Application Properties Analysis as a Dielectric Capacitor of End-of-Life Tire-Reinforced HDPE
Abstract
1. Introduction
2. Capacitive Variables, Materials, and Applications
2.1. Relative Permittivity or Dielectric Constant εr or k
2.2. Loss Angle δ, Dielectric Loss Factor, Tg δ and Loss Ratio εr″
2.3. Dielectric Materials
2.4. Capacitive Application
- -
- Charge storing capacity: this is the basic characteristic of a capacitor. The capacitor consists of two metal conducting plates and a dielectric sandwiched between them. When provided with the supply voltage capacitors are charged and can be used as a charge storage device.
- -
- Direct current (DC) blocking: the capacitor blocks DC and allows the AC signal to pass.
- -
- Signal decoupling: decoupling capacitor’s job is to suppress high-frequency noise in power supply signals.
- -
- Signal filtering: capacitors are used as filters in some circuits as they are used to filter the output signal of a rectifier.
- -
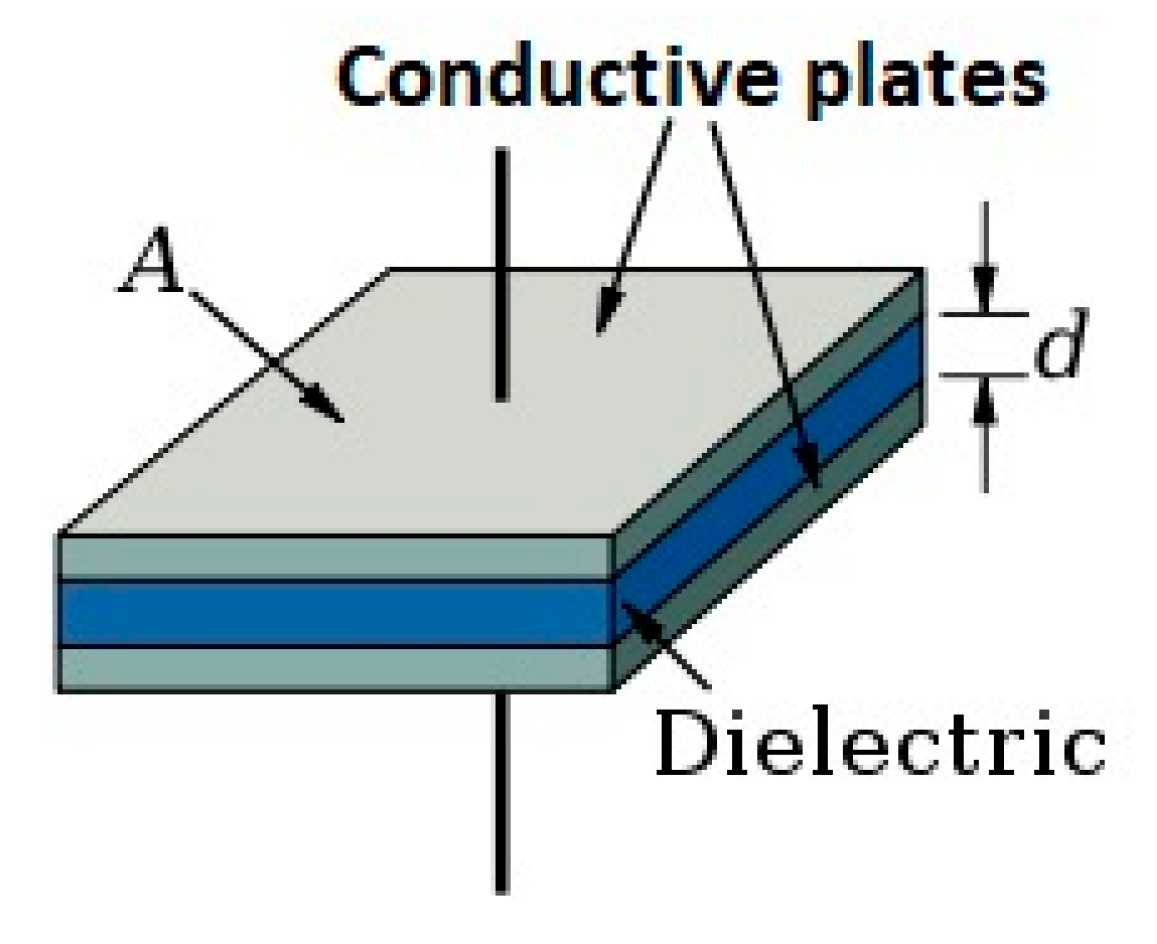
- Capacitor construction: a capacitor is constructed anytime two electrical conductors or conducting plates are separated by a dielectric or insulator (Figure 1).
