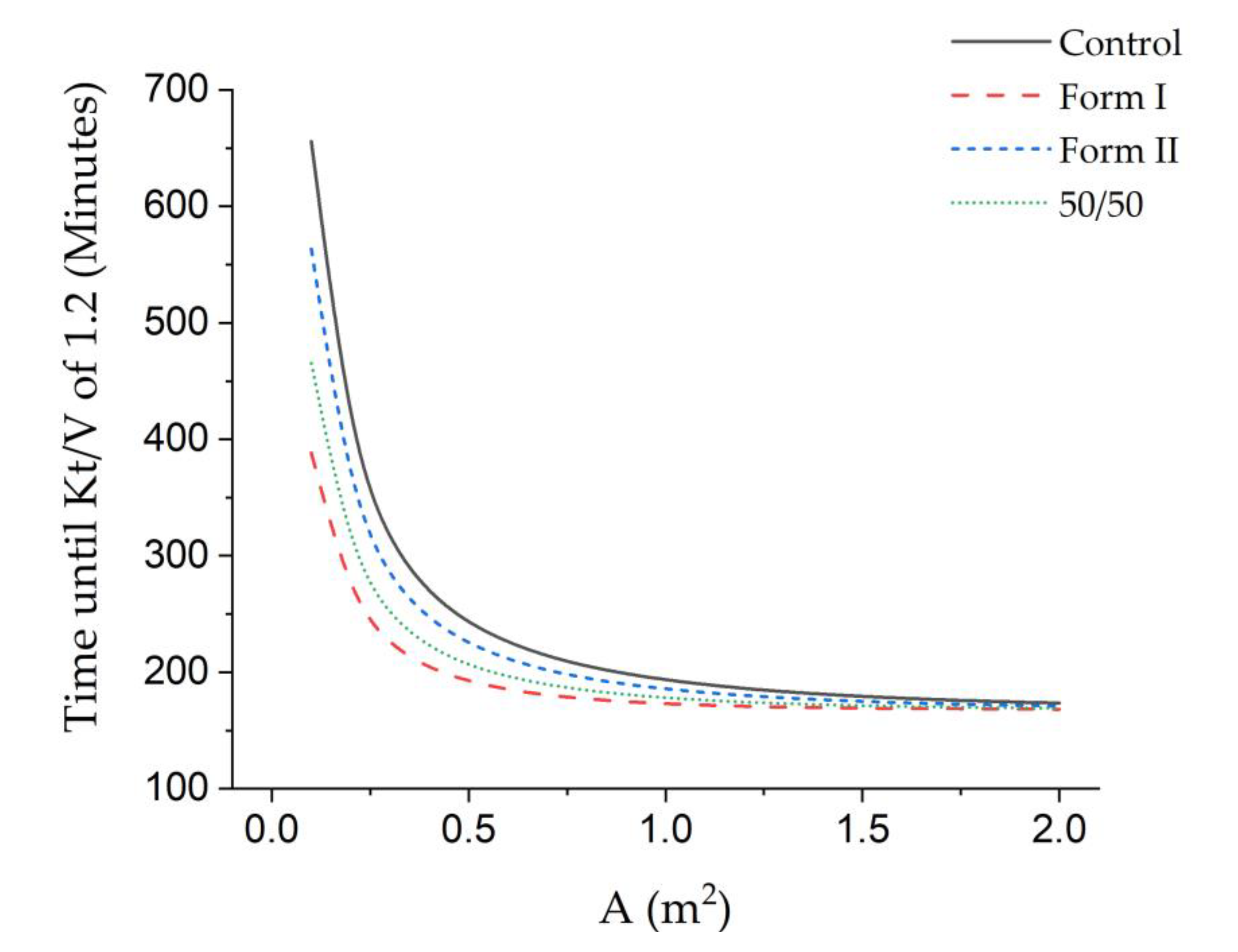
3.3. Theoretical Urea Clearance and Treatment Time
4. Conclusions
5. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
List of Variables:
| (A) membrane area, m2 |
| (cf) the concentration of the filtrate mg/mL |
| (cp) the concentration of the permeate mg/mL |
| (K) clearance, mL/min |
| (Kexperimental) experimental clearance, mL/min |
| (KoA) is the mass transfer area coefficient mL/(m2*min) |
| (KTheoretical) determined clearance values, mL/min |
| (KUF) pure water flux of the membrane at constant pressure (L/hr/m2/PSI) |
| (MW) molecular weight, Daltons |
| (N) represents the solute transport rate mg/min |
| (P) pressure, PSI |
| (Q) represents the flow rate, mL/min |
| (QUF) permeate flow rate, mL/min |
| (R) hollow fiber radius, µm |
| (Rp) pore radius, µm |
| (Rs) theoretical solute radius, µm |
| R = KoA A /Qf, dimensionless |
| (So) sieving coefficient, dimensionless |
| Subscript (f) represents the feed |
| Subscript (d) represents dialysate |
| Subscript (o) represents out |
| Subscript (i) represents in |
| (T) hollow fiber thickness, µm |
| (λ) the ratio of the solute radius to the pore radius, dimensionless |
| (Z) ratio of the blood flow rate, dimensionless |
References
- Saritas, T.; Floege, J. Cardiovascular Disease in Patients with Chronic Kidney Disease. Herz 2020, 45, 122–128. [Google Scholar] [CrossRef]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C. Chronic Kidney Disease and the Risks of Death, Cardiovascular Events, and Hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Force, U.P.S.T.; Grossman, D.C.; Bibbins-Domingo, K.; Curry, S.J.; Barry, M.J.; Davidson, K.W.; Doubeni, C.A.; Epling, J.W.; Kemper, A.R.; Krist, A.H.; et al. Behavioral Counseling to Promote a Healthful Diet and Physical Activity for Cardiovascular Disease Prevention in Adults without Cardiovascular Risk Factors: US Preventive Services Task Force Recommendation Statement. JAMA 2017, 318, 167. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, N.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; De Ferranti, S.; Després, J.-P.; Fullerton, H.J.; et al. Heart Disease and Stroke Statistics—2016 Update: A Report from the American Heart Association. Circulation 2016, 133, e38–e360. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Ma, W.; Tang, H.; Liu, D. Severe COVID-19: A Review of Recent Progress with a Look toward the Future. Front. Public Health 2020, 8, 189. [Google Scholar] [CrossRef]
- Shafi, T. Hemodialysis: Prescription and Assessment of Adequacy. Renal Urology News 2017. Available online: https://www.renalandurologynews.com/home/decision-support-in-medicine/nephrology-hypertension/hemodialysis-prescription-and-assessment-of-adequacy/ (accessed on 14 June 2020).
- Colton, C.K.; Lysaght, M.J. Membranes for Hemodialysis. In Replacement of Renal Function by Dialysis; Springer: Dordrecht, The Netherlands, 1996; pp. 103–113. [Google Scholar]
- Humes, H.D.; Fissell, W.; Tiranathanagul, K. The future of hemodialysis membranes. Kidney Int. 2006, 69, 1115–1119. [Google Scholar] [CrossRef] [Green Version]
- Kim, T.R.; Hadidi, M.; Motevalian, S.P.; Zydney, A.L.; Sunohara, T. Transport Characteristics of Asymmetric Cellulose Triacetate Hemodialysis Membranes. Blood Purif. 2017, 45, 46–52. [Google Scholar] [CrossRef]
- Zhang, Q.; Lu, X.; Liu, J.; Zhao, L. Preparation and Preliminary Dialysis Performance Research of Polyvinylidene Fluoride Hollow Fiber Membranes. Membranes 2015, 5, 120–135. [Google Scholar] [CrossRef] [Green Version]
- Hulko, M.; Haug, U.; Gauss, J.; Boschetti-De-Fierro, A.; Beck, W.; Krause, B. Requirements and Pitfalls of Dialyzer Sieving Coefficients Comparisons. Artif. Organs 2018, 42, 1164–1173. [Google Scholar] [CrossRef]
