Effect of Different Phosphate Glass Compositions on the Process-Induced Macromolecular Dynamics of Polyamide 66
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Melt Processing and Characterization Methods
2.2.1. Internal Batch Mixing
2.2.2. Differential Scanning Calorimetry (DSC)
2.2.3. Dynamic Mechanical Analysis (DMA)
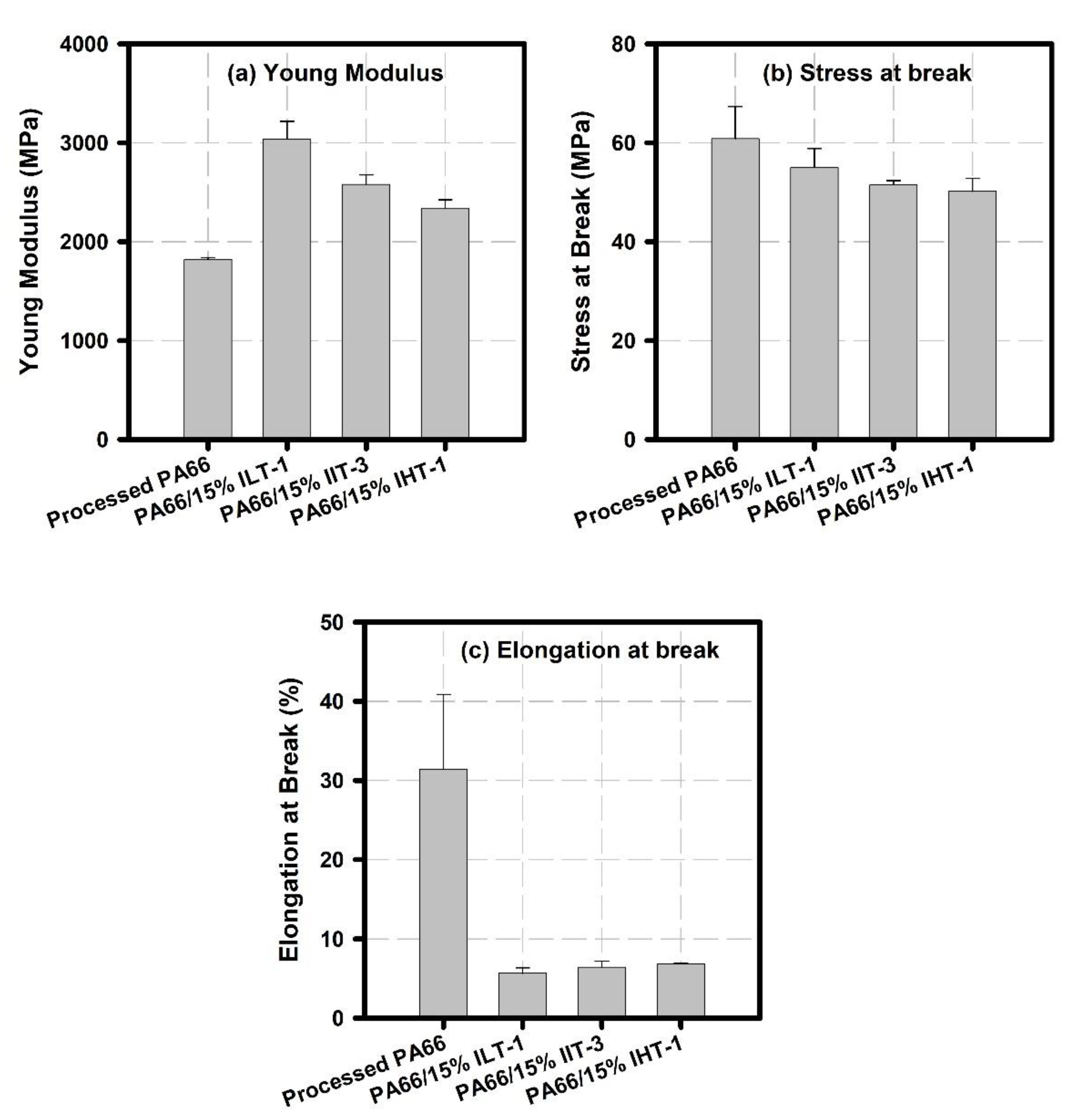
2.2.4. Tensile Strength
2.2.5. Compressing Molding
2.2.6. Broadband Dielectric Spectroscopy (BDS) Measurements
3. Results and Discussion
3.1. Differential Scanning Calorimetry (DSC) Analysis
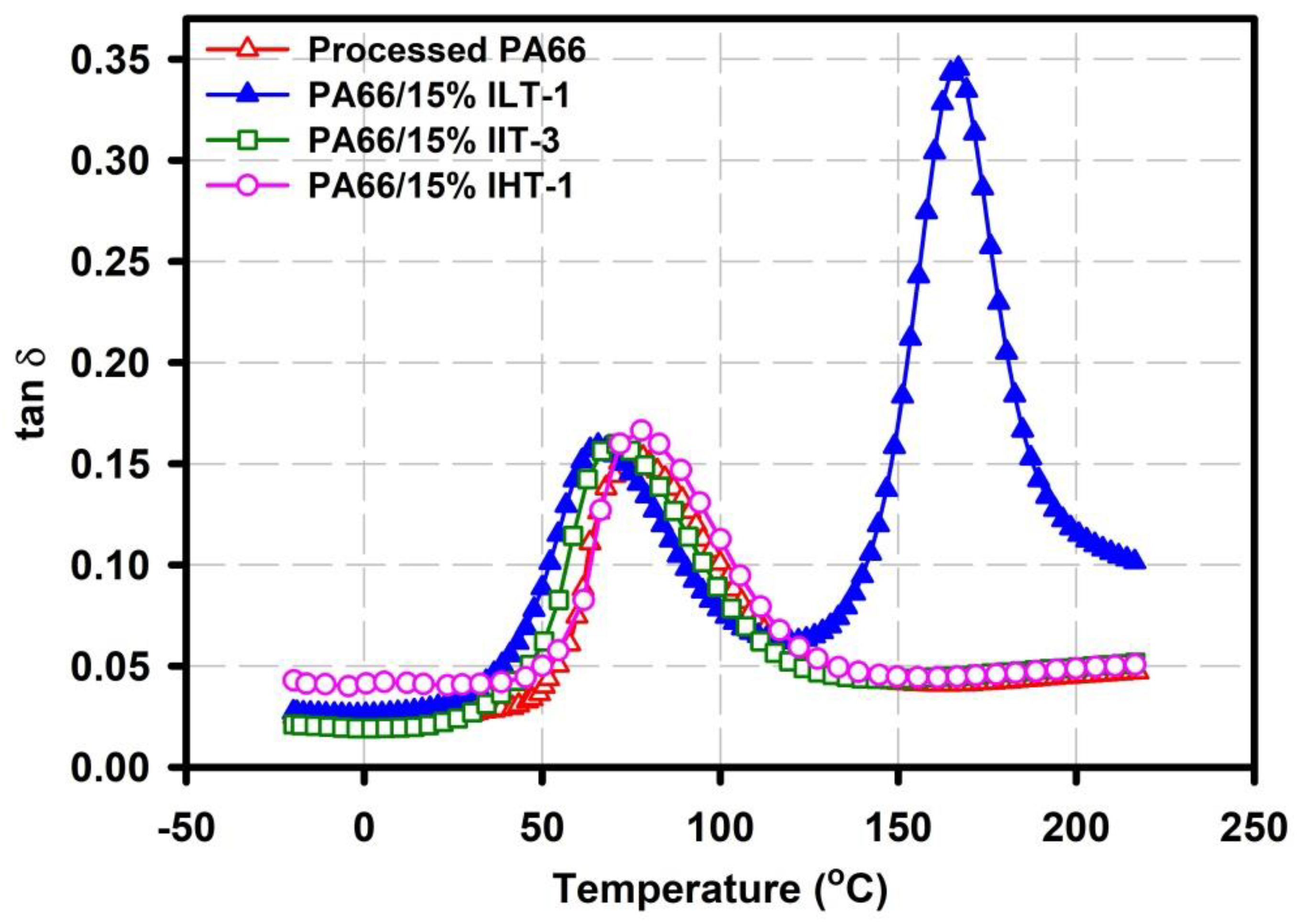
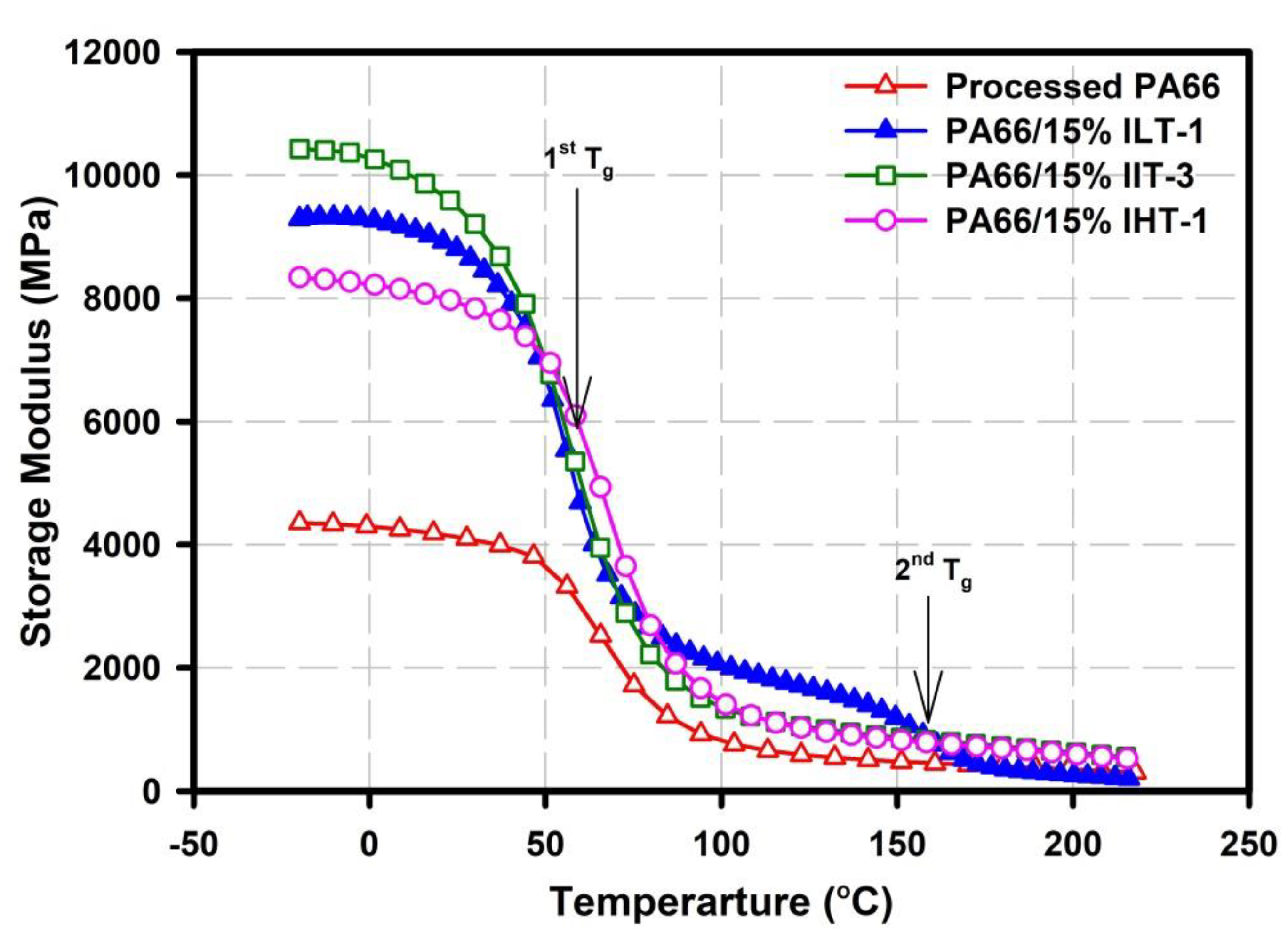
3.2. Dynamic Mechanical Analysis (DMA)
3.3. Dielectric Spectroscopy Analysis
3.3.1. γ relaxation
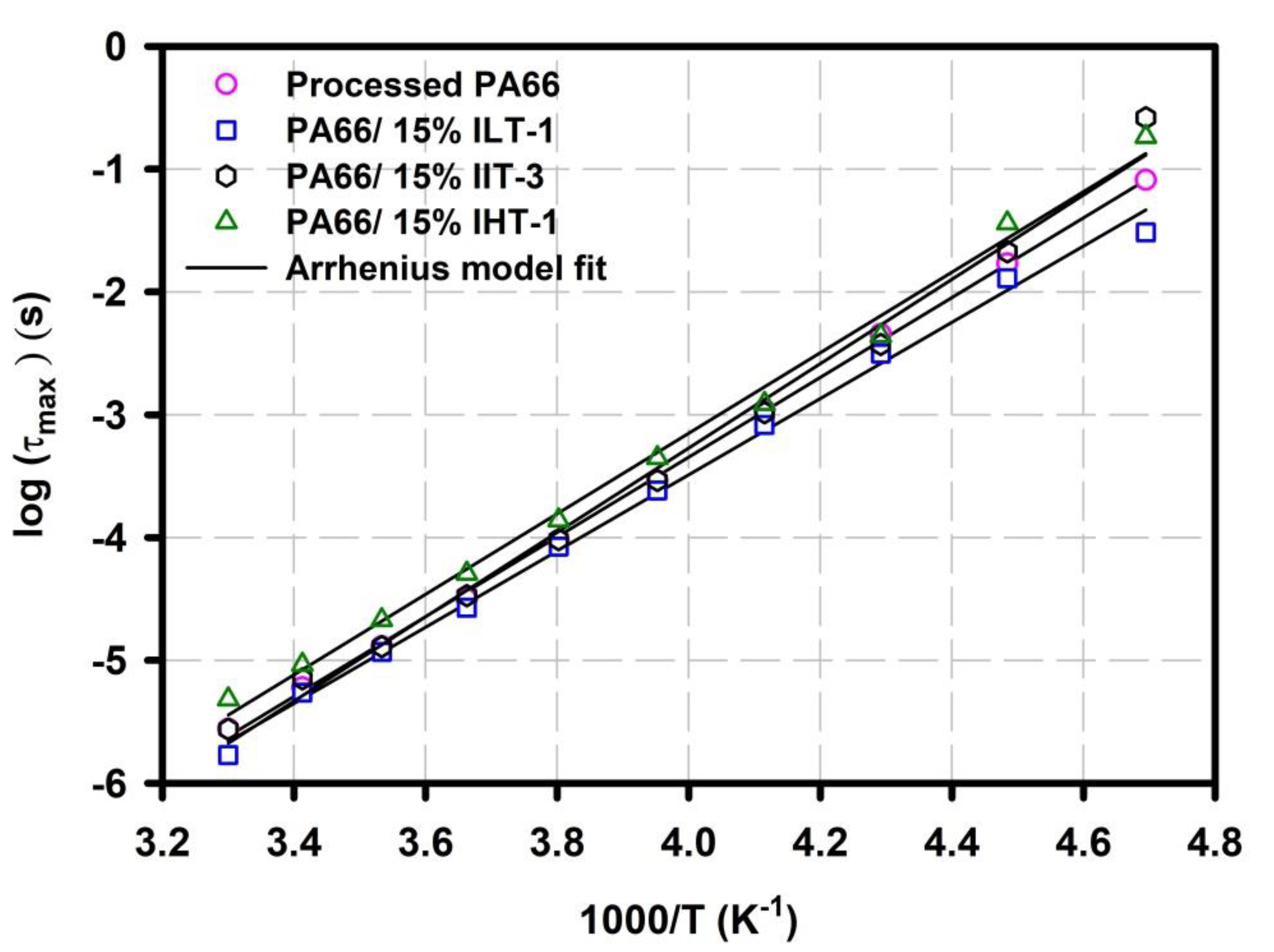
3.3.2. α Relaxation: Glass Transition Chain Dynamics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fulchiron, R.; Belyamani, I.; Otaigbe, J.U.; Bounor-Legaré, V. A simple method for tuning the glass transition process in inorganic phosphate glasses. Sci. Rep. 2015, 5, 8369. [Google Scholar] [CrossRef] [PubMed]
- Loong, C.-K.; Suzuya, K.; Price, D.; Sales, B.C.; Boatner, L.A. Structure and dynamics of phosphate glasses: From ultra- to orthophosphate composition. Phys. B Condens. Matter 1997, 241, 890–896. [Google Scholar] [CrossRef]