3. Materials and Methods
3.1. Samples
3.2. Equipment and Methods
4. Results and Comments
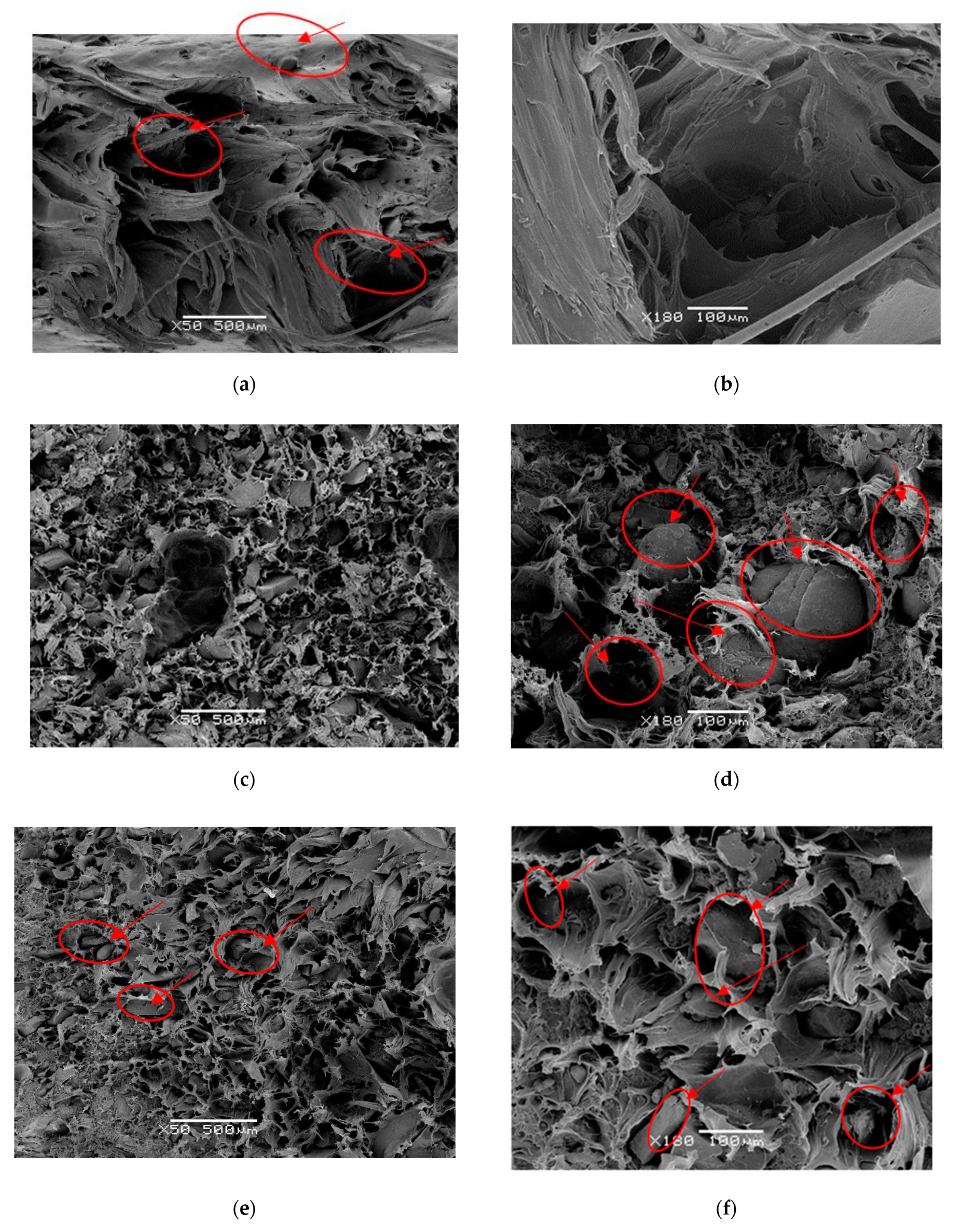
4.1. Microstructure Analysis
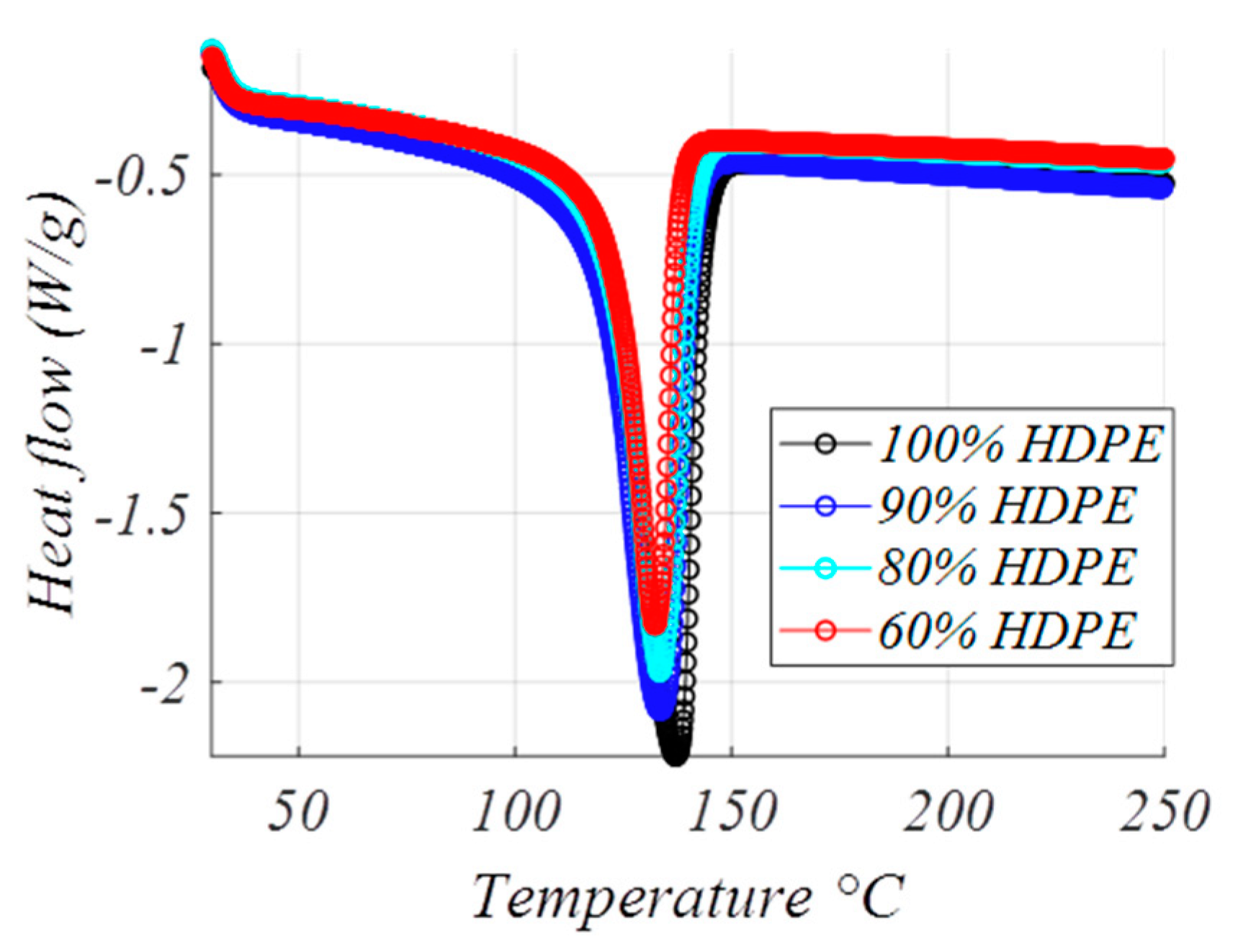
4.2. Thermal Study
4.3. Dielectric Analysis of High-Density Polyethylene (HDPE) and HDPE/Ground Tire Rubber (GTR) Compounds
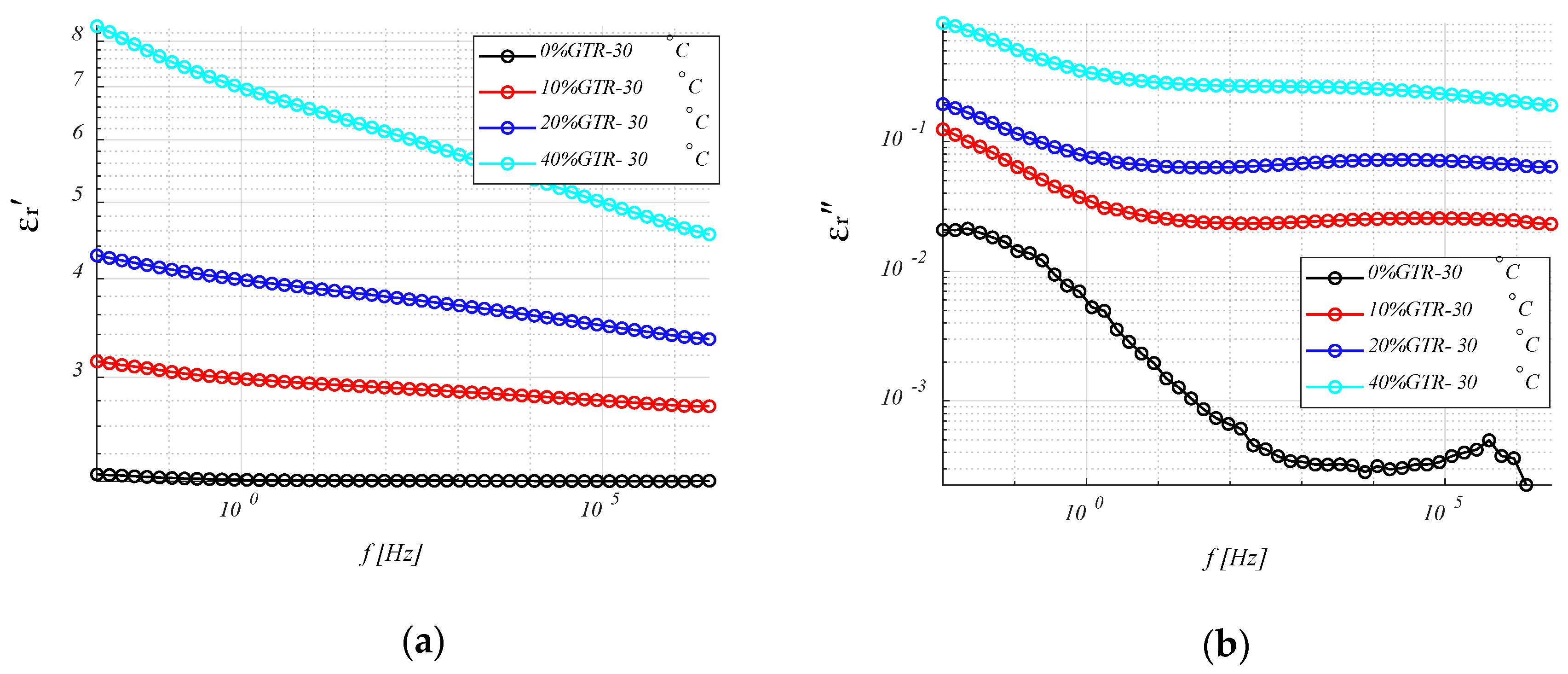
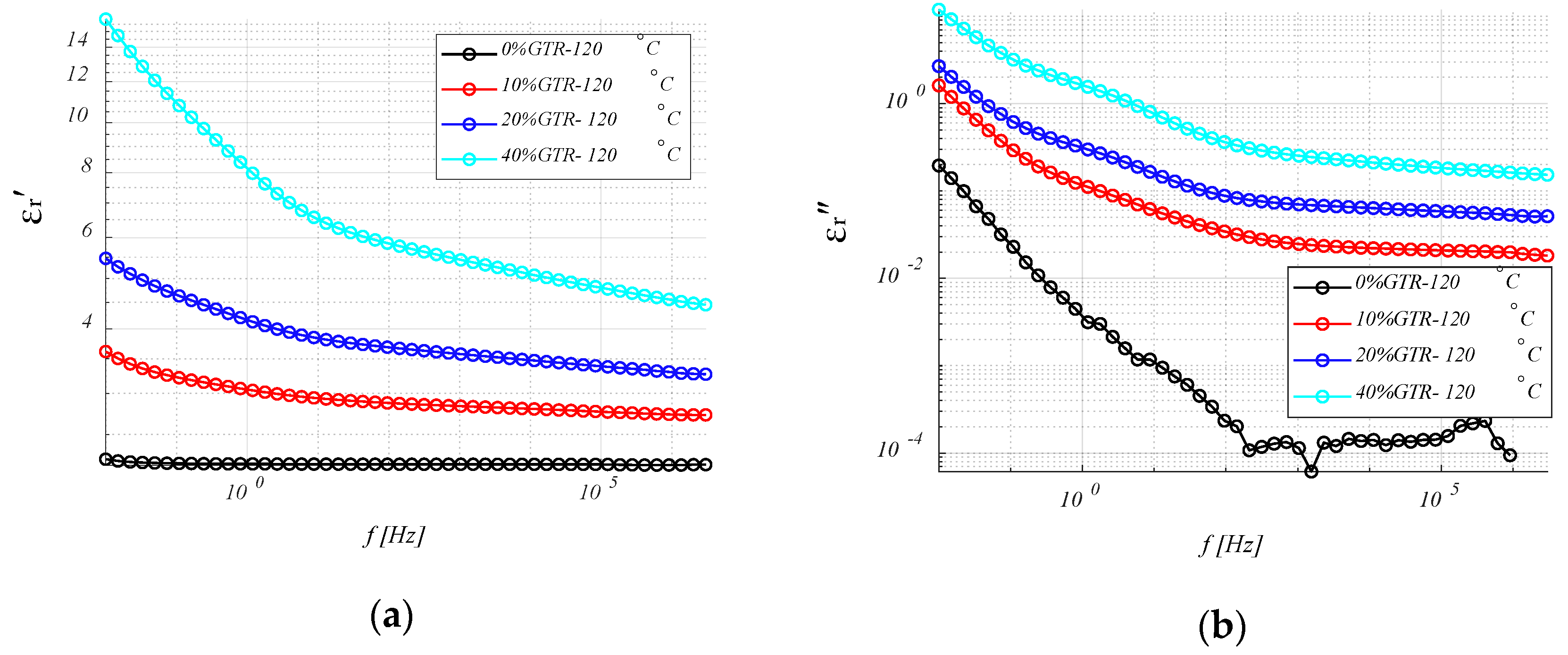
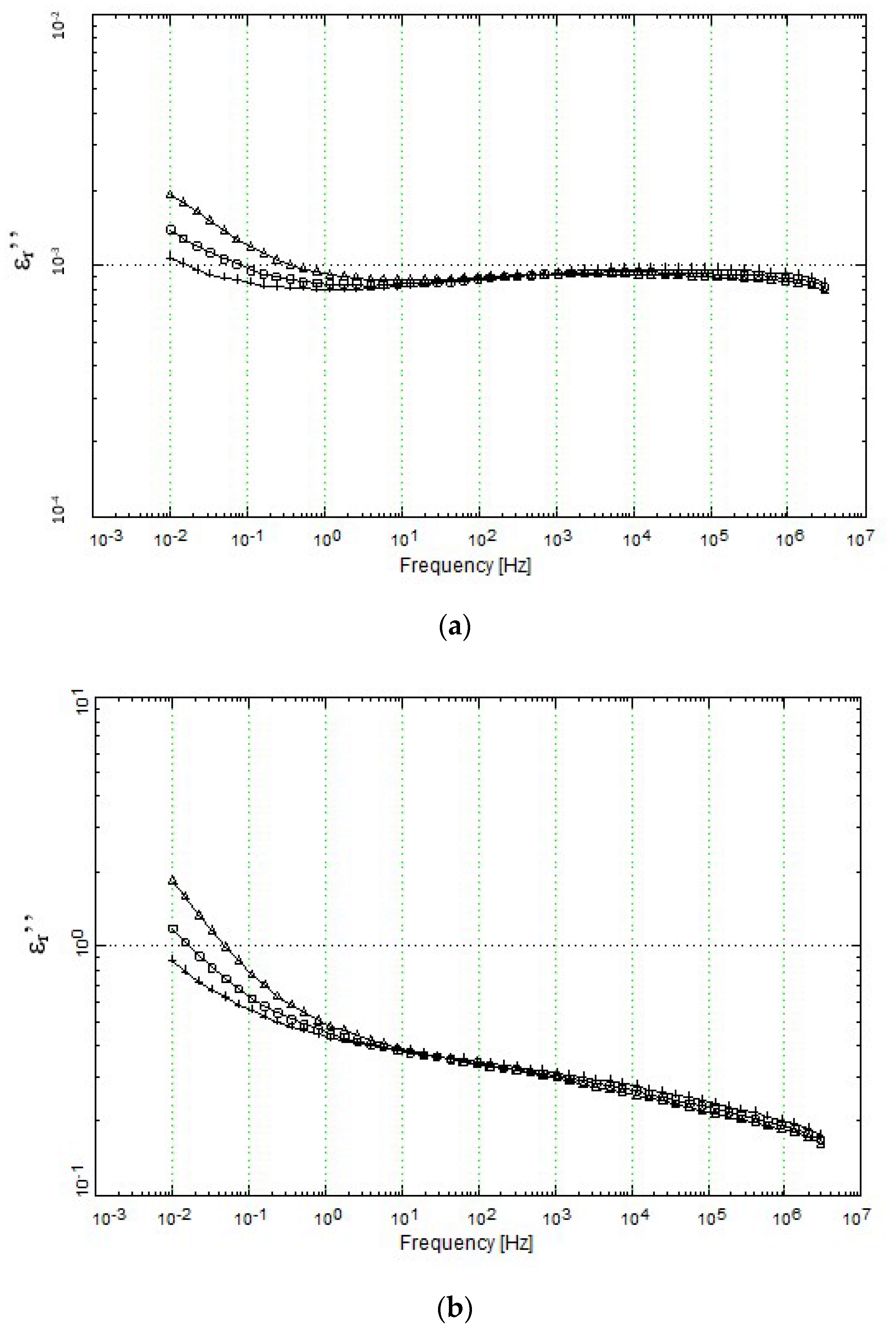
4.3.1. Permittivity (ε′ and ε″) Analysis.
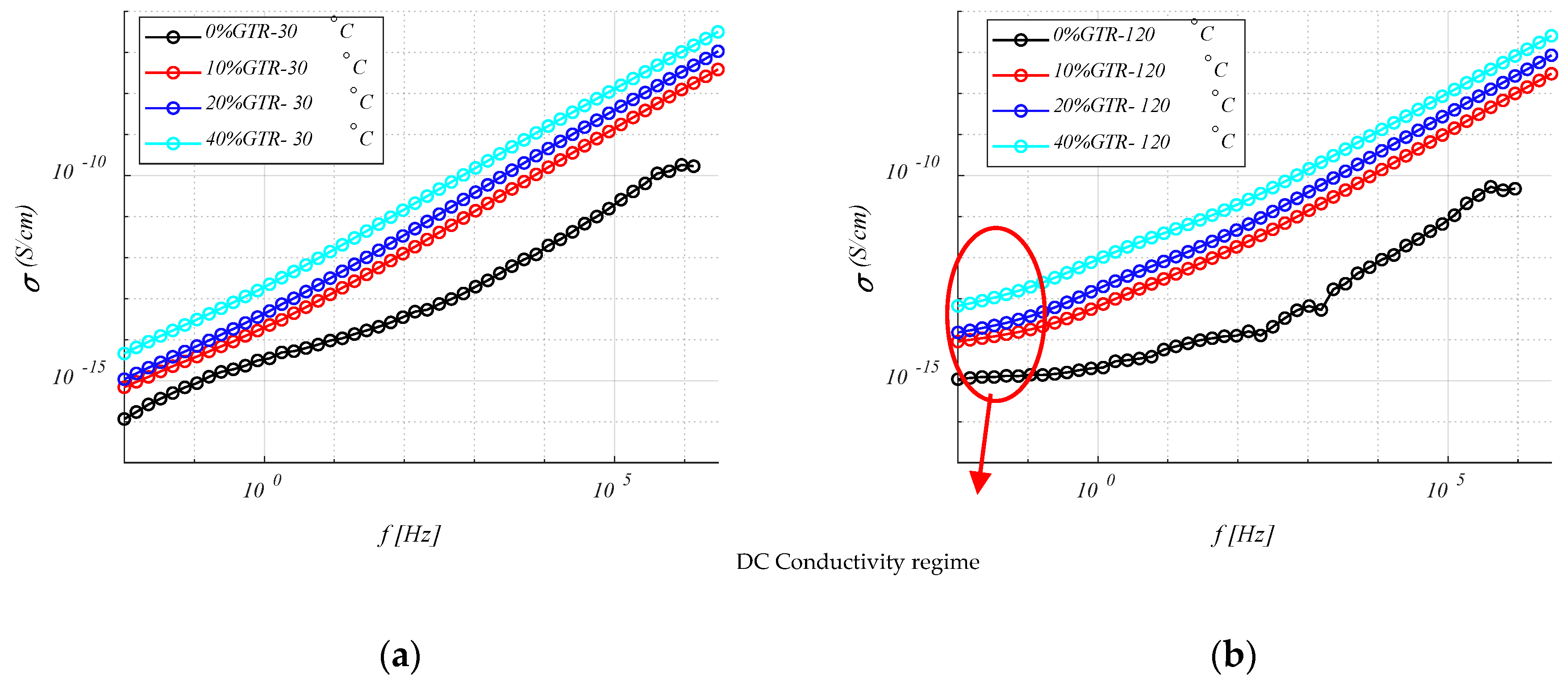
4.3.2. Conductivity () Analysis
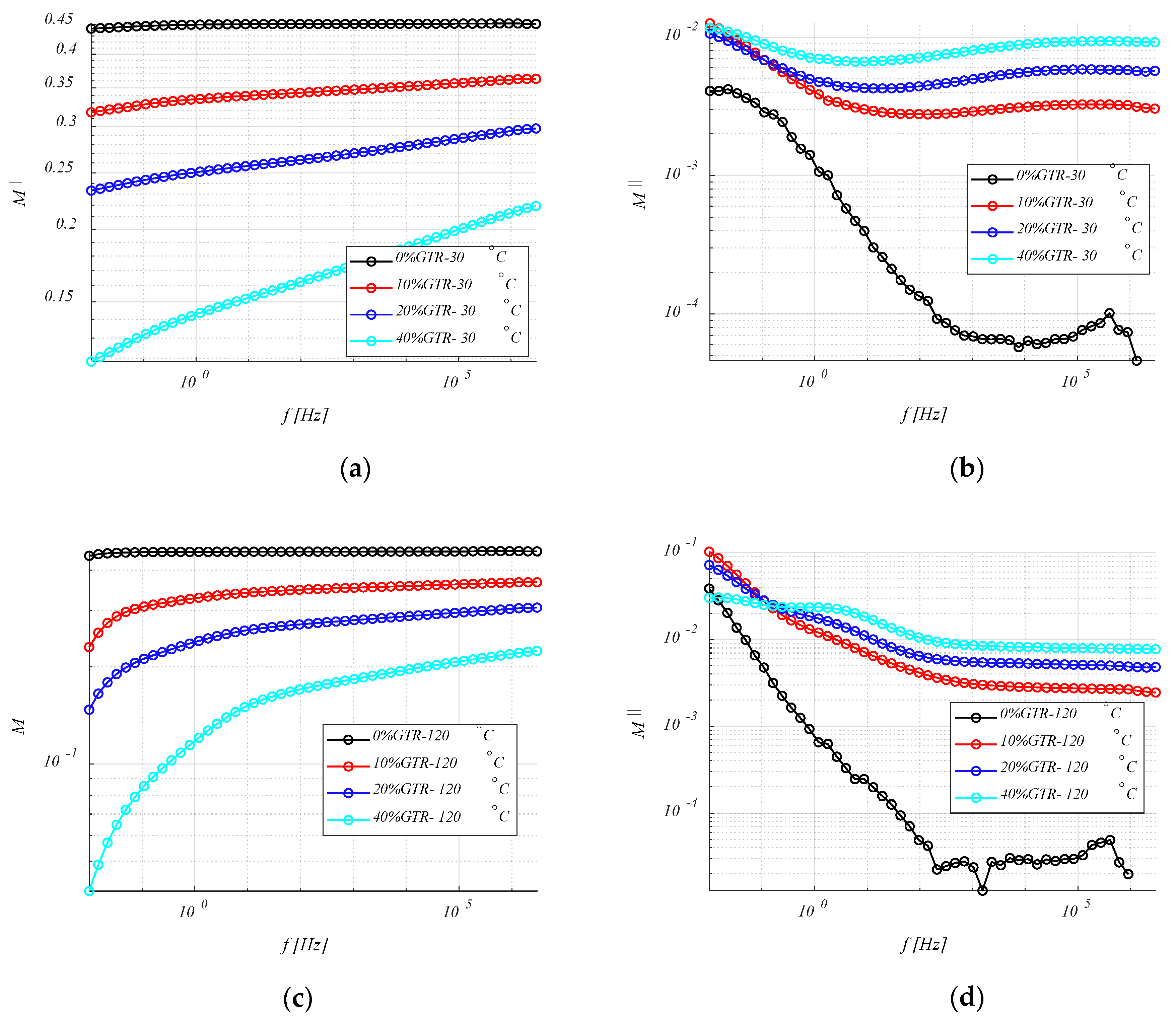
4.3.3. Electrical Modulus (M′ and M″)
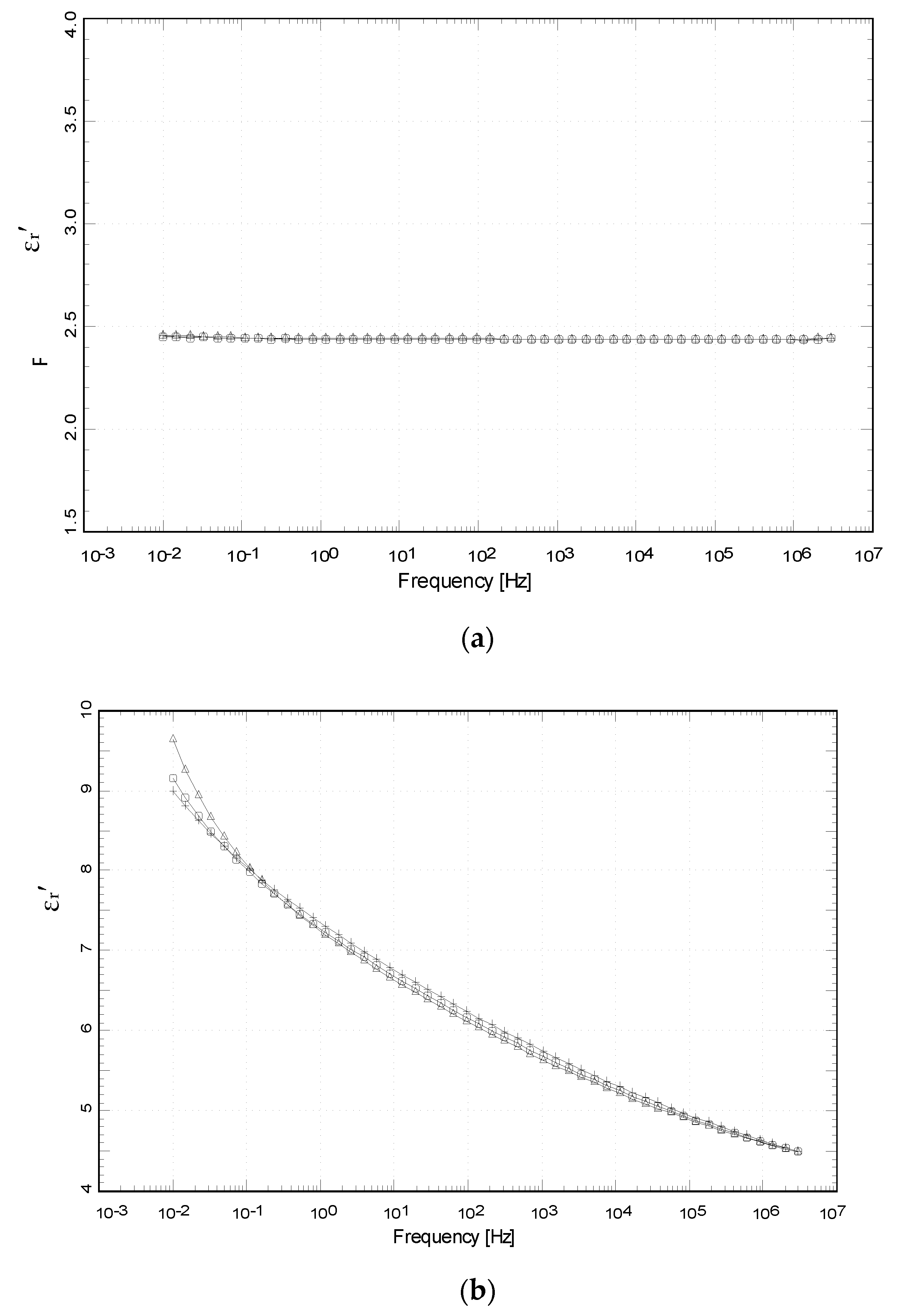
4.3.4. Relative Permittivity, εr′ (or K)
4.3.5. Loss Ratio, εr″
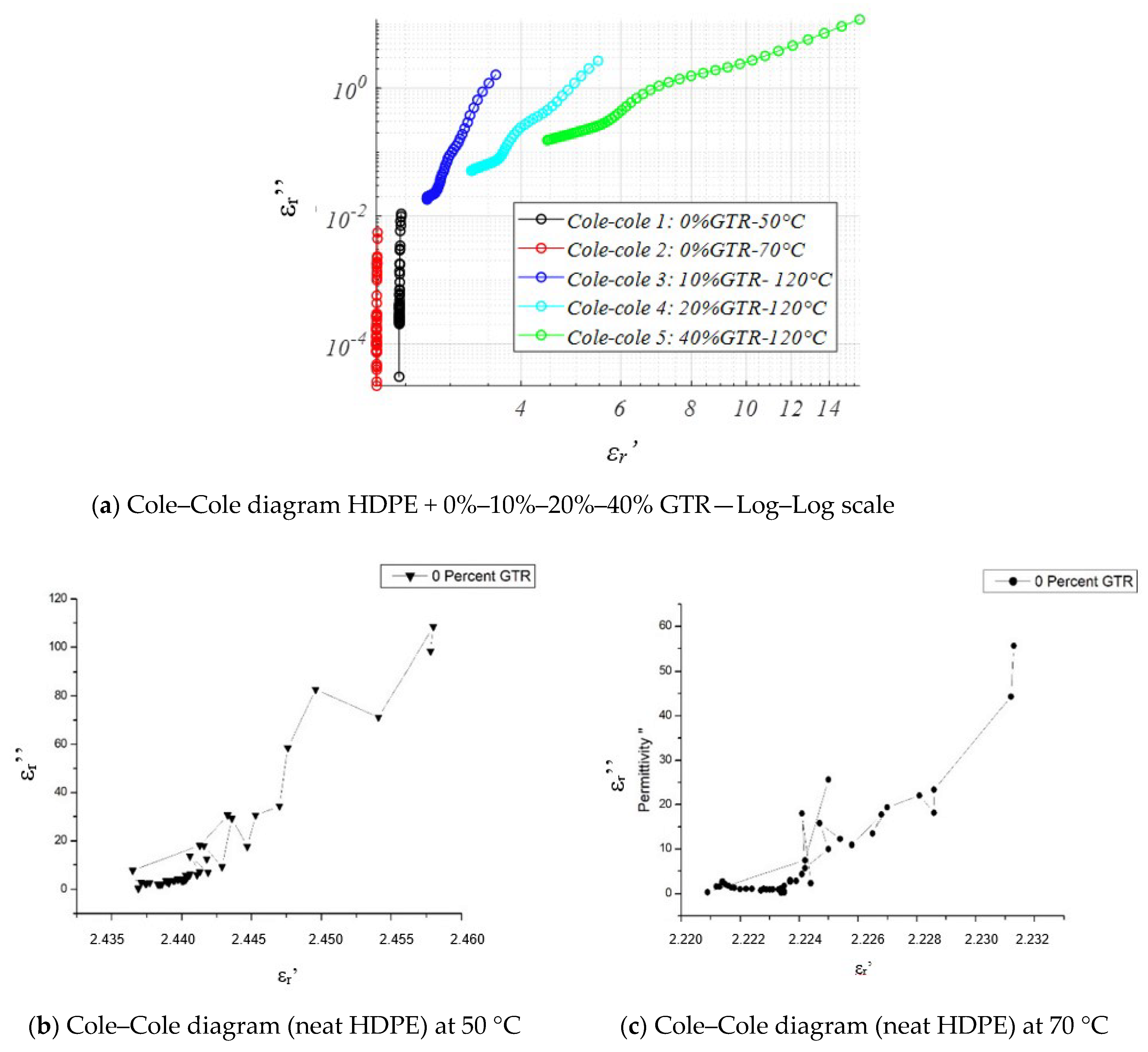
4.3.6. Cole–Cole Diagram and Analysis
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rodgers, B.; Waddell, W. Tire Engineering. In Science and Technology of Rubber; Mark, J.E., Erman, B., Eirich, F.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 619–661. [Google Scholar]
- Marín-Genescà, M.; García-Amorós, J.; Mujal-Rosas, R.; Vidal, L.M.; Arroyo, J.B.; Colom, X. Ground Tire Rubber Recycling in Applications as Insulators in Polymeric Compounds, According to Spanish UNE Standards. Recycling 2020, 5, 16. [Google Scholar] [CrossRef]
- Behnood, A.; Olek, J. Stress-dependent behavior and rutting resistance of modified asphalt binders: An MSCR approach. Constr. Build. Mater. 2017, 157, 635–646. [Google Scholar] [CrossRef]