- Storr, M.; Ward, R.A. Membrane innovation: Closer to native kidneys. Nephrol. Dial. Transplant. 2018, 33, iii22–iii27. [Google Scholar] [CrossRef]
- Hill, K.; Walker, S.N.; Salminen, A.; Chung, H.L.; Li, X.; Ezzat, B.; Miller, J.J.; Desormeaux, J.S.; Zhang, J.; Hayden, A.; et al. Second Generation Nanoporous Silicon Nitride Membranes for High Toxin Clearance and Small Format Hemodialysis. Adv. Heal. Mater. 2020, 9, e1900750. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.W. Overview of Membrane Science and Technology. Membr. Technol. Appl. 2004, 1–14. [Google Scholar] [CrossRef]
- Hakim, R.M. Clinical implications of hemodialysis membrane biocompatibility. Kidney Int. 1993, 44, 484–494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duranti, E. Importance of Development of Dialysis Membranes for Purification of Small, Middle, High Molecular Weight and Protein-Bound Uremic Toxins: The Asymmetric Cellulose Triacetate (SOLACEA™-H). Dial. Trans OA 2018, 1, 180002. [Google Scholar]
- Yang, Q.; Mi, B. Nanomaterials for Membrane Fouling Control: Accomplishments and Challenges. Adv. Chronic Kidney Dis. 2013, 20, 536–555. [Google Scholar] [CrossRef] [PubMed]
- Okita, Y.; Fujisawa, S.; Saito, T.; Isogai, A. TEMPO-Oxidized Cellulose Nanofibrils Dispersed in Organic Solvents. Biomacromolecules 2011, 12, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Fukuzumi, H.; Saito, T.; Okita, Y.; Isogai, A. Thermal stabilization of TEMPO-oxidized cellulose. Polym. Degrad. Stab. 2010, 95, 1502–1508. [Google Scholar] [CrossRef]
- Dachavaram, S.S.; Moore, J.P.; Bommagani, S.; Penthala, N.R.; Calahan, J.L.; Delaney, S.P.; Munson, E.J.; Batta-Mpouma, J.; Kim, J.; Hestekin, J.A.; et al. A Facile Microwave Assisted TEMPO/NaOCl/Oxone (KHSO 5) Mediated Micron Cellulose Oxidation Procedure: Preparation of Two Nano TEMPO-Cellulose Forms. Starch Stärke 2019, 72, 1900213. [Google Scholar] [CrossRef]