- Belyamani, I.; Kim, K.; Rahimi, S.K.; Sahukhal, G.S.; Elasri, M.O.; Otaigbe, J.U. Creep, recovery, and stress relaxation behavior of nanostructured bioactive calcium phosphate glass-POSS/polymer composites for bone implants studied under simulated physiological conditions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 2419–2432. [Google Scholar] [CrossRef]
- Lapa, A.; Cresswell, M.; Jackson, P.; Boccaccini, A. Phosphate glass fibres with therapeutic ions release capability—A review. Adv. Appl. Ceram. 2019, 119, 1–14. [Google Scholar] [CrossRef]
- Belyamani, I.; Otaigbe, J.U.; Fielding, W.R. Development of new sustainable inorganic flame retardant additive system for polyamide 6,6 with improved performance. Polym. Eng. Sci. 2014, 55, 1741–1748. [Google Scholar] [CrossRef]
- Urman, K.; Otaigbe, J.U. New phosphate glass/polymer hybrids—Current status and future prospects. Prog. Polym. Sci. 2007, 32, 1462–1498. [Google Scholar] [CrossRef]
- Urman, K.; Otaigbe, J.U. Novel phosphate glass/polyamide 6 hybrids: Miscibility, crystallization kinetics, and mechanical properties. J. Polym. Sci. Part B Polym. Phys. 2005, 44, 441–450. [Google Scholar] [CrossRef]