- Colom, X.; Cañavate, J.; Carrillo, F.; Lis, M.J. Acoustic and mechanical properties of recycled polyvinyl chloride/ground tire rubber composites. J. Compos. Mater. 2013, 48, 1061–1069. [Google Scholar] [CrossRef]
- Ramarad, S.; Khalid, M.; Ratnam, C.; Chuah, A.L.; Rashmi, W. Waste tire rubber in polymer blends: A review on the evolution, properties and future. Prog. Mater. Sci. 2015, 72, 100–140. [Google Scholar] [CrossRef]
- Mujal-Rosas, R.; Marín-Genescà, M.; García-Amorós, J.; Salueña-Berna, X.; Colom, X. Influence on the mechanical properties of various polymeric composites reinforced with GTR particles. Afinidad 2019, 76, 588. [Google Scholar]
- Mujal-Rosas, R.; Orrit-Prat, J.; Marin-Genesca, M.; Rahhali, A.; Colom, X. Dielectric and mechanical properties of high-density polyethylene (HDPE) reinforced with used tires (GTR). Afinidad 2010, 67, 7–13. [Google Scholar]
- Marín-Genescà, M.; García-Amorós, J.; Mujal-Rosas, R.; Salueña Berna, X.; Massagués Vidal, L. Comparison of Mechanical and Electrical Characteristics of Various Polymers Blended with Ground Tire Rubber (GTR) and Applications. Appl. Sci. 2019, 9, 1564. [Google Scholar] [CrossRef]
- Mather, P.J.; Thomas, K.M. Carbon black/high density polyethylene conducting composite materials: Part I Structural modification of carbon black by gasification in carbon dioxide and the effect on the electrical and mechanical properties of the composite. J. Mater. Sci. 1997, 32, 401–407. [Google Scholar] [CrossRef]