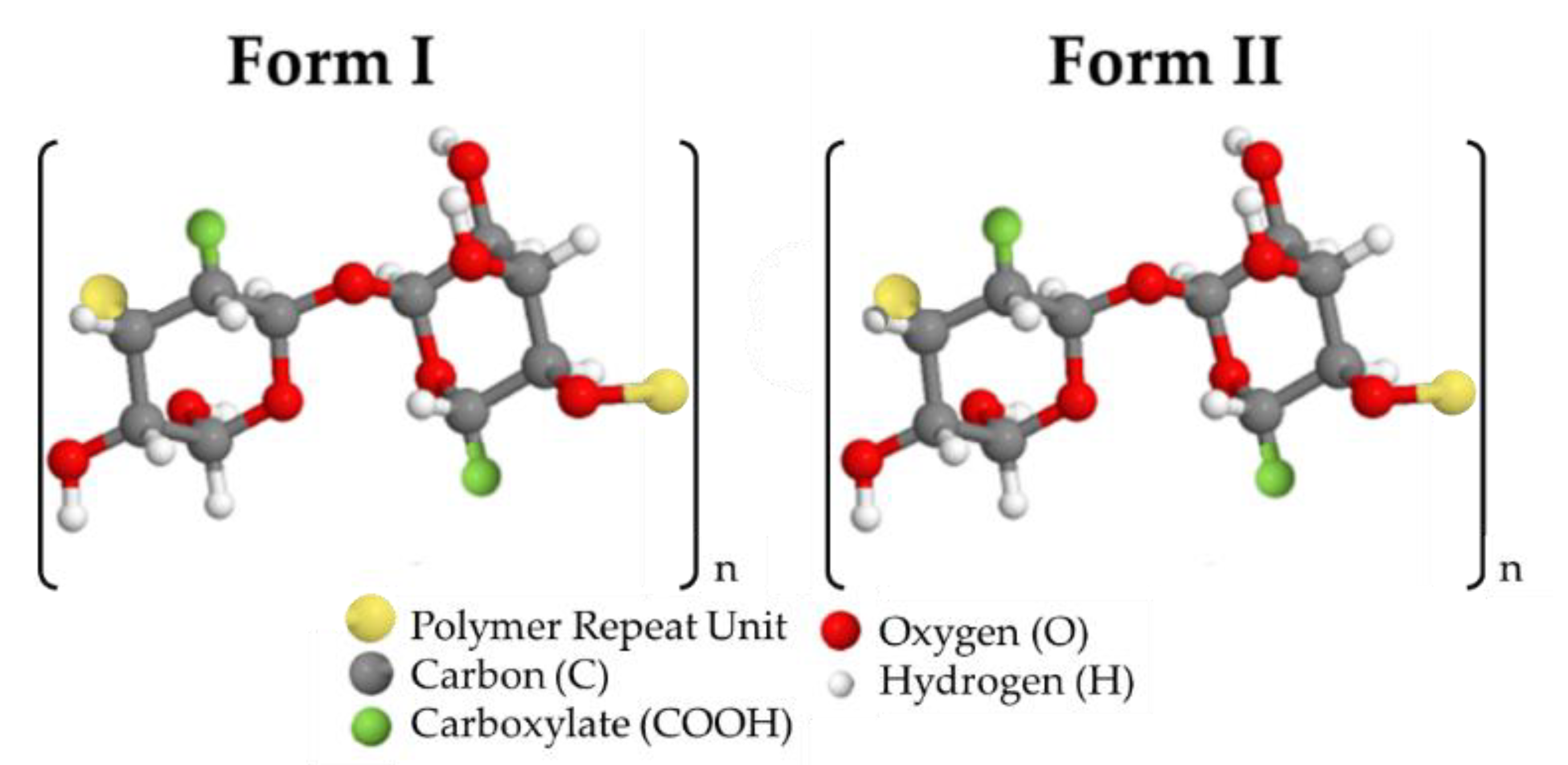
- Ii, J.M.; Dachavaram, S.; Bommagani, S.; Penthala, N.; Venkatraman, P.; Foster, E.J.; Crooks, P.A.; Hestekin, J.A. Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterials form I and form II. Molecules 2020, 25, 1847. [Google Scholar] [CrossRef] [Green Version]
- Mollahosseini, A.; Abdelrasoul, A.; Shoker, A. A Critical Review of Recent Advances in Hemodialysis Membranes Hemocompatibility and Guidelines for Future Development. Mater. Chem. Phys. 2020, 122911. [Google Scholar] [CrossRef]
- Hestekin, C.N.; Hestekin, J.A.; Paracha, S.; Morrison, G.; Pakkaner, E.; Moore, J.; De Souza, L.S.; Stephens, S.; Atchley, C.; Kurtz, I. Simulating nephron ion transport function using activated wafer electrodeionization. Commun. Mater. 2020, 1, 1–10. [Google Scholar] [CrossRef]
- Oshvandi, K.; Kavyannejad, R.; Borzuo, S.R.; Gholyaf, M. High-Flux and Low-Flux Membranes: Efficacy in Hemodialysis. Nurs. Midwifery Stud. 2014, 3, 21764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eknoyan, G.; Beck, G.J.; Cheung, A.K.; Daugirdas, J.T.; Greene, T.; Kusek, J.W.; Allon, M.; Bailey, J.; Delmez, J.A.; Depner, T.A.; et al. Effect of Dialysis Dose and Membrane Flux in Maintenance Hemodialysis. N. Engl. J. Med. 2002, 347, 2010–2019. [Google Scholar] [CrossRef] [PubMed]
- Rheosense, I. Application Note: Viscosity Measurement of a Model Protein Solution of BSA; RheoSense Inc.: San Ramon, CA, USA, 2008. [Google Scholar]
- Minton, A.P.; Edelhoch, H. Light scattering of bovine serum albumin solutions: Extension of the hard particle model to allow for electrostatic repulsion. Biopolymers 1982, 21, 451–458. [Google Scholar] [CrossRef]