- Serio, L.; Gawne, D.T.; Bao, Y. Effect of tin fluoride content on the structure and properties of phosphate glass—Polyamide 11 hybrids. Eur. Polym. J. 2018, 99, 134–141. [Google Scholar] [CrossRef]
- Rawal, A.; Urman, K.; Otaigbe, J.U.; Schmidt-Rohr, K. Detection of Nanometer-Scale Mixing in Phosphate-Glass/Polyamide-6 Hybrids by1H−31P NMR. Chem. Mater. 2006, 18, 6333–6338. [Google Scholar] [CrossRef]
- Rawal, A.; Fang, X.-W.; Urman, K.; Iverson, D.; Otaigbe, J.U.; Schmidt-Rohr, K. Promotion of the γ-phase of polyamide 6 in its nanocomposite with phosphate glass. J. Polym. Sci. Part B Polym. Phys. 2008, 46, 857–860. [Google Scholar] [CrossRef]
- Rawal, A.; Kong, X.; Meng, Y.; Otaigbe, J.U.; Schmidt-Rohr, K. Reduced Crystallinity and Mobility of Nylon-6 Confined near the Organic–Inorganic Interface in a Phosphate Glass-Rich Nanocomposite Detected by1H–13C NMR. Macromology 2011, 44, 8100–8105. [Google Scholar] [CrossRef]
- Meng, Y.; Otaigbe, J.U. Mechanism of Unexpected Viscosity Decrease of Nylon 6 Melts by Low-Tg Inorganic Tin Fluorophosphate Glass during Processing. Appl. Rheol. 2011, 21. [Google Scholar] [CrossRef]
- Urman, K.; Madbouly, S.; Otaigbe, J.U. Unusual accelerated molecular relaxations of a tin fluorophosphate glass/polyamide 6 hybrid studied by broadband dielectric spectroscopy. Polymer 2007, 48, 1659–1666. [Google Scholar] [CrossRef]