- Mather, P.J.; Thomas, K.M. Carbon black/high density polyethylene conducting composite materials: Part II The relationship between the positive temperature coefficient and the volume resistivity. J. Mater. Sci. 1997, 32, 1711–1715. [Google Scholar] [CrossRef]
- Zhang, Q.M.; Li, H.; Poh, M.; Xia, F.; Cheng, Z.Y.; Xu, H.; Huang, C. An all-organic composite actuator material with a high dielectric constant. Nature 2002, 419, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Nan, C.W.; Shen, Y.; Ma, J. Physical properties of composites near percolation. Ann. Rev. Mater. Res. 2010, 40, 131–151. [Google Scholar] [CrossRef]
- Facchetti, A.; Yoon, M.H.; Marks, T.J. Gate dielectrics for organic field-effect transistors: New opportunities for organic electronics. Adv. Mater. 2005, 17, 1705–1725. [Google Scholar] [CrossRef]
- Chu, B.; Zhou, X.; Ren, K.; Neese, B.; Lin, M.; Wang, Q.; Zhang, Q. A dielectric polymer with high electric energy density and fast discharge speed. Science 2006, 313, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Bauer, F.; Fousson, E.; Zhang, Q.M. Recent advances in highly electrostrictive P (VDF-TrFE-CFE) terpolymers. IEEE Trans. Dielectr. Electr. Insul. 2006, 13, 1149–1154. [Google Scholar] [CrossRef]
- Tummala, R.R.; Laskar, J. Gigabit wireless: System-on-a-package technology. Proc. IEEE 2004, 92, 376–387. [Google Scholar] [CrossRef]
- Dang, Z.M.; Zheng, Y.; Xu, H.P. Effect of the ceramic particle size on the microstructure and dielectric properties of barium titanate/polystyrene composites. J. Appl. Polym. Sci. 2008, 110, 3473–3479. [Google Scholar] [CrossRef]