- Huang, Y.-S.; Jeng, U.-S.; Shiu, Y.-J.; Lai, Y.-H.; Sun, Y.-S. Charge interaction and temperature effects on the solution structure of lysozyme as revealed by small-angle X-ray scattering. J. Appl. Crystallogr. 2007, 40, s165–s169. [Google Scholar] [CrossRef] [Green Version]
- Ronco, C.; Brendolan, A.; Everard, P.; Irone, M.; Ballestri, M.; Cappelli, G.; Inguaggiato, P.; Bellomo, R. Cellulose triacetate: Another membrane for continuous renal replacement therapy. J. Nephrol. 1999, 12, 241–247. [Google Scholar]
- Foley, G. Membrane Filtration: A Problem Solving Approach with MATLAB; Cambridge University Press: Cambridge, England, 2013. [Google Scholar]
- Anderson, R.B.; Bayer, J.; Hofer, L. Linear Solutions of Fick’s Law. Ind. Eng. Chem. Process. Des. Dev. 1965, 4, 167–171. [Google Scholar] [CrossRef]
- Dahe, G.J.; Singh, R.P.; Dudeck, K.W.; Yang, D.; Berchtold, K.A. Influence of non-solvent chemistry on polybenzimidazole hollow fiber membrane preparation. J. Membr. Sci. 2019, 577, 91–103. [Google Scholar] [CrossRef]
- Bai, L.; Wu, H.; Ding, J.; Ding, A.; Zhang, X.; Ren, N.; Li, G.; Liang, H. Cellulose nanocrystal-blended polyethersulfone membranes for enhanced removal of natural organic matter and alleviation of membrane fouling. Chem. Eng. J. 2020, 382, 122919. [Google Scholar] [CrossRef]
- Bai, H.; Wang, X.; Zhou, Y.; Zhang, L. Preparation and characterization of poly (vinylidene fluoride) composite membranes blended with nano-crystalline cellulose. Prog. Nat. Sci. 2012, 22, 250–257. [Google Scholar] [CrossRef] [Green Version]
- Setiawan, L.; Wang, R.; Shi, L.; Li, K.; Fane, A. Novel dual-layer hollow fiber membranes applied for forward osmosis process. J. Membr. Sci. 2012, 421, 238–246. [Google Scholar] [CrossRef]
- Ayyaru, S.; Dinh, T.T.L.; Ahn, Y.-H.; Linh, D.T.T. Enhanced antifouling performance of PVDF ultrafiltration membrane by blending zinc oxide with support of graphene oxide nanoparticle. Chemosphere 2019, 241, 125068. [Google Scholar] [CrossRef]