- Kremer, F.; Schönhals, A. Broadband Dielectric Spectroscopy; Springer: Berlin, Germany, 2003. [Google Scholar]
- Ahmed, H.M.; Windham, A.D.; Al-Ejji, M.M.; Al-Qahtani, N.; Hassan, M.K.; Mauritz, K.A.; Buchanan, R.K.; Buchanan, J.P. Preparation and Preliminary Dielectric Characterization of Structured C60-Thiol-Ene Polymer Nanocomposites Assembled Using the Thiol-Ene Click Reaction. Materials 2015, 8, 7795–7804. [Google Scholar] [CrossRef] [PubMed]
- Levchik, S.; Costa, L.; Camino, G. Effect of the fire-retardant ammonium polyphosphate on the thermal decomposition of aliphatic polyamides. Part III—Polyamides 6.6 and 6.10. Polym. Degrad. Stab. 1994, 43, 43–54. [Google Scholar] [CrossRef]
- Jahromi, S.; Gabriëlse, W.; Braam, A. Effect of melamine polyphosphate on thermal degradation of polyamides: A combined X-ray diffraction and solid-state NMR study. Polymer 2003, 44, 25–37. [Google Scholar] [CrossRef]
- Wang, C.; Wu, Y.; Li, Y.; Shao, Q.; Yan, X.; Han, C.; Wang, Z.; Liu, Z.; Guo, Z. Flame-retardant rigid polyurethane foam with a phosphorus-nitrogen single intumescent flame retardant. Polym. Adv. Technol. 2017, 29, 668–676. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Zhang, J.; Xiao, R. Preparation and characterizations of inherent flame retarded polyamide 66 containing the phosphorus linking pendent group. Polym. Adv. Technol. 2017, 29, 951–960. [Google Scholar] [CrossRef]
- Qiu, S.; Ma, C.; Wang, X.; Zhou, X.; Feng, X.; Yuen, K.K.R.; Hu, Y. Melamine-containing polyphosphazene wrapped ammonium polyphosphate: A novel multifunctional organic-inorganic hybrid flame retardant. J. Hazard. Mater. 2018, 344, 839–848. [Google Scholar] [CrossRef]
- Tang, W.; Cao, Y.; Qian, L.; Chen, Y.; Qiu, Y.; Xu, B.; Xin, F. Synergistic Charring Flame-Retardant Behavior of Polyimide and Melamine Polyphosphate in Glass Fiber-Reinforced Polyamide 66. Polymers 2019, 11, 1851. [Google Scholar] [CrossRef]
- Haberkorn, H.; Illers, K.; Simak, P. Calorimetric, X-ray and infra-red investigations on poly(hexamethylene adipamide). Polym. Bull. 1979, 1, 485–490. [Google Scholar] [CrossRef]
- Rong, M.; Zeng, H. Polycarbonate-epoxy semi-interpenetrating polymer network: 2. Phase separation and morphology. Polymer 1997, 38, 269–277. [Google Scholar] [CrossRef]
- Madbouly, S.A.; Ougizawa, T. Isothermal Crystallization of Poly(ε-caprolactone) in Blend with Poly(styrene-co-acrylonitrile): Influence of Phase Separation Process. Macromol. Chem. Phys. 2004, 205, 1923–1931. [Google Scholar] [CrossRef]