- Pramanik, N.C.; Seok, S.I. High-dielectric constant inorganic-organic hybrid materials prepared with sol-gel-derived crystalline BaTiO3. J. Appl. Phys. 2008, 47, 531–537. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Tanase, T.; Tabata, T.; Miwa, T.; Konno, M. Fabrication and dielectric properties of the BaTiO3-polymer nanocomposite thin films. J. Eur. Ceram. Soc. 2008, 28, 117–122. [Google Scholar] [CrossRef]
- Bai, Y.; Cheng, Z.Y.; Bharti, V.; Xu, H.S.; Zhang, Q.M. High-dielectric-constant ceramic-powder polymer composites. Appl. Phys. Lett. 2000, 76, 3804–3806. [Google Scholar] [CrossRef]
- Paik, K.W.; Hyun, J.G.; Lee, S.; Jang, K.W. Epoxy/BaTiO3 composite films and pastes for high dielectric constant and low-tolerance embedded capacitors fabrication in organic substrates. IEEE Trans. Electron. Packag. Manuf. 2005, 28, 297–303. [Google Scholar]
- Rao, Y.; Ogitani, S.; Kohl, P.; Wong, C.P. Novel polymer–ceramic nanocomposite based on high dielectric constant epoxy formula for embedded capacitor application. J. Appl. Polym. Sci. 2002, 83, 1084–1090. [Google Scholar] [CrossRef]
- Li, H.; Liu, F.; Fan, B.; Ai, D.; Peng, Z.; Wang, Q. Nanostructured ferroelectric-polymer composites for capacitive energy storage. Small Methods 2018, 2, 1700399. [Google Scholar] [CrossRef]