- Manickam, S.S.; McCutcheon, J.R. Understanding mass transfer through asymmetric membranes during forward osmosis: A historical perspective and critical review on measuring structural parameter with semi-empirical models and characterization approaches. Desalination 2017, 421, 110–126. [Google Scholar] [CrossRef]
- Bonert, M.; Saville, B.A. A Non-Dimensional Analysis of Hemodialysis. Open Biomed. Eng. J. 2010, 4, 138–155. [Google Scholar] [CrossRef] [PubMed]
- Sanaei, P. Mathematical Modeling of Membrane Filtration; Digital Commons, New Jersey Institute of Technology: Newark, NJ, USA, 2017. [Google Scholar]
- Cabasso, I.; Robert, K.Q.; Klein, E.; Smith, J.K. Porosity and pore size determination in polysulfone hollow fibers. J. Appl. Polym. Sci. 1977, 21, 1883–1900. [Google Scholar] [CrossRef]
- Himmelfarb, J.; Ikizler, T.A. Hemodialysis. N. Engl. J. Med. 2010, 363, 1833–1845. [Google Scholar] [CrossRef]
- Mandolfo, S.; Malberti, F.; Imbasciati, E.; Cogliati, P.; Gauly, A. Impact of Blood and Dialysate Flow and Surface on Performance of New Polysulfone Hemodialysis Dialyzers. Int. J. Artif. Organs 2003, 26, 113–120. [Google Scholar] [CrossRef]
- Khandpur, R.S. Handbook of Biomedical Instrumentation; McGraw-Hill Education: New York, NY, USA, 1987. [Google Scholar]
- Azar, A.T.; Canaud, B. Hemodialysis System. In Modelling and Control of Dialysis Systems; Springer: Berlin/Heidelberg, Germany, 2013; pp. 99–166. [Google Scholar]

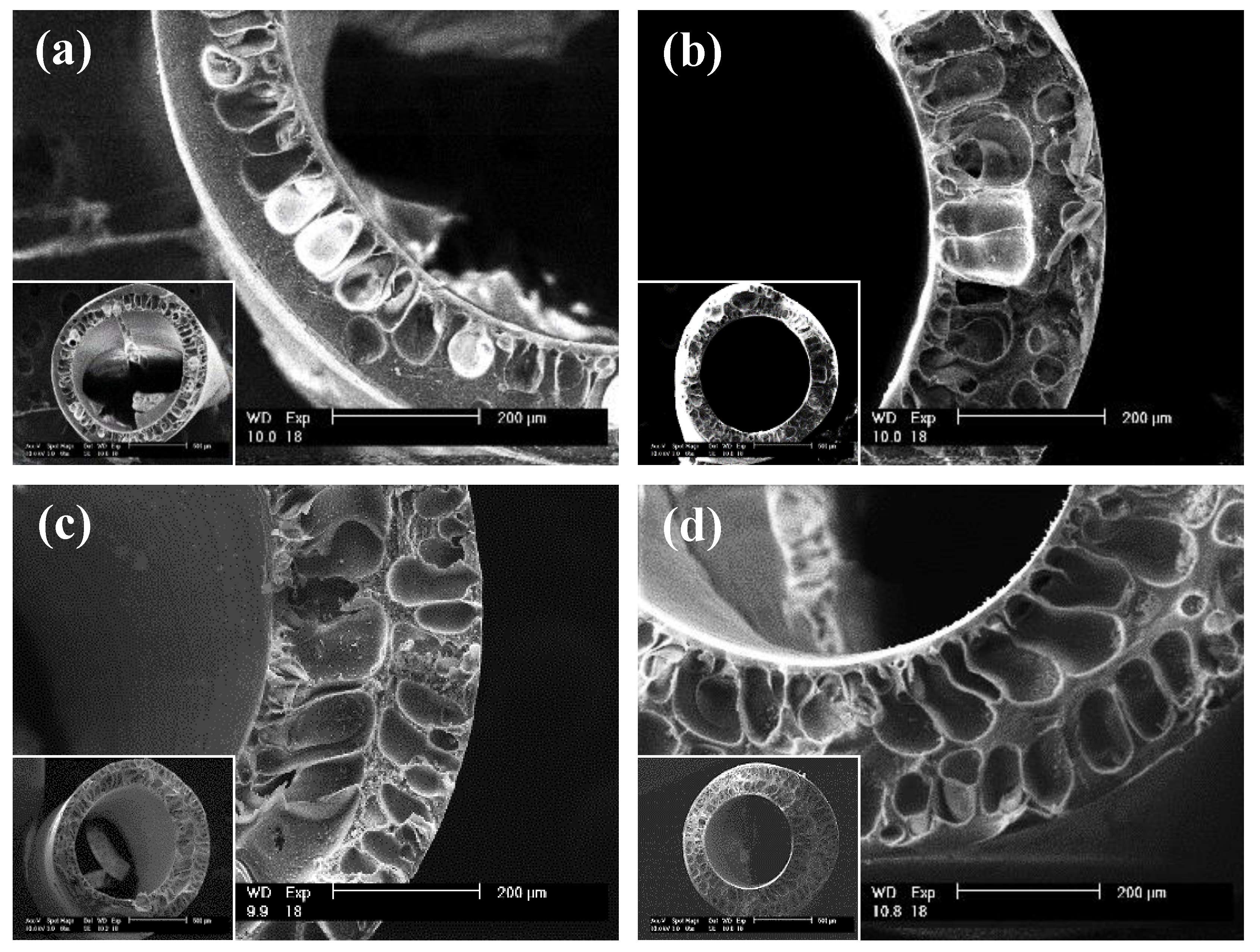
| Membrane Material | A (m2) | R (µm) | T (µm) | KUF Pure Water (L/hr/m2/PSI) | Rp (nm) |
|---|---|---|---|---|---|
| Control | 0.0115 | 568 | 192 | 109 ± 18 | 8.9 ± 1.2 |
| Form I | 0.0112 | 578 | 211 | 144 ± 39 | 9.8 ± 1.6 |
| Form II | 0.0118 | 554 | 229 | 111 ± 19 | 9.9 ± 1.4 |
| 50/50 | 0.0105 | 513 | 226 | 121 ± 18 | 11.6 ± 0.1 |
| Urea | Lysozyme | BSA | |||||