- Xing, P.; Dong, L.; An, Y.; Feng, Z.; Avella, M.; Martuscelli, E. Miscibility and Crystallization of Poly(β-hydroxybutyrate) and Poly(p-vinylphenol) Blends. Macromolecules 1997, 30, 2726–2733. [Google Scholar] [CrossRef]
- Madbouly, S.A.; Mansour, A.A.; Abdou, N.Y. Molecular dynamics of amorphous/crystalline polymer blends studied by broadband dielectric spectroscopy. Eur. Polym. J. 2007, 43, 1892–1904. [Google Scholar] [CrossRef]
- Mauritz, K.A. Dielectric relaxation studies of ion motions in electrolyte-containing perfluorosulfonate ionomers. 4. Long-range ion transport. Macromology 1989, 22, 4483–4488. [Google Scholar] [CrossRef]
- Vogel, H. The law of the relation between the viscosity of liquids and the temperature. Phys. Z. 1921, 22, 645–646. [Google Scholar]
- Fulcher, G.S. Analysis of recent measurements of the viscosity of glasses. J. Am. Ceram. Soc. 1925, 8, 339–355. [Google Scholar] [CrossRef]
- Tammann, G.; Hesse, W. Die Abhängigkeit der Viscosität von der Temperatur bie unterkühlten Flüssigkeiten. Zeitschrift Für Anorganische Und Allgemeine Chemie 1926, 156, 245–257. [Google Scholar] [CrossRef]