- Rao, Y.; Ogitani, S.; Kohl, P.; Wong, C.P. High dielectric constant polymer-ceramic composite for embedded capacitor application. In Proceedings of the International Symposium on Advanced Packaging Materials Processes, Properties and Interfaces (Cat. No.00TH8507), Braselton, GA, USA, 6–8 August 2000; pp. 32–37. [Google Scholar] [CrossRef]
- Nayak, S.; Kumar Chaki, T.; Khastgir, D. Development of Poly (Dimethylsiloxane)/BaTiO3 Nanocomposites as Dielectric Material. In Advanced Materials Research; Trans Tech Publications Ltd.: Schwyz, Switzerland, 2013; pp. 897–900. [Google Scholar]
- ASTM D-150. Standard Test Methods for AC Loss Characteristics and Permittivity (Dielectric Constant) of Solid Electrical Insulation, 2018 ed.; ASTM: West Conshohocken, PA, USA, 1 May 2018. [Google Scholar]
- Song, Y.; Zheng, Q. Conduction stability of high-density polyethylene/carbon black composites due to electric field action. Eur. Polym. J. 2005, 41, 2998–3003. [Google Scholar] [CrossRef]
- Geuskens, G.; Gielens, J.L.; Geshef, D.; Deltour, R. The electrical conductivity of polymer blends filled with carbon black. Eur. Polym. J. 1987, 23, 993–995. [Google Scholar] [CrossRef]
- Montanari, G.C.; Mazzanti, G.; Palmieri, F.; Motori, A.; Perego, G.; Serra, S. Space-charge trapping and conduction in LDPE, HDPE and XLPE. J. Phys. D Appl. Phys. 2001, 34, 2902. [Google Scholar] [CrossRef]
- Marín-Genescà, M. Characterization of Electrical Behavior, Mechanical and Thermal of Compounds Polymeric and with Mixtures from Recycling. September de 2011. ISBN: 9788469501146. Available online: https://www.tesisenred.net/handle/10803/37918#page=1 (accessed on 9 September 2020).