|---|---|---|---|---|---|---|---|
| Sample | KoA | Kexperimental (mL/min) | KoA | Kexperimental (mL/min) | KoA | Kexperimental (mL/min) | SC |
| Control | 968 | 9.4 | 115 | 1.3 | <1 | <1 | 0.0080 |
| Form I | 1996 | 20.8 | 261 | 2.9 | 225 | 2.5 | 0.0091 |
| Form II | 1177 | 14.5 | 201 | 2.4 | 499 | 5.8 | 0.0111 |
| 50/50 | 1528 | 16.2 | 388 | 4.0 | 478 | 5.0 | 0.0125 |
| Sample | KTheoretical (mL/min) | KTheoretical (mL/min/m2) | Theoretical Treatment Time (hours) |
|---|---|---|---|
| Control | 9.2 | 211 | 3.98 |
| Form I | 20.6 | 284 | 2.95 |
| Form II | 12.2 | 254 | 3.31 |
| 50/50 | 15.9 | 257 | 3.26 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, J.P., II; Robling, K.; Romero, C.; Kiper, K.; Dachavaram, S.S.; Crooks, P.A.; Hestekin, J.A. Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterial Ultrafiltration and Dialysis Mixed-Matrix Hollow Fiber Membranes. Polymers 2020, 12, 1348. https://doi.org/10.3390/polym12061348
Moore JP II, Robling K, Romero C, Kiper K, Dachavaram SS, Crooks PA, Hestekin JA. Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterial Ultrafiltration and Dialysis Mixed-Matrix Hollow Fiber Membranes. Polymers. 2020; 12(6):1348. https://doi.org/10.3390/polym12061348
Chicago/Turabian StyleMoore, John P., II, Kristyn Robling, Cristian Romero, Keturah Kiper, Soma Shekar Dachavaram, Peter A. Crooks, and Jamie A. Hestekin. 2020. "Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterial Ultrafiltration and Dialysis Mixed-Matrix Hollow Fiber Membranes" Polymers 12, no. 6: 1348. https://doi.org/10.3390/polym12061348
APA StyleMoore, J. P., II, Robling, K., Romero, C., Kiper, K., Dachavaram, S. S., Crooks, P. A., & Hestekin, J. A. (2020). Oxone®-Mediated TEMPO-Oxidized Cellulose Nanomaterial Ultrafiltration and Dialysis Mixed-Matrix Hollow Fiber Membranes. Polymers, 12(6), 1348. https://doi.org/10.3390/polym12061348