- Madbouly, S.A.; Otaigbe, J.; Hassan, M.K.; Mauritz, K. Broadband Dielectric Spectroscopy of mPP/PC Blend Prepared Via In Situ Polymerization and Compatibilization. Abstr. Pap. Am. Chem. Soc. 2006. [Google Scholar]
- Pratt, G.J.; Smith, M.J.A. A Dielectric Investigation of Miscibility and Morphology in Engineering Thermoplastics. Polym. Int. 1997, 43, 137–142. [Google Scholar] [CrossRef]
- Rhoades, D.W.; Hassan, M.K.; Osborn, S.J.; Moore, R.B.; Mauritz, K.A. Broadband dielectric spectroscopic characterization of Nafion® chemical degradation. J. Power Sources 2007, 172, 72–77. [Google Scholar] [CrossRef]
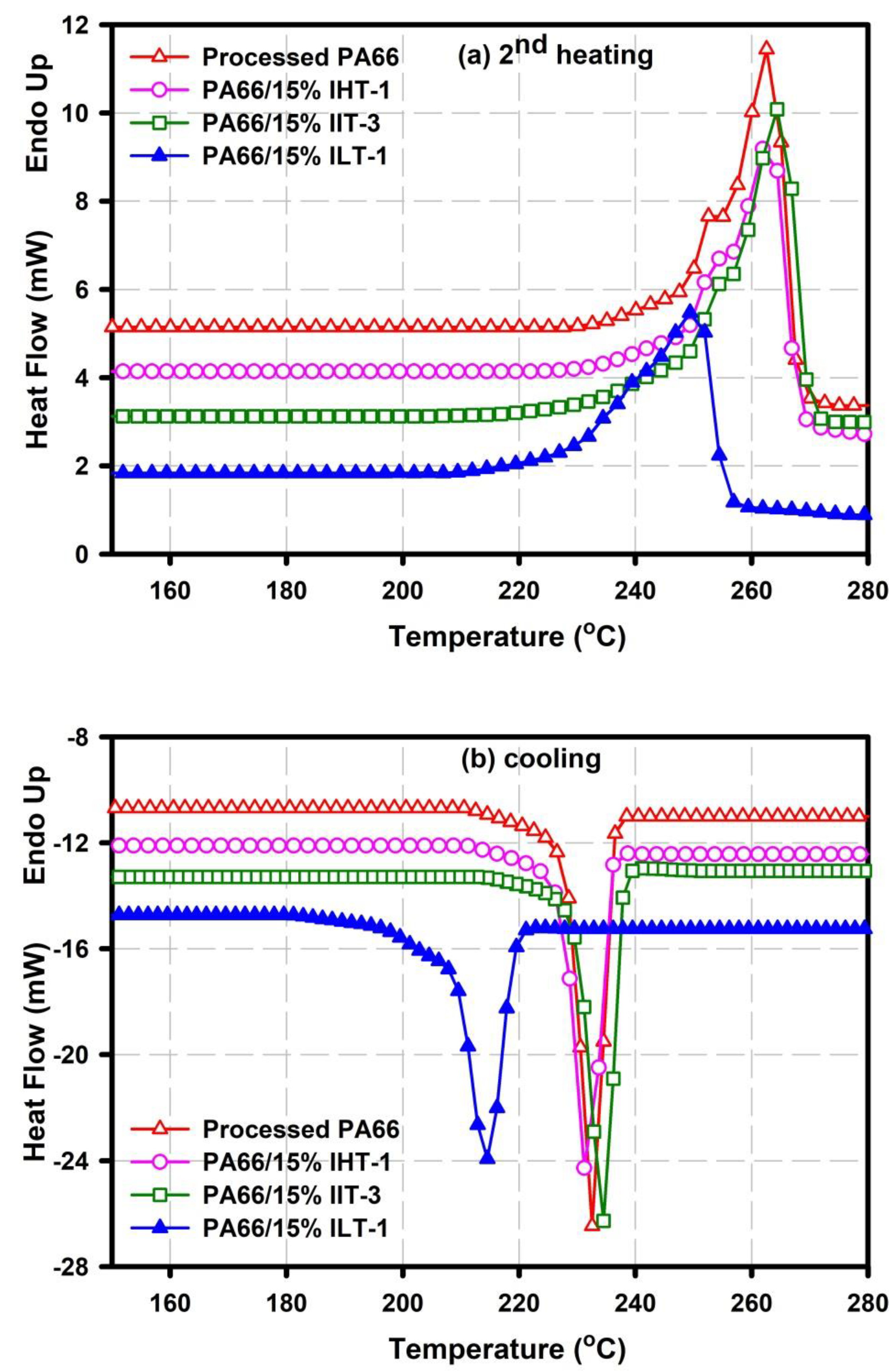
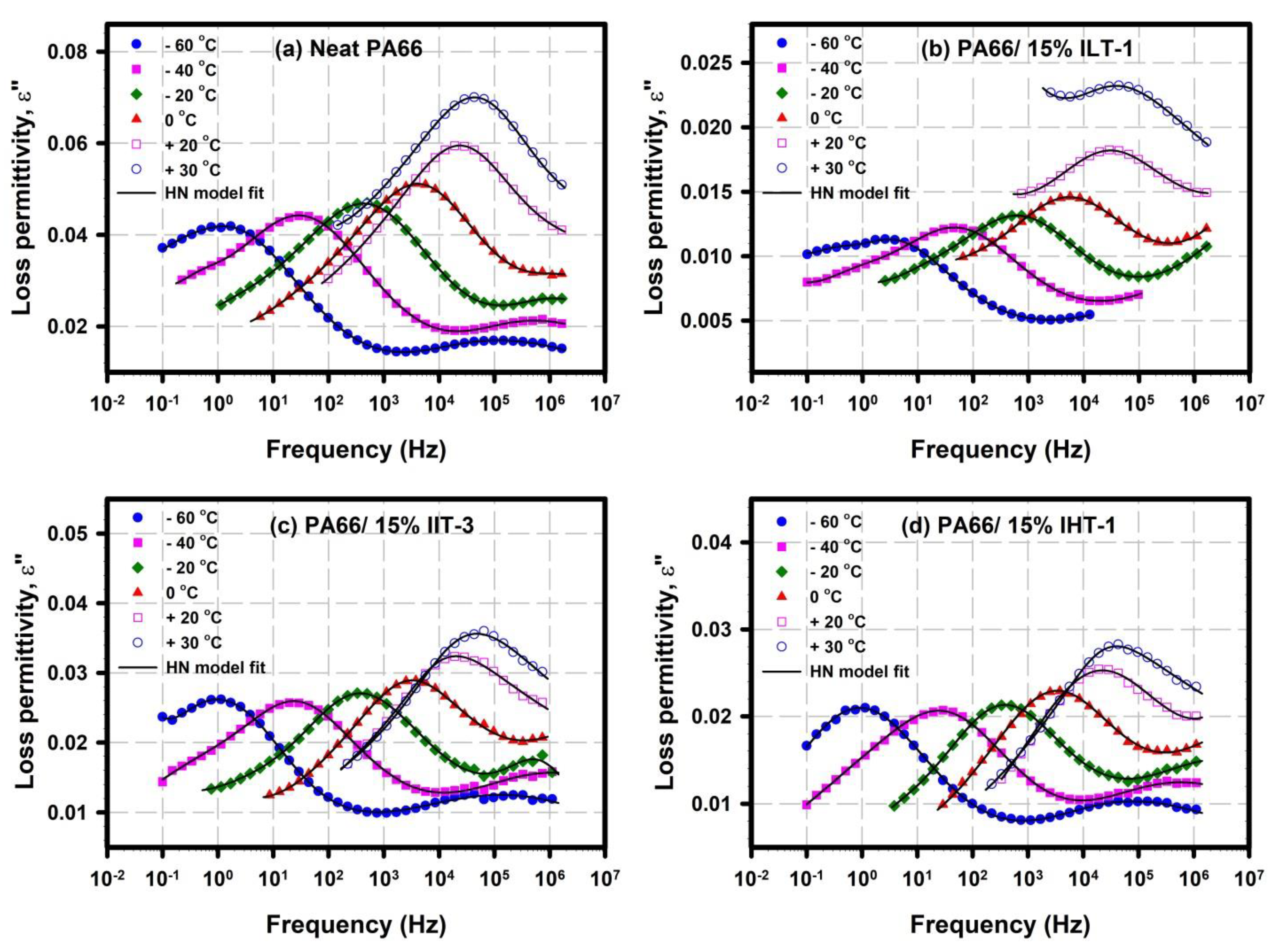
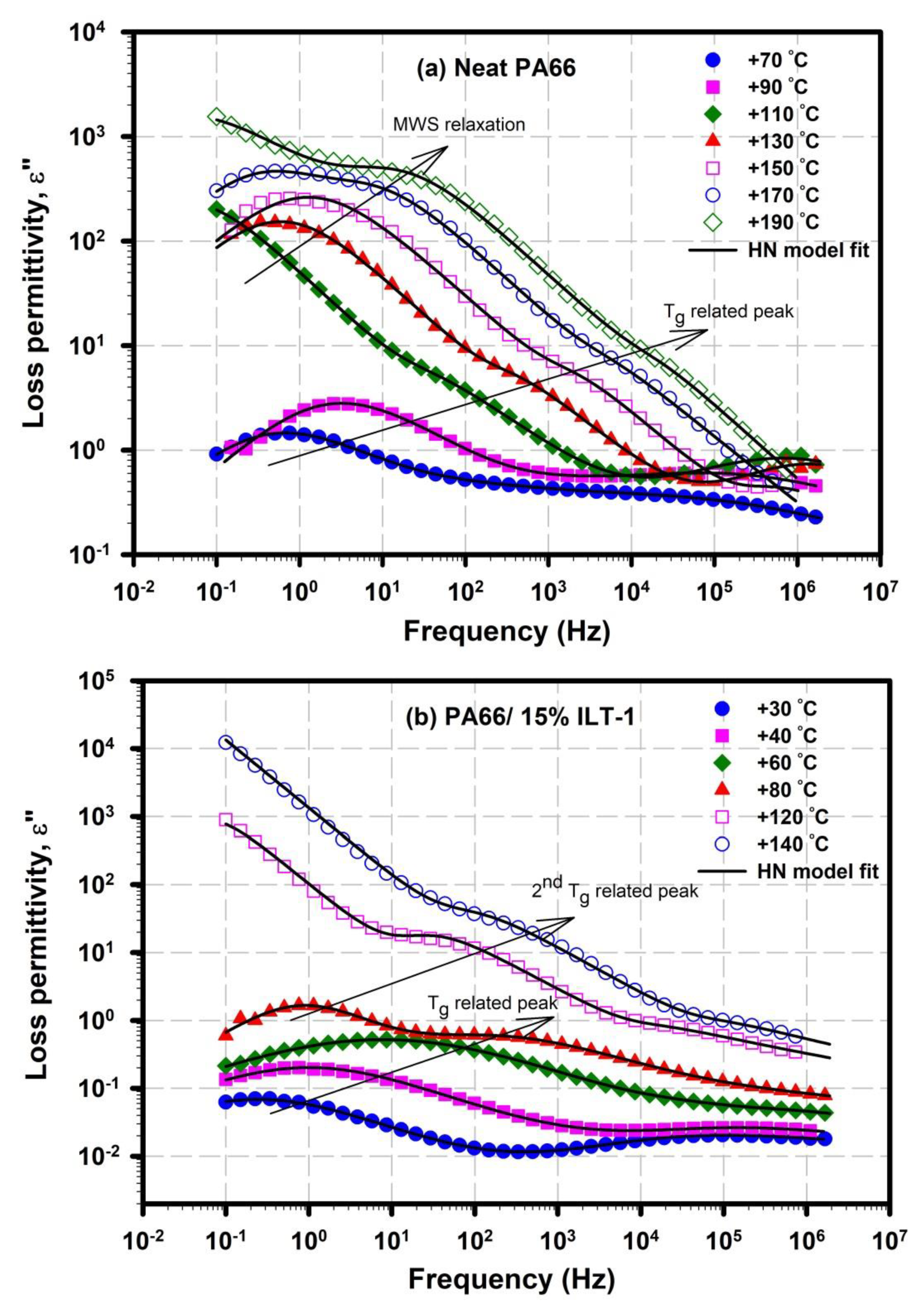
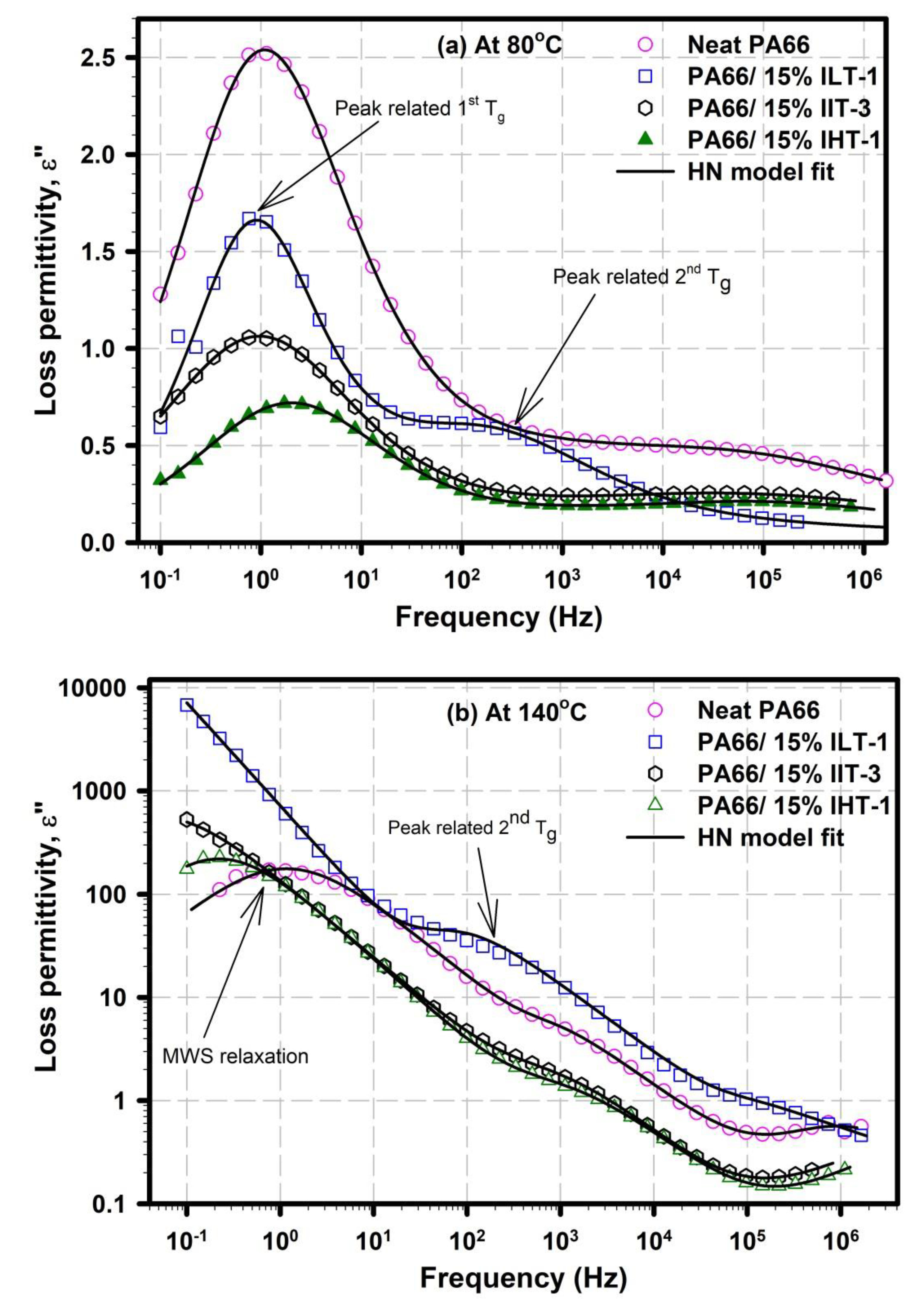
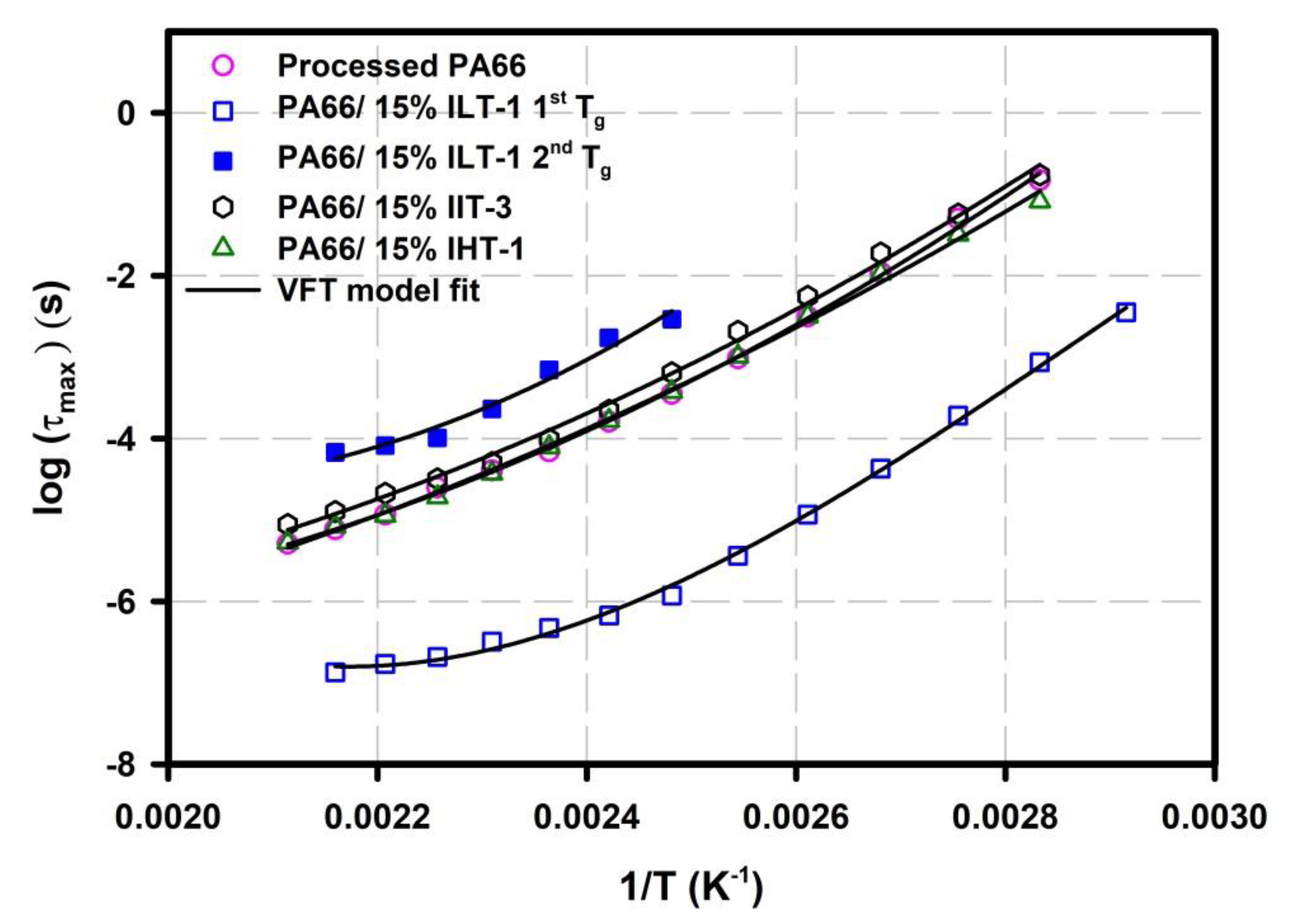
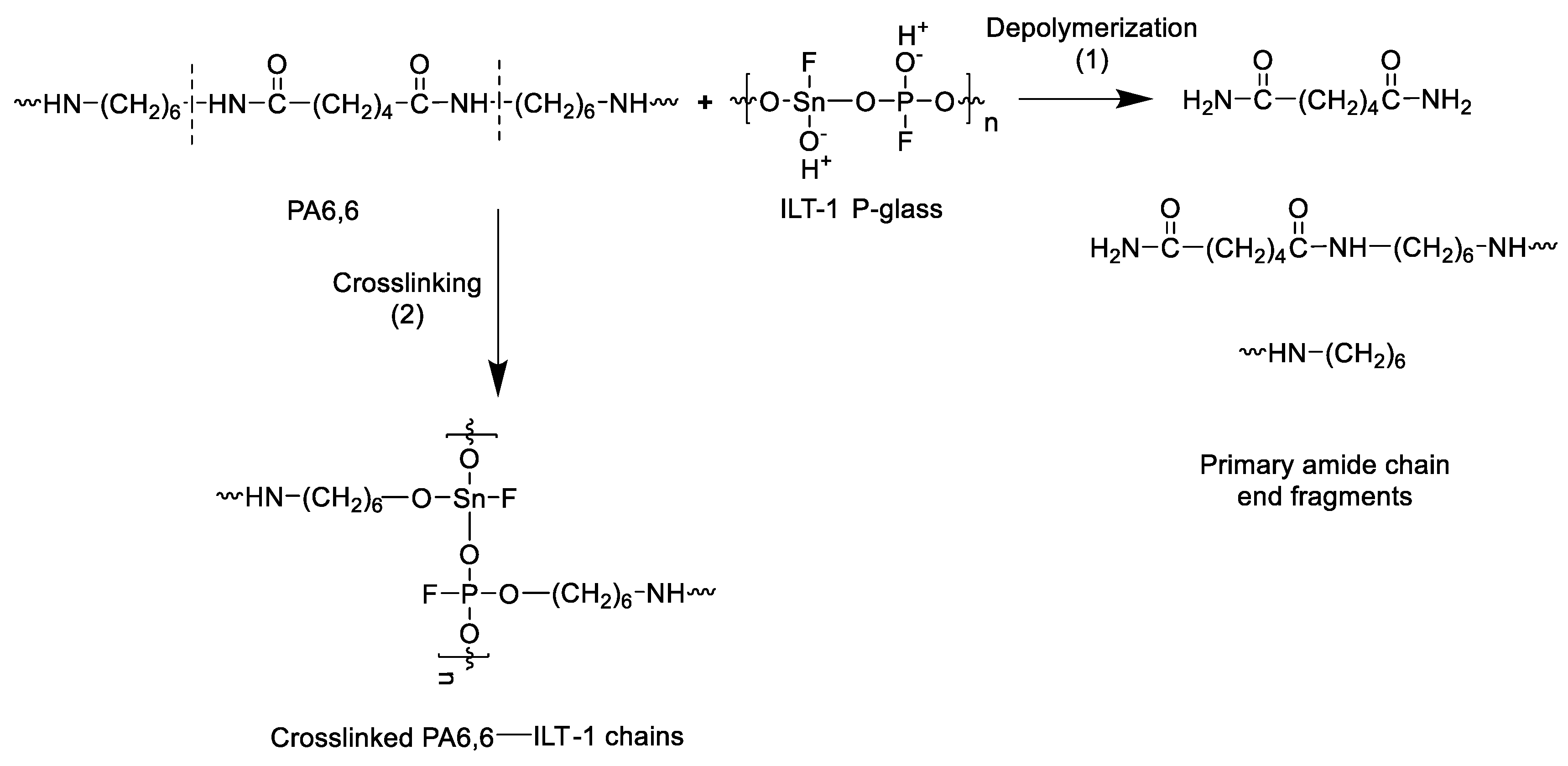
| Sample | Tmonset (°C) | Tm peak (°C) | Tconset (°C) | Tc peak (°C) | Xc (%) |
|---|---|---|---|---|---|
| Processed neat PA66 | 230 | 263 | 242 | 232 | 34.10 |
| PA66/15% ILT-1 | 214 | 250 | 221 | 215 | 15.05 |
| PA66/15% IIT-3 | 227 | 265 | 240 | 234 | 35.80 |
| PA66/15%IHT-1 | 230 | 262 | 242 | 230 | 33.50 |
| Sample | Activation Energy (kJ/mol) | Fit R2 |
|---|---|---|
| Processed neat PA66 | 62.1 | 0.99964 |
| PA66/15% ILT-1 | 59.4 | 0.99818 |
| PA66/15% IIT-3 | 65.7 | 0.99612 |
| PA66/15%IHT-1 | 62.7 | 0.99755 |