- Chen, Q.; Wang, Y.; Zhou, X.; Zhang, Q.M.; Zhang, S. High field tunneling as a limiting factor of maximum energy density in dielectric energy storage capacitors. Appl. Phys. Lett. 2008, 92, 142909. [Google Scholar] [CrossRef]
- Grove, T.T.; Masters, M.F.; Miers, R.E. Determining dielectric constants using a parallel plate capacitor. Am. J. Phys. 2005, 73, 52–56. [Google Scholar] [CrossRef]
- Colom, X.; Marín-Genesca, M.; Mujal, R.; Formela, K.; Cañavate, J. Structural and physico-mechanical properties of natural rubber/GTR composites devulcanized by microwaves: Influence of GTR source and irradiation time. J. Compos. Mater. 2018, 52, 3099–3108. [Google Scholar] [CrossRef]
- Colom, X.; Carrillo, F.; Cañavate, J. Composites reinforced with reused tyres: Surface oxidant treatment to improve the interfacial compatibility Composites: Part A. Appl. Sci. Manuf. 2007, 38, 44–50. [Google Scholar] [CrossRef]
- Colom, X.; Cañavate, J.; Carrillo, F.; Velasco, J.I.; Pagès, P.; Mujal, P.; Nogués, F. Structural and mechanical studies on modified reused tyres composites. Eur. Polym. J. 2006, 42, 2369–2378. [Google Scholar] [CrossRef]
- Lin, T.A.; Lin, M.C.; Lin, J.Y.; Lin, J.H.; Chuang, Y.C.; Lou, C.W. Modified polypropylene/thermoplastic polyurethane blends with maleic-anhydride grafted polypropylene: Blending morphology and mechanical behaviors. J. Polym. Res. 2020, 27, 34. [Google Scholar] [CrossRef]
- Wunderlich, B. Macromolecular Physics, 1st ed.; Academic Press: New York, NY, USA, 1973; ISBN 9780323145909. [Google Scholar]
- Wochowicz, A.; Eder, M. Distribution of lamella thicknesses in isothermally crystallized polypropylene and polyethylene by differential scanning calorimetry. Polymer 1984, 25, 1268–1270. [Google Scholar] [CrossRef]
- Billings, M.J.; Smith, A.; Wilkins, R. Tracking in Polymeric Insulation. IEEE Trans. Electr. Insul. 1967, EI-2, 131–137. [Google Scholar] [CrossRef]
- Li, S.; Yu, S.; Feng, Y. Progress in and prospects for electrical insulating materials. High Volt. 2016, 1, 122–129. [Google Scholar] [CrossRef]
- Teyssedre, G.; Laurent, C. Charge Transport Modeling in Insulating Polymers: From Molecular to Macroscopic Scale. IEEE Trans. Dielectr. Electr. Insul. 2005, 12, 857–875. [Google Scholar] [CrossRef]
- Crone, B.K.; Campbell, I.H.; Davids, P.S.; Smith, D.L. Device model for single carrier organic diodes. J. Appl. Phys. 1997, 82, 6319–6325. [Google Scholar]
- Liu, X.H.; Krückel, J.; Zheng, G.Q.; Schubert, D.W. Electrical conductivity behavior of sheared poly(methylmethacrylate)/carbon black composites. Compos. Sci. Technol. 2014, 100, 99–104. [Google Scholar] [CrossRef]
- Zhao, J.; Dai, K.; Liu, C.; Zheng, G.; Wang, B.; Liu, C.; Chen, J.; Shen, C.A. comparison between strain sensing behaviors of carbon black/polypropylene and carbon nanotubes/polypropylene electrically conductive composites. Compos. A Appl. Sci. Manuf. 2013, 48, 129–136. [Google Scholar] [CrossRef]
- Prodromakis, T.; Papavassiliou, C. Engineering the Maxwell–Wagner polarization effect. Appl. Surf. Sci. 2009, 255, 6989–6994. [Google Scholar] [CrossRef]
- Marín-Genescà, M.; García-Amoròs, J.; Mujal-Rosas, R.; Massagués, L.; Colom, X. Study and Characterization of the Dielectric Behavior of Low Linear Density Polyethylene Composites Mixed with Ground Tire Rubber Particles. Polymers 2020, 12, 1075. [Google Scholar] [CrossRef]
- O’Neill, R.; Bowman, M.; Gregg, J.M. Dielectric enhancement and Maxwell–Wagner effects in ferroelectric superlattice structures. Appl. Phys. Lett. 2000, 77, 1520. [Google Scholar] [CrossRef]
- Petrovic, M.M.V.; Bobic, J.D.; Ramoška, T.; Banys, J.; Stojanovic, B.D. Antimony doping effect on barium titanate structure and electrical properties. Ceram. Int. 2011, 37, 2669–2677. [Google Scholar] [CrossRef]
- Naskar, A.K.; Khastgir, D.; Bhowmick, A.K.; De, S.K. Effect of Chlorination of Ground Rubber Tire on its Compatibility with Poly(vinyl chloride): Dielectric Studies. J. Appl. Polym. Sci. 2002, 84, 993–1000. [Google Scholar] [CrossRef]
- Saad, A.L.G.; Aziz, H.A.; Dimitry, O.I.H. Studies of Electrical and Mechanical Properties of Poly(vinylchloride) Mixed with Electrically Conductive Additives. J. Appl. Polym. Sci. 2004, 91, 1590–1598. [Google Scholar] [CrossRef]
- Williams, G. Dielectric Properties of Polymers. Encycl. Mater. Sci. Technol. 2017, 7642–7648. [Google Scholar] [CrossRef]
- Hill, R.M.; Dissado, L.A. Debye and non-Debye relaxation. J. Phys. C Solid State Phys. 1985, 18, 3829. [Google Scholar] [CrossRef]
- Medina, N.F.; Garcia, R.; Hajirasouliha, I.; Pilakoutas, K.; Guadagnini, M.; Raoul, S. Composites with recycled rubber aggregates: Properties and opportunities in construction. Constr. Build. Mater. 2018, 188, 884–897. [Google Scholar] [CrossRef]
- Karger-Kocsis, J.; Mészáros, L.; Bárány, T. Ground tire rubber (GTR) in thermoplastics, thermosets, and rubbers. J. Mater. Sci. 2013, 48, 1–38. [Google Scholar] [CrossRef]
- Ikeda, Y.; Kato, A.; Kohjiya, S.; Nakajima, Y. Rubber Science; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Rajan, V.V.; Dierkes, W.K.; Joseph, R.; Noordermeer, J.W. Science and technology of rubber reclamation with special attention to NR-based waste latex products. Prog. Polym. Sci. 2006, 31, 811–834. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Kucinska-Lipka, J.; Janik, H.; Balas, A. Progress in used tyres management in the European Union: A review. Waste Manag. 2012, 32, 1742–1751. [Google Scholar] [CrossRef]
- Amari, T.; Themelis, N.J.; Wernick, I.K. Resource recovery from used rubber tires. Resour. Policy 1999, 25, 179–188. [Google Scholar] [CrossRef]
- Van Beukering, P.J.; Janssen, M.A. Trade and recycling of used tires in Western and Eastern Europe. Resour. Conserv. Recycl. 2001, 33, 235–265. [Google Scholar] [CrossRef]
- Akiba, M.; Hashim, A.S. Vulcanization, and crosslinking in elastomers. Prog. Polym. Sci. 1997, 22, 475–521. [Google Scholar] [CrossRef]
- Fazli, A.; Rodrigue, D. Waste Rubber Recycling: A Review on the Evolution and Properties of Thermoplastic Elastomers. Materials 2020, 13, 782. [Google Scholar] [CrossRef] [PubMed]
- Mujal-Rosas, R.; Marin-Genesca, M.; Garcia-Amoros, J.; Colom, X.; Salueña-Berna, X. Comparison of the mechanical and dielectric characteristics of various polymers mixed with ground-tired rubber (GTR) for its application as industrial work footwear insulator. DYNA 2020, 95, 61–67. [Google Scholar] [CrossRef]
- Nakajima, N. Science and Practice of Rubber Mixing; Smithers Rapra Publishing: Shawbury, UK, 2000. [Google Scholar]
- Mente, P.; Motaung, T.; Hlangothi, S. Natural rubber and reclaimed Rubber composites—A Systematic Review. Polym. Sci. 2016, 2, 1–19. [Google Scholar]
- Zhang, J.F.; Zheng, Q.; Yang, Y.Q.; Yi, X.S. High-density polyethylene/carbon black conductive composites. I. Effect of CB surface modification on its resistivity–temperature behavior. J. Appl. Polym. Sci. 2002, 83, 3112–3116. [Google Scholar] [CrossRef]
- Zhang, J.F.; Zheng, Q.; Yang, Y.Q.; Yi, X.S. High-density polyethylene/carbon black conductive composites. II. Effect of electron beam irradiation on relationship between resistivity–temperature behavior and volume expansion. J. Appl. Polym. Sci. 2002, 83, 3117–3122. [Google Scholar] [CrossRef]
- Tang, H.; Chen, X.; Luo, Y. Studies on the PTC/NTC effect of carbon black filled low density polyethylene composites. Eur. Polym. J. 1997, 33, 1383–1386. [Google Scholar] [CrossRef]
- Mallette, J.G.; Quej, L.M.; Marquez, A.; Manero, O. Carbon black-filled PET/HDPE blends: Effect of the CB structure on rheological and electric properties. J. Appl. Polym. Sci. 2001, 81, 562–569. [Google Scholar] [CrossRef]
- Feller, J.F.; Linossier, I.; Levesque, G. Conductive polymer composites (CPCs): Comparison of electrical properties of poly (ethylene-co-ethyl acrylate)-carbon black with poly (butylene terephthalate)/poly (ethylene-co-ethyl acrylate)-carbon black. Polym. Adv. Technol. 2002, 13, 714–724. [Google Scholar] [CrossRef]
- Elsen, W. Application of Capacitors in Low Frequency and Long Time Constant Circuits. IEEE Trans. Compon. Parts 1964, 11, 216–219. [Google Scholar] [CrossRef]

| Material | ε′ at 1 MHz |
|---|---|
| Alumina | 4.5–8.4 |
| Ambar | 2.6 |
| Pyrex glass | 3.8–6 |
| Mica | 2.5–7 |
| Neoprene | 4.1 |
| Nylon | 3.4 |
| Polyethylene | 2.4–2.7 |
| Polyvinyl chloride | 3 |
| Teflon | 2 |
| Properties | Method | Unit | Value |
|---|---|---|---|
| GENERAL | |||
| Fluidity Index (2.16 Kg/190 °C) | ISO 1133 | g/10 min | 3.0 |
| Density at 23 °C | ISO 1183 | g/cm3 | 0.902 |
| MECHANICS | |||
| Tensile strength | ISO 527 | Mpa | 21 |
| Traction elongation | ISO 527 | % | 700 |
| Elastic modulus in flexion | ISO 178 | Mpa | 1000 |
| Formulation (% HDPE) | Δh (J/g Mixture) | Δh (J/g HDPE) | χ (%) | Tmax (°C) | Imax (A) |
|---|---|---|---|---|---|
| 100 | 205.9 | 205.9 | 70.3 | 137 | 380 |
| 90 | 183.8 | 204.3 | 69.7 | 135.2 | 272 |
| 80 | 162.1 | 202.7 | 69.1 | 135 | 265 |
| 60 | 127.8 | 212.9 | 72.7 | 132.3 | 189 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marín-Genescà, M.; García-Amorós, J.; Mujal-Rosas, R.; Massagués Vidal, L.; Colom Fajula, X. Application Properties Analysis as a Dielectric Capacitor of End-of-Life Tire-Reinforced HDPE. Polymers 2020, 12, 2675. https://doi.org/10.3390/polym12112675
Marín-Genescà M, García-Amorós J, Mujal-Rosas R, Massagués Vidal L, Colom Fajula X. Application Properties Analysis as a Dielectric Capacitor of End-of-Life Tire-Reinforced HDPE. Polymers. 2020; 12(11):2675. https://doi.org/10.3390/polym12112675
Chicago/Turabian StyleMarín-Genescà, Marc, Jordi García-Amorós, Ramon Mujal-Rosas, Lluís Massagués Vidal, and Xavier Colom Fajula. 2020. "Application Properties Analysis as a Dielectric Capacitor of End-of-Life Tire-Reinforced HDPE" Polymers 12, no. 11: 2675. https://doi.org/10.3390/polym12112675
APA StyleMarín-Genescà, M., García-Amorós, J., Mujal-Rosas, R., Massagués Vidal, L., & Colom Fajula, X. (2020). Application Properties Analysis as a Dielectric Capacitor of End-of-Life Tire-Reinforced HDPE. Polymers, 12(11), 2675. https://doi.org/10.3390/polym12112675