| Sample | τ0 (s) | Ea (eV) | Tv (K) |
|---|---|---|---|
| Neat PA66 | 3.082 × 10−13 | 0.4397 | 161.4 |
| PA66/15% ILT-1 1st Tg | 1.000 × 10−20 | 0.8799 | 90.50 |
| PA66/15% ILT-1 2nd Tg | 3.261 × 10−06 | 0.0229 | 371.5 |
| PA66/15% IIT-3 | 1.691 × 10−11 | 0.3003 | 204.0 |
| PA66/15%IHT-1 | 2.904 × 10−12 | 0.3537 | 184.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belyamani, I.; K. Hassan, M. Effect of Different Phosphate Glass Compositions on the Process-Induced Macromolecular Dynamics of Polyamide 66. Polymers 2020, 12, 1179. https://doi.org/10.3390/polym12051179
Belyamani I, K. Hassan M. Effect of Different Phosphate Glass Compositions on the Process-Induced Macromolecular Dynamics of Polyamide 66. Polymers. 2020; 12(5):1179. https://doi.org/10.3390/polym12051179
Chicago/Turabian StyleBelyamani, Imane, and Mohammad K. Hassan. 2020. "Effect of Different Phosphate Glass Compositions on the Process-Induced Macromolecular Dynamics of Polyamide 66" Polymers 12, no. 5: 1179. https://doi.org/10.3390/polym12051179
APA StyleBelyamani, I., & K. Hassan, M. (2020). Effect of Different Phosphate Glass Compositions on the Process-Induced Macromolecular Dynamics of Polyamide 66. Polymers, 12(5), 1179. https://doi.org/10.3390/polym12051179